Abstract
This was a randomized, double-blind study designed to evaluate the comparative efficacy and tolerability of the 40-mg dose of eletriptan and the 2.5-mg dose of naratriptan. Patients (n = 548) meeting International Headache Society (IHS) criteria for migraine were randomized to treat a single migraine attack with either eletriptan 40 mg, naratriptan 2.5 mg, or placebo. Headache response rates at 2 h and 4 h, respectively, were 56% and 80% for eletriptan, 42% and 67% for naratriptan (P < 0.01 for both time-points vs. eletriptan), and 31% and 44% for placebo (P < 0.0001 vs. both active drugs at both time-points). Eletriptan also showed a significantly greater pain-free response at 2 h (35% vs. 18%; P < 0.001) as well as lower use of rescue medication (15% vs. 27%; P < 0.01) and higher sustained headache response at 24 h (38%) compared with naratriptan (27%; P < 0.05) and placebo (19%; P < 0.01). Both eletriptan and naratriptan were well tolerated. The results confirm previous meta-analyses that have suggested the superiority of eletriptan vs. naratriptan in the acute treatment of migraine.
Introduction
Migraine is a multidimensional illness with a clinical presentation that includes headache pain, nausea, photophobia, phonophobia, and impaired functioning. Improvement in each of these symptom dimensions contributes to the global effectiveness of any acute treatment. Systematic reviews of efficacy (1–3) have suggested that various triptans may have differential advantages in one or more of these outcomes, which include rapid onset of efficacy, ability to achieve a pain-free state, reduction in associated symptoms, low recurrence rate, and tolerability.
While systematic reviews and meta-analyses provide useful supportive evidence to assist physicians in making a rational choice between triptans and tailoring treatment to each patient's clinical needs, such reviews are no substitute for placebo-controlled head-to-head comparator trials. Depending on the comparator being evaluated, a triptan will face stringent tests for different clinical outcomes. For example, naratriptan, perhaps due to its longer half-life of 6 h (4), has consistently demonstrated lower recurrence rates than other triptans in head-to-head trials: 17–27% lower than sumatriptan (5, 6), and 12% lower than the 10-mg dose of rizatriptan (7). In contrast, naratriptan has a relatively slow onset of action, which may be due in part to a slower time-to-maximal concentration (Tmax) of 2–3 h, making it less effective in achieving a rapid response, but perhaps contributing to its status as one of the best-tolerated of all triptans (2).
Eletriptan is a newer triptan with rapid and consistent absorption, high bioavailability, and potent agonist activity at 5-HT1B/1D receptors (8–10). Among newer triptans, eletriptan has been the most extensively studied in double-blind, placebo-controlled head-to-head comparator studies. To date, eletriptan has demonstrated superior efficacy in 3 head-to-head trials vs. sumatriptan (11–13), and one each vs. Cafergot® (14) and zolmitriptan (15). The efficacy advantage of eletriptan compared to sumatriptan, zolmitriptan, and Cafergot® appears to be consistent across most of the key clinical outcome dimensions of migraine, including rapid headache response, relief of associated symptoms, functional response, and the ability to sustain response. The current study was designed to evaluate the efficacy of the 40-mg dose of eletriptan compared with naratriptan 2.5 mg at acute time-points up to 4 h postdose (the primary endpoint used in the naratriptan development program). Because of the low headache recurrence rate and low adverse event profile associated with naratriptan treatment, we were especially interested in assessing the comparative ability of the standard 40-mg starting dose of eletriptan in the additional areas of minimizing recurrence, achieving sustained response, and yielding high overall levels of patient satisfaction.
Patients and methods
Patients
Male or female adults, aged 18–80 years, were eligible for study entry if they met International Headache Society (IHS) criteria for migraine with or without aura (16) and reported a minimum of 1 acute migraine attack every 6 weeks. Patients were excluded from the study if they reported (1) coronary artery disease, heart failure, uncontrolled hypertension or abnormal ECG; (2) frequent migraine or concomitant nonmigrainous headache (>6 per month), migraine variants (e.g. familial hemiplegic or basilar migraine), and/or migraines which, in the clinical judgement of the investigator, had consistently failed to respond to adequate medical therapy; (3) hypersensitivity or known contra-indication to treatment with eletriptan or naratriptan; (4) concomitant use of potent CYP3A4 inhibitors or use of MAO inhibitors in the 2 weeks prior to study entry; (5) any clinically significant medical illness or laboratory abnormalities; (6) severe reduction in gastrointestinal absorption; (7) misuse or abuse of alcohol or other substances, including analgesics or ergotamine; (8) use of any experimental drug within the past month; (9) (if female) current pregnancy, breast-feeding, or not using a medically accepted form of contraception.
The study was conducted at 42 centres in the UK and Latin America according to the standards set forth in the Declaration of Helsinki (1996 revision) and consistent with all ICH Good Clinical Practice guidelines. The protocol was approved by an Ethics Review Committee at each site. Study procedures were explained to prospective patients and written informed consent was obtained prior to study entry. The screening medical evaluation consisted of a physical examination, vital signs, 12-lead ECG, and urine pregnancy testing.
Study design
This study was a double-blind, placebo-controlled, parallel-group, outpatient study with patients randomized in a 2 : 2 : 1 ratio to treat one attack with either eletriptan 40 mg, naratriptan 2.5 mg, or placebo. A double-dummy design was used, with each patient taking one tablet (either eletriptan 40 mg or matching placebo), and one capsule (either naratriptan 2.5 mg or matching placebo). Blinded naratriptan was shown to be equivalent to the commercial tablet based on the results of an in vitro dissolution study conducted in deionized water (data on file, Pfizer Inc, New York, USA), as well as a bioequivalence study in which AUC and Cmax values were within standardized norms in healthy subjects (data on file, Pfizer Inc). In addition to bioequivalence, naratriptan used in the current study was clinically equivalent to commercial naratriptan 2.5 mg as demonstrated by comparison of current study results to those reported in the naratriptan US label (USPI) for the key efficacy parameters of headache response at 4 h (60–66% across 3 studies in the USPI vs. 67% in the current study).
Patients were instructed to take study medication when they experienced a typical migraine attack of at least moderate severity, and within 4 h of the onset of headache pain. Patients who experienced aura were instructed to wait until the aura phase ended before taking study treatment. Treatment with study medication was not permitted if the patient had used an analgesic in the previous 4 h, or had taken another triptan or ergotamine-containing or ergot-type medication (e.g. dihydroergotamine) in the previous 48 h.
Patients volunteered adverse events in response to investigator questioning at the follow up visit, and these were recorded on the case report forms. Adverse events were recorded regardless of whether, in the judgement of the investigator, they were considered to be related to study treatment (this comprised the ‘all-causality’ total of adverse events recorded). Use of rescue medications was also recorded in the diary.
Patients were permitted to take rescue medication if they failed to achieve a headache response (defined below) by 4 h postdose. They were not permitted to take any other triptan, ergotamine, or ergotamine-like substance for 24 h postdose. Patients who achieved a 2-h headache response, but experienced a recurrence of moderate to severe headache, were permitted to take a second dose of study medication, provided that at least 4 h had elapsed since the first dose was taken. The time of recurrence and the second dose, and information on rescue medication, were noted in the diary card. Headache intensity was recorded immediately prior to taking the second dose of study medication. Rescue medication was permitted 2 h after this second dosing if needed. Within 48 h of study treatment, the patient was asked to contact the investigator or his representative to review adverse event information, and to schedule the final appointment, which took place within 14 days of the index attack.
Evaluation of efficacy
The
Statistical analyses
The primary comparison was between eletriptan 40 mg and naratriptan 2.5 mg, and consisted of the proportion of subjects with a headache response at 2 h after the first dose of study treatment for the migraine attack. The study was powered to detect as significant a 15-point difference in 2-h headache response rates between eletriptan and naratriptan. It was estimated that a sample size of at least 182 subjects per treatment group would be required to ensure 80% power based on a 2-tailed comparison, and with an alpha of 0.05. Assuming that 80% of randomized patients would meet all eligibility criteria, and have efficacy data available, a total of 550 patients were needed for enrolment.
Baseline characteristics of the sample were compared for homogeneity across treatment groups. All efficacy analyses were performed on the ITT sample, defined as all subjects who took at least one dose of study medication and had a valid baseline and at least one postbaseline evaluation. The primary analysis was the 2-h headache responder rate for the ITT group. This analysis was conducted using a categorical linear model based on the SAS procedure CATMOD, which included terms for treatment and baseline severity. In case of a statistically significant between-treatment difference at baseline, adjustments were made. Secondary endpoints were analysed using a categorical linear model based on SAS CATMOD procedure, which includes terms such as treatment and baseline severity.
All statistical tests of significance were performed at the 5% level of significance, and were 2-sided tests (unless otherwise specified). No adjustment was made for multiple comparisons. Pair-wise comparisons were not done unless the overall comparison of treatment groups was significant.
Results
Baseline characteristics of patients
A total of 548 patients were randomized to one of three groups and 483 patients (88%) received treatment for a single migraine headache (Fig. 1). Demographic and clinical characteristics were similar across each treatment group (Table 1), and were broadly similar to the clinical profile of patients entering studies for acute treatment of migraine.
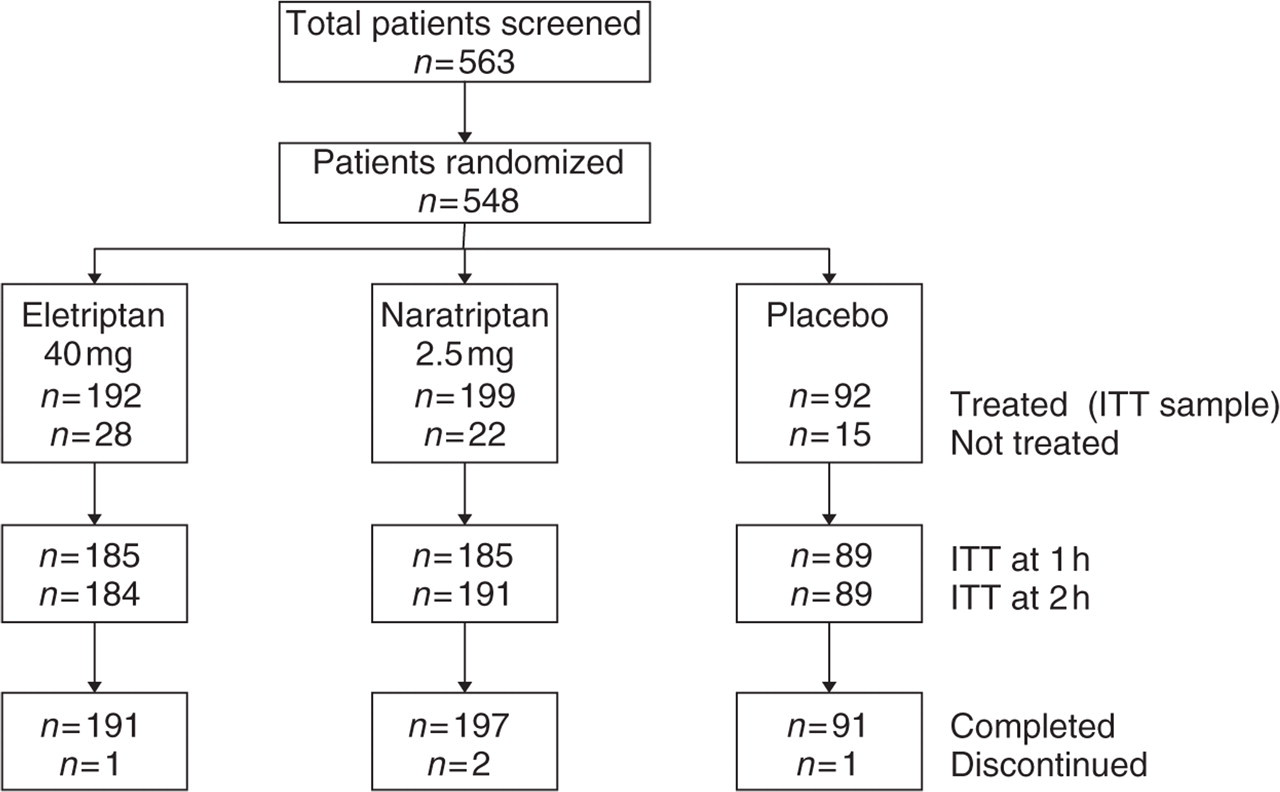
Patient disposition.
Clinical characteristics of the patient sample

∗Time since first diagnosis.
†Average over 3 months prior to study entry.
Headache response and pain-free response
Headache response at 2 h, the primary efficacy variable, was significantly higher on eletriptan compared to naratriptan (56% vs. 42%, P < 0.01; Fig. 2). Headache response rates at 2 h were both significant vs. placebo for eletriptan (P < 0.0001) and for naratriptan (P < 0.05).

Headache response rates 0.5 h to 4 h postdose. • Eletriptan 40 mg, ▪ Naratriptan 2.5 mg, ○ Placebo. Eletriptan/naratriptan vs. placebo ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001; Eletriptan vs. naratriptan † P < 0.05, †† P < 0.01.
Headache response was also significantly higher for eletriptan at 1 h and 4 h, respectively, compared with both naratriptan (34% vs. 25%, P < 0.05; 80% vs. 67%, P < 0.01) and placebo (21%, P < 0.01; 44%, P < 0.0001; Fig. 2). Headache response rates were not significantly different from placebo at 30 min for either eletriptan (12% vs. 5%; P = 0.063) or for naratriptan (9%; P= 0.391 vs. placebo).
Eletriptan showed higher pain-free rates (Fig. 3) at both 2 and 4 h (35% and 56%) compared with both naratriptan (18%; P < 0.001 and 41%, P < 0.01) and placebo (19%, P < 0.001; 24%, P < 0.0001). By 1 h, pain-free rates were significantly higher for eletriptan (12%) compared with naratriptan (6%; P < 0.05). Pain-free response for naratriptan was significantly higher than placebo at 4 h (P < 0.01) but not at 2 h.

Pain-free rates 0.5 h to 4 h postdose. • Eletriptan 40 mg, ▪ Naratriptan 2.5 mg, ○ Placebo. Eletriptan/naratriptan vs. placebo ∗P < 0.05, ∗∗P < 0.01; Eletriptan vs. naratriptan †† P < 0.01, ††† P < 0.001.
Consistent with the high 4-h headache response rate, only 15% of patients on eletriptan used rescue medication (Fig. 4) compared to 27% taking naratriptan (P < 0.01), and 49% on placebo (P < 0.0001).

Use of rescue medication and sustained headache response at 24 h. □ Placebo, ░ Naratriptan 2.5 mg, ▪ Eletriptan 40 mg. Eletriptan/naratriptan vs. placebo ∗P < 0.05; ∗∗P < 0.01; Eletriptan vs. naratriptan †P < 0.05.
Headache recurrence and sustained headache and pain-free response
Among patients who achieved a 2-h headache response, headache recurrence rates were consistently low for eletriptan (29%), naratriptan (26%), and placebo (28%), with no significant differences among the 3 treatment groups. Similarly, the proportion of patients taking a second dose of study medication for headache recurrence was lower for eletriptan and naratriptan (19% and 18%, respectively) than for placebo (26%).
The proportion of patients reporting sustained headache response at 24 h was significantly higher for eletriptan (38%; Fig. 4) compared with both naratriptan (27%; P < 0.05) and placebo (19%; P < 0.01). The difference in sustained response was not significant for naratriptan vs. placebo.
The proportion of patients reporting a sustained pain-free response at 24 h was significantly higher for eletriptan (22%) compared with both naratriptan (11%; P < 0.05) and placebo (12%; P < 0.05).
Nausea
Absence of nausea at 2 h (Fig. 5) was not significantly different for eletriptan (73%) compared with naratriptan (68%; P= 0.09) or placebo (66%; P= 0.07). The frequency of vomiting was too low for meaningful analysis.

Absence of nausea and functional status at 2 h. □ Placebo, ░ Naratriptan 2.5 mg, ▪ Eletriptan 40 mg. Eletriptan/naratriptan vs. placebo ∗∗∗P < 0.001; Eletriptan vs. naratriptan † P < 0.05.
Functional improvement
Patients treated with eletriptan showed significantly better functional improvement at 2 h (Fig. 5) compared with both naratriptan (60% vs. 52%; P= 0.014) and placebo (44%; P < 0.001). The difference in functional status was not significantly different for naratriptan vs. placebo.
Tolerability and safety
Rates for all causality treatment-emergent adverse events were similar for eletriptan, naratriptan, and placebo (31%, 28%, 35%). Eletriptan was associated with a higher percentage of treatment-related adverse events than naratriptan or placebo (26%, 16%, 16%). However, treatment-emergent adverse events (Table 2) were typically transient and mild-to-moderate in intensity. One patient on eletriptan, 2 on naratriptan, and 1 on placebo discontinued the study prematurely. None of these premature discontinuations were due to adverse events or were study-drug related (3 were due to protocol violations; 1 was lost to follow-up).
Incidence of treatment-emergent adverse events∗

Values are given as %.
∗All causality, with incidence ≥ 3%; Between-treatment group comparisons were not significant on chi square.
Patient preference for migraine treatment
Patient ratings of treatment acceptability (recorded at 24 h for current vs. prior migraine treatments; Fig. 6) were significantly higher for eletriptan compared to both naratriptan (68% vs. 50%; P < 0.001) and placebo (31%; P < 0.0001). Naratriptan also showed significantly higher acceptability compared to placebo (P < 0.05).
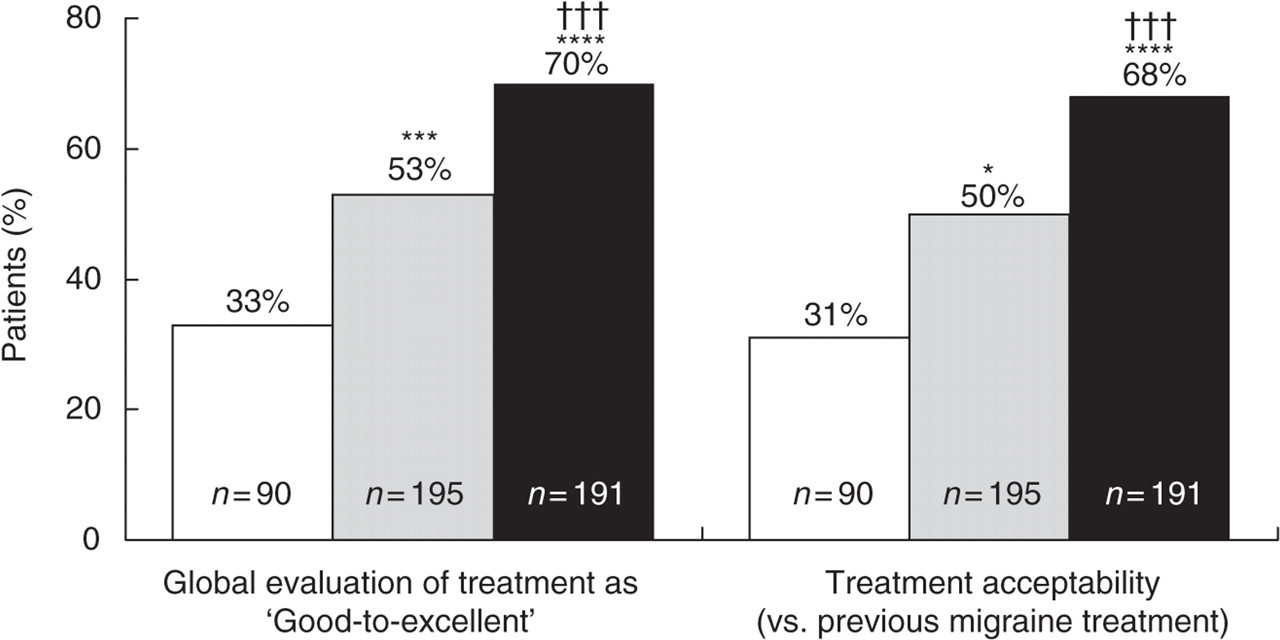
Global patient treatment evaluations at 24 h. □ Placebo, ░ Naratriptan 2.5 mg, ▪ Eletriptan 40 mg. Eletriptan/naratriptan vs. placebo ∗P < 0.05, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001; Eletriptan vs. naratriptan ††† P < 0.001.
Finally, patients were asked to provide a 7-point global rating of study treatment (Fig. 6). The proportion of patients reporting treatment to be ‘good-to-excellent’ was significantly higher for eletriptan (70%) compared to both naratriptan (53%; P < 0.001) and placebo (33%; P < 0.0001). Naratriptan also showed significantly higher global ratings compared to placebo (P < 0.001).
Discussion
The results of this double-blind, placebo-controlled, head-to-head comparator trial found eletriptan 40 mg to have significantly greater efficacy than the 2.5-mg dose of naratriptan across a range of clinically important outcome measures. As expected based on Tmax, eletriptan showed significantly greater early (1-h) headache response than did naratriptan (Fig. 2). The early efficacy advantage in favour of eletriptan continued to be significant at both 2 h and 4 h. Pain-free efficacy was also greater for eletriptan at both 2 h and 4 h (Fig. 3). The superior acute efficacy profile of eletriptan was associated with both a reduced need for rescue medication (Fig. 4), and a significantly more rapid return to normal functioning (Fig. 5). The one exception to the efficacy advantage of eletriptan vs. naratriptan was relief of nausea, which showed no difference among the 3 study treatments. This may have been, in part, attributable to the approximately 20-point lower baseline incidence of nausea in the current study (55%) compared to other recent naratriptan and eletriptan clinical trials (5, 7, 11, 14).
In addition to significantly more rapid onset of efficacy, treatment with eletriptan was associated with higher rates of sustained headache response (Fig. 4). Sustained response is a derived measure requiring that a patient achieve a response within 2 h, have no recurrence, and use no rescue medication and no second dose of study medication. Sustained response is highly correlated with pharmacoeconomic indices. A meta-analysis by Ferrari et al. (2) found naratriptan to have a lower headache recurrence rate at 24 h than sumatriptan, rizatriptan, zolmitriptan, and almotriptan. Only eletriptan was reported to have an equivalently low headache recurrence rate. The results of the current head-to-head comparator study were consistent with the meta-analytic result. The higher initial headache response of eletriptan, coupled with an equivalently low recurrence rate, resulted in significantly superior sustained response at 24 h compared to naratriptan.
The other most frequently noted therapeutic advantage of naratriptan is its tolerability (2). In the current study, all causality adverse events were similar across treatment groups. Although eletriptan was associated with a higher percentage of treatment-related adverse events than naratriptan or placebo, most adverse events were reported as being transient and mild-to-moderate in intensity.
The tolerability exhibited by the 40-mg dose of eletriptan, together with its superior efficacy, resulted in significantly superior global patient ratings of therapeutic effect (good-to-excellent, 70% vs. 53%; P < 0.001), as well as preference compared to previously utilized migraine treatments (68% vs. 50%; P < 0.001; Fig. 6). The wide margin in favour of eletriptan on these 2 patient-rated global measures is consistent with previous research (17), which has found satisfaction with migraine treatment to be correlated much more highly with efficacy parameters (e.g. rapid onset, sustained response) than with tolerability.
In summary, the results of this head-to-head comparator trial found the 40-mg dose of eletriptan to have significantly more rapid and sustained efficacy compared to naratriptan 2.5 mg, with only modestly lower tolerability, making eletriptan the preferred treatment choice on global ratings at 24 h.
Footnotes
Acknowledgements
This study was funded by a grant from Pfizer, Inc. We would like to express our appreciation to the patients who participated in the study, and to the coordinators at each site who supervised the conduct of the study. We would also like to express our appreciation to the Investigators who participated:
