Abstract
Consciousness takes two forms, transitive and intransitive. Transitive consciousness is a matter of being conscious of something or other whereas intransitive consciousness has no object, as being conscious or awake. Of the different forms of transitive consciousness, perceptual, somatic, kinaesthetic and so on, cognitive neuroscience has concentrated on determining the neural concomitants of perceptual consciousness. To be conscious of a percept is to be aware of it and this requires attending to it. This work sets out a hypothesis as to what brain areas are involved in a schizophrenia subject attending and becoming aware of hallucinations. First, the different areas of cortex that support different visual and auditory illusions of percepts are considered. Next it is argued that endogenous activity in these areas of cortex give rise to hallucinations of percepts that are similar to the percepts that these same areas support during illusions. The basis of such endogenous activity, it is suggested, is to be found in the paucity of afferent synapses to these cortical areas. This may occur as a consequence of loss and regression of synapses due to a degenerative disease or because of abnormal synapse formation and regression during childhood and adolescence, as is likely to be the case in schizophrenia. Finally the neural basis of attention and awareness of these hallucinations are considered for subjects suffering from schizophrenia, and a set of important questions posed that await elucidation through future experimental studies.
In a previous essay in this journal an hypothesis was elaborated that schizophrenia arises as a consequence of a failure of molecules (most of which were originally discovered in the peripheral nervous system) that guide synapse formation [1]. The present essay is concerned with how this loss of synapses could give rise to distortions of consciousness, such as hallucinations, a characteristic symptom in schizophrenia.
Consciousness
There are two forms of consciousness, transitive and intransitive [2]. Transitive consciousness is a matter of being conscious of something or other, or of being conscious that something or other is thus or otherwise. Intransitive consciousness, by contrast, has no object. It is a matter of being conscious or awake, as opposed to being unconscious or asleep. To say, on awakening after an operation, ‘I am conscious’ does not impress as an occasion during which something mysterious has occurred. There is nothing intrinsically private to one when turning to the nurse and saying ‘I am now conscious, can I have a cup of tea?’. If a veterinarian anaesthetizes a cat, we say it is now unconscious, and after the operation, that it has gained consciousness. Only a sentient animal can be ‘unconscious’. This particular form of consciousness we may refer to as intransitive, that is, it is something that a person or animal may lose (as when being anaesthetized) and then subsequently recover (as when regaining consciousness as the anaesthetic wears off). To be unconscious is to be incapable of perceiving the environment and to be insensible to stimuli. Being unconscious and being asleep are clearly different because in the latter we can easily be awakened.
During the waking hours we experience, are conscious of, different things at different times, for a period. Such consciousness, when our attention is caught by something and held, so we become aware of it, is clearly different from intransitive consciousness, for it has an object and so is called ‘transitive consciousness’. There are many forms of transitive consciousness, such as perceptual consciousness, where one becomes conscious of what one perceives, as becoming conscious of the ticking clock, which is not the same as perceiving the ticking clock; somatic consciousness, to become conscious of a sensation such as the pain of a toothache capturing our attention; kinaesthetic consciousness of one's cramped limb; affective consciousness, as becoming conscious of one's current emotions, for example anger or annoyance; reflective consciousness, to become conscious of something that is not present, such as an honour due to one; conscious of one's actions, as in cracking a joke; conscious of one's motives, to become conscious of feeling jealous of Daniel for the honours he has received; self-consciousness, as in becoming aware of being the centre of attraction and on introspective reflection on one's motives, successes and failures. Clearly all these forms of transitive consciousness are dependent on intransitive consciousness. The concept of transitive consciousness is clearly allied to that of becoming aware, of noticing, that is, of having one's attention caught. Paying attention often involves taking an interest in something, thinking about it. Lack of attention is linked to unawareness, failure to notice, absent mindedness. That which makes what we do a case of attending in one or other of the forms already enumerated is that when we do it, something is made the centre, object or topic of whatever it is we are doing.
Cognitive neuroscientists attribute consciousness to humans and other animals on the grounds of their behaviour in the context of their lives. They investigate the neural networks in the brain that make it possible for an animal to have and exhibit forms of feeling, perceiving and purposiveness. Neuroscientists would regard it as unintelligible to claim that an animal that manifests powers of perception, memory and emotions is unconscious. By establishing inductive correlations between such facts of consciousness as seeing features of different kinds in the visual field and the firing of certain neurons, neuroscientist infer that when these neurons are observed to fire, the animal, possessing normal visual capacities, sees. The question does not arise as to whether the animal is conscious because it is clear that it enjoys perceptual and affective experiences. The challenge to cognitive neuroscience is to first establish the neural correlates of various forms of perception, sensation, affective feeling and mood. Then unravel the neural activity accompanying, attending to, and becoming aware of something within these psychological categories. Here the focus is then to determine the neural activity associated with attention and awareness. In cognitive neuroscience the experimental paradigm defines what the experimenter means when he uses the terms ‘attention’ and ‘awareness’. It is then important to consider the relationship between such words and phrases used in the laboratory and those in general parlance in order to ensure that incoherences do not arise when in general discussion it is claimed that the neuroscientists have discovered the neural concomitants of ‘attention’ and ‘awareness’.
Perceptual consciousness
Of the different forms of transient consciousness, cognitive neuroscientists have mostly sought the neural concomitants of perceptual consciousness. Perception per se is not a form of consciousness, but becoming aware and retaining an awareness is. Whatever one is perceptually conscious of one is also aware of. It should be noted in passing, however, that one may be aware of things of which one is not conscious, for example the things that one has been informed of, but in this case we are not talking of perceptual awareness or consciousness that is the subject of the present considerations. The phrase ‘to become conscious of’ belongs to a small family of cognitive verbs, that includes ‘to become aware of’, ‘to realize’, and ‘to notice’.
Awareness and perception
To be successful in a perceptual activity such as looking, scenting, tasting, listening or sniffing amounts to seeing, smelling, tasting, hearing or smelling; that is, recognizing, detecting, discerning or distinguishing percept. To become and to be conscious of such things as colours, smells, and sounds, is not an alternative to perceiving them. Furthermore, what one is perceptually conscious of is not something over and about some of the things one perceives. It is only those things that we perceive and realize we perceive that are things of which we are conscious. It is obvious that whatever one is perceptually conscious of, it is something perceived, although not everything one perceives is something of which one is conscious. For example while reading the above sentence I am not conscious of reading, which is undoubtedly a process that involves perception of the written word. That is, much of what is perceived is not even noticed, let alone attended to, so we are not conscious of it.
The act of perception is accompanied by activity in specific sensory areas of the cortex, each associated with a specific sensory modality. The establishment of this fact was a major triumph of 20th century neuroscience [3]. Such cortical activity is a necessary condition for us to be able to act, while being unaware or conscious of the action, most of the time, as when driving a car. Certain specific sensory areas must of course be active if we are to be aware or conscious of driving the car, as would occur if a car in front of us suddenly stopped and we had to avoid a collision. Cognitive neuroscientists have shown that conditions of visual awareness are accompanied by enhanced activity in fronto-parietal areas of the cortex, distinct from those involving attention. The question arises here as to whether, besides the parts of the nervous system supporting the perceptual faculties engaged in whatever one is perceptually conscious of, there could be said to be a single part of the cortex that supports our capacity to be aware or conscious of that which is perceived. This is probably not the case. When seeing Daisy in the corner of the room, without becoming aware of her, and therefore walking towards her, activation of visual cortical areas 17, 18 and extending down into the temporal cortex are active but not the fronto-parietal area of the cortex. If, in contrast, on entering the room we see Daisy and walk towards her, that is we are aware and conscious of her, the fronto-parietal area of the cortex is active. Binocular rivalry experiments are ones in which we do not concentrate our attention on an object, and so become conscious of it, but rather the object imposes itself upon our attention. Seeing Daisy on entering the room is not necessarily to become aware of Daisy, but it is a condition of being aware of her. Seeing the change in perceptibilia during binocular rivalry experiments is what catches our attention and therefore awareness and so, as experiments indicate, is accompanied by activity in the fronto-parietal area of the cortex.
Attention and perception
We cannot remain conscious of something that no longer holds our attention or occupies our thoughts. So we are perceptually conscious of that which catches and holds our attention. The emphasis here is on that which ‘captures and holds our attention’ because if we are looking at a painting or talking to somebody we are not conscious of the painting or of the person unless our attention is focused or riveted on the painting or the person. So what we become and are conscious of is not that which we intentionally attend to but rather that which catches and holds our attention. While looking at the texture of the paint on the surface of a wall in search of signs of decay one may become conscious of the poor rendering of a small section of mortar but not of the surface of the wall being scanned. In other words we do not become conscious of the particular attribute of an object to which we are intentionally attentive but we do become conscious of those features of the object that have brought about a shift in our attention and which then holds our attention. It is that which intrudes and impresses itself on us, and with which we are not purposefully occupied at all, such as a distracting feature that catches and holds our attention, like the creaking of a door or the smell of a coffee aroma. It is this that we become conscious of. The phrase ‘conscious of [the name or description of a material object or person]’ specifies what object or person has caught and held our attention.
When cognitive neuroscientists attempt to measure the neuronal concomitants of ‘attention’, involving visual or auditory perception, they use experimental designs that aim to distinguish between attending to the object or person and being aware of the object or person that is being conscious of them. In this case, when a subject is asked to attend to the left-hand lower quadrant of a blank screen occupying the visual field, an intentional act to so attend is carried out that requires us to rivet our attention on that particular quadrant. When this is performed, as we will see in the following section, fronto-parietal areas of the cortex are excited, and these areas overlap for attending either in the visual or auditory modalities.
Visual and auditory illusions
The question asked here is whether areas of cortex that subsume specialized sensory modalities must be active in order to experience, be aware of, an illusion in that modality. The answer to this question is necessary before an enquiry is made into the neural concomitants of hallucinations.
Visual illusions
Visual illusions are distortions or misconceptions of a visual scene currently being viewed. Psychologists use several different methods to achieve such illusions. Chief among these are the awareness of illusory objects as in the Kanizsa illusion, in which one seems to perceive a non-existent rectangle emerging from four corner discs; the McCulloch after-effect in which one perceives, while viewing achromatic oriented contours, illusory colours; the motion after-effect in which, after prolonged viewing of the motion of an object in one direction, a stationary object subsequently viewed appears to be moving in the opposite direction; and synaesthesia, such as seeing colours when viewing achromatic letters. All of these conditions produce awareness of an illusory visual phenomenon without any changes in the objects being viewed. Here evidence is presented that shows that there is a very good correlation between the type of reported awareness of an illusion and activity in the appropriate specialized area of visual cortex that is normally activated when actually viewing the substance of the illusion.
Kanisza illusions
The Kanisza figure consists of four corner discs and some lines, lying on a ground of even light intensity. One is aware, however, of an illusory bright square, which seems to lie over the other patterned elements. At the time of becoming aware of the illusory contours in a newly presented Kanisza figure, records of primate cortical electrical activity show that it begins in neurons in the primary visual cortex (V1) as well as in the surrounding (extrastriate) cortex (V2), but with different delays. In V1 superficial cortical layers the delay is 100 ms and in the deep layers the delay is 120–190 ms, whereas in V2 superficial layers the delay is much shorter than in V1, namely 70 ms, and in deep layers, 95 ms [4]. As these neurons fire when the primate signals it is aware of real contours, as well as illusory contours, then awareness of illusory contours is accompanied by appropriate activity in those parts of the visual cortex that are active during normal vision of the objects projected onto the retina. The earlier firing of neurons in higher visual areas such as V2 (i.e. further along the visual pathway from the retina), compared with lower ones such as V1, suggests a top-down effect; that is, the illusion is accompanied by interaction between higher and lower areas of the visual pathway.
McCullough illusions
These arise when looking at a test stimulus consisting, for example, of a pattern of black and white lines, some vertical and some horizontal. Before doing so the visual system is adapted by looking for 10 s at a pattern of red vertical lines and then for 10 s at a pattern of green horizontal lines (each against a black background), with this procedure repeated by alternating between the two for approximately 5 min. On then looking at an achromatic test stimulus one is aware that the vertical lines are faintly greenish and the horizontal lines faintly reddish (i.e. the white areas surrounding the vertical lines are tinged green and the white areas surrounding the horizontal lines are tinged red). The coloured illusions have then become ‘tied’ to the opposite orientations of the coloured lines used in the conditioning period. Awareness of these illusory effects correlates with increased activity in extrastriate (outside V1) visual areas involved in colour vision, namely the lingual and fusiform gyri (V4) in humans, as has been determined using functional magnetic resonance imaging (fMRI) [5], [6].
Movement illusions
After watching movement in a single direction for a long time and then subsequently looking at a stationary scene it seems to move in the opposite direction. This is often referred to as the ‘waterfall effect’ because on staring at a waterfall for a prolonged period of time and then looking at a stationary image, such as the trees at the base of the waterfall, one is aware of them moving upwards for a few seconds. In humans the visual cortical area V5 is activated during motion in the visual field of a subject. Using fMRI shows that such motion in one direction leads to a gradual decrease in the activity of V5, whereas following such decreased activity, if a motion in the opposite direction is presented a vigorous response is elicited from V5 over and above that obtained during the initial motion [7]. It is this imbalance in the neuronal population level responses that favours the excitability of the non-adapted neurons during the test period, which underlies the movement after-effect.
Synaesthesia
Subjects with synaesthesia have the illusory experience of a perception or a sensation in a modality different from the one being stimulated as a consequence of attending to something. For example, grapheme-colour synaesthetes experience particular colours associated with specific achromatically presented numbers or letter characters. In colour-hearing synaesthesia subjects have colour experiences when hearing spoken words. During speech the cortical colour centre in such subjects, V4, is active according to fMRI, but it is not active when imagining colours [8]. These fMRI studies also show that V4 is active when graphemes of words or numbers are presented to grapheme-colour synaesthetes [9]. Of interest in this case is that the area of cortex activated during perception of word forms is adjacent to the colour area in V4 (so-called hV4; [10]). It has been proposed that during normal development these two areas are connected and only later removed by the process of synapse regression [1].
Young infants confuse the inputs from different senses, suggesting that at an early age we are all synaesthetic (reviewed in [11]). For example, at 12 months of age an infant will look longer at a novel object rather than one they had explored with their mouths earlier, whereas preterm 12-month-olds do not [12]. It is probable that the baby is just developing separate schemas for listening and seeing and manipulating, suggesting that during this period each sensory modality does not involve sensory receptors uniquely projecting to particular sensory cortices. This is supported by observations made during early infancy that show that electrically recorded evoked responses to spoken words can be recorded from the occipital (visual) cortex as well as over temporal (auditory) cortex [13]. Visual stimuli give rise to evoked responses recorded over other areas of cortex than those devoted to vision [14]. This suggests that a newborn does not have distinct sensory modalities; rather, these are intermingled in a synaesthetic confusion. If this is the case then a normal newborn perceives a jumble of sensations in the context of a particular perception.
Mature synaesthetic subjects may fail to have such connections between primary visual cortex and auditory cortex removed, so that they experience colours when viewing numbers or letters [11], [15] (for a recent review, see [16]). There is anatomical evidence that transient synaptic connections exist in the monkey's brain at birth [17–20], although there is yet no evidence for such connections between primary visual cortex and auditory cortex [21]. Evidence exists for the regression of most of the synaptic projections from higher areas to V4 during development [22], although it is not known to what extent failure of synapse regression occurs in synaesthetes, with the potential to leave different regions of the brain that subserve different modalities directly connected (for a review of these considerations, see articles in [23]). In summary, although there is at present both behavioural and electrophysiological evidence for synaesthesia in the newborn, there is as yet no anatomical evidence to suggest transient direct connections between different parts of cortex subserving different sensory modalities, which are permanent rather than transient in synaesthetes.
Binocular rivalry
Although not an illusory phenomenon, binocular rivalry is of interest in enforcing the argument that perceptual experiences involve activity in the appropriate part of the brain that supports that kind of experience. If two different images are presented stereoscopically, then the view presented to one eye will dominate that of the other for a period of time before it alternates. In such binocular rivalry, if a stimulus consisting of dissimilar images is presented separately to each eye, then one is aware of perceptual alterations between each monocular view every few seconds [24]. Of interest is that if rival house and face stimuli are presented, the switch in awareness of these occurs at the same time as the fMRI signal increases in either the fusiform face area or the parahippocampal place area, whichever is appropriate for the experienced image [24].
Auditory illusions
Auditory cortex is composed of Heschl's nucleus, the planum temporale, the lateral superior temporal gyrus, the dorsal bank of the superior temporal sulcus, the ventral bank of the superior temporal sulcus and the lateral middle temporal gyrus (Figure 1b) [25], [26]. Speech perception studies using fMRI suggest that the part of the auditory cortex concerned with unmodulated broad spectrum noise is to be found in Heschl's gyrus, whereas frequency-modulated tones rather than noise are to be found in planum temporale as well as the dorsolateral superior temporal gyrus; that is, in areas surrounding Heschl's gyrus (Figure 1b) [27]. The part of the auditory cortex activated by speech (i.e. containing phonetic information), rather than by frequency-modulated tones is found in the superior temporal gyrus that extends into the superior temporal sulcus, with only the anterior part of this sulcus active if the phonetics are intelligible [28]. Interestingly this sulcus also shows sustained activation during silent rehearsal of speech [29]. There is then a hierarchy of auditory processing, starting in medial and intermediate parts of Heschl's gyrus, sometimes called the primary auditory cortex, to regions that support advance phoneme–auditory processing in the superior temporal gyrus, including the superior temporal sulcus [30].
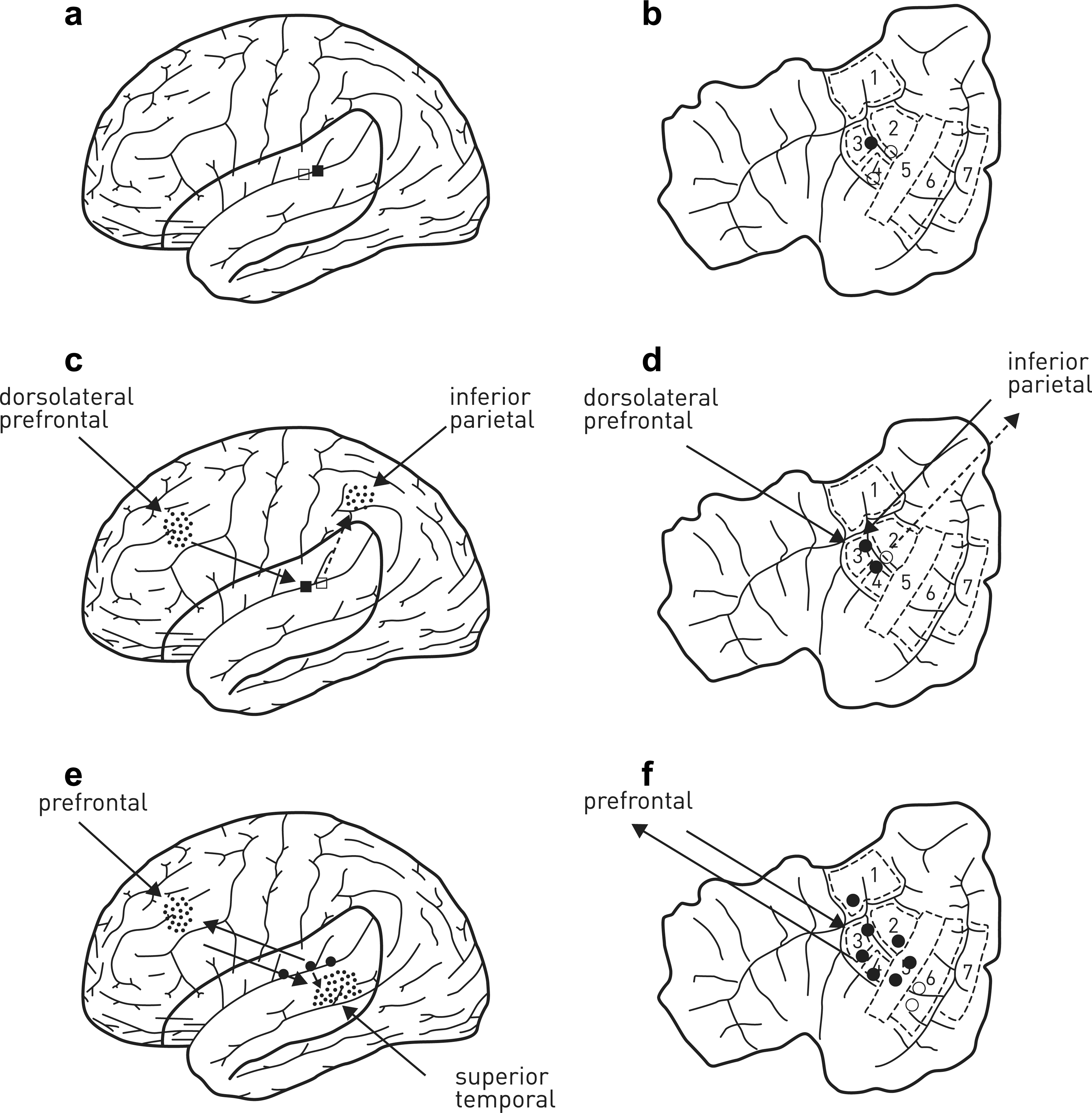
Attention and awareness of auditory percepts. In the absence of attention, (a) activity in auditory cortex is low and (b) confined to the early auditory pathway, namely Heschl's gyri (areas 3,4). (c) Excitation of auditory cortex on hearing an auditory percept may be accompanied by activity increases in the inferior parietal and the dorsolateral prefrontal cortex if the percept is attended to, with the possibility that salience of the percept activates inferior parietal cortex whereas the intention to attend to the percept involves activation of the dorsolateral prefrontal cortex. Such attention leads to enhanced excitation in Heschl's gyrus (areas 3,4 in d) and to some extent in the planum temporale (area 2). If attention is caught and held, as in (d), it is possible that activity in the superior temporal and prefrontal cortex is enhanced (e). Whether there is a threshold at which activity in these areas increases explosively, like that in the brain areas subserving awareness in the visual cortex (Figure 2c), is not known. (f) What is known is that the growth of activation in superior temporal and prefrontal cortex leads to the enhancement of activity in auditory cortex, spreading from Heschl's gyrus (areas 3,4), into all of planum temporale (area 2), lateral superior temporal gyrus (area 5), as well as perhaps into the dorsal and ventral banks of the superior temporal sulcus (areas 6,7) and the anterior supramarginal gyrus (area 1). The increase in the extent of recruitment of extra-Heschl's gyrus and planum temporale as well as of activity in the prefrontal and superior temporal cortex is associated with being conscious of the auditory percept; that is, the subject reports being aware.
Continuity illusion
If a sound being listened to is interrupted by silence then it is perceived as discontinuous. What perhaps is not realized is that if noise replaces the silent period then the sound being listened to is often heard as being uninterrupted, depending on the frequency characteristics of the noise with respect to the sound. So when parts of a speech are deleted comprehension declines, whereas if the deleted parts are filled in with appropriate loud noise speech comprehension is restored. This phenomenon in the auditory modality is similar to that of visual illusory contours. Studies using fMRI have been made of activity in the cortex of subjects during trials in which they perceive this illusory tone. The perception of the illusion correlates with changes in neural activity in early auditory cortical regions in Heschl's gyrus [31]. Experiments that show the importance of primary auditory cortex during experience of auditory illusions were designed in which a target tone, varying in frequency between 500 Hz and 3223 Hz, is interrupted for 600 ms by a noise burst that contains a notch or band-stop filter, parameterized across zero, 0.25, 1.25, and 2.0 octaves. The noise and the notch are centred on the target frequency. At the highest masking level, when there is no notch, the interrupted targets are perceived as continuity illusions, which are accompanied by changes in Heschl's gyrus. It is very likely that these fMRI responses reflect changes in neuronal activity because neurons in monkey primary auditory cortex (A1) fire impulses during continuous presentation of the tone interrupted by intense noise, as if they were responding to presentation of the tone without any interruptions [32].
Auditory illusions and mismatch negativity
Mismatch negativity is an event-related potential that is recorded when there is a change in a standard continuous regular auditory stimulus sequence to a deviant sequence. This potential is very likely generated pre-attentively, without any associated awareness, in auditory cortex [33]. Mismatch negativity can reflect the neural concomitants of an auditory illusion, as is shown in the following [34]. Consider experimental conditions in which a tone stimulus is interrupted both in the prior standard and later deviant intervals, and one in which the tone is continuous in the standard interval but interrupted in the deviant interval. Noise bursts are introduced that are filtered in some cases in the same frequency range as the tones, and in other cases in a higher frequency region that does not spectrally overlap with the tones. The mismatch negativity in the case of a continuous tone in the standard interval, interrupted in the deviant interval by a noise burst in a different frequency region from the tone, gives the largest mismatch negativity (here the percept changes from a continuous tone to an interrupted tone). This mismatch negativity is larger than that in the case of a continuous tone in the standard interval followed by a discontinuous tone filled in by a noise burst in the deviant interval (here the percept does not change from a continuous tone). For the cases involving discontinuous tones in the standard intervals, when the gap is filled by a noise burst for which the frequency is centred about that of the tone in the deviant interval (here the percept is one that changes from that of an interrupted tone to one in which the tone is continuous), the mismatch negativity is greater than that for the case in which the noise burst is not centred on the tone frequency (in this case the percept is one of a discontinuous tone followed by a discontinuous tone). These results support the idea that mismatch negativity gives a measure of the continuity illusion.
The mismatch negativity response then gives a measure of the deviance in the auditory modality between the memory of a standard sound and that of a subsequent deviant sound. It is accompanied by activity in the dorsal superior temporal gyrus rather than in the ventral superior temporal gyrus that is activated by shifts in attention to task-irrelevant auditory factors [35].
Summary
The question posed at the beginning of this section on visual and auditory illusions can be answered in the affirmative, given the observations outlined in the previous sections. The area of cortex that subserves a specialized sensory modality must minimally be active in order to experience, be aware of, an illusion in that modality. Note that this establishes only the cortical activity that is necessary to be aware of an illusion but does not claim that such activity is sufficient to be aware of an illusion. The sufficient cortical activity for supporting awareness is considered in a following section. This enquiry into some of the neuronal concomitants of illusions provides the necessary foundation for consideration of the neuronal systems supporting hallucinations that are now described.
Visual and auditory hallucinations
Visual hallucinations
Claude Bonnet hallucinations
Visual hallucinations represent images generated by endogenous electrical activity unrelated to the objects currently being viewed, whereas visual illusions, as we have seen, are distortions or misconceptions of a visual scene currently being viewed. Claude Bonnet syndrome involves visual hallucinations that cover the range from simple flashes to the appearance of scenes, animals and people. These generally occur in subjects that have a deteriorating visual pathway, such as occurs in macular degeneration, optic neuritis and retinitis pigmentosa. The pathological visual experiences of these subjects are associated in such a way that they can be grouped into categories: hallucinations of grotesque, disembodied and distorted faces with prominent eyes and teeth; hallucinations of extended landscapes; of small figures in costumes wearing hats; and hallucinations of objects remaining in the field of view after the patient has looked away or returning after the patient has looked away (so-called perseveration and delayed palinopsia [36]).
It was established in the section on visual illusions that there are correlations between particular kinds of visual illusions and activity in the appropriate visual centres that normally subserve non-illusory phenomena of the same kind. It is anticipated then that visual hallucinations involve something going awry in these centres. This is the case because the different categories of hallucinations have been correlated with spontaneous activity in the particular parts of cortex known to support capacities for identifying faces, landscapes/figures, and those utilizing eye-centred reference frames. These are the superior temporal sulcus, ventral occipito-temporal lobe and the visual parietal lobe [36]. For example fMRI studies of subjects experiencing hallucinations involving faces show heightened activity in the fusiform face area [37].
Auditory hallucinations
Types of auditory hallucinations
Auditory hallucinations, generated by spontaneous endogenous electrical activity, cover the range from the less often experienced simple clicking and humming sounds to the more frequently experienced hallucinations of haunting voices, of voices telling one where to go and what to do as well as the voices of children crying (table 7 in [38]). These hallucinations generally occur in subjects with an identifiable lesion somewhere in the auditory pathway [39], typically due to haemorrhaging or to a glioma in sites such as the superior temporal gyrus [40] (see also table 5 in [38]). Such auditory post-lesion hallucinations are very similar to those first reported for patients consequent on direct electrical stimulation of their exposed temporal cortex during operations, often for intractable epilepsy [41]. It is interesting in this regard that the superior temporal gyrus is established as involved in speech perception [25] (for a review see [30]). Auditory hallucinations also occur in approximately 10% of the normal population for whom no lesion has been identified. Half of these have hallucinatory experiences each day, at a rate higher in adolescence than in later life [42], [43].
Auditory hallucinations in schizophrenia
The most common and positive symptom in schizophrenia, involving approximately 70% of all patients, is auditory hallucinations of speech [44]. Positron emission tomography studies show that speech hallucinations experienced by schizophrenia subjects are accompanied by excitation in the auditory cortex [45]. Studies with fMRI show that in schizophrenia not only is the primary auditory cortex in Heschl's gyrus active, as it is in auditory illusions, but also the auditory cortical areas in the superior temporal gyrus [46] (for a review see [40]). Confirmation of the importance of this gyrus in auditory hallucinations is provided by observations using trans-cranial magnetic stimulation in which the magnetic coils are arranged in such a way as to direct the field into the left superior temporal gyrus. Such fields suppress auditory hallucinations for periods of up to 3 months, bringing great relief to patients [47]. It is interesting to note that the part of the secondary auditory cortex in planum temporale, next to Heschl's gyrus (Figure 1b), is activated according to fMRI, on the perception of real voices located in external space in normal subjects [48], [49] but not in schizophrenia subjects [50].
Summary
Both visual and auditory illusions involve activity triggered by percepts in centres of visual and auditory cortex that subserve normal visual and auditory percepts. The observations reported here support the hypothesis that hallucinatory visual and auditory experiences arise from spontaneous endogenous activity in these centres, unrelated to percepts. Of great interest is that identifiable lesions in specific centres within the visual or auditory pathways give rise to hallucinations of a type found in subjects with schizophrenia, such as of grotesque, disembodied and distorted faces, as well as of voices telling one where to go and what to do. This suggests the hypothesis that lesions in specific centres, be they molecular or cellular, are responsible for different experiences during hallucinations in schizophrenia.
Deafferentation hypothesis for visual and auditory hallucinations
It is most likely that visual and auditory hallucinations involve experiences that arise as a consequence of spontaneous activity in these regions of the cortex that subserve the normal experience of percepts. Consideration is now given to the mechanisms that might give rise to this spontaneous activity, especially in the context of deafferentation of neural networks in schizophrenia [51].
Deafferentation of visual centres
Burke has put forward a deafferentation hypothesis for Claude Bonnet hallucinations in order to explain why certain parts of visual cortex are spontaneously active during these hallucinations; that is, engage cortical networks in bursts of activity without an input [52]. He points out the similarities between hallucinations and local seizures caused by bursts of activity originating in a group of neurons that have been isolated either by deafferentation or by trauma or tumours. It is known that almost any region of cortex can be made hyperexcitable by partial or complete isolation [53]. Burke's hypothesis, which is akin to that suggested some time ago by Konorski [54], postulates that hallucinations can be regarded as local paroxysms in the sensory systems arising from deafferentation. Such deafferentation may occur as a consequence of age-related degeneration, direct injury to the cerebral cortex or more peripheral visual structures or, in the case of hallucinations in a disease such as schizophrenia, to failure of synapse formation during childhood and excess regression of synapses during adolescence [1].
Deafferentation of auditory centres
Studies with fMRI show a 20% reduction of grey matter in the planum temporale of patients with schizophrenia [55], probably due to a loss of synapses as indicated by the decrease in synaptic proteins [56] (for review see [1]). This loss in planum temporale is likely to have implications for the generation of auditory hallucinations, because it is known from fMRI studies that the planum temporale is critically involved in the perception of real voices located in external space. It is therefore of considerable interest that there are correlations between the reduced size of planum temporale in schizophrenia subjects and the extent of auditory mismatch [57], as well as between the extent of grey matter loss in Heschl's gyri and mismatch negativity [58]. Deafferentation of planum temporale in schizophrenia subjects, due to excess loss of synapses during adolescence, or indeed failure to form adequate numbers of synapses during childhood, may lead to spontaneous activity in the planum temporale, giving rise to auditory hallucinations (for a model of this process see [59], [60]).
If the loss of synapses is taken as a measure of the onset and development of the frequency of auditory hallucinations then the number of synapses may be used to give a measure of the development of schizophrenia. As the extent of synapse loss increases, primarily during adolescence, a subject either enters the prodromal period without a later full onset of schizophrenia, or enters this period and proceeds into a mild onset, or passes through the prodromal period to onset and maximal expression of the illness. These suggestions may be compared with those in which the rate of synapse loss in schizophrenia is the same as that of normals during the adolescent period, but in the case of schizophrenia it follows a failure of initial synapse formation during early childhood [61] (for a review see [1]). In this case mild, moderate and severe schizophrenia still arises, but at later times during development. It is important to determine whether the fewer synapses found in the planum temporale of subjects with schizophrenia occurs because of a failure of synapse formation or excess synapse regression.
Summary
Consideration of visual and auditory illusions as well as hallucinations in the previous sections led to the hypothesis that hallucinations in schizophrenia arise because of a lesion in specific cortical regions that give rise to the components of the hallucination. It is suggested in this section that the lesion is one that constitutes deafferentation of specific regions. Whether this takes the form of loss of the different afferent connections to the regions or to a general loss of synaptic connections within the regions, or both, remains to be determined.
Determination of the brain areas involved in ‘attention’
Visual attention
To be conscious, that is, to be aware, is to have one's attention caught and held. But one can be attentive without being aware. Indeed separate neural networks subserve visual attention, and these networks are found in the fronto-parietal region of the brain, centred in the lateral and medial sub-regions. This is shown by experiments in which brain activity is recorded with fMRI from subjects performing a cued spatial attention task. In this task single letter cues (L, R, P) are given at central fixation that instruct the subject to either covertly attend to a location in the left (L) or right (R) lower visual field or to detect a possible (P) faint dot target there or to interpret the cue but then not attend to the target [62]. In some trials there is no target, so that any brain response is only due to the cue. At the end of the trial the letters ‘REP’ are presented in the mid-line lower visual field, at which point the subject presses a button to indicate target detection on that trial. The following named procedures were used in these experiments: ‘interpret-cue’, the letter ‘p’ is presented instructing the subject not to attend to a target on that trial; ‘attention-cue-plus-target trials’, target presented at randomized times (900–1900 ms) after the onset of the cue; ‘attend cue only trials’ and ‘interpret cue only trials’, no target presented so the brain response is due to the cue only; ‘no stimulation trials’, periods of fixation only. The images obtained with fMRI in these different trials after being subtracted from each other, show that medial frontal and parietal regions are involved in attention (Figure 2b). All of the interpret cues and other interpret-cue-only trials activate the parietal and frontal areas (Figure 2b). Frontal and parietal regions that are activated by the interpret-cues are lateral, whereas more medial regions are activated by the attend-cues relative to the interpret-cues. Thus specific attentional orienting is accompanied by activity in medial frontal and parietal regions (Figure 2b). This functional specialization for the different aspects of cue processing in the lateral and medial subregions of the superior frontal and parietal cortex is shown by the fMRI signal time courses in the superior frontal and parietal cortex during these trials. The amplitude of these changes is much larger in medial areas for the attend-cue trials compared with the interpret-cue trials. In contrast, the responses to the interpret-cue trials were nearly as large as those for the attend-cue-only and the attend-cue-plus-plus-target trials in the lateral areas, whereas the interpret-cue responses were smaller than for the attend-cue trials in the medial areas. The conclusion of these studies attempting to identify the regions of the brain concerned with visual spatial attention, summarized in Figure 2(b), is that these are to be found as medial subregions of the medial frontal (including frontal eyefields) and parietal cortex [63–65] (for a review see [66]).
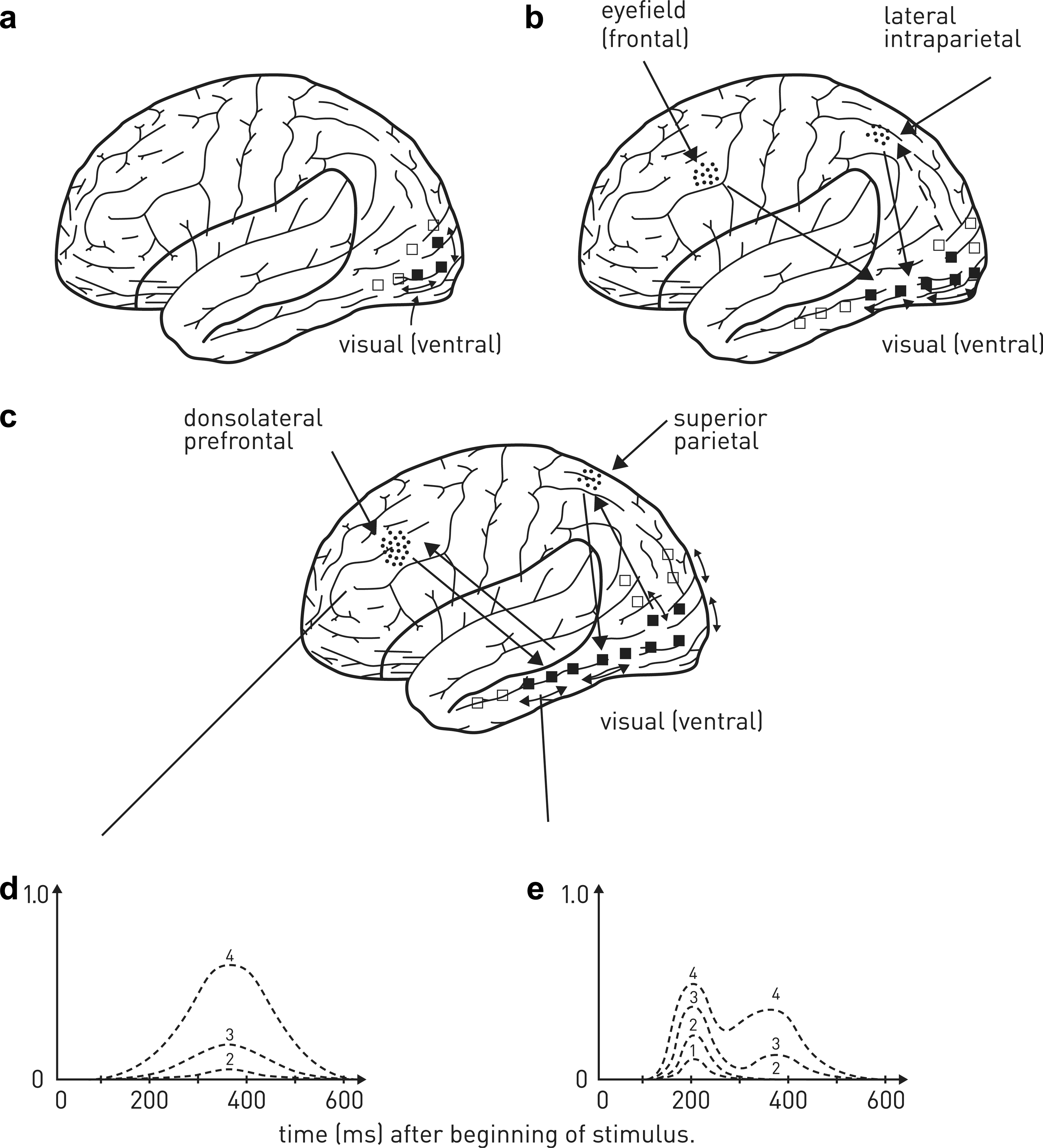
Attention and awareness of visual percepts. (a) In the absence of attention, percepts evoke only slight activity in the visual extrastriate (outside primary visual cortex, V1) pathways, subserving, for example, the detection of motion or colour. (b) If the visual percepts are salient, networks in lateral intraparietal cortex are activated that most likely increase the extent of the extrastriate visual pathway activity; slightly later the frontal eyefield attentional area of the brain is active that also contributes to activity in the extrastriate pathway. (c) If attention is caught and held as in (b) it is possible that activity in the superior parietal and dorsolateral prefrontal cortex is enhanced, as is shown in (d) for the dorsolateral prefrontal cortex. This is graded with an increase in extrastriate visual cortex activity (e, first peak near 200 ms; signals 1,2), until a threshold is reached (d, signal 3), at which time there is a sudden non-graded large increase in activity in dorsolateral prefrontal cortex (signal 4, d,e) as well as in superior parietal cortex, which drives a very large late increase in extrastriate visual cortex (e; approx. 350 ms; signal 4). The increase in the extent of recruitment of extrastriate visual cortex and of activity in the dorsolateral prefrontal and superior parietal cortex is associated with being conscious of the percepts; that is, the subject reports awareness of these.
Within this parieto-frontal network it is likely that visual target selection involving attention to a salient target is associated first with firing of neurons in the lateral intraparietal area (Figure 2b) [67–69]. In contrast to this, top-down selection, involving volitional acts of attention, is associated first with firing of neurons in the frontal eye fields (Figure 2b) [70].
Auditory attention
It is known that patients with parietal lobe damage may neglect auditory or visual modalities or both [71]. Early fMRI studies indicated that the regions of the brain that must be active for one to attend to an auditory spatial stimulus are similar to those involved in attending to a visual spatial stimulus (Figure 1c,2b) [72]. Recently Woldorff and colleagues. used an attend-cue experimental paradigm for subjects exposed to acoustic stimuli like that which they used in their experiments on visual stimuli [73]. The fMRI responses throughout the brain for the different cue conditions indicated that activation of frontal and parietal areas as well as of left and right auditory cortices occurs both in interpret-cue-only trials as well as in attend-cue-only trials (although the interpret-cue-only trials did not activate as many areas as the attend-cue-only trials). Attend-cue-only trials were looked at in relation to interpret-cue-only trials in order to determine which parts of the brain must be active in auditory spatial attention. The results showed that auditory spatial attention involves activation bilaterally of the frontal and superior parietal gyri, which is similar to the same areas activated in visual spatial attention [74], [75]. In general, activation for visual spatial attention in the fronto-parietal cortex is not as superior and medial as that for auditory spatial attention (Figure 1c,2b). Note also should be made of activation in other areas during auditory attention such as the anterior cingulate cortex, the middle cingulate cortex, the anterior insula and at the putamen/caudate nuclei. Of particular interest is the fact that cue-triggered orienting of auditory spatial attention leads to enhancement of activity in the auditory cortex, and this also occurs on those occasions in which no targets are present (Figure 1b,d). Thus the fronto-parietal cortices involved in auditory spatial attention are likely to enhance activity in the auditory cortex.
Attention to auditory phenomena is supported by activity in the inferior parietal and inferior prefrontal cortices [74], [75]. These cortical areas enhance activation by auditory stimuli in the superior temporal gyrus (area 5, Figure 1b) and left inferior frontal cortex, and in the case of the phonetics of speech in the precentral gyrus as well as supramarginal gyrus (area 1, Figure 1b), with lexicality (words over pseudo-words) in the left lateral prefrontal cortex [76]. In the absence of attention, activity in relation to auditory phenomena remains in the early auditory areas around Heschl's gyrus (Figure 1b). So, as in vision, without attention engaged, the activity enhancement to a sensory experience is confined to early perceptual–mediating neural systems. An exception arises, however, if novel sounds are presented, such as rotated speech and to some extent pseudo-words, in which case attention is caught and focused on the auditory task even though attention has been directed to a task in a different sensory modality [76] (see Figure 1c,d for a possible saliency of the auditory percept activity in the inferior parietal cortex supporting attention and feeding back to excite further the auditory cortex).
Summary
Visual attention is supported by activity in lateral intraparietal and frontal (eyefield) cortical areas and that for auditory attention supported by activity in inferior parietal and dorsolateral prefrontal cortex.
Determination of brain areas involved in awareness
Visual awareness
What are the neural correlates in the cortex of being aware of an object in its spatial extent?
Recently, high-density (128 electrode) sets of electroencephalographic recordings have been made from subjects carrying out particular psychophysical tests designed to identify the temporal and spatial evolution of electrical signals (sampled at 250 Hz) in the cortex that accompany the subject's awareness of an object when the subject attends to part of the visual field; that is, their being conscious of the object [77]. These recordings are made when there is an object in the part of the visual field attended to, when there is no object in the attended field and when an object in the attended visual field does not enter awareness, because it is masked. In this latter case the subject is asked to fixate on a point on a screen, and after a variable delay a target object (a numeral) is presented for 16 ms in one of the screen's quadrants, followed by a letter mask for 250 ms. The letter mask consists of four alphabetical letters crowding the numeral. Six conditions of delay between the presentation of the target and the mask are used (the stimulus-onset asynchrony or SOA), namely 16 ms, 33 ms, 50 ms, 66 ms, 83 ms or 100 ms and one mask only condition (without the target numeral). Following each such stimulus, subjects are asked to perform two consecutive tasks: (i) a forced-choice comparison of the target numeral with the numeral 5; and (ii) evaluation of subjective target visibility using a continuous scale labelled ‘not seen’ at the left and ‘maximal visibility’ at the right. The results of such experiments for the condition when the subject reported maximal visibility of the numeral (which occurred at SOA of 100 ms) indicated two phases of cortical activation: a first phase, before 300 ms, when activation progresses from the occipital pole towards both parietal and ventral sites (Figure 2c,e; signals 1–3); and a second phase, after 300 ms, when there is a sudden onset of high-amplitude activity, which is very prominent in ventral prefrontal cortex, and is accompanied by a concomitant reactivation of all previous posterior sites (Figure 2c–e; signal 4).
Masked stimuli that are insufficient for the subject to be aware of the target evoke activation in extrastriate visual cortex, but their intensity is attenuated in higher visual areas (Figure 2c–e; signals 1,2), whereas stimuli insufficient for the subject to be aware of the target numeral generate a sudden increase in activation in the superior parietal and dorsolateral prefrontal areas (Figure 2c,d; signal 4), followed by a large increase in activation in extrastriate visual areas (areas 19, 20, 21; Figure 2c,e; signal 4 at >300 ms)). Studies with fMRI confirm that activation of superior parietal and dorsolateral prefrontal cortex are necessary for visual awareness, together with, of course, activation of a distributed set of regions for the different components of the visual scene in primary visual cortex and ventral visual areas (for a review see [78]).
These observations and their interpretation are supported by a further electroencephalographic study (20 electrodes) using a similar experimental masking paradigm to relate awareness of a target object to cortical excitation [79], [80]. Electrodes were used to record event-related potentials over occipital, posterior temporal and parietal cortex. Left-hemisphere waveforms were obtained when the target was in the right visual field, and right-hemisphere waveforms when the target was in the left field. These waveforms consist of a small early positive component at 80–120 ms (P1), followed by, in general, a larger negative component at 130–200 ms (N1), with this followed by a very large positive waveform at 290–700 ms (P3). Attending to a target in space (spatial attention), without any object presented or any masking, increased the amplitude of P1 and N1 amplitudes over occipital and posterior temporal regions so that the effect of spatial attention is to amplify activity in these regions. Selection negativity (SN), defined as the negativity between 160 ms and 300 ms (i.e. nearly all of the N1 waveform component) did not differ between masked and unmasked stimuli, so it is related to attention rather than awareness. SN, however, did differ in latency between the unattended and the attended field, this being longer in the unattended field, as expected, if it is related to attention. In contrast to this, the visual awareness negativity, defined as the negativity between 120 ms and 290 ms (i.e. taking in nearly all of the N1 waveform component and occurring at times that include those for the SN waveform component) increases significantly for masked versus unmasked conditions, particularly over occipital cortex, as does the very large late positivity. These observations indicate that electrophysiological correlates of attention (SN) are elicited independently of awareness of a target, whereas the electrophysiological correlates of awareness of the target occur independently of manipulations of spatial and non-spatial attention [79]. Electrical correlates related to reflection on the aware target, are associated with working memory in prefrontal cortex, and occur as a late positive potential.
A technical issue here is that if the frequency range of electrophysiological recordings in the EEG reflects that of local field potentials due to synaptic currents, then the psychophysical experiments relating attention and awareness to specific brain regions are approximately consistent with those using fMRI, whereas if high-frequency components that take in action potential firing are used, this is not the case [81]. Such a difference might be expected because the fMRI haemodynamic signal is very likely to be causally driven by excitatory glutamatergic synaptic transmission, not by action potentials [82], [83]. Magneto-encephalographic recordings of activity during psychophysical experiments attempting to identify the brain regions involved in attention and awareness give rise to similar conclusions to those arrived at using electroencephalographic and fMRI recording methods [84], supporting the conclusion that the activity of interest is due to synaptic currents.
The fact that different types of illusions are accompanied by activation of areas of the brain that are also active when actually perceiving or experiencing the subject matter of the illusion does not mean that awareness of these is just dependent on such activation. The phenomenon of extinction shows this. For example, patients with right parietal cortex damage are often not aware of presentations to areas of their left visual field when the same image is presented simultaneously to the right visual field; that is, there is often left visual field extinction. fMRI, however, shows that there is elevated activity in the right fusiform face area of these patients, whether they report awareness of a face presented to their left visual field or not [85]. Interestingly, when the patient is aware of the left visual stimulus in bilateral trials, greater activity is detected by fMRI in the right ventral visual cortex but now elevated activity is also recorded in the left frontal and parietal areas [85]. Two points emerge from such studies. First, category-specific areas of visual cortex are still activated by extinguished stimuli (one that the subject is unaware of), so that such activation is not sufficient for awareness. Second, parietal and pre-frontal areas are, in addition, activated when the patients are aware, suggesting that these areas form a network that must be engaged to support awareness [86].
Auditory awareness
Auditory awareness requires activation in dorsolateral prefrontal cortex and superior temporal cortex whereas visual awareness, as we have seen, requires activation in dorsolateral prefrontal cortex and superior parietal cortex (Figure 1e,2c) [87]. Whether this auditory awareness involves the triggering of a late surge of activity in prefrontal and superior temporal cortex is not known, although it is very likely to be responsible for an increase in the intensity and spread of activity in auditory cortex (Figure 1e).
Why should subjects with schizophrenia be aware of hallucinatory voices as alien? The auditory cortex in planum temporale allows one to locate voices in external space and this cortex does not function properly in schizophrenia. One argument is that there is failure of corollary discharges involving the planum temporale in subjects with schizophrenia [88]. Corollary discharges are thought to be normally required in order to be able to distinguish between percepts arising from one's own activities and those arising independently in one's environment. The concept of corollary discharge is that it is an efferent copy of a motor command sent to the related sensory system in order for one to be alerted to the motor act carried out, such as when lifting a weight. Without the collateral discharge it would appear as if the motor act is imposed on one rather than being voluntary. Corollary discharge is taken to be the reason you cannot tickle yourself; sensory perception of the tickle is damped out by the corollary discharge accompanying intention to carry out the motor act of tickling. If a stroke interferes with the production of a corollary discharge then even lifting one's arm appears to involve lifting a very heavy weight – as if someone was resisting you lifting your arm. In the case of the auditory system a corollary discharge might exist whereby signals that arise when one is about to speak are sent to the auditory cortex, where they are compared with the signals relating to hearing the speech [89]. In this model of auditory corollary discharges, a motor command to the sensorimotor system accompanying speech generates an efference copy that, through a corollary discharge, is compared with the auditory feedback of the heard speech. Self-produced speech sounds are correctly predicted by the efference copy, with this lack of discrepancy detected by the comparator giving rise to suppression of auditory cortex activity to the self-produced sound. These ideas are supported by experiments in which the N1 event-related potential due to a 1000 Hz tone probe (baseline), in the absence of speech, is decreased in normal subjects when they talk aloud, but it is not decreased in patients with schizophrenia. During normal speech the corollary discharge to auditory cortex dampens its activity (indicated by the N1 potential) and this supports the interpretation that the speech emanates from oneself and not from outside. If, however, the corollary discharge mechanism fails, as indicated by the failure to suppress the N1 potential, then this supports the misinterpretation that the self-initiated speech can be attributed to an outside source that does not exist. The failure of mismatch negativity in patients with schizophrenia also points to abnormalities in auditory cortical mechanisms [90]. There appears then to be a problem in the corollary discharge mechanism in patients with schizophrenia whereby the feedback from the neural pathway that underpins the effort or intention to use a word is faulty, so the words do not appear to be self-generated; that is, they appear alien.
Summary
Visual awareness requires activity in dorsolateral prefrontal and superior parietal cortex. Auditory awareness requires activity in prefrontal and superior temporal cortex.
Conclusion: attention and awareness in schizophrenia
When attention is caught and held activation in cortical regions subserving a percept is greatly amplified, leading to a burst of activity in the networks subserving awareness that is consciousness. The hypothesis proposed here is that in schizophrenia endogenous spontaneous activity occurs in a cortical region associated with a particular percept as a consequence of that region being deafferented. For example, if this deafferentation occurs in the anterior temporal cortex, hallucinations involving grimacing faces may occur. In contrast, if deafferentation occurs in the planum temporale then hallucinations of declamatory voices may occur. The salience of these hallucinations can activate lateral intraparietal cortex in the case of vision (Figure 2b) or perhaps dorsolateral prefrontal cortex in the case of audition (Figure 1c), leading to amplification of the endogenous activity in the visual or auditory areas. This can in turn activate prefrontal/superior parietal regions that support visual awareness (Figure 2c) or prefrontal/superior temporal areas that support auditory awareness (Figure 1e) of the hallucinations. Initial tests of this hypothesis require answers in the near future to the following questions: (i) do the different categories of auditory hallucinations (e.g. voices of children crying) pertain to different regions of auditory cortex as do different categories of visual hallucinations to different regions of visual cortex; (ii) to what extent is there synapse loss in such regions or in the main afferent projections to these regions; (iii) can the site of mismatch negativity be better spatially resolved so as to assist in identifying the region(s) of auditory cortex that have gone awry in schizophrenia; is this region deafferented; and (iv) finally, is the activity in the fronto-parietal cortex devoted to auditory attention and in the frontal and superior temporal cortex supporting awareness normal in subjects suffering from schizophrenia.
