Abstract
Background: In visual metacontrast masking, the visibility of a brief target stimulus can be reduced substantially if it is preceded (forward masking) or followed (backward masking) by a non-overlapping mask. These effects have been attributed to inhibitory processes within the visual system. Two previous studies have used metacontrast masking to assess inhibitory function in migraine and control groups, however, each used different types of masking and obtained different results.
Subjects and Methods: Forward, backward and combined forward and backward masking were compared in migraine (15 with visual aura, 15 without) and control (n = 15) groups. Baseline trials were also included (target only).
Results: For all types of masking, both migraine groups were more accurate than the control group. When performance for the masking trials was expressed relative to baseline, however, there were no significant group differences. Performance in certain conditions nevertheless correlated significantly with migraine frequency and with the recency of the last attack.
Conclusions: The inhibitory processes involved in the masking tasks employed in this study do not appear to be impaired in migraine. Their better overall performance may reflect a sensitivity difference, perhaps as a consequence of a heightened neuronal response, which varies with the migraine cycle.
Introduction
There have been numerous psychophysical, electrophysiological and imaging studies of visual function in migraine, in between attacks, which have been conducted in an attempt to refine models of neural processing underlying this condition. Much of this work has focused on cortical function and whether neuronal excitability differs between migraine and control groups. There is compelling evidence that cortical processing is abnormal in migraine, although the precise form of the abnormality is still under debate.
The dominant model is one of cortical hyperexcitability in migraine, which has been attributed to damage to GABAergic neurons resulting in a lack of cortical inhibition (1–6). Other proposed causes of hyperexcitability include increased excitation from elevated glutamate, low magnesium, mitochondrial abnormalities, a channelopathy or receptor hypersensitivity (7–12).
Alternatively, cortical hypoexcitability, rather than hyperexcitability, has been proposed in migraine. Much of the evidence that is consistent with hypoexcitability has come from electrophysiological studies (13–16), although there is also some psychophysical support (17–19). Hypoexcitability could arise as a consequence of low pre-activation levels from hypofunctioning aminergic or serotonergic pathways, reduced mitochondrial energy reserves, increased cortical inhibition, or reduced excitation.
Other researchers have suggested general models of abnormal cortical excitability in migraine may not be appropriate, as there is evidence that cortical excitability itself may change at different phases of the migraine cycle (20–28). Again, alternative models have been proposed, some suggesting an increase in excitability prior to an attack (27), and others suggesting a normalisation prior to an attack, or a decrease in excitability to hypoexcitability from increased inhibition (23–25,28).
Another line of research has focused on pre-cortical visual processing abnormalities in migraine and has highlighted differences presumed to reflect processing within the lateral geniculate nucleus (LGN) or retina (29–33) but see Harle and Evans (34). Rather than appealing to models of a general neuronal dysfunction in migraine (whether generally hyper- or hypo-excitable) to explain differences between migraine and control groups, these studies have proposed circumscribed deficits, such as retinal lesions (31), or changes within particular neuronal pathways that depend on the task used (29,30,32,33,35–37).
From the studies cited above, it is clear that the nature, extent, and time-course of visual processing abnormalities in migraine are still debated. A difficulty encountered when trying to find psychophysical evidence to support any model of cortical, or pre-cortical, abnormality is determining how the proposed abnormality would affect performance. Shepherd (38) commented that sometimes the practical consequences of a proposed abnormality are not presented in detail and, consequently, that opposing models (such as cortical hyper- vs hypo-excitability) can be formulated to make exactly the same predictions for a particular visual task.
Against this background, it is suggested that, to refine models of neural function in migraine, the precise form, location within the visual system (or other neocortical areas), and functional consequences of any proposed abnormality, should be made explicit. Further, the stage that each participant is in their migraine cycle should be recorded (interictal vs within 48 h either side of a migraine attack). Finally, where possible, experimental paradigms and tasks should be selected where the circuitry underlying a key stage or stages of the driven neural response can be identified and, therefore, where clear predictions from a particular model are possible (33,38). Visual metacontrast masking has been used as one such task (1,2).
In visual metacontrast masking, a brief target stimulus is either preceded (forward masking) or followed (backward masking) by a masking stimulus (Figure 1). Although the target and mask stimuli do not overlap temporally or spatially, nevertheless, the mask can interfere with the perception of the target and even render it invisible (1,2,39–42). The perceptual interference has been attributed to interference within the cortex ever since it was shown that the visibility of a target presented to one eye can be affected by the presentation of a mask to the other eye (43,44): inputs from the two eyes are first combined within the primary visual cortex (V1) (45). More specifically, the reduction in visibility has been attributed to inhibitory processes within the visual system and, particularly for simple displays such as that shown in Figure 1, to inhibition within the early visual cortex (41,42,46).
The target and mask stimuli, after Palmer et al. (1) The upper image shows the target as a complete circle, the lower shows the target as a circle with an arc removed from the right hand side. Both types of target had a diameter of 7.5 mm (0.86° visual angle). The larger (non-overlapping) annular mask had an inner diameter of 9 mm (1.03°) and an outer diameter of 15 mm (1.72°).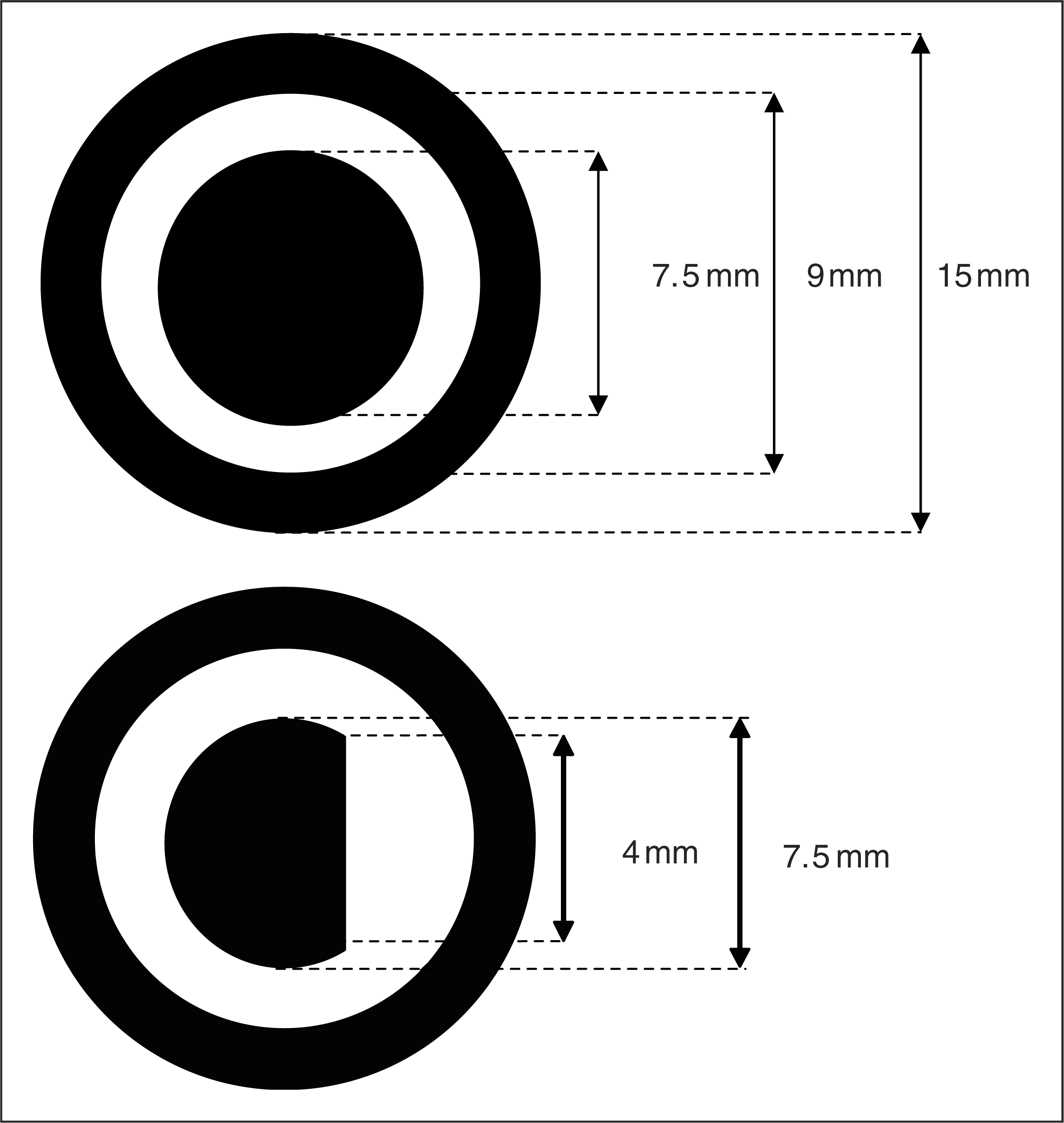
Inhibition within V1 features in several accounts of migraine pathophysiology (3,4,6,8); therefore, a study of visual metacontrast masking in migraine should help to clarify its importance. Two previous studies have used metacontrast masking to assess inhibitory function in migraine and control groups; however, each used different types of masking and obtained different results (1,2). Palmer et al. (1) employed backward metacontrast masking and proposed that, if there is a lack of inhibition in migraine, and if backward masking reflects inhibitory processes, there should be less masking effects for migraine participants. They should, therefore, perform better than the control participants. Palmer et al. (1) employed brief (13 ms) targets and masks with onset lags of 0 ms (simultaneous presentation), 39, 78 and 156 ms (‘stimulus onset asynchronies’; SOA). Their target and mask stimuli are presented in Figure 1.
Palmer et al. (1) included migraine with visual aura (MA), migraine without aura (MO) and control (C) groups. For each group, performance was substantially impaired for the 39 ms target-mask SOA, falling close to chance levels. Performance for the 78 ms target-mask SOA was also somewhat impaired, compared to the 0 and 156 ms target-mask SOA conditions. In line with their predictions, they found less masking effects in MA, but comparable masking effects in their MO and C groups. They further reported a positive correlation between migraine frequency and overall accuracy in MA: the more frequent the attacks the less the effect of the mask.
In a later study, Huang et al. (2) combined forward and backward masking on each trial: the mask was presented briefly before the target, removed, and then displayed again after the target. Huang et al. (2) used symmetrical SOAs between each mask and the targets of 0, 34, 100 or 500 ms. They also reported substantial masking with the 34 ms SOA. Unlike Palmer et al. (1), however, Huang et al. (2) found no significant differences between their MA and C groups although there was a trend for greater masking in migraine (they did not test people with migraine without aura).
The results reported by Palmer et al. (1) and Huang et al. (2) are not directly comparable as they used different types of masking, different stimuli, and different tasks. This study re-examines visual metacontrast masking to try to reconcile the results from these two studies and to examine whether it is a paradigm that can help assess models of abnormal inhibitory visual function in migraine. In this study, forward, backward and combined forward and backward masking were compared in migraine and control groups.
Subjects and Methods
Participants
Participant details in the form of group mean, SD and ranges

Display
The stimuli were created using an experimental script developed in Matlab 7.7 (The MathWorks, Natick, MA, USA) in conjunction with the Psychophysics Toolbox set of functions (48,49). The stimuli were presented on a LaCie Diamondtron 22-inch CRT monitor connected to an Apple Macintosh G4 computer running MacOS X. The CRT monitor had a spatial and temporal resolution of 1024 × 768 pixels, and 100 Hz, respectively. Participants were seated 50 cm from the monitor in an otherwise dark room.
Following Palmer et al. (1), the stimuli consisted of an inner target and an outer non-overlapping and non-contiguous mask (Figure 1). The mask consisted of a black ring (outer diameter 15 mm, inner diameter 9 mm, luminance 1.0 cd/m2). The target was either a whole circle (diameter 7.5 mm, luminance 1.0 cd/m2) or a circle with an arc removed from the right hand side. Both the targets and masks were presented on a light grey background (luminance 108 cd/m2, Michelson contrast 98%). Targets and masks appeared either both above or both below a central black fixation point, positioned so that the nearest outside edge of the mask was 100 mm (1.15°) from the central fixation point. The location of each target and mask combination (above or below fixation) was determined randomly.
Procedure
On each trial, participants were asked to focus on a central fixation point, which was present at all times. They were instructed to press one of two labelled keys to indicate whether the target was seen as a circle with an arc removed on the right hand side, or a whole circle (‘/’, on the left of the keyboard, and ‘Z’, on the right), using their left and right hands, respectively. These keys were selected so that there was congruency between the hand used to report the circle had an arc removed and the side of the target where the important feature was located (the missing arc) so as to avoid the Simon effect (50,51).
This two-alternative forced choice task differed to the task used by Palmer et al. (1), where participants were only required to respond when the target appeared to have one side removed. Their procedure was modified as it was considered likely that it would produce more responses deemed to represent that the circle appeared whole, particularly on ambiguous trials, compared to the forced choice procedure employed here. Participants were asked to respond as quickly and accurately as they could and both reaction times and accuracy were recorded.
Each trial started with the central fixation point for 1000 ms. On backward (BW) masking trials, participants were then presented with the target followed by the mask, while, in forward (FW) masking trials, the mask preceded the target. When both forward and backward masking were combined (B), a mask was followed by the target, which was then followed by a further mask. The fixation point then remained on screen until a response was made. BW, FW and B masking trials were presented in separate blocks.
The ‘target only’ level denotes the baseline trials where the target was presented alone, without a mask.
The order of masking conditions (FW, BW, B) was counterbalanced, and the presentation of each SOA within a block of trials was randomised. For each masking condition, 40 practice trials were followed by 320 experimental trials. This number of trials was chosen based on pilot data, which showed that performance declined if more trials were included. There were 80 trials for each SOA.
Statistical analysis
For the experimental trials, accuracy (mean proportion correct) and reaction times (mean for correct responses only) were calculated for each participant and each SOA in each masking condition. Accuracy was calculated by counting the number of correct responses and dividing by the total number of trials for that condition (80). Thus, the mean proportion correct combines correct responses irrespective of whether each response was that the target was intact or had an arc removed from the right hand side, or whether the target appeared above or below fixation.
The statistical analyses were performed using SPSS v17.0 (SPSS Inc., Chicago, IL, USA). Group differences for the baseline trials, when there was no mask, were assessed with a one-way analysis of variance (ANOVA), with Group as a between-subjects factor (MA, MO, C). For the masking trials, group differences and the effects of SOA were assessed with three separate mixed ANOVAs, one for each masking condition, with the between-subjects factor Group (MA, MO, C) and the within-subjects factor SOA (FW trials: 30, 90, 130 ms; BW trials: 0, 40, 70, 130 ms; B trials: 10, 30, 90, 130 ms, see Table 2).
Each migraine participant provided estimates of their migraine frequency, migraine duration (years experienced) and the days elapsed since the last attack (Table 1). Differences between the two migraine groups (MA and MO) were assessed with three separate Mann–Whitney U-tests, one for each migraine attribute. Spearman’s rank correlation coefficients were used to assess the relationships between the migraine groups’ overall performance for each type of masking trial (averaged across SOA) and each migraine attribute. Non-parametric statistics were performed for these analyses due to the approximate nature of self-reported estimates of each migraine attribute, and due to the range and distribution of the estimates so obtained.
Results
Accuracy rates for the forward and backward masking conditions are shown in Figure 2. Overall, the data for the control group (filled circles) show the expected trends: first, the greatest masking occurred for backward masking with target-mask SOAs between 40 ms and 70 ms. Performance declined substantially at these SOAs, resulting in a characteristic U-shaped function. Second, forward masking was weaker than backward, and occurred only at shorter mask-target SOAs. It is also clear from Figure 2 that the migraine groups performed better than the control group in both masking conditions (filled squares and diamonds), partly replicating the results reported by Palmer et al. (1).
The proportion of correct target judgments for each group in the forward and backward masking conditions (filled symbols) as a function of mask-target, or target-mask, stimulus onset asynchronies, respectively. Performance on the baseline trials are denoted by the open symbols. MA, squares; MO, diamonds; C, circles. Error bars denote one standard error.
Accuracy rates for the condition that combined forward and backward masking are shown in Figure 3 (filled symbols). Performance with the shortest SOA (10 ms) is similar to performance with the simultaneously presented masks and targets (plotted in Figure 2). Within each group, comparable masking occurs for the remaining SOAs as the group averages fall on approximately horizontal lines. Group differences are also apparent, with both migraine groups (filled squares and diamonds) again performing better than the control group (filled circles).
The proportion of correct target judgments for each group in the condition that combined forward and backward masking (filled symbols) and for the baseline trials (open symbols, same as in Figure 2). MA, squares; MO, diamonds; C, circles. Error bars denote one standard error.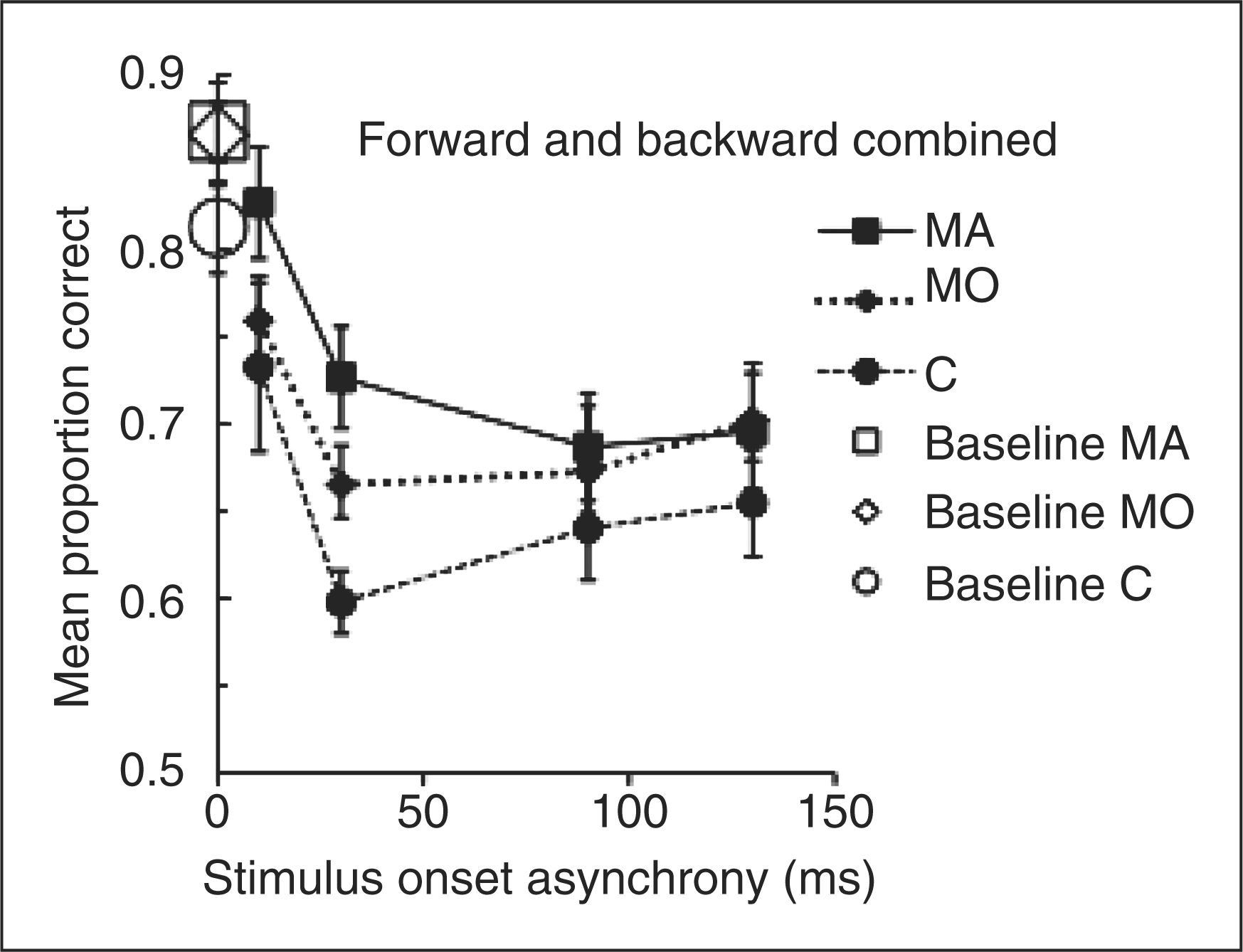
These data do not necessarily indicate that visual metacontrast masking is weaker in migraine; however, since the migraine groups’ performance on the baseline trials (open symbols in Figures 2 and 3), when no mask was present, was also better than the control group’s performance. A one-way ANOVA (factor: Group) on the baseline trial data revealed a significant effect (F(2,42) = 4.3; P < 0.05). Post-hoc Tukey’s HSD tests showed that each migraine group performed better than the control group (MA vs C: P = 0.048, MO vs C: P = 0.034) but did not differ from each other (P > 0.9). To eliminate these baseline differences from the subsequent analyses of the masking trial data, each person’s baseline performance was subtracted from their performance in each masking condition for each SOA.
When baseline performance is taken into account in this way, the group differences for each masking condition largely disappear (Figures 4 and 5). This was confirmed in three separate mixed two-factor ANOVAs (factors: Group, SOA). For each masking condition there was a significant main effect of SOA (FW: F(1.3,54)
1 = 10.8, P < 0.005; BW: F(2.2,90) = 15.8, P < 0.001; B: F(1.8,75) = 11.5, P < 0.001) but no significant main effects involving Group (FW: F(2,42) = 0.03, P > 0.9; BW: F(2,42) = 0.07, P > 0.9; B: F(2,42) = 0.5 P > 0.6). For forward masking only, the Group by SOA interaction approached significance (FW: F(2.6,54) = 2.5, P = 0.07; BW: F(4.3,90) = 0.2, P > 0.9; B: F(3.6,75) = 0.8, P > 0.5).
Mean accuracy scores for the forward and backward masking conditions taking baseline performance into account. Baseline accuracy (when no mask was present) was subtracted from the accuracy for each SOA and for each participant individually. Error bars denote one standard error. Mean accuracy scores for the condition that combined forward and backward masking, taking baseline performance into account. Baseline accuracy (when no mask was present) was subtracted from the accuracy for each SOA and for each participant individually. Error bars denote one standard error.

All data were normally distributed (Kolmogorov–Smirnov tests), but the degrees of freedom have been adjusted for unequal variance using the Greenhouse–Geisser correction.
Parallel analyses were run on reaction times for each group in each condition, which produced no significant effects involving Group (all F values <0.6, P > 0.5).
Palmer et al. (1) also reported a significant association between overall performance (averaged across SOA) and the frequency of migraine, using the number of attacks per year as a correlate. Here, three migraine attributes were correlated with overall performance: migraine frequency (per year); days elapsed since last attack; migraine duration (years with migraine). Since there were no significant differences between the migraine groups with and without aura and each migraine attribute (three separate Mann–Whitney U-tests, P > 0.25), the migraine groups were combined to increase the sample size for the correlations.
Spearman correlation coefficients between overall performance (averaged across SOAs for each masking condition) and migraine frequency (freq) and between overall performance and the time elapsed since the last attack. Overall performance for each masking condition has been adjusted for baseline performance

Discussion
The primary aim of this study was to compare visual metacontrast masking using forward, backward, and combined forward and backward masking in migraine and control groups, to try to reconcile the different results reported by Palmer et al. (1) and by Huang et al. (2) This study has replicated the study by Palmer et al. (1) since it also found better overall accuracy for the MA group for the backward masking trials, compared to the MO and control groups. Here, both migraine groups (MA and MO) performed better than the control group (Figure 2), although the only statistically significant difference was between the MA and control groups.
Although this study has replicated Palmer et al. (1), the present results nevertheless suggest that backward masking is not reduced in the migraine groups tested here. Palmer et al. (1) did not include baseline trials, when no mask was present. Their inclusion in the present study revealed that both migraine groups’ performance was also better than the control group’s performance. Thus, both migraine groups were better at discriminating the briefly displayed targets. When these baseline differences were taken into account, there were no significant differences between the groups (MA, MO, C) in each masking condition (Figure 4).
However, overall performance was correlated with migraine frequency in each masking condition, as was also reported by Palmer et al. (1) for backward masking. These were positive correlations, which indicates that the more frequent the migraine attacks, the better the overall performance (i.e. the less the effects of the masks). The time elapsed since the last attack also correlated with overall performance for the combined forward and backward masking trials, which were the most difficult of the three masking conditions. This negative correlation indicates that the more recently migraine had been experienced, the better the overall performance (i.e. the less the effects of the mask).
This study has also replicated the results reported by Huang et al. (2) for the combined forward and backward masking condition. Huang et al. (2) assessed baseline performance in a pre-test and adjusted the difficulty of their task, prior to performing the masking trials, such that each participant’s baseline accuracy was greater than 90% correct. They subsequently found no significant differences between their MA and control groups in the masking trials (they did not include a MO group). In the present study, both migraine groups performed better than the control group for the combined forward and backward masking condition (Figure 3) but, once again, there were no significant group differences (MA, MO, C) when the baseline differences in performance were taken into account (Figure 5).
Huang et al. (2) did not report the results of the adjustments made to the target-only (baseline) stimuli for each participant following their pre-test, or whether there were group differences. They only presented accuracy data for the target-only trials once task difficulty had been adjusted for each individual, to show that the performance of their MA and C groups had been equated. It is not known, therefore, whether their MA group performed better than the control group for the un-adjusted target-only trials during the pre-test, as found here (Figure 3).
Cortical inhibition and masking
As described in the Introduction, the reduction in target visibility in simple forward or backward masking displays (such as those used by Palmer et al. (1) or Huang et al. (2)) has been attributed to inhibitory processes early in the visual system (41,42,46). Until recently, the inhibition was ascribed to early cortical circuits following: (i) demonstrations of dichoptic masking, where the target and mask are presented to different eyes (43,44); and (ii) early reports of neurophysiological correlates of forward and backward masking using single cell recordings at the level of the primary visual cortex (V1) (42,52), but not at the LGN (53), but see Macknik and Martinez-Conde (46,54).
For example, Macknik and Livingstone (42) compared perceptual masking in humans with neurophysiological recordings from area V1 in awake and anaesthetised monkeys. They found that neurons in V1 had a bi-phasic response to a briefly presented stimulus, responding transiently when it appeared (an on-response) and when it was removed (an after-discharge/off-response). They further reported that the time-course of inhibition of the on- and off-responses of V1 cells that responded to a target stimulus, by forward and backward masks, matched the time course of forward and backward masking in humans. Other research has also reported psychophysical masking effects in primates matches those in humans (52).
Macknik and Livingstone (42) concluded both on- and off-responses are important to the visibility of a target and masking is likely to occur if either is inhibited. Thus, backward masking was attributed to cortical inhibition of the transient after-discharge of neurons that respond to the target by the activity of neurons that respond to the subsequent mask. Equivalently, forward masking was attributed to cortical inhibition of the on-response of neurons that respond to the target by the activity of neurons that respond to the preceding mask.
Based on Macknik and Livingstone’s research (42), Palmer et al. (1) concluded that the reduced masking in the MA group was consistent with dysfunctional inhibitory mechanisms in V1. They attributed the dysfunctional inhibition to a disruption of GABAergic inhibitory control, which may be caused by the visual aura. They also reported that MA patients who were taking sodium valproate, a GABA agonist, showed larger masking effects and indeed their performance did not differ from the control group. They suggested that valproate restored a normal inhibitory potential and so increased masking effects in the medicated MA group. They related a general cortical hyperexcitability, which may be reversed pharmacologically, indirectly to these suggestions. Future research that examines the effects of sodium valproate, or other prophylactic medication, on performance for both target-only (baseline) and masking trials is needed to develop these ideas.
Huang et al. (2) combined their study with functional magnetic resonance imaging. They presented their participants with displays that were similar to those used in their psychophysical test of masking, but were passively viewed during the scans. They reported less activation in V1 in the mask-target-mask condition than in a control condition in which the (much smaller) fixation dot replaced the target (mask-fixation-mask), which they argued showed that masking does indeed involve inhibitory processes. Following Macknik and Livingstone (42), they suggested less activation in the mask–target–mask condition resulted from a strong overall suppression of neuronal activity due to reciprocal inhibition between the first mask’s off-response and the target’s on-response and between the target’s off-response and the second mask’s on-response.
Since there were no statistically significant group differences, Huang et al. (2) concluded that cortical inhibitory control is not abnormal in migraine, at least for the neural pathways involved in masking. They further reported no evidence for hyperneuronal activity in MA and concluded hyperexcitability, or dysfunctional inhibition, if it exists, occurs only for specific conditions. The evidence their study provides for or against hyperneuronal activity in MA is undetermined, however, without knowing whether the adjustments needed to achieve their high baseline accuracy rates (> 90%) differed between their MA and C groups.
The present results also suggest that inhibitory control within the neural pathways involved in masking is not abnormal in migraine, since there were no significant differences between the groups (MA, MO, C) in any of the masking conditions once the data were adjusted for baseline performance. These results do not, however, support the conclusion that inhibition within the neural pathways involved in masking in V1 is normal in migraine. The traditional explanation that forward and backward masking reflect cortical inhibitory circuits now appears correct only for dichoptic displays, where the target is presented to one eye and the mask is presented to the other (46,55). More recent work has indicated that when masking displays are presented monoptically (as was used in the present study and in the work it was based on (1,2)), cells that respond to the targets can be inhibited by both forward and backward masks at the level of the LGN, V1, and higher visual cortical areas (46). Monoptic presentation occurs when displays are viewed monocularly, or when the same display is presented to the two eyes. Future research could use dichoptic masking to assess cortical inhibitory circuits in migraine without sub-cortical involvement.
Baseline and overall performance
The migraine groups’ better baseline performance, and better overall performance in each masking condition, compared to the control group, could reflect a difference in sensitivity between the migraine and control groups, or some non-specific difference, such as motivation. For example, the migraine groups’ better performance may reflect a heightened cortical response to these briefly presented stimuli, which boosts the signal to noise ratio within the visual system. The correlations with migraine frequency and the time elapsed since the last attack could, further, indicate that the difference in sensitivity may vary with the migraine cycle, as has been shown with other psychophysical and electrophysiological measures (24–32). Alternatively, motivation and vigilance are required to detect the shape of the briefly displayed targets, particularly when they are masked. It is possible that the migraine groups’ better performance reflects a greater motivation to perform well when faced with a difficult task, compared to the control group, because of their personal interest in contributing to migraine research. Further, those with frequent migraine, or who had recently experienced migraine, may be the most motivated, which could account for the correlations between overall performance and migraine frequency or the days elapsed since the last attack.
However, when other demanding visual psychophysical tasks have been used to compare migraine and control groups, better performance by the migraine group is not usually reported (reviewed by Chronicle and Mulleners (56) and Harle et al. (57)), despite presumably similar motivational differences between the migraine and control groups. For example, processing of spatial frequency (58–60), flicker (59,61), colour (32,35–37,62), form (63) and motion (61,63,64) have all been reported to be impaired in migraine in that they fail, on average, to detect or discriminate stimuli that would be faintly discernible to people without migraine.
On the other hand, none of the studies just cited used displays as brief as the target and mask displays presented here (10 ms). Shepherd (19,38) reviewed three models of hyperexcitability in migraine: (i) increased background neuronal noise in the visual system combined with a greater response to a visual stimulus; (ii) increased background neuronal noise in the visual system without a heightened response to a visual stimulus; and (iii) a greater response to a visual stimulus without increased background neuronal noise. Shepherd (19,38) proposed a short-lived version of the third model, which predicts better detection of visual stimuli with short presentation times, but impaired performance when presentation time increases as the initially heightened response cannot be maintained. This model is consistent with the better performance found for both migraine groups on the baseline trials (see also Antal et al. (64)). Future research could test this model by reassessing differences between migraine and control groups using a series of brief and longer display durations. This model cannot be used to formulate predictions for a masking study until ‘short-lived’ can be defined.
Conclusions
The overall better performance in migraine may reflect a difference in visual sensitivity, such as a short-lived heightened response to visual stimuli, rather than a more general difference such as motivation. Although the contribution of any heightened response to the overall better performance remains uncertain, the inhibitory processes involved in monoptic masking do not appear to be impaired in migraine in the interictal period (see also McColl and Wilkinson (12)). In this study, participants were not recruited if they had experienced migraine within the 48 h prior to the test session, and none reported experiencing a migraine in the subsequent 48 h. Future research could examine dichoptic masking, to isolate cortical circuits, and whether performance changes in the peri-attack interval.
Footnotes
Acknowledgements
This work was funded by Action Medical Research, Migraine Action Association, Search, the University of London Central Research Fund and Birkbeck College Faculty of Science. The research was completed at Birkbeck College, but was prepared for publication while Dr Shepherd was employed at the University of Sydney.

