Abstract
Neurobiological models of obsessive–compulsive disorder (OCD) emphasize excessive activity of frontostriatal brain circuits, including the anterior cingulate cortex (ACC), both at rest and during cognitive task performance and symptom provocation [1–4]. But despite the fact that consistent identification of ACC functional abnormalities have been identified in OCD, very few studies have examined whether there are also biochemical abnormalities in the region.
Proton magnetic resonance spectroscopy (1H-MRS) offers a non-invasive means for obtaining information about brain biochemistry in vivo, and allows such measures to be related to the various clinical manifestations of OCD. In this context, two of the most widely studied metabolites have been glutamate–glutamine (Glx) and N-acetylaspartate (NAA). Elevated Glx levels have been found in the caudate of treatment-naïve patients (both paediatric and adult), but these seem to normalize after treatment with selective-serotonin re-uptake inhibitors (SSRIs) [5–9]. Similar Glx elevations have been reported in orbitofrontal regions in adult OCD patients [10], although Rosenberg et al. recently found that Glx concentrations in the ACC of paediatric OCD patients were decreased rather than increased, even when medication-naïve, suggesting that the nature of glutamatergic pathology in this disorder may be regionally specific [11]. Reduced levels of NAA, considered to be a marker of neuronal viability [12], have also been found in OCD patients [13–15] and although they also normalize with SSRI treatment, they are not correlated with symptomatological improvement [14].
Thus, although both Glx and NAA abnormalities respond to treatment, only the former appears to be related to symptom improvement [5–9]. Several limitations of this work, however, restrict the conclusions that can be drawn, particularly as they pertain to the ACC. First, studies have varied with respect to the side (left only, right only, or bilateral) and subregion of the ACC sampled. Given that the ACC is a functionally heterogeneous region [16], subtle, regionally specific, alterations may be obscured unless key subregions are assessed independently. Second, few studies have explicitly examined gender differences. Gender has been shown to influence both clinical and biological heterogeneity in OCD [17], thus representing an important variable for consideration. Finally, most studies have focused on one or a few metabolites, and have failed to exploit the potential of 1H-MRS to provide measures of other markers of cellular function and integrity, such as choline-containing metabolites (Cho; a measure of lipid turnover), creatine plus phosphocreatine (Cr; a measure of cellular energetics) and myoinositol-containing compounds (mI; among other things, an index of glial metabolism). There has been no study to date that has comprehensively examined each of these metabolites across the various subregions of the ACC in OCD.
In this context we analysed data acquired from the dorsal and rostral ACC in 20 male and female OCD patients and 26 healthy comparison subjects. Data from the dorsal ACC have been previously published, although the effects of gender and hemispheric laterality were not examined [15]. Thus, in the present investigation we probed ACC biochemistry from the dorsal and rostral subregions of the ACC of each hemisphere, which are thought to be specialized for cognitive and affective functioning, respectively [16]. Using a 3 T scanner allowed us to resolve signals from Glx, NAA, mI, Cr and Cho, facilitating a comprehensive in vivo assay of ACC biochemistry in OCD.
Methods
Subjects
Twenty OCD patients and 26 healthy volunteers were recruited from the general community through advertisements in local newspapers and community services. All patients were required to (i) have a current IQ of >80; (ii) be medication-free or stable on their medication dose for at least 1 month; and (iii) have no other current Axis I psychiatric diagnosis. All subjects with a history of significant head injury, neurological disease, electroconvulsive therapy, impaired thyroid function, or steroid use were excluded. After complete description of the study to the subjects, written informed consent was obtained. Demographic and clinical characteristics of the samples are given in Table 1.
Subject characteristics (mean±SD)
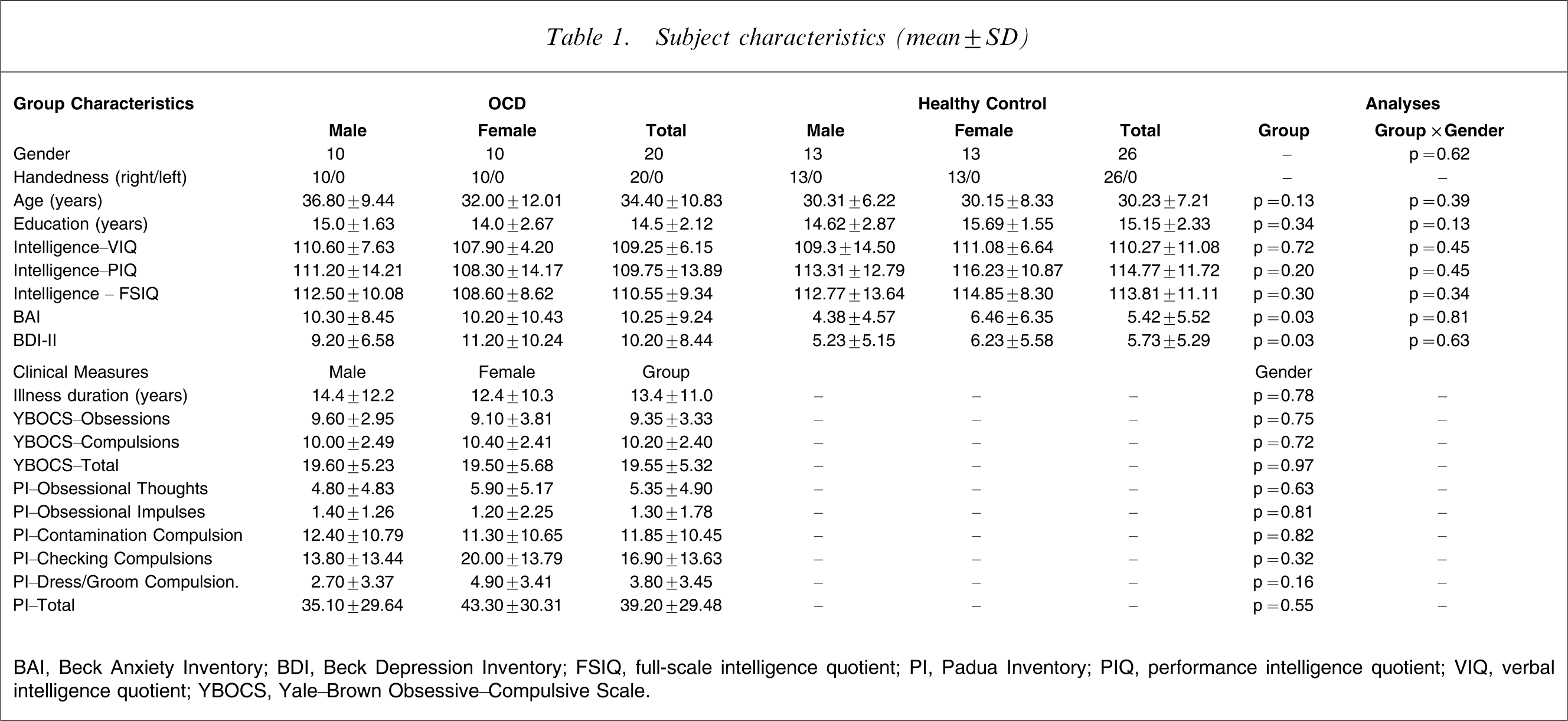
BAI, Beck Anxiety Inventory; BDI, Beck Depression Inventory; FSIQ, full-scale intelligence quotient; PI, Padua Inventory; PIQ, performance intelligence quotient; VIQ, verbal intelligence quotient; YBOCS, Yale–Brown Obsessive–Compulsive Scale.
Clinical and neuropsychological assessment
Diagnoses were established using the Structured Clinical Interview for DSM-IV Axis I Disorders, Patient Edition [18]. At the time of scanning, the patient group reported overall mild to moderate OCD symptoms (indexed by the Yale–Brown Obsessive–Compulsive Scale, YBOCS; characterized by contamination and checking compulsions; indexed using the Padua Inventory, PI). Patients were also characterized by moderate cognitive and affective symptoms of depression and physiological and psychological symptoms of anxiety (measured by the Beck Depression Inventory, BDI-II; and the Beck Anxiety Inventory, BAI). Twelve of the patients were on stable doses (at least 2 months) of psychotropic medication at the time of scanning (fluoxetine, n = 4; fluvoxamine, n = 1; citalopram, n = 2; venlafaxine, n = 1; clomipramine, n = 4). Two patients had received adjuvant medications (e.g. benzodiazepines) within the past 6 months. General intelligence (IQ) was assessed using the Wechsler Abbreviated Scale of Intelligence (WASI) [19].
1H-MRS imaging acquisition
Magnetic resonance imaging (MRI) data were acquired using similar methods to those described in our previous studies [15], [20], [21]. The previously analysed 1H-MRS data were processed using a bilateral dorsal ACC voxel of OCD patients to examine its correlations with haemodynamic responses during cognitive task performance [15]. We now extend this work in the current analysis by examining each hemisphere independently, and also by analysing spectra for the rostral ACC, which we have not previously investigated.
A 3 T GE Signa Horizon LX human imaging scanner (GE Healthcare, Milwaukee, WI, USA) was used. Subjects were immobilized, using a Velcro strap over the forehead. Volume-localized 1H-MRS was recorded from four separate locations using a standard short-echo point resolved spectroscopy sequence (TR = 3000 ms, TE = 30 ms, with a nominal voxel size of approx. 6.5 cm3). For the dorsal ACC (dACC) measurements we acquired 64 transients, whereas rostral ACC (rACC) required 128 transients from each hemisphere due to susceptibility arising from the ethmoid sinuses, orbits, and vasculature in the region. The region of interest (ROI) had to be re-positioned for some individuals in order avoid major vasculature and to minimize the influence of this on susceptibility. The total scanning time for the 1H-MRS was approximately 30 min. Figure 1 illustrates the typical location and spatial geometry of the volume localized voxels.
(a) Functional subdivisions of the anterior cingulate cortex (ACC) on the basis of functional imaging studies and voxel placements in the current study. The coronal sections through the (A) dorsal ACC (dACC) and (B) rostral ACC (rACC) regions of interest further highlight the areas of brain coverage by the magnetic resonance spectroscopy (MRS) voxels. (b) Sample MRS spectrum from the dorsal ACC region highlighting the five main metabolite peaks: Cho, choline-containing compounds; Cr, creatine plus phosphocreatine; Glx, glutamate–glutamine; NAA, N-acetylaspartate; mI, myoinositol. The MRS voxels were placed in each hemisphere, separated by the medial wall boundary of the frontal lobes, so as to encompass the dACC and rACC. For the dACC, the posterior boundary of the voxel was approximately 10 mm posterior to a vertical line from the anterior commissure orthogonal to the anterior–posterior commissure line. The inferior border was approximately 5 mm superior to the top edge of the corpus callosum while the medial border was 1–2 slices lateral to the parasagittal slice on the T1 structural image for each hemisphere. For the rACC, the posterior boundary was the front edge of the corpus callosum while the inferior border was the anterior–posterior commissure line. The medial border was again 1–2 slices lateral to the parasagittal slice on the T1 structural image for each hemisphere.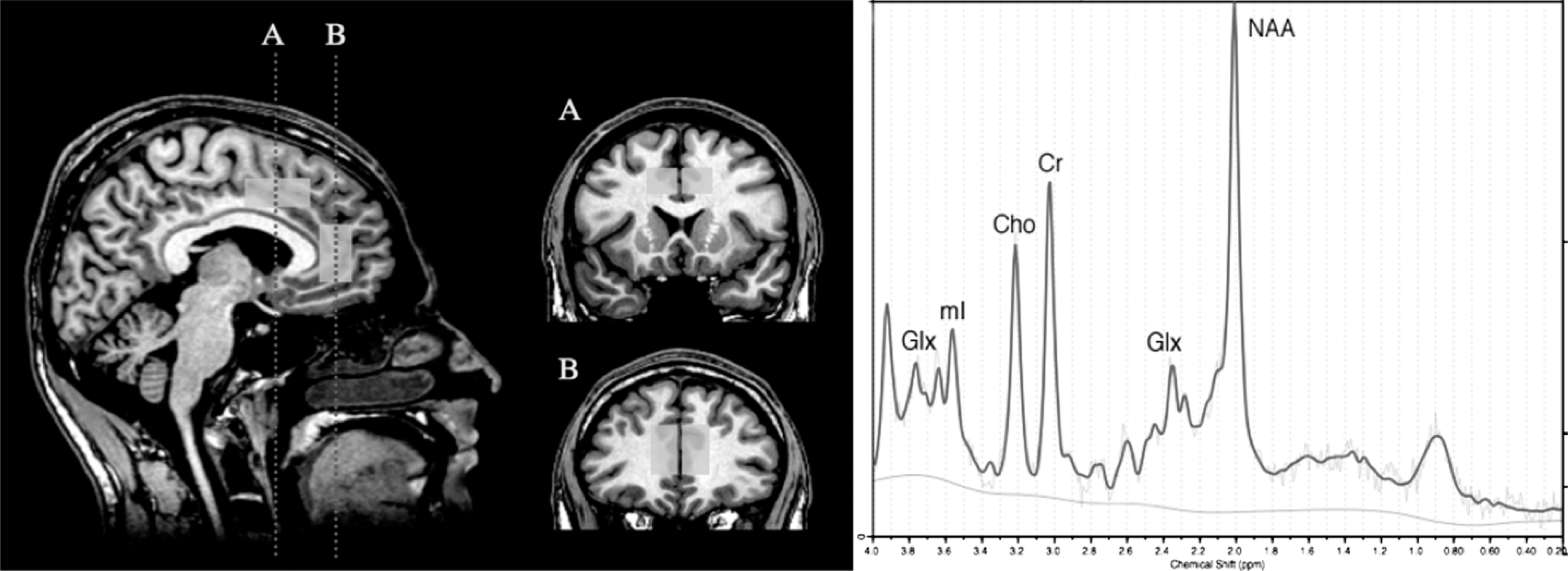
We measured absolute levels of NAA, Glx, Cr, Cho, and mI from each of the four volume-localized spectra with LCModel software [22]. This software utilized a library of reference spectra in a basis set recorded specifically for the scanner. The LCModel fitting algorithm uses the multiple peaks contributing to an individual metabolite spectrum (including macromolecule contribution to the spectra) to estimate the tissue content of each metabolite [22]. Results are presented in institutional units approximating millimolar concentration (mmol/L), using the tissue water signal as an internal standard. The residual signal corresponds to, and is fitted by, additional broad peaks representing unknown metabolites and components such as macromolecules with short T2 relaxation times.
Statistical analysis
Individual demographic measures of gender, age, education, and IQ were compared across groups using χ2 test and independent sample t-tests comparing the two groups, in SPSS version 11.0 (SPSS, Chicago, IL, USA).
The metabolite content of voxels was corrected for differences in tissue composition using segmentation information derived from coronal 3-D spoiled gradient-recalled echo in the steady state images recorded during the same scanning session. The brain component was separated from non-brain in the structural images, using the Brain Extraction Tool of FSL (www.fmrib.ox.ac.uk). The images were then segmented into grey matter (GM), white matter and cerebrospinal fluid using the FAST module of FSL. The proportion (probability) for each of the three tissue types was then determined for the volume and position of each MRS voxel, using a module [23] [Masterton RAJ et al.: unpublished data, 2005] written in IDL for iBrain (www.brain.org.au/software.html). Where there was a correlation >0.2 (Spearman's rank-order correlation coefficients, two-tailed) for the amount of GM in the voxel and any metabolite, we corrected for the influence of grey matter using a previously described formula [24], cMRS = mMRS – (gradient [mGM – meanGM]), where cMRS is corrected MRS; mMRS is the measured MRS concentration; gradient is the gradient of the regression line between the GM tissue volume of interest and the average amount of GM in the entire sample of subjects; mGM is the measured GM fraction; and meanGM is the mean GM tissue volume of the entire sample. These corrected metabolite concentrations were used for subsequent analyses.
The 1H-MRS parameters used for the present study provided robust signals for both the healthy control and OCD groups in the dACC and rACC. Specifically, healthy controls had a dACC and rACC signal-to-noise ratio of 16.13 (SD = 2.22) and 14.59 (SD = 3.99) and a full width at half maximum peak height (FWHM) of 0.06 ppm (SD = 0.01) and 0.07 ppm (SD = 0.02), respectively. OCD patients had a dACC and rACC signal-to-noise ratio of 16.1 (SD = 2.8) and 15.7 (SD = 5.5) and an FWHM of 0.06 ppm (SD = 0.01) and 0.07 ppm (SD = 0.03), respectively. None of these measures was different between the two groups (p = 0.45–0.96) suggesting that the quality of the data is comparable across the two groups. The output from LCModel includes the Cramer–Rao lower bound, which is a measure of reliability of the fit. Typically, values <30 are considered to be reliable for less abundant metabolites such as mI and Glx, while values <20 are considered to be a good fit for more abundant metabolites such as NAA, Cr and Cho. Only one metabolite (mI) for one voxel of a single individual fell well short of these criteria (i.e. >30) and was entered as ‘missing’. The mean Cramer–Rao lower bounds (averaged across all regions and hemispheres) for NAA, Cr, Cho, mI and Glx were 5.6, 5.4, 5.9, 10.5 and 10.5 (SD = 1.87, 1.06, 1.33, 1.99 and 3.06), respectively, for healthy controls, and 5.8, 5.2, 5.4, 7.7 and 9.7 (SD = 1.94, 0.92, 1.17, 1.99 and 2.63), respectively, for OCD patients.
Metabolites were analysed individually, using two-way ANOVA with two repeated measures: hemisphere (left and right) and region (dACC and rACC). We tested main effects and interactions in the overall model with α = 0.05 and examined significant effects with post-hoc pairwise contrasts evaluated against a Bonferroni-adjusted alpha to correct for multiple comparisons. Only effects involving diagnostic group differences were examined in this manner because these were the effects of interest in the current study (as opposed to effects involving hemisphere or region). Effect sizes, expressed as Cohen's d [24], are also provided where appropriate. Spearman's rank-order correlation coefficients were used to examine the associations between 1H-MRS metabolite levels obtained from within each of the ROI and clinical measures of OCD symptom dimensions and symptom severity. We statistically covaried for the effects of state-anxiety and depressive symptomatology in these biochemistry-symptom correlational analyses. As with previous studies [10], we wanted to investigate primary OCD symptomatology that could not be explained by levels of state-related anxiety and depression.
Results
Creatine and choline
There was a significant main effect of hemisphere for both Cr (F(1,42) = 9.42; p = 0.004; d = 0.44) and choline (F(1,42) = 9.74; p = 0.003; d = 0.39). No diagnosis effect or interactions with diagnosis were found for these metabolites. These findings indicate that across the entire sample, concentrations of both Cr and Cho were higher in the left hemisphere (Table 2).
Metabolite levels in the ACC (mean±SD)
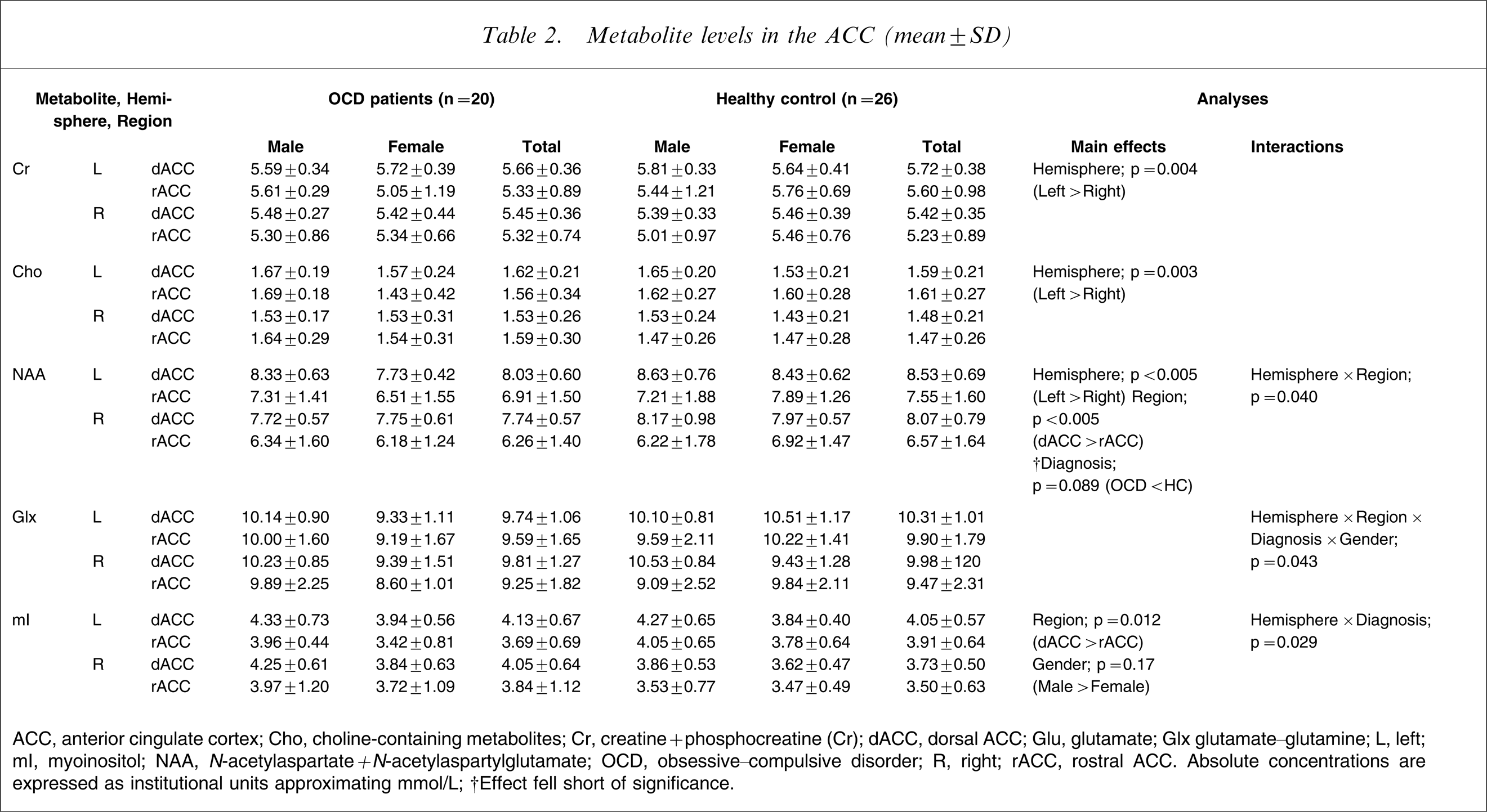
ACC, anterior cingulate cortex; Cho, choline-containing metabolites; Cr, creatine + phosphocreatine (Cr); dACC, dorsal ACC; Glu, glutamate; Glx glutamate–glutamine; L, left; mI, myoinositol; NAA, N-acetylaspartate + N-acetylaspartylglutamate; OCD, obsessive–compulsive disorder; R, right; rACC, rostral ACC.
Absolute concentrations are expressed as institutional units approximating mmol/L.
†Effect fell short of significance.
Myoinositol
There was a main effect of region (F(1,41) = 6.99; p = 0.012; d = 0.47) and gender (F(1,41) = 6.16; p = 0.017; d = 0.75), as well as a hemisphere×diagnosis interaction (F(1,41) = 5.10; p = 0.029, corrected). Post-hoc pairwise contrasts comparing the two groups within each hemisphere showed that OCD patients had higher concentrations of mI in the right ACC (collapsed across dorsal and rostral subregions) compared with healthy controls, although this just failed to reach significance (F(1,41) = 3.92; p = 0.054; d = 0.62).
N-acetylaspartate
There was a main effect of hemisphere (F(1,42) = 27.17; p < 0.005; d = 0.63), and region (F(1,42) = 43.67; p < 0.005; d = 1.18), as well as a hemisphere×region interaction (F(1,42) = 4.15; p = 0.04). The main effect of diagnosis trended towards significance (F(1,42) = 3.03; p = 0.089; d = 0.55).
Glutamate and glutamine
There were no main effects of hemisphere, region or gender for Glx, but there was a four-way hemisphere×region×gender×diagnosis interaction (F(1,42) = 4.37; p = 0.043). Inspection of the data and effect size analyses showed that, compared to female controls, female OCD patients had significantly reduced Glx in all subregions except the right dACC (Figure 2). Post-hoc analysis of glutamate alone (after removing metabolite information for two individuals due to fitting error of >30%) indicated a trend-level diagnosis×gender interaction (F(1,40) = 3.86; p = 0.057; d = 0.58) indicating that female OCD patients had less overall glutamate than their healthy counterparts in all ACC subregions.
Glutamate–glutamine (Glx) concentrations in (a) female and (b) male obsessive–compulsive disorder (OCD) patients, as well as healthy controls (HC) across each subregion of the anterior cingulate cortex (ACC). The left dorsal ACC (dACC) of female OCD patients was significantly lower in Glx, compared to their female control counterparts. There were no significant differences for male OCD patients. (c) This is further highlighted in the effect size analysis. Solid black lines, effect size analysis for glutamate alone.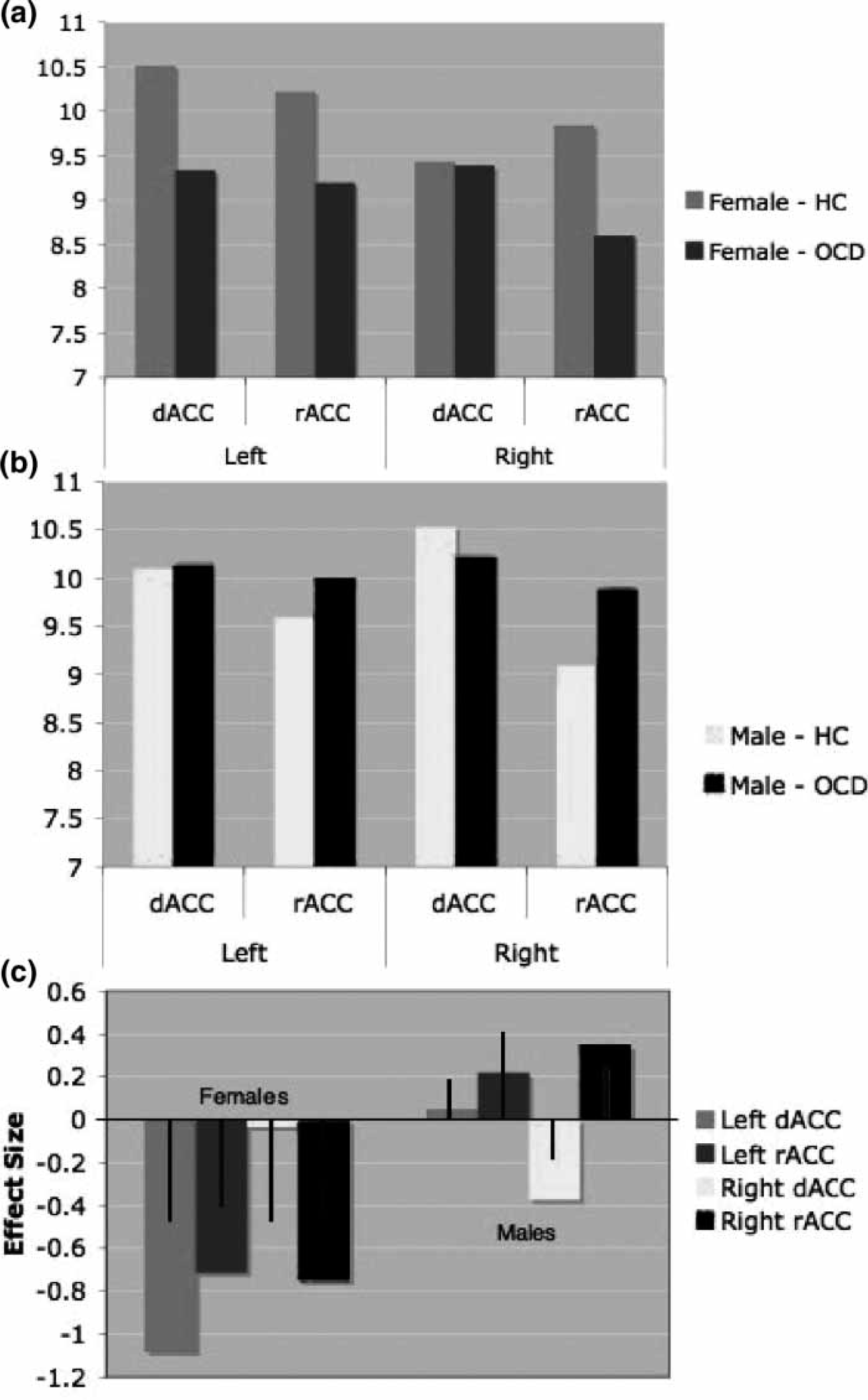
Effects of psychotropic treatment
Twelve of the OCD patients were receiving some form of psychotropic therapy at the time of testing, which may have affected their brain biochemistry. A re-analysis of the data using repeated-measures ANOVA comparing the 12 medicated OCD patients to the eight unmedicated patients separately for each metabolite indicated no significant effects. Due to the small sample sizes in these comparisons we additionally ran non-parametric tests (Mann–Whitney U-test) for each metabolite, none of which indicated any significant effects, including Glx (p = 0.16). There were no significant differences in symptom severity between the medicated and non-medicated groups.
Biochemical-symptom associations
A correlational analysis was conducted separately for male and female subjects to determine if the identified patient–control differences in Glx, mI and NAA metabolites were related to clinical symptom severity and symptom dimensions. These analyses indicated gender specific (i.e., female only) associations between ACC biochemistry and OCD symptomatology. Levels of Glx across the ACC subregions were strongly and positively associated with OCD symptom severity (particularly the obsessions subscale and the YBOCS-total score) in female patients (Table 3; Figure 3). Only the right dACC NAA was correlated with obsession scores; there were no significant associations for mI in either male or female subjects.
Scatterplot of the association between the Yale–Brown Obsessive–Compulsive Scale (YBOCS)-total score and concentrations of total anterior cingulate cortex glutamate–glutamine (Glx) for female obsessive–compulsive disorder patients (the correlation is partialled for the effects of state anxiety and depressive symptoms).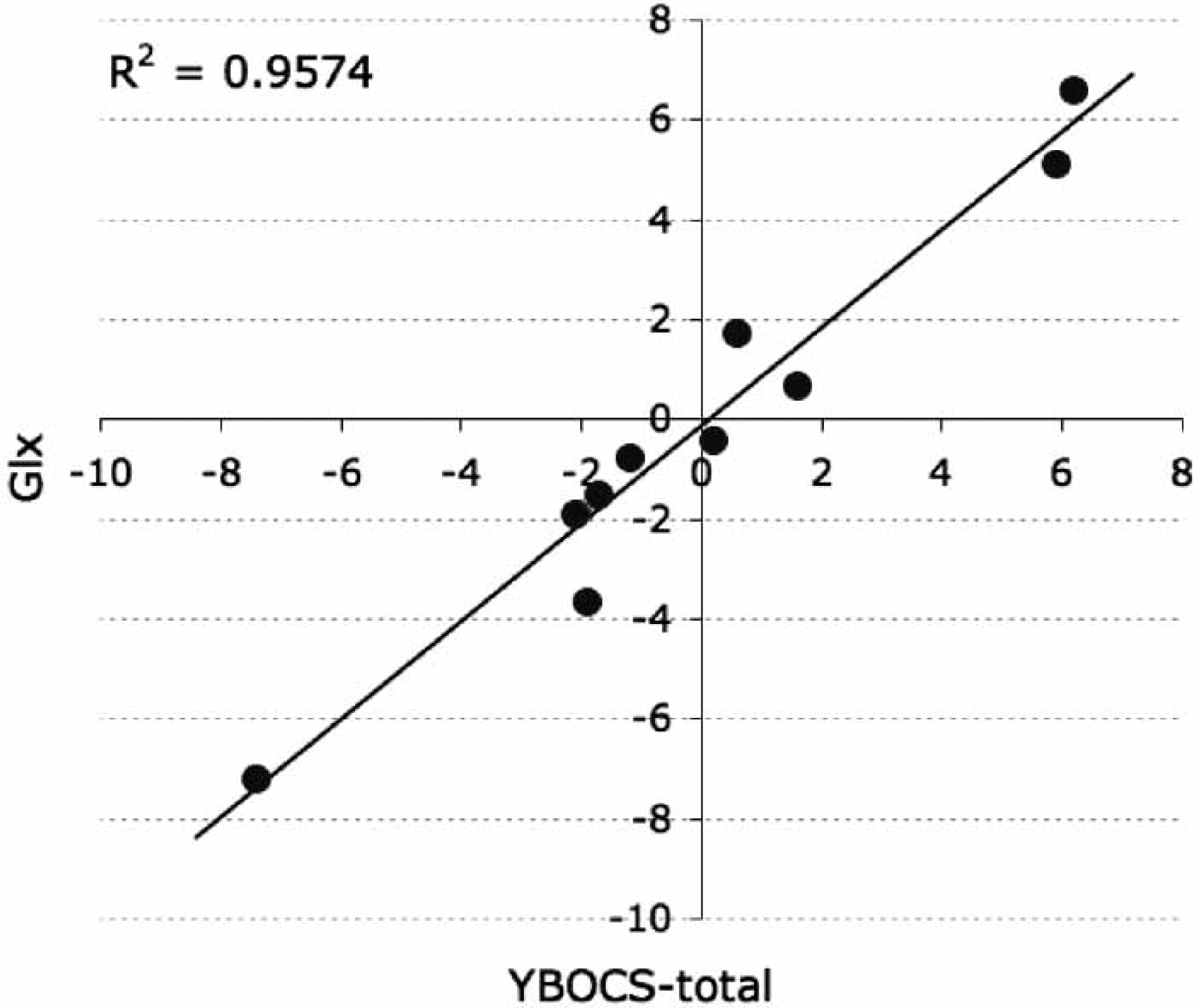
correlations of biochemical and clinical characteristics for OCD patients
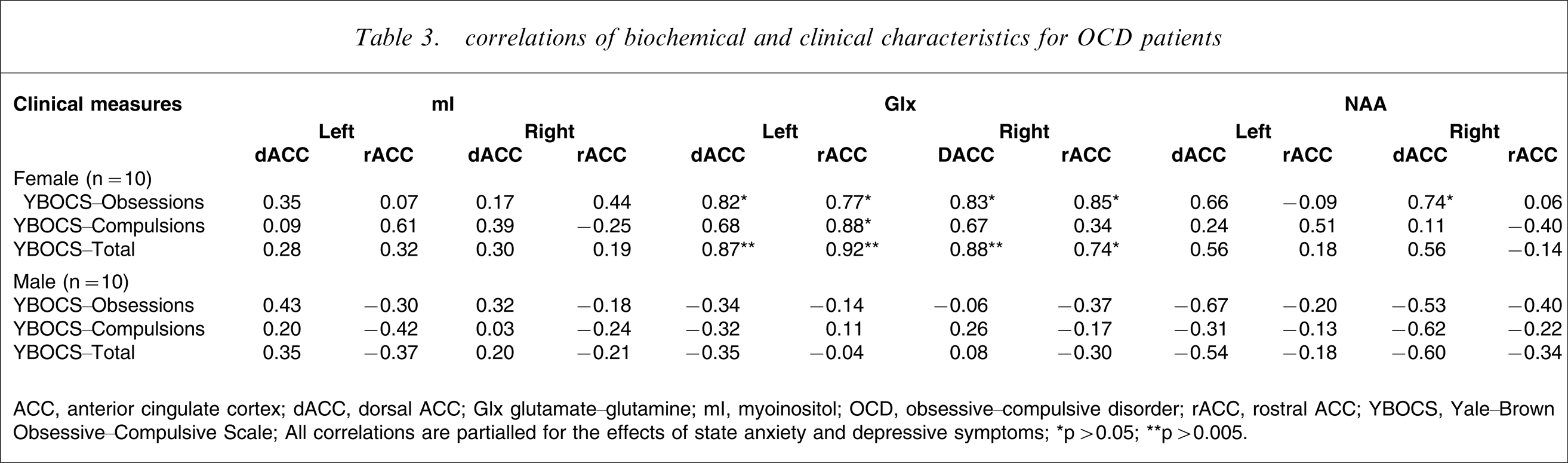
ACC, anterior cingulate cortex; dACC, dorsal ACC; Glx glutamate–glutamine; mI, myoinositol; OCD, obsessive–compulsive disorder; rACC, rostral ACC; YBOCS, Yale–Brown Obsessive–Compulsive Scale.
All correlations are partialled for the effects of state anxiety and depressive symptoms.
∗p > 0.05; ∗∗p > 0.005.
Discussion
We examined the specificity of brain metabolite disturbances with respect to gender, hemisphere and subregion of the ACC in patients with OCD. We observed a significant reduction in the concentrations of Glx in all but one of the ACC subregions investigated in female OCD patients, elevated concentrations of mI in the right ACC of both male and female patients, and a trend towards a global reduction in NAA in both male and female patients. Although the mI alterations did not correlate with any symptom measures, individual differences in ACC Glx levels in female OCD patients were strongly correlated with clinical symptom severity, across nearly all symptom dimensions. There was only a limited correlation between NAA levels and symptom measures. The present findings do not represent state-related anxiety or depressive symptoms at the time of scanning because these variables were measured and statistically controlled in the analyses. Therefore, the Glx-symptom association identified in female OCD patients is likely to reflect a more enduring illness characteristic.
Although others have reported positive correlations between Glx levels and measures of symptom severity [7], [10], they did not examine gender-related differences. There is now increasing recognition that gender may have a significant influence on the clinical and biological heterogeneity of OCD [17]. For example, there are slightly more female OCD patients in adult samples [25]; female patients are more likely to exhibit contamination obsessions and respond better to treatment [26], [27]; and there are sexually dimorphic patterns of genetic susceptibility [17] including glutamate transporter genes [28] in OCD. In light of the present findings, future studies should consider more carefully the putative relationship between gender and OCD psychopathology.
The present finding of decreased Glx concentrations in OCD patients is consistent with the only other study to examine this metabolite in the ACC [11]; but that study, which examined only one subregion (the rostral ACC) in a paediatric sample, did not find an effect of gender. Although the Glx alterations support recent speculations implicating glutamatergic dysfunction in OCD [29–31], the direction of both the present findings and those of Rosenberg et al. [11] (i.e. decreased rather than increased) is inconsistent with a straightforward hyperglutamatergic model and contrasts with the excessive levels of Glx reported in orbitofrontal and striatal regions of OCD patients [7], [10]. It is unclear whether Glx alterations reflect explicit changes in specific physiological processes (e.g. differences in the cycling of glutamate from neurons to glia, and anaplerotic metabolism) or cell population (e.g. differences in the relative distribution of neurons relative to glia) [32]. This reduced Glx concentration in the ACC could represent a mechanism for counterregulating hyperactivity in the striatum of patients with OCD. But evidence of a similar pattern of reduced ACC Glx concentrations in adults and children with major depressive disorder [11], [33–35], together with the absence of these abnormalities in male OCD patients, suggest that this phenomenon is not specific to OCD.
OCD patients (both male and female) also had elevated concentrations of mI in their right ACC. This metabolite can act as a cerebral osmolyte, governing processes intrinsic to cellular homeostasis, such as the accumulation of intracellular water, and forms a core component of the phosphatidylinositol second messenger system (PI-cycle) [36]. Abnormalities in cellular mI levels and/or PI-cycle regulation have been implicated in the pathophysiology and treatment of OCD, as well as other psychiatric disorders, including bipolar disorder, major depressive disorder, panic disorder and schizophrenia [37]. As such, this neurochemical abnormality is likely to reflect a general pathophysiology related to altered cellular homeostasis, rather than one specific to OCD.
OCD patients had reduced NAA concentrations, but a direct comparison with controls fell short of significance. In our previous analysis of this OCD group and a subsample of the healthy controls [15], NAA was reduced, although we did not examine the rostral ACC and did not investigate the effects of gender or hemispheric laterality. The failure to identify a significant difference in the present study is most likely due to the stringent statistical thresholds used to correct for multiple comparisons. Two other studies have demonstrated reduced NAA levels in this region in adult patients [13], [14]. NAA is produced within neuronal mitochondria and is thought to reflect neuronal density, mitochondrial metabolism and functional viability [38], but whether reduced levels reflect neuronal loss or a state of (potentially reversible) neuronal dysfunction remains unclear. Interestingly, although NAA abnormalities in OCD respond to pharmacological treatment [14], they do not seem to be associated with changes in symptomatology [8], [10], [13], [14]. One speculation is that abnormalities in the glutamatergic system are more relevant to OCD symptomatology, whereas NAA changes are more closely associated with functional abnormalities in the region [15]. The lack of significant change in other brain metabolites measured by MRS indicates that processes, such as altered lipid turnover (choline-containing metabolites), and cellular energetics (creatine plus phosphocreatine), are unlikely to be core characteristics of OCD pathophysiology.
There are several limitations in the present study. First, we conducted a large number of comparisons both across and within groups. This was necessary, given the number of voxels from which we acquired data, and the number of brain metabolites and clinical measures investigated. But the analyses were corrected for multiple comparisons, suggesting that the findings are robust (also supported by the relatively large effect sizes). A second limitation was our focus on the ACC. We did not acquire MRS data from other brain regions that are also important in the neurobiology of OCD (e.g. the orbitofrontal cortex, striatum) [39]. Thus, the anatomical specificity of the present MRS findings is uncertain. Third, we examined unresolved Glx with only post-hoc analysis of glutamate alone for several reasons: (i) the separation and quantification of glutamate and glutamine are technically difficult (i.e. overlapping resonances); (ii) Glx provides a more robust fit compared with glutamate alone; and (iii) we wanted to be consistent with previous literature. Nevertheless, further 1H-MRS work that separates the glutamate and glutamine signals and also assesses concentrations of glutathione (a product of glutamate metabolism that serves as a cellular antioxidant) will facilitate identification of the physiological processes associated with the changes in Glx reported here [40]. Finally, despite the fact that we obtained large effects sizes, some of the analyses (e.g. comparison of medicated vs unmedicated patients) may have been underpowered due to the small sample size and/or confounded by patients receiving adjuvant medications (e.g. benzodiazepines). In this context, the use of Bonferroni corrections may have been overly conservative, hence we have concurrently reported effect size statistics to provide an index of the magnitude of the changes observed. Future studies examining the relationship between OCD-related biochemical changes and how these vary as a function of symptom profile, treatment history and gender will represent a useful extension of the present study.
Conclusions
The primary finding is the observation of gender-specific alterations in ACC Glx in OCD patients. Importantly, this Glx disturbance is strongly predictive of symptom severity, and is not explained by levels of state-related anxiety and depressive symptoms. Importantly, MRS-detected metabolite abnormalities correlate with the severity of OCD-related impairment, suggesting that this technique may be of use in objectively monitoring patient progress and assessing the effectiveness of various treatments.
Footnotes
Acknowledgements
This research was supported by grants from the National Health and Medical Research Council (NHMRC) of Australia (I.D. 236175), the Colonial Foundation and the Ian Potter Foundation. Dr Yücel is supported by an NHMRC Clinical Career Development Award (ID 509345). Dr Harrison is supported by an NHMRC Training Award (ID 400420). Dr Wood is supported by an NHMRC Clinical Career Development Award and a NARSAD Young Investigator Award. Dr Fornito was supported by a JN Peters Fellowship and NHMRC CJ Martin Fellowship (ID: 454797). The authors would also like to thank Miss Kerrie Clarke, Professor Mike Kyrios and Dr Sunil Bhar for their efforts with patient recruitment, as well as the Mental Health Research Institute for research and administrative support.
