Abstract
Background
Dilatation of cranial vessels has been proposed as a part of the cascade that initiates an episode of migraine. This is based on the observation that intravenous administration of several substances with vasodilator properties can trigger migraine-like symptoms in migraineurs.
Methods
We used in vitro myography of human cerebral arteries and in vitro pressurized arteriography of rat middle cerebral artery (MCA) to evaluate the vasomotor responses of cerebral arteries to increasing concentrations of vasoactive substances used to elicit migraine-like attacks.
Results
All substances except carbachol induced a strong vasodilatory response when applied to the abluminal side of a rat MCA but negligible response when applied to the luminal side. Luminal carbachol gave a strong dilatory response but a weak response at the abluminal side. The prostaglandins PGE2 and epoprostenol constricted the rat MCA while human cerebral arteries relaxed. The pEC50 of carbachol, histamine, epoprostenol, VIP and sildenafil differed significantly between cerebral arteries from man and rat. The differences in pEC50 for SNP, αCGRP, PACAP-27 and PACAP-38 were not significant between the species. PGE2 had no noticeable effect on human arteries in vitro.
Conclusion
All tested substances with the exception of VIP and carbachol have been found to elicit migraine-like attacks in migraineurs. Since these two agents have vasodilatory effects in humans, it suggests that vasodilatation is not the only reason for eliciting a migraine-like attack in migraineurs. In addition, there are significant species differences that show the importance of performing experiments in human vessels.
Introduction
Migraine affects up to 15% of the world’s population, making it the most common neurological disorder. Demographically, Caucasians are more often affected than members of other ethnic groups, smokers more often than nonsmokers and people of low education more so than those of high education (1). Genetic as well as environmental factors contribute to the development of the condition in an individual.
The condition has been known and studied for centuries, but the etiology of the associated symptoms is still the subject of much debate. Some theories hold that the origin of migraine-associated pain is to be found in the synapses of the trigeminal system. Certain stimuli cause substances to be released from the synapses, causing cerebral blood vessels to dilate and trigger the pain by activating nociceptors in the process (2). A possible origin of these stimuli may be nuclei located in the brain stem and diencephalon (3). Other theories claim that a migraine attack is initiated by peripheral stimulation, with nitric oxide (NO) being a key mediator (4). Whatever the cause of origin, it has been shown that calcitonin gene-related peptide (CGRP) is released from the trigeminal ganglion and its ramifications during an episode of migraine (5) and that this release is required for the migraine-related symptoms to fully emerge. A new generation of anti-migraine pharmaceutics in the form of CGRP-receptor antagonists shows promising clinical results (6).
Previously published observations of different substances’ abilities to elicit a migraine-like pain when infused intravenously into human test subjects. Data for PACAP-27 were not available at the time of this publication.
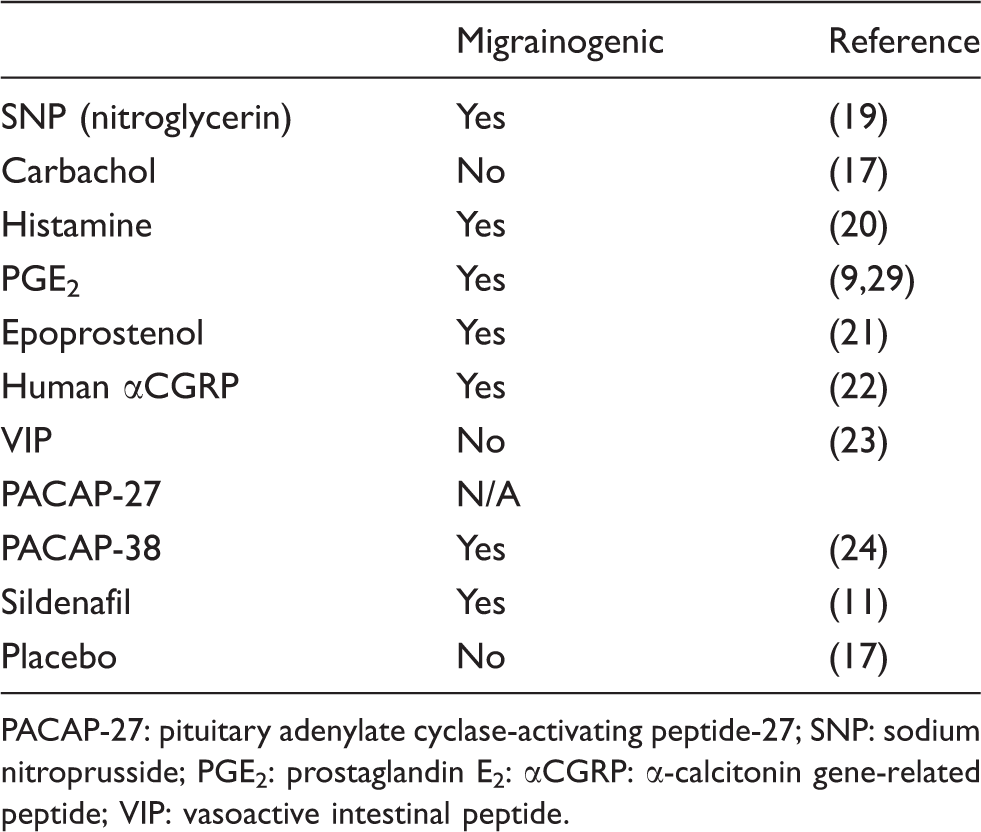
PACAP-27: pituitary adenylate cyclase-activating peptide-27; SNP: sodium nitroprusside; PGE2: prostaglandin E2: αCGRP: α-calcitonin gene-related peptide; VIP: vasoactive intestinal peptide.
The present study was designed to evaluate several pharmacologic substances suggested to be involved in migraine pathophysiology regarding their ability to dilate cerebral arteries. We hypothesized that the blood-brain barrier (BBB) would prevent them from reaching the smooth muscle cells of the tunica media or the sensory nerves of the outer wall. To examine this question, we conducted a series of in vitro experiments where we applied the substances to cerebral arteries from rat and humans and studied the vasomotor responses. For the rat cerebral arteries, we used a pressurized arteriography system, allowing us to apply substances to either the luminal or the abluminal side of the arteries thereby addressing if the BBB would make a difference. Abluminal application of a substance would mimic the release of said substance from nerve endings inside of the BBB. This approach has not been used with human cerebral vessels because of the uneven quality of the samples. Instead, the human arterial segments were examined in a sensitive myograph, placed in a tissue bath and subjected to the tested substances. Due to the scarcity of samples of human cerebral arteries taken from live patients, we have borrowed results from other publications whenever possible and used the human tissue available to us to complement these data with novel experiments.
Materials and methods
Preparation of rat cerebral arteries
The Animal Protocol Review committee at the University of Lund approved the experimental protocol (M111-04).
Male Sprague-Dawley rats (250–350 g) were anesthetized using CO2 and decapitated. The brain was immediately removed and placed in a cold (4°C) buffer solution of the following composition: NaCl 119 mM, NaHCO3 15 mM, KCl 4.6 mM, MgCl2 1.2 mM, NaH2PO4 1.2 mM, CaCl2 1.5 mM and glucose 5.5 mM.
With the aid of a dissecting microscope, a segment of the middle cerebral artery (MCA) was carefully harvested from each animal, beginning at the circle of Willis and extending 5–8 mm distally.
Preparation of human cerebral arteries
Macroscopically healthy cortical branches of human cerebral arteries were obtained from patients undergoing neurosurgery because of tumors or epilepsy. Following their removal, the vessels were immediately placed in cold, sterile Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Invitrogen, Carlsbad, CA, USA) and transported to the laboratory, where they were carefully dissected free from connective tissue, leaving the vessels with intact intima, media and adventitia.
All procedures were carried out strictly within national laws and guidelines and approved by the Ethical Committee at the University of Lund (LU-818-01).
Pressurized arteriography experiments with rat MCA
A segment of the MCA (1–2 mm in length) was mounted in a pressurized arteriography system (Living Systems, Burlington, VT, USA) as previously described (7). Micropipettes were inserted into both ends of the MCA and secured with 11-0 nylon ties. The MCA was bathed in the described buffer solution (37°C) and equilibrated with a gas mixture consisting of 5% CO2 and 95% O2, resulting in a pH of 7.4. Transmural pressure of the MCA was maintained at 85 mmHg by raising reservoirs connected to the micropipettes to the appropriate height above the MCA. Luminal perfusion was adjusted to 0.1 ml/min by setting the two reservoirs at different heights. Pressure transducers on either side of the MCA provided direct measurement of perfusion pressure across the MCA.
The vessel was magnified 600-fold using a microscope connected to a digital camera (Axis®, Lund, Sweden) and a computer. The program Mary® (Nihil KB, Lund, Sweden) saved pictures at intervals of one second and measured the outer diameter of the vessel during the experiments.
Following a resting period, the pressurized vessels attained a stable tone, considered to be due to shear stress. Any MCA that did not develop spontaneous tone of at least 10% compared to the initial diameter within 1 hour (h) was excluded from further experiments. After another period of 30 minutes (min), the MCA segments were exposed to luminal adenosine triphosphate (ATP) (10−5 M) in order to evaluate the function of the endothelium. A dilation of at least 10% of the resting diameter was considered indicative of a functional endothelium and preserved the structural integrity of the BBB. Experimental protocols were not initiated until the MCA diameter was stable over a 15-min period. The testing of the vascular response to a substance was carried out by adding the substance to the luminal perfusate (in a concentration range of 10−12 to 10−6 M). In addition, the substance was added abluminally in the same concentration range to test the direct smooth muscle effect, since this route of administration avoids interaction with the endothelium. The substances tested were sodium nitroprusside (SNP), carbachol, histamine, prostaglandin E2 (PGE2), epoprostenol, human α-calcitonin gene-related peptide (αCGRP), vasoactive intestinal peptide (VIP), pituitary adenylate cyclase-activating peptide-27 (PACAP-27), PACAP-38, sildenafil and a placebo buffer consisting of a 6% dimethyl sulfoxide (DMSO) solution.
At the end of each experiment, the vessel segments were exposed to a calcium-free buffer solution that allowed us to recalibrate the value of the maximum relaxant capability of each MCA segment.
Figure 1 shows pictures taken during the different phases of the experiment following the above protocol.
Pictures taken of a rat middle cerebral artery (MCA) suspended in an arteriography system. The images show the different phases of a typical experiment.
Myograph experiments with human cerebral arteries
The human cerebral arteries obtained were cut into 2 mm long circular segments and placed in ice-cold buffer solution aerated with a gas consisting of 5% CO2 and 95% O2 (8). In order to determine vessel tension, each segment was mounted on two metal wires, 40 µm in diameter, in a myograph (Model 610 M; Danish Myo Technology, Aarhus, Denmark). One wire was connected to a force displacement transducer attached to an analogue-digital converter unit (ADInstruments, Oxford, UK). The other wire was connected to a micrometer screw, allowing fine adjustments of vascular tone by varying the distance between the wires. Measurements were recorded on a computer using a PowerLab unit (ADInstruments).
The segments were immersed in a temperature-controlled buffer solution (37°C). The buffer was continuously aerated with oxygen enriched with 5% CO2, resulting in a pH of 7.4. The vessels were stretched to an initial resting tone of 2 mN and then allowed to stabilize at this tone for 1 h. The contractile capacity was determined by exposing the vessels to an isotonic solution containing 63.5 mM of K+, obtained by partially replacing NaCl with KCl in the above buffer. The contraction induced by K+ was used as a reference for the contractile capacity. Only vessels responding by contraction of at least 2 mN to K+ were included in the study.
In order to study the relaxant effect of different substances, the MCA was precontracted with the thromboxane receptor agonist (TxA2) U499166. This resulted in a stable tension lasting for at least 30 min without significant fall in tone. Concentration-response curves were obtained by cumulative application of the tested substance (Sigma, St. Louis, MO, USA) in a concentration range of 10−12 to 10−5 M.
Data analysis
For data obtained from experiments performed in the pressurized arteriography system, the changes in the measured diameters of the vessel segments are expressed as a percentage of the resting diameter. Dilation is given as a percentage of the spontaneous tension obtained through perfusion (0%) and the maximum relaxation obtained at the end of the experiment to a calcium-free medium (100%). For the experiments performed with the myograph, the amount of relaxation induced by each concentration of substance was calculated as a percentage of maximum vessel diameter restored from the TxA2 receptor-induced precontraction. Values in all tables and diagrams are given as mean ± SEM. The number of experiments = n. Emax% in the arteriography experiments denotes the percentage of the maximum response of ATP a substance is able to induce. In the myograph experiments, Emax% denotes the percentage of the precontraction a substance is able to restore toward the baseline. The pEC50 denotes the negative logarithm of the concentration needed of a substance to elicit half of Emax%. A t test was performed to test the difference in Emax% between luminal and abluminal application of a substance to rat MCA. The hypothesis that the pEC50 for a given substance is different between humans and rats was also tested for significance with the Student’s t test with Bonferroni correction. A p value of <0.05 is given when values are significantly different.
All concentrations expressed indicate the final concentration in the luminal or abluminal compartments of the arteriography system and in the tissue baths of the myograph.
Results
Pressurized arteriography experiments with rat MCA
The results obtained from testing the different pharmacologic substances in the pressurized arteriography system are displayed in Table 2 and in Figure 2.
Maximum response (Emax%) elicited luminally and abluminally by the different substances in rat middle cerebral artery (MCA). Asterisks denote the level of statistical significance (p < 0.05) of the difference between the two modes of application. The number of experiments is given in Table 2. The Emax% and pEC50 of all substances tested luminally and abluminally on rat MCA in the arteriograph. Responses below 10% are difficult to use properly for calculation of pEC50. In cases where Emax% is below 10%, the pEC50 is designated as not demonstrable (ND). Values are given as mean ± SEM, n = number of arteries. MCA: middle cerebral artery; Emax%: maximum response; pEC50: negative logarithm of the concentration needed of a substance to elicit half of Emax%; SNP: sodium nitroprusside; PGE2: prostaglandin E2; αCGRP: α-calcitonin gene-related peptide; VIP: vasoactive intestinal peptide; PACAP-27: pituitary adenylate cyclase-activating peptide-27; DMSO: dimethyl sulfoxide.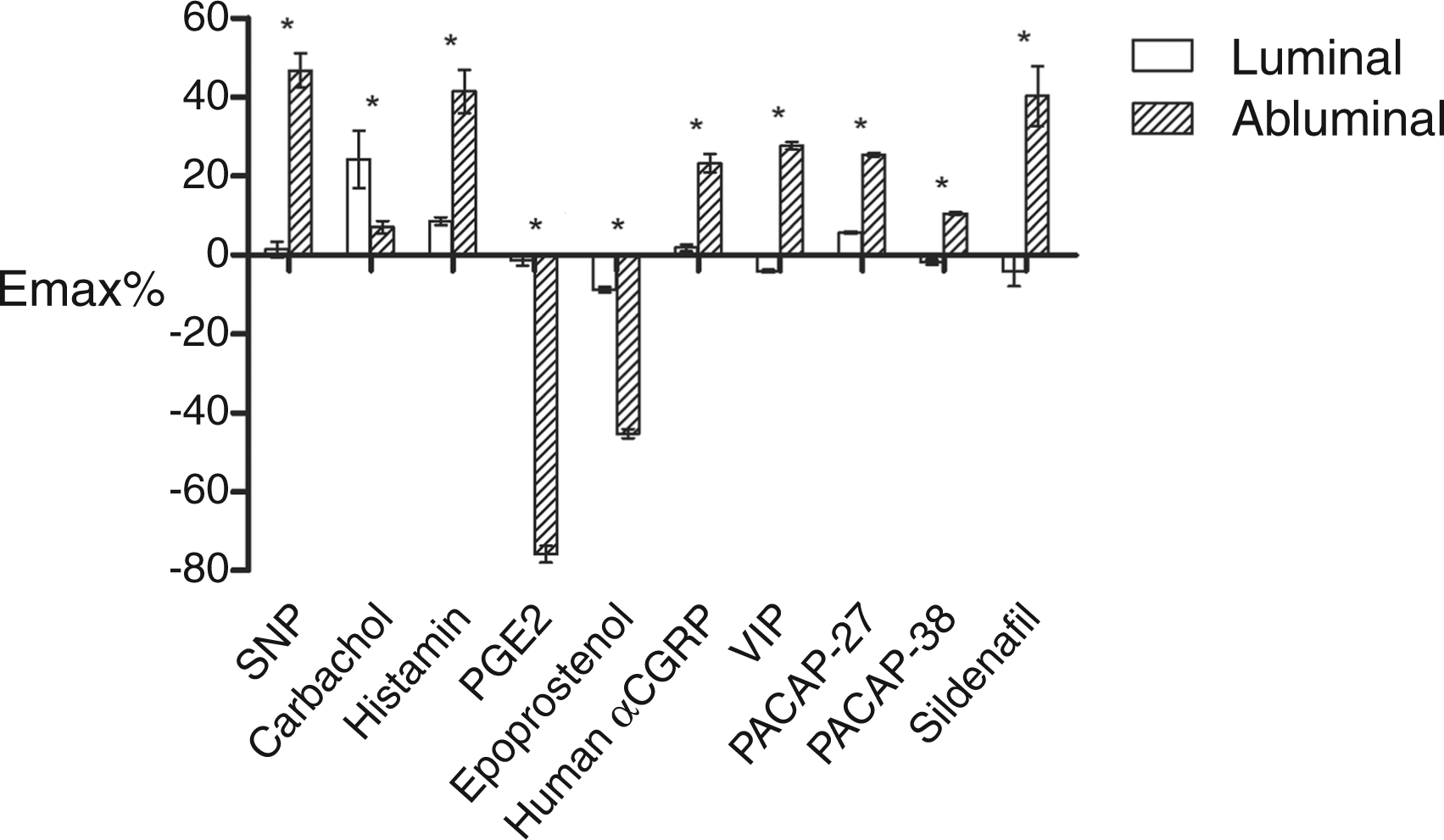

All substances tested, except for carbachol, elicited a demonstrable vasomotor response from the rat MCA only when administered abluminally. Inversely, carbachol was the only substance that gave rise to a dilation of significant magnitude (Emax% = 24.2 ± 7.3) when applied to the luminal side of the vessel. A strong response from a luminally applied substance indicates that the receptor site is located on the endothelial cells, while a response from an abluminally applied substance indicates that the receptors are located on the smooth muscle cells of the tunica media, provided that the substance is unable to cross the BBB.
Two of the vasodilators, PGE2 and epoprostenol, were shown to contract, rather than dilate, the rat MCA (Emax% = −75.8 ± 2.1 and −45.3 ± 1.1, respectively). The difference in Emax% between luminal and abluminal application of a substance was significant for each of the tested substances (Figure 2).
Myograph experiments with human cerebral arteries
The Emax% and pEC50 of different substances tested on human MCA in the myograph. The data for SNP, carbachol, PGE2, PACAP-38 and sildenafil were produced in-house, while the rest were borrowed from previous publications. Values are given as mean ± SEM, n = number of arteries.
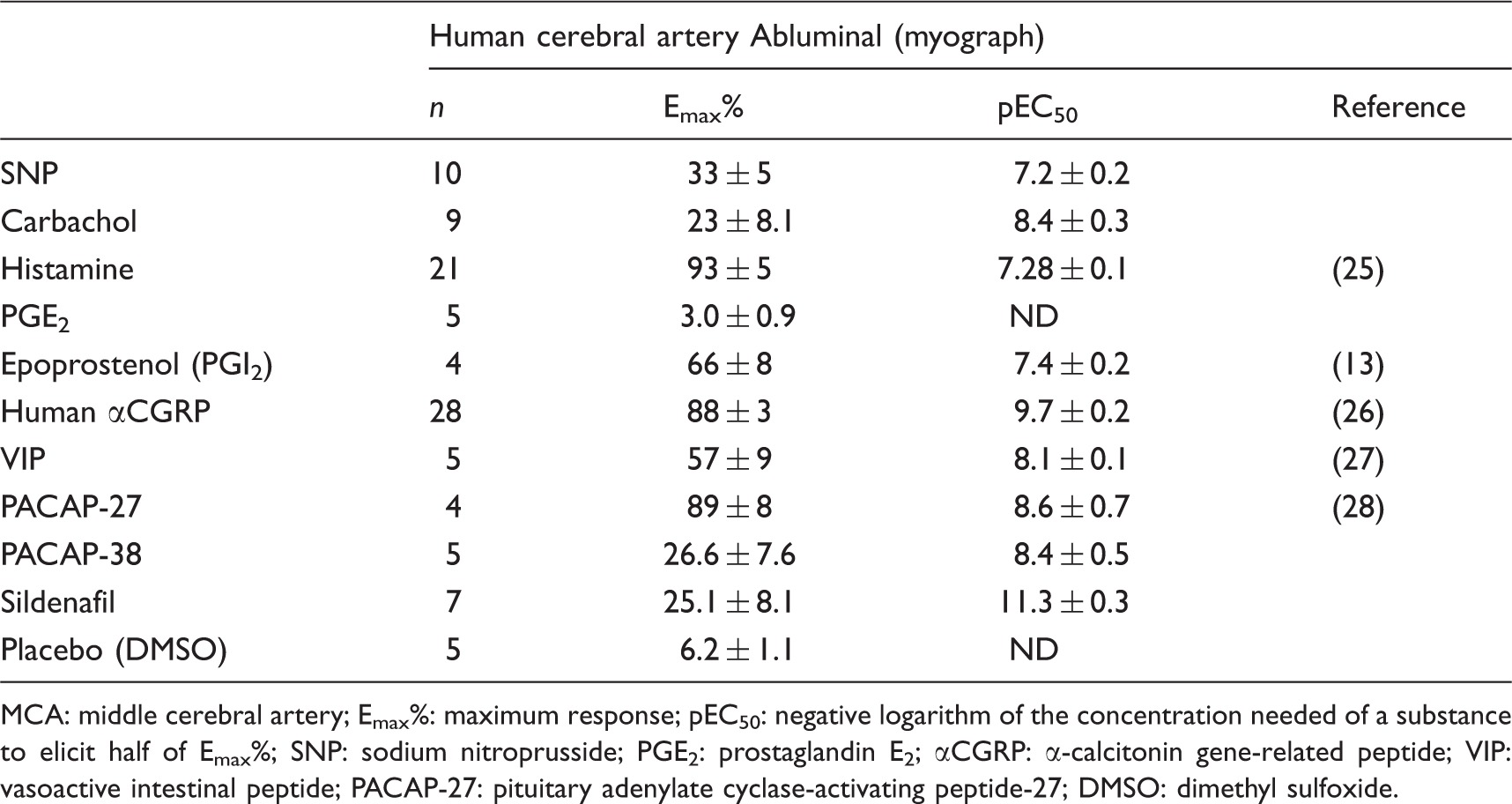
MCA: middle cerebral artery; Emax%: maximum response; pEC50: negative logarithm of the concentration needed of a substance to elicit half of Emax%; SNP: sodium nitroprusside; PGE2: prostaglandin E2; αCGRP: α-calcitonin gene-related peptide; VIP: vasoactive intestinal peptide; PACAP-27: pituitary adenylate cyclase-activating peptide-27; DMSO: dimethyl sulfoxide.
The pEC50 from human cerebral arteries was shown to be significantly different from the corresponding values in rat for carbachol, histamine, epoprostenol, VIP and sildenafil (all with p < 0.05). The substances that, when compared between species, did not reveal significant differences in responses were SNP, αCGRP, PACAP-27 and PACAP-38. PGE2 did not reveal a significant vasomotor effect on the studied human arteries in vitro (Figure 3).
Comparison between the pEC50 for the different substances’ ability to dilate human and rat cerebral arteries. The rat pEC50 values refer to abluminal administration of substances, with the exception of carbachol, which refers to luminal administration. In the case of epoprostenol, the pEC50 for rat refers to contraction of the middle cerebral artery (MCA) rather than dilation. PGE2 is absent since it gave no significant vasomotor response in human arteries that could be used to calculate pEC50. Asterisks denote the level of statistical significance of the difference between the two species (p < 0.05 or NS = not significantly different). For number of experiments, see Tables 2 and 3.
Discussion
The present study is the first to perform a systematic comparison between different molecules used to trigger migraine attacks in patients and their vascular responses in isolated human cerebral arteries. In addition, it was performed in order to increase the understanding of the mechanisms activated on intravenous administration of migraine-inducing substances. Table 1 summarizes the published results from a series of in vivo experiments where the development of migraine-like symptoms was examined in test subjects subjected to intravenous infusion of various vasodilators. These results are compared with our data to further elaborate on the relationship between migraine and vasodilators such as NO.
It should be pointed out that in this paper we have studied only cerebral arteries. The BBB of the cerebral arteries is an obstacle to the passage of the various substances across the BBB to the tunica media in vivo, and this is partly modeled by the perfused MCA in the rat experiments. With this in mind, we saw a fairly good correlation between the results obtained from human brain vessels and from rat MCA when substances were given abluminally, although there was a significant difference in the magnitude of response between the two species in most cases.
Human cerebral arteries were significantly more sensitive (higher pEC50) as compared to corresponding values in rat for carbachol, histamine, epoprostenol, VIP and sildenafil while there was no species difference for SNP, αCGRP, PACAP-27 and PACAP-38 (Figure 3). PGE2 did not have any significant vasomotor effects on human cerebral arteries in vitro. However, previous in vivo experiments with human test subjects have shown that an intravenous infusion of PGE2 induces vasodilatation in both the MCA (detected as a decrease in velocity of blood flow) and the superficial temporal artery (STA), which is not part of the BBB (9). That gives us a difference in response between human and rat arteries during in vitro experiments and a difference in response for human arteries between in vitro and in vivo experiments. The reason for this difference is unknown.
The vasodilatory effects of VIP and PACAP-27 on meningeal arteries (pEC50 = 8.4 ± 0.1 and 7.7 ± 0.2, respectively) have been described in a recent publication (10). The difference in pEC50 of these substances on cerebral arteries is small.
An interesting observation is that sildenafil caused a fairly strong dilation of the human vessels in vitro, but had no effect on cranial vessels without BBB when given in vivo (11). It has been suggested that sildenafil differs in effect between extracerebral and cerebral arteries and that this rules out a cerebrovascular site of action in migraine-inducing responses (11). Our results clearly show that it has to do with the site of administration and that previous in vivo responses on vasculature demonstrate only that it does not pass the BBB and that there are sites that can be activated by sildenafil. The present data are in agreement with a basic study by Kruuse et al. (12) where the presence of the phosphodiesterase 5 (PDE5) protein was confirmed by Western blot in human cerebral arteries.
A surprising result was that the PGI2 agonist epoprostenol did not induce vasodilation, but rather induced vasoconstriction in the rat MCA. This was in contrast to findings in human cerebral arteries in which epoprostenol induced strong vasodilation (13). Previous in vivo experiments have shown that the PGI2 analogue iloprost dilates rat meningeal (dura mater) arteries (14), and we expected abluminal epoprostenol to do the same to rat MCA. The cause of these conflicting results is open to debate, but a possible explanation is a difference in receptor affinity between epoprostenol and iloprost or a difference in receptor expression between the middle meningeal artery (MMA) and MCA in the rat. The results from human vessels were obtained using natural PGI2, rather than synthetic analogues, and the difference in response compared to our experiments with epoprostenol on rat MCA could in this case too be due to a difference in receptor affinity for the two analogues used. Another possible reason is that the receptor subtype distribution might be different among the two species: rat and human.
In rat MCA, PGE2 acted as a vasoconstrictor when given abluminally, but since no significant contraction was induced by luminal PGE2, we can rule out an endothelial response and any involvement from putative endothelial PGE2-receptors. Our results are contradicted by previously published results, where PGE2 dilated rat MCA and MMA (15). PGE2 failed to induce any significant response in human arterial segments in our in vitro experiments, but we know that PGE2 dilates the human cerebral artery and other cranial arteries in vivo (9). The reasons for these differences between species and methodology are not known to us, but future investigations will expand on the subject.
The dilatation of the rat MCA induced by luminal carbachol provides another interesting observation. Acetylcholine analogues like carbachol do not induce migraine, but they induce vasodilation by the formation of NO in the vascular endothelial cells (16). Carbachol binds to muscarinic receptors on the endothelium at the luminal side of the vessel, which explains the weak response to abluminally applied carbachol. This means that carbachol is an effective vasodilator in any cranial vessel, regardless of whether that vessel is part of the BBB or not. The substances listed in Table 1 show no correlation between having a migrainogenic effect and having vasodilator effect mediated via the endothelial NO pathway. Epoprostenol, αCGRP, PACAP-38 and possibly histamine induce “migraine-like” responses, but do not act via formation of NO. Carbachol, as mentioned, stimulates production of NO but does not induce migraine (17). This challenges the theory that NO-induced vasodilation is a prominent mediator of migraine via an endothelial mechanism (4).
Conclusion
SNP, carbachol, histamine, αCGRP, VIP, PACAP-27, PACAP-38 and sildenafil all have a dilatory effect on human and rat cerebral arteries (Table 2, Table 3). The pEC50 was shown to be significantly different for some of these substances, but the overall similarity in vascular response still indicates that the rat model is a good surrogate for the human model when conducting vasodilator experiments.
Substances that induced fundamentally different responses when applied to rat and human cerebral arteries were epoprostenol and PGE2. Epoprostenol acts as a vasodilator on human cerebral arteries, but as a vasoconstrictor on rat MCA (Table 2). PGE2 induced no response whatsoever in human cerebral arteries in vitro, but it constricted the rat arteries. Some controversy surrounds our claim regarding the vasoconstriction properties of PGE2, in particular when tried on rat MCA, since previously published observations demonstrate the opposite reaction (15). Even if these results conflict with our data, they still show that a response was induced by PGE2 in rat arteries, as opposed to the lack of response we observed in human arteries. Due to these differences between species, it is not recommended to conduct experiments with epoprostenol and PGE2 using a rat model as a substitute for human material.
The comparison between the vascular responses of different vasomotor agents and their ability to induce migraine-like attacks following intravenous infusion rule out the vascular endothelial NO as a common denominator. This view is based on the fact that many migrainogenic substances are independent of endothelial NO formation for their vasomotor effect. Also, carbachol is not migrainogenic, despite being a potent NO-dependent dilator of human cerebral arteries with full luminal access to its endothelial receptor site. This conclusion is supported by recent clinical trials where a selective inducible NO synthase (iNOS) inhibitor was found to be without effect in relieving acute migraine attacks (18). These recent findings add further weight to the view that migraine is not primarily a cerebrovascular disease, but that the trigeminal system has a significant role in the pathophysiology (3).
Clinical implications
Vasodilation has been suggested as one key player in the induction of migraine attacks. We have compared several molecules used in studies aimed at inducing migraine-like attacks and find no single mechanism shared by them. Nitric oxide has been suggested as one unifying molecule; however, the agents eliciting migraine-like attacks do not have this ability. The data rather suggest that vascular effects may exacerbate an attack in a migraine-prone patient.
Footnotes
Funding
This research received support from the Swedish Research Council (grant no. 5958).
Conflict of interest
None declared.
