Abstract
A new formulation of zolmitriptan has been developed that dissolves on the tongue without the need for additional fluid intake. In this double-blind, parallel study, 471 patients were randomized to receive the zolmitriptan orally disintegrating tablet 2.5 mg(n=231) or matching placebo (n=240) to treat a single moderate or severe migraine. Headache relief following zolmitriptan 2.5 mg (63%) was significantly greater than with placebo (22%) at 2 h post-dose (primary endpoint; P < 0.0001). The zolmitriptan orally disintegrating tablet was also significantly more effective than placebo for 1-, 2- and 4-h pain-free response (8% vs. 3%, P =0.0207, 27% vs. 7%, P < 0.0001, and 37% vs. 11%, P < 0.0001, respectively). Of those patients stating a preference, 70% of patients preferred the orally disintegrating tablet to a conventional tablet. Zolmitriptan orally disintegrating tablets are an effective and convenient alternative to a conventional tablet, allowing migraine attacks to be treated anytime a migraine strikes, which can facilitate earlier treatment.
Introduction
Migraine is a common neurological condition that affects 5–17% of the general population and is about 3–4 times more common in women than in men (1, 2). Because of the chronic and debilitating nature of the disorder, migraine has a significant socioeconomic impact and adversely affects patients' quality of life (3–5). It is also an unpredictable disorder, with attacks that can occur at anytime, and patients therefore need a medication that enables them to treat their migraine headache anytime a migraine strikes. Importantly, recent clinical research shows that treating a migraine early, when the pain is mild, results in more effective response to therapy (6), an effect also observed previously in a long-term study of zolmitriptan (7). Consequently, medications are needed that patients can take (even in the absence of water) as soon as it is evident that the headache is a migraine.
Zolmitriptan is a selective 5-HT1B/1D receptor agonist that appears to have the ability to inhibit trigeminovascular activation both peripherally and centrally (8–10). The conventional oral zolmitriptan tablet is effective and well tolerated in the acute treatment of migraine (11–13), with an onset of effect reported as early as 45 min after dosing (14, 15).
A unique orally disintegrating tablet of zolmitriptan that dissolves on the tongue without additional fluid intake has been developed. This formulation offers a convenient alternative for patients who prefer not to take tablets. Furthermore, because fluid intake is not required, it may allow patients to take their medication earlier in the course of a migraine attack when water or other liquids may not be available.
Pre-clinical studies showed that the pharmacokinetic profile of the zolmitriptan orally disintegrating tablet is similar to that of the conventional tablet (16). The AUC and Cmax of zolmitriptan are similar following administration of conventional zolmitriptan tablets and the orally disintegrating formulation. Similar to the Tmax delay seen with other orally disintegrating tablets (17), Tmax of the parent compound was observed somewhat later with the zolmitriptan orally disintegrating tablet compared with the conventional tablet. Importantly, the AUC, Cmax and Tmax for the active N-desmethyl metabolite of zolmitriptan are similar for the two formulations, suggesting that they will exhibit a similar efficacy profile.
To study the clinical efficacy and tolerability of the zolmitriptan orally disintegrating tablet, adult migraine patients were recruited into a double-blind, randomized, multicentre trial that compared zolmitriptan 2.5 mg orally disintegrating tablet with placebo for the acute treatment of a single moderate to severe migraine attack.
Methods
Patients
Female and male patients aged 18–65 years with an established diagnosis of migraine (as defined by International Headache Society criteria) (18) were recruited. All patients were required to have an age at migraine onset of < 50 years, to have experienced ≥1 migraine headache per month during the 3 months prior to the study, and to be able to distinguish between migraine and non-migraine headaches.
Patients were excluded if they had a history of basilar, ophthalmoplegic or hemiplegic migraine, or if they reported non-migraine headaches on ≥ 6 days per month in the preceding 6 months. Other exclusion criteria were: pregnancy, lactation or inadequate contraception in female patients; history or symptoms of ischaemic heart disease (or other vascular disease, including Prinzmetal's angina), dysrhythmias or cardiac accessory pathway disorders (e.g. Wolff-Parkinson–White syndrome); uncontrolled hypertension; use of monoamine oxidase A inhibitors, methysergide or methylergonovine within 2 weeks before randomization; a recent history of alcohol or drug abuse; known hypersensitivity or adverse reaction to study medication or other 5-HT1B/1D receptor agonists; phenylketonuria; severe hepatic impairment; or participation in another clinical trial during or within 30 days prior to this study.
This study was designed and conducted in compliance with the ethical principles of Good Clinical Practice and the Declaration of Helsinki, and with the approval of the local ethics committees. All patients provided written informed consent before study procedures were initiated.
Study design and treatments
This was a multicentre, randomized, double-blind, placebo-controlled study conducted in Canada, South Africa and the United Kingdom between March and October 1999. After a screening visit to determine eligibility for the study, 573 patients were randomized to receive either zolmitriptan 2.5 mg orally disintegrating tablets or orally disintegrating placebo tablets for the acute treatment of a single moderate or severe migraine headache. The placebo tablets were matched for taste, size and shape as compared with the active treatment but did not include zolmitriptan.
Patients were randomized in blocks of four in a 1 : 1 ratio. Details of the treatment codes were given to the investigators in sealed envelopes, which could be opened only if knowledge of the trial medication was essential to optimize patient management. Patients were instructed to treat only a migraine of moderate or severe intensity, to have had at least 24 h since a previous migraine, to take the medication within 4 h of migraine onset or within 4 h of waking with a migraine, and not to sleep within 4 h of dosing.
The trial medication was to be placed on the tongue and allowed to dissolve. If required, a second tablet of trial medication or escape medication (including non-steroidal anti-inflammatory drugs, analgesics, sedatives or antiemetics, approved for individual patients in consultation with the investigator before randomization) could be taken from 2 h after the first dose.
Before administration of study medication, use of other acute migraine treatments was restricted: triptans or ergot derivatives were not permitted within the previous 24 h; opiates and analgesics were not permitted within the previous 12 and 6 h, respectively.
Efficacy and tolerability assessment
The response to study medication was recorded on patient diary cards. The primary endpoint was headache relief at 2 h after treatment, defined as a reduction in migraine pain from moderate or severe at baseline to mild or none. Secondary outcomes were headache relief from 30 min to 4 h post-dose, the proportion of patients pain free from 1 h to 4 h post-dose and the proportion of patients with improvement in headache pain. This was defined as at least a 1-point reduction on the 4-point migraine headache rating scale (severe, moderate, mild or no pain) at 30 min and 1 h after the first dose. Patient preference for the orally disintegrating tablet as compared with a conventional tablet was also recorded. Adverse events occurring within 24 h of treatment were recorded on the patient diary cards and solicited by investigators at the follow-up visit.
Statistical analysis
Sample size calculations assumed a response rate at 2 h of 47% for placebo and 65% for zolmitriptan orally disintegrating tablet. It was calculated that 380 patients would be required to detect this 18% difference between treatment groups (α=0.05, power=0.90), allowing for an estimated withdrawal rate of 10%.
Statistical analysis was based on the intent-to-treat population of patients who treated at least one migraine headache of moderate or severe intensity and had at least one efficacy evaluation. Patients who took a second treatment 2 h after the initial dose (trial medication or rescue therapy) were considered non-responders for all subsequent assessments.
With the exception of patient treatment preference and adverse events data, all endpoints were analysed by a logistic regression model using PROC LOGISTIC in SAS™. Results were presented as odds ratios with 95% confidence intervals and P-values. All tests were considered statistically significant at P < 0.05 and all hypothesis tests were two-sided.
Results
Patient characteristics
A total of 573 patients were randomized to the study, of which 102 patients did not treat a migraine headache within the time period of the trial (Fig. 1). The safety population therefore comprized 471 patients who treated an attack with study medication. One patient in the placebo group withdrew consent after treating a migraine and had no efficacy assessments. This patient was excluded from the intent-to-treat population, which consisted of 470 patients: 231 in the zolmitriptan 2.5 mg group and 239 in the placebo group. Demographic and baseline migraine characteristics were similar in the two treatment groups (Table 1).
Demographics and baseline migraine characteristics of the ITT population


Flow of patients through the study.
Headache relief
Patients who received the zolmitriptan orally disintegrating tablet had significantly greater headache relief at 2 h compared with placebo recipients (138/220 (63%) vs. 53/236 (22%); P < 0.0001; odds ratio 6.1, 95% CI 4.0, 9.3). Headache relief with zolmitriptan was consistent across patient subgroups (e.g. age, gender, presence of menses, migraine onset while waking or sleeping, presence of aura and headache-associated symptoms such as phonophobia or nausea at baseline).
Zolmitriptan orally disintegrating tablets demonstrated an early onset of action in some patients. Compared with the placebo group (10%), a higher proportion of patients in the zolmitriptan group (16%) reported headache relief 30 min after the first dose, which approached statistical significance (P=0.0538; odds ratio 1.7, 95% CI 1.0, 3.1). Significantly more patients also reported headache relief at 1 h (45% vs. 19%, P < 0.0001; odds ratio 3.5, 95% CI 2.3, 7.3; Fig. 2) and 4 h (51% vs. 14%, P < 0.0001; odds ratio 6.3, 95% CI 4.0, 9.8) with zolmitriptan than with placebo.

Percentage of patients with headache relief 30 min, 1 h and 2 h after an initial orally disintegrating tablet of zolmitriptan 2.5 mg (▪) or placebo (□). The total number of patients within each treatment group with an assessment at that time-point is given. ∗∗∗P < 0.0001 vs. placebo.
Over the 24-h period following treatment, headache relief was maintained (headache relief at 2 h with no recurrence for 24 h) in 40% of patients who received a single dose of the zolmitriptan orally disintegrating tablet compared with only 12% of orally disintegrating placebo tablet recipients.
Absence or reduction of pain
Pain-free status was achieved by a significantly greater proportion of patients in the zolmitriptan group than in the placebo group at 1 h (8% vs. 3%; P=0.0207; odds ratio 3.1, 95% CI 1.2, 7.9), 2 h (27% vs. 7%; P < 0.0001, odds ratio 4.7, 95% CI 2.6, 8.4) and 4 h (37% vs. 11%; P < 0.0001; odds ratio 4.9, 95% CI 3.0, 8.0) post-dose (Fig. 3).
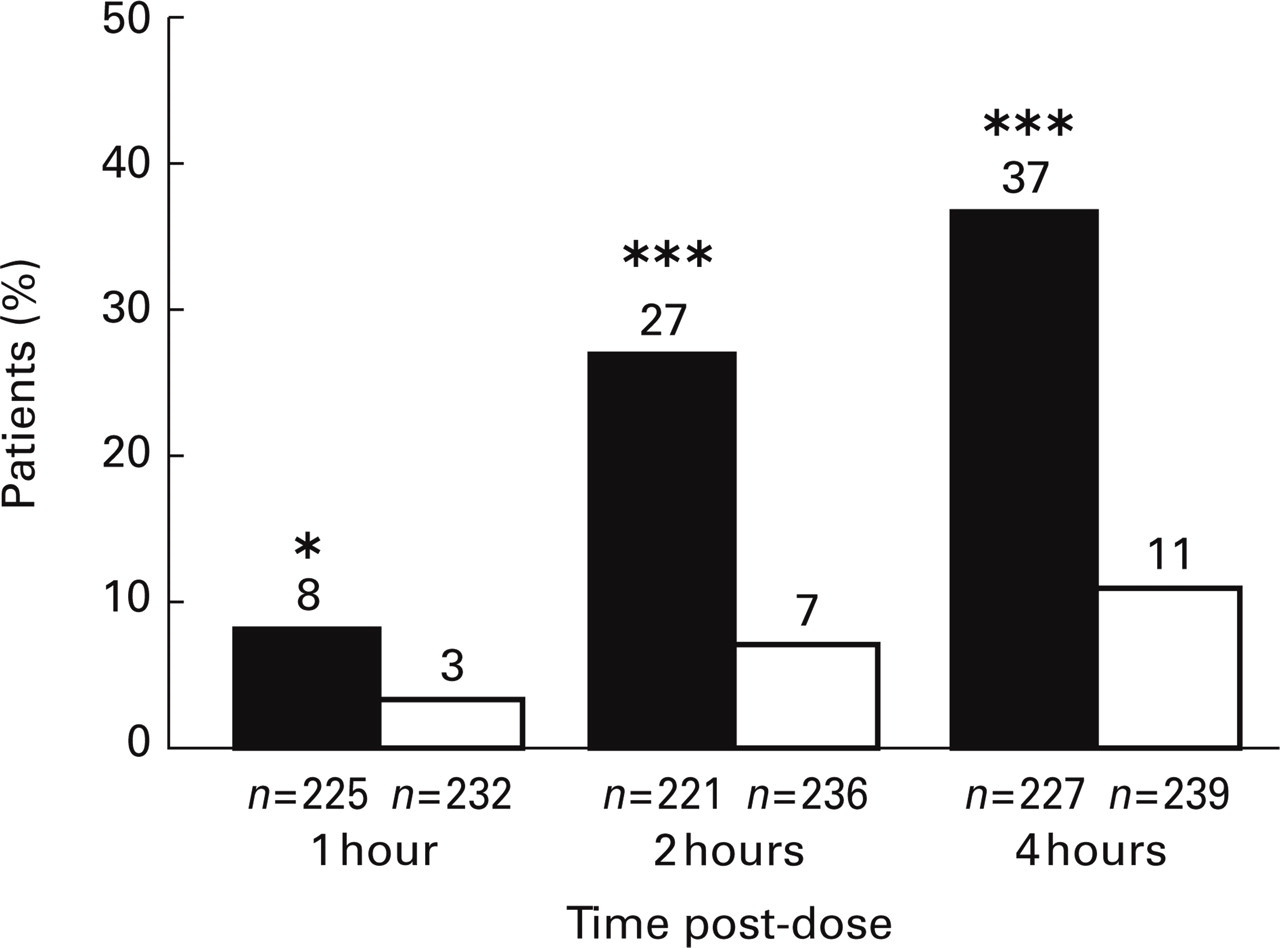
Percentage of patients pain free at 1, 2 and 4 h after an initial orally disintegrating tablet of zolmitriptan 2.5 mg (▪) or placebo (□). The total number of patients within each treatment group with an assessment at that time-point is given. ∗P = 0.0207; ∗∗∗P < 0.0001 vs. placebo.
Zolmitriptan orally disintegrating tablets began to provide headache relief within 30 min as measured by at least a one-point improvement in headache pain intensity compared with placebo (22% vs. 15%; P=0.0385; odds ratio 1.7, 95% CI 1.0, 2.7). By 1 h post-dose, 51% and 29% of zolmitriptan and placebo recipients, respectively, reported improvement in headache pain (P=0.0001; odds ratio 2.7, 95% CI 1.8, 3.9). Similar differences between treatments were seen at 2 and 4 h post-dose but these data were not analysed for statistical differences between groups.
Migraine-associated symptoms
Photophobia, phonophobia and/or nausea were associated with the majority of headaches before study treatment. Improvement in these migraine-associated symptoms was greater in the zolmitriptan recipients compared with the placebo group at 1 and 2 h post-dose (Table 2).
Proportion of patients free from photophobia, phonophobia or nausea at 1 and 2 h following treatment with zolmitriptan 2.5 mg or placebo orally disintegrating tablets in those patients with symptoms at baseline

∗ n, number of patients with improvement; N, total number of patients with data available at that time-point.
Persistence and recurrence: use of rescue medication
Patients were allowed to take a second dose or rescue medication for treatment of either persistent or recurrent headache of any severity (i.e. mild, moderate or severe). The results show that the median time to use of rescue medication or a second dose of study medication was 2 h and 10 min for patients in the placebo-treatment group and 5 h and 45 min in the zolmitriptan orally disintegrating tablet group. The overall number of patients taking a second dose or escape medication was 56.7% for the zolmitriptan group, compared with 84.9% for the placebo group.
Patient preference
Four hundred and sixty-nine patients provided treatment preference data, of whom 70% preferred to take the orally disintegrating tablet formulation compared with a conventional tablet. Similarly, 78% of patients stated that the orally disintegrating tablet was convenient and 70% said the orally disintegrating tablets could be taken earlier in the course of a migraine headache than a normal tablet (Fig. 4). The orally disintegrating tablet was considered easy to handle by 92% of patients, while 80% liked the orange taste of the tablet. The presence of nausea at baseline had no effect on the proportion of patients preferring the zolmitriptan orally disintegrating tablet over the conventional tablet (70% vs. 69% for patients with and without nausea at baseline, respectively).

Patient ratings of treatment preference and acceptability of the orally disintegrating formulation.
Tolerability
Zolmitriptan orally disintegrating tablets were well tolerated. Adverse events were reported by 63 (27%) patients in the zolmitriptan group and by 29 (12%) patients receiving placebo; the most frequently occurring events are presented in Table 3. The profile of adverse events was consistent with those reported with this drug class (19), including asthenia, throat tightness, somnolence and dizziness. No patients were withdrawn because of adverse events and no serious drug-related events were reported.
Incidence of adverse events reported by > 2% of patients in either treatment group (safety population)

Discussion
This multicentre, placebo-controlled study shows that the zolmitriptan 2.5 mg orally disintegrating tablet is effective and well tolerated for the acute treatment of migraine. The 2-h headache relief rate observed in the present study (63%) is consistent with the response rates previously reported for the conventional zolmitriptan 2.5 mg tablet, which range from 59% to 67% (11–14). Significant improvement over placebo was also observed at 1 h and 4 h for headache relief, which is consistent with previous reports of the onset of efficacy observed with the conventional 2.5 mg tablet (14, 15).
A statistically significant drop by at least one point in headache pain severity was reported at 30 min post-dose, suggesting early reduction in pain intensity following treatment. This improvement in the reduction in pain intensity was also evident 1, 2 and 4 h following treatment.
Although collected for descriptive purposes only, the orally disintegrating tablet was preferred by patients over their previous conventional tablet formulations. Other favourable characteristics included convenience and ease of use.
The value of a medication that is preferred by patients and easy to take without water is that it may allow some patients to treat their attacks earlier. Recent studies suggest that the response to therapy increases if patients treat migraine early following onset (6). The convenience of an orally disintegrating tablet may facilitate the likelihood of early intervention, and studies need to be done to further understand treatment behaviour of patients with migraine to see if this is likely to occur.
Another important consideration is that patients stratified to appropriate care are less likely to lapse from care if their medication is clinically effective from the onset (20). Some traditional care strategies delay a patient's chance of receiving migraine-specific medications, thereby delaying the time until an effective treatment is given (21).
As there is no universal approach to the pharmacotherapy of migraine, the choice of migraine-specific treatment should be individualized based on the patient's needs, migraine characteristics and patient preference. By increasing the available treatment options and delivery formulations it is possible to better match the patient's needs and preferences to a specific migraine therapy. This study shows that the zolmitriptan orally disintegrating tablet is an effective and well-tolerated acute treatment for migraine. The orally disintegrating tablet offers a convenient alternative for patients who cannot swallow or prefer not to swallow conventional tablets, for patients who want the choice to take their medication discreetly or for patients with nausea (which may be aggravated by the intake of fluid). Furthermore, the zolmitriptan orally disintegrating tablet has the advantage that it may be taken immediately, without the need for additional fluids, anytime a migraine headache strikes.
Footnotes
Acknowledgements
This study was supported by AstraZeneca Pharmaceuticals. The authors gratefully acknowledge Adis International Inc. and Starr Pearlman PhD for their technical support in preparing this manuscript.
