Abstract
Orientation discrimination is a visual task dependent on inhibitory mechanisms in the visual cortex. In this study, orientation discrimination thresholds for bar and grating patterns were measured at two visual field locations in subjects with migraine with (n = 20) and without aura (n = 20) and in migraine-free control subjects (n = 20). No statistically significant differences were found between migraine groups and the control group on either task at foveal or peripheral visual field locations. No significant correlations were found between psychophysical thresholds and age, total lifetime auras or total lifetime migraine episodes. However, a trend was seen toward slightly impaired performance on the two foveal tasks in a subgroup of subjects with the highest total lifetime aura count. Thus we have found no convincing evidence that impaired cortical inhibitory mechanisms are a predisposing characteristic in migraine, but cannot rule out the possibility that cortical inhibitory mechanisms may be adversely affected by repeated visual auras.
There are many indicators that the visual system is implicated in migraine (see Chronicle (1) for review). In addition to the visual auras and photophobia that characterize many migraine episodes, there are indications that migraineurs may have heightened visual sensitivity or otherwise altered visual function interictally (2–4). Electrophysiological (5–7), psychophysical (8, 9) and transcranial magnetic stimulation (10) studies have all suggested that the visual cortex may be hyperexcitable, although there are conflicting findings in each of these domains (11–13).
One proposed mechanism which could underlie such excitability would be a reduction in the strength of cortical inhibitory processes. One form of this hypothesis argues that repeated visual aura may result in actual damage to GABA-ergic cortical neurones (14). Were this the case, one would predict increasing visual abnormality with duration of disease and frequency of auras. An alternative form of the hypothesis would argue that an abnormality in either GABA-ergic cell number or in some functional property of these neurones would be a predisposing factor for migraine. One approach to testing this hypothesis is to examine visual performance on tasks known to depend on cortical inhibitory circuitry.
Orientation tuning is a well-documented property of visual cortical neurones (15). Most neurones in primary visual cortex (Brodmann's area 17) respond maximally to a bar or grating of a given orientation; response amplitude falls off as stimulus orientation is rotated away from this optimal position. Although the basis of orientation selectivity has been hotly debated in the literature (16–19), there is very strong evidence that intracortical inhibition plays a fundamental role, as application of the GABA antagonist bicuculline dramatically broadens the tuning function of both simple (20) and complex cells (21) in area 17. Since the ability to make fine orientation-based judgements depends on the steepness of the slope of these tuning functions (22), psychophysical tasks which depend on the ability to make fine orientation judgements might be expected to reflect the strength of cortical inhibition.
To date, there has been only one examination of orientation discrimination in migraineurs (23). In her study, which compared migraine with aura with headache-free controls, an orientation discrimination deficit was seen in the migraine group only for very low spatial frequency patterns (0.5 c/deg) viewed foveally. McKendrick attributes this deficit to impaired magnocellular function because the magnocellular system is tuned to somewhat lower spatial frequencies than the parvocellular system (see (24) for review of magno-/parvo-pathways).
In the present study we have measured orientation discrimination thresholds in migraineurs with and without aura and in control subjects without migraine. All measurements were made interictally. Because the two major cortical cell types (simple and complex cells), although both orientation-selective, may be implicated in different visual functions (contour analysis vs texture analysis) (25), we used two orientated stimuli (bars and gratings) to anticipate the possibility of a deficit limited to only one of these orientated cell groups.
It has been suggested that visual problems associated with migraine aura might occur selectively in the visual field affected by the aura (7). To examine this issue, we planned to make measurements at 6° eccentricity in each lateral hemifield as well as foveally. However, there is a potential problem with this approach which had to be addressed first. There is a large literature (e.g. (26, 27)) on human visuospatial perception showing hemispheric differences in performance in neurologically normal right-handed subjects (performance of individual left-handed and ambidextrous subjects may show similar, reversed or no asymmetries). An underlying hemispheric asymmetry would complicate the interpretation of the relationship between aura side and orientation thresholds. Whether such field differences occur for orientation discrimination was not conclusive from the existing literature (28–30). Data from a pilot study in our laboratory showed no hemispheric differences in right-handers on an orientation task similar to the one described below. We first reconfirmed this result in the right-handed control subjects in the present study. Once this was established, we added left-handed subjects to our control group to match the subjects in our migraine groups.
Subjects and methods
Subjects
Participants were recruited through local neurologists and through advertisements on the McGill University campus. All potential subjects participated in a screening interview to assess headache and aura symptoms, as well as exclusionary criteria, which included a history of neurological or other disorders which could affect performance on visual tasks (e.g. diabetes, glaucoma, optic neuritis). The use of prophylactic medications for migraines or other neuroactive drugs (e.g. beta blockers, anti-depressants, anxiolytics and feverfew) was another basis of exclusion. All subjects were tested for handedness using a questionnaire (31) which assesses the degree of right or left handedness based on which hand subjects normally use to perform 18 tasks.
The migraine group consisted of 20 subjects with migraine with aura (MA) and 20 with migraine without aura (MO), according to the International Headache Society (32) criteria. A group of 20 non-migraine control subjects (C) who did not suffer from frequent or severe headaches and fitted other inclusionary criteria (described above) were also tested. Since our MA group contained several subjects who were left-handed or ambidextrous, we included two ambidextrous controls, and also recruited control subjects to match our migraine groups as closely as possible in age. A summary of relevant subject characteristics is provided in Table 1.
Group characteristics
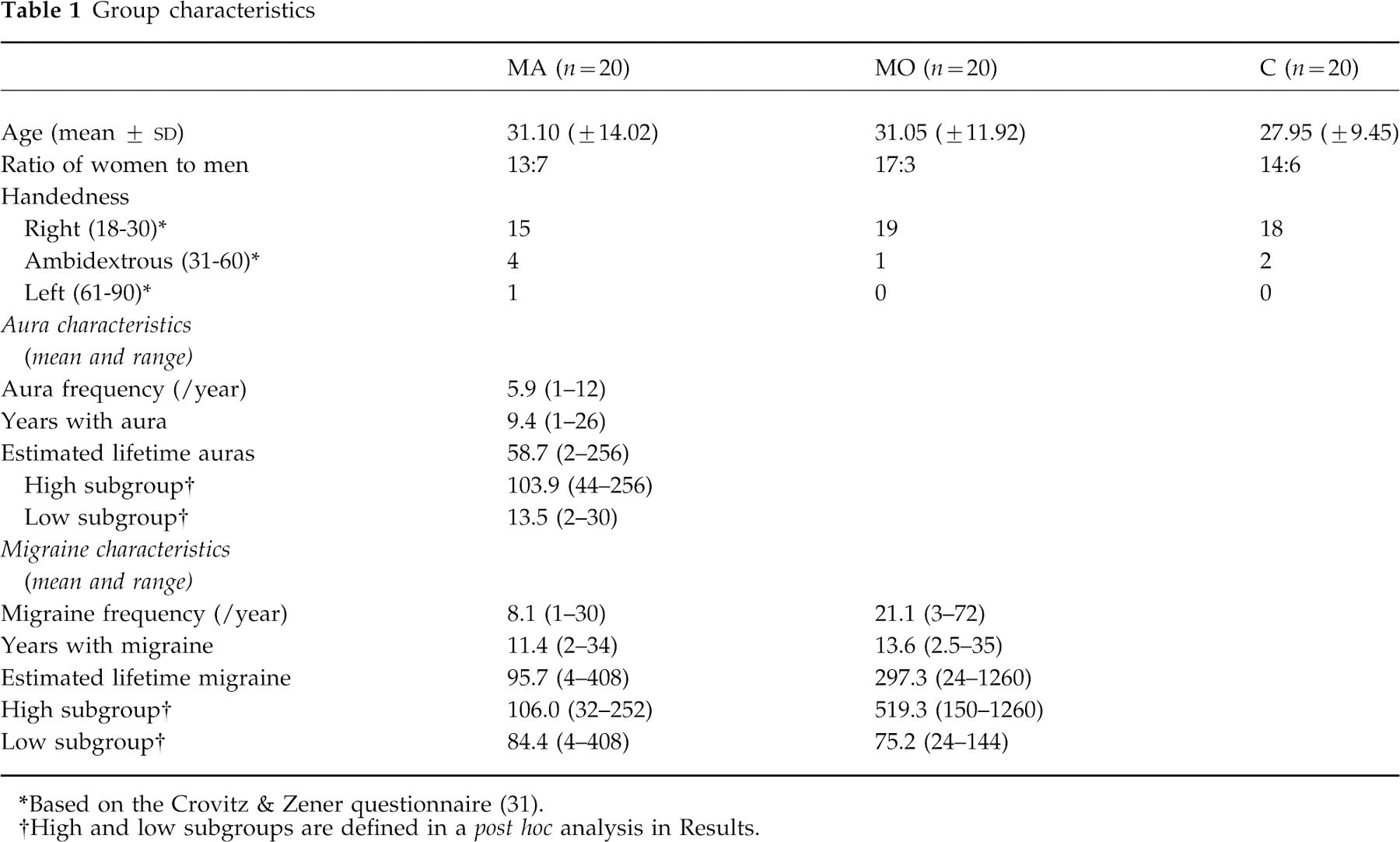
*Based on the Crovitz & Zener questionnaire (31).
†High and low subgroups are defined in a post hoc analysis in Results.
All procedures were approved by the Ethics Committee for research with human subjects of McGill University (Department of Psychology).
Stimuli and threshold measurement
The two types of orientated stimuli used in this study are illustrated in Fig. 1. The bar stimulus (top) is mathematically defined as the 4th spatial derivative of a Gaussian in luminance cross-section. This stimulus was chosen because its luminance distribution approximates the excitatory and inhibitory subregions of a typical on-centre/off-surround cortical simple cell (15), and should therefore be an optimal stimulus for this cell class. The grating stimulus is a sinusoidal grating multiplied by a circular Gaussian function, which produces a gradual fall-off in stimulus contrast at the edges of the stimulus. In the vision literature, this pattern is technically known as a Gabor patch and would be expected to be a stronger stimulus for cortical complex cells than the bar stimulus, because these cells summate orientation information non-linearly over an extended receptive field (33). It should be noted that because the Gaussian window is circular, the overall shape of the grating stimulus provides no orientation information; orientation judgements must be based on the orientation of the individual grating bars. Stimuli with sinusoidal luminance profiles and Gaussian blurring are used in vision research to avoid artefacts caused by sharp edges at stimulus boundaries and to circumvent the resolution limitations of computer monitors.

Examples of the bar (a) and grating (b) stimuli. The subject's task was to indicate whether the top of the pattern tilted to the left or right of true vertical.
Stimuli were presented on a Macintosh 17-in colour monitor with 1026 × 756 pixel resolution and 75 Hz frame rate, controlled by a 7100 Power Macintosh. Viewing distance was 1 m for the peripheral test conditions and 1.5 m for the foveal test conditions. Because the grain of visual analysis becomes coarser with retinal eccentricity, the stimuli used for testing at 6° eccentricity were scaled to compensate for the fall-off in visual acuity with eccentricity. The spatial frequency of the foveal grating stimulus was 9.0 cycles per degree (c/deg) and of the peripheral grating stimluli was 3.6 c/deg. The diameter of the grating patch was approximately 0.8° in the foveal and 2° in the peripheral condition. The length of the bar stimulus was approximately 1.3°, and the width of the white central portion was 4.6 min of arc foveally; peripherally values of 2° and 7 min of arc were used. The average luminance of the computer screen was 35 candelas/m2. The test room was dimly illuminated by diffuse lighting placed so as to avoid any reflections on the computer screen.
Procedure
Testing took place in our laboratory during a migraine-free period (no migraine for at least 3 days prior to the appointment). After obtaining written informed consent, subjects were seated in front of the computer monitor with their chin placed in a chinrest to maintain the appropriate viewing distance. Subjects then received instructions and a brief training session for the two-alternative forced choice paradigm that was used to determine orientation sensitivity thresholds.
Testing was conducted in two blocks: both foveal tasks were presented in the first test block with order of bar vs grating randomized across subjects. In the second test block, peripheral testing was carried out with bar and grating stimuli, again with order randomized. Foveal thresholds were assessed before the peripheral thresholds, rather than using a fully counterbalanced design, because the peripheral tasks are more difficult in terms of their fixation requirements (see below). The foveal testing gave subjects experience with the basic task before adding the complexity of monitoring two peripheral locations simultaneously. Each test block was preceded by a brief practice session, and subjects were given a break between the two threshold assessments within each block (bar and grating) as well as between blocks.
For foveal testing, the subject fixated a small fixation spot on an otherwise homogeneous grey screen, and initiated a trial by pressing a designated key on the keyboard. The fixation point disappeared and one of the eight test stimuli appeared at fixation for 150 ms, after which it was replaced by the fixation point. The subject was required to indicate whether the top of the bar or grating tilted left or right, guessing if they were uncertain (forced choice). Eight stimulus orientations (four tilted left and four tilted right of vertical) were presented 25 times each in random order (method of constant stimuli) for a total of 200 trials per threshold measurement.
For peripheral testing, the subject again fixated a small fixation spot at the centre of the computer screen, and initiated trials as above. However, in this case the stimulus appeared in one of two symmetric locations 6° to the left or right of fixation, with side determined randomly from trial to trial. The stimulus exposure time was again 150 ms, too brief to permit the subject to foveate the stimulus before it disappeared. Subjects were instructed to maintain fixation on the central spot at all times and to avoid trying to predict where the stimulus would appear.
In the peripheral conditions, thresholds were evaluated separately for left and right visual fields, with 25 presentations of each of eight orientations in each visual field for a total of 200 trials for each of the two interleaved threshold measurements. The range of stimulus values used for each condition was determined for each subject on the basis of performance in the training sessions, such that the values tested would include at least one highly discriminable angle and at least one subthreshold angle.
For each test condition, the percentage of correct trials was calculated for each stimulus orientation and these data were fitted with a cumulative normal function. The standard deviation of the function, which indicates the tilt angle at which the subject successfully identified the direction of tilt on 68% of trials, was taken as the orientation threshold.
Results
Interhemifield comparisons
Because of the possibility of interhemispheric differences on visual tasks, we first compared the performance of our normal right-handed control subjects (n = 18) in left and right hemifields on each of the orientation tasks (bar and grating). As assessed by paired t-tests, performance was not found to differ significantly between hemifields on either task (grating task: t17 = 1.37, P > 0.05; bar task: t17 = 0.16, P > 0.05). This is consistent with a previous pilot study in our lab using a task very similar to the grating task used here. Therefore, we do not think including left-handed and ambidextrous individuals in our control and migraine groups obscured potential interfield differences.
In designing this study, our intention had been to make within-subject comparisons between hemifields in MA subjects with aura consistently in one hemifield, and this was one reason we included the two peripheral (6° eccentric) conditions in our study. However, as the study proceeded it became clear from the interview data that this analysis was not viable. Of our 20 MA subjects, one subject was unable to give a retrospective report of aura side and 10 reported auras occurring in both hemifields, either within the same episode or in different episodes. When we examined the orientation discrimination data for the remaining nine subjects who stated retrospectively that their auras were limited to one hemifield, we found that three showed worse performance on the side of their auras in both tasks, three showed poorer performance on the side opposite to their auras, and three showed poorer performance in the left visual field on one task and in the right visual field on the other. All interfield differences were small—both the mean and the range of interfield differences were comparable to those of the control group for both bar and grating conditions. Finally, six subjects in this study have more recently participated in an ongoing study of aura which involves recording information about aura side at the time of the episode. Three of these six show inconsistencies between their retrospectively reported aura side at initial interview, and aura side as recorded during episodes. In view of these observations, we concluded that our data set does not provide a basis for testing the relationship between aura side and visual hemifield performance. Therefore, in order to obtain more accurate threshold estimates in these difficult peripheral tasks, we combined the data for left and right visual hemifields, yielding a single peripheral score for each subject for each of the two visual tasks. All further analyses were based on these averaged peripheral scores. Thresholds for foveal and peripheral testing with the grating and bar stimuli are shown in Fig. 2.
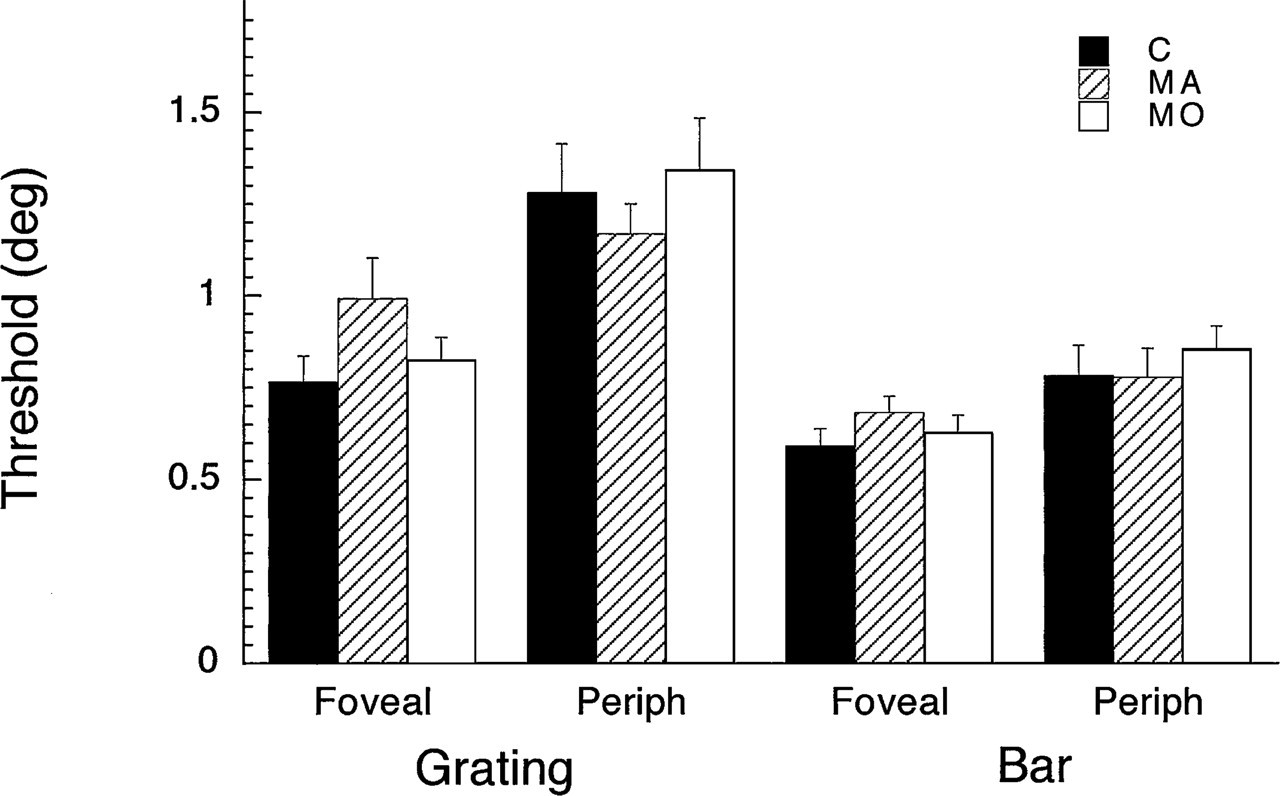
Thresholds (group means and standard errors) for the grating and bar stimuli at foveal and peripheral (6° eccentric) visual field locations. C, Control; MA, migraine with aura; MO, migraine without aura.
Foveal and peripheral orientation thresholds
We then used Dunnett's test of planned comparisons between each
experimental group and a single control group to examine the effects of migraine
condition on performance on each test/position combination separately. Because
our underlying hypothesis was directional (that migraine-related loss of
inhibition would result in significantly poorer orientation discrimination in
migraineurs than in the control group), we used one-tailed tests to increase the
power of rejecting the null hypothesis if indeed a difference exists. Despite
our lenient criteria, none of the four contrasts between MA and C reached
statistical significance at the 0.05 level, although the comparison for the
foveal grating stimulus was very close to significance (critical t for d.f.
60,2 = 1.95; t-values for MA vs
C: foveal grating (t = 1.93), foveal bar (t = 1.43), peripheral grating (t =
−0.65), peripheral bar (t = −0.04)). None of the
t-values for comparisons between MO and control reached
significance (range: t = 0.36–0.70). Since multivariate analysis may
reveal effects which do not reach significance in individual univariate
comparisons, we re-examined the data using
The sample size used in this study (20/group) would allow us to detect a between
group difference of about σ (the within group
Relationship between orientation thresholds and migraine history
If, as suggested by Chronicle & Mulleners (14), the occurrence of migraine auras brings about a loss of GABA-ergic cortical neurones, the analysis of group means we have performed above might fail to reveal this effect because of the wide range in both aura frequency and number of years since aura onset seen in our subjects (see Table 1). One might, however, predict a positive correlation between visual thresholds and total number of auras experienced, even in the absence of overall group differences. Since cumulative measures of aura frequency are likely to be positively correlated with age, we first asked whether there was a relationship between age and any of our four measures of orientation discrimination. We found no significant correlations between age and any of our four threshold measures for non-migraine controls or for either of the migraine groups (P > 0.05 in all cases).
We made crude estimates of lifetime aura and migraine frequencies by multiplying reported annual rates by the number of years since onset. However, while estimates of number of years since migraine/aura onset are likely to be quite accurate, frequency estimates are much more open to error, especially as subjects often report that aura patterns have varied considerably over years as a function of hormonal factors, life events, etc. Because of our lack of confidence in the accuracy of these estimates, we used Spearman's correlation for ranks (ρ), which assumes only that the ranking of subjects is accurate and makes no assumptions about the absolute number of episodes. When the data for the MA group were examined with respect to total lifetime auras, all correlations were positive (range: ρ = 0.18–0.40) but only one approached statistical significance (peripheral gratings: P = 0.08). The same question was asked of total migraine episodes in both the MA and MO groups. For the MA groups, the outcome was the same as for total auras—no significant correlations. When we asked the same question of the MO group, we noted to our surprise that the correlations were all negative. However, none reached statistical significance.
In view of the weak but positive correlations between total lifetime auras and
our threshold measures, we carried out one final analysis of our data dividing
our MA and MO groups into high and low subgroups (n = 10/group)
based on total lifetime auras for MA and total lifetime migraines for MO (see
Table 1
for characteristics of each subgroup). Because the high groups in both cases
were on average older than the corresponding low groups, we subdivided the
control group on the basis of age. (Mean ages for high subgroups: MA = 33.9
years; MO = 39.9 years; C = 34.1 years. Mean ages for low subgroups: MA = 28.3
years; MO = 25.2 years; C = 21.8 years.) The data for the high and low subgroups
are displayed in Fig.
3. For the two foveal conditions, it is striking that thresholds for the
low MA group are nearly identical to those for the low MO and low C groups. The
weak trends we saw in our overall data set (Fig. 2) appear to arise from the
performance of the high subgroup of MA subjects, i.e. those individuals with the
highest total lifetime auras. Data on the peripheral tasks do not present such a
clear picture. While the MA groups show a similar trend for the peripheral
grating task, the fact that the low MA group performed so much better than the
other groups makes this observation difficult to interpret. In the case of the
peripheral bar task, the two MA subgroups and the two control groups showed
nearly identical performance, whereas the MO group showed the negative
relationship between total migraines and thresholds noted in the discussion of
correlations above. Given the post hoc nature of this analysis,
applying the planned comparisons used earlier in the study is not justified.
Instead, one-way

Data from Fig. 2 divided into high (□) and low subgroups (▪) as described in the text. Left panels, foveal thresholds; right panels, peripheral thresholds. C, Control; MA, migraine with aura; MO, migraine without aura.
Discussion
In the present study, orientation discrimination was used as a measure of inhibition within the striate cortex because of the extensive evidence that the narrowness of orientation tuning of cortical cells depends in large part on inhibitory intracortical mechanisms (17, 20, 21). The thresholds obtained foveally using the bar stimulus (group averages: 0.5–0.70°) are very comparable to those reported in the vision literature (36). Peripheral thresholds for the bar stimulus were slightly higher than foveal in all three groups but still within the range reported for normal subjects in the literature (37, 38). Making psychophysical judgements of peripheral stimuli while maintaining fixating is difficult for naive observers, so it would not be surprising if thresholds were slightly elevated.
Thresholds obtained for foveal grating stimuli were slightly higher than for foveal bar stimuli in all three groups (group means: 0.8–1.00°). However, these values are within the range reported in the literature (39–41). Interestingly, peripheral thresholds for this task were markedly higher than either the foveal thresholds for gratings or the peripheral thresholds for bars in all three groups of subjects. To the best of our knowledge, this condition has not been examined in the vision literature and merits further investigation. It is well known that lateral interactions affect a variety of aspects of discrimination of textural stimuli in the visual periphery (25, 42); therefore this may account for the elevated thresholds seen in our study.
Since the thresholds reported here for the normal control group fall within or very close to the normal range for the three conditions which have been examined in the literature, we feel confident that our technique provides a sufficiently sensitive measure of this visual ability in untrained normal and clinical populations to justify its use as a marker of cortical inhibition.
Cortical hyperexcitability and inhibition
Based on several lines of evidence, Welch and his collaborators have argued persuasively that migraineurs display cortical hyperexcitability within migraine episodes, and probably interictally as well (43, 44). Our results suggest that this hyperexcitability does not arise as a consequence of reduced intracortical inhibition. This finding is in line with other recent work in our laboratory (45). Our results leave open the possibility that reduced intracortical inhibition is a characteristic of migraine with aura (or of all migraine) if one or more of the following are true: (i) the effects are very small, and might therefore be revealed with much larger migraine groups. However, to increase statistical power sufficiently to detect differences of the size that appear possible in our MA group would require group sizes of at least 50, and it would still be questionable how important such a small effect would be to the migraine process; (ii) reduced inhibition is due to phasic changes in neuromodulatory mechanisms, occurring only during and in the period immediately preceding an episode; (iii) inhibition plays multiple roles in neocortical functioning and it is possible that the circuitry mediating orientation sensitivity is not the relevant circuitry in migraine. However, in another recent study in our laboratory we have also failed to find evidence of impaired contrast gain controls (45), another inhibition-based mechanism which is not orientation-dependent. The same experimental conditions did however provide evidence of a modest hyperexcitability suggesting that hyperexcitability does not depend on changes in inhibition.
Thus, while a negative finding is much more difficult to interpret than a positive finding, we believe that it can safely be asserted based on the results of our study that neither MA nor MO are characterized by a persistent large reduction in intracortical inhibition.
The Chronicle–Mulleners hypothesis of aura-induced damage
Chronicle & Mulleners (14) have proposed that repeated migraine aura might actually cause damage to inhibitory cortical neurones. This possibility must be given serious evaluation because it implies increasing neuronal damage with increasing aura frequency, and that an aspect of migraine which has generally been considered to be benign may in fact compromise normal visual function. Most migraine therapy is targeted at alleviating headache, not aborting auras, and in fact, some acute migraine medications are given at the onset of the headache for best effect (i.e. after the aura) (46). Moreover, there is a significant population of individuals who experience migraine aura without headache who generally receive no treatment, on the assumption that with the rare exception of migrainous stroke (47–49), aura has no lasting deleterious effects. Reports of transient changes in visual sensitivity after migraine attacks (50, 51) and more recent indications of persistent precortical field defects in migraine with aura (23) indicate that this possibility must be taken seriously.
In order to best test this hypothesis, one would ideally wish to examine a very large population of MA subjects well distributed across the range of total lifetime number of auras. In such a group one would expect to find a strong positive correlation between one's measure of impaired inhibition and lifetime auras. As discussed in Results, total auras is very difficult to estimate accurately retrospectively, because migraine frequency fluctuates dramatically across years as a result of hormonal factors, life stressors, and probably many other factors not yet understood. In our study we did see weak positive correlations between ranked estimated lifetime auras and each of our measures, but these did not reach statistical significance. It is possible that this is because our groups contained relatively few older subjects with long migraine histories. However, when we subdivided our MA group into high and low lifetime aura incidence, it became clear that the modest trends toward worse foveal performance in MA were restricted to the subset of individuals with the highest lifetime incidence. This lends some support to the Chronicle & Mulleners position, and strongly indicates the need for further research on this issue.
Foveal vs peripheral and magno- vs parvo-effects
The weak trends we saw in our MA group occurred for foveal testing only. In a recent dissertation study (23), McKendrick also examined orientation discrimination in migraine with aura using stimuli similar to our grating stimuli (Gabor patches). She reports a significant impairment in migraineurs only for very low spatial frequency gratings (0.5 c/deg) and only for foveal viewing. This deficit was tentatively attributed to a magnocellular pathway deficiency. Foveal viewing of higher spatial frequencies (4 c/deg) and peripheral viewing (10° into the upper field) of either spatial frequency elicited comparable performance in controls and migraineurs. Clearly, further investigation will be required to determine whether the foveal representation is particularly affected in migraine, as assessed by orientation sensitivity. There is no obvious explanation as to why this would be the case.
The grating stimuli used in our study had a foveal spatial frequency of 9 c/deg, and were suitably scaled in peripheral viewing to correct for the change in visual acuity with eccentricity. While our tasks were not designed to test the magno-/parvo-cellular distinction, the line and grating patterns used in this study would stimulate both magno- and parvo-cellular pathways, approximately equally ((52); Wilson, personal communication). Thus it is possible that the impairment noted by McKendrick at very low spatial frequencies reflects a precortical (retina or LGN) abnormality in the magnocellular input to cortical orientation tuned mechanisms and does not reflect impaired intracortical processing. However, we are cautious about emphasizing the magno–parvo distinction, because based on calculations using Wilson's retinal model (52), both of McKendrick's stimuli (0.5 and 4.0 c/deg) should have been much stronger magnocellular than parvocellular stimuli, yet a deficit was seen only for the 0.5 c/deg pattern. The resolution of this issue must await further testing with stimuli better designed to isolate these two precortical input systems.
Aura laterality
Despite the widespread depiction of visual auras as typically restricted to a single hemifield, our experience in our ongoing aura mapping study indicates that in fact many migraineurs experience auras in both hemifields with moderate frequency despite their retrospective descriptions of restriction to a single side. The unreliability of retrospective reports with respect to aura and headache laterality has been discussed by other investigators (53). While it would be appealing to be able to demonstrate within-subject differences in migraine-related visual effects, we would suggest that this can only feasibly be done if prospective aura laterality data are collected from the subjects over an extended time period using aura diaries or a similar tool.
Summary
In summary, we have failed to demonstrate a statistically significant deficit in orientation discrimination in migraine with aura which would be indicative of impaired cortical inhibitory mechanisms as part of the underlying pathophysiology of migraine aura. However, our study does provide weak support for the possibility that a long history of frequent migraine aura may lead to weakened inhibitory processes. Subsequent work in this vein should focus on the population of individuals with longstanding, frequent and stable aura patterns. We would point to the population of individuals with auras without headache as a particularly interesting population in this respect, since recent case self-studies in the literature (54, 55) as well as our experience with our subject population indicate that these individuals tend to have highly stereotyped auras over periods of many years. Finally, the weak trends we saw in our aura group were not mirrored by the migraine without aura group in any conditions. Therefore our study provides no support for the suggestion that reduced cortical inhibition is an important mechanism producing heightened cortical excitability in all migraine but aura symptoms accessible to conscious awareness in only a subset of migraineurs.
Footnotes
Acknowledgements
This work was supported by the Medical Research Council of Canada (grant no. 14034 to F.W.) and by the G.W. Stairs Fund of McGill University. We are grateful to Dr F. Andermann, Dr W. E. S. Connelly, Dr L. Durcan, Dr G. Francis, Dr I. Woods, Dr T. Kirkham, Dr S. Fyles, Dr J. Rubin, Dr S. Murphy, and Dr P. Tellier (Director, McGill Student Health) for referring their patients to our project on cortical inhibition and migraine. We would especially like to acknowledge the participation of the late Dr J. Meloche (Director) and of the entire staff of Clinique de la Migraine de Montréal. Thanks also to our Research Coordinator Anna Feindel BA, RN, for her role in participant recruitment and clinical interviews.
