Abstract
This study was designed to investigate whether small ubiquitin-like modifier (SUMO) conjugation is activated after focal cerebral ischemia. Transient ischemia induced a dramatic increase in SUMO2/3 protein conjugates. The most pronounced changes were found in the parietal cortex. SUMO2/3 conjugation was particularly high in neurons located at the border of the middle cerebral artery territory where sumoylated proteins translocated to the nucleus. Considering the marked effect of SUMO conjugation on the function of target proteins, it is very likely that the postischemic activation of sumoylation has a significant effect on the fate of neurons exposed to transient ischemia.
Keywords
Introduction
Conjugation of target proteins with small ubiquitin-like modifier (SUMO) is a posttranslational protein modification that has recently been identified (Matunis et al, 1996; Mahajan et al, 1997). Whereas ubiquitin targets proteins for degradation at the proteasome, sumoylation has a major impact on the stability, subcellular localization, and activity of cellular proteins including transcription factors, transporters, and membrane channel proteins (Hay, 2005). Four SUMO proteins have been identified, SUMO1–4. Similar to ubiquitin, SUMO is bound to lysine residues of target proteins in a complex process involving activating, conjugating, and ligating enzymes. Recently, we have observed that sumoylation is massively activated after transient forebrain ischemia (Yang et al, 2007), but SUMO conjugation has not been investigated before in experimental models of stroke. Here, we show that transient focal cerebral ischemia of 90mins duration dramatically activates SUMO2/3 conjugation. Vascular occlusion of only 15mins is sufficient to massively activate this process.
Materials and methods
Animal Experiments
The following study was approved by the Duke University Animal Care and Use Committee (Durham, NC, USA). Male Wistar rats of 10 weeks of age were housed in a temperature-controlled environment with a 12 h light-dark cycle. Animals were fasted with free access to water 12 h before starting the experiment. Five animals were used for each experimental group (control, 15 or 90mins ischemia and 30mins or 6 h of reperfusion). Transient focal cerebral ischemia was induced by unilateral occlusion of the right middle cerebral artery (MCA). Rats were anesthetized in a chamber with 5% isoflurane in 100% oxygen. The trachea was intubated, and lungs mechanically ventilated with 40% oxygen/balance nitrogen. The inspired isoflurane concentration was reduced to 1.5% to 2%. A thermistor was subcutaneously implanted beneath the right temporal muscle adjacent to the skull, and pericranial temperature was maintained at 37.5°C ± 0.2°C by surface heating or cooling. Surgery was performed with aseptic techniques, and all surgical fields were infiltrated with 0.25% bupivacaine. The tail artery was cannulated to monitor mean arterial pressure and sample blood. Middle cerebral artery occlusion was performed as described elsewhere (Sakai et al, 2007). At the end of the experiments, animals were deeply anesthetized with isoflurane and decapitated. Control animals were deeply anesthetized with isoflurane and decapitated. Brains were quickly removed, frozen in 2-methylbutane cooled to −40°C, and stored at −80°C until being analyzed.
Western Blot Analysis
Brains were cut in a cryostat set at −20°C up to the plane of the striatum. Samples were taken from the right ischemic and left contralateral hemispheres from the parietal cortex, the striatum, and the penumbra. Frozen tissue specimens were weighed in the cold, and the same volume of extraction buffer per milligram tissue was used for homogenization. Protein extraction and Western blot analysis was performed as described before (Yang et al, 2007). For analysis of ischemia-induced changes in levels of SUMO2/3 conjugates, the high-molecular-weight area in each lane was cropped and analyzed, as indicated in the legends to the respective figures.
Microscopy
Brains were fixed with paraformaldehyde and embedded in paraffin. After deparaffinization, immunohistochemistry was performed using the Immunoperoxidase Secondary Detection System (Chemicon, Billerica, MA, USA). Sections were treated with 3% hydrogen peroxide and incubated with blocking buffer at room temperature. Sections were incubated with SUMO1 or SUMO2/3 antibodies (1:500) for 16 h at 4°C. Sections were then incubated with biotinylated secondary antibody, followed by incubation with streptavidin horseradish peroxide. A diaminobenzidine substrate was used to visualize the target antigen. For confocal microscopy, sections were subjected to immunofluorescence staining in a procedure similar to the immunohistochemistry protocol. After overnight incubation with a mixture of rabbit anti-SUMO2/3 (1:500) and mouse anti-NeuN (1:500; Chemicon), sections were incubated with Alexa Fluor 594-conjugated goat anti-rabbit IgG (1:500; Invitrogen) and Alexa Fluor 488-conjugated goat anti-mouse IgG (1:500; Invitrogen) at room temperature. DNA was stained with DAPI (4,6-diamidino-2-phenylindole) in a mounting medium (Santa Cruz Technology, Santa Cruz, CA, USA). Images were captured on a LSM 510 confocal microscope (Carl Zeiss Microimaging) using a × 40/1.3 Plan Neofluar oil objective.
Statistical Analysis
One out of five animals of the 90 mins MCA occlusion and 30mins reperfusion groups did not respond to filament insertion and was therefore excluded from statistical analysis. Data are presented as means ± s.d., with four (90 mins ischemia and 30 mins of reperfusion) or five independent brain samples per group. Statistically significant differences between groups were evaluated by Student's t-test for paired samples. A P-value of ≤95% was taken to indicate significant differences.
Results
The pattern of SUMO conjugation present in control brains was identical to the pattern found in the contralateral hemisphere of rats subjected to transient focal cerebral ischemia (data not shown). Changes in SUMO conjugation in the ischemic hemisphere were therefore related to the extent of SUMO conjugation in the nonischemic contralateral hemisphere. Transient focal cerebral ischemia did not cause major changes in SUMO1 conjugation (data not shown), but it induced a dramatic increase in SUMO2/3 conjugation (Figure 1). A massive increase in SUMO2/3 conjugation was already observed after 90 mins ischemia and 30 mins of reperfusion (Figure 1A). The increase in protein sumoylation was most pronounced in the parietal cortex of the ischemic hemisphere and markedly less so in the striatum and penumbra. Small ubiquitin-like modifier-2/3 conjugation increased 20.9 ± 1.6, 5.0 ± 1.1, and 4.3 ± 2.1-fold in the parietal cortex, striatum, and penumbra of the ischemic hemisphere, respectively, whereas free SUMO2/3 levels (the protein band at approximately 17kDa) decreased to about 50, 70, and 50% of control in these regions. Changes in SUMO2/3 conjugation and in free SUMO2/3 protein levels were even more dramatic after 6 h of reperfusion (Figure 1B). The dramatic increase in SUMO2/3 conjugation, most evident in the parietal cortex, appeared as a smear at high molecular weight, indicating many sumoylated proteins. Furthermore, several protein bands that were only faint in samples from the left nonischemic hemisphere were clearly more prominent in samples from the ischemic right hemisphere (e.g., the band at approximately 100 kDa). To elucidate the possible significance of the SUMO conjugation for the pathological process induced by transient focal cerebral ischemia, we performed the following experiments and analyses: we exposed animals to a short preconditioning period of transient MCA occlusion, we performed immunohistochemistry to identify the region where SUMO conjugation is most markedly activated after ischemia, and we used confocal microscopy to clarify whether sumoylation is activated in neurons.
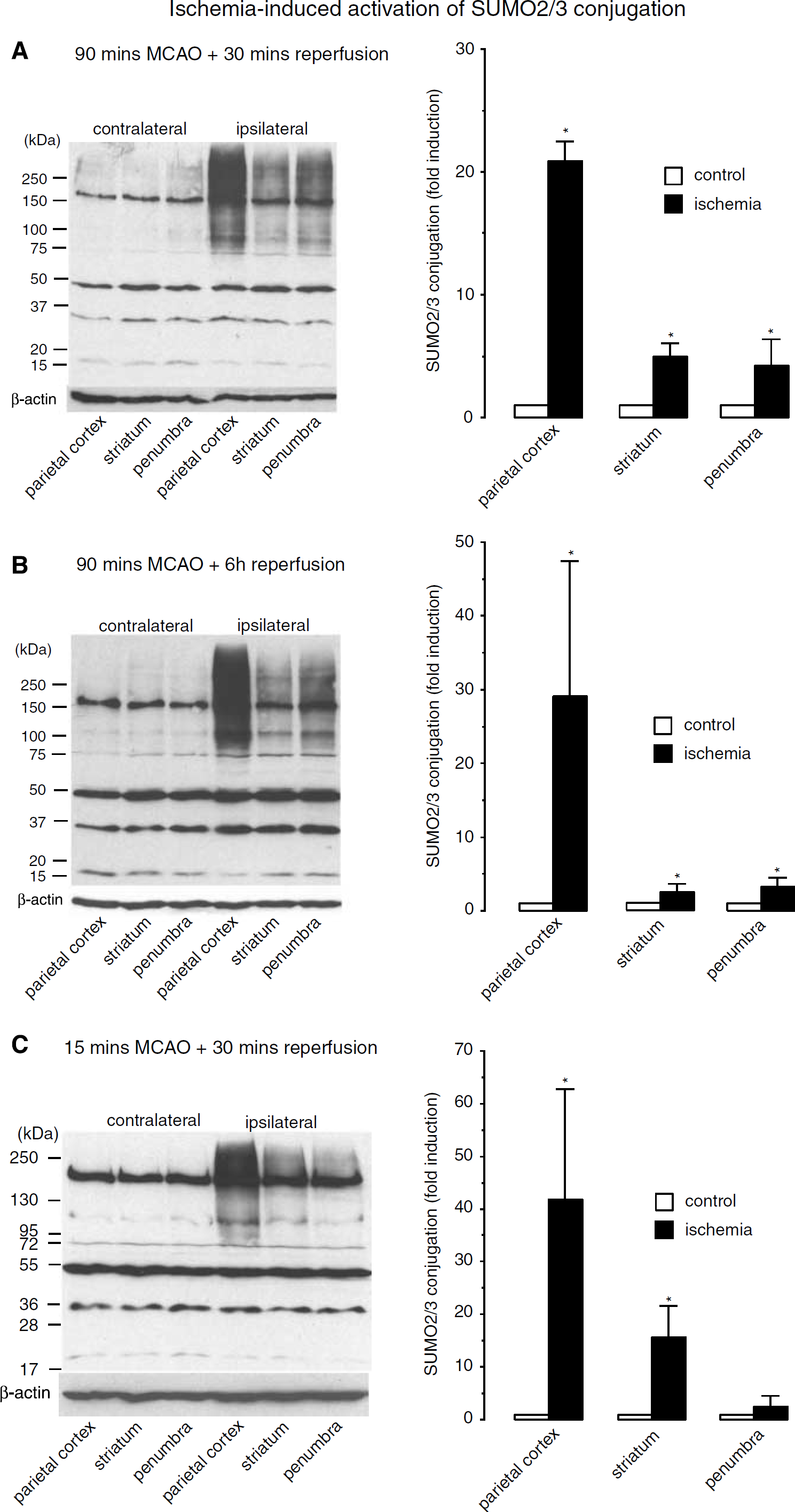
Effect of transient MCA occlusion on protein modification with SUMO2/3. Rats were subjected to 15 or 90 mins right MCA occlusion and 30 or 6 h of reperfusion. Brain samples were taken from the parietal cortex, striatum, and penumbra of the right ischemic and left nonischemic hemispheres. The pattern of SUMO2/3 conjugation was evaluated by Western blot analysis. Ischemia-induced changes in the pattern of SUMO2/3 conjugation were evaluated after 90 mins MCA occlusion and 30 mins of reperfusion (
The pattern of SUMO2/3 conjugation induced by 15mins MCA occlusion and 30mins reperfusion is illustrated in Figure 1C. For quantification, the molecular weight area above the strong band at about 170kDa in each lane was cropped and analyzed. Small ubiquitin-like modifier-2/3 conjugation in the ipsilateral ischemic hemisphere was found to be increased 42 ± 21.0, 16 ± 6.0, and 3 ± 2.0-fold in the parietal cortex, striatum, and penumbra, compared to the same regions in the contralateral hemisphere (Figure 1C). When we analyzed SUMO2/3 conjugation in animals subjected to 90 MCA occlusion and 30 mins reperfusion in the same way, we found 41 ± 11.7, 9 ± 7.0, and 14 ± 8.0-fold increases in levels of SUMO2/3-conjugated proteins.
In the next step, we performed immunohistochemistry and confocal microscopy to identify the brain regions exhibiting the most pronounced increase in SUMO conjugation and to verify whether SUMO2/3 conjugation is activated in neurons after ischemia. In the contralateral nonischemic hemisphere, SUMO2/3-conjugated proteins were present in the cytoplasm and, to a much lesser extent, also in the nucleus (Figure 2A). After ischemia, SUMO2/3-conjugated proteins were highly concentrated in the nucleus of cells located in the border of the MCA territory.
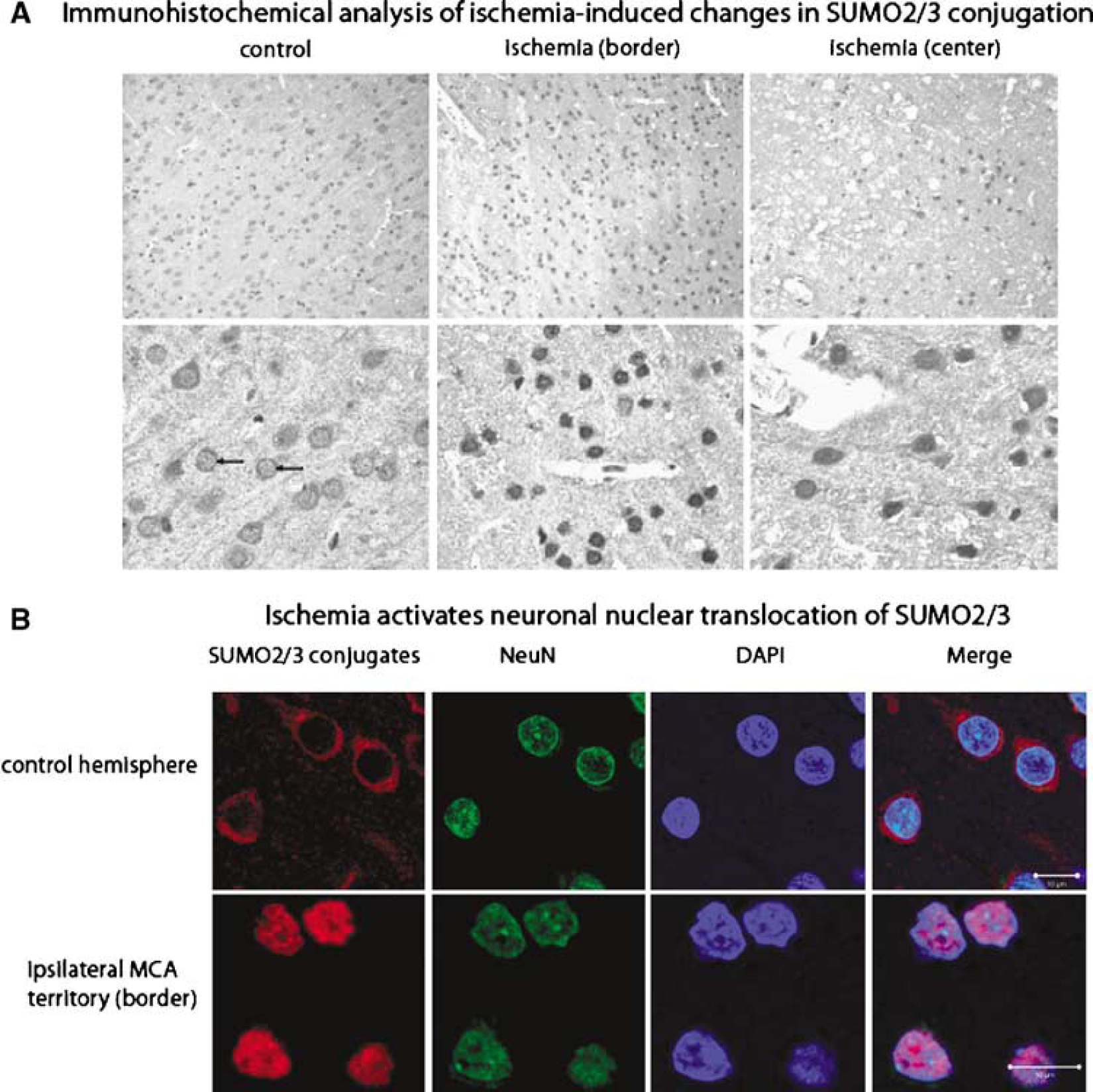
Histochemical analysis (
To verify whether SUMO2/3 conjugation was activated in neurons after transient focal cerebral ischemia, we used confocal microscopy (Figure 2B). The SUMO2/3 antibody was used to identify sumoylated proteins, DAPI staining of DNA was used to identify cells with intact nuclear membranes, and NeuN staining was used to identify neurons. Analysis was performed at the border of the MCA territory where cells showed strong nuclear SUMO2/3 immunoreactivity (Figure 2A). In control cells, SUMO2/3 immunoreactivity was detectable in the cytoplasm but not in the nucleus. Transient cerebral ischemia induced a dramatic translocation of SUMO-conjugated proteins from the cytoplasm to the nucleus. All DAPI-stained cells that were positive for NeuN showed strong SUMO immunoreactivity (Figure 2B).
Discussion
In a recent publication, the group of John Hallenbeck investigated changes in SUMO conjugation during hibernation in ground squirrels (Lee et al, 2007). SUMO conjugation was massively activated in hibernation torpor, and the authors concluded that activation of protein sumoylation may help brain cells to survive the otherwise lethally low levels of blood flow reduction during the state of hibernation. This assumption was corroborated by the observation that SUMO overexpression made SHSY5Y cells in culture more tolerant to transient oxygen/glucose deprivation (Lee et al, 2007). If activation of protein sumoylation does indeed make cells more tolerant to the severe form of metabolic stress induced by transient ischemia, it could open up fascinating new avenues for therapeutic intervention. To date, experimental stroke research has focused mainly on identifying proteins and signal-transduction pathways involved in the pathological process triggered by a transient interruption of blood supply and culminating in neuronal cell death, in the hope of designing new drugs that could block this process. An alternative strategy to protect brain cells from ischemic cell death would be to activate processes that make cells more tolerant to ischemia. Elucidating the mechanisms involved in stress-induced activation of SUMO conjugation could help to achieve this goal.
It is well established that the localization, activity, and stability of transcription factors, nuclear pore proteins, and other nuclear proteins that play a role in genome stability is modified by SUMO conjugation (Melchior et al, 2003; Ulrich, 2005). Small ubiquitin-like modifier deletion impairs silencing at heterochromatin regions, and sumoylation has been shown to link methylation of DNA to histones (Shin et al, 2005; Uchimura et al, 2006). This suggests that sumoylation could be involved in the regulation of heterochromatin formation and gene silencing. Although most of the transcription factors investigated so far are inactivated by SUMO conjugation, the heat shock factor and the hypoxia-inducible factor are activated by sumoylation. To elucidate the possible impact of this process on the fate of cells exposed to transient ischemia, it will be of significant interest to identify proteins SUMO2/3 conjugated after ischemia. Furthermore, assuming a protective function of the sumoylation process, it would be of tremendous importance for stroke research to develop strategies to activate SUMO conjugation and thus to make the brain more resistant to the severe form of metabolic stress caused by a transient interruption of blood supply.
Footnotes
Acknowledgements
We are grateful to Dr John M Hallenbeck, National Institutes of Health, for providing SUMO1 and SUMO2/3 polyclonal antibodies.
