Abstract
Study Design:
Systematic review.
Objectives:
The aim was to determine the fusion efficacy of allograft and demineralized bone matrix (DBM) in lumbar instrumented and noninstrumented fusion procedures for degenerative lumbar disorders.
Methods:
A literature search was conducted using the PubMed and Cochrane databases. To be considered, publications had to meet 4 criteria: patients were treated for a degenerative lumbar disorder, a minimum group size of 10 patients, use of allograft or DBM, and at least a 2-year follow-up. Data on the study population, follow-up time, surgery type, grafting material, fusion rates, and its definition were collected.
Results:
The search yielded 692 citations with 17 studies meeting the criteria including 4 retrospective and 13 prospective studies. Six studies used DBM and 11 employed allograft alone or in the combination with autograft. For the allograft, fusion rates ranged from 58% to 68% for noninstrumented and from 68% to 98% for instrumented procedures. For DBM, fusion rates were 83% for noninstrumented and between 60% and 100% for instrumented lumbar fusion procedures.
Conclusions:
Both allograft and DBM appeared to provide similar fusion rates in instrumented fusions. On the other hand, in noninstrumented procedures DBM was superior. However, a large variation in the type of surgery, outcomes collection, lack of control groups, and follow-up time prevented any significant conclusions. Thus, studies comparing the performance of allograft and DBM to adequate controls in large, well-defined patient populations and with a sufficient follow-up time are needed to establish the efficacy of these materials as adjuncts to fusion.
Introduction
Lumbar spine fusion with and without instrumentation is frequently a treatment of choice for various spinal pathologies. Several studies have demonstrated an increase in the number of cases and associated costs in the past few decades. 1 -3
Various factors including surgical technique, primary or revision surgery, use of instrumentation, grafting materials, and patient comorbidities have an impact on fusion success. For instance, deleterious effects of cigarette smoking on spinal fusion have been highlighted in several studies. 4 -6 Graft materials play a crucial role in bone remodeling, and the adequate choice is dependent on patient’s condition as well as the surgical approach. Ideally, the graft material should be osteoconductive, osteoinductive, and osteogenic. Autologous iliac crest bone graft (ICBG) represents the only stand-alone graft with all 3 components needed for fusion. Studies have shown that ICBG performed better in single- and 2-level fusions than in 3 or more level procedures. 7 -18 Additionally, studies have shown that the fusion rates with ICBG are often lower in noninstrumented than in instrumented lumbar procedures. 8,11 -13 While ICBG has all 3 desired graft properties, several drawbacks including donor comorbidities, limited supply, and various complications have been noted. 19 -21
As a result, the use of alternative materials such as allograft, demineralized bone matrix (DBM), synthetic materials (calcium sulfates, calcium phosphates, or hydroxyapatite), growth factors, and cell- or platelet-based therapies has greatly increased. Each of these graft materials has certain pros and cons that will guide patient selection. Allograft is readily available in large amounts and does not carry ICBG-related complications, in particular the harvest morbidity. During preparation, allografts are depleted of cells and growth factors and primarily provide osteoconduction with minor osteoinductivity. Its main disadvantages are immunogenicity and disease transmission. 22 Based on the preparation procedure, allografts can be divided into 3 groups: fresh-frozen, freeze-dried, and DBM. Fresh-frozen allografts provide the highest mechanical stability, but at the same time they carry the highest risk of disease transmission. On the other hand, DBM has certain osteoinductive capabilities in addition to osteoconduction. 23 However, studies have shown a large lot-to-lot variability. 24 Various studies have looked at the use of allograft in lumbar spine in combination with ICBG or as a stand-alone graft. In posterolateral instrumented fusion, An et al reported no fusion with freeze-dried allograft, while Gibson et al found similar outcomes and revision rates at 6-year follow-up. 25,26 Freeze-dried allografts in combination with autograft bone have been also used in deformity corrections, achieving over 90% fusion rates. 27,28 At the same time several studies have looked at the DBM fusion potential in lumbar spine. Both Vaccaro et al and Cammisa et al reported similar fusion rates between DBM mixed with bone marrow aspirate or ICBG and ICBG alone. 17,29 Despite the large number of clinical studies utilizing those graft materials, the existing reviews are often narrative rather than systematic, 30 -36 or the level of evidence is limited due to a small sample size, short follow-up times, lack of appropriate controls, or incomplete outcome information. 19,21,30,36 -41
The purpose of this systematic review was to determine the fusion efficacy of allograft and DBM in lumbar instrumented and noninstrumented fusion procedures for degenerative lumbar disorders.
Materials and Methods
Search Strategy
A clinical epidemiologist performed a literature search using PubMed and 5 databases of the Cochrane Library in November 2013 and January 2014, respectively (Figure 1). Since the employed terminology is extremely diverse, the search strings were kept general to avert the risk of missing eligible publications. In order to be included in the review, allograft bone and DBM had to be used in fusion procedures for degenerative conditions of the lumbar spine. Any type of allograft bone or DBM was eligible for inclusion in the review as long as it was not used in combination with freshly harvested ICBG. The reason for this exclusion was that (1) if ICBG autograft was part of the graft, it would remain unclear whether the effect on fusion stemmed from the autograft or the alternative material, and (2) the goal of the review was to evaluate the potential of allograft and DBM as substitutes for ICBG.
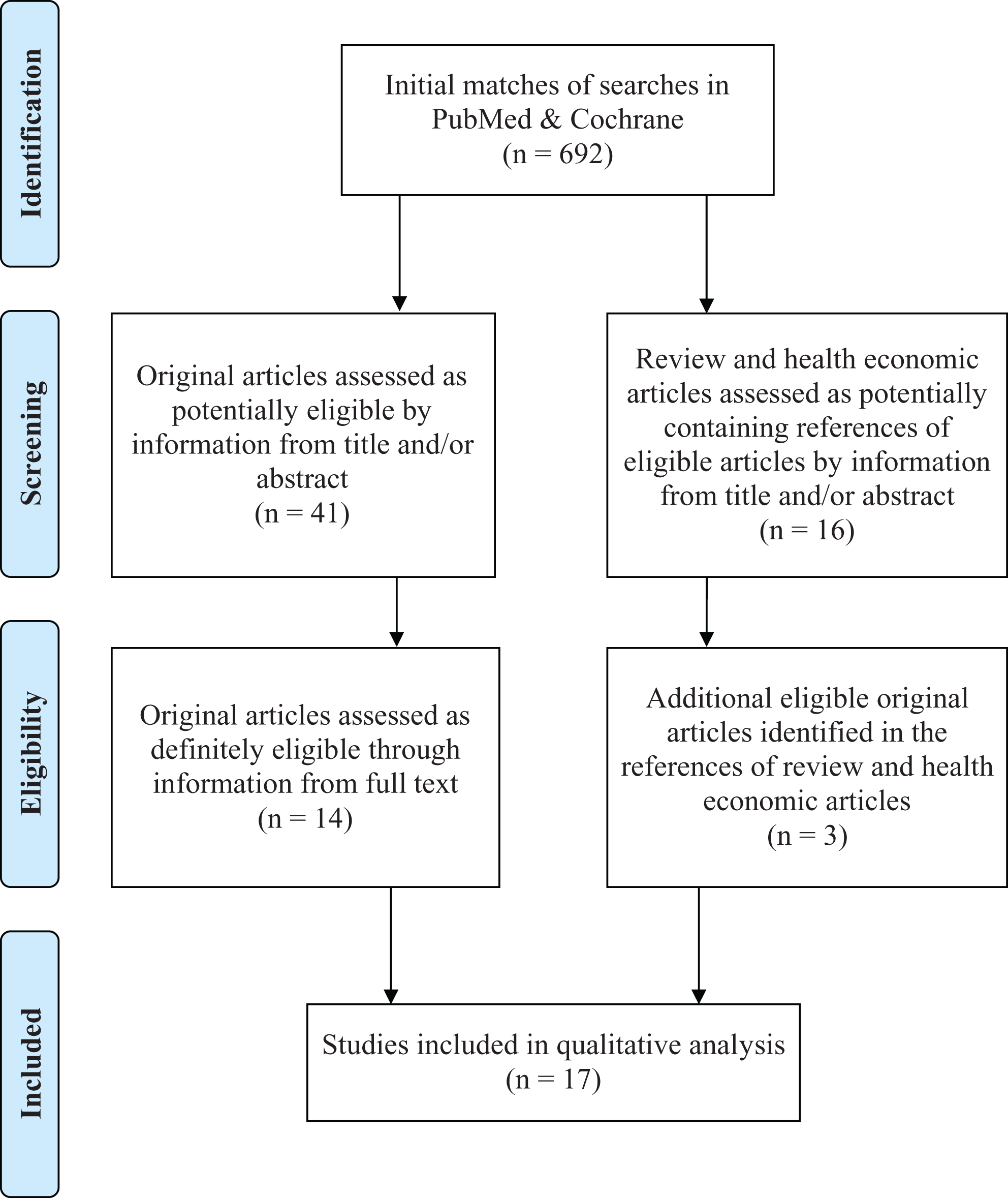
PRISMA 2009 flow diagram. From Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: the PRISMA statement.
Study Selection
Titles and abstracts of the initial matches were independently screened by 2 reviewers to identify eligible original publications. The initial search included original articles and articles with a focus on health economic aspects. When the title or the abstract were not sufficient to determine the eligibility, the full text was used. Whenever the 2 reviewers had a disagreement concerning the eligibility, a third reviewer decided.
Inclusion and Exclusion Criteria
Original publications were included if the following inclusion criteria were met:
Original publications were excluded if any of the following criteria applied:
Data Extraction
Information on study population, follow-up time, intervention (type of surgery and allograft used), fusion success, and its definition were collected. A standardized data extraction form was used. Any study characteristics that could have led to bias, for example, details about lost to follow-up or potential conflict of interest, were also captured.
Data Analysis
Due to the high variability among studies with regard to study focus, procedural type, definition of fusion, as well as the indication for surgery, all analysis were done on an individual level.
Results
A total of 692 potentially eligible original articles were identified (Figure 1). From those, 41 articles were retrieved for full-text evaluation and 14 were deemed appropriate for inclusion. Additionally, the full text of 16 review and health economic articles was screened for further eligible references and another 3 articles were included in the review. Subsequently, all eligible articles were screened for overlapping patient populations. Since there was no population overlap, all 17 articles were included in this review. No randomized controlled trial (RCT) comparing allograft or DBM to autograft met the eligibility criteria (Table 1).
Overview of Results.
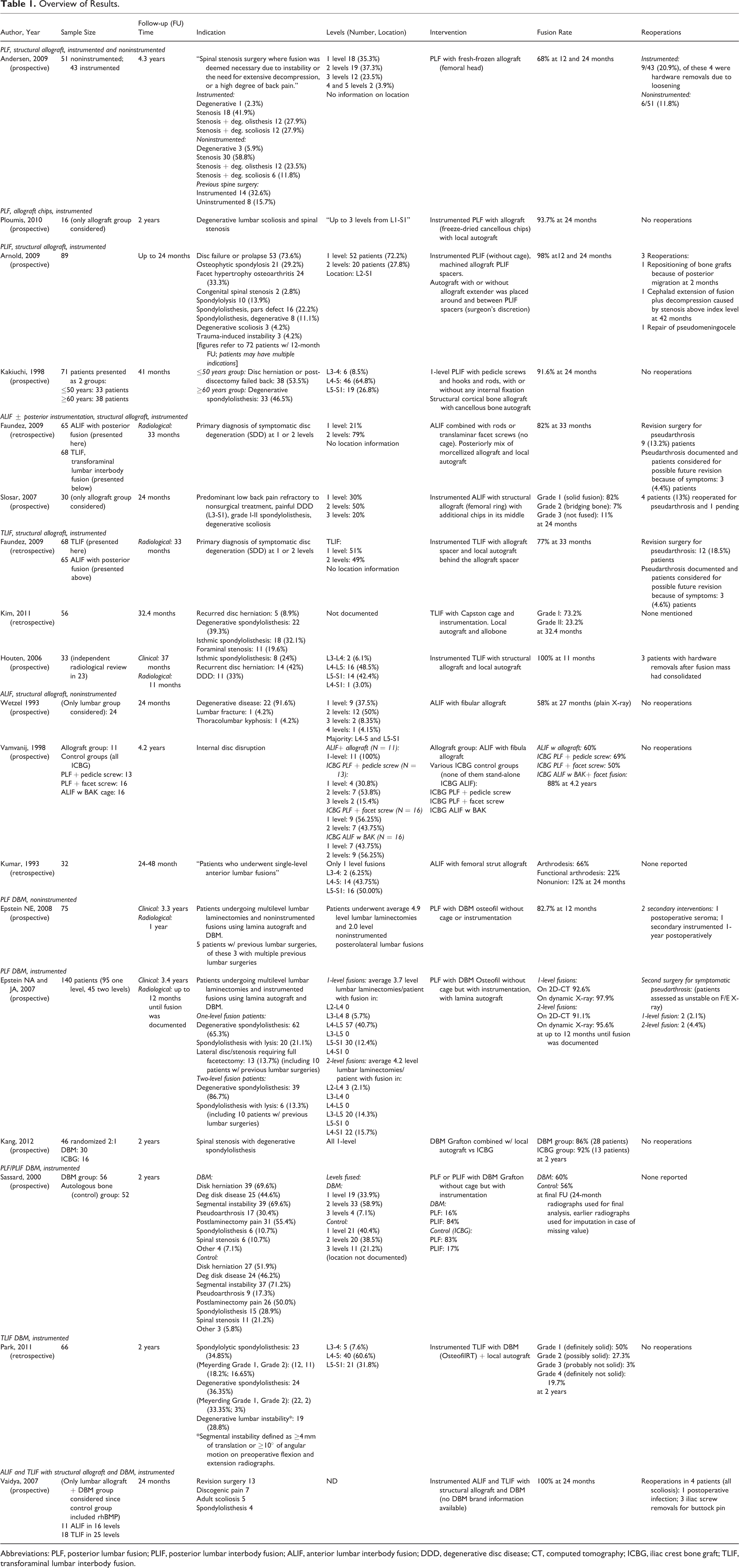
Abbreviations: PLF, posterior lumbar fusion; PLIF, posterior lumbar interbody fusion; ALIF, anterior lumbar interbody fusion; DDD, degenerative disc disease; CT, computed tomography; ICBG, iliac crest bone graft; TLIF, transforaminal lumbar interbody fusion.
Allograft
Eight prospective and 3 retrospective studies comparing allograft to autograft in posterior and anterior fusion with and without instrumentation were included. Only 5 out of 11 studies had groups with allograft alone. In the prospective study done by Andersen et al, 94 patients underwent instrumented (54.3%) or noninstrumented (45.7%) posterior lumbar fusion (PLF) procedures with fresh frozen femoral head allograft. 42 At 12 and 24 months, a fusion rate of 68% for noninstrumented and 81% for instrumented fusion was reported (Tables 2 and 3). Additional 3 prospective studies on PLF 43 and posterior lumbar interbody fusion (PLIF) 44,45 enrolled 16 to 89 patients and graft material included autograft mixed with various forms of allograft (freeze-dried, 43 machined, 44 or cortical 45 ). In all 3 studies patients underwent instrumented procedures, resulting in fusion rates between 91.6% and 98% at 24 months. For anterior fusion procedures 2 retrospective 46,47 and 3 prospective studies 48 -50 looked at the use of allograft in instrumented 46,48 and noninstrumented 47,49,50 fusion. Four out of 5 studies used structural allograft alone, 47 -50 with 2 studies using fibular 49,50 and 2 femoral bone graft. 47,48 In a case-control study done by Faundez et al, the type of structural allograft used for anterior interbody fusion included tricortical iliac crest allograft in 41 patients (59.7%), milled femoral ring in 12 (17.9%), patella allograft alone in 11 (16.4%), and a combination of patella allograft and iliac crest allograft in 4 patients (6%). 46 Two instrumented studies reported 82% fusion rates at 24 and 33 months, similar pseudarthrosis rates (13%), and significant improvements in the clinical outcomes. 46,48 On the other hand, in the 3 noninstrumented anterior lumbar interbody fusion (ALIF) studies fusion rates were lower 58% to 66% and one study reported that 85% of the cases had graft subsidence. 47,49,50 Although the fusion rates were lower than in instrumented studies, all 3 studies reported improvement in the clinical outcomes and return to work.
Definition of Fusion and Fusion Success.

Abbreviations: PLF, posterior lumbar fusion; PLIF, posterior lumbar interbody fusion; ALIF, anterior lumbar interbody fusion; CT, computed tomography; TLIF, transforaminal lumbar interbody fusion.
Availability of Power Calculation.
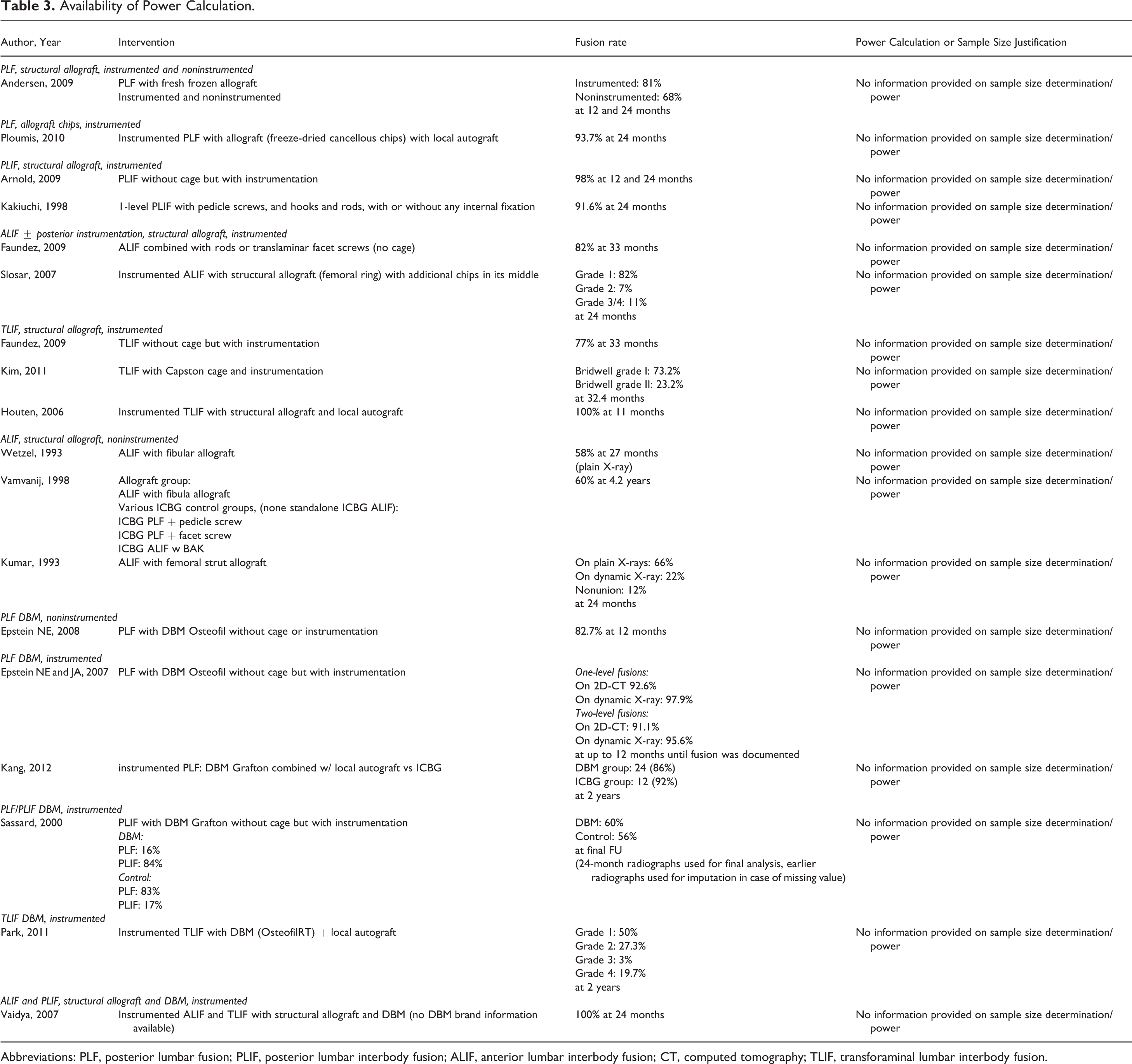
Abbreviations: PLF, posterior lumbar fusion; PLIF, posterior lumbar interbody fusion; ALIF, anterior lumbar interbody fusion; CT, computed tomography; TLIF, transforaminal lumbar interbody fusion.
Three studies reported on the use of allograft in transforaminal lumbar interbody fusion (TLIF) procedures. Faundez et al used a boomerang-shaped allograft spacer in 2 slightly different designs with 61.5% of the patients receiving a semilunar femoral ring graft and the other 38.5% having a split femoral ring allograft. 46 At 33 months, the authors reported 77% fusion rates and improvement in the Short Form-36 (SF-36) score (Tables 2 and 3). 46 Two additional nonrandomized studies utilizing TLIF with combination of local autograft and structural allograft were included in this review. 51,52 The studies enrolled 33 to 68 patients and there was no control group. Fusion rates were 73.2% at 32.4 months 51 and 100% at 11 months. 52 Both studies reported improvements in the clinical outcomes assessed by Oswestry Disability Index (ODI), Visual Analogue Scale, or Prolo Scale. 51,52
Demineralized Bone Matrix
Six studies on DBM, 1 noninstrumented 53 and 5 instrumented, 9,54 -57 were included in this review. Five (4 prospective and 1 retrospective) studies looked at DBM in posterior fusion, 9,53 -56 and one prospective study utilized DBM for both anterior and posterior procedures. 57
In the noninstrumented PLF study, 75 patients received lamina autograft and DBM paste at a 1:1 ratio (Osteofil, Medtronics Sofamor Danek, Memphis, TN). 53 At 12 months, 82.7% of the patients were deemed to be fused and the authors reported an improvement in the SF-36 score at the 1- and 2-year follow-up. 53 In 2 prospective studies patients underwent PLF with DBM and local graft. 9,54 In the study done by Epstein et al, 95 patients underwent single- and 45 two-level PLF with lamina autograft and DBM paste in a 50:50 ratio (Osteofil, Medtronics Sofamor Danek). 54 Fusion rates assessed on 2D computed tomography were 92.6% for 1-level and 91.1% for 2-level procedures up to 12 months postoperatively (Tables 2 and 3). When the fusion was assessed on dynamic X-rays, the rates were 97.9% for 1-level and 95.6% for 2-level procedures. Kang and coworkers enrolled 46 patients randomly assigned to Grafton DBM Matrix (Medtronics, Memphis, TN) with local bone (30 patients) or ICBG (16 patients). 9 At the 2-year follow-up, fusion rates of the DBM and ICBG groups were similar (86% vs 92%) along with the ODI and SF-36 score. 9
Three studies, 2 prospective and 1 retrospective, utilized a combination of DBM and autograft in PLF/PLIF, TLIF, or ALIF procedures. 55 -57 In a prospective case-control study, 56 patients underwent PLF or PLF/PLIF with Grafton DBM and local autograft, and 52 patients received ICBG. 55 At 24 months, the results showed a 60% fusion rate in the DBM and a 56% fusion rate in the control group (Tables 2 and 3). However, this difference was not statistically significant. 55 On the other hand, 100% fusion rates and minimal graft subsidence were reported by Vaidya et al, where patients underwent either ALIF (11) or TLIF (18) procedures with DBM and allograft. 57 In a retrospective study, 66 patients underwent TLIF with a combination of DBM paste (OsteofilRT DBM paste; Regeneration Technologies Inc, Alachua, FL) and local autograft. 56 At the 2-year follow-up, solid fusion was achieved in 77% of the patients; however, there were no significant differences in the clinical outcomes between patients with solid fusion and nonunion (Tables 2 and 3). 56
Study Demographics and Surgery
A sufficient description of patient baseline characteristics (age, gender distribution, and diagnoses) was given in 11 out of 17 citations. 9,42 -44,46,48,50,52,54,55 Yet only 7 articles explicitly described the inclusion and exclusion criteria for study participation. 9,44,46,48,50,51,55 None of the publications provided a power calculation or justified their sample size.
The number of fused levels was provided by 15 out of 17 studies, 9,42 -50,52 -55,57 but only one third of the studies specified the exact locations. 45,47,52 -54
Outcome Assessment
In the majority of studies, the fusion assessment was performed by an independent or blinded observer. 9,43,44,46 -48,50,52 -55,57 The definition of fusion varied between the studies, with no studies having the same fusion assessment protocol (Table 2). When analyzing the individual fusion definitions in relation to the achieved fusion rates, there were no discrepancies between the fusion percentage and the assessment stringency (Table 2).
Lost to Follow-up
In general, the rates of lost to follow-up were quite variable. Eight studies that documented radiological and clinical outcomes at various time points did not make any statements with regards to “lost to follow- up.” 43,45,47,49,50,53,54,57 On the other hand, in the studies done by Kim et al 51 and Houten et al, 52 all of the patients had complete follow-ups for both radiological and clinical outcomes. Andersen et al reported clinical outcomes after a mean of 4.3 years and radiological outcomes up to 24 months. The follow-up rate in this study was 79%. 42 This was the sole study that provided information on why patients were lost to follow-up and also analyzed the characteristics of patients included in the follow-up versus those lost to follow-up. In the studies conducted by Arnold et al 44 and Sassard et al, 55 the number of patients available for follow-up decreased from 12 to 24 months, from 83% to 54% 44 and from 81% to 76%, 55 respectively. Similar results for the clinical outcomes were observed in a study conducting by Kang et al. 9 In the study done by Faundez and coworkers, radiological data was available for 59% of the ALIF and 54% of TLIF patients at 33 months, and the clinical follow-up rate was 64%. 46 Slosar and coworkers followed-up patients up to 24 months for both radiological and clinical outcomes, and only 3 patients dropped over the 24-month time period. 48
Conflict of Interest Declaration
In 7 of the 17 articles, no conflict of interest statements was provided. 42,45 -47,50 -52 Six studies were funded by the industry, 9,48,49,53 -55 and in 2 publications, some authors declared a consultancy role for the manufacturer of the used allograft. 44,48
Discussion
The purpose of this review was to determine the fusion efficacy of allograft and DBM in lumbar instrumented and noninstrumented fusion procedures for degenerative lumbar disorders. Seventeen studies met the inclusion criteria. However, a large variation in fusion rates, a lack of control group, and follow-up time prevented any significant conclusions. There were no obvious differences between allograft and DBM with regard to instrumented fusion procedures. For the allograft, the fusion rates were between 58% and 68% for noninstrumented lumbar fusions 42,47,49,50 and between 73% and 100% for instrumented fusions with and without additional local autograft. 42 -46,48,51,52 Regarding DBM, one study reported fusion rates of 82.7% for noninstrumented fusion, 53 with the remaining studies reporting between 60% and 100% for instrumented procedures with or without autograft. 9,54 -57
Autograft is the only graft material that has all 3 characteristics of the ideal graft: osteoconductivity, osteogenecity, and osteoinduction. Despite being considered the gold standard, a wide range of fusion rates has been reported in the literature. The use of autograft in instrumented lumbar fusion procedures has led to fusion rates between 54% and 99%, 7 -18,29 and for noninstrumented fusion between 30% and 100%. 10,11,58 -64
Based on those findings, allograft and DBM use had fusion ranges that overlap with autograft fusion rate ranges. Those results are in line with the recommendations created by Fischer and coworkers using Guyatt criteria and focusing on non–bone morphogenetic protein graft materials. 39 In their review, a 1B grade was given to allograft use in PLF and ALIF approaches, reflecting a strong recommendation with a clear benefit to most of the patients. For DBM, the grading was dependent on whether DBM was used as a substitute or extender. The authors gave a 1C grade for PLF and ALIF surgeries due to the lack of RCTs.
Although the allograft and DBM can provide comparable fusion rates, our review found a large variability between the studies. There are several reasons that may have contributed to the heterogeneity, including the variability in the surgical approaches used to achieve fusion. Surgical techniques have been a subject of previous research and some of the approaches have demonstrated a clear advantage over others. Future prospective longitudinal studies are needed to establish what approach and material are best for a patient.
The diversity in diagnosis as well as the number of surgical levels was high, leading to too many confounding variables. Studies on allograft included spinal stenosis, degenerative lumbar scoliosis, spondylolisthesis, disc herniation or prolapse, trauma, and several others. 42 -52 The number of fused levels varied between single- to 5-level fusion, sometimes within the same study. Similar to the allograft studies, diagnosis heterogeneity was seen in the studies focusing on DBM. 9,53 -57 Furthermore, most of the studies with DBM included patients with failed surgery or pseudarthrosis as part of their cohorts. If recorded, the number of fused levels ranged between 1 and 5 vertebra. Another limitation in all of the studies was the methodological weakness. Most of the studies were purely observational without a control group and included poorly defined and/or mixed indications, procedures, and implants. None of the publications provided a power analysis or sample size justification.
Although most of the studies described the protocol for fusion assessment, there was no consistency in the definition of fusion. The differences in the individual fusion definitions revealed that patients deemed as fused in one study would be assessed as partially or inadequately fused in another study (Table 2). Only in 4 studies fusion rates were assessed by multiple observers, surgeons, and/or radiologists 43,53,56,57 ; however, blinded assessment was done in only 2 studies. 43,53 None of the 17 studies included in this review reported intra- or interobserver kappa values on fusion assessment reliability.
Aside from the surgical and clinical inconsistencies, the DBM preparation techniques are likely a strong contributor to the differences in fusion rates. Different methods of processing and sterilization influence DBM osteoinductivity, which together with the type of carrier and donor’s variability contribute to the wide range in fusion rates.
Conclusion
Based on an analysis of 17 studies, this review found a large variation in the fusion rates, surgical approaches, lack of control groups, and follow-up time preventing any significant conclusions. No RCT comparing allograft or DBM to autograft as an adjunct to fusion met the eligibility criteria for this review. Well-designed RCTs employing standardized surgical techniques, addressing the clinical safety and efficacy of allograft and DBM compared to autograft in large, homogeneous patient populations, and a sufficient follow-up time are needed to unequivocally establish the usefulness and efficacy of these materials as an adjunct to fusion.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Financial activities outside of submitted work: ZB-Xenco Medical (consultancy) and AO Spine (consultancy, past). JBP and ER—Nothing to disclose. HJM—Dr Meisel is a consultant (money paid to institution): Regenerate Life Sciences GmbH for Zyga, DiFusion (ongoing), Co.don (paid to Dr Meisel, past); royalties from Medtronic, Fehling Aesculap (past); owns stocks (money paid to institution): Regenerate Life Sciences GmbH in DiFusion. STY—Dr Yoon owns stock in Phygen, Alphatec, Meditech; royalties: Meditech Advisors, Stryker Spine (paid directly to institution/employer), grant from AOSpine (paid directly to institution/employer), research support from Biomet (research support given to AREF), nonfinancial research support from Nuvasive and Medtronic. JAY—Royalties: NuVasive, Osprey Medical, Amedica, Integra; Stock ownership: Benvenue Medical, Paradigm Spine, Promethean Surgical Devices, Spinal Ventures, VertiFlex, Spinicity, ISD, Providence Medical; Private investments: Amedica, VertiFlex, Benvenue, NuVasive; Consulting: Integra, NuVasive, Amedica, HealthTrust; Board of directors: Durango Orthopedic Associates (None); Research support (staff and/or materials): Globus Medical (paid directly to institution/employer), NuVasive (paid directly to institution/employer), VertiFlex (paid directly to institution/employer), Integra (paid directly to institution/employer). DSB—Consultant: Vallum; Royalties: Amedica, DePuy Synthes, Medtronic; Fellowship support: AOSpine (paid directly to institution). JCW—Royalties: Aesculap, Biomet, Amedica, Seaspine, Synthes; Stock ownership: Fziomed; Private investments: Promethean Spine, Paradigm Spine, Benevenue, NexGen, Vertiflex, Electrocore, Surgitech, Expanding Orthopaedics, Osprey, Bone Biologics, Curative Biosciences, Pearldiver; Board of directors: North American Spine Society (Second Vice President), North American Spine Foundation (nonfinancial), Cervical Spine Research Society (travel expenses), AO Spine/AO Foundation (honorariums for board position); Fellowship support: AO Foundation (spine fellowship funding paid to institution).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by AOSpine and departmental funds. AOSpine is a clinical division of the AO Foundation—an independent medically guided nonprofit organization. The AOSpine Knowledge Forums are pathology focused working groups acting on behalf of AOSpine in their domain of scientific expertise. Each forum consists of a steering committee of up to 10 international spine experts who meet on a regular basis to discuss research, assess the best evidence for current practices, and formulate clinical trials to advance spine care worldwide. Study support is provided directly through AOSpine’s Research Department.
