Abstract
Reactive astrocytes are thought to protect the penumbra during brain ischemia, but direct evidence has been lacking due to the absence of suitable experimental models. Previously, we generated mice deficient in two intermediate filament (IF) proteins, glial fibrillary acidic protein (GFAP) and vimentin, whose upregulation is the hallmark of reactive astrocytes.
Introduction
Astroglial cells are the most abundant cells in the central nervous system (CNS) and are believed to play a major role in the brain and spinal cord pathologies. Although never proved directly, astrocytes are thought to exert a neuroprotective effect in stroke by shielding neurons from oxidative stress (Kraig et al, 1995). This hypothesis is based on the ability of astrocytes to engage in spatial buffering, to transport and metabolize amino acids, glucose, and other key molecules, and to upregulate antioxidants and free radical scavengers in the ischemic region.
Reactive gliosis is a response of astrocytes to CNS injury, including brain ischemia, mechanical trauma, neurodegenerative diseases, and tumors. Reactive astrocytes undergo changes in morphology and in their expression of a wide range of molecules (Eddleston and Mucke, 1993; Ridet et al, 1997). The hallmark of reactive gliosis, regardless of its origin, is increased expression of glial fibrillary acidic protein (GFAP) and vimentin in reactive astrocytes. Glial fibrillary acidic protein and vimentin are building blocks of intermediate filaments (IFs), which, together with microtubules and actin filaments, constitute the cytoskeleton.
We and others have generated
In this study, we subjected
Materials and methods
Mice
Single-mutant (
Induction of Ischemia: Proximal Middle Cerebral Artery Transection
Focal cerebral ischemia was induced in six WT and six
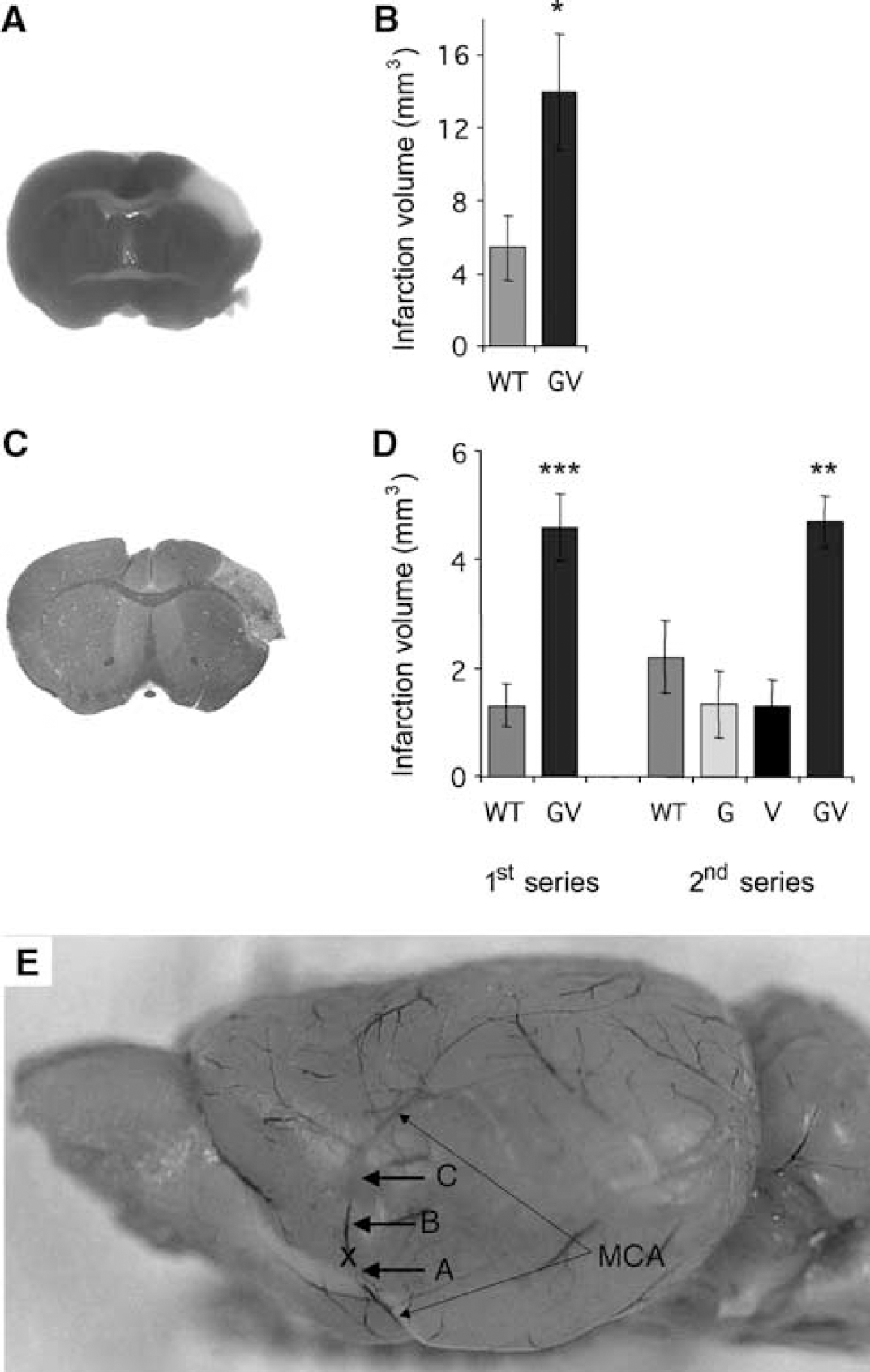
Infarct area is 2.5- to 3.5-fold larger in
Induction of Ischemia: Distal Middle Cerebral Artery Transection
Distal MCA transection was performed as described (Nagai et al, 1999) in two independent series of experiments. One set of experiments was performed in 10 WT and 11
Measurement of Infarct Volume 7 Days after Proximal Middle Cerebral Artery Transection
Deeply anesthetized mice were killed by decapitation. Fresh frontal brain slices (0.5 mm thick) were cut on a Vibratome, incubated for 30 mins in 0.125% triphenyltetrazolium chloride solution (T4375; Sigma-Aldrich, St Louis, MO, USA) in buffer containing 1.35% dimethylsulfoxide, 2 mmol/L MgCl2, 0.1 mol/L Na2HPO4, and 0.1 mol/L NaH2PO4 (pH 7.4) at 37°C, and fixed in 4% buffered formaldehyde (pH 7.4) (Fotheringham et al, 2000). In each slice, the infarct area in white matter was measured with a Nikon SMZ-U stereomicroscope and image analysis software (Easy Image, Bergström Instrument, Göteborg, Sweden). The infarct volume was calculated by integrating infarct areas on all adjacent brain slices with detectable infarction. The extent of brain edema was judged by comparing the areas of the ischemic and contralateral hemispheres.
Measurement of Infarct Volume 7 Days after Distal Middle Cerebral Artery Transection
Deeply anesthetized mice were perfused through the left ventricle with 4% phosphate-buffered formaldehyde (pH 7.4). The brains were postfixed overnight and embedded in paraffin. Frontal sections (8 μm thick) were stained with hematoxylin and erythrosine. Infarct areas were assessed by delineating the ischemic region, which could be clearly discriminated by its lighter appearance and high proportion of cells with pycnotic nuclei. The infarct area was measured on all sections (320 μm apart) on which it was detectable (4 to 10 sections per mouse) as described above, and the total infarct volume was calculated.
Blood Pressure and Heart Rate Monitoring
Blood pressure and heart rate were monitored in five WT and four
Comparison of Cerebrovascular Architecture
The MCA territory was determined as described (Maeda et al, 1998). Briefly, the cerebrovasculature of four WT and four
Data Analysis
Data are expressed as mean±s.e.m. The two-tailed
Immunohistochemistry
Coronal cryosections (35 μm), made from perfused and postfixed brains of four WT and three
Astrocyte-Enriched Cultures
Primary astrocyte-enriched cultures were prepared from postnatal day 1
Immunocytochemistry
Cells were fixed in methanol at −20°C for 5 mins and washed with PBS. After nonspecific binding was blocked with 5% normal goat serum (Dako) for 15 mins at room temperature, cells were incubated with rabbit antibodies against ETBRs (1:100; Alomone Labs) and mouse antibodies against GFAP (clone GA5, 1:100; Sigma-Aldrich) and then with Alexa 488-conjugated anti-rabbit and Alexa 568-conjugated anti-mouse antibodies (1:500; Molecular Probes). All antibodies were diluted in PBS.
Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis and Immunoblotting for Endothelin B Receptors
Medium was removed from confluent cultures of astrocytes (10 to 14 days after plating). After two washes with PBS, cells were harvested by scraping into sodium dodecyl sulfate-extraction buffer consisting of protease inhibitor cocktail (Roche, Mannheim, Germany), 1 mmol/L Na+ orthovanadate, and 1 mmol/L phenylmethanesulfonyl in 2% sodium dodecyl sulfate, and the cell lysate was frozen at −80°C. Thawed lysates were sonicated for 5 secs, and the protein content was determined with the Bicinchoninic Acid Protein Assay (Sigma-Aldrich). Protein (50
Reverse Transcription and Quantitative Real-Time PCR
cDNA was generated using the iScript cDNA Synthesis Kit (Bio-Rad Laboratories, Hercules, CA, USA) with a mixture of random hexamers and oligo(dT) primers, according to the manufacturer's instructions, except that for ETBR and Cx43 expression analyses, the incubation time at 42°C was increased from 30 to 60 mins. The reverse transcription was run in duplicate in 10-μL reactions (Stahlberg et al, 2004) using 1.5 μg of total RNA extracted from primary astrocyte cultures as described below. The cultures were prepared from four WT and four
Scrape Loading/Dye Transfer
Astrocyte gap-junctional communication (AGJC) was assessed by using the scrape loading/dye transfer technique as described (Blomstrand et al, 1999). In brief, confluent astrocyte cultures from five WT and four
Glutamate Uptake
Fresh brain slices microdissected from five WT and nine
Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis and Immunoblotting for Glutamate Transporter-1
Brain slices (approximately 2 × 2 mm) taken from the cortex of the left hemisphere of six WT and nine
DNA Array Analysis
RNA extraction, DNase treatment, poly (A+) RNA enrichment, probe synthesis, column chromatography, and hybridization of the probes to the array were performed as described in the Atlas Pure Total RNA Labeling System User Manual and the Atlas cDNA Expression Arrays User Manual (Clontech Laboratories, Palo Alto, CA, USA). Confluent astrocyte cultures were harvested by scraping into RNase-free PBS and centrifuged at 500g for 5 mins at 4°C. The arrays were exposed to phosphor screens (Molecular Dynamics, Buckinghamshire, UK) overnight. The screens were scanned in a Storm instrument (Storm 820, Molecular Dynamics), and the images were analyzed with AtlasImage 1.5 (Clontech Laboratories).
Results
Larger Infarct Volume in GFAP–/–Vim–/– than WT Mice after Proximal Middle Cerebral Artery Transection
Seven days after proximal MCA transection, infarct volume was 257% higher in
Larger Infarct Volume in GFAP–/–Vim–/– than WT Mice after Distal Middle Cerebral Artery Transection
To determine if the transection point affects the difference in the infarct volume between WT and
Median Blood Pressure and Heart Rate and Cerebrovascular Architecture are not Altered in GFAP–/–Vim–/– Mice
To address a possible primary effect of the absence of GFAP and Vim on blood pressure and cerebrovascular architecture, we recorded blood pressure and heart rate during and after the MCA transection and compared the anastomoses between branches of the MCA and the anterior cerebral artery. During the MCA transection and for 60 mins thereafter, mean blood pressure and heart rate did not differ in WT and
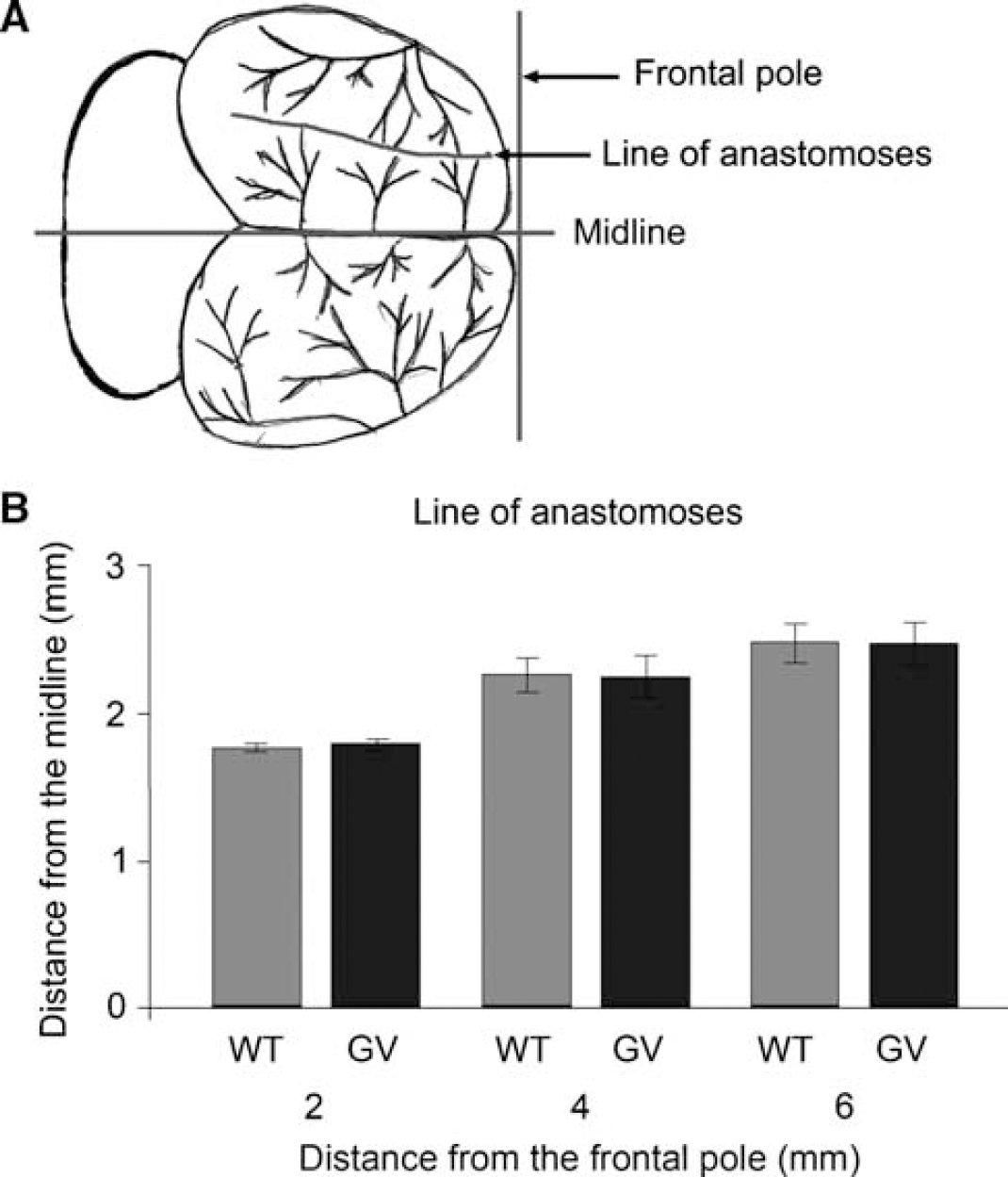
The territory supplied by the MCA is comparable in WT and
Normal Number of Astrocytes and Microglia in the Penumbra of GFAP–/–Vim–/– Mice
Next, we examined the penumbra and the infarct area histologically. Except for the larger infarct area in
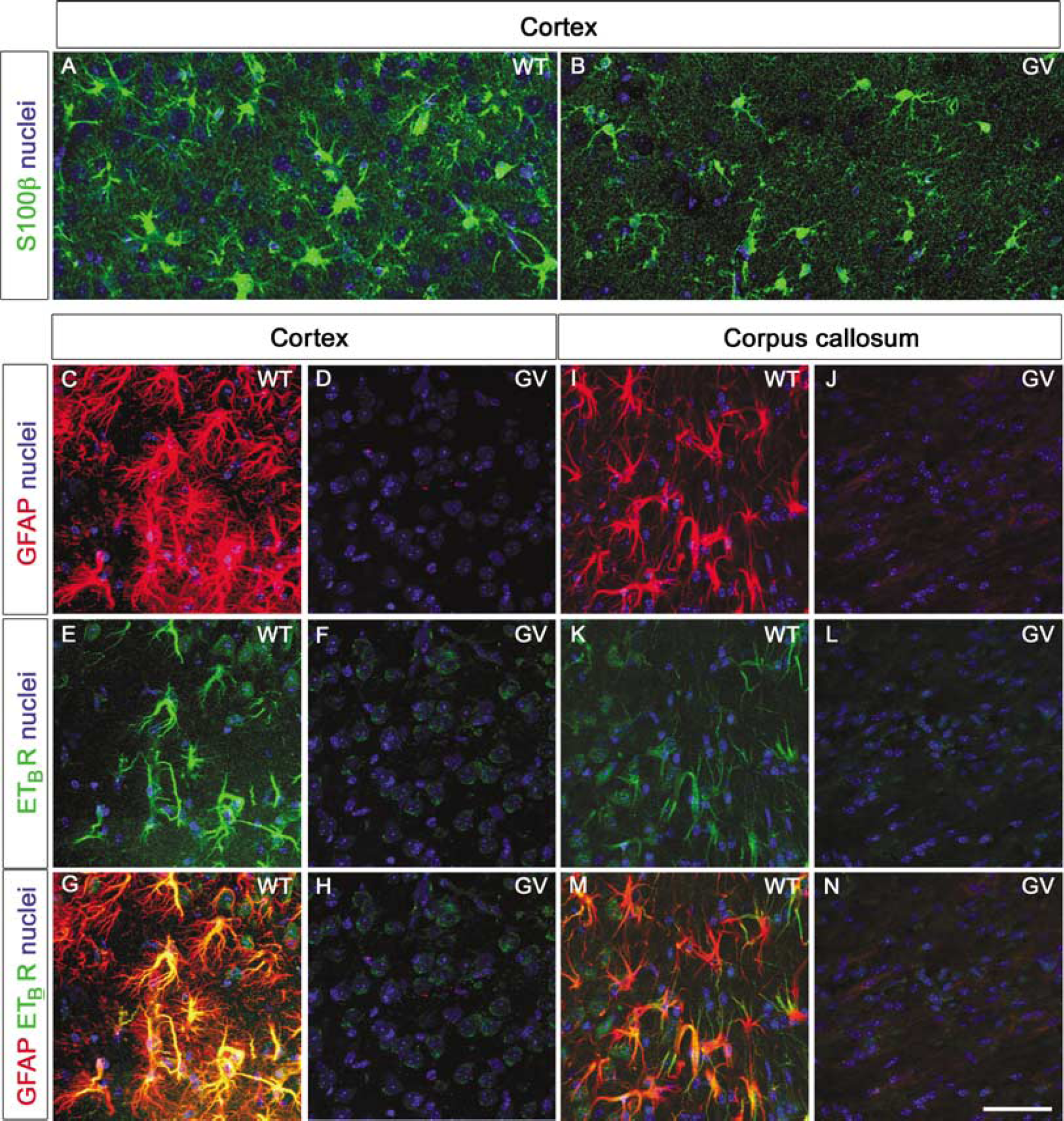
Astrocytes around the ischemic lesion show ETBR immunoreactivity in WT but not
Absence of Endothelin B Receptor-Immunoreactivity in Astrocytes in the Ischemic Penumbra of GFAP–/–Vim–/– Mice
Endothelin B receptor expression by astrocytes in the injured CNS was proposed as one of the steps leading to astrocyte activation and reactive gliosis (Koyama et al, 1999). In the ischemic penumbra and in the corpus callosum 7 days after MCA transection, ETBRs were highly expressed on reactive astrocytes in WT mice (Figures 3E and 3K) but were essentially undetectable on astrocytes of
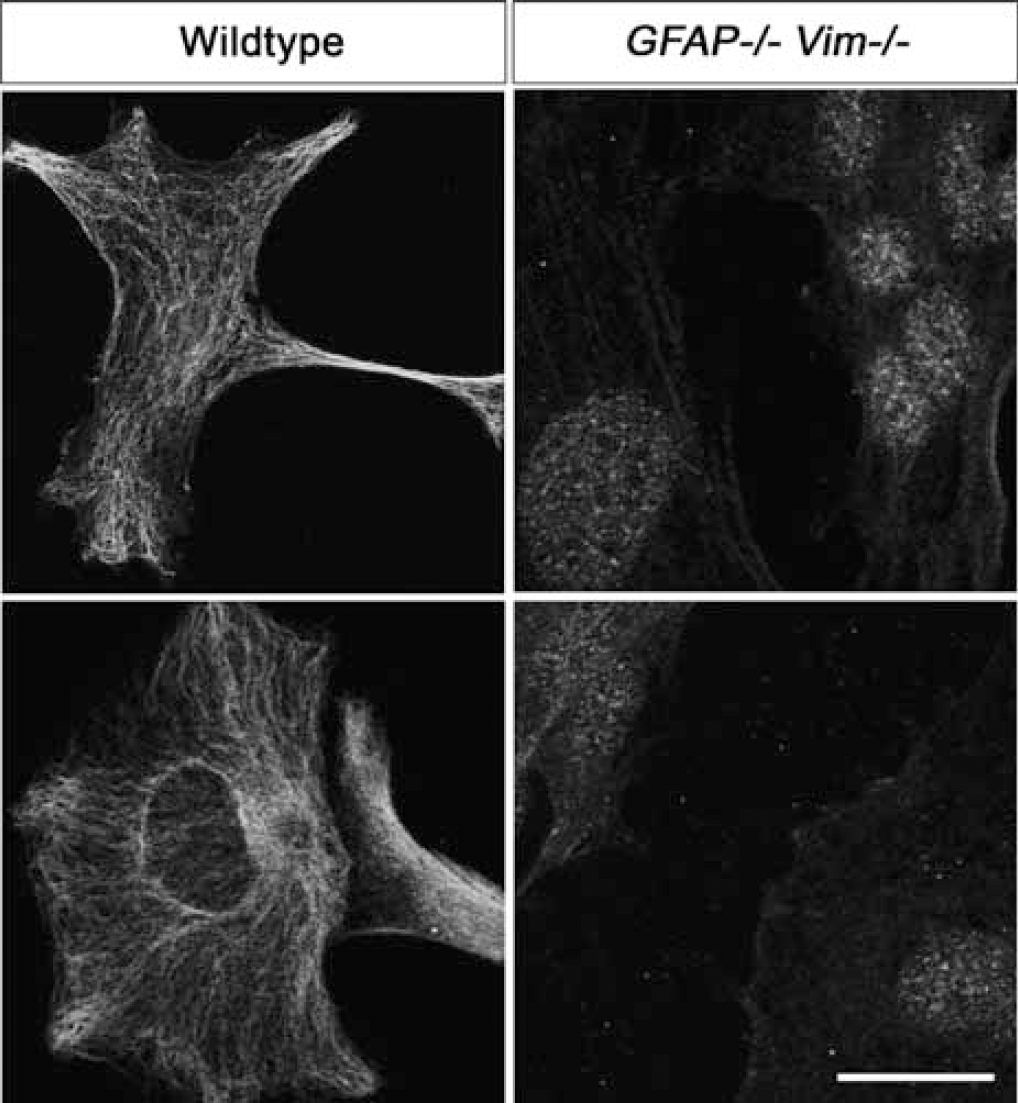
ETBR immunoreactivity had a filamentous appearance in the cytoplasm of cultured WT astrocytes but was absent in the cytoplasm of
Endothelin B Receptor Immunoreactivity Colocalizes with Intermediate Filament Bundles in Reactive Astrocytes
To investigate further the relationship between ETBR and astrocyte IFs, we analyzed their distributions in cultured astrocytes by laser-scanning confocal microscopy. This analysis revealed remarkable colocalization between bundles of IFs and ETBR immunoreactivity in WT astrocytes, with the ETBR immunoreactivity decorating bundles of IFs (Figure 5).
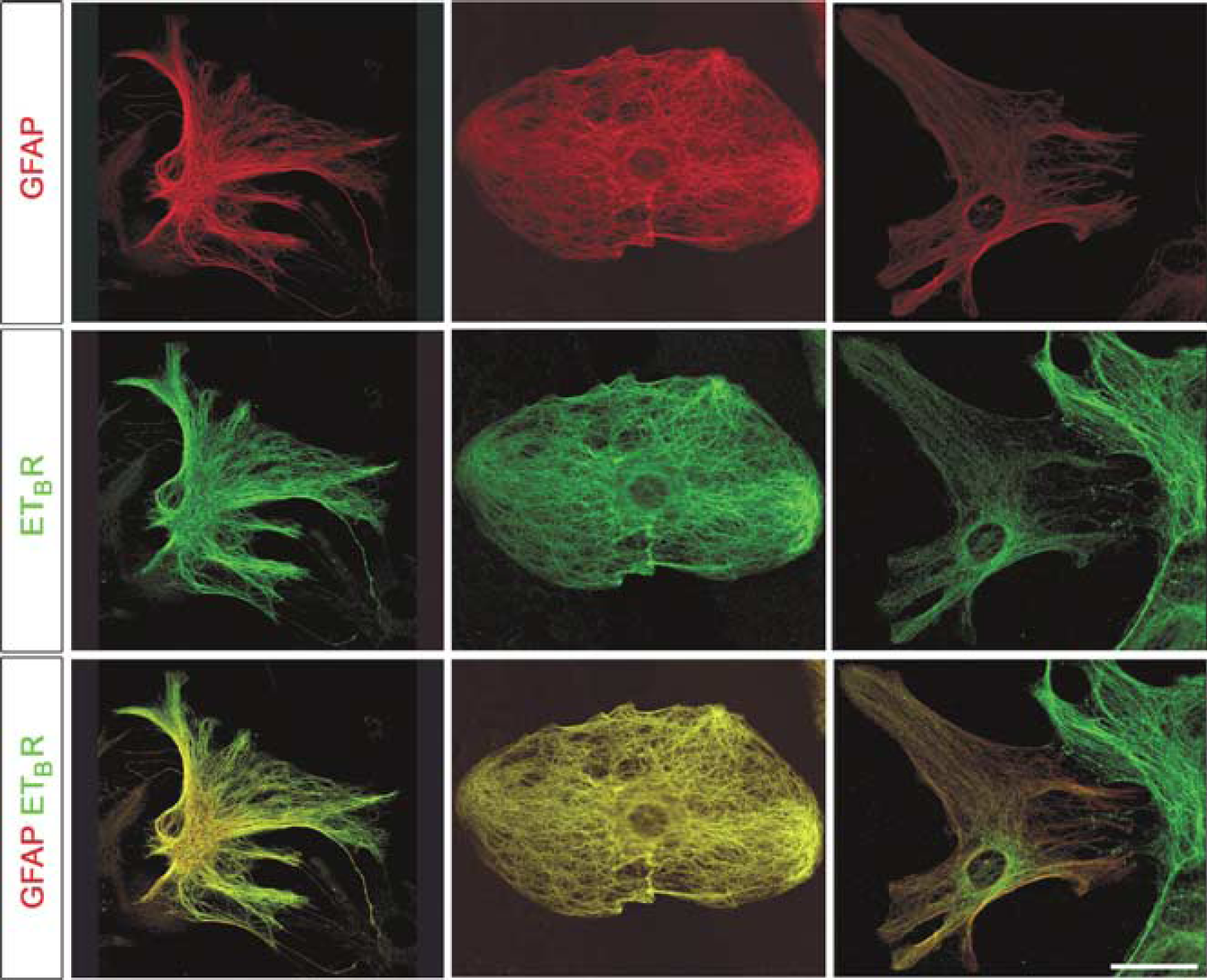
ETBR and bundles of IFs colocalize in cultured WT astrocytes. Laser-scanning confocal microscopy revealed a filamentous appearance of ETBR immunostaining, which colocalized with GFAP-positive bundles of IFs. Red, GFAP; green, ETBR. Scale bar = 20 μm.
Intermediate Filaments Determine Endothelin B Receptor Distribution in Reactive Astrocytes
To determine if IFs are required for the production, stability, or distribution of ETBR in reactive astrocytes, we performed quantitative real-time PCR and Western blot analyses on cultured astrocytes from
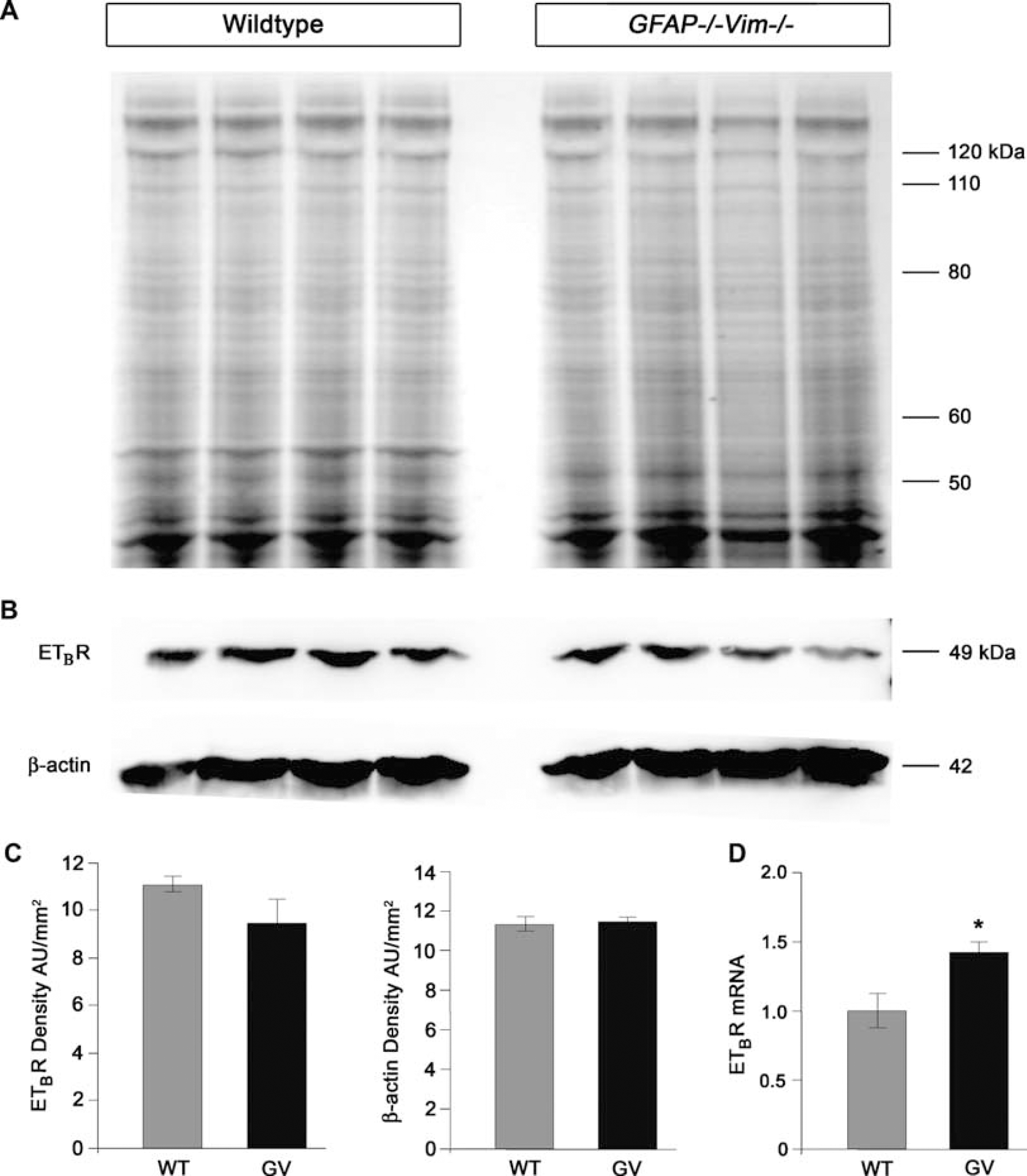
ETBR protein levels are comparable in WT and
Endothelin-3-Induced Blockage of Gap Junctions is Attenuated in GFAP–/–Vim–/– Astrocytes
Endothelins are well-known blockers of astrocyte gap-junctional communication (AGJC) (Blomstrand et al, 1999). To determine if the altered cytoplasmic distribution of ETBRs affects AGJC and thus the function of the astrocyte syncytium, we stimulated ETBRs with the selective ligand endothelin-3. No significant difference in basal AGJC was observed between WT and
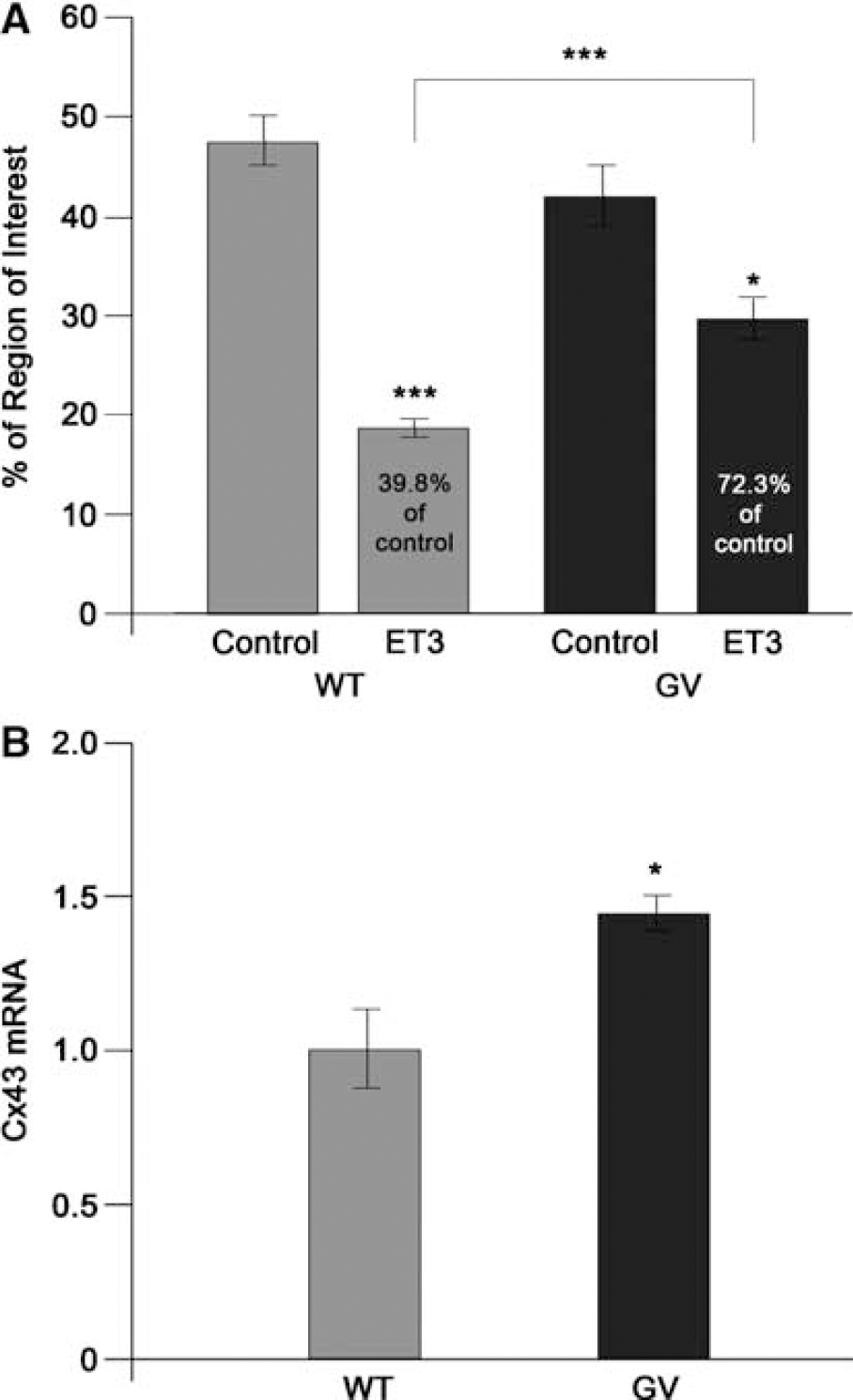
Reduction in Glutamate Transport in GFAP–/–Vim–/–Mice
The ability of astrocytes to remove glutamate released by neurons was proposed to reduce infarct size by limiting the excitotoxic cell death (reviewed in Nedergaard and Dirnagl, 2005). To determine if glutamate transport is altered in
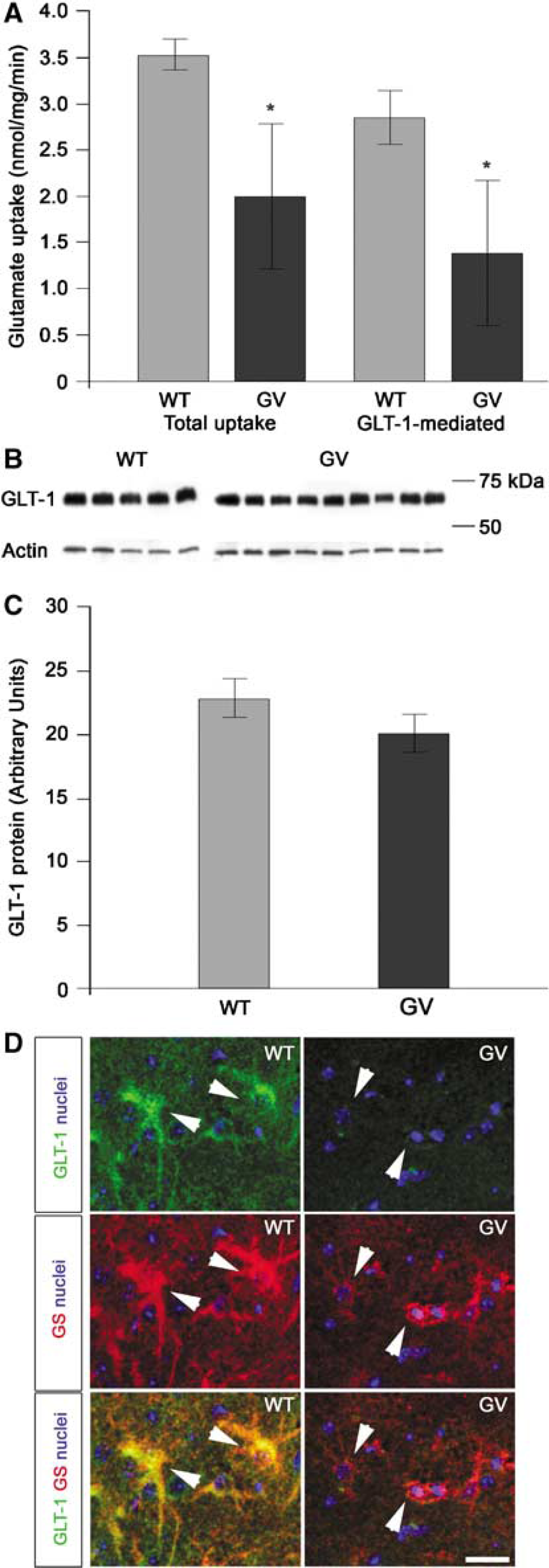
Total and GLT-1-mediated glutamate uptake is decreased in the cortex of
Plasminogen Activator Inhibitor-1 is Downregulated in GFAP–/–Vim–/– Astrocytes
To address further, at a molecular level, the effect of attenuated reactive gliosis, we performed microarray analysis with the Atlas cDNA Expression Arrays (Clontech Laboratories). The expression of 1,200 genes was analyzed in primary astrocyte cultures derived from WT and
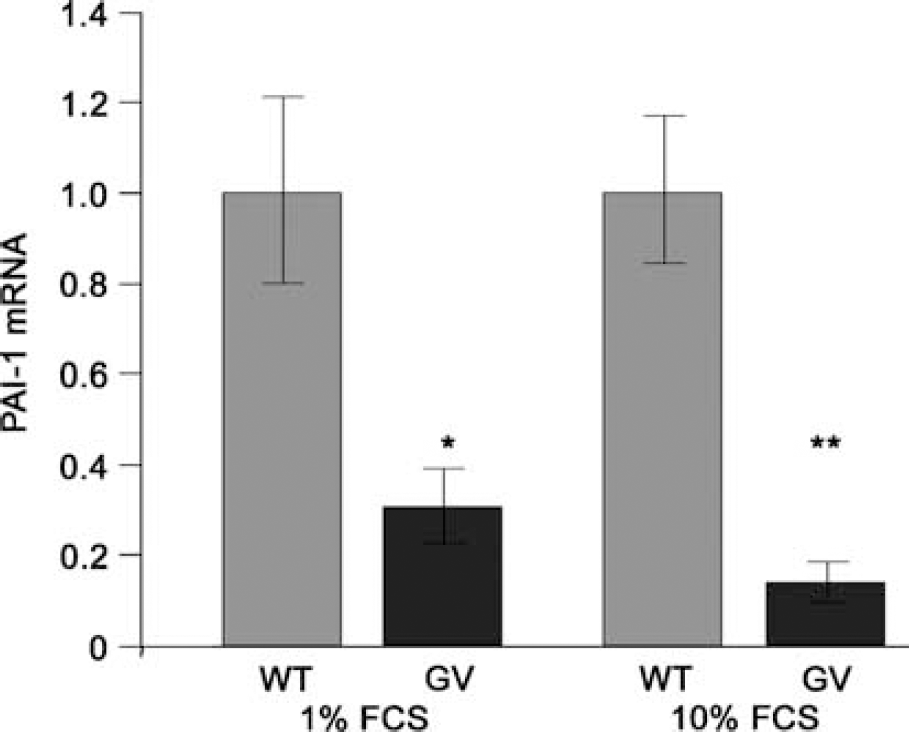
Plasminogen activator inhibitor-1 mRNA levels are lower in
Discussion
In the absence of a suitable experimental model, direct proof that astrocytes have a protective effect in the ischemic brain has largely been lacking. Attempts to generate mammalian models in which astrocytes would be globally or regionally eliminated resulted in phenotypes too severe or complex for studying the role of astrocytes in the pathogenesis of CNS diseases. In mice expressing herpes simplex virus thymidine kinase from the GFAP promoter, ablation of dividing astroglial cells by treatment with ganciclovir during early postnatal development led to severe developmental abnormalities (Delaney et al, 1996). The same approach was applied in adult mice to selectively eliminate the subpopulation of dividing reactive astrocytes after neurotrauma and resulted in a massive invasion of leukocytes in response to dying astrocytes, increased neurodegeneration, and failed blood—brain barrier (BBB) repair (Bush et al, 1999), implying neuroprotective activities of astrocytes in the lesioned area (Faulkner et al, 2004).
Upregulation of IFs is a hallmark of reactive gliosis after trauma or stroke and is a key step in astrocyte activation (Pekny et al, 1999b). To address the role of reactive astrocytes in stroke, we subjected
In
Vimentin is also expressed in other cell types in the CNS, particularly endothelial cells, oligodendroglia, and microglia. Endothelial cells in
How might the absence of IFs in reactive astrocytes explain the increased infarct size in
We saw a close colocalization between bundles of IFs and ETBR immunoreactivity in cultured WT astrocytes and altered ETBR immunoreactivity in
Astrocytes play a major role in the transport and metabolism of a range of molecules, in particular those utilized in nutrition, cell—cell signaling, and neurotransmission. A failure to carry out these functions may constitute a major pathogenic component in stroke and other CNS pathologies (reviewed in Kraig et al, 1995). In primary cultures of reactive astrocytes from WT,
Among 1,200 genes whose expression was compared between WT and
Reactive astrocytes play a key role in the reconstruction of damaged BBB after both trauma and stroke. In a previous study with the
In response to CNS injury, astroglial cells migrate over considerable distances to the injured region, where they join the local pool of reactive astrocytes (Johansson et al, 1999). We have shown that partial or complete deficiency of IFs in reactive astrocytes
The ability of astrocytes to regulate their volume by releasing osmotically active molecules, such as taurine, is considered to be a mechanism for counteracting cytotoxic brain edema in stroke (Kimelberg, 1991). When subjected to hypotonic stress,
Previously, we showed that attenuation of reactive gliosis in
In summary, our findings provide
Footnotes
Acknowledgements
We thank Dr Anders Hamberger, Dr John Eriksson, and Hanna-Mari Pallari for their input in this project and Dr Martin Rydmark for his advice on stereomeric evaluation of the infarct volume.
