Abstract
The efficacy, safety and tolerability of the 5-HT1B/1D receptor agonist eletriptan (40 mg and 80 mg) in acute treatment of migraine was evaluated in a multinational, randomized, double-blind, parallel-group, placebo-controlled, three-attack study treating 1153 patients. In the initial attack, significantly more eletriptan patients reported headache relief and complete pain relief at 2 h vs. placebo (40 mg 62% and 32%, 80 mg 65% and 34%, placebo 19% and 3%; P < 0.0001). Headache relief occurred faster after eletriptan, with more patients at both doses reporting relief 30 min (P < 0.01) and 1 h (P < 0.0001) after treatment than after placebo. There was a significantly lower recurrence rate with eletriptan 80 mg compared with placebo (P < 0.01). Adverse events for all treatments were generally mild or moderate and self-limiting. Eletriptan 40 mg and eletriptan 80 mg both appear to be effective and well-tolerated acute migraine treatments.
Introduction
The introduction of sumatriptan, the first of the targeted 5-HT1B/1D receptor agonists, represented a substantial therapeutic advance over the non-specific treatments previously available. However, pharmacokinetic and clinical data have indicated that oral sumatriptan has some limitations in terms of the onset of action, efficacy and headache recurrence rates (1). Eletriptan is a novel, potent, selective 5-HT1B/1D receptor agonist that has shown efficacy in the acute treatment of migraine over the dose range 20–80 mg in placebo-controlled clinical trials (2, 3). Eletriptan is characterized by a high affinity at both 5-HT1B and 5-HT1D receptor subtypes (4, 5), and demonstrates rapid association and slow dissociation from the 5-HT1D receptor in vitro (4). It has been shown to be selective for cranial vs. coronary vasculature in animal models (6) and in isolated human tissue (7). In pharmacokinetic studies, eletriptan has been shown to have fast and consistent absorption (8, 9), bioavailability of 50% (8) and a plasma half-life of 4–5 h (9). A recent comparative clinical study (3) has shown that eletriptan 40–80 mg had a faster onset of response and improved overall patient acceptability compared with oral sumatriptan 100 mg. Furthermore, eletriptan 80 mg was more effective in the relief of headache than sumatriptan 100 mg at 1 h and 2 h following dosing (3).
The objective of the study reported here was to further characterize the efficacy and tolerability of 40 mg and 80 mg eletriptan for treatment of three consecutive migraine attacks in a large population.
Methods
This multinational, randomized, double-blind, placebo-controlled, parallel-group study evaluated the safety and efficacy of oral eletriptan (40 mg or 80 mg) across three attacks for the acute treatment of migraine.
Male and female patients aged over 18 years, who had a history of at least one migraine attack every 6 weeks as defined by the International Headache Society (IHS) diagnostic criteria for migraine with or without aura (10), were enrolled by centres in Australia, Europe and South Africa. Exclusion criteria included: patients with concomitant frequent non-migrainous headache (i.e. more than six attacks per month on average); atypical migraine that had consistently failed to respond to migraine therapy; or a history of migraine with prolonged aura, familial hemiplegic migraine, basilar migraine or migrainous infarction (10). In addition, patients with clinical evidence or history of other clinically significant (including cardiovascular) disease, severely limited gastrointestinal absorption, documented drug allergy, drug or alcohol misuse, or who intended to donate blood for up to 1 month following completion of the study, or were likely to be non-compliant with the study procedures, were excluded. Female patients who were pregnant, breast-feeding or not taking effective contraceptive precautions were not considered for entry. Concomitant treatment with any investigational drug, including eletriptan, in the month before entry was prohibited. Treatment with analgesics or ergotamine on more than 2 days in any 7-day period during the study was prohibited. Patients taking migraine prophylactic treatments were eligible for entry; however, treatment with analgesics or anti-emetics in the 6 h before dosing or with sumatriptan or ergot derivatives in the 48 h before dosing was not permitted. The study was approved by the ethics committee responsible for each centre and performed in accordance with the Declaration of Helsinki (Hong Kong revision, 1989). All patients gave written informed consent before entry.
Study procedure
Patients were evaluated for eligibility at a screening visit that included a medical history, review of systems and a migraine treatment history. A physical examination and a 12-lead resting ECG were performed, and blood samples were taken for measurement of routine laboratory safety variables. A urine pregnancy test was performed for all female patients of childbearing potential.
Eligible patients were randomized to one of three double-blind study treatments: eletriptan (40 mg or 80 mg) or matching placebo, in the proportion of 2 : 2 : 1. Patients were instructed to take the study drug as soon as possible but within 6 h of onset of the attack, provided that the headache was moderate or severe, that the headache had not spontaneously improved, and provided that any aura phase had ended. Symptom intensity was assessed immediately before and at 0.5, 1, 2, 4 and 24 h after dosing. For patients who did not respond to the initial dose of study medication, or who experienced headache recurrence, the protocol allowed the administration of a second dose of eletriptan or placebo between 2 h and 24 h after the first dose. The results presented in this paper relate to the efficacy of the first dose only for each of the three attacks: the second dose results form part of a prospectively planned meta-analysis across all eletriptan phase III trials, which will be presented separately. Rescue medication other than sumatriptan or ergot derivatives was permitted, but only from 2 h after any second dose of study medication.
At the time of initial randomization, patients were instructed in the completion of the migraine diary. Patients were instructed to rate the intensity of their headache pain and their degree of functional impairment using a 4-point scale (severity of headache pain: 0 = no pain to 3=severe pain; functional impairment: 0=normal activity to 3=unable to perform activities and requiring bed rest), and to indicate whether nausea, vomiting, photophobia or phonophobia were present.
Patients were also instructed to record any adverse events and concomitant medication use from the start of the study to the final visit day. Adverse events for a particular attack were considered to have occurred if they happened from the time the medication was taken until 7 days afterwards. If another attack (not a recurrence) occurred before the end of the 7-day period, the window for an adverse event from the previous attack was closed and a new one was begun. In addition, patients were asked, 24 h after taking a dose of study medication, to rate the acceptability/preference for study treatment by answering the question: ‘Given the choice between this and any other medication to treat a migraine attack, would you take this again?’
Patients returned to the clinic for assessment between 1 and 7 days after taking study treatment for the first attack. Physical examination, ECG and laboratory safety assessments were repeated; all adverse events, use of rescue medication and changes in concomitant medication were noted, and migraine diaries and all study medication containers, and any unused study medication, were collected by the investigator. Patients were then issued additional diaries and study medication for the next two attacks. Patients who had received eletriptan for the first attack were allocated the same dose for the next two attacks. Patients who had previously received placebo were allocated to treatment with eletriptan, either 40 mg or 80 mg, in accordance with the initial segmented randomization schedule, for the next two attacks.
For each of the next two attacks, patients were asked to take the study medication as soon as possible after the onset of each migraine attack, provided the criteria specified for the first attack were also satisfied. Symptoms were recorded in the migraine diary at 2, 4 and 24 h after dosing. Treatment response was assessed by patients at 2 h after dosing for each attack. As for the first attack, a second dose was permitted if the patient had failed to respond to the initial dose or had suffered a subsequent recurrence at any time during the period 2–24 h after dosage, and rescue medication was only permitted from 2 h after the second dose.
Patients returned for a final assessment 7 to 14 days after the third attack, or within 16 weeks of the baseline visit, whichever occurred sooner. Patients who did not treat the required number of attacks within 16 weeks were withdrawn from the study. However, all patients with baseline and treatment data for one or more attack were included in the intent-to-treat (ITT) analysis. Physical examination, ECG and laboratory safety assessments were repeated, and all adverse events and changes in concomitant medication were noted. The investigator collected migraine diaries, all study medication containers and any unused study medication.
Statistical analysis
The primary efficacy endpoint was headache response, defined as a reduction in the intensity of headache pain from moderate or severe at baseline to mild or no pain, at 2 h after the first dose taken for the first attack. All analyses were performed on the intent-to-treat (ITT) population on an attack-by-attack basis. Response rates were calculated and analysed using logistic regression and analysis of covariance (
Comparisons of pain-free responder rates, the incidence of nausea, vomiting, photophobia and phonophobia, and headache recurrence rates after each attack, were made using the same procedure outlined above. Headache recurrence was defined as the return of a moderate-to-severe headache, from a previously improved level of mild or no headache, between 2 h and 24 h after taking study medication. Functional impairment data were analysed using logistic regression techniques. Safety data were summarized descriptively for all patients who received at least one dose of the study medication.
Results
Of a total of 1365 patients screened for the study by 75 centres (68 in Europe, three in South Africa and four in Australia), 1354 patients were subsequently randomized to the study treatment. A total of 1153 patients took the study treatment: 238 received placebo, 453 received eletriptan 40 mg and 462 received eletriptan 80 mg (Fig. 1). Among 201 patients who did not take study medication, the most frequent reason was that the patient did not experience an attack within the study period. Others included patient being lost to follow-up, or withdrawn due to a protocol violation.
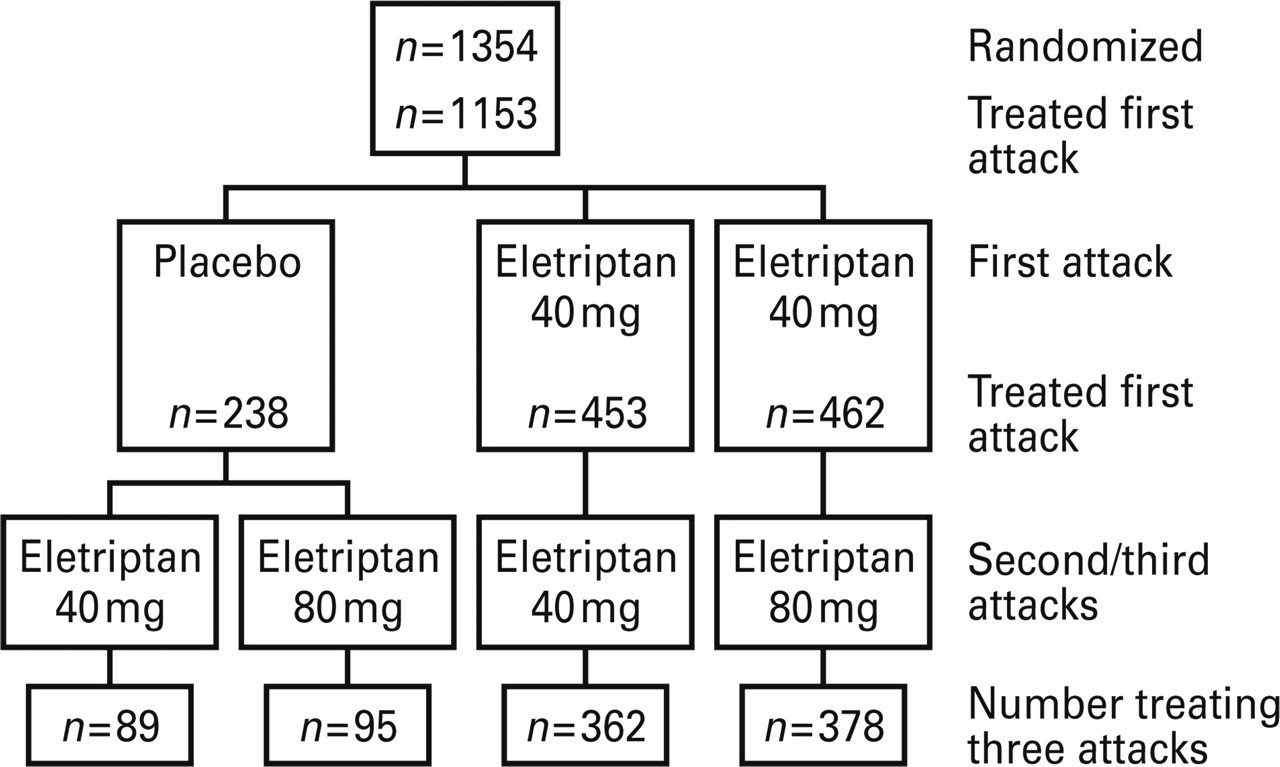
Allocation of patients to treatment groups.
All three treatment groups appeared to be similar with respect to demographic and baseline migraine symptoms (Table 1). In each of the treatment groups there was a similar proportion of female patients (more than 80%). Migraine without aura was the most common diagnosis (range across treatment groups 64–67% of patients), although a substantial proportion of patients (range 24–29%) also reported migraine both with and without aura. Duration since diagnosis was also similar across treatment groups and diagnoses (range 0.3–58 years). Patients had reported approximately nine attacks on average (range across treatment groups 8.7–9.9) over the previous 3-month period, the majority of which were of at least moderate intensity (range 85.6–90.7%). Most patients had previously received sumatriptan for treatment of their migraine (range 68.0–73.4%). Baseline migraine symptoms for the first attack were similar in each group; across all three treatment groups, 59% of patients reported moderate headache pain and 41% reported severe headache pain, and most patients (more than 60%) reported nausea, photophobia and phonophobia during attacks.
Patient demographics and baseline characteristics of all treated patients∗

∗Treatment group defined by initial dose for first attack.
†Functional impairment was rated using a 4-point scale: 3=requiring bed rest; 2=severely impaired functioning; 1=activities reduced; 0=normal functioning.
In total, 924 (80%) patients completed the study (184 in the placebo group, 362 in the eletriptan 40 mg group and 378 in the eletriptan 80 mg group). A total of 148 patients (36 in the placebo group, 57 in the eletriptan 40 mg group and 55 in the eletriptan 80 mg group) discontinued after the first attack. The most common reason for discontinuation was that the subject did not treat any further attacks during the study period. Reasons for discontinuation that were considered treatment-related were insufficient response, adverse events and laboratory abnormalities. A higher proportion of patients in the placebo group than either active treatment group withdrew at this time due to insufficient response (19% in the placebo group vs. 7% in the 40 mg group and 5% in the 80 mg group). Of the remaining patients, 79 patients discontinued before the final attack (including 18 in the placebo group) and two withdrew after the final attack.
Headache response
Headache response rates were significantly higher at 0.5 h, 1 h and 2 h after a single dose of eletriptan 40 mg or 80 mg than placebo for the first attack (Fig. 2). The percentages of patients who were pain-free were significantly higher at 1 h and 2 h after a single dose of eletriptan 40 mg or 80 mg than following placebo. At 2 h after the first dose, 62% of patients in the 40 mg group and 65% of patients in the 80 mg group reported a headache response compared with only 19% on placebo (P < 0.0001 for each comparison). In addition, 32% of patients in the 40 mg group and 34% of patients in the 80 mg group reported that they were completely pain-free at 2 h compared with only 3% in the placebo group (P < 0.0001 for each comparison) (Fig. 2a). The onset of the response was rapid, with a statistically significant headache response being observed as early as 0.5 h; 8% and 11% of patients on eletriptan 40 mg or 80 mg, respectively, reported a headache response by the 0.5 h time-point, compared with 2% on placebo (P < 0.01 and P < 0.001, respectively) (Fig. 2b). At 1 h, 33% of patients in both eletriptan groups reported a headache response compared with only 9% in the placebo group (both, P < 0.0001). In terms of pain-free response, 6% of eletriptan 40 mg and 12% of eletriptan 80 mg patients were pain-free at 1 h, compared with 0% in the placebo group (both P < 0.0001).
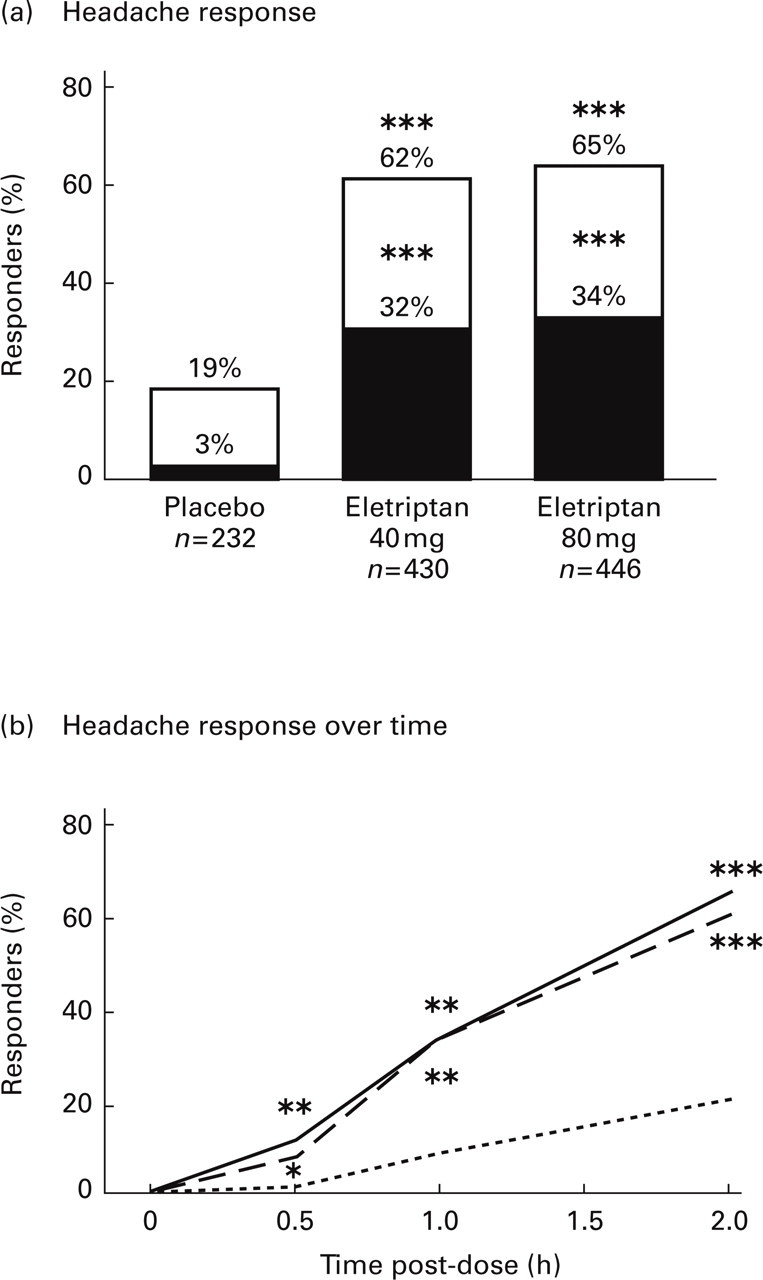
(a) Proportion of patients reporting a headache response rate or being pain-free at 2 h following treatment of the first attack with a single oral dose of eletriptan 40 mg or 80 mg or placebo: n=number of subjects in the ITT group with an assessment at 2 h. □ Mild or no pain; ▪ Pain-free. (b) Headache response rate over time following treatment of the first attack with a single oral dose of eletriptan 40 mg (–––) or 80 mg (—); Placebo (–––). Headache response is defined as a change from moderate-to-severe pain at baseline (pre-dose) to mild pain or no pain at the relevant time-point. ∗P < 0.01; ∗∗P < 0.001, ∗∗∗P < 0.0001, eletriptan vs. placebo: n=number of subjects in the ITT group at baseline.
Relief of functional impairment
At baseline, 88% to 89% of patients in all groups had functional impairment of grades 2 or 3 (do nothing or very little, 58–61%; required bed rest, 28–30%). At 2 h after treatment, significantly higher proportions of patients receiving eletriptan 40 mg or 80 mg than those receiving placebo reported that they were able to perform some activities or resume normal activity (58% on eletriptan 40 mg and 62% on eletriptan 80 mg vs. 20% on placebo; P < 0.0001 for each comparison vs. placebo) (Table 2 and Fig. 3). The functional response was fast in onset, with significantly more patients in the eletriptan 40 mg and 80 mg groups than in the placebo group reporting a functional response at the 0.5 h (9–10% vs. 3%) and 1 h (31–32% vs. 13%) time-points (Table 2 and Fig. 3).
Functional response at 0.5, 1 and 2 h post-dose: ITT population (1st attack, 1st dose)

P-value vs. placebo:
∗ P≤0.01,
∗∗ P < 0.01.

Functional response rate over time following treatment of the first attack with a single oral dose of eletriptan 40 mg (––– n=461) or 80 mg (––– n=452) or placebo (––– n=238). Functional response is defined as a change from being able to do nothing or requiring bed rest at baseline (pre-dose) to normal function or being able to do something at the relevant time-point. (Statistical analyses were only performed at the 2 h time-point) n=number of subjects in the ITT group at baseline. ∗∗∗P < 0.0001 vs. placebo.
Relief of other migraine-associated symptoms
Patients treated with eletriptan 40 mg or 80 mg reported a significantly lower incidence of nausea, photophobia and phonophobia at 2 h after dosing compared with placebo patients (P < 0.0001 for each eletriptan dose vs. placebo; Fig. 4). Baseline rates of vomiting were too low (4%−8%) to permit an analysis of treatment effects.
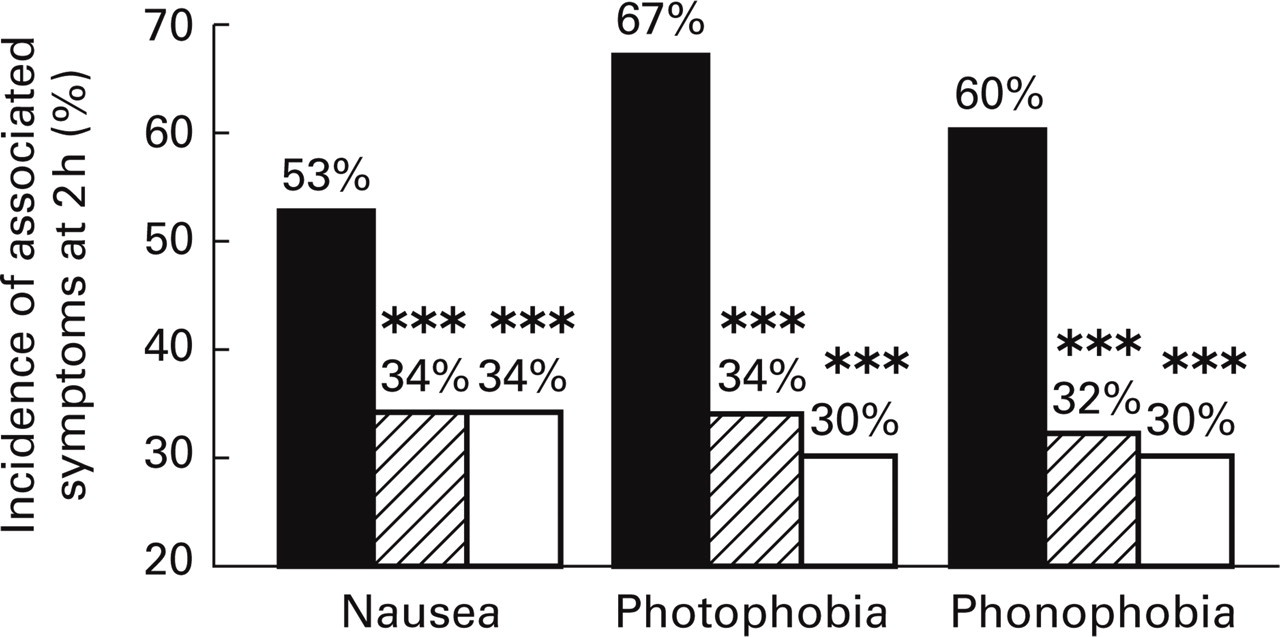
Incidence of associated symptoms at 2 h post-dosing. ▪ Placebo; (horizontal lined square) Eletriptan 40 mg; □ Eletriptan 80 mg.
Headache recurrence
Of those patients with a headache response within 2 h, headache recurrence was reported by significantly fewer patients on eletriptan 80 mg than by those receiving placebo; 21% in the 80 mg group compared with 40% in the placebo group had migraine recurrence within 24 h after dosage for the first attack (P < 0.01) (Fig. 5). The recurrence rate on eletriptan 40 mg (30%) was also lower than for the placebo group, but did not reach statistical significance. A post hoc analysis revealed that significantly fewer patients treated with eletriptan 80 mg experienced headache recurrence compared with those treated with eletriptan 40 mg (P < 0.005). The median time to recurrence was substantially longer after both eletriptan 40 mg and 80 mg than placebo (16.7 and 19.0 h, respectively, vs. 5.0 h).

Headache recurrence rates. A headache recurrence was defined as return of moderate or severe headache within 24 h of dosing in patients who had shown a response (mild or no headache) at 2 h after the first dose. Numbers of patients (n) represent patients with a headache response at 2 h who had an assessment (∗∗P < 0.01, eletriptan vs. placebo).
Use of a second dose
Subjects were allowed to take a second dose of the study drug for either non-response to the initial dose or for treatment of headache recurrence. In the first attack, fewer patients in the eletriptan 40 mg and 80 mg groups (52% and 45%, respectively) took a second dose compared with patients in the placebo group (86%). The mean number of doses of study medication used in 24 h to treat an attack was lower for both eletriptan 40 mg (1.52) and 80 mg (1.44) compared with placebo (1.86), although no formal statistical analyses were performed for these data.
Use of rescue medication
Rescue medication could be used 2 h after a second dose of eletriptan taken for non-response (at 2 h after first dose) or for headache recurrence (between 2 h and 24 h after first dose). In the first attack, 21% of patients taking eletriptan 40 mg and 17% of those taking 80 mg eletriptan took rescue medication, compared with 57% of patients taking placebo. These differences were statistically significant (P < 0.05).
Efficacy across all attacks
Within each eletriptan dose group, response rates were comparable across all three attacks. In each attack, 57–62% of patients taking 40 mg and 61–65% of those taking 80 mg eletriptan reported headache response at 2 h after treatment (Fig. 6a). Similar proportions of patients were able to resume their activities by this time-point (56–58% for 40 mg and 58–62% for 80 mg eletriptan) (Fig. 6b).
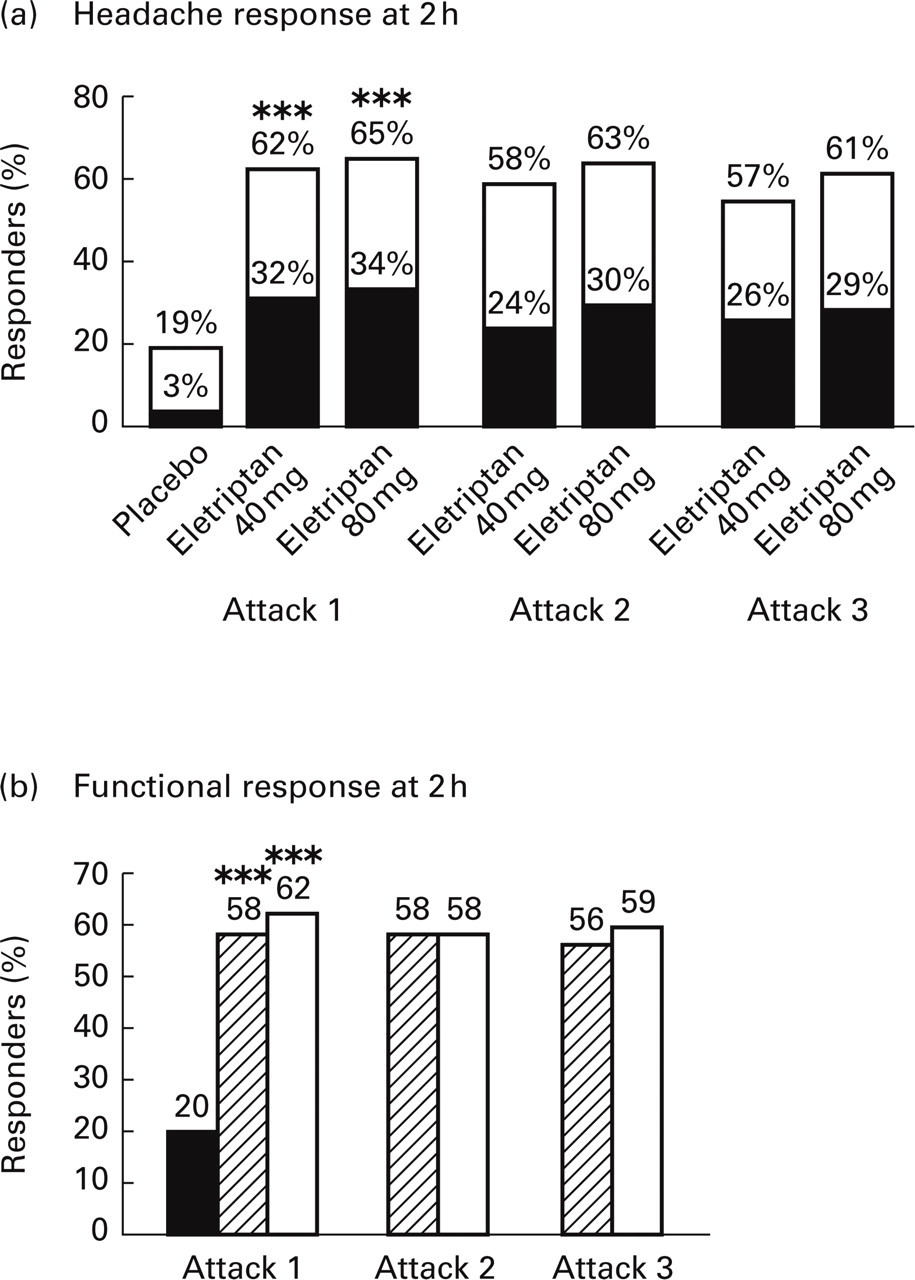
(a) Headache response rates at 2 h and (b) functional response rates at 2 h across all three treated attacks. Headache response is defined as a change from moderate to severe pain at baseline (pre-dose) to mild pain (□) or no pain (▪) at 2 h. Functional response is defined as a change from severe functional impairment or bed rest at baseline (pre-dose) to normal function or reduced activities at 2 h. ▪ Placebo; (horizontal lined square) Eletriptan 40 mg; □ Eletriptan 80 mg. ∗∗∗P < 0.0001, eletriptan vs. placebo (statistical analyses were only performed for the first attack data).
Safety and tolerability
The incidence of adverse events is presented in two ways: during the first 2 h after the initial dose (Table 3), and up to 7 days after the study medication was taken. If another attack occurred before 7 days then the window for an adverse event from the previous dose was closed and a new one was begun. Treatment-emergent adverse events reported over 24 h provide a more definitive assessment of treatment tolerability, but interpretation of the results is complicated in a substantial proportion of patients by the use of a second dose of study medication, as well as by use of rescue medication. Examination of all-causality adverse events using both methods of evaluation showed a modest increase for both doses of eletriptan compared with placebo. The incidence of adverse events for each dose of eletriptan showed a small but progressive reduction following treatment of the second and third attack compared with the incidence reported during the initial treated attack (Table 3).
Frequency of adverse events reported across three attacks∗

∗Frequency shown is all causality in the first 2 h after the first dose used to treat each attack; all events with incidence ≥4% in any treatment group are shown.
The most frequently reported treatment-related adverse event was asthenia. Other commonly reported adverse events reported by subjects taking eletriptan included dizziness and somnolence. While the types of adverse events were generally similar in the placebo group, the frequency was lower. Most adverse events were mild-to-moderate in intensity and transient. The incremental increase in adverse events observed on the 80 mg dose of eletriptan compared with the 40 mg dose was modest, ranging from 1% to 9%. There was one serious adverse event judged by the investigator to be treatment-related after a patient received four 80-mg doses during three attacks over a 24-day study period. A 50-year-old woman was hospitalized for 4 days because of transiently elevated liver function. Tests for hepatitis A, B and C were found to be negative but 10 days later she was found to be positive for Epstein Barr virus. The incidence of adverse events for each dose of eletriptan, both for all causality and treatment-related events, was substantially lower following treatment of the second and third attack than those observed during the initial treated attack.
The percentage of patients who discontinued due to adverse events was low in all three treatment groups. Overall, 20 patients (2%) discontinued during the course of treatment across three attacks due to adverse events that were felt to be treatment-related: 12 patients (3%) after taking a dose of eletriptan 80 mg, six patients (1%) after taking a dose of eletriptan 40 mg, and two patients (1%) after taking a dose of placebo. The most commonly reported adverse events leading to withdrawal were nausea, asthenia and chest symptoms, either alone or in association with other events. With the exception of asthenia reported by one patient in the placebo group, all events resolved following withdrawal.
There was no evidence of any clinically significant trends for changes in blood pressure, pulse rate, ECG or laboratory safety variables.
Acceptability of treatment
Treatment acceptability for patients taking one or two doses was high and significantly better after either eletriptan 40 mg or 80 mg than placebo (78% and 83% vs. 38%, P < 0.001 for each analysis).
Discussion
The results of this large multinational, multicentre study demonstrate that a single dose of oral eletriptan 40 mg or 80 mg is a highly effective, fast-acting and well-tolerated acute treatment for a migraine attack of moderate-to-severe intensity.
Variations in the placebo response can make comparison between studies of anti-migraine drugs difficult. The placebo-subtracted response or ‘therapeutic gain’ can be used to make a more meaningful comparison. The headache responses observed in this study translate into a high therapeutic gain for both doses of eletriptan: 43% for 40 mg and 46% for 80 mg eletriptan. These are among the highest therapeutic gains reported for 5-HT1B/1D agonists at the 2 h time-point (13). Previous studies have reported therapeutic gains with eletriptan 80 mg ranging from 33% to 53%, rizatriptan 10 mg from 32% to 41%, zolmitriptan from 19% to 34%, sumatriptan 100 mg from 26% to 33% and naratriptan 2.5 mg from 15% to 21% (14, 15). Placebo-subtracted headache response rates at the 1 h time-point were 24% for both doses of eletriptan.
The proportion of patients who were able to resume at least some daily activities at each time-point closely reflected the proportion reporting symptom relief. More patients on eletriptan 40 mg or 80 mg reported a functional response at 0.5 h, 1 h and 2 h compared with patients on placebo, although formal statistical analyses were only performed on the 2 h time-point data for this parameter (58% and 62% for 40 mg and 80 mg, respectively, vs. 20% for placebo; P < 0.0001 for each).
These findings reflect those from an earlier double-blind, phase II trial in which a statistically significant headache response vs. placebo was observed by 1 h following oral dosing with eletriptan 40 mg or 80 mg (3). Placebo-adjusted headache response rates at 1 h and 2 h after treatment in these trials were also comparable. A rapid onset of action, as observed for eletriptan in both of these studies, is consistent with the favourable pharmacokinetic profile of eletriptan: modest lipophilicity (16), good bioavailability (9) and rapid absorption following oral dosing in healthy volunteers (Tmax 1 h) (9). The potential for a rapid onset of action is particularly important for an acute migraine treatment, as patients rate the ability to provide rapid symptom relief as the most desirable attribute of an anti-migraine drug (17, 18).
The results of the present study also suggest that eletriptan has the ability to provide long-lasting relief from the symptoms of an acute attack of migraine. In those patients who had a headache response within 2 h of treatment, a lower incidence of headache recurrence was reported by patients receiving either dose of eletriptan, although only the rate in the 80-mg eletriptan group (21%) was statistically significant compared with the rate in the placebo group (40%). Furthermore, the time to recurrence in those patients who did experience it was considerably longer in both eletriptan groups (16.7 h and 19.0 h for 40 mg and 80 mg, respectively) than in the placebo group (5.0 h), although no formal statistical analyses were performed for this parameter. The comparative results with placebo should be interpreted with caution, as differential recurrence rates for study drug vs. placebo are perhaps the least reliable acute migraine outcome measures, as the small sample size of responders in the placebo group, and fluctuations in headache severity in this group, may yield variable results.
The recurrence rates observed following eletriptan treatment in the present trial compare favourably with those reported in short-term trials of other 5-HT1B/1D agonists that have shorter half-lives than eletriptan. Recurrence rates of 41% to 47% following 10 mg rizatriptan have been reported in a range of studies (19, 20), while recurrence rates following zolmitriptan doses of 2.5 mg and 5 mg ranged between 35% and 40% in a series of trials (21). In each study these recurrence rates were similar to those observed in the placebo groups. Although direct comparative studies are required before conclusions can be drawn about the relative merits of these drugs with respect to recurrence, it is possible that the longer pharmacokinetic half-life of eletriptan (4–5 h) and its relatively slow dissociation rate from the 5-HT1D receptor (4) are responsible for the lower incidence and delayed onset of headache recurrence rates observed in the study. In this respect, it is interesting that certain doses of naratriptan, with a half-life of 6 h (22), appear to be associated with low rates of headache recurrence in some studies (23), although the primary efficacy of naratriptan is relatively low. It should be noted that a linear dose response for headache recurrence has not been established for naratriptan. For example, Dahlöf et al. reported that naratriptan 0.5, 1.0, 2.5, 5 and 10 mg produced a u-shaped headache recurrence rate curve with respective rates of 25%, 31%, 41%, 21% and 27%. In this study, headache recurrence was actually highest at the optimal dose of 2.5 mg. However, no statistical analysis was conducted on these results (24).
Most data relating to consistency of efficacy of 5-HT1B/1D agonists come from open design, long-term extension studies. Because of its design, the present double-blind placebo-controlled study could not accurately measure consistency. However, it should be noted that the proportions of patients reporting a headache response or functional response in each individual attack were comparable.
Both doses of eletriptan were generally well-tolerated in this study and adverse events were rarely severe. The incidence of treatment-related adverse events was somewhat higher with eletriptan 80 mg than with eletriptan 40 mg, consistent with previously published data (3). However, treatment acceptability of eletriptan was similarly high for both doses, at about 80%. Treatment acceptability, or willingness to use the therapy again, can be viewed as a global measure of the risk : benefit ratio of a drug; the high acceptability of the 80 mg dose in this study, despite the higher incidence of adverse events, suggests that it provides additional efficacy benefits over the 40 mg dose.
Footnotes
Acknowledgements
Financial assistance for this study was provided by Pfizer Central Research, Sandwich, Kent, UK.
Appendix: Clinical investigators
The principal clinical investigators were as follows:
Australia: R Burns, Bedford Park; R Stark, Prahran; J Joubert, Richmond; J Karrasch, Kippa-Ring.
Belgium: J Schoenen, Liegne; G Monseu, Braine L'Alleud; J Jacquy, Charleroi.
Czech Republic: P Docekal, Prague; D Fibich, Prague; O Keller, Praha; E Havrdova, Praha.
Denmark: J Olesen, Glostrup; P Arlien-Soborg, Hvidovre; K Jensen, Hilleroed; TS Jensen, Esbjerg; KM Olsen, Esbjerg.
France: JM Senard, Toulouse; H Massiou, Paris; M Lanteri-Minet, Nice; C Sereni, Paris; S Donnadieu, Paris.
Germany: A Beckmann-Reinholdt, Konigstein; JP Jansen, Berlin; H Gobel, Kiel; HD Beikufner, Eckernforde; C Albert, Hannover; H Hoffmann, Bad Schwartau; S Strache, Berlin; M Foh, Fulda; L Ost-Muller, Ottweiler; P Franz, Berlin; J Alder, Ludwigshafen; H Hornlein-Rummel, Oldenburg; R Lubbe, Osnabruck; B Steinberg, Hamburg.
Hungary: G Gacs, Budapest; I Szirmai, Budapest.
Israel: R Hering, Kfar Saba; A Reches, Jerusalem; A Kuritzky, Petah-Tikvah; A Korczyn, Tel Aviv.
Italy: G Di Battista, Roma; C Firenze, Perugia; M Farinelli, Arenzano; C Benedetto, Torino; R Viviani, Torino; GC Manzoni, Parma; F Facchinetti, Modena; G Sandrini, Pavia.
Netherlands: E Siebenga, Gouda; LJMM Mulder, Rotterdam; PMM Van Erven, Breda; RJW Witteveen, Liederdorp.
Norway: I Monstad, Elverum; K Nestvold, Nordbyhagen; LJ Stovner, Trondheim; JP Larsen, Stavanger.
Poland: J Kwasucki, Warsaw; ML Wender, Poznan; L Klimczak, Warsaw.
South Africa: N Shah, Durban; MK Baker, Pretoria; H Staub, Durban.
Spain: S Gimenez-Roldan, Madrid; M Noya, Santiago de Compostela (Coruna); E Diez Tejedor, Madrid.
Sweden: C Dahlöf, Göteborg; S Boes Hansen, Växjö; A Henriksson, Luleå; CG Cederberg, Vällingby.
United Kingdom: TJ Steiner, London; LJ Findley, Romford; AJ Dowson, Guildford; JD Gibson, Plymouth; JN Blau, London; PJ Goadsby, London.
