Abstract
Clinical and experimental data support a role for the intact cortex in recovery of function after stroke, particularly ipsilesional areas interconnected to the infarct. There is, however, little understanding of molecular events in the intact cortex, as most studies focus on the infarct and peri-infarct regions. This study investigated neuronal immunoreactivity for hypoxia-inducible factor-1α (HIF-1α) and vascular endothelial growth factor (VEGF) receptor-2 (VEGFR-2) in remote cortical areas 3 days after a focal ischemic infarct, as both HIF-1α and VEGFR-2 have been implicated in peri-infarct neuroprotection. For this study, intracortical microstimulation techniques defined primary motor (M1) and premotor areas in squirrel monkeys (genus
Keywords
Introduction
After stroke, peri-infarct regions have reduced oxygen delivery as a result of a reduction in blood flow surrounding the infarct (Marti et al, 2000; Wei et al, 2001). This lack of oxygen (i.e., hypoxia) places peri-infarct neurons at risk of apoptotic or necrotic cell death. This study investigated the neuronal patterns of immunoreactivity for hypoxia-inducible factor-1α (HIF-1α) protein and vascular endothelial growth factor (VEGF) receptor-2 (VEGFR-2) to address how hypoxia affects neurons in a non-human primate model of focal cortical ischemia. The neuronal immunoreactivity of HIF-1α and VEGFR-2 is expected to increase in areas of cortex that become hypoxic, due to injuries such as stroke, based on previous findings (Bergeron et al, 1999; Chavez and LaManna, 2002). Unique to this investigation was the evaluation of both HIF-1α and VEGFR-2 in presumably non-hypoxic neurons interconnected to and functionally associated with the infarct. Neurons projecting to an area of infarct may be at risk of cell death due to a loss of retrograde neurotrophin transport (Skaper and Walsh, 1998), whereas neurons receiving projections from an infarct are at risk of death because of lost excitatory input (Stein, 1998). The survival of these interconnected neurons may be critical, however, as clinical and experimental evidence suggests that intact ipsilesional regions of the brain undergo molecular, anatomic, and physiologic changes in response to the infarct and can aid functional recovery (Dancause et al, 2005; Frost et al, 2003; Liu and Rouiller, 1999; Seitz et al, 1998).
The HIF-1α subunit functions as the main regulator for oxygen homeostasis and is stabilized under hypoxic conditions in the peri-infarct region, but not in contralateral or control cortices, after stroke (Bergeron et al, 1999; Marti et al, 2000). One transcriptional target of post-stroke HIF-1α is the gene encoding VEGF, which has been implicated in the post-stroke recovery processes of angiogenesis and neuroprotection (Sun et al, 2003; Wang, 2003; Zhang et al, 2000). Neurons express one of four receptors for VEGF, VEGFR-2, throughout adulthood in both the cortex and hippocampus (Yang et al, 2003). After stroke, VEGFR-2 is the main receptor for the action of VEGF, though the receptor does not have a promoter region to allow for upregulation by HIF-1α. Instead, VEGF directly upregulates VEGFR-2 mRNA through either increased mRNA stability or gene transcription (Shen et al, 1998). Experimental data show that neurons can upregulate VEGFR-2 expression as soon as 1 h post-infarct (Hayashi et al, 2003), and clinical data suggests that upregulation can be maintained through 150 days post-stroke in the infarct and peri-infarct regions, but not in the contralateral cortex (Issa et al, 1999).
We previously examined neuronal VEGF protein expression levels in infarct, peri-infarct, and remote cortical regions at 3 days after a focal ischemic infarct in motor cortex (Stowe et al, 2007). Neurons in both the ventral premotor (PMv) hand representation, a remote region axonally interconnected to the infarct, and the primary motor (M1) hindlimb representation, significantly increased immunoreactivity for VEGF protein. This study continues the investigation into remote molecular events in these functionally defined motor areas by examining neuronal immunoreactivity for HIF-1α protein and VEGFR-2 in adjacent cortical sections within the same animals.
Materials and methods
Surgical and Mapping Procedures
All surgical procedures were conducted under aseptic conditions and in accordance with protocols approved by the University of Kansas Animal Care and Use Committee (KU IACUC protocol no. 2004-1470). Seven adult squirrel monkeys (genus
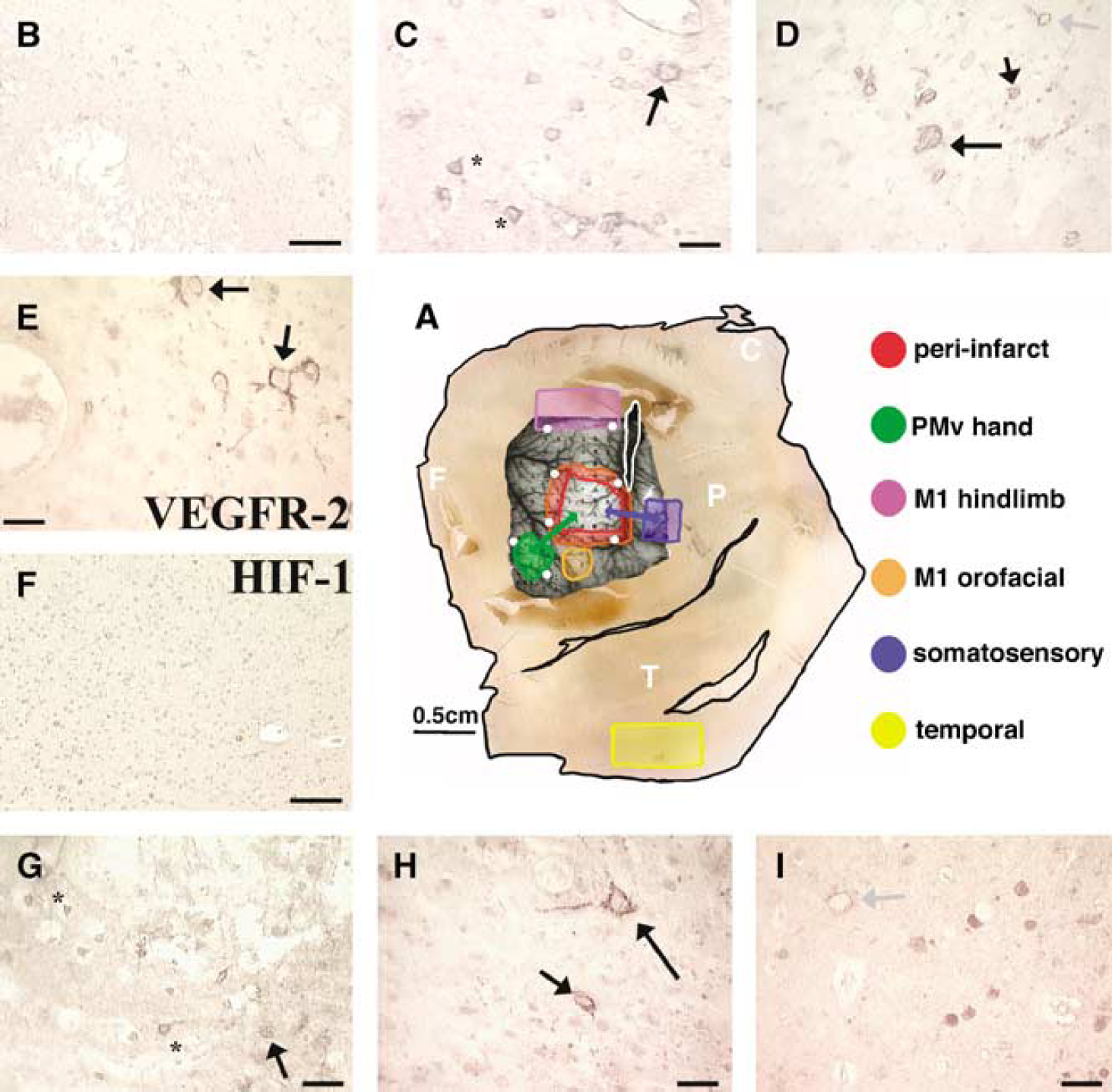
(
Methods are identical to previous published methods (Stowe et al, 2007). Briefly, a craniotomy contralateral to the preferred hand exposed areas of primary and secondary motor cortices, as well as primary somatosensory cortex. Intracortical microstimulation mapping techniques were used to identify the borders of the M1 hand, the PMv hand, and the lateral extent of the M1 hindlimb representations. One-micron polystyrene beads (Polysciences Inc., Warrington, PA, USA) were injected around each of these movement representations to identify during further processing and analysis. Microforceps connected to a bipolar electrocoagulator induced an infarct through permanent occlusion of cortical vessels supplying the M1 hand area in the experimental groups (animals 601, 563A, 602, and 653). This technique consistently produces infarcts throughout the gray but not the underlying white matter (Nudo et al, 1996). Control animals (animals 155, 597, and 598) did not undergo the infarct procedure.
Histologic Procedures
Three days after the surgical procedures, animals were deeply anesthetized and perfused through the left ventricle (0.1 mol/L PBS, pH 7.25; 3% paraformaldehyde). The cerebral cortex was dissected from surrounding tissue, flattened between glass slides, and kept in 3% paraformaldehyde/0.1 mol/L PBS for 72 h (Dancause et al, 2006b). Each hemisphere was dehydrated, paraffin-embedded, and sectioned tangential to the cortical surface at a 12-μm thickness on a rotary microtome. Sections were exposed to primary antibodies to stain for (a) neurons (mouse anti-NeuN; 1:100; Chemicon); (b) HIF-1α protein (mouse anti-HIF-1α; 1:200; Chemicon); and (c) VEGFR-2 (mouse anti-VEGFR-2; 1:200; Chemicon). Procedures for blocking and secondary labeling followed the Rapid Antibody Procedure in the mouse Vectastain ABC Elite kits (Vector labs, Burlingame, CA, USA).
Stereology
In each animal, five serial sections, each 10 sections apart, were randomly selected for analysis and visualized on an Axioplan 2 Microscope (Zeiss, Jena, Germany). Borders of the motor areas were identified in every section using the polystyrene bead injection sites (Stowe et al, 2007) or by topographical references based on criteria established in prior studies in this laboratory and others (Dancause et al, 2006b; Huang et al, 1988). Cell numbers were estimated using the Optical Fractionator (West et al, 1991). One marker was placed on every neuron with an intact cell membrane, and another marker on each neuron that also had positive staining for HIF-1α protein or VEGFR-2 expression. The coefficient of error (CE); (Gundersen et al, 1999) for total neuron counts was between 0.03 to 0.08 for all areas in either control or experimental animals for both HIF-1α and VEGFR-2 estimations, excluding the M1 hand infarct in the experimental animals. This area had a CE range of 0.08 to 0.16, due to the low number of cells counted in the infarct region. We determined the ratio of the mean CE2 to the coefficient of variance (CV)2 to establish the contribution of stereological analysis to the observed group variance (Gundersen et al, 1999; Slomianka and West, 2005). The CE2/CV2 for all cortical regions was between 0.01 and 0.25 for both the HIF-1α and the VEGFR-2 data sets, indicating that stereological procedures contributed little to the observed group variances.
Statistical Analysis
As each physiologic region varied in surface area both within and between animals, raw neuronal population estimates were converted to neurons per unit volume (total neuron estimation/region of interest in cubic mm) to facilitate comparison. One-way analysis of variance compared the neurons/mm3 between groups (experimental and control; arcsin-transformed) for each physiologic region (JMP statistical software, Cary, NC, USA). The ratio of neurons that were immunoreactive for the antigen of the protein or receptor of interest to total neuronal population was calculated for each physiologic region in each group. Values are reported as mean±standard deviation (s.d.). The correlation between estimates of neuron numbers immunoreactive for VEGF and VEGFR-2 within each physiologic area was assessed using linear regression analysis on the ratios of arcsin-transformed neuronal densities of VEGF or VEGFR-2 to total neurons. Statistical outcomes were considered significant for
Results
Identification of Intracortical Microstimulation-Defined Representations and the Area of Infarct
Intracortical microstimulation mapping procedures identified the M1 hand, PMv hand, and M1 hindlimb representations (Figure 1A). Examination of the tissue showed that the area of infarct was almost exclusively confined to the M1 hand representation in all experimental cases. The infarct was readily distinguished from surrounding tissue based on blood-filled vessels, an artifact of the electrocoagulation method employed. Neuronal staining within the infarct was markedly reduced, and most neurons that did stain positive for NeuN had shrunken and irregularly shaped cell bodies. NeuN staining patterns were identical to previously published results (Sondell et al, 2000; Stowe et al, 2007). Staining patterns for HIF-1α protein in neurons were similar to previous results in rodents (Jin et al, 2000). Hypoxia-inducible factor-1α protein was confined to a peri-cytoplasmic location in cells that also positively stained for NeuN (Figure 1F—I). Staining of VEGFR-2 resulted in intense staining patterns in the cell body and occasional dendrites, predominantly in cells that also positively stain for NeuN (Figure 1B—E). Sections processed without primary antibodies did not show discernable staining (data not shown). The M1 orofacial region was used to control for motor cortex plasticity and proximity to the infarct, as it was the closest remote area to the peri-infarct region. Because of previous evidence of a lack of penumbra using the bipolar electrocoagulation method (Frost et al, 2003), and analysis of the M1 orofacial region as a control, we are confident that changes in ‘remote’ ipsilesional areas were not a result of peri-infarct hypoxia or diffusion of growth factors from the peri-infarct region into the surrounding cortex.
Significant Neuronal Loss Was Confined to the Primary Motor Hand Area Infarct
Design-based stereological analysis yielded two population estimates, one for total neurons (NeuN-positive cells) and another for neurons that associated with either HIF-1α (NeuN-positive cells that were immunoreactive for HIF-1α protein; Table 1) or VEGFR-2 (NeuN-positive cells that were immunoreactive for VEGFR-2; Table 1). Although only neurons were investigated in this series of experiments, the results do not preclude significant changes occurring in other cell types within the cortex. Estimates for neuronal populations were similar to previous results (Stowe et al, 2007). Conversion of the estimates to neuronal densities (neurons/mm3) in each region of interest facilitated comparison across cortical regions and between animals.
Population densities and percent ratios for neurons immunoreactive for either HIF-1α or VEGFR-2 from total neuronal populations
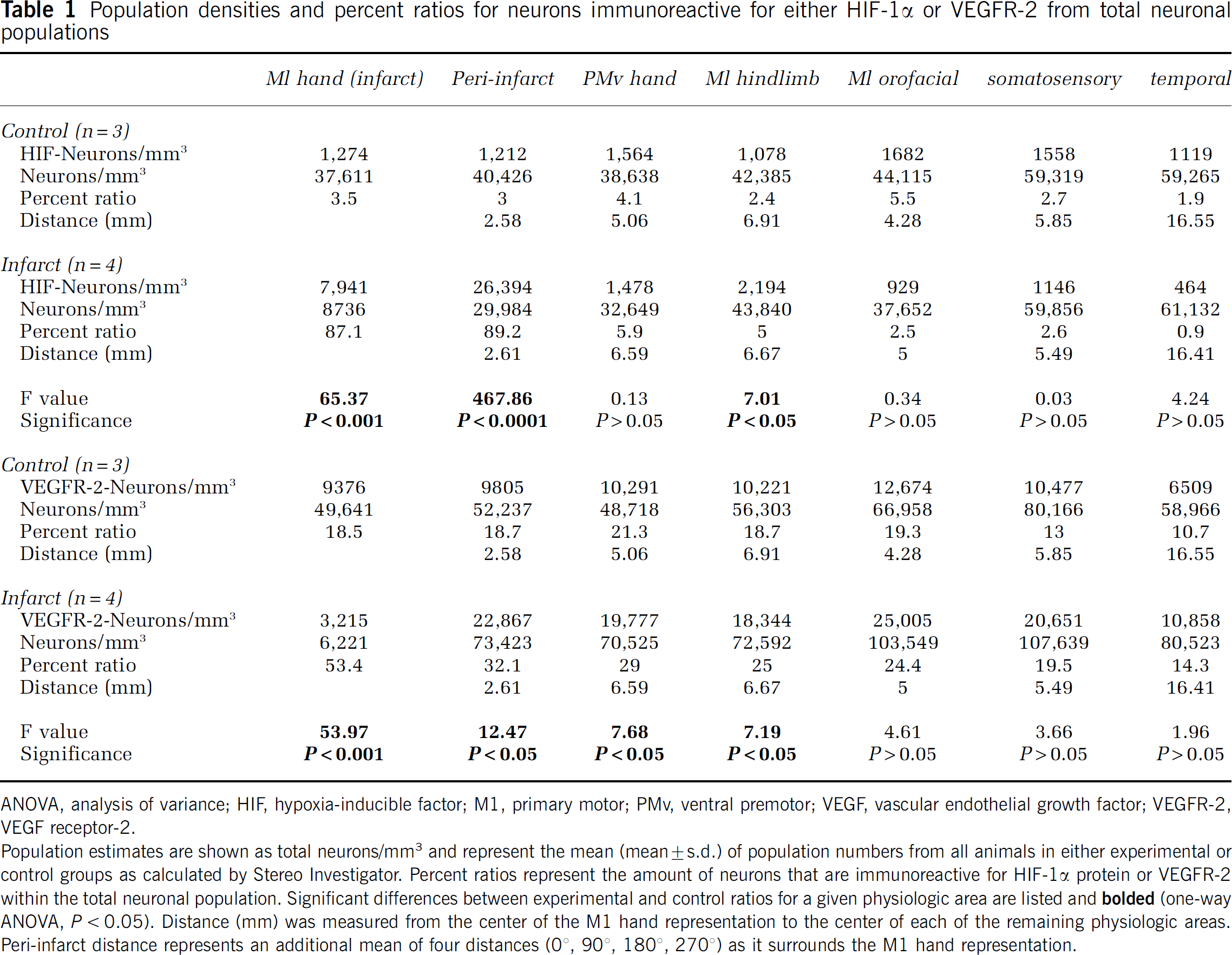
ANOVA, analysis of variance; HIF, hypoxia-inducible factor; M1, primary motor; PMv, ventral premotor; VEGF, vascular endothelial growth factor; VEGFR-2, VEGF receptor-2.
Population estimates are shown as total neurons/mm3 represent the mean (mean±s.d.) of population numbers from all animals in either experimental or control groups as calculated by Stereo Investigator. Percent ratios represent the amount of neurons that are immunoreactive for HIF-1α protein or VEGFR-2 within the total neuronal population. Significant differences between experimental and control ratios for a given physiologic area are listed and
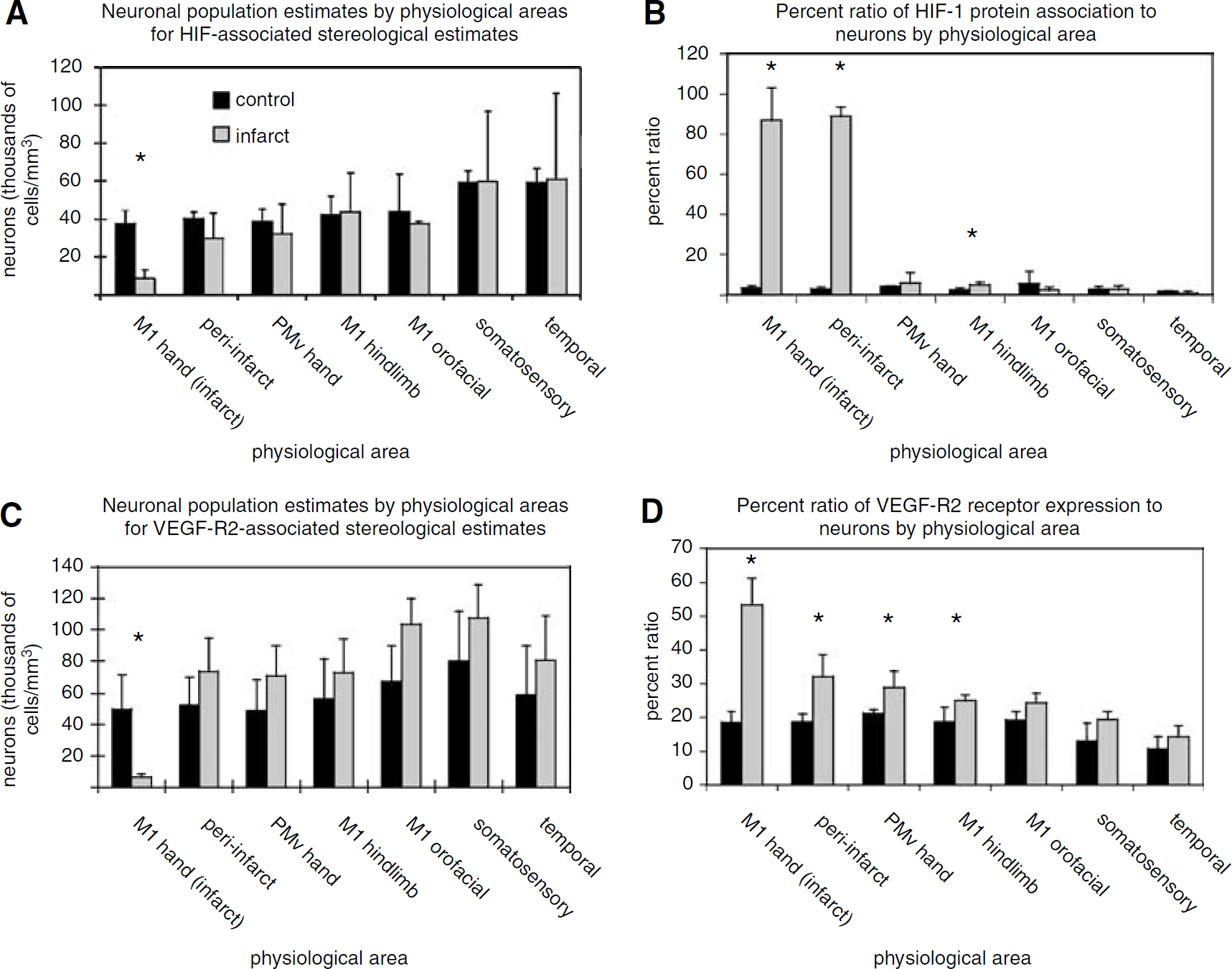
In the HIF-1α data set, the mean neuronal density within the M1 hand representation was significantly reduced in experimental versus control conditions (
(
Similarly, in the VEGFR-2 data set, the only significant decrease in mean neuronal densities between control and experimental conditions again occurred within the M1 hand representation (
Neurons Under Control Conditions were Immunoreactive for Both Hypoxia-Inducible Factor-1α and Vascular Endothelial Growth Factor Receptor-2 in All Examined Areas of the Cortex
The ratio of neurons immunoreactive for either HIF-1α or VEGFR-2 was derived from population estimates described above and found in Table 1. In general, fewer than 5% of neurons were immunoreactive for HIF-1α protein under control conditions, regardless of cortical region. Approximately 19% of the total neurons in the primary and secondary motor cortices were immunoreactive for VEGFR-2 under control conditions. The somatosensory (13.0%) and the temporal (10.7%) regions each had fewer neurons immunoreactive for VEGFR-2 compared with motor cortex.
Neurons Have Significantly Greater Immunoreactivity for Hypoxia-inducible factor-1α Protein at 3 Days After an Infarct
After the M1 hand area infarct, the mean proportion of neurons immunoreactive for HIF-1α protein was significantly greater in three cortical areas: the M1 hand, the peri-infarct, and the M1 hindlimb regions (Table 1 and Figure 2). Although there was a significant reduction in total neurons in the M1 hand representation after infarct, neurons increased immunoreactivity for HIF-1α from 3.5±0.8% in the control group to 87.1±16.3% in the neurons remaining at 3 days post-infarct (
Neurons Significantly Increased Vascular Endothelial Growth Factor Receptor-2 Immunoreactivity After an Infarct
After an M1 hand representation infarct, the mean proportion of neurons immunoreactive for VEGFR-2 significantly increased in four cortical areas: the M1 hand, the peri-infarct, the PMv hand, and the M1 hindlimb regions (Table 1 and Figure 2). The significant increase in VEGFR-2 immunoreactivity in the M1 hand representation was in a significantly reduced total neuron population. The percentage of neurons in the M1 hand area that were immunoreactive for VEGFR-2 increased from 18.5±3.3% in the control group to 53.4±7.9% in the experimental group (
Vascular Endothelial Growth Factor Receptor-2 Immunoreactivity Significantly Correlated to Neuronal Vascular Endothelial Growth Factor Receptor Protein Levels
Under both experimental and control conditions, there was a significant correlation between the ratio of neurons immunoreactive for VEGFR-2 (present results) to the ratio of neurons immunoreactive for VEGF protein (Stowe et al, 2007). Under control conditions and across all cortical areas, the correlation between VEGF and VEGFR-2 was significant, with an
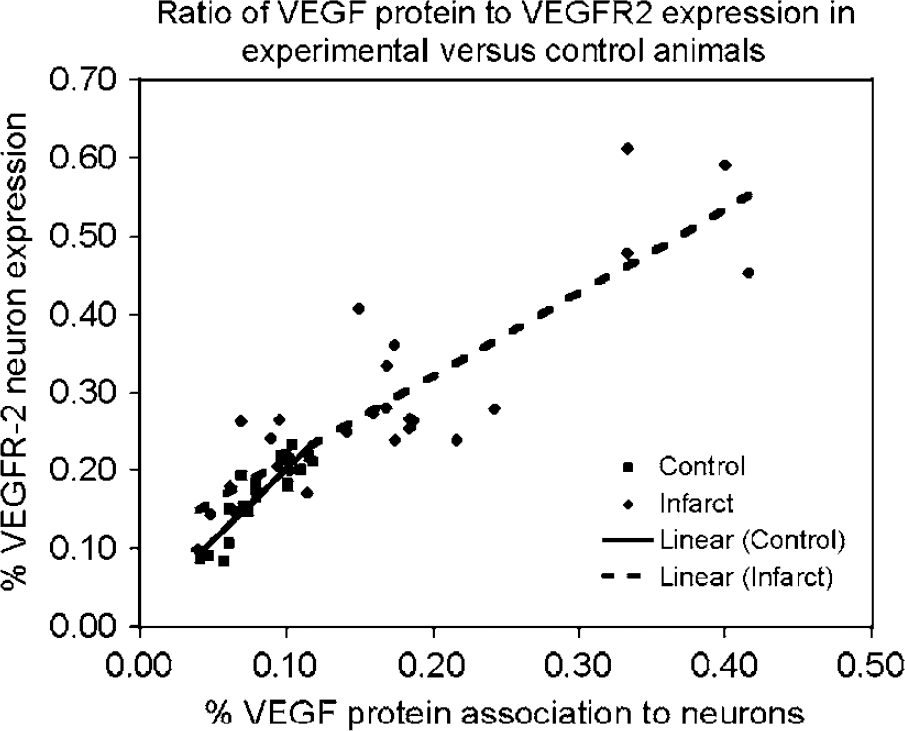
Linear regression analysis based on arcsin-transformed population densities for neurons immunoreactive for VEGF and VEGFR-2 in all physiologic areas of the cortex for either experimental or control conditions for each animal. The
Discussion
The Role of hypoxia-inducible factor-1α in Neuroprotection After Stroke
In this study in non-human primates, neurons significantly increased immunoreactivity for HIF-1α in both the peri-infarct and infarct (i.e., M1 hand representation) regions, in agreement with previous rodent studies (Chavez and LaManna, 2002; Jin et al, 2000). Although the neuronal densities in the M1 hand infarct reflect a 77% loss of neurons, nearly 9 out of every 10 (87%) viable neurons remaining stained for HIF-1α protein, versus only 4% in the control M1 hand area. This ratio represented more than a 20-fold increase in HIF-1α immunoreactivity. Evidence supports a role for HIF-1α in the upregulation of genes promoting both neuroprotection (Jin et al, 2000; Semenza, 2004) as well as apoptosis (Carmeliet et al, 1998; Helton et al, 2005). Despite evidence that increased activation of HIF-1α reduces infarct volume (Siddiq et al, 2005), Helton et al (2005) suggest that the predominant role of HIF-1α during hypoxia/ischemia is proapoptotic, and that the
Neuronal Vascular Endothelial Growth Factor Receptor-2 Expression Occurred in Functionally Related Remote Areas
This study investigated neuronal immunoreactivity for VEGFR-2 in remote ipsilesional physiologic areas that may be involved in recovery of function. Previous studies in the squirrel monkey showed that the PMv hand representation is heavily interconnected to the M1 hand representation (Dancause et al, 2006a) and, at 3 months after an M1 hand area infarct, PMv hand neurons undergo neuroanatomic and neurophysiologic plasticity (Dancause et al, 2005; Frost et al, 2003). Despite verification of PMv plasticity in both non-human primate and clinical data (Seitz et al, 1998), the understanding of molecular events, in response to an infarct, in this or other remote regions of the intact cortex, is minimal.
There was a significant increase in the proportion of neurons immunoreactive for VEGFR-2 in the PMv hand representation by 3 days post-infarct. Because VEGFR-2 is not under the transcriptional regulation of HIF-1α, but instead upregulated by VEGF itself (Kremer et al, 1997; Shen et al, 1998), it is possible that the increased VEGFR-2 immunoreactivity in PMv was a result of a VEGF-induced neuroprotective cascade initiated by interconnected neurons. Neuronal levels of VEGFR-2 in the M1 hindlimb representation also underwent a significant increase in neuronal immunoreactivity after infarct. It is doubtful, however, that a significant number of M1 hindlimb neurons were at risk of neuronal death due to the ischemic injury, as neurons in this region are not heavily interconnected to the area of infarct (Hatanaka et al, 2001). Instead, increases in collateral blood flow from the anterior cerebral artery may have upregulated VEGF, and thus VEGFR-2 production, in this area (Milkiewicz et al, 2001).
Vascular Endothelial Growth Factor Receptor-2 Expression Highly Correlated to Neuronal Vascular Endothelial Growth Factor Levels Throughout the Cortex
The level of neuronal VEGF protein immunoreactivity (Stowe et al, 2007) correlated to VEGFR-2 expression levels in neurons in both motor and non-motor cortices under either control and experimental conditions. During control conditions, there was a 1:2 ratio of VEGF/VEGFR-2 in neurons. At 3 days after an M1 hand area infarct, however, the linear fit evolved into a ratio of 2:3 for VEGF/VEGFR-2. The results suggest that increased VEGF protein levels can drive increased VEGFR-2 expression on neurons, albeit in a time-delayed manner. The data seem to reflect a time point where the VEGF protein is still promoting upregulation of VEGFR-2. Despite the uniformity of VEGF/VEGFR-2 ratios when all regions are pooled, there are differences in the magnitude of increases when considering specific regions. In the infarct and peri-infarct regions, the overall magnitude of percentage change increased over control values by 35 and 13%, respectively, for VEGFR-2 immunoreactivity and 26 and 9%, respectively, for VEGF immunoreactivity. In both of these areas, there was a greater magnitude increase of neuronal VEGFR-2 expression over VEGF protein immunoreactivity.
Within the PMv hand representation, however, both VEGFR-2 and VEGF protein immunoreactivity increased 8% over control values, and in the M1 hindlimb, neuronal immunoreactivity for VEGFR-2 actually increased 2% less than the magnitude increase of VEGF immunoreactivity in neurons. The equality of magnitude in the PMv region suggests a delayed response to the infarct, a reasonable hypothesis if the stimulus driving the molecular response in this region is not due to the direct insult of hypoxia/ischemia, but instead due to neurons interconnected to the infarct. These comparisons reveal a lack of uniformity in the magnitude of upregulation in the ipsilesional cortex. In conclusion, our analyses of molecular plasticity in neurons from interconnected ipsilesional regions, remote from the ischemic injury but still at risk for cell death, suggest that a uniquely prolonged ‘window of opportunity’ may exist during the subacute stage of recovery for directed cytoprotective therapies in the intact cortex.
Footnotes
Acknowledgements
We thank Erica Blumberg and Robert Cross for assistance in data acquisition and analysis. We also thank Dr Lutz Slomianka, University of Zurich, Switzerland, for continued guidance with the stereological protocols and analysis.
