Abstract
Auditory verbal hallucinations (AVHs) occur in two-thirds of patients diagnosed with schizophrenia [1, 2]. Persistent AVHs lasting for more than 2 years are significantly associated with depression and anxiety in patients with schizophrenia [3]. Because of frequent resistance to antipsychotic treatment [4], AVHs have been associated with considerable psychosocial distress, violent behaviour, and even suicide attempts [5].
The evaluation of AVHs relies inherently on subjective descriptions provided by the patients who experience these symptoms. Despite the potential neurobiological significance of subjective experiences [6], these phenomena remain challenging to assess via behavioural observation. In addition, methodologies that present the sums of scaled scores tend to lose information [7]. The application of auditory hallucination scales that merely list various types of AVHs to evaluate dimensional changes in AVHs may inevitably result in the loss of information [8]. Numerous phenomenological variables pertaining to AVHs have been identified [9, 10], but studies of the clinical significance of such phenomenological variables are still scarce [11, 12].
The measurement of only the reported physical characteristics of AVHs (e.g. frequency or loudness) may not reflect the actual clinical status of patients who report AVHs [13]. The particular emotional or cognitive characteristics of AVHs (e.g. derogatory contents, negative emotions, and critical cognitive appraisals) carry a stronger influence on the behaviours of patients than do the particular characteristics of non-verbal hallucinations (e.g. musical hallucinations or mechanical noises) [5]. Haddock et al. developed the Psychotic Symptom Rating Scales (PSYRATS) based on previous studies of the phenomenology of positive symptoms; these contain subscales for auditory hallucinations and delusions [8]. The clinical use of PSYRATS has provided useful symptom-oriented information when it has been combined with broader measures such as the Positive and Negative Syndrome Scale (PANSS) [14]. The usefulness of PSYRATS had been also explored in patients who suffer from acute exacerbations [15].
Because specific psychological symptoms are not exclusively tied to specific mental health diagnoses, the aetiology and clinical expression of symptoms with identical names can differ completely when manifested as components of different diagnostic entities [16]. Furthermore, significant changes in the internal structure of psychotic symptoms can occur during the long-term course of this condition [17]. Patients with first-episode schizophrenia may show dynamic changes in symptom structure compared to patients at a chronic stage [18]. Thus, comparing first-episode and chronic schizophrenia groups may provide complementary information on symptom formation and stabilization.
We used a multivariate statistical approach to evaluate clinically significant dimensional changes in the AVHs characteristics of schizophrenia. Data from both first-episode and chronic groups were used to obtain complementary information pertinent to the effects of the course of illness on the dimensions of AVHs in schizophrenia.
Methods
Subjects
Inpatients who were diagnosed with schizophrenia and who reported active AVHs were recruited from Seoul National University Hospital and Gachon University Gil Medical Center, Korea. Active AVHs were operationally defined as including both the experience of hearing ‘voices’ and a minimum score of 4 on the hallucination item (P3) of the PANSS [19]. Two groups of inpatients with acute exacerbations of psychotic symptoms were eligible for participation: (i) drug-naïve patients with first-episode schizophrenia (illness duration <2 years); and (ii) drug-free patients with chronic schizophrenia (illness duration >10 years). Drug-free patients were defined as those who had not received antipsychotic treatment for >4 weeks before the study. Drug-free patients with chronic schizophrenia were recruited to investigate the influence of illness duration on the dimensional properties of AVHs after controlling for the effects of medication.
The inclusion criteria were: DSM-IV diagnosis of schizophrenia; age 15–65 years; presence of active AVHs; and fluency in written and spoken Korean. Patients were excluded if there was any evidence of DSM-IV-defined substance dependence (except nicotine and caffeine); mental retardation; pregnancy or lactation; otological conditions; and neurological disorders, including epilepsy, stroke, or severe head trauma. Patients receiving non-psychiatric medications with potential effects on psychiatric symptoms were also excluded.
Measures and procedures
The diagnosis of schizophrenia was confirmed by at least two psychiatrists using a semi-structured interview based on DSM-IV criteria. Patients were evaluated at baseline and at 6 month follow up. The severity of overall psychiatric symptoms was rated using PANSS and the Clinical Global Impression–Severity scale (CGI-S). The raters underwent training with standardized videotaped interviews to achieve optimal reliability. Prior to the study's initiation, the inter-rater reliability, determined by intra-class correlations, reached 0.89–0.95 for all instruments.
The 11 items on the Psychotic Symptom Rating Scales–Auditory Hallucination Subscale (PSYRATS-AHS) were used to evaluate multifaceted characteristics of AVHs [8]. The item structure of PSYRATS-AHS is consistent with the results of phenomenological studies [9]. PSYRATS was also examined for reliability and validity among Korean patients with psychotic disorders [20, 21]. Three factors were identified in a study of 71 patients with psychotic disorders: (i) physical characteristics, (ii) emotional characteristics, and (iii) cognitive interpretations [8].
Patients continued to be treated by their primary psychiatrists, who were independent of the study protocol. The follow-up interview was conducted with patients who were clinically stable and experiencing persistent AVHs after 6 months. Clinically stable patients were defined as follows: (i) no change in the regimen of antipsychotic treatment for >2 months; and (ii) no change in CGI-S scores for >2 months, indicating a plateau of clinical response to medication. Only patients who completed both baseline (active phase) and 6 month follow-up (stable phase) evaluations were included in the final analyses.
The study protocol was reviewed and approved by the Institutional Review Board. This study was conducted in accordance with the latest version of the Declaration of Helsinki. Written informed consent was obtained from each patient prior to enrolment.
Statistical analysis
Data on demographic variables and baseline scores on the clinical rating scales were compared using χ2, Fisher exact, and t-tests, as appropriate. To evaluate the stabilization of psychiatric symptoms, between-group differences in the score changes of clinical rating scales were examined using repeated-measures analysis of variance (RM ANOVA), with clinical status (first-episode vs chronic schizophrenia) as the between-subjects factor and time (baseline vs 6 month follow up) as the within-subjects factor.
We used hierarchical clustering (HC) and multidimensional scaling (MDS) to assess the internal structure of AVHs as a function of stage in the course of the illness. In addition, the overall structures of item clusters at baseline and at 6 months were constructed for both groups at each visit to determine treatment-induced changes in the internal structure of AVHs.
Ward's method of HC is designed to optimize the minimum variance within clusters [22], thereby reducing the information loss associated with cluster fusion. Squared Euclidean distance was used as the similarity measure. Ward's method outperforms other clustering methods in controlling the problem of cluster overlap.
The geographical presentation of MDS was used to convey additional information [23]. By modelling multivariate data as distances among points (variables) in geometric space [24], MDS provides graphical information on the essential properties of clusters, including density, variance, shape, and separation [22]. The S-Stress level (Stress) was used as a badness-of-fit measure and the R-squared index (RSQ) as a goodness-of-fit measure, both of which range from 0 (perfect fit for Stress; worst possible fit for RSQ) to 1 (worst possible fit for Stress; perfect fit for RSQ). Relatively high lack of fit (e.g. Stress >0.2 or RSQ <0.6) indicates too few dimensions in a given MDS solution [25]. All statistical analyses, including MDS and HC, were performed using SPSS 13.0 for Windows (SPSS, Chicago, IL, USA).
Results
Demographic and clinical characteristics
A total of 40 patients with first-episode (n=21) or chronic (n=19) schizophrenia completed both baseline and 6 month follow-up evaluations. Forty-eight patients with first-episode schizophrenia participated in the baseline assessment; four of these (8.3%) were lost to follow up and 23 (47.9%) reported the disappearance of AVHs at the 6 month follow-up visit. Thus, 21 (43.7%) reported persistent AVHs. Nineteen patients with chronic schizophrenia took part in the baseline assessment and none of the patients reported disappearance of voices during the 6 months of follow up. The baseline demographic and clinical characteristics of the two groups are presented in Table 1. The characteristics of the two groups were similar, except with regard to age and illness duration.
Subject details (mean±SD)

Changes in overall psychopathology and global functioning
The scores of the clinical rating scales are presented in Table 2, as well as the results of significance tests for group×time interactions in RM ANOVA. There were no significant differences between the two groups in the baseline scores of the clinical rating scales. Significant improvements in the severity of psychopathology, measured by the PANSS total score and CGI-S score, were observed between baseline and 6 month follow up (F1,38=124.1, p<0.001 for PANSS total score; F1,38=175.8, p<0.001 for CGI-S score, respectively), but no significant group×time interaction was observed. The severity of AVHs also decreased from baseline to the 6 month visit (F1,38=91.6, p<0.001) without significant group×time interaction.
Clinical rating scales scores
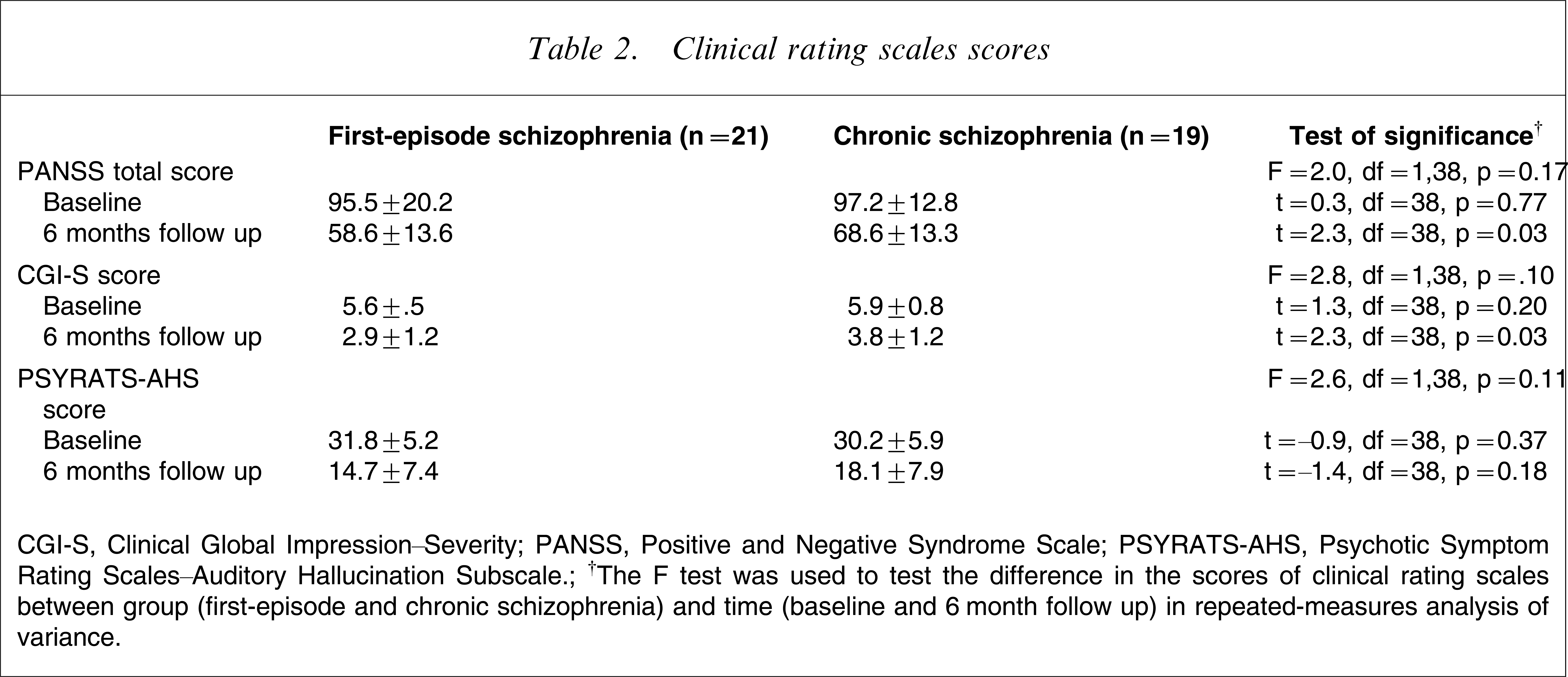
CGI-S, Clinical Global Impression–Severity; PANSS, Positive and Negative Syndrome Scale; PSYRATS-AHS, Psychotic Symptom Rating Scales–Auditory Hallucination Subscale.
†The F test was used to test the difference in the scores of clinical rating scales between group (first-episode and chronic schizophrenia) and time (baseline and 6 month follow up) in repeated-measures analysis of variance.
Overall changes in the internal structure of AVHs
The results of HC using Ward's method for 40 patients diagnosed with schizophrenia are shown in Figure 1. The baseline and 6 month follow-up dendrograms demonstrate a general decomposition of 11 items into two main clusters: emotional and cognitive. The items comprising physical characteristics did not form a separate cluster at baseline. At 6 months, dendrograms had longer internal nodes and shorter external nodes, indicating more distinctive and cohesive clustering: decreased distances between items in the same cluster were observed at 6 month follow up compared to baseline. Two structural changes occurred from baseline to 6 month follow up. First, ‘frequency’ moved from the cognitive to the emotional domain; thus, all items pertaining to physical characteristics, except ‘location’, formed a cluster with emotional variables. Second, ‘disruption to life caused by voices’ was shifted from the cognitive to the emotional domain.
Changes in the cluster structures of auditory verbal hallucinations in schizophrenia patients (n=40) between (a) baseline and (b) 6 month follow up analysed by hierarchical clustering using Ward's method. C, cognitive; E, emotional; P, physical.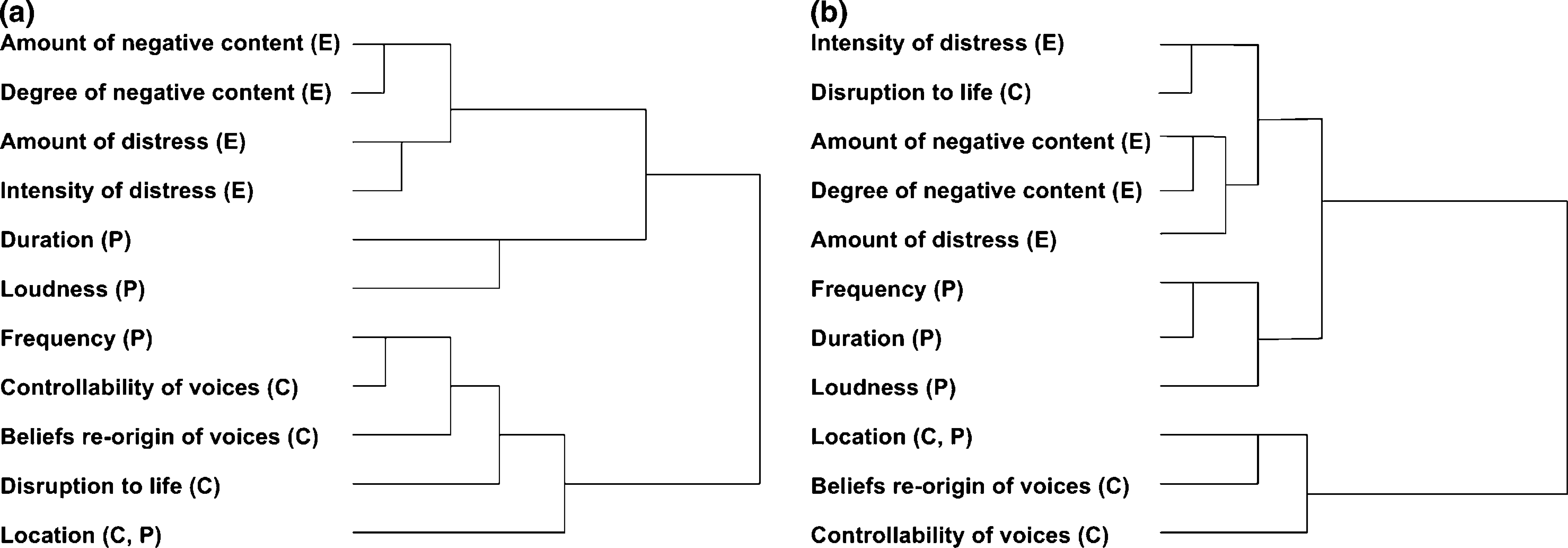
Comparison between first-episode and chronic schizophrenia
In first-episode schizophrenia the cluster structure at baseline was disorganized; thus, no interpretable cluster was observed. This disorganized pattern of baseline cluster structure was also observed in the data from the first-episode group who reported temporary AVHs. In contrast, two main clusters were partially perceptible at baseline in subjects with chronic schizophrenia. The cluster structures of the two groups at 6 month follow-up visits are shown in Figure 2. The distinction between cognitive and emotional clusters became obvious in both groups with treatment, indicating more dynamic changes in cluster structures within the first-episode group compared to the chronic group. Physical characteristics remained unstable from baseline through 6 month follow up. The ‘disruption to life caused by voices’ item was positioned close to emotional variables in both groups after stabilization of the cluster structure was achieved.
Stabilized cluster structures of auditory verbal hallucinations in patients with (a) first-episode schizophrenia (n=21) and (b) chronic schizophrenia (n=19) analysed by hierarchical clustering using Ward's method. C, cognitive; E, emotional; P, physical.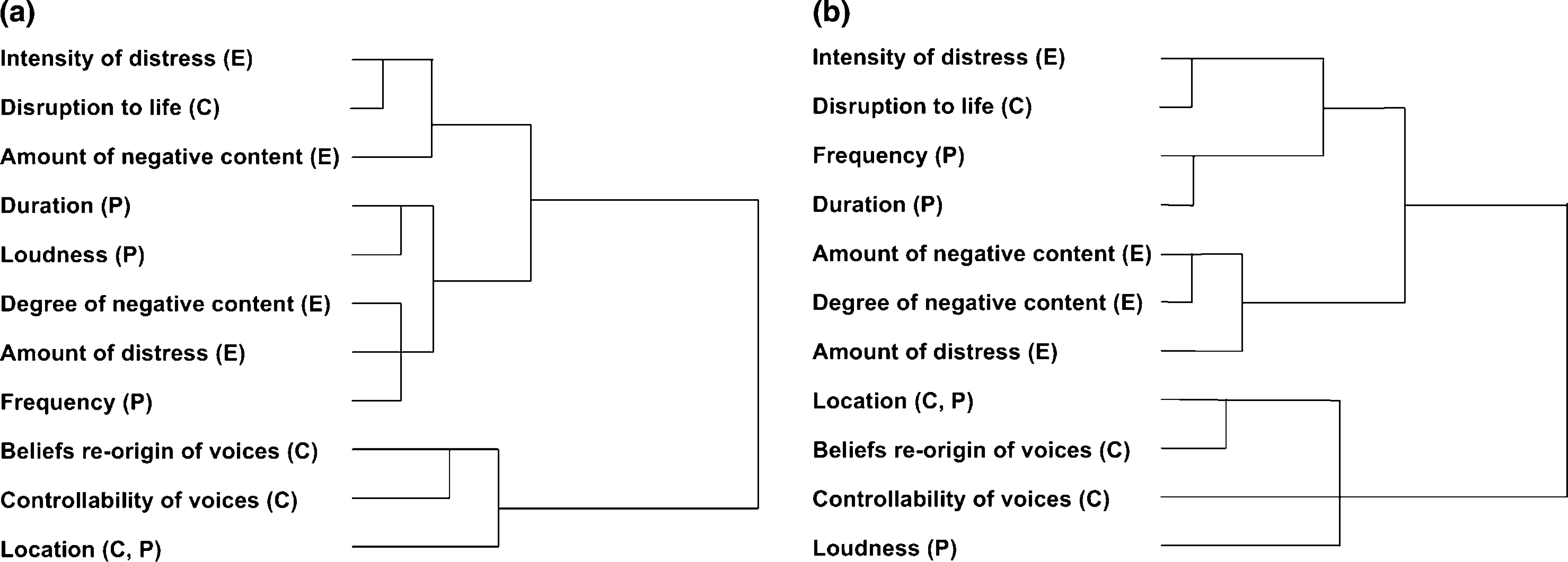
In terms of the Stress and RSQ values, 2-D solution of MDS showed a fairly good fit in both groups at baseline and at 6 month follow up. At baseline, phenomenological variables forming loose clusters were scattered on the MDS map in both groups, but interpretable clusters were noted in the chronic schizophrenia group. The cohesiveness of cluster structure increased in both groups with antipsychotic treatment. Although HC yielded a two-cluster solution as shown in Figure 2, the first cluster showed underlying structure of two large subclusters including items of emotional or physical characteristics. Thus, three circles representing cluster–subcluster structure were imposed on the MDS map to compare local and global properties of MDS configurations between the two groups in detail (Figure 3). The circles were used to indicate the hyperspherical form of clusters produced by Ward's method. Compared to the baseline, the shape of clusters became more spherical, with increased density and decreased variance, thereby indicating coherent and organized clusters. The location of physical variables did not fit into the subclusters of their respective domains in both groups.
Treatment-induced stabilization in 2-D structure for auditory verbal hallucination in patients with (a) first-episode schizophrenia (n=21) and (b) chronic schizophrenia (n=19). Dotted circles indicate underlying item assemblies as assessed by hierarchical cluster analysis.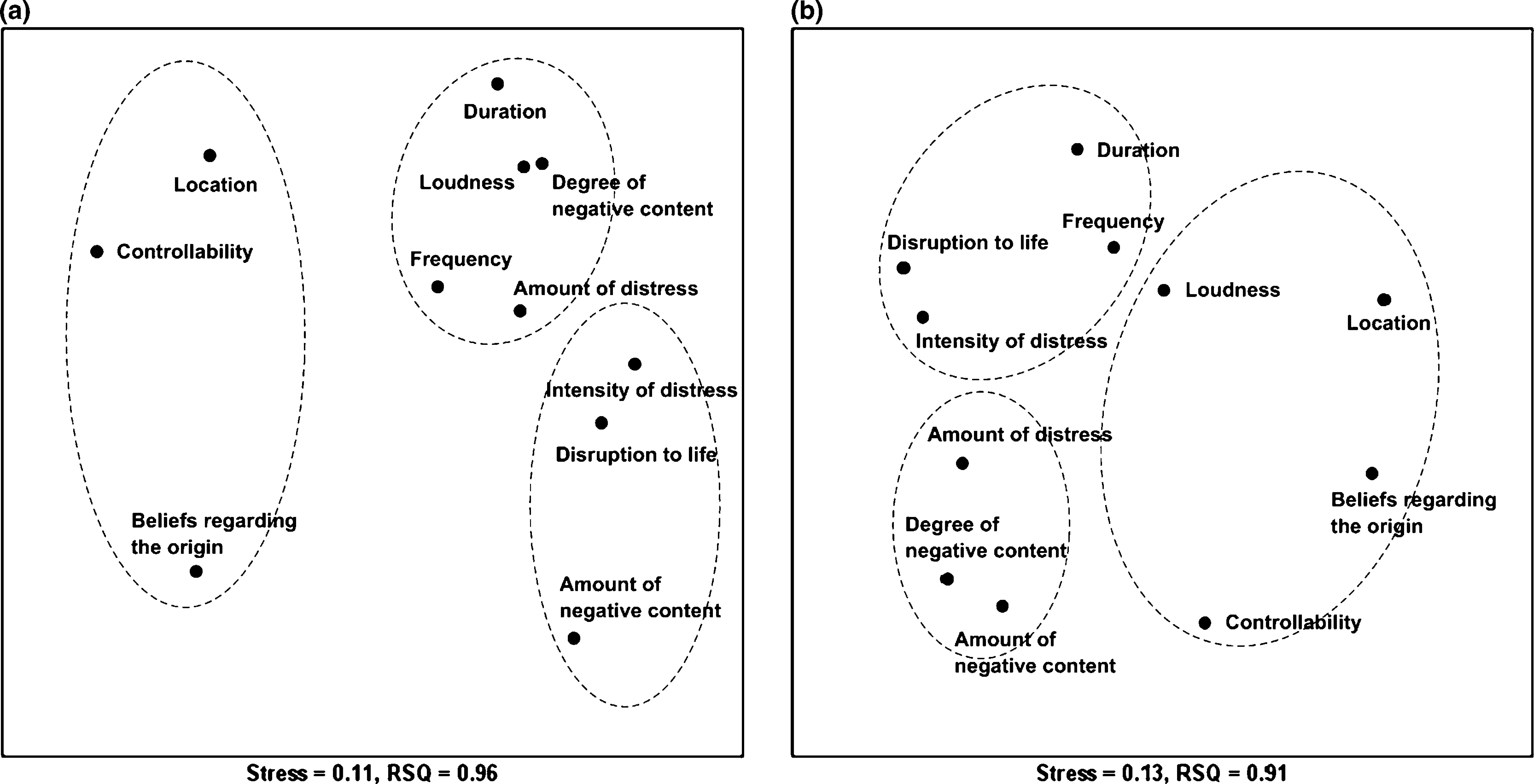
Relationship between cluster structure and symptom severity
The clinical significance of the PSYRATS-AHS clusters was explored in relation to the PANSS and CGI-S. Based on the cluster structure, four items (i.e. ‘amount of negative content of voices’, ‘degree of negative content’, ‘amount of distress’, and ‘intensity of distress’) were summed as the emotional cluster score; and two items (i.e. ‘beliefs regarding the origin of voices’ and ‘controllability of voices’) were summed as the cognitive cluster score. Spearman's rho was used as a correlation coefficient. In first-episode schizophrenia the cognitive cluster score was significantly correlated with the PANSS total score (0.45) at baseline. At 6 month follow up both cluster scores were significantly correlated with the PANSS hallucination item, the PANSS positive subscale, the PANSS total, and the CGI-S score (0.72, 0.66, 0.52, and 0.64 for the emotional cluster; 0.83, 0.73, 0.61, and 0.56 for the cognitive cluster). In subjects with chronic schizophrenia both cluster scores were significantly correlated with the PANSS total (0.53 and 0.59, respectively) at baseline. At 6 month follow up both cluster scores were significantly correlated with the PANSS hallucination item (0.68 and 0.66, respectively).
Discussion
We observed the stability of two main domains (i.e. emotional and cognitive characteristics) of AVHs at an interval of 6 months from the time of baseline measurements. The changes in the configurations of the MDS map indicate the stabilization of cluster structures after 6 months of antipsychotic treatment. Physical characteristics of AVHs (i.e. frequency, loudness, and duration) did not form a stable cluster. Psychosocial disruption was initially associated with frequency and with cognitive interpretations of AVHs (i.e. location and beliefs about the origin and controllability of voices), but it highly correlated with the emotional characteristics of AVHs (i.e. negative contents of voices and distress caused by voices) after the stabilization of cluster structure. This finding is consistent with the results of previous phenomenological studies that suggested the importance of emotional response to persistent AVHs in socio-occupational functioning in patients with chronic psychotic disorders [2]. These changes in the patterns of clustering also suggest the inadequacy of clinical evaluations based on all-or-none phenomena or merely on the physical characteristics of AVHs. In addition, there is a possibility that the cognitive or emotional characteristics of AVHs may have differential effects on psychosocial disruption as a function of the clinical status of the patients.
Subgroup comparisons were performed to assess the effects of the course of illness on the structure of AVHs. The changes in the patterns of clustering and the configurations in the MDS map were similar in both groups, with the exception of the disorganized pattern of clustering in the first-episode group at baseline. As expected, more dynamic changes in the pattern of clustering were observed in the first-episode group. In addition, the results of the correlation analysis suggest differences in the clinical significance of AVHs between the two groups, with greater fluidity of symptom structures during the early course of psychotic disorders.
At 6 month follow up the emotional and cognitive clusters were both well correlated with the overall severity of psychiatric symptoms in the first-episode schizophrenia group. These clusters were significantly correlated only with corresponding PANSS items in the chronic schizophrenia group. These findings may reflect the fluidity of symptom structures during the early course of psychotic disorders.
Since Jaspers first differentiated pseudohallucinations from true hallucinations [26], there have been consistent debates among mental health professionals about the clinical importance of the ‘location of voices’ [26, 27]. The term ‘location’ refers to whether AVHs are located internally or externally. We found that location consistently formed a cluster with two essential items of cognitive interpretation after symptom stabilization: beliefs regarding the origin of voices and the controllability of voices. Internal auditory hallucinations, so-called pseudohallucinations, have been often recognized as either a sign of insight or as evidence of thought disorder [28]; the results of principal components analysis of PSYRATS-AHS suggested the correlation of ‘location’ with the two domains (i.e. emotional and cognitive) simultaneously [8]. A recent study of the phenomenology of AVHs also identified the inner–outer location as an independent dimension of AVHs [10]. Although there is an uncertain association between location and overall severity of AVHs [12], the inner or outer location may carry differential effects on a patient's cognitive interpretation of AVHs [12]. The clustering pattern suggests that internal AVHs would be associated with improved insight and a subjective feeling that the voices are controllable.
We explored the underlying symptom structure of AVHs through HC and MDS. Compared with factor analysis, HC has an advantage in producing non-overlapping clusters presented through a dendrogram, as well as in handling a data set drawn from a small sample [22]. Clustering for this study sample was achieved by classifying 11 phenomenological characteristics of AVHs (i.e. 11 items on PSYRATS-AHS) into meaningful subsets. HC using Ward's method created item clusters of hyperspherical form resulting in the highest internal (within-cluster) homogeneity, thereby maximizing between-cluster heterogeneity [22]. Despite its structure-seeking properties, the overall relationship between items of different clusters cannot be fully explored on HC. In contrast, MDS is generally used to display the degree of similarity of two items as a geometric picture. Therefore, MDS can demonstrate the semantic configuration based on the similarities between pairs of objects (e.g. items or variables) [24]. In the current study each item of PSYRATS-AHS was represented by a point in a 2-D space. Imposing underlying clusters on MDS solution was useful in interpreting the interrelation of items with regards to cluster structure.
The present study had certain limitations. First, the statistical techniques used herein are inherently exploratory and heuristic [23]. Despite enormous information about underlying clusters, HC and MDS are not able to provide the level of probabilistic significance associated with formal statistical inference. Second, the sample size was relatively small because of the research decision to maximize the sample homogeneity. Finally, the effect of antipsychotic treatment was assessed in the absence of a control group.
To date, most studies of the phenomenology of AVHs have been conducted with cross-sectional designs using heterogeneous sample populations without regard for the stage of illness. By using two subgroups at different stages of illness, the present results may help clinicians to understand the patterns of dimensional changes in AVHs during antipsychotic treatment [29]. Clinical assessments that are focused primarily on the physical characteristics of AVHs may fail to detect the changes in essential properties of AVHs. These results lend support to the usefulness of a dimensional approach in assessing subjective experiences of persistent AVHs. Clinical assessment of AVHs can be made not only by using the standard rating scales but also by questioning the subjective experience of AVHs. Exploring emotional or cognitive aspects of AVHs might be of help in improving the therapeutic alliance and planning an effective treatment strategy. Evaluations of the multidimensionality of AVHs would be essential in designing integrative treatment approaches to schizophrenia and in improving the quality of life of patients [30].
Footnotes
Acknowledgements
This study was supported by Grant No. M103KV010012-06K2201-01210 from the Brain Research Center of the 21st Century Frontier Research Program funded by the Ministry of Science and Technology, Republic of Korea. The Ministry of Science and Technology had no further role in study design; in the collection, analysis and interpretation of data; in the writing of the report; or in the decision to submit the paper for publication.
