Abstract
The present study examined the extent to which migraineurs demonstrate interictal visual cortical hyperexcitability as a result of poor inhibitory control in the visual system. We employed a well-established psychophysical measure of inhibition, visual contrast gain control. The task involved detecting a briefly presented target that was superimposed on a highly excitable high contrast masking pattern. The strength of inhibition was assessed by comparing target detection thresholds with and without the operation of gain controls. Migraineurs with and without aura (n = 25, n = 22, respectively) were compared with those with no history of migraine (n = 25). Our results do not indicate a loss of inhibition in migraine; the strength of inhibitory feedback contrast gain controls was similar between migraineurs and controls. We did however, find a statistically greater masking effect in migraineurs compared with controls in the zero delay condition, suggesting cortical hyperexcitability in migraine. Possible mechanisms of cortical hyperexcitability are discussed in light of the results.
A number of abnormalities in visual function have been described in migraine, some of which indicate hyperexcitability of the visual pathways (for review see 1–3). Clinical observations suggest that hyperexcitability occurs not only during an attack, typically in the form of photophobia, but also between episodes. Compared with headache-free subjects, migraineurs are more sensitive to light (4), and indicate more intense illusions and greater visual discomfort to high contrast grating patterns (5–8), lower critical flicker fusion rates (9), and speeded performance on low-level visual tasks (10), although the latter finding was not replicated in a study by Palmer & Chronicle (11). In addition, background gratings have been shown to hinder detection of low contrast stimuli (12) and letter detection (13).
Electrophysiological investigations suggest that the hyperexcitability in migraine occurs at the cortical level. Migraineurs show augmented amplitude on Visual Evoked Potential (VEP) measures using flash or checker-board stimuli (7, 14–19) (though not all studies show this effect (20–22), increased contingent negative variation amplitudes (23, 24), and enhanced photic driving responses on EEG measures (25), all of which imply visual cortical hyperexcitability in migraine. Transcranial magnetic stimulation work has also provided support for visual cortical excitability in migraine: Aurora et al. (26) measured the intensity of occipital cortical stimulation required to generate visual phosphenes and found that a subset of visually sensitive MA subjects required a lower cortical stimulation threshold compared with controls, although again there is evidence to the contrary (27).
The observed hyperexcitability may reflect a direct augmentation of excitatory mechanisms, either at the level of the glutaminergic thalamocortical synapse, or at the level of recurrent excitatory circuits known to operate within the visual cortex (28). A wide variety of abnormalities, for example reduced magnesium levels, could heighten this recurrent excitation. An alternative route to cortical hyperexcitability would be through a reduction in local cortical inhibition. One variant of this notion, proposed by Chronicle & Mulleners (29), posits that inhibitory function may be compromised by repeated visual auras, as a consequence of hypoxia-induced damage to GABAergic neurones.
Inhibition is widespread in the neocortex comprising about 25% of primate cortical neurones overall, although a slightly lower percentage in primate visual cortex (30). To date, the only documented neurochemical substrate for cortical inhibition is via GABAergic neurones. Intracortical inhibition has long been seen as playing a critical role in keeping excitation in check; blocking GABAergic inhibitory synapses with the GABAa antagonist bicuculline in slice preparations of human neocortex results in epileptiform activity in response to stimulation (31). Hypoxia-induced loss of GABAergic neurones in early development (32, 33) has been suggested to underlie the clinically observed link between hypoxia at birth and convulsive disorders in children (34, 35). In the visual cortex, inhibition plays a wide variety of roles in visual function. For example, it contributes to the tuning of neural receptive fields, sharpening their specificity for a variety of visual features such as stimulus orientation (36, 37).
It is not presently possible to distinguish behaviour-ally among the various possible non-inhibitory sources of visual hyperexcitability. However, it should be possible to distinguish between reduced inhibition on the one hand, and other excitatory changes on the other, by examining performance on visual tasks known to involve cortical inhibition. We have recently attempted to do this by measuring orientation discrimination thresholds in migraineurs (38) and were unable to detect any evidence of visual impairment in migraineurs relative to non-headache controls, although a subset of individuals with aura showed a weak trend in this direction. In the present study we approach the same issue using a different task, one that assesses the integrity of the contrast gain control mechanisms—another cortical mechanism believed to depend on inhibition.
Because neurones have a limited dynamic range (i.e. a maximum response rate of about 400 Hz), the nervous system uses inhibition to adjust incoming stimulation to produce a match between the response range of the neurones and the current stimulus range. For example, if one awakens in the middle of the night and turns on a light, one is momentarily dazzled by the bright light, but rapidly adjusts. Initially, the responses of retinal neurones, adjusted to darkness, are saturated by the new ambient light level (i.e. most are firing at their maximum rate) and thus cannot distinguish between gradations in luminance. However, the operation of lateral inhibitory circuits in the retina rapidly adjusts the sensitivity of the system, allowing the range of luminances in the image to be appreciated. This is luminance gain control.
At the cortical level, a similar gain adjustment occurs for contrast. Contrast refers to the range of luminances present in the image, rather than the absolute luminances. Thus a visual scene with many bright and dark areas would have a high average contrast, whereas one with more subtle changes (e.g. a scene in mist) would have low average contrast, even when the average luminance of the two scenes was equal. Single cortical neurones show characteristic contrast response functions which are adjusted dynamically depending on the mean contrast level (39–41). Unlike the orientation mechanisms discussed above, the neurochemical basis of these gain controls has not been clearly demonstrated. However, behavioural measures of this function display a clear signature of inhibition—they operate divisively. That is, the response—at the level of single cells (41), the human VEP (42) and psychophysically assessed visual behaviour (43)—is scaled by a constant multiple. The only known mechanism for performing neural division is inhibition (44). Moreover, there is evidence that rapid gain controls operate in a feedback fashion (43). That is, when an abrupt change from low to high average contrast occurs, there is a brief period where subtle differences in contrast at the high contrast end of the range are difficult to detect because neural responses elicited by these stimuli are approaching saturation. However, within 150 ms there is a shift such that previously undetectable contrast differences can now be seen. The task we use in the present study takes advantage of this dynamic change reflecting inhibitory feedback to assess the integrity of contrast gain control in subjects with and without migraine.
In considering the weakened inhibition hypothesis, two issues must be considered. First, would one expect to see equivalent effects in migraine with and without aura? The very fact that the cortex of migraineurs with visual aura is prone to this form of spreading activation during episodes would lead one to predict differential excitability between groups. However, to date the evidence is mixed. Some of the existing evidence for cortical hyperexcitability cited above shows effects in both migraine groups (4, 7, 8, 12, 18), although the effects in MA are generally stronger, whereas others have shown effects only in MA (13). A number of studies either did not distinguish between MA and MO (5, 6, 15, 17, 19), or tested MA groups only (10, 16), so do not provide further insight on this issue. The second issue to be considered is whether hyperexcitability is an intrinsic feature of the migraineur's visual system. Alternatively, does it arise as a consequence of repeated migraine attacks, or as hypothesized by Chronicle & Mulleners (29), as a result of repeated visual auras?
The present study was designed to address these issues by comparing groups of migraineurs with and without visual aura with headache-free control subjects on a visual masking task. The task we use has two components: the first assesses interictal hyperexcitability and the second assesses the strength of inhibition in the form of contrast gain control.
Subjects and methods
Participants
All potential observers took part in an extensive clinical interview including a detailed description of their health and headache history. We assessed hand dominance as a clinical guide for laterality using a handedness questionnaire ((45); scores < 30 strongly right-handed, > 60 strongly left-handed). All subjects had normal or corrected-to-normal vision as defined by Snellen Acuity of at least 20/25. Subjects were remunerated $10 for their participation. All procedures were approved by the Human Research Ethics Committee of the McGill University Department of Psychology.
Exclusion criteria
Subjects were excluded from the study if they had a history of any of the following: optic neuritis, glaucoma (including family history), cluster headache, multiple sclerosis, severe head or neck injury, cardiovascular disease, hypertension, diabetes, epilepsy, or any known neurological condition that could affect vision. They were also excluded if they were taking any headache prophylactic medication, anti-depressants, or anxiolytics.
Migraine-free control subjects (n = 25; 17 female, eight male)
Control subjects were recruited through advertisements on the McGill University campus and in the Montreal community and had no history of migraine, or of frequent severe headaches of other types (defined as more than two per month, on average). The average age of the control subjects was 27.4 years (
Migraine subjects
Migraine subjects were recruited through local neurologists as well as by advertisement within McGill University and the Montreal community. Based on interview data, migraine participants were classified into two groups on the basis on the criteria established by the International Headache Society (45): migraine with aura (MA), and migraine without aura (MO). If migraine subjects were not referred to the study by a neurologist, we obtained a medical confirmation of a specific migraine diagnosis from the subject's physician with the subject's consent. In some cases (three MA, four MO), we were unable to acquire such information, but included such participants based on the following strict criteria: longstanding migraine, no recent change in migraine characteristics and a familial component to their migraines. Participants had to have been migraine-free for at least 7 days before testing.
Migraine with aura (n = 25; 19 female, six male)
The mean age of the MA subject group was 30.7 years (
Migraine without aura (n = 22); 20 female, two male)
The mean age of the group of MO subjects was 29.0 years (11.3) and their average handedness score was 25.2 (12.3). None of the MO subjects in our sample reported experiencing visual or other aura symptoms.
There was no significant difference in ages or handedness between subject groups (F(2.69) = 0.65, P = 0.52; F(2.69) = 0.30, P = 0.74). Each subject group contained one experienced psychophysical observer who was familiar with the goals of the task. All other participants were inexperienced observers and were naive as to the purpose of the experiment.
Apparatus and stimuli
An Apple Macintosh II computer controlling two Apple monochrome monitors was used to present the mask and target stimuli. The monitors had refresh rates of 66.7 Hz, 8-bit grey-scale resolution, and were linearized by using a subset of 150 linearly spaced grey levels. The resolution of each monitor was 640 pixels in width × 480 pixels in height and each subtended a visual angle of 12° × 9° at a viewing distance of 1 m. Mean screen luminance was 90 cd/m2. Both screens remained at mean luminance except when the target and masking stimuli were presented. The target and mask patterns were presented independently on separate monitors and were optically superimposed by use of a prism beam splitter. Because the two images were superimposed, the maximum contrast possible was limited to 50%.
The target and mask stimuli are shown individually and superimposed in Fig. 1a–c. The excitatory masking pattern was a square (2° × 2°) patch of three cycles per degree (c/deg) sinusoidal grating orientated 15° relative to the vertical and was presented at a contrast of 37.5%. The target pattern was a vertically orientated localized patch with a bright centre and darker flanking lobes. A detailed description of the target can be found in Wilson & Humanski (46). Electrophysiological studies on primates have shown that similar patterns stimulate visual neurones that are orientation and spatial frequency selective, such as simple cells of area V1 in striate cortex (47). An orientation difference between the mask and target was introduced to assess the masking effect over a large range of local spatial phases (48).

(a) The target stimulus, technically the sixth derivative of a spatial Gaussian function with a peak spatial frequency of 3 c/deg. (b) The masking stimulus, a 3 c/deg sinusoidal grating. (c) The masking and target superimposed.
Procedure
There were three experimental conditions (one baseline and two masking conditions), in each of which we determined the lowest contrast at which subjects could reliably detect the target (target ‘contrast threshold’). Thresholds were measured using a two-alternative temporal forced choice method where subjects indicated by a key press which of two presentation intervals contained the target pattern. Whether the target appeared in the first or second interval was determined randomly on each trial.
In the baseline unmasked condition (see Fig. 2a), in one interval the target pattern was presented on a uniform grey background for 30 ms, and in the other interval, the grey background appeared without the target. This threshold value provided a measure of baseline contrast sensitivity to the briefly presented target. In the masking conditions, in each interval the mask appeared for 400 ms, with the 30-ms target superimposed in either the first or second interval. In one masking condition, the mask and target appeared simultaneously with zero onset delay such that contrast gain controls would not have time to operate (see Fig. 2b), whereas in the second masking condition, a 150-ms delay was introduced between the onset of the mask and the presentation of the target to allow full action of contrast gain controls (see Fig. 2c).

A schematic of the presentation of the stimuli in the three experimental conditions. (a) Baseline unmasked condition; the target stimulus appeared in either interval 1 or interval 2. (b) Masking condition with zero delay between mask-target presentation. (c) Masking condition with a 150-ms delay between presentation of the mask and target. For (b) and (c) the masking pattern appeared in interval 1 and interval 2, but the target stimulus appeared in either interval 1 or interval 2. Although the target is shown as occurring in interval 1 in this figure, from trial to trial the target occurred randomly in either interval 1 or interval 2.
Subjects were seated with their head comfortably positioned on a chin rest and viewed the display monocularly with their dominant eye (determined by two simple pointing tests) from distance of 1 m. During experimental runs, observers were asked to fixate on the centre of the screen. Each trial was initiated by a button press made by the subject. A brief tone signalled the onset of each stimulus interval. We used the method of constant stimuli which involved randomly presenting five target contrasts 25 times per contrast level within a given run. At the end of each condition, a Quick function (49) was fitted to the data using a maximum likelihood estimation technique. The target contrast threshold was taken to be the 75% correct estimate.
The order in which testing conditions were run was the same for each subject: masking condition with zero delay, masking condition with delay, baseline unmasked condition. Participants were given short breaks between testing conditions. To ensure subjects could perform the task and to reduce practice effects, all subjects participated in three short practice runs in the zero offset condition before formal testing began: (i) using a lower contrast mask (20%) and a range of high target contrasts; (ii) using the same low contrast mask and lower target contrasts; (iii) using the same mask contrast as that in the actual test (37.5%) with a range of high and low target contrasts. To assess the contribution of practice effects across test conditions, pilot work was conducted on one MA subject (author S.L.M.) in which multiple consecutive runs were made in the zero delay masking condition. Although there were small practice effects, no significant changes in threshold was observed with practice in this testing condition. Therefore, we can be confident that any reductions in masking effect observed in the second testing condition (150 ms delay) are a result of feedback contrast gain controls and not practice effects.
Dependent measures
On the basis of Bowen & Wilson's (50) masking terminology, the masking effect here is defined in terms of a threshold shift: the masked target threshold divided by the unmasked baseline target threshold. Thus, a threshold shift of 1.0 would indicate no change in threshold and therefore, no masking effect. In contrast, a threshold shift > 1.0 would indicate that target threshold is elevated by that factor, and a shift < 1.0 would indicate that the mask facilitates target detection (i.e. reduces target threshold). Our measure of inhibition is defined as the ratio of the threshold shift in the no delay masking condition to that of the masking condition with the 150-ms delay allowing for action of contrast gain controls. The greater the ratio, the stronger the gain control and the stronger the inhibition. A ratio equal to or approximately 1, where masking effects are comparable in the two masking conditions, suggests that gain controls are not functioning at all.
Statistical analysis
We wished to evaluate two main hypotheses concerning subject group differences. First, do migraineurs, irrespective of aura status, have heightened visual cortical excitability compared with migraine-free controls? Second, to what extent do the two clinical groups of migraineurs (MA, MO) differ in hyperexcitability? To analyse such group differences in our dependent measures, we performed two orthogonal planned comparisons. The first planned comparison tested for effects as a result of having migraine (i.e. MA and MO groups weighted equally (MA + MO) compared with controls). The second compared MA with MO to address any difference that may come from the overt aura process.
Specific hypotheses
If the visual cortex is hyperexcitable in migraine, we expect the following: (i) in the masking condition with zero delay, where contrast gain controls do not have time to operate, masking effects (i.e. threshold shifts) will be greater in MA and MO subjects compared with migraine-free controls (i.e. the mask will exert a greater excitatory effect on the visual cortical channels that mediate detection of the target); (ii) if contrast gain controls, which require time to operate, are weak or dysfunctional in migraine, the ratio of the threshold shift in the condition with zero delay compared with that with a delay will be smaller in migraineurs compared with controls.
Results
Baseline target contrast threshold
The average contrast threshold for the unmasked target was lower in the migraine groups (MA 2.99 ± 0.99%; MO 3.02 ± 0.99%) than controls (3.50 ± 1.48%). However, the orthogonal planned comparisons were not significant (F(1.69) = 2.80, P = 0.09 for NC vs. (MA + MO); F(1.69) = 0.01, P = 0.93 for MA vs. MO). Thus, even though the migraineurs were slightly more sensitive at detecting the briefly presented target compared with controls, the difference was not statistically significant.
Measure of visual cortical hyperexcitability: masking effect with zero mask-test delay
In agreement with previous research, we found a large masking effect for every subject. On average, controls subjects had masked thresholds that were 5.9 (± 1.7) times greater than the unmasked baseline thresholds. Both migraine groups showed greater threshold shifts than the control group (see Fig. 3). Orthogonal planned comparisons revealed a statistically significant difference between NC vs. (MA + MO) (F(1.68) = 4.8, P = 0.03), but no significant difference between MA and MO groups (F(1.68) = 0.24, P = 0.63).
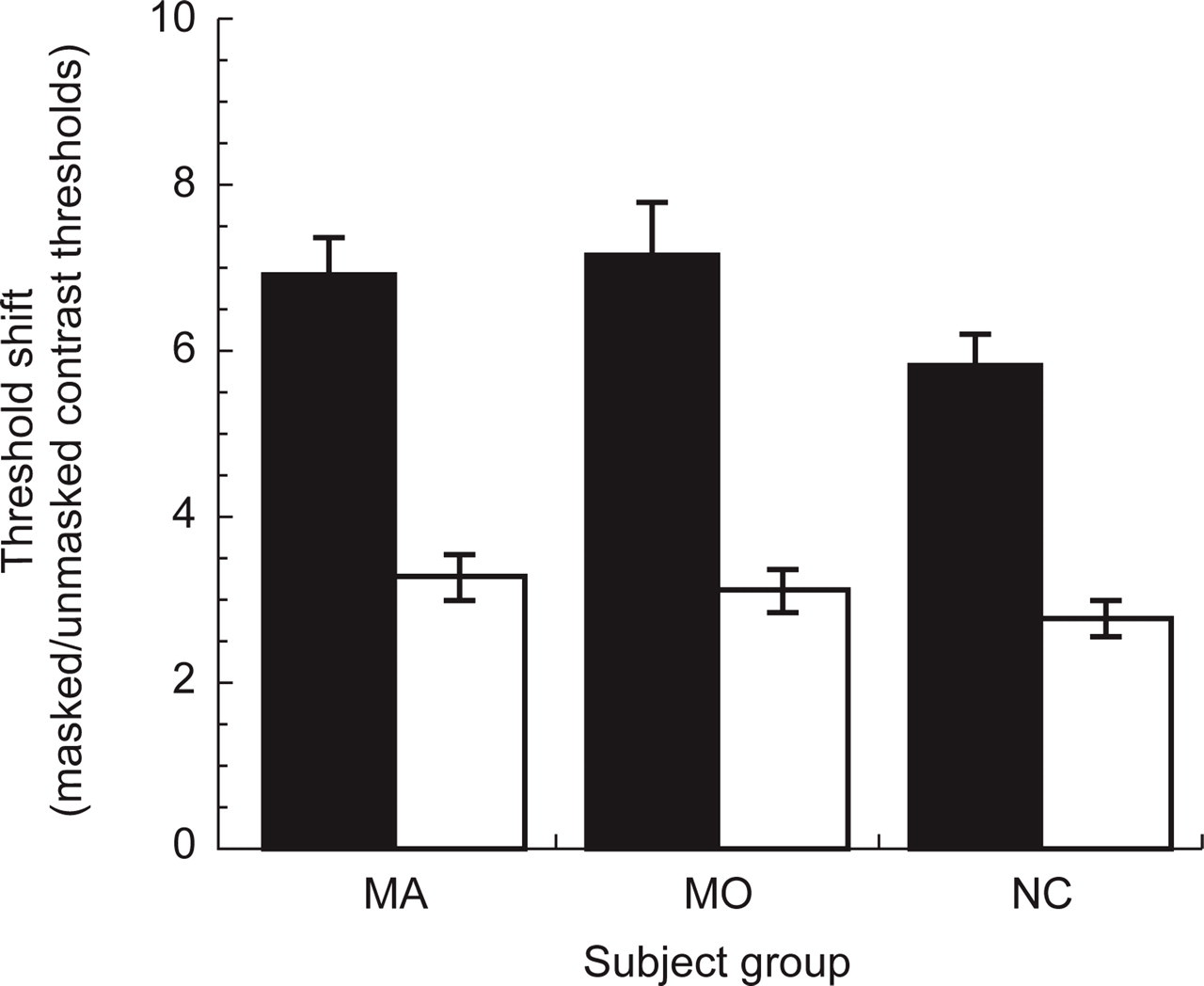
Mean threshold shifts (ratio of masked to unmasked contrast thresholds) in control subjects (NC), and migraine subjects with (MA) and without aura (MO). ▪, Data for the condition with no delay between the onset of the mask and target pattern (where contrast gain controls do not have time to operate); □, data for the 150-ms delay (inhibitory contrast gain controls operate in full).
In summary, we found a robust masking effect that is larger in migraineurs than in migraine-free control subjects.
Measure of inhibition: reduction in masking effect
The mean masking effect in the zero delay condition (5.86 ± 1.7) was larger than the condition with the delay (2.79 ± 1.1), resulting in an average ratio in threshold shifts of 2.2 (± 0.7) in control subjects. Contrary to our hypothesis, migraineurs did not show smaller ratios. The ratios of threshold shifts between the zero delay and 150 ms delay conditions in MA and MO subjects were 2.3 (± 0.67) and 2.5 (± 1.05), respectively. None of the planned comparisons were significant (F(1.68) = 0.61, P = 0.44 for NC vs. (MA + MO) and F(1.68) = 1.34, P = 0.25 for MA vs. MO).
In summary, we found no evidence to support a difference between migraineurs and normal controls on this measure of inhibition.
Effect of frequency of auras and migraine episodes on dependent measures
If cortical hyperexcitability is a precursor to and underlying cause of migraine, one would expect to find evidence of this in all migraineurs independent of the length of time they have suffered from the disorder, and for this reason we included in our study individuals with a wide range of migraine durations. However, a hypothesis such as that of Chronicle & Mulleners (29), that migraine episodes (in their case, specifically visual auras) cause hyperexcitability, would predict an increasing effect with length of disease and total lifetime number of aura/migraine episodes. The latter figure is very difficult to estimate due to general problems with retrospective estimates (e.g. inaccurate memory), and due to the fact that episode frequency often varies greatly over an individual's lifetime as a function of hormonal factors, life events, etc. We have made crude estimates of lifetime aura and migraine incidence based on subjects' reported number of episodes/year, and total years with migraine. However, we feel these estimates can only safely be used as a basis for ranking subjects in terms of migraine and aura episodes, and not as a basis for statistical analyses based on ratio measures. Therefore we used Spearman's rank correlation analysis to examine the relationship between our measure of cortical inhibition and rank based on total estimated visual auras in the MA group, and rank based on total estimated migraine episodes in both MA and MO groups. These analyses failed to reveal any statistically significant relationship between our measures of inhibition and excitation, and number of auras or number of migraine episodes (see Fig. 4).

The relationship between the estimated number of total lifetime migraine episodes for migraine subjects with aura (MA) (Δ) and migraine subjects without aura (MO) (○) and the measures of excitation and inhibition are shown on the top left and top right panels, respectively. The relationship between the estimated total number of auras experienced (by MA subjects) and the measures of excitation and inhibition are shown in the lower left and lower right panels, respectively. Simple regression lines are shown for MA (—) and MO (––––). The range of the slopes of the regression lines was −0.0016 to +0.0002. r 2 values ranged from 0.0004 to 0.1.
We also conducted Pearson correlations between age at first migraine, age at first aura, number of years with migraine, or number of years with aura and the dependent measures, as these independent variables can be estimated with much greater accuracy than total lifetime episodes. Again, we did not find any statistically significant relationships.
Discussion
The results of the present study provide further evidence of visual cortical hyperexcitability in migraine. Migraineurs were more sensitive to the high contrast masking pattern, with the result that their thresholds for detecting a target superimposed on a larger mask was significantly poorer than controls. Our results do not however, indicate a loss of inhibition in migraine; the strength of inhibitory feedback contrast gain controls was similar in migraineurs and controls.
On the basis of Chronicle & Mulleners' (29) hypo-thesis, weakened inhibition might be expected to occur exclusively in MA, as a result of the damaging effects of repeated visual auras on inhibitory neurones. Contrary to their hypothesis, we found no evidence of reduced inhibition in migraineurs with or without aura. The strength of our measure of inhibition was similar in MA and MO groups. Normal inhibitory contrast gain control in both MA and MO argues against a reduction in cortical inhibition as an intrinsic characteristic of migraine. Furthermore, the finding of no significant difference between MA and MO, and the fact that for MA subjects we found no relationship between the lifetime incidence of auras and our measure of inhibition, do not support the notion that visual auras cause impaired cortical inhibition. Two other studies have reported mixed results: one found evidence of abnormal inhibition in MA subjects on a metacontrast backward masking task (51), and another did not find strong evidence of this on a test of orientation discrimination (38).
The finding that MA and MO subjects had strikingly similar masking effects in the zero delay condition is consistent with the idea that hyperexcitability is characteristic of both MA and MO. Enhanced excitability in both MA and MO is also suggested by the findings of a potentiated visual evoked potential response (VEP) in MA and MO during prolonged visual stimulation (52), and lower thresholds in MA and MO than in controls for producing visual phosphenes by transcranial magnetic stimulation (53). Moreover, similarities between MA and MO at the onset of an episode have been described (54, 55). Schoenen et al. (55) reported unilateral depression of neural activity in MA and MO using topographical EEG mapping, and Barkley et al. (54) suggested suppression of spontaneous neural activity in MA and MO on the basis of findings from neuromagnetic field measures. Reductions in regional cerebral blood flow during an attack have been well documented in MA (56–61), but have not been reported in MO on the basis of single-photon emission tomography (SPECT) (62, 63) or perfusion weighted imaging (64). However, recently a positron-emission tomography study captured the early stages of a spontaneous migraine attack without aura (65) and found bilateral spreading hypoperfusion in the visual cortex. Overall, these findings suggest some commonalities in the mechanisms underlying the two types of migraine.
If hyperexcitability occurs as a result of repeated migraine attacks, irrespective of the overt aura process, we would expect to find a correlation between lifetime incidence of migraine and our measures of excitability, but this correlation was not significant in our study. Thus, our results point to hyperexcitability as an intrinsic feature of migraine that does not appear to be dependent on repeated migraine attacks. However, because our estimate of lifetime migraine incidence was crude and our samples were not selected specifically to test this hypothesis and therefore were not evenly distributed, we feel this remains an open question.
Hyperexcitability may fluctuate over time interictally as a function of other variables and their complex interactions. Changes in hormones (66, 67), stress levels (68), and diet (69) could modulate neurotransmitter systems and potentially give rise to cortical hyperexcitability. Subjects might be expected to show the least hyperexcitability immediately after a migraine episode, as there is evidence in the literature of transiently depressed visual function in this period (70). On this basis we did not test subjects in the 7 days immediately following an episode. One might expect the greatest hyperexcitability immediately prior to an episode. We did not collect post-test follow-up data on the timing of occurrence of our subjects' next episodes, so we cannot estimate what proportion, if any, of our subjects were in the immediate premigraine period. However, given the wide variation in our subjects' migraine frequency, it is unlikely that this would have been true of a significant proportion of the group, unless our testing procedures actually triggered episodes. The important point is that despite the possibility of within-subject fluctuations over time, our measure of hyper-excitability was sensitive enough to detect a difference between migraineurs and controls interictally.
Our measure of inhibition, based on mechanisms which have been demonstrated electrophysiologically to be present in V1 (41) but not at the level of the LGN (40), would have clearly localized a deficit (had it existed) to striate cortex. The source of a behaviourally measured hyperexcitability is however, more difficult to localize. As mentioned earlier, this could result from enhanced geniculo-cortical input reflecting changes at either that synapse or at earlier levels in the visual system. Alternatively, it could reflect altered intracortical mechanisms affecting the strength of recurrent excitatory networks which serve to amplify stimulation (28). This could involve, for example, altered levels of activity in serotonergic or other brainstem mechanisms which modulate the activity of cortical neurones, or abnormalities in magnesium levels which would affect the efficacy of NMDA receptor-mediated glutaminergic circuits.
The task used in the present study was designed on the basis of the known physiology of primary visual cortex, and classic visual aura symptoms are strongly suggestive of V1 involvement (71, 72). However, Grusser (71) and Baumgartner (73) speculated that the migraine process may also affect extrastriate visual cortical areas, and recent imaging data implicate a large region of occipital cortex in the aura process (61, 64). Therefore, tasks designed to probe functions specific to extrastriate areas might provide additional indicators of hyperexcitability. McKendrick (74) employed limited-lifetime random dot kinematograms, thought to probe the human analogue of primate motion-sensitive area MT, and observed significant elevations in motion coherence thresholds in migraine patients with aura compared with control subjects. Our stimuli were not designed to probe extrastriate areas, but we are currently investigating this possibility further.
In conclusion, the present results do not support the hypothesis of weakened inhibitory processes in the visual system of migraineurs during their headache-free periods. Contrast gain controls functioned at normal capacity in both MA and MO subjects. We did find modest evidence that migraineurs both with and without aura have more difficulty than controls in detecting a low contrast target in the presence of an excitatory masking pattern, suggesting that hyperexcitability may be characteristic of migraine. Although the results point in the direction of hyperexcitability as an intrinsic feature of migraine, we cannot rule out the possibility that it occurs a result of repeated episodes.
Footnotes
Acknowledgements
This research was supported by grants to F.W. by the Medical Research Council of Canada (grant no. 14034). We would like to thank Anna Feindel B.A. R.N., for assisting with subject recruitment and clinical interviewing. We would also like to thank the following neurologists and ophthalmologists for referring patients to our studies: The late Dr J. P. Meloche, Dr F. Andermann, Dr L. Durcan, Dr G. Francis, Dr I. Woods, Dr T. Kirkham, Dr S. Fyles, Dr J. Rubin, Dr S. Murphy, Dr W. E. S. Connolly; as well as Dr P.-P. Tellier and staff at McGill Student Health Services.
