Abstract
The aim of this study was to examine efficacy and tolerability of eletriptan in patients switched from barbiturate-containing combinations (Fiorinal®, Fioricet®). Migraineurs (n = 160) meeting IHS criteria, with unsatisfactory response in the past year to butalbital-containing combinations, treated up to 16 attacks over 3 months with eletriptan 40 mg. Assessments included headache response and pain-free rates and functional impairment at baseline and 2 h postdose, and global ratings of treatment satisfaction at 24 h. At 2 h postdose, average headache response and pain-free rates were 71% (95% CI, 69-74%) and 37% (95% CI, 35-40%), respectively; 68.5% of patients (95% CI, 65-72%) reported functional response. Within-patient analysis found no efficacy diminution over time (no tolerance). Average headache recurrence rate was 20% (95% CI, 18-23%). Eletriptan was well-tolerated; 6 (3.7%) patients discontinued due to adverse events. There were no serious treatment-related adverse events. We conclude that in poor responders to butalbital-caffeine combinations, switching to eletriptan 40 mg was well-tolerated and efficacious.
Introduction
Rational diagnosis and treatment of migraine depend on operational diagnostic criteria, which were introduced in 1988, and subsequently revised (1), and the use of treatments that have demonstrated efficacy and tolerability in randomized clinical trials (RCTs) that are double blind and placebo controlled (2). The advent of standardized diagnostic criteria and the proliferation of RCTs during the past decade have made it possible to establish evidence-based treatment guidelines (3). Despite the dissemination of these guidelines (4), a wide gap remains between recommendations for treatment and actual clinical practice (5, 6).
Of special concern are migraine treatments that continue to be widely used despite minimal evidence of efficacy based on well-controlled RCTs. Important examples are the barbiturate-containing combination products, such as Fiorinal® (aspirin, butalbital and caffeine), and Fioricet® or Esgic® (acetaminophen, butalbital and caffeine). In a recent US epidemiological survey (7), butalbital-containing combination products were the single most frequently prescribed therapy for migraine, used by 36% of patients taking prescription medicines for their headaches.
No placebo-controlled RCTs have ever been reported demonstrating the efficacy of Fiorinal, Fioricet or other barbiturate-containing drugs in the treatment of migraine and only one positive trial has been reported for Fiorinal with codeine (3, 8). Consequently, migraine treatment guidelines have assigned these combination products the lowest possible efficacy rating (3). Further complicating this clinical picture is the extent to which butalbital-containing analgesics may be associated with tolerance, physical dependence, and withdrawal reactions (9, 10); adverse behavioural effects (11) and overdose risk (12).
It should be noted that butalbital is not the only problematic ingredient in Fiorinal/Fioricet combination products. Regular use of caffeine has been associated not only with tolerance but also with withdrawal reactions in which headache is the most prominent clinical feature (13, 14). Butalbital- and caffeine-containing combination products are common treatments in patients diagnosed with chronic daily headache, but the extent to which these agents may contribute to the pathogenesis of this frequent condition (prevalence, 4–5%) remains to be fully characterized, both epidemiologically and pathophysiologically (15–18).
Given the wide use of barbiturate-containing drugs such as Fiorinal and Fioricet in the United States, and their unproven benefit in migraine, switching patients to migraine-specific triptans may be expected to occur with increasing frequency as practice guidelines are implemented more fully. And yet, no study that systematically examines the efficacy and tolerability of making this switch has been published.
Eletriptan is a newer triptan with rapid and consistent absorption, high oral bioavailability, and potent agonist activity at 5-hydroxytryptamine (5-HT1B/1D) receptors (19–21). Eletriptan has shown high response rates in patients who were poor responders to both nonsteroidal anti-inflammatory drugs (NSAIDs) (22) and sumatriptan (23).
The goal of the current study was to evaluate the effectiveness of eletriptan as an acute treatment for migraine in patients who were currently being treated with butalbital-caffeine combination products and who reported unsatisfactory results using these drugs. We report the results of an open-label, multiple-attack study designed to mirror clinical practice and to provide systematic information on the efficacy and tolerability of switching to eletriptan in this patient group of poor responders to barbiturate-containing drugs such as Fiorinal/Fioricet.
Patients and methods
Patients
Men and women aged 18–50 years were eligible to enter the study if they met International Headache Society (IHS) criteria for migraine, with or without aura, for more than 1 years (1), and reported experiencing between two and eight migraine headaches per month, with impairment in usual activities occurring during a typical migraine. Inclusion in the study was limited to patients who were treated in the past year with aspirin/butalbital/caffeine or acetaminophen/butalbital/caffeine combination therapy (Fiorinal and/or Fioricet or the generic equivalent) and who failed to achieve subjective relief of their migraines with this treatment. A treatment failure of butalbital-containing medication was ascertained by patient self-report and could indicate that it was not efficacious or possessed intolerable side-effects. Such a definition of ‘treatment failure’ was designed to mimic the typical practice of switching from one abortive medication class to another.
Patients were excluded from the study if one or more of the following criteria were present:
migraine headaches that failed to respond to two or more types of headache medications, or adequate trials of two or more previous triptans;
atypical migraines or migraine with prolonged aura, familial hemiplegic migraine, basilar migraine, migrainous infarction, aura without headache or less than 24 h of freedom from headache between attacks;
more than six nonmigrainous headaches per month or chronic daily headache;
contra-indications to the use of eletriptan or other triptans, including coronary artery disease, peripheral vascular disease, significant arrhythmias, heart failure or uncontrolled hypertension (blood pressure > 150/95 mmHg);
concomitant use of potent CYP3A4 inhibitors;
any clinically significant medical illness or electrocardiogram (ECG) or laboratory abnormalities;
severe reduction in gastrointestinal absorption (e.g. gastrectomy);
evidence of alcohol or substance abuse and medication overuse of analgesics or ergotamine (>100 pills per month);
women who were pregnant [confirmed by serum human chorionic gonadotropin beta-subunit (β-HCG) at screening], breast-feeding or not using a medically accepted form of contraception if sexually active and potentially fertile.
The study was conducted at 12 headache centres in the United States and was approved by the local ethics committee, in accordance with the Declaration of Helsinki. Patients were recruited from the practices of each investigator. Whether eligible patients were recruited sequentially was not recorded. All patients provided written informed consent.
The screening medical evaluation consisted of a physical examination, vital signs, 12-lead ECG and laboratory testing, including blood chemistries, complete blood count with differential and serum β-HCG.
Study design
This was an open-label, switch study of patients reporting inadequate response to barbiturate-containing drugs such as Fiorinal or Fioricet. Patients treated up to 16 migraine attacks over 3 months with eletriptan 40 mg. Patients were instructed to take study medication when they experienced a typical migraine attack of moderate or severe intensity that was not improving. Patients took study treatment when the aura phase had ended, and within 4 h of the onset of headache pain. Treatment with study medication was not permitted if the patient had used an analgesic or antiemetic during the previous 6 h or had taken another triptan or ergotamine-containing or ergot-type medication (e.g. dihydroergotamine) during the previous 24 h. If necessary to treat nonresponse, patients were permitted to take rescue medication, provided that at least 2 h had elapsed since the second dose and provided that the rescue drug did not include a triptan, ergotamine-like compounds, methysergide or Midrin® (isometheptene mucate, dichloralphenazone and acetaminophen).
Patients recorded migraine-related symptoms in a diary at baseline (immediately predose) and at 2 and 24 h postdose. Use of rescue medications was also recorded in the diary. Patients were asked to return for a final assessment at the study centre within 14 days of the index attack.
Evaluation of efficacy
Efficacy parameters consisted of the following: (1) headache response; (2) pain-free response; (3) pain relief, rated at 2 h on a five-point scale (0, complete relief/100%; 1, a lot of relief/75%; 2, some relief/50%; 3, a little relief/25%; 4, no relief); (4) functional impairment, rated at 2 h on a four-point scale (1, normal; 2, some impairment; 3, severe impairment; 4, bed rest); (5) subjective satisfaction with eletriptan treatment, assessed at 2 h on a six-point scale (1, extremely satisfied; 2, very satisfied; 3, satisfied; 4, neither satisfied or dissatisfied; 5, somewhat dissatisfied; 6, very dissatisfied); (6) work productivity, assessed at visits two and four using the Migraine-Related Work Productivity Questionnaire (WPQ) (24); (7) health-related quality of life (QOL) using the Migraine-Specific Quality of Life Questionnaire (MSQ) (25).
Statistical analysis
Descriptive statistics were performed on the baseline characteristics of the sample. No inferential statistics were necessary because this was an open-label study without a control group. In addition to analysing data from the efficacy parameters, the following four derived end points were calculated:
Headache recurrence, defined as the return of a moderate to severe headache (from a previously improved level of mild or no headache) between 2 and 24 h after ingestion of study medication;
Sustained headache response, consisting of patients who achieved a headache response at 2 h, had no recurrence, and did not use additional antimigraine medications for 24 h;
Sustained pain-free, consisting of those patients who were pain-free at 2 h, had no recurrence, and did not use additional antimigraine medications for 24 h;
Functional response, defined as improvement from a baseline impairment of severe/bed rest, to ‘mild impairment’ or ‘normal level of functioning’.
Consistency of response was analysed as the per cent of patients achieving headache response at 2 h in 1/3, 2/3, 3/3 attacks for participants who treated the first 3 attacks.
Results
Baseline characteristics
Dates of the study were from 6 December 1999 to 6 November 2000. A total of 171 patients were screened, 160 of whom received at least one dose of study medication and thus constitute the ITT sample. The majority (85%) of study patients were women, with a mean age of 37 years (range, 18–57 years). Patients were predominantly white (88%) and well-educated, with 83% having at least some college education. Forty-six per cent were currently married and 23% were divorced, separated, or widowed. Mean duration since diagnosis of migraine was 15.5 years. Patients reported having a mean of approximately 4 migraine attacks per month, with 29% experiencing an aura prior to at least some of their attacks. Overall, a total of 1485 migraine attacks were treated with eletriptan, which is a mean (± SD) of 10.0 ± 4.6 attacks per patient. Forty per cent (63/156) of patients treated 12 or more attacks with eletriptan during the course of the study, while 59.6% (93/156) of patients treated less than 12 attacks. The 63 individuals reporting high attack frequency accounted for 59.5% (884/1485) of all treated attacks.
Headache response and pain-free rates
The average headache response rate for eletriptan 40 mg at 2 h, averaged across all treated attacks, was 71%[95% confidence interval (CI) of 69–74%] for all 1485 treated attacks. Figure 1 summarizes headache response and pain-free rates for each treated attack. Among the subgroup of patients with frequent attacks, defined as ≥12 attacks in the 12-week study period, the average 2-h headache response rate was 76% (670/884; 95% CI, 73–79%). For patients treating less than 12 attacks, the average 2-h headache response rate was 65% (388/601; 95% CI, 61–68%). Within-patient consistency was analysed in patients (n = 129) with 3-attack data available. On this analysis, response on eletriptan was achieved by 88% (113/129) of patients in 1/3 attacks, 67% (87/129) in 2/3 attacks, and 44% (57/129) in 3/3 attacks.

Proportion of patients reporting headache (□) and pain-free (▪) response at 2 h postdose of eletriptan 40 mg: by attack data. ∗Number of patients with available data.
Among the total population, the average pain-free rate at 2 h was 37% (551/1485; 95% CI, 35–40%). Similarly, the average pain-free rate among study patients experiencing frequent (≥12) attacks was 41.5% (367/884; 95% CI, 38–45%). For patients treating fewer than 12 attacks, the pain-free rate was 31% (184/601; 95% CI, 27–34%).
Functional response
Eletriptan treatment was associated with rapid improvement in functioning, with functional response rates at 2 h, averaged across all treated attacks of 69% (95% CI, 65–73%). Figure 2 summarizes the functional response rate at 2 h for each treated attack.

Functional response at 2 h postdose of eletriptan 40 mg: by attack data. ∗Number of patients with available data.
Tachyphylaxis analysis
To evaluate whether tolerance developed to the therapeutic effect of eletriptan, the three-attack average headache response rate at 2 h was determined for the first three and last three attacks in the subgroup of patients who treated at least 12 attacks (n = 63). This analysis showed that average headache response rates did not decrease as treatment progressed from the first three attacks (72%; 95% CI, 64–80%) to the last three attacks (79%; 95% CI, 70–85%). Average pain-free rates also showed no decrease as treatment progressed from the first three attacks (37%; 95% CI, 30–47%) to the last three attacks (41%; 95% CI, 33–51%).
Rescue medication and sustained response
Rescue medication was used in 24% (353/1485; 95% CI, 22–26%) of all treated attacks (Fig. 3). Study patients with frequent attacks reported the same per-attack need for rescue medication [average, 21% (186/884); 95% CI, 18–24%].

Rescue medication, recurrence, sustained response and sustained pain-free response.
Overall, the average headache recurrence rate was 20% (212/1049; 95% CI, 18–23%), resulting in an average sustained headache response rate of 53% (779/1476; 95% CI, 50–55%) and an average sustained pain-free rate of 32% (468/1481; 95% CI, 29–34%) (Fig. 3).
Disposition of patients, tolerability, and safety
Of the total ITT sample of 160 patients, 34 (21.3%) discontinued treatment prematurely, 6 (3.8%) due to an adverse event. The specific adverse events cited as leading to discontinuation in each of these patients consisted of the following: meningioma (not treatment-related), fatigue, nausea, increased migraine (2 patients), multiple adverse events (fatigue, dizziness, ear pain) and death by accidental hanging. The patient who died had no history of depression or suicide attempts, and the autopsy revealed no substance abuse or use of concomitant medication. The patient died 15 days after the last known dose of study medication. In the opinion of the investigator, the event was not related to study medication.
Treatment-emergent all-causality adverse events, regardless of whether they were related to eletriptan, are shown in Table 1. All adverse events were transient, and the majority were mild to moderate in intensity.
All-causality adverse events∗

Values are percentages. All causality, with incidence ≥2%.
Hypertonia consists of muscle tension and related symptoms.
Quality of life and work productivity
The effect of study treatment on headache-related QOL was evaluated using the MSQ. Scores on the MSQ prior to treatment with eletriptan are shown in Table 2. Since the precise timing of use of Fiorinal/Fioricet treatment was not recorded, it cannot be determined whether the baseline MSQ scores specifically reflect the QOL benefit of Fiorinal or Fioricet treatment. It should also be noted that there was a gender difference in the MSQ, with men exhibiting less impairment than women at baseline (total score, 67.2 ± 17.4 vs. 55.7 ± 19.4) and after treatment (total score, 76.7 ± 17.9 vs. 63.25 ± 20.3). At the end of 3 months, treatment with eletriptan was associated with an improvement in all three MSQ dimensions, both for the combined sample (Table 2) and for each gender individually.
Effect of eletriptan treatment on MSQ scores

Treatment with eletriptan was also associated with improvement in the level of impairment on the WPQ, from a baseline score of 27.9 ± 13.8 to a post-treatment score of 25.4 ± 13.5 (P < 0.001). Changes in MSQ and WPQ were moderately correlated (Spearman coefficient, – 0.319; P < 0.001).
Patient global assessment of migraine treatment
For approximately two thirds (971/1479) of the treated attacks, patients rated themselves as satisfied to extremely satisfied with eletriptan treatment (Fig. 4). The mean satisfaction rate, calculated for each patient instead of by-attack, was 3.16 (1, extremely satisfied; 2, very satisfied; 3, satisfied; 4, neither satisfied or dissatisfied; 5, somewhat dissatisfied; 6, very dissatisfied). On average, 62% of patients reported being satisfied-to-extremely satisfied.
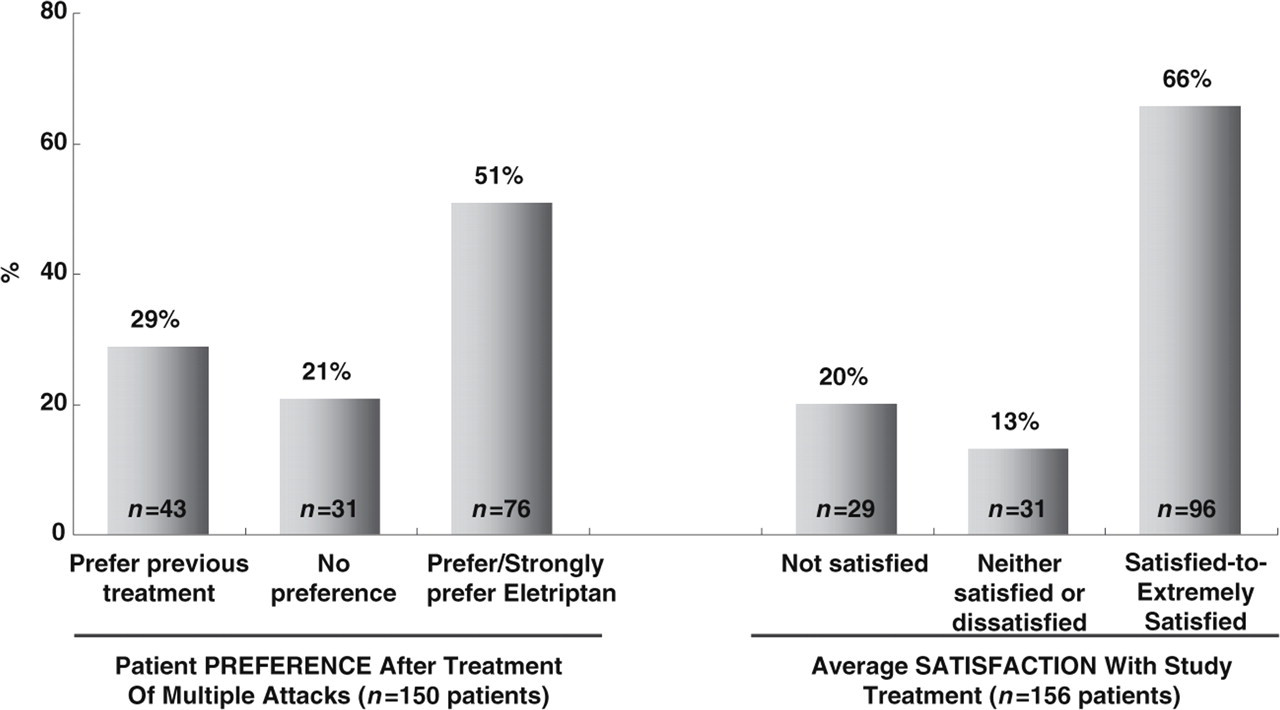
Average patient preference and satisfaction with long-term eletriptan therapy.
Discussion
In the current study, patients who were dissatisfied with barbiturate-containing drugs such as Fiorinal or Fioricet and did not achieve adequate relief with one of these combinations were switched to open-label treatment with 40 mg of eletriptan for up to 16 attacks over 3 months. Eletriptan 40 mg was a highly effective acute treatment of migraine in this patient group who reported poor response to barbiturate-containing drugs such as Fiorinal or Fioricet, with an average 2-h headache response rate of 71% and an average pain-free rate of 37%. Consistent with headache response rates, average functional response rate was 69% at 2 h. Eletriptan was well tolerated, with a low incidence of adverse events.
The goals of the current study were to provide clinicians with systematic efficacy, tolerability, and satisfaction data on switching to treatment with eletriptan in patients who were unable to achieve a satisfactory response to butalbital-caffeine-analgesic combination products; the study does not provide information on the efficacy of eletriptan in patients who overuse Fiorinal or Fioricet. We were especially interested in patient satisfaction after switching to eletriptan because patients who rely on barbiturate-containing combination products, similar to those who rely on narcotic analgesics, may become physically and psychologically dependent on these medications, making the switch to alternative therapy difficult, even if the alternative therapy has higher efficacy. The results of the current study showed an improvement in QOL measures and a high level of satisfaction with eletriptan treatment (66%) when satisfaction was assessed concurrently at 2 h postdose and was averaged across all attacks.
Overall, 21% of patients discontinued eletriptan prior to the end of 6 months of study treatment; this is in the middle of the 15–35% attrition range that has been reported in most long-term studies of triptans (26–30). Patient satisfaction with eletriptan, combined with an attrition rate that is in the expected range for this type of long-term treatment study, indicates that the majority of patients who have an unsatisfactory response to barbiturate-containing combination products will respond favourably to eletriptan.
Multiple-attack studies provide important information on consistency of response, including whether response is maintained at a high level among patients with frequent migraine attacks, and whether tolerance develops after repeated treatment (tachyphylaxis). In the current study, the average headache response rate was 71% in the total patient sample, which was similar to the average response rate of 76% reported by patients with a high attack frequency (i.e. ≥1 attack per week). Using traditional criterion for consistency, eletriptan had an 88% response rate on 1/3 attacks, a 67% response rate on 2/3 attacks, and a 44% response rate on 3/3 attacks. These results are comparable to results from a meta-analysis of triptan consistency data by Ferrari et al. (31) who reported 1/3 response rates of approximately 80–90%, 2/3 rates of approximately 60–70%, and 3/3 rates of approximately 30–40%. Rizatriptan reported higher consistency rates, but based on a very different study design.
To minimize possible bias associated with attrition and differential attack frequency over a 3-month treatment period, a within-patient tachyphylaxis analysis was performed in the subgroup of patients for whom both early and late headache response data were available and who treated at least 12 attacks. The results of this analysis confirmed that no tolerance to eletriptan therapy develops, either in terms of headache response (72% vs. 79%) or pain-free rate (37% vs. 41%). Not only was the efficacy of eletriptan sustained across attacks, but also eletriptan treatment showed a low average recurrence rate (20%) and a high average level of sustained response (53%).
A recent analysis of headache patients who were high utilizers of emergency department services found that 52% were currently using butalbital-containing analgesics (32). The majority of these patients were also using narcotics and/or benzodiazepines, suggesting a pattern of reliance on symptomatic medicines that do not specifically target underlying migraine mechanisms. The results of the current study provide preliminary evidence that patients having an inadequate response to one type of symptomatic therapy, containing butalbital, can be effectively switched to eletriptan, resulting in high rates of headache response with low risk of recurrence. It should be noted that pain-free rates on eletriptan in previous open-label studies have ranged from 48%, in a recent Excedrin® switch study (33), to 58% in a crossover comparison with subcutaneous sumatriptan (34). The lower pain-free rates (average = 37%) observed in the current study suggest that patients using Fiorinal or Fioricet represent a difficult to treat clinical subgroup. This is consistent with data showing that use of barbiturate-containing compounds is associated with an increased risk of chronic daily headache (35). Thus, the current results, which indicate that patients on Fiorinal or Fioricet can be effectively switched, and maintained, on eletriptan, are encouraging.
Finally, it should be noted that eletriptan treatment was associated with significant improvement in the level of impairment on the WPQ, from a baseline score of 27.9 ± 13.8 to a post-treatment score of 25.4 ± 13.5 (P < 0.001).
The main limitation of the current study is that it was not designed as a randomized, double-blind, placebo-controlled, head-to-head trial. Although open-label trials have been reported to produce results that are comparable to RCTs, especially those using operationally defined entry criteria and standardized assessments (35, 36), nonetheless, the current results must be interpreted with this limitation in mind. An additional study limitation is that the six-point drug satisfaction scale is not fully symmetrical. Patients wishing to state satisfaction with eletriptan are provided three options (‘satisfied’, ‘very satisfied’ and ‘extremely satisfied’), while patients wishing to record dissatisfaction are provided only two options (‘somewhat dissatisfied’ and ‘very dissatisfied’). To correct for this scaling error, Fig. 4 shows a combined satisfaction rate for eletriptan, but a scaling bias may still exist.
In conclusion, while butalbital-caffeine combination products are commonly used for the treatment of migraine, they have unproven clinical efficacy and may be associated with psychological and physical dependence. In this open-label trial, switching patients from such combination products to eletriptan 40 mg was associated with consistently high headache response rates, low recurrence rates and good tolerability and patient acceptance.
Footnotes
Acknowledgements
We would like to express our appreciation to the investigators who participated in this study: James Couch, MD (University Hospital, Oklahoma City, OK); Seymour Diamond, MD (Diamond Headache Clinic, Chicago, IL); Arthur Elkind, MD (Elkind Headache Center, Mt. Vernon, NY); Jerome Goldstein, MD (San Francisco Clinical Research Center, San Francisco, CA); David Kudrow, MD (California Medical Clinic for Headache, Encino, CA); Elizabeth Loder, MD (Spaulding Rehabilitation Hospital, Boston, MA); Vincent Martin, MD (Cincinnati Headache Center, Cincinnati, OH); Ninan Mathew, MD (Houston Headache Clinic, Houston, TX); Joel Saper, MD (Michigan Head, Pain & Neurological Institute, Ann Arbor, MI); Timothy Smith, MD (Ryan Headache Center, Chesterfield, MO); Paul Winner, MD (Premier Research Institute, West Palm Beach, FL); Roger Cady, MD (Headache Care Center, Springfield, MO).
This study was supported by a grant from Pfizer Inc.
