Abstract
Calcitonin gene-related peptide (CGRP) has been detected in increased amounts in external jugular venous blood during migraine attacks. However, it is unknown whether this is secondary to migraine or whether CGRP may cause headache. In a double-blind crossover study, the effect of human αCGRP (2 μg/min) or placebo infused intravenously for 20 min was studied in 12 patients suffering from migraine without aura. Headache intensity was scored on a scale from 0 to 10. Two patients were excluded due to severe hypotension and one because she had an infection. In the first hour median peak headache score was 1.0 in the hαCGRP group vs. 0 in the placebo group (P < 0.01). During the following 11 h all patients experienced headaches after hαCGRP vs. one patient after placebo (P = 0.0004). The median maximal headache score was 4 after CGRP and 0 after placebo (P = 0.006). In three patients after hαCGRP, but in no patients after placebo, the delayed headache fulfilled the IHS criteria for migraine without aura. As intravenous administration of hαCGRP causes headache and migraine in migraineurs, our study suggests that the increase in CGRP observed during spontaneous migraine attacks may play a causative role.
Introduction
Calcitonin gene-related peptide (CGRP) immunoreactive fibres originating in the trigeminal ganglion innervate cranial cerebral blood vessels (1). In animals, stimulation of these sensory nerve fibres has been shown to cause antidromic release of CGRP with subsequent vasodilatation of the cerebral vasculature (2). CGRP is also one of the mediators of neurogenic inflammation. This phenomenon has been implicated in the pathogenesis of migraine headache in an impressive range of previous studies (3). CGRP levels are increased in the blood from the external jugular vein but not in cubital venous blood during migraine attacks (4). The 5HT1B/D receptor agonist, sumatriptan, which aborts migraine attacks, normalizes CGRP levels (5). Whether this cephalic CGRP release plays a role in the pathophysiology of migraine or only represents a consequence of the migraine attack is unknown. In previous studies where CGRP was infused into non-migraineurs, a feeling of fullness in the head but no headache was reported (6, 7). To elucidate the role of CGRP in migraine we therefore decided to study whether intravenous CGRP infusion in migraine sufferers causes more headache or migraine than placebo using a double blind, crossover design.
Material
Twelve volunteers (11 females, 1 male; mean age 39.5 years, range 31–47 years; mean weight 69.7 kg, range 51–89 kg), all suffering from episodic migrainep without aura according to the classification of the International Headache Society (8), but otherwise healthy, were included. The patients had between six migraine attacks per month and six migraine attacks per year (extremes included). Patients had less than 5 days of tension-type headache per month.
The subjects were told not to have headache on arrival and they were told not to take medication, or consume coffee, tea, alcohol or tobacco for 12 h before the study. They were not allowed to take a triptan for 24 h or ergotamine for 48 h before the study. Exclusion criteria were: use of any kind of daily medication, including prophylactic headache therapy but excluding oral contraceptives; pregnancy or breast-feeding; excessive use of analgesics or alcohol; serious somatic or psychiatric disorders; ischaemic heart disease; or a supine systemic blood pressure above 160/90 or below 110/75 mmHg at entry into the study.
Patients were informed that they were free to withdraw at any time and all patients signed written informed consent. The study was approved by the local ethical committees of Copenhagen and of Copenhagen County and complied with the Declaration of Helsinki.
Design, procedure and methods
In a double-blind crossover study 12 patients were randomized to receive human α CGRP (hαCGRP) 2 µg/min or placebo (0.9% NaCl) intravenously (i.v.) for 20 min. The infusions were given on two different study days at least 1 week apart. Patients were randomly assigned by computer (Med.Stat®, version 2.12). Randomization and preparation of study drugs were done by medical staff not involved in the study. Five patients started with placebo and seven with CGRP. This quota was chosen by the computer and not known before the study.
When subjects arrived at the laboratory a cannula (Viggo Venflon® 1.4 mm) was placed in the right cubital vein for hαCGRP or placebo infusion. Baseline values of headache intensity and characteristics, blood pressure and heart rate were recorded after 30 min of rest in the supine position. Then hαCGRP or placebo were infused intravenously for 20 min by a volume-directed pump (Braun Perfuser®).
Headache intensity and characteristics were recorded before, every 10 min during, and after hαCGRP/placebo infusion until 80 min after start of the infusion. After discharge the patients recorded headache intensity and characteristics and medication use once every hour for the following 11 h. Patients were given a sumatriptan 100-mg tablet to be taken if needed.
Headache intensity was scored on a verbal rating scale from 0 to 10; 0 = no headache, 1 = pre-pain (a feeling of something unusual in the head, but not headache), 5 = a headache of medium severity, 10 = the worst possible headache. Other headache characteristics necessary for precise classification of the induced headache, according to the criteria of the International Headache Society (IHS), were also recorded. At every recording it was evaluated whether the headache fulfilled the IHS criteria for migraine. Blood pressure and heart rate were measured with an automatic inflatable arm cuff (Omega 1400, Invivo Research Laboratory, New York, USA) every 5 min.
The mean maximal blood velocity in the middle cerebral artery (MCA) on the usual headache side was measured with transcranial Doppler (DWL, Belinea, Germany) every 10 min throughout the study. The regional cerebral blood flow (rCBF) was measured with Xe-133 inhalation and a brain dedicated SPECT-system, before, during the last 5 min of infusion and during the last 5 min of the in-hospital study period (55–60 min after end of infusion). The Doppler measurement was only performed on the usual headache side due to the very small space around the head in the SPECT scanner. The end-tidal pCO2 was measured simultaneously with the TCD and CBF measurements throughout the study period. The results from the haemodynamic part of the study will be published elsewhere.
Statistics
Headache scores are given as median and range. Headache was calculated as immediate, i.e. occurring during infusion or in the 40 min thereafter, or as delayed. The latter occurred 60 min to 12 h after the beginning of the infusion. The reason for this division is that headache after glyceryl trinitrate and histamine is clearly biphasic (9, 10) and a similar pattern was expected to occur after hαCGRP. The difference between the number of hαCGRP-treated patients and placebo-treated patients who experienced immediate headache and delayed headache was tested with a Fishers exact test (Med.Stat®, version 2.12).
All statistical calculations below were done by using the Statgraphics® 7.0 software package. Differences in maximal headache score and total headache experienced (the sum headache score) in the first 60 min and 60 min to 12 h after the beginning of the hαCGRP/placebo infusion were analysed with Wilcoxon's rank test for paired data. Friedman's test was used to test for time differences in headache intensity within each group.
Hemodynamic responses were calculated in percentage of baseline, and are given as mean and standard error of the mean (± SEM).
Differences in blood pressure and heart rate over time within the group were analysed with analysis of variance (
Differences in peak value of the blood pressure and heart rate between hαCGRP and placebo treatment were tested with a paired t-test. In all tests P < 0.05 was considered statistically significant.
Results
Two patients were excluded from the calculations below due to symptoms of severe hypotension causing the infusion to be terminated ahead of time. Signs and symptoms were pallor, cold sweat, stomach ache, nausea and palpitations. The situation was restored after placing the patients in a head-down tilt after 7 and 20 minutes, respectively. The induced hypotension demonstrates that the hαCGRP dose used is the maximally tolerated dose. Furthermore, one patient was excluded because she had an infection (with high fever and headache) after discharge from the clinic (placebo treatment). All patients were excluded before the code was broken.
Immediate headache
Immediate headache was defined as headache occurring during the hαCGRP/placebo infusion or 40 min thereafter.
Eight out of nine patients treated with hαCGRP scored headache more than 0, in four of these patients the headache score was 1. None of the headaches fulfilled the IHS criteria for migraine without aura. The median duration of the immediate headache was 30 min. The patient without immediate headache later developed a migraine headache. In comparison only one of the nine patients treated with placebo experienced headache score 1 during this period (P = 0.0034, Fishers exact test) (Figs. 1 and 2).
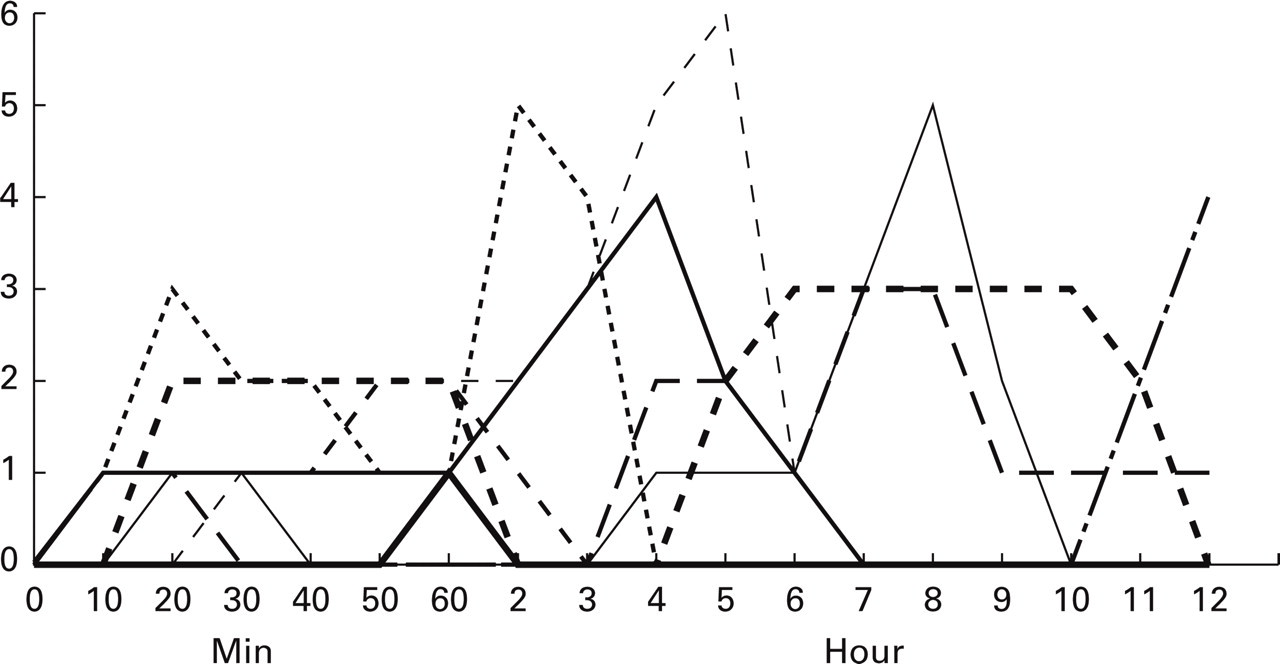
The individual headache scores after CGRP in the first 60 min after start of the infusion (immediate headache) and in the subsequent 11 h (delayed headache).
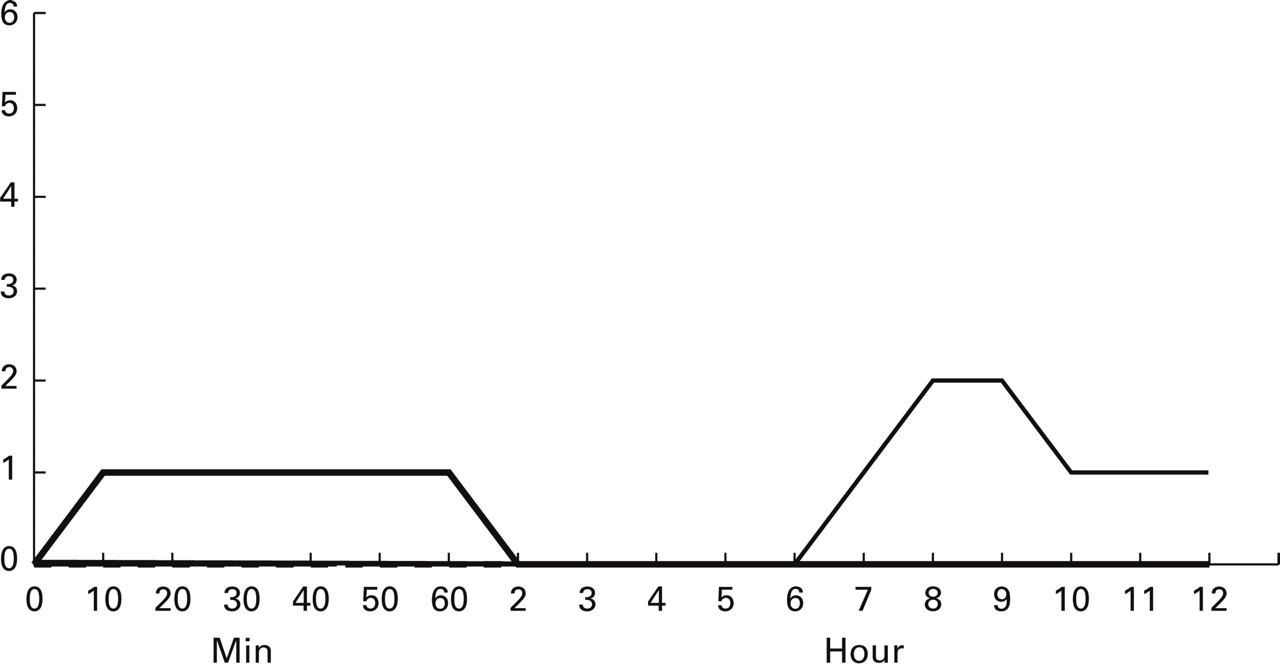
The individual headache scores after placebo in the first 60 min after start of the infusion (immediate headache) and in the subsequent 11 h (delayed headache).
The median maximal headache score was 1 (range 0–3) after hαCGRP infusion and 0 (range 0–1) after placebo (P = 0.01, Wilcoxon) (Fig. 1). The sum of headache scores in this period was 45 after hαCGRP and 6 after placebo (P = 0.01, Wilcoxon).
Delayed headache
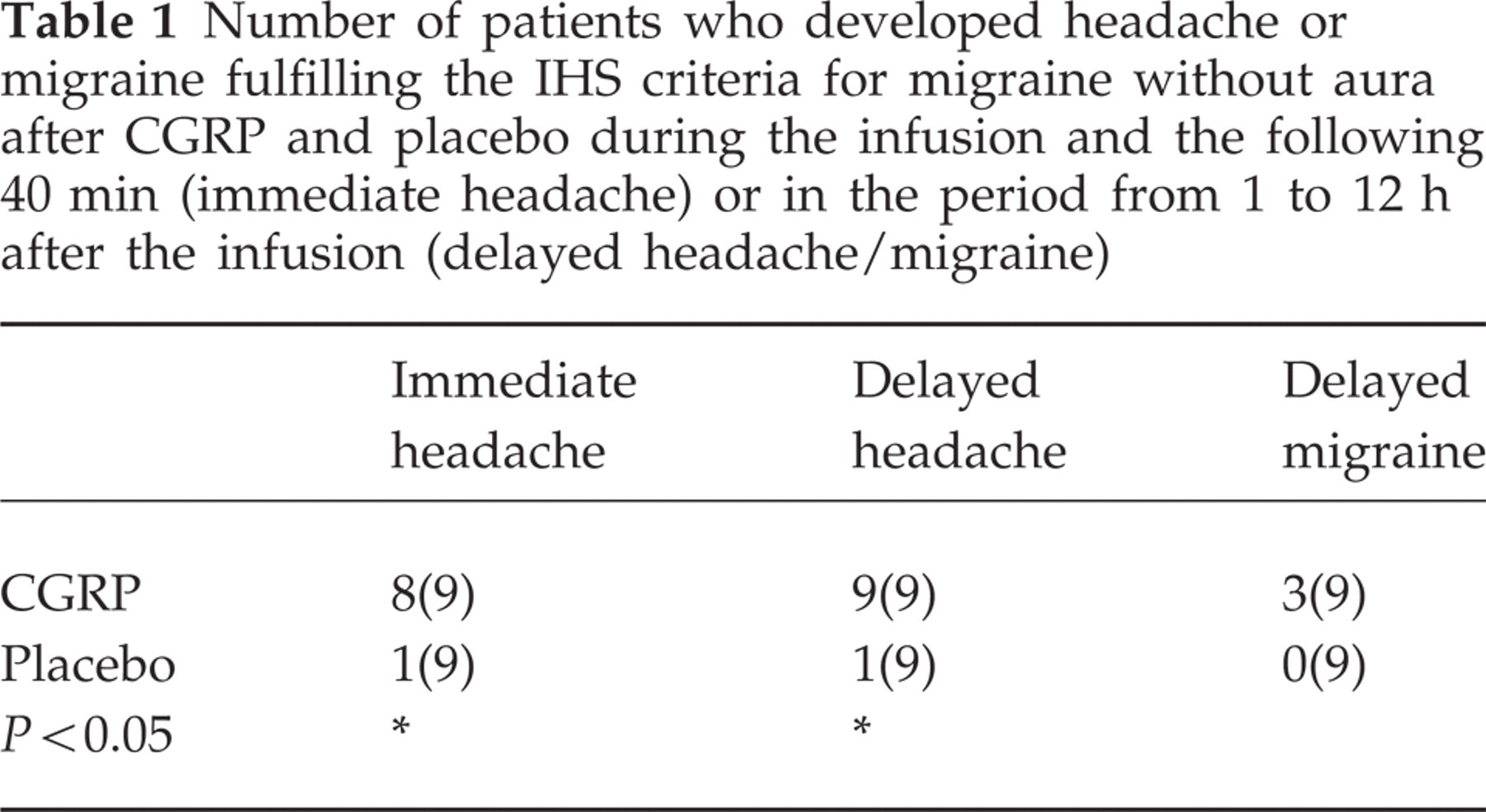
Delayed headache was defined as headache in the period from 1 to 12 h after the beginning of hαCGRP/placebo infusion. All nine patients treated with hαCGRP continued to have a headache or developed a delayed headache vs. one of the placebo-treated patients (P = 0.0004, Fishers exact test). In three of the hαCGRP-treated patients the delayed headache fulfilled the IHS criteria for migraine without aura but not in any patients after placebo (P = 0.21, Fishers exact test) (Table 1).
Number of patients who developed headache or migraine fulfilling the IHS criteria for migraine without aura after CGRP and placebo during the infusion and the following 40 min (immediate headache) or in the period from 1 to 12 h after the infusion (delayed headache/migraine)
One of these patients did not experience an immediate headache. The second patient scored maximum 3 and later 1 during the immediate headache phase. Two hours after infusion it became a migraine headache with maximum score 5. The third patient scored maximum 2 during the immediate headache phase and the headache continued to increase to maximum score 6, 5 hours after the beginning of the infusion.
The median maximal headache score after hαCGRP was 4 (range 1–6). It occurred at a median of 5 h (range 1–12 h) after the beginning of the infusion. The median maximal headache score after placebo was 0 (range 0–2) (P = 0.006, Wilcoxon) (Figs. 1 and 2). The total headache experienced (sum headache score) in this time period was 100 in hαCGRP-treated patients and 8 in placebo-treated patients (P < 0.01, Wilcoxon). For further information about headache characteristics and accompanying symptoms see Table 2.
(a) Clinical characteristics of patients’ usual migraine. (b) Clinical characteristics of maximal headache score from 1 to 12 h after start of the CGRP infusion
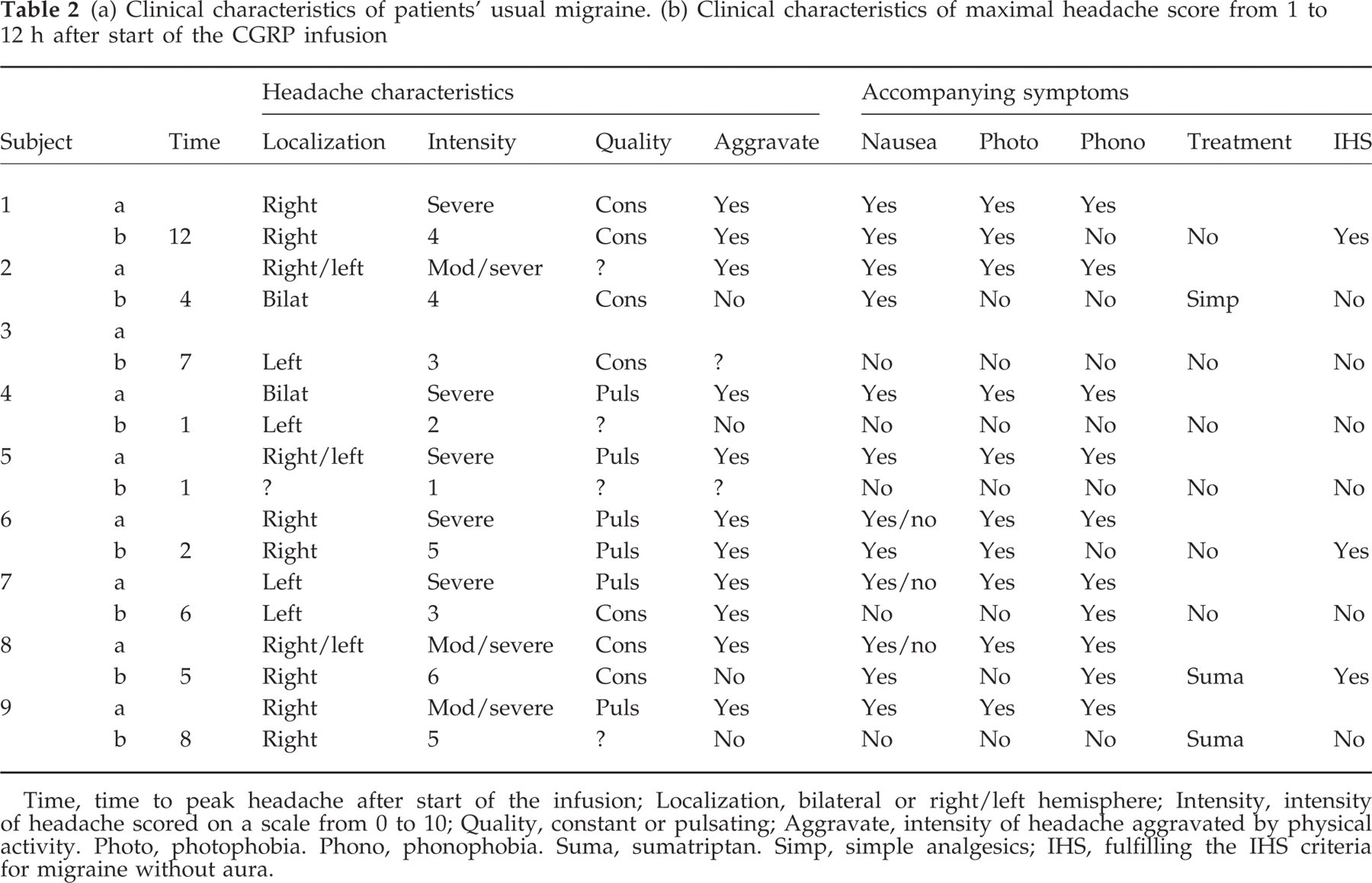
Time, time to peak headache after start of the infusion; Localization, bilateral or right/left hemisphere; Intensity, intensity of headache scored on a scale from 0 to 10; Quality, constant or pulsating; Aggravate, intensity of headache aggravated by physical activity. Photo, photophobia. Phono, phonophobia. Suma, sumatriptan. Simp, simple analgesics; IHS, fulfilling the IHS criteria for migraine without aura.
Blood pressure
The mean arterial blood pressure (MABP) decreased significantly over time after hσCGRP (P < 0.0001), but not after placebo (P = 0.66,
Blood pressure (mmHg) and heart rate (bpm) before, and 15, 20 and 80 min after start of the CGRP/placebo infusion
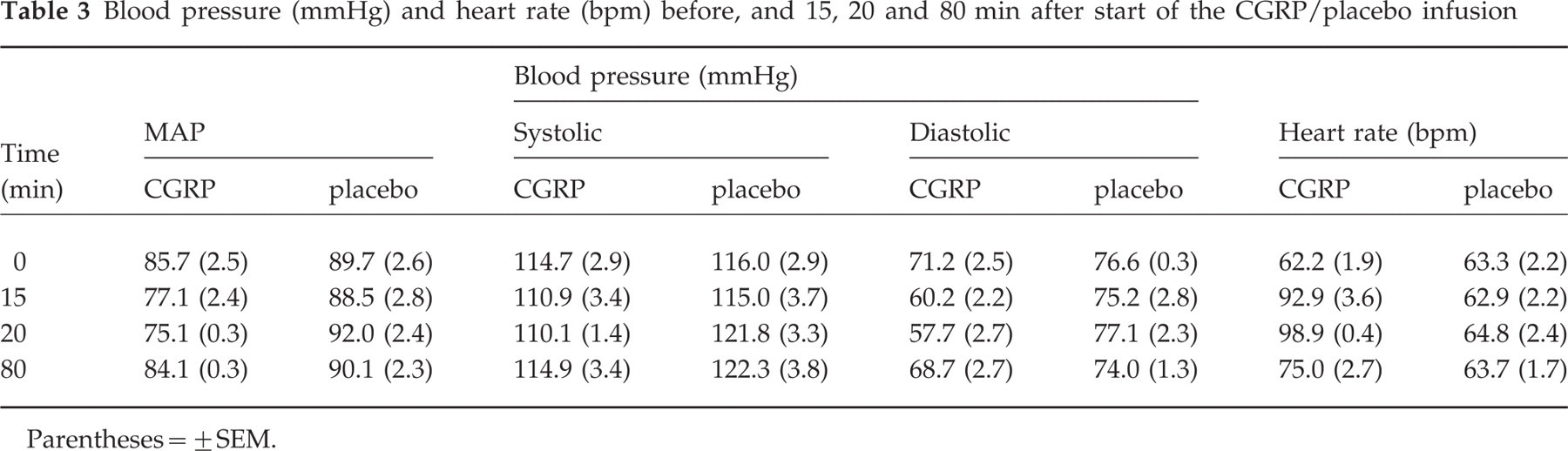
Parentheses = ± SEM.
Heart rate
When patients were treated with hαCGRP, their heart rate changed significantly over time (P < 0.0001,
Adverse experiences
Flushing after hαCGRP was pronounced and appeared only in the face, neck and upper chest. Flushing and a feeling of warmth appeared 10 min after the beginning of hαCGRP in all patients (time of the first observation) and disappeared at a median of 70 min after the beginning of infusion (range 20–80 min). There was no flushing when patients were treated with placebo.
Discussion
Calcitonin gene related peptide (CGRP) seems to be a very interesting molecule in the pathogenesis of migraine. It is the most abundant peptide transmitter in perivascular sensory trigeminal nerve fibres, where it is colocalized with substance P and other neurotransmitters (1). These transmitters are liberated into the perivascular space and from there may diffuse into venous blood, where they are found in increased concentration in situations of trigeminal activation. Liberation of sensory neurotransmitters is normally due to peripheral noxious stimulation but is also observed in the somewhat artificial situation of electrically induced retrograde activity in sensory nerve fibres (5, 11, 12). The latter phenomenon has been used in the neurogenic inflammation model of migraine, which has led to substantial progress in the understanding of the trigeminal vascular system (3). Finally, and most importantly, it has been demonstrated that the level of CGRP in blood from the external jugular vein, which drains extracranial tissue including the dura and the Gasserian ganglion, is increased during spontaneous migraine attacks and during electrical stimulation of the trigeminal ganglion in humans during neurosurgical operations (2, 4). It has been difficult, however, to evaluate the importance of CGRP excretion as measurements of CGRP during acute migraine attacks are usually not possible until several hours into the attacks.
If CGRP were liberated before the onset or at a very early stage of an attack, it would suggest a causative role. However, the increase could equally well occur at a later stage of the attack and might then merely reflect pain activation. The fact that specific migraine medication (sumatriptan) seems to eliminate the increase in CGRP (5) while treatment with the morphinomimetics seems not to affect CGRP (13) suggests that the CGRP increase may be more than just an epi-phenomenon. However, CGRP is not algogenic when injected into the temporal muscle (14), which seems to make a direct nociceptive action of CGRP less likely also in migraine.
We chose a direct approach in order to evaluate the importance of CGRP in migraine. If CGRP plays a causative role, it should be possible to induce a vascular headache by an infusion of CGRP. In the present study, we demonstrated the significant role of CGRP and could compare the response to cerebral heamodynamic parameters. The latter results are to be published elsewhere, but briefly the results were no change in the regional cerebral blood flow during or after CGRP infusion, but a decrease in mean maximal blood velocity in the middle cerebral artery (Vmean) of 13.5%. As tissue blood flow was unchanged, velocity changes were inversely proportional to the cross-sectional area of the artery. CGRP thus dilated the middle cerebral artery. The haemodynamic changes showed a maximum at the end of the infusion and returned to normal within 1 h after the termination of the infusion. For reasons of feasibility no haemodynamic measurements were made at later stages when the patients developed delayed headache.
CGRP causes vascular headache and migraine
The outcome of the present study is very clear. CGRP caused headache in virtually all migraine sufferers, whereas placebo did not. The headache occurred during the infusion and disappeared or diminished after the infusion. However, after a median of 5 h, a stronger headache developed in all nine subjects. This headache, as shown in Table 2, had most of the characteristics of migraine and in three of these subjects it fulfilled diagnostic criteria of the International Headache Society for migraine without aura (8). Thus, CGRP is not only able to cause immediate headache but it also induces migraine-like disorders or migraine without aura in migraine sufferers. The time profile of the induced headache is virtually identical to the time profile previously observed with glyceryl trinitrate and histamine. These substances also induced an immediate headache, a decrease in headache and then, after one to several hours (median time of 5 h and 5.5 h, respectively), a delayed headache which often fulfilled diagnostic criteria for migraine without aura (9, 10).
During CGRP infusion we found a median peak headache of 1 (out of 10). Thus, despite the fact that CGRP was given in the maximal tolerated dose, the immediate headache induced by CGRP was milder than the immediate headache induced by GTN and histamine (9, 10). Correspondingly, the dilatation of the middle cerebral artery was of smaller magnitude (9, unpublished observation). CGRP caused a much stronger decrease in blood pressure than the two other substances, which may have reduced the immediate headache. It has previously been shown that histamine-induced headache is much milder if blood pressure is allowed to fall (15).
The delayed headache after CGRP was perhaps less severe and occurred in a smaller proportion of patients than after histamine and GTN. The difference was, however, not as marked as with immediate headache. Since no head-to-head direct comparison between CGRP-induced headache and GTN/histamine-induced headache has been made, it is difficult to know whether this difference is significant.
The very distinctive temporal profile of headache induced by GTN/histamine and now also CGRP makes it virtually certain that there must be underlying pathophysiological mechanisms that are shared. We have previously argued that nitric oxide (NO) may be the common mediator of GTN- and histamine-induced migraine (16). Subsequently, we have shown that the nitric oxide synthase (NOS) inhibitor L-NMMA is effective in the treatment of acute migraine attacks (17). It therefore seems intuitively likely that NO may also underlie CGRP-induced headache. In previous studies of isolated cephalic blood vessels from humans, CGRP interacted with a vascular smooth muscle receptor coupled to cyclic adenosine monophosphate (cAMP) and no effect of removal of the endothelium could be demonstrated (18–20). However, in recent years endothelial CGRP receptors have been demonstrated in an increasing number of blood vessels from different species. For example, the effect of CGRP in rat aorta and hindquarter arteries depends on an intact endothelium and is antagonized by NOS inhibitors, suggesting an endothelial receptor-mediated NO-dependent effect (21, 22). It has also been demonstrated that the CGRP-induced increase in human forearm blood flow can be inhibited by approximately 50% after a non-selective NOS-inhibitor, LNMMA (23). Finally, using molecular biological techniques, mRNA for the subcomponents CRLR, RAMP1 and RAMP of CGRP and adrenomedullin receptors have been demonstrated in the endothelium of human cranial blood vessels (I. Jansen-Olesen, personal communication). It is possible therefore that intravascularly infused CGRP induces migraine via endothelial receptors coupled to activation of endothelial NOS.
Another possibility is that CGRP is the common mediator of GTN, histamine and CGRP induced migraine. GTN has been shown to liberate CGRP from pial arterioles, a response partly responsible for the dilatory activity of GTN in the pial window model (24). NO has also been shown to liberate CGRP in other vascular beds (25). Whether histamine has a similar role is not clear.
If CGRP is the final common pathway for these experimental migraine headaches it remains to be explained why the induced migraine occurs several hours after the infusion. For NO an autoinduction of the formation of NO has been seen, resulting in prolonged production of NO after GTN (26), but no data are available to suggest autoinduction of CGRP.
Another explanation for the delay could be that sensitization is involved. Both NO and CGRP have been proposed to sensitize nerve terminals and the time delay may be counted in hours (9, 27, 28). According to this mechanism the vascular dilatation is long gone when sensitization develops and it must be the normal pulsations that then drive the pulsating headache. We are only aware of one study of sensitization during experimental migraine (29). More studies in this field would be of great interest.
Significance of the present results
The documentation in the present study of a direct headache-inducing potential of the messenger molecule CGRP has important implications. It shows that the previous finding of increased CGRP in external jugular venous blood is probably not an epiphenomenon but reflects direct participation of CGRP in migraine mechanisms.
Although the triptans represent a major step forward in the treatment of acute migraine attacks, these substances are not ideal. In clinical trials they appear better than they actually are because a successful result was defined as a decrease in pain intensity from 2 or 3 to zero or 1 on a 3-point pain scale. In other words, patients did not have to become pain free in order to be scored as having a successful outcome. Furthermore, approximately 30% of patients have recurrence after treatment with triptans. Using novel, more strict, outcome parameters such as complete freedom from pain or sustained response over 24 h (response without recurrence and without rescue medication) the response rate of triptans ranges between 20% and 50%. Therefore, the interest in developing drugs with a different mechanism of action has lately increased.
Unfortunately, several targets have recently proven useless, such as substance P antagonists (NK1 antagonists), endothelin 1 receptor antagonists and a novel class of agents with extreme potency in inhibiting neurogenic inflammation exemplified by CP122 288 (30–32). In view of these negative results it is stimulating that the present results demonstrate a possible causative role of CGRP in migraine. This finding greatly increases the likelihood that a CGRP antagonist may be effective in the treatment of migraine attacks. Several drugs that in various ways antagonize CGRP development (33) are presently known to be in pre-clinical and clinical trials but no results are available in the public domain.
Footnotes
Acknowledgements
The study was supported financially by The Horslev Foundation, the Cool Sorption Foundation and the Boehringer Ingelheim Foundation.
