Abstract
Study Design
Systematic review.
Objectives
The study’s primary objective was to determine how osteobiologic choice affects fusion rates in patients undergoing anterior cervical discectomy and fusion (ACDF). The study’s secondary objectives were to 1) determine the optimal timing of fusion assessment following ACDF and 2) determine if osteobiologic type affects the timing and optimal modality of fusion assessment.
Methods
A systematic search of PubMed/MEDLINE was conducted for literature published from 2000 through October 2020 comparing anterior fusion in the cervical spine with various osteobiologics. Both comparative studies and case series of ≥10 patients were included.
Results
A total of 74 studies met the inclusion criteria. Seventeen studies evaluated the efficacy of autograft on fusion outcomes, and 23 studies assessed the efficacy of allograft on fusion outcomes. 3 studies evaluated the efficacy of demineralized bone matrix, and seven assessed the efficacy of rhBMP-2 on fusion outcomes. Other limited studies evaluated the efficacy of ceramics and bioactive glasses on fusion outcomes, and 4 assessed the efficacy of stem cell products. Most studies utilized dynamic radiographs for the assessment of fusion. Overall, there was a general lack of supportive data to determine the optimal timing of fusion assessment meaningfully or if osteobiologic type influenced fusion timing.
Conclusions
Achieving fusion following ACDF appears to remain an intricate interplay between host biology and various surgical factors, including the selection of osteobiologics. While alternative osteobiologics to autograft exist and may produce acceptable fusion rates, limitations in study methodology prevent any definitive conclusions from existing literature.
Introduction
Anterior cervical discectomy and fusion (ACDF) remains the gold standard for the treatment of symptomatic cervical degenerative disc disease with a demonstrable track record of excellent clinical outcomes. Fusion rates are quite high in most cases, however, in longer fusions, high rates of pseudarthrosis remains a surgical quandary.1,2 Fortunately, asymptomatic pseudarthrosis has been shown to be a relatively common clinical entity 3 and with improving surgical technique, rates of fusion actually appear to be improving. However, pseudarthrosis may nonetheless be associated with significant patient morbidity with up to 37% patients with pseudarthrosis requiring revision surgery by the 7 years follow-up. 4
When performing ACDF, choice of osteobiologic appears to be an important part of the equation in creating the ideal fusion environment. Autologous iliac crest bone graft (ICBG) has long remained the gold standard media for fusion as it exhibits all three quintessential characteristics needed for bony fusion (i.e. osteogenic, osteoconductive, and osteoinductive properties) while simultaneously evading the risk of disease transmission and immunogenecity. However, donor site morbidity and a finite quantity are well recognized significant downsides to use of ICBG.5,6
Therefore, increasing interest in the development of the optimal ICBG alternative has arisen to circumvent these aforementioned disadvantages. Examples of these include allograft, ceramics and bioactive glasses, synthetic osteo-inductive proteins and peptides, and stem cells --- each, of course, with their respective strengths and weaknesses.7-11 However, despite a substantial amount of respective published data, no clear consensus exists regarding the optimal osteobiologic for use in ACDF.
Consequently, we performed a systematic review to critically evaluate the current evidence on use of osteobiologics in ACDF. In this context, with the recent explosion in the number of available osteobiologics, in addition to determining between group differences in rates of fusion, we sought to quantify any disparities that existed in regards to time to apparent fusion. Further, we thought it prudent to qualify if the osteobiologic utilized potentially affects the choice of optimal imaging modality that should be utilized to confirm fusion. Therefore, the primary objective of the study was to determine how osteobiologic choice affects fusion rates in patients undergoing anterior cervical discectomy and fusion (ACDF). Secondary objectives of the study were to (1) determine the optimal timing of fusion assessment following ACDF and (2) to determine if osteobiologic type affects the timing and optimal modality of fusion assessment.
Materials and Methods
Electronic Literature Search
A systematic search of PubMed/MEDLINE was conducted for literature published through October 2020 by 2 independent reviewers. The search strategy included use of controlled vocabulary (MeSH terms) as well key words (Appendix A). After identifying appropriate studies based on title and abstract, full manuscripts were then reviewed. Only English language clinical studies evaluating the efficacy of various osteobiologics in the setting of ACDF were included in the review. All basic science studies were therefore excluded. Reference lists of full-text manuscripts and relevant systematic reviews were additionally reviewed to identify any additional pertinent studies not previously identified. A full list of study inclusion and exclusion criteria can be reviewed in Appendix B. The search flow chart with articles excluded or included was reported in a PRISMA flowchart (Figure 1). Diagram of review process utilizing Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.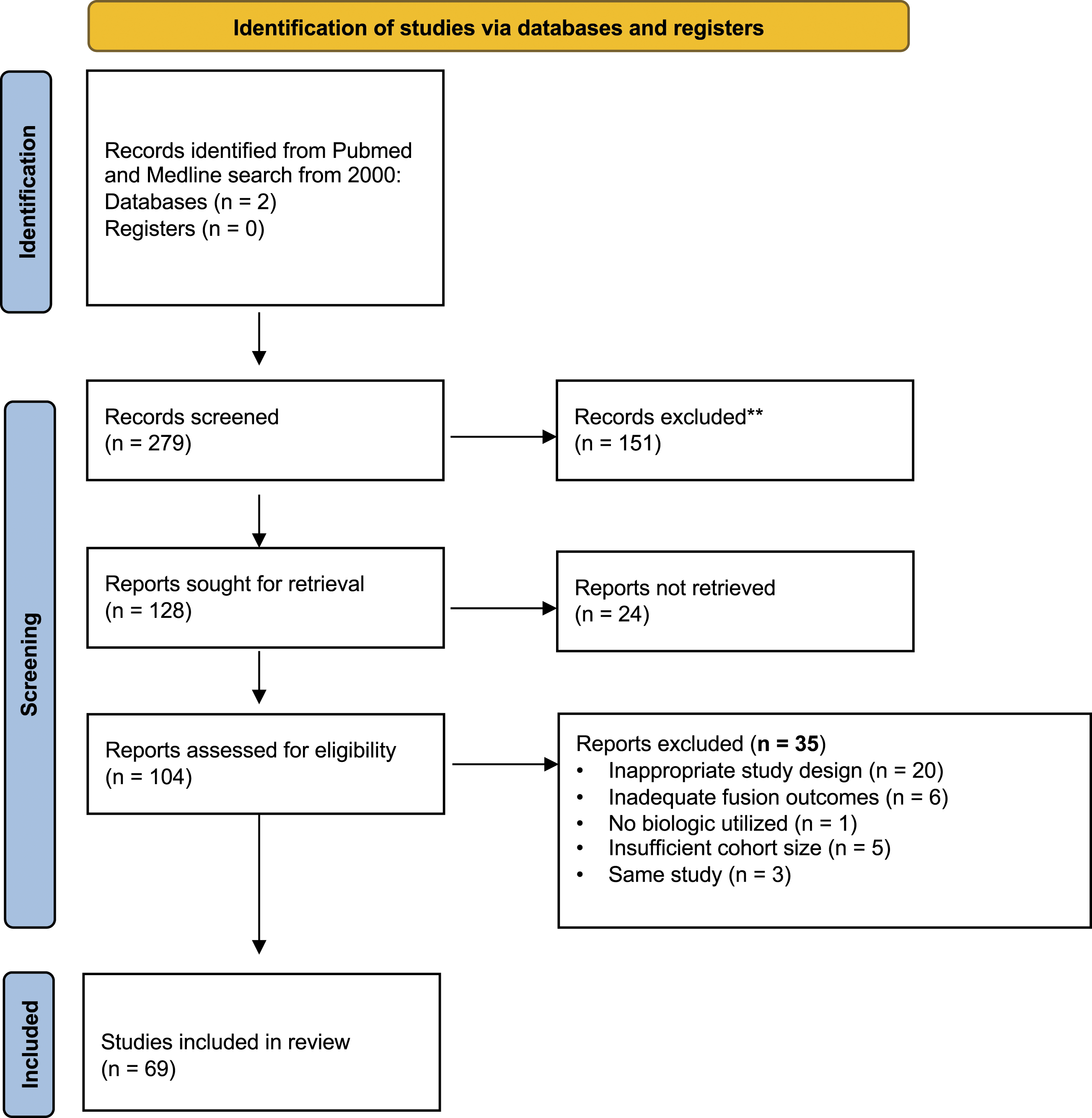
Data Collection
If available, the following data was extracted: (1) study design, (2) patient characteristics (patient demographics, risk factors for non-union, smoking status, underlying diagnosis), (3) interventions and comparative groups (if applicable), (4) fusion outcomes (including time interval of fusion assessment – if available), (5) risk factors for pseudarthrosis (6) methodology used to assess for fusion (as one our secondary objectives was to determine the optimal fusion assessment modality based on osteobiologic selection, we made note of the specific fusion assessment techniques utilized by each individual study i.e. plain radiographs versus computed tomography), (7) fusion outcomes, (8) length of follow-up.
Quality Assessment Methods
The quality of nonrandomized studies (comparative and non-comparative) was assessed using the Methodological Index for Non-Randomized Studies (MINORS). 12 The methodological quality of the randomized studies and their risk of bias were using RevMan 5.4.0 software, according to the risk of bias assessment of the Cochrane handbook. 13 The assessment criteria consisted of 5 items: Selection bias (allocation concealment and random sequence generation); Performance bias (blinding of participants and personnel); Detection bias (blinding of outcome assessors); Attrition bias (dropouts and exclusion addressing and intention to treat analysis); Reporting bias (selective or nonselective reporting). Each study was evaluated as High, Low, or Unclear risk of bias for each item.
Results
Search Strategy
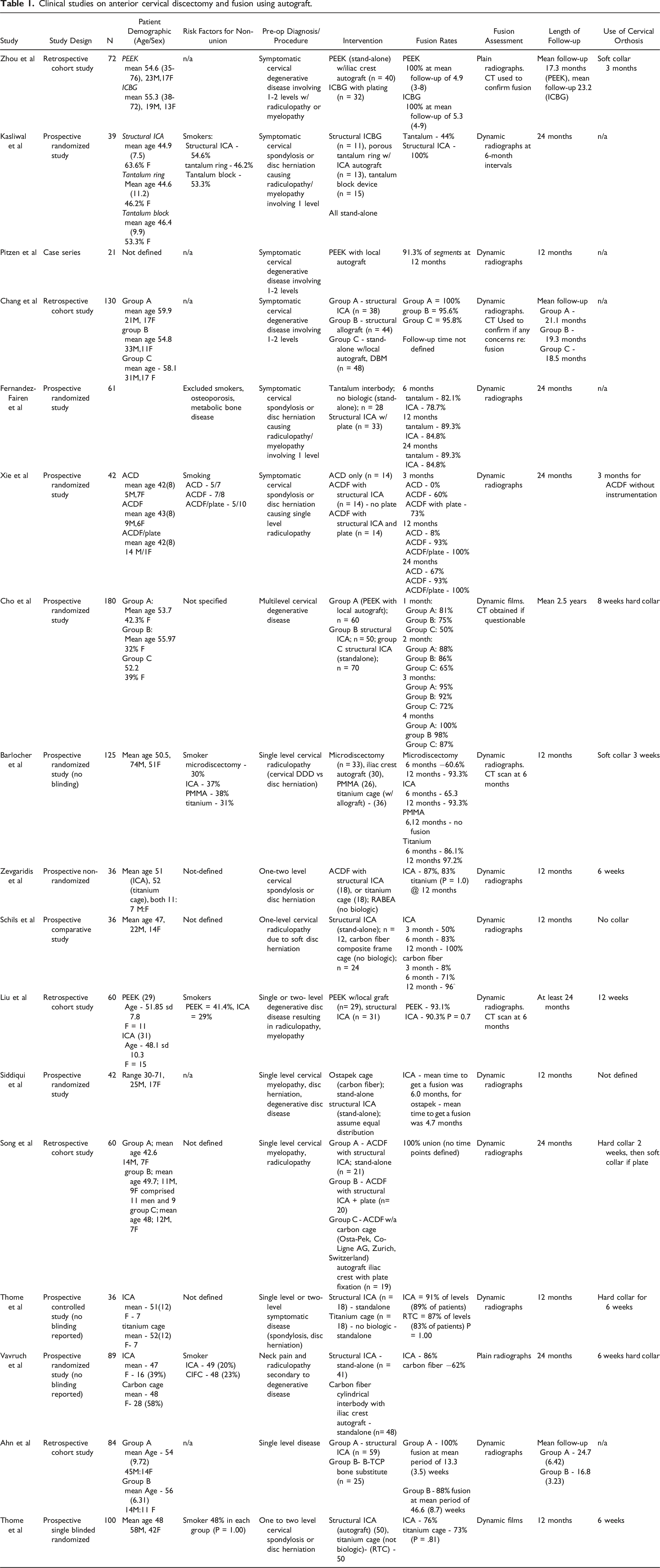
Clinical studies on anterior cervical discectomy and fusion using autograft.
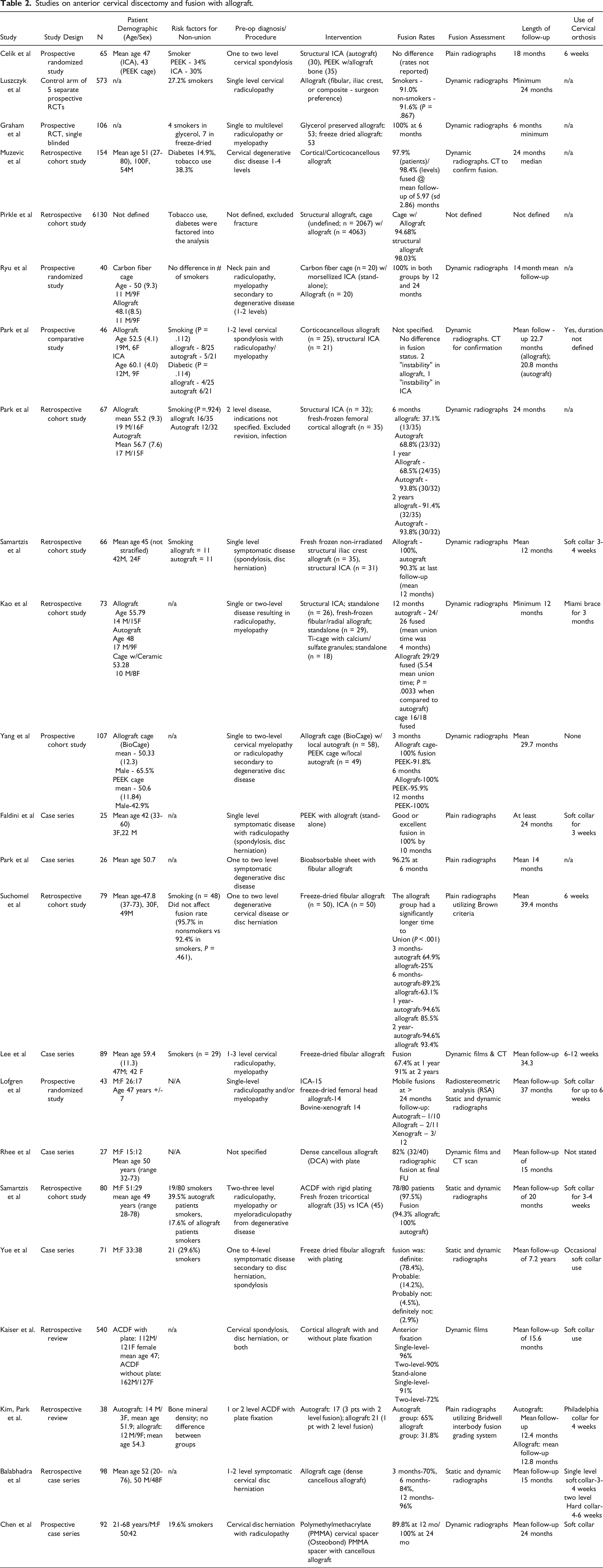
Studies on anterior cervical discectomy and fusion with allograft.
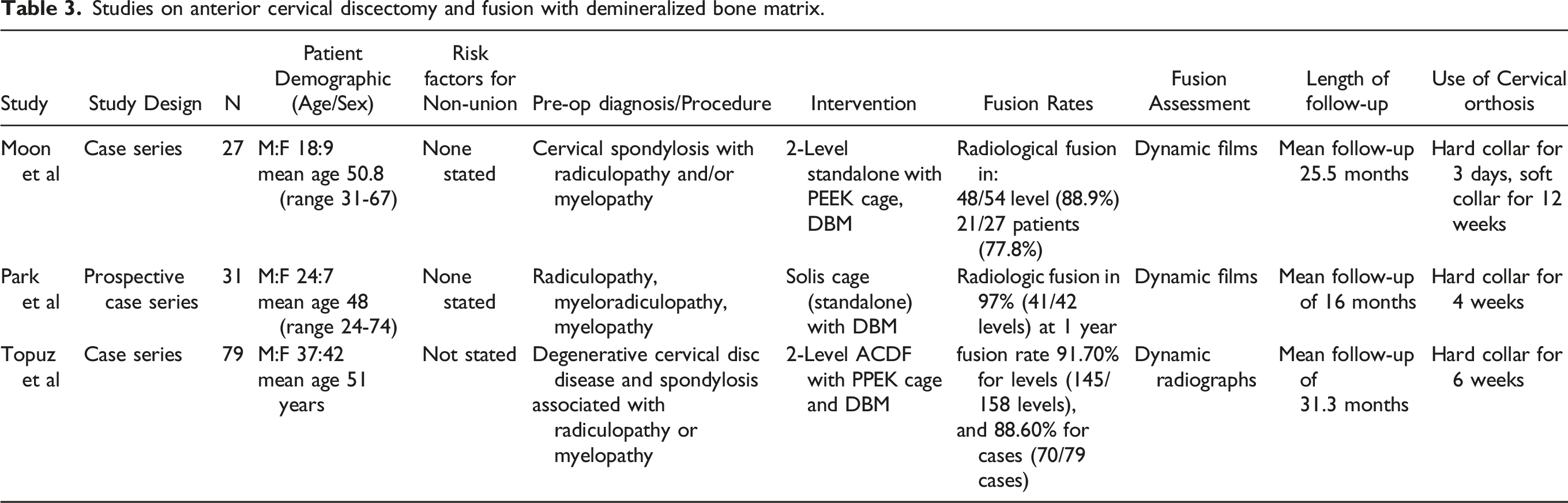
Studies on anterior cervical discectomy and fusion with demineralized bone matrix.
Efficacy of Osteobiologics on Fusion Outcomes
Autograft
There was a total of 17 studies that evaluated the efficacy of autograft on fusion outcomes (Table 1).14-30 There were 7 prospective randomized studies, 5 retrospective comparative studies, 4 prospective comparative studies, and 1 case series. All studies had at least 12 months of follow-up. Fusions rates ranged from 76% to 100% at final follow-up. All studies except one utilized ICBG. 16 Fifteen of 17 studies utilized dynamic radiographs to evaluate fusion status. One study used computed tomography alone. 24 Vavruch et al used static films alone. 28
Allograft
There was a total of 23 studies that evaluated the efficacy of allograft on fusion outcomes (Table 2).31-53 There were 5 prospective randomized studies, 9 retrospective comparative studies, 2 prospective comparative studies, and 7 case series. All studies had at least 6 months of follow-up. Fusions rates ranged from 31.8% to 100% at final follow-up. Two studies utilized PEEK with allograft,31,42 17 studies utilized some form of structural allograft, 1 study utilized an allograft cage with local autograft, 41 1 with allograft cage alone, 52 and 1 study utilized a PMMA spacer with cancellous allograft. 53 Seventeen studies utilized dynamic radiographs to evaluate fusion status. 5 studies used only static imaging alone.31,42-44,51 One study did not define how they evaluated fusion. 35
Demineralized Bone Matrix
There was a total of 3 studies that evaluated the efficacy of demineralized bone matrix on fusion outcomes (Table 3).54-56 All 3 studies were case series and had at least 12 months follow-up. Fusion rates ranged from 77.8 to 97.0% at final follow-up. All 3 studies used dynamic radiographs to evaluate fusion.
rhBMP-2
There were a total of 7 studies that evaluated the efficacy of rhBMP-2 on fusion outcomes (Supplemental Table 4) These included one prospective randomized study, 2 prospective cohort studies, 2 retrospective cohort studies, and 2 retrospective case series. All studies had at least 6 months follow-up and most studies had greater than 24 months of mean follow up. Fusions rates ranged from 95% to 100% at final follow-up. 5 studies utilized PEEK with rhBMP-2,57-63 1 study utilized fibular allograft with rhBMP-2, 62 and 1 study utilized structural allograft with rhBMP-2. 63 To assess for radiographic fusion, 1 study utilized a combination of static radiographs and CT, 57 2 studies utilized a combination of static and dynamic radiographs,58,61 3 studies utilized a combination of dynamic radiographs and CT59,62,63 and 1 study utilized CT alone. 60
Ceramics and Bioactive Glasses
There was a total of 14 studies that evaluated the efficacy of ceramics and bioactive glasses on fusion outcomes (Supplemental Table 5).64-77 These included 6 prospective randomized studies, one prospective cohort study, 4 retrospective cohort studies, and 3 case series. All studies had at least 6 months of follow-up, although most had significantly longer follow-up, ranging up to several years. Fusion rates ranged from 62.5% to 100% at final follow up. A variety of combinations of implants and commercially available ceramics and bioactive glasses were utilized in the included studies. 5 studies utilized block hydroxyapatite,67,70,71,73,76 3 studies utilized PEEK with beta-tricalcium phosphate,68,72,74 one study utilized PEEK with calcium phosphate ceramic, 64 one study utilized Silicon Nitride, 66 , one study utilized acrylic with tricalcium phosphate, 65 one study utilized PEEK with hydroxyapatite, 75 one study utilized titanium cage with beta-tricalcium phosphate, 69 and one study utilized tubular titanium cages packed with either hydroxyapatite or beta-tricalcium phosphate. 77 To assess for radiographic fusion, seven studies utilized a combination of dynamic radiographs and CT,66,67,69,72,74,75,77 3 studies utilized dynamic radiographs alone,70,73,76 one study utilized static radiographs alone, 65 one study utilized a combination of static radiographs and CT, 71 one study utilized a combination of static and dynamic radiographs, 64 and one study utilized CT alone 68
Synthetic Peptides
A single study included evaluated the efficacy of synthetic peptides, specifically I-factor or P-15, on fusion outcomes (Supplemental Table 6). 7 The study by Arnold et al. is a prospective randomized study with 2 years of follow up. Fusion rates for I-factor were 97.3% at final follow up. The investigators utilized cortical ring allograft filled with i-factor. Fusion was assessed using static and dynamic radiographs, and CT scan.
Stem Cell Products
There was a total of 4 studies that evaluated the efficacy of stem cell products on fusion outcomes (Supplemental Table 7).78-81 These included one retrospective cohort study, and 3 prospective case series. All of these studies had a minimum of 12 months of follow up. Fusion rates ranged from 87.7% to 95% at final follow-up. 3 studies utilized PEEK interbody device with allograft cellular bone matrix.79-81 One study utilized structural allograft with cellular bone matrix. 78 To assess for radiographic fusion, all 4 studies utilized a combination of dynamic radiographs and CT scan.
Optimal Timing of Fusion Assessment
Autograft
4 studies looked specifically at timing of fusion in the setting of ICBG. Zhou et al reported a 100% fusion rate at a mean follow-up of 5.3 months, 14 Cho et al reported a 98% and 87% fusion rate by 4 months with and without plate fixation, 20 respectively. Siddiqui et al noted a mean time to full fusion of 6 months 25 and Ahn et al demonstrated a 100% fusion rate at a mean of 3.5 months follow-up. 29
Allograft
Three studies evaluated time to fusion with use of allograft. Muzevic et al demonstrated a 97.9% fusion rate at a mean time of 6.0 months (sd 2.9) utilizing corticocancellous allograft. 34 Kao et al demonstrated a mean union time of 5.5 months with use of fresh frozen fibula and radial allograft when compared to autograft (mean 4.0 months; P = .003). 40 Finally, Suchomel et al found that allograft had a longer time to fusion than autograft with freeze-dried fibular allograft not achieving fusion until approximately 2 years compared to 1 year with autograft. 44
rhBMP-2
Two studies evaluated time to fusion with use of rhBMP-2. Stachniak et al. demonstrated a 95% fusion rate at 6 months, and 100% fusion rate at 9 months in patients that had undergone 2- or 3-level ACDF using PEEK interbody cages with rhBMP-2. 60 Vaidya et al. evaluated for radiographic evidence of fusion at multiple time points, demonstrated increasing percentages of patients that had achieved “probable fusion” at each successive time point, with all 38 (100%) patients achieving probable fusion by 12 months. 57
Ceramics and Bioactive Glasses
Seven studies evaluated time to fusion with use of ceramics and bioactive glasses. In their study of PEEK with Triosite (HA/B-TCP) compared to PEEK with ICA, Cho et al. demonstrated fusion rates of 100% at 6 months. 64 Prior to the six-month time point, fusion rates increased with time. Furthermore, they were noted to be reduced, with time to fusion delayed, with increasing number of levels fused. 64 Arts et al. demonstrated increasing fusion rates at multiple successive time points with use of Silicon Nitride interbody device compared to PEEK with local autograft. Fusion rates were shown to be 66.7%, 76.3%, 81.4% and 93% at 3, 6, 12 and 24 months, respectively. 66 In their study of B-TCP packed in PEEK interbody devices compared to structural ICA, Park et al. demonstrated fusion in 19/24 subjects at 12 months, which increased to 22/24 subjects fused at 24 months. 74 In their study of titanium interbody devices with B-TCP compared to ICA, Yamagata et al. demonstrated 21.9% and 60.9% rates of osseous union and partial union, respectively, at 1 year. These rates increased to 54.7% and 35.9% for osseous union and partial union, respectively, at 2 years. 69 Sugawara et al. demonstrated increasing rates of fusion at multiple successive time points for both B-TCP and HA. For B-TCP, fusion rates were 46%, 69% and 94% at 6 months, 1 year and 2 years, respectively. For HA, fusion rates were 24%, 49% and 90% at 6 months, 1 year and 2 years, respectively. 77 Dai and Jiang demonstrated increasing rates of fusion with time in subjects undergoing ACDF with PEEK or carbon fiber cage and B-TCP, both with, and without anterior plating. At 3 months, subjects with anterior plating demonstrated fusion rates of 98.1%, while those without anterior plating demonstrated fusion rates of 72.3%. Fusion rates in both groups increased to 100% at 6 months. 72 In their study of coralline HA, Agrillo et al. demonstrated fusion rates of 70% and 90% in smokers and nonsmokers, respectively, at 6 months. Fusion rates increased to 100% in both smokers and nonsmokers at 12 months. 73
Synthetic Peptides
A single study evaluated time to fusion with use of synthetic peptides, specifically i-factor. Arnold et al. demonstrated fusion rates of 88.9% at 12 months. This rate increased to 97.3% at 24 months. 7
Stem Cell Products
Two studies evaluated time to fusion with use of cellular bone allograft. Peppers et al. demonstrated fusion rates of 65.7% at 6 months. This rate increased to 89.4% at 12 months. 79 Vanichkachorn et al. demonstrated fusion rates of 78.6% at 6 months. This rate increased to 93.5% at 12 months. 80
Methodological Quality of Included Studies
The vast majority of studies were non-randomized. Risk of bias in published clinical trials for each study group is demonstrated in Supplemental Figures 3 through 12.
In general, the quality of the included non-randomized studies which evaluated the reviewed biologics ranged from moderate to high. As presented in Figure 2, MINORS scores ranged from 11 to 16 for non-comparative studies and 24 to 27 for comparative studies. Only 4% (2/50) comparative studies received a score of 24/24. No non-comparative studies received a score of 16/16. Summary of methodological index for non-randomized studies (MINORS) Grading.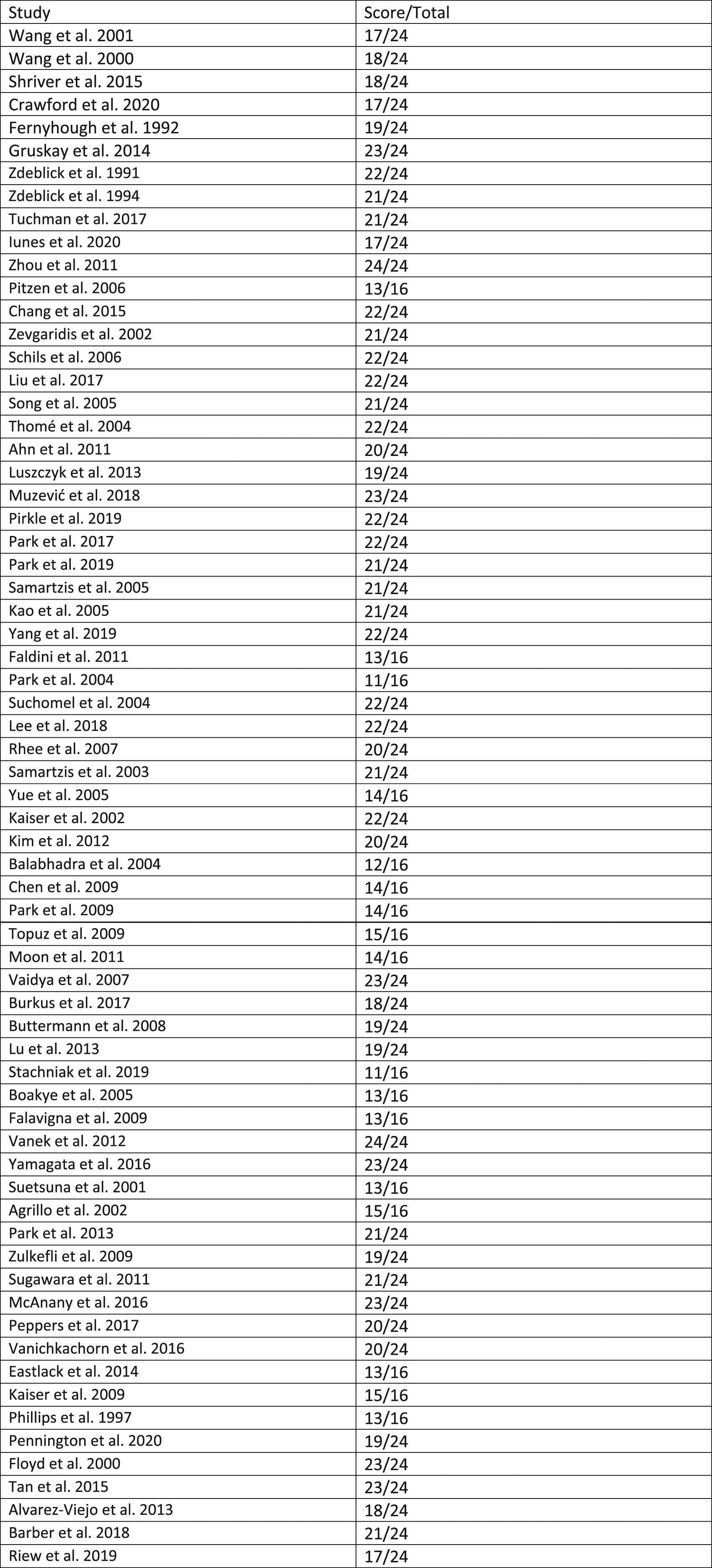
Discussion
Despite high rates of fusion following ACDF, pseudarthrosis nonetheless remains a significant surgical dilemma that may contribute to significant patient morbidity3,82,83 with published revision rates of up to 37% in patients with persistent pseudarthrosis. 84 Revision surgery is associated with poorer patient perceived outcomes when compared to primary surgery. 84 Further, these surgical episodes are costly. This further incentivizes efforts to maximize fusion outcomes in this increasingly cost-conscious health care landscape. Again, while ICBG remains the gold standard for fusion, it comes at the cost of substantial morbidity and finite quantity. Rates of morbidity have been reported at rates of up to 39% with associated complications including pain, hematoma, pelvic wing fracture, and localized nerve injury.5,85 Therefore, the identification of a suitable alternative to ICBG has spurred exponential growth in the field of osteobiologics. Ultimately, it appears that while there exist acceptable alternatives, to date, none clearly show superiority to structural autograft.
Allograft has long been hailed as an excellent alternative to ICBG. Lack of donor site morbidity, decreased blood loss, and reduced operative times are all benefits of allograft use when compared to ICBG.8,48,86 Comparative improvements in post-operative functional outcomes may counter-balance the slightly inferior fusion outcomes that have been reported with allograft use. Further a wide spectrum of allograft preparations (bone chips, strips, powders, and interbody implants) allows for significant versatility in application. For instance, demineralized bone matrix (DBM) is an allograft formulation that consists of an osteoconductive scaffold of collagen and proteins that comes in various preparations that may facilitate its use during surgery. 55 Unique to DBM is that a varying degree of osteoinductive properties may persist due to remnant growth factors depending on how it is prepared.
Beta-tricalcium phosphate (B-TCP), hydroxyapatite (HA), and beta-calcium pyrophosphate are examples of ceramics that have been utilized in the setting of spinal fusion. Ceramics are highly biocompatible scaffolds that mimic the inorganic phase of bone with excellent osteoconductive properties. Ease of applicability, infinite supply, and synergism with other osteobiologics are significant advantages of ceramic use.87,88 A further substantial benefit is their innate customizability to optimize mechanical and fusion properties (pore size, modulus of elasticity, etc.). 1
Bone morphogenetic proteins (BMPs) are glycoproteins that belong to the transforming growth factor-β (TGF-β) class of proteins. While their functions are numerous, from a fusion standpoint, they are integral to the signaling cascades of bone growth and remodeling. rhBMP-2 has been shown to be an effective alternative to autologous bone grafts in achieving spinal fusion59,89 although it’s FDA approved applications are limited. Finally, BMP-2 has been associated with severe side-effects possibly leading to life threatening dysphagia when used in the setting of ACDF.59,90 This has also led the FDA to put out an advisory on the use of this osteobiologic in ACDF, which is also an off-label application. 91
Recently, use of stem cells spinal fusion has garnered significant interest. Stem cells are believed to exhibit significant osteogenic potential similar to ICBG although variability of potency is of notable concern.92,93 In general, autologous stem cells are most commonly retrieved from the iliac crest or vertebral bodies. The aspirate is then typically concentrated via centrifugation to isolate mesenchymal stem cells (MSCs) which are then utilized for fusion purposes oftentimes in conjunction with an osteoconductive carrier. To potentially circumvent potency-related variability, the development of allogeneic stem cell products has garnered significant interest, of late. 81 These products are typically harvested from cadaveric donors and have been shown to exhibit all 3 optimal graft properties.
Fusion Rates
Published fusion rates show significantly variability even with use of the same biologic. For instance, in the setting of allograft, fusion rates ranged from 31.8% to 100% at final follow-up. Perhaps the one exception to this may be rh-BMP-2 which demonstrated over 7 studies a fusion rate of 95-100% at final follow-up. That being said, the potential risk of morbidity remains a significant concern. 59 Ultimately, much of the disparity in the remaining data is likely the result of heterogeneity in study design, surgical technique (including implant selection), and method of fusion assessment amongst other factors. Ultimately, while it appears that acceptable alternatives to structural autograft exist, to date, none have been demonstrated to be superior.
Timing of Fusion
Data regarding time to fusion is limited in the current literature. Existing data suggests that fusion can occur as early at 3.5 months 29 with use of autograft in single level disease. However, the data is variable, with another study showing that it may take up to 1 year or longer to achieve fusion. 44 Furthermore, while it appears that, for instance, autograft may yield faster time to fusion when compared to allograft,40,44 limitations in study design and quality of data reporting prevent any definitive conclusions from being drawn.
Radiological Assessment
Across included studies, there was significant heterogeneity in the method of assessment of radiographic fusion. Modalities utilized in the included studies to evaluate for radiographic fusion include static radiographs, dynamic radiographs, computed tomography, or some combination thereof. Furthermore, qualitative assessments of fusion employed were not homogenous between studies, as a variety of assessment criteria were utilized to define fusion in the reviewed studies. Ultimately, we were unable to determine if osteobiologic type affects the optimal modality of fusion assessment.
From a strictly radiologic standpoint, each modality has inherent limitations. While plain radiography is readily accessible, the ability to discern bridging trabecular bone across a disc space is imperfect and may lead to over-, or underestimation of radiographic fusion rates. Computed tomography is considered the “gold standard” for assessing fusion from a radiologic standpoint; however, a large number of studies did not routinely utilize CT to assess fusion. This was likely due to issues related to unnecessary radiation exposure, and issues related to convenience, costs, and resources. On this note, however, Riew et al recently demonstrated similar accuracy between dynamic radiographs and CT scan when utilizing strict measurement criteria, 94 but these criteria have not yet been widely adopted in the literature.
Study Limitations
There are significant limitations to this systematic review. Lack of stratification in regards to the number of levels fused and lack of standardized surgical techniques, use of numerous adjunctive biologics in conjunction with studied biologics, are significant confounding factors that critically limit the quality of data to date. Furthermore, despite many studies defining specific lengths of final follow-up, in many instances there was lack of clarity regarding the specific time point at which fusion was actually determined. This is particularly true of studies that used multiple imaging modalities to determine fusion status. We could only assume if it was not specifically defined, then the stated rates reflect assessment of fusion at final follow-up. However, we cannot state this with certainty. Subsequently, effective comparisons between homogenous groups at equivalent timepoints were not possible, as exclusion of poorer quality of data would have further limited the number of data points for comparison. Consequently, we view this systematic review as a very important eye-opening depiction of the unfortunate current state of our biologics literature as it pertains to ACDF. It underscores the critical importance of collective improvements in future study methodologies moving forward when attempting to determine osteobiologic efficacy.
Conclusions
Achieving fusion following ACDF appears to remain an intricate interplay between host biology and a variety of surgical factors with biologic selection existing as just one part of this complex puzzle. Structural autograft has long remained the gold standard for fusion optimization. In this context, while alternative biologics exist that may produce acceptable fusion rates, significant limitations in study methodology prevent any definitive conclusions from being drawn from existing literature, specifically, as it relates to identifying the optimal biologic to promote fusion, the optimal timing of fusion assessment, and understanding whether osteobiologic type influences the optimal assessment strategy for fusion. Increased homogeneity in study design between scientific studies and an overall higher level of evidence is an absolute must in order to best answer these latter questions. Future studies should keep these limitations close in mind when developing their respective study protocols.
Supplemental Material
Supplemental Material - Comparison of Different Osteobiologics in Terms of Imaging Modalities and Time Frames for Fusion Assessment in Anterior Cervical Discectomy and Fusion: A Systematic Review
Supplemental Material for Comparison of Different Osteobiologics in Terms of Imaging Modalities and Time Frames for Fusion Assessment in Anterior Cervical Discectomy and Fusion: A Systematic Review by Andrew S. Chung, Robert Ravinsky, Ronit Kulkarni, Patrick C. Hsieh, Jacobus J Arts, Ricardo Rodrigues-Pinto, Jeffrey C. Wang, Hans Joerg Meisel, Zorica Buser, AO Spine Knowledge Forum Degenerative in Global Spine Journal
Footnotes
Author Contributions
ASC: Study design, data analysis and interpretation, manuscript preparation, manuscript revision, PH: Study design, data analysis and interpretation, manuscript preparation, manuscript revision, RK: Data analysis and interpretation, manuscript preparation, manuscript revision, RR: Study design, data analysis and interpretation, manuscript preparation, manuscript revision, CA: Study design, data analysis and interpretation, manuscript revision, RRP: Study design, data analysis and interpretation, manuscript revision, JCW: Study design, data analysis and interpretation, manuscript preparation, manuscript revision, HJM: Study design, data analysis and interpretation, manuscript revision, ZB: Study design, data analysis and interpretation, manuscript preparation, manuscript revision
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This supplement was organized and financially supported by AO Spine through the AO Spine Knowledge Forum Degenerative, a focused group of international spine experts.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
