Abstract
The proinflammatory cytokine interleukin (IL)-1 mediates several forms of experimentally induced acute brain injury and has been implicated in chronic neurodegenerative disorders. The IL-1 receptor antagonist, IL-1RA, protects rodents against ischaemic brain injury, but its molecular mass (17 kDa) potentially limits the brain penetration of peripherally administered IL-1RA. We therefore sought to identify whether therapeutically effective concentrations of IL-1RA in the rat were also achieved in brain of patients with subarachnoid haemorrhage (SAH), using a peripheral administration regime that had proved to be safe and reduce peripheral inflammation in patients after stroke. An intravenous bolus of IL-1RA, followed by infusion, was administered to rats after induction of focal cerebral ischaemia. The effects of IL-1RA on brain ischaemia and the concentrations achieved in cerebrospinal fluid (CSF), were determined. Interleukin-1 receptor antagonist was similarly administered to patients with SAH, and CSF was sampled via external ventricular drains. In rats, IL-1RA significantly reduced brain injury induced by focal cerebral ischaemia. The plasma IL-1RA concentrations reached 12±2 μg/mL by 30 mins, and CSF concentrations were maintained between 91 and 232 ng/mL between 1 and 24 h of infusion. In patients with SAH, IL-1RA reached a steady-state plasma concentration of 22 ± 4 μg/mL by 15 mins, and CSF concentrations were maintained at 78 to 558 ng/mL between 1 and 24 h. Intravenous delivery of IL-1RA leads to CSF concentrations in patients comparable to those that are neuroprotective in rats, and might therefore be of therapeutic benefit.
Introduction
Growing evidence implicates inflammation and inflammatory molecules such as interleukin (IL)-1 in central nervous system disease (Lipton, 1999; Ginsberg, 2002; Allan and Rothwell, 2003; Lees et al, 2006; Emsley et al, 2007). This cytokine is induced rapidly in response to experimental brain insults and, when administered to rodents centrally or peripherally, enhances brain injury (Rothwell et al, 1997a, b ; Allan et al, 2005). Interleukin-1 receptor antagonist (IL-1RA) is a potent and highly selective, competitive IL-1 antagonist, which appears to block all actions of IL-1 and is also induced by tissue injury (Hannum et al, 1990; Loddick et al, 1997; Pinteaux et al, 2006). In rodents, IL-1RA markedly inhibits neuronal injury induced by focal or global, reversible or permanent cerebral ischaemia, excitotoxicity, traumatic brain injury, and seizures (Relton and Rothwell, 1992; Touzani et al, 1999).
Interleukin-1 receptor antagonist has proved safe in patients with sepsis (Fisher et al, 1994), and is licensed for clinical use in rheumatoid arthritis (Nuki et al, 2002). We have tested its safety in a small, randomized, double-blinded study, where IL-1RA was administered as an intravenous bolus, followed by infusion, to patients within 6 h of stroke compared with placebo (Emsley et al, 2005). No serious adverse events were observed and IL-1RA markedly attenuated the levels of circulating inflammatory molecules and cells, which have been correlated with poor clinical outcome. These data suggest that IL-1RA may be beneficial in brain disorders associated with ischaemic or excitotoxic processes and brain trauma. Although brain penetration of IL-1RA might be expected to be limited, because of its size (17 kDa), several studies have reported that very high doses of IL-1RA, given subcutaneously, are neuroprotective in rodents (Martin et al, 1994; Relton et al, 1996), and an active transport mechanism into brain has been reported in rats (Gutierrez et al, 1994). The hypothesis underlying this study was that peripherally administered IL-1RA would enter the cerebrospinal fluid (CSF) to decrease the extent of ischaemic damage in rats subjected to stroke and would penetrate the CSF in patients with subarachnoid haemorrhage (SAH) at similar concentrations. We therefore induced focal cerebral ischaemia (middle cerebral artery occlusion (MCAo)) in rats to assess the neuroprotective effect, and the plasma and CSF concentrations of IL-1RA achieved, after administration of the IL-1RA via a similar intravenous regime to that used in the human safety study (Emsley et al, 2005). To study the pharmacokinetics of IL-1RA in humans, we administered IL-1RA to patients with SAH, where placement of external ventricular drains (EVDs) for clinical management allowed sampling of CSF. The results showed that intravenous treatment with IL-1RA was safe in SAH and achieved steady-state CSF concentrations comparable to those that were neuroprotective in rats.
Materials and methods
Rats
Male rats (Sprague—Dawley, Charles River Laboratories, Margate, UK) were maintained on pelleted diet (Bantin and Kingman Universal, Aldbrough, UK) and kept under the same housing conditions in a 12-h light/dark cycle, fasted overnight before use. All procedures were performed in accordance with the United Kingdom Animals (Scientific Procedures) Act (1986) under Project Licence no. 40/02538. They were anesthetized with 4% halothane and maintained by inhalation of 1% halothane, 70% N2O, and 30% O2. The rectal and tympanic temperature was kept at 37 ± 0.5°C with a thermostatically controlled heating blanket, but they received no ventilator support at any time during the procedures.
Middle Cerebral Artery Occlusion
Fourteen rats were analysed in the neuroprotective study. They were subjected to MCAo by retrograde insertion of an occlusive intraluminal thread, via the external carotid artery into the internal carotid artery, according to the method described by Zea Longa et al, (1989). Successful MCAo was defined as clinical evidence of middle cerebral artery territory infarction (contralateral hemiparesis) after reperfusion and no evidence of SAH (indicating arterial perforation) on histologic analysis. For investigation of the neuroprotective effect of IL-1RA, animals were killed at 24 h by exposure to increasing concentrations of CO2, followed by dislocation of the cervical spine. After craniectomy and removal of the hindbrain, the forebrains were frozen on dry ice and transferred to −20°C for storage. Coronal sections of 20 μm were cut through the frozen brain every 500 μm. The sections were placed on 3-aminopropyltriethoxy-silane-coated slides and subsequently stained with cresyl violet. Northern Eclipse Image Analysis Program software (Empix, Mississauga, ON, Canada) and Microsoft Excel were used to calculate lesion volumes, which were adjusted for edema.
Pharmacokinetics in Rats
Twenty-seven rats, similarly subjected to MCAo, were used in the IL-1RA pharmacokinetic study. The IL-1RA (as r-met-huIL-1RA: Kineret®; Amgen, Thousand Oaks, CA, USA) was delivered intravenously as a bolus (10 mg) and intravenous infusion (Alzet osmotic minipump model 5001d, 0.8 mg/h; Alzet, Cupertino, CA, USA) via a dedicated intravenous line placed surgically in the right external jugular vein. Blood was obtained by cardiac puncture and CSF by cisterna magna puncture under terminal anaesthesia at 30 mins (n = 4), 1 (n = 4), 2 (n = 3), 3 (n = 4), 4 (n = 3), and 24 h (n = 9). Samples were collected in pyrogen-free heparinized tubes (10 U/mL) and centrifuged for 15 mins at 2,000g. Plasma was stored at −70°C until analysis. Assays for IL-1RA were performed using an enzyme-linked immunosorbent assay, as described previously (Emsley et al, 2005).
Subarachnoid Haemorrhage Patients
Six female and two male patients were recruited to the study from the Intensive Care or Neuro High Dependency Units at Hope Hospital, Salford, UK between September 2002 and January 2005. Patients were Caucasian, aged 39 to 69 years (mean age = 50.5 years), with a mean weight of 70 kg (range = 64 to 150 kg). Patients with confirmed aneurysmal SAH requiring insertion of an EVD for treatment of hydrocephalus were approached for inclusion in the study. The Salford and Trafford NHS Trust Local Research Ethics Committee and the University of Manchester Research Ethics Committee approved the study. Informed written consent was obtained from patients or, where this was not possible, written assent obtained from their representative. Patients were excluded from study participation if they were known to have a clinically significant concurrent medical condition, or had participated in a clinical trial in the previous 30 days. Pregnant or breastfeeding patients, and patients with a history of sensitivity to products derived from Escherichia coli or any component of IL-1RA were also excluded. Physical examinations were performed, and demographic details and clinical histories were collected. At presentation, 2 of the 8 patients were in each of the four World Federation of Neurosurgeon Score categories (Teasdale, 1988). The presenting Glasgow Coma Score (Teasdale and Jennett, 1974) ranged from 8 to 15, 6 of the 8 patients being grade XIII and above. The distribution of subarachnoid blood on computed tomography varied greatly, but the majority of patients had thin or thick vertical patterns of haemorrhage (revised Fisher grades II and III (Fisher et al, 1981). All patients scored 100/100 on the functional ability Barthel index (Mahoney and Barthel, 1965). Six patients had a modified Rankin score (van Swieten et al, 1988) of 0, and two had a score 1 before the study. Participants were sequentially coded A—H.
Pharmacokinetics in Patients with Subarachnoid Haemorrhage
All patients received the IL-1RA (as Kineret®) within 5 days of SAH. Baseline blood samples were obtained within 6 h before IL-1RA infusion for measurement of IL-1RA, hematological, and biochemical profiles. Participants received an intravenous bolus injection (100 mg) of IL-1RA via a dedicated, intravenous cannula. This was immediately followed by a continuous intravenous infusion at 2 mg/kg per h (in 1 L, 0.9% sterile NaCl) for 24 h, via a volumetric infusion pump (Graseby, Watford, UK), except for patient B, who entered the study with impaired renal function (plasma creatinine concentration > 177 μmol/L) and received 1 mg/kg per h infusion. Samples of 3 mL CSF (from the EVD) and 5 mL blood were obtained for measurement of IL-1RA. Samples were collected in pyrogen-free heparinized tubes (10 IU/mL) and centrifuged for 15 mins at 2,000g. The plasma was stored at −70°C until analysis. Assays for IL-1RA were performed as described above. Albumin in CSF samples was measured using Micro-albumin CP reagent sets for plasma and urine (ABX Pentra, Montpellier, France) on a Cobas Bio analyzer (Roche Diagnostics, Bugess Hill, UK), and serum albumin was measured in the routine clinical biochemistry laboratory. An assessment of blood—brain barrier (BBB) function was estimated using a ratio of CSF albumin (mg/L) to serum albumin (g/L) using a value of > 9 to signify albumin leakage into the CSF and BBB breakdown (Al Kassab et al, 1981).
Safety in Patients
The occurrence of adverse events was monitored, with particular attention to infection, and full physical examination was performed on days 0, 1, 2, 3, 4, 10, and 30. Haematology and blood biochemistry were monitored at these time points, and CSF was obtained for microscopy and bacteriological culture, where available via EVD. All adverse events were reported to an independent data monitoring committee (P Kirkpatrick and E Warburton, University of Cambridge, UK) for review.
Analysis and Statistics
Data comparing groups were analysed using unpaired Students' t-test. A two-tailed probability of less than 5% was assigned to indicate statistical significance. The half-life (t1/2) of IL-1RA in plasma and CSF was calculated using the formula t1/2 = 0.693/k, where k, is the first-order elimination constant. Graphical representation of data and area under curve analysis was performed using GraphPad Prism software (GraphPad, San Diego, CA, USA).
Results
Intravenous Infusion of Interleukin-1 Receptor Antagonist is Neuroprotective in Rats
Mortality and exclusions were similar in both groups (30%). Exclusions were based on predetermined criteria to assure that only animals with successful MCAo were included. An intravenous bolus of IL-1RA administered at the time of occlusion, followed by an intravenous infusion of IL-1RA, significantly reduced lesion volume by 32% in comparison to vehicle treatment (Figure 1). This reduction in lesion volume was attributable to a significant reduction in cortical lesion volume in IL-1RA-treated animals of 41% (93 mm3) compared with vehicle-treated animals. Interleukin-1 receptor antagonist had no statistically significant neuroprotective effect in the subcortex.
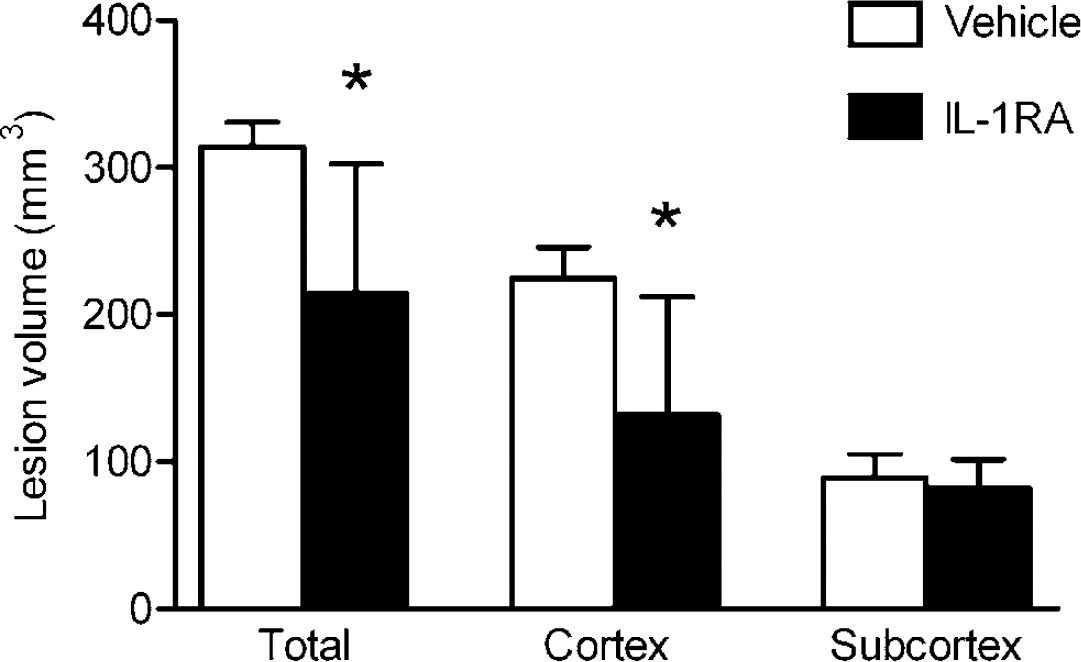
Effect of an intravenous bolus and infusion of IL-1RA on lesion volume induced by MCAo in rats. Interleukin-1 receptor antagonist significantly reduced total lesion volume (whole hemisphere) by 32% (*P < 0.02) and cortical lesion volume by 41% (P < 0.02). There was no statistically significant reduction in subcortical lesion volume (P = 0.47); n = 7 (IL-1RA) and n = 7 (vehicle). Bars denote s.d.
Intravenously Infused Interleukin-1 Receptor Antagonist Enters the Cerebrospinal Fluid of Rats
Human IL-1RA in plasma and CSF was not detectable before treatment. After intravenous bolus injection and infusion of IL-1RA, concentrations in plasma increased to a maximum of 12 ± 2 μg/mL by 30 mins. Cerebrospinal fluid concentrations increased more slowly to 232 ± 103 ng/mL at 1 h, and were maintained at a mean concentration of152 ng/mL (range = 91 to 232 ng/mL) between 1 and 24 h (Figure 2). The half-lives of IL-1RA in plasma and CSF were 45 mins and 4.7 h, respectively. Combined data indicate that the CSF concentration at plateau is 5.1% of that in plasma.
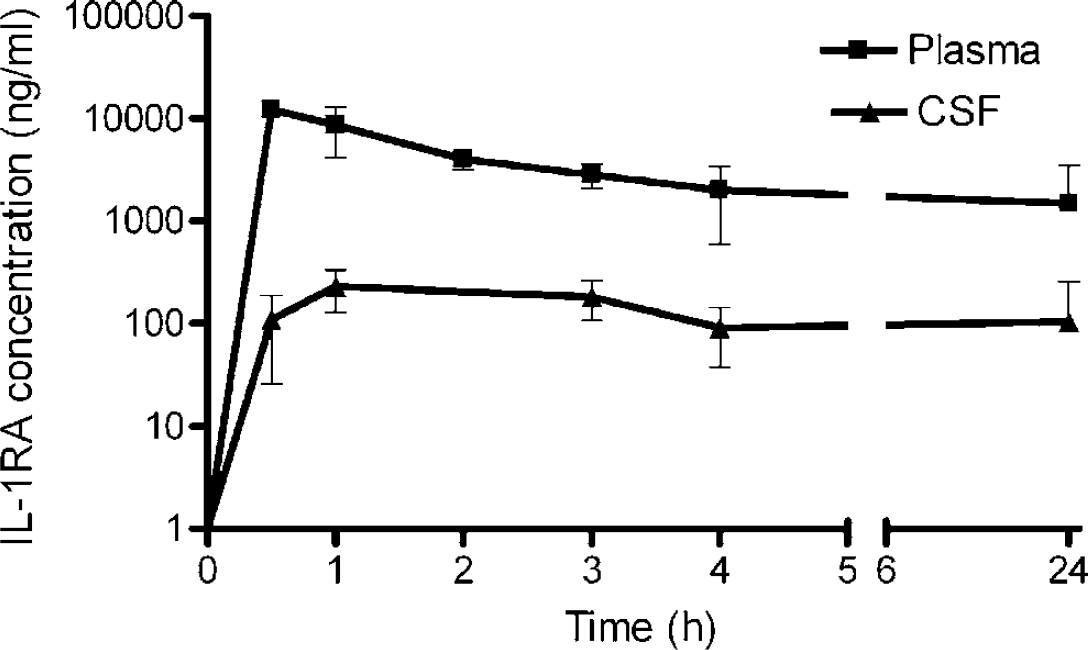
Concentration of IL-1RA in blood (plasma) and CSF after intravenous bolus (10 mg) and infusion of 0.8 mg/h in rats. Data are expressed as mean concentration of IL-1RA ± s.d. 30 mins (n = 4), 1 (n = 4), 2 (n = 3), 3 (n = 4), 4 (n = 3), and 24 h (n = 9).
Intravenous Infusion of Interleukin-1 Receptor Antagonist did not Raise Safety Concerns in Patients with Subarachnoid Haemorrhage
Six patients completed the full 24-h infusion of IL-1RA. Patient B developed diabetes insipidus and hypernatraemia, and the infusion was stopped at 8 h. Patient F developed severe cardiac instability, requiring large concentrations of positive inotropic agents, and the infusion was stopped at 11 h. Pharmacokinetic data for patients B and F were therefore excluded from analysis. In the 30 days of the study, the single serious adverse event (death) was not attributable to test treatment, since the patient was extremely unwell because of haemorrhage before study entry. In the same 30-day time scale, there were 12 adverse events in 6 patients. Of these, eight were infections (two CSF, four respiratory, one urinary, and one gastrointestinal). The three remaining adverse events were haematemesis, diabetes insipidus, and bleeding from a surgical wound. All adverse events were referred to an independent data monitoring committee who deemed that none of these were attributable to test treatment. For the duration of the study, IL-1RA had no significant effect on haemoglobin concentration or circulating leukocyte or platelet numbers. In addition, there was no significant alteration in biochemical, renal, or hepatic parameters related to infusion of IL-1RA, and it had no apparent effect on heart rate, blood pressure, or aural temperature during the course of the study.
Intravenous Infusion of Interleukin-1 Receptor Antagonist in Patients with Subarachnoid Haemorrhage Achieved Similar Cerebrospinal Fluid Concentrations to Those that were Therapeutically Effective in Rats
The mean plasma concentration of IL-1RA at baseline was 780 ± 660 pg/mL and in CSF was 880 ± 720 pg/mL. After infusion of IL-1RA, the mean plasma concentration increased to 22 ± 4 μg/mL at 15 mins, was maintained at 20 μg/mL (range = 14 to 33 μg/mL) until the infusion was terminated at 24 h, and then decreased with an half-life of 22 mins (Figure 3). The concentrations of IL-1RA in CSF increased to 78 ng/mL at 1 h, and were maintained at 383 ng/mL (range = 78 to 558 ng/mL) between 1 and 24 h, before falling with an initial half-life of 2.5 h. These data indicated that the CSF concentration at plateau was 2.5% of that in plasma. Cerebrospinal fluid albumin concentrations were available for the first five patients, and CSF/serum albumin ratios at baseline (11 ± 4) suggested some breakdown of the BBB (11 to 14.6 in 4, and 3.6 in 1 patient). There was no apparent relationship between this ratio and IL-1RA entry to the CSF, the 24 h concentrations of IL-1RA being 129 and 105 μg/mL in patients with the lowest (3.6) and highest (14.6) ratios, respectively, and 594 μg/mL IL-1RA in a patient with a ratio of 12. The 24-h ratio (9 ± 6) did not suggest further plasma leakage during IL-1RA infusion.
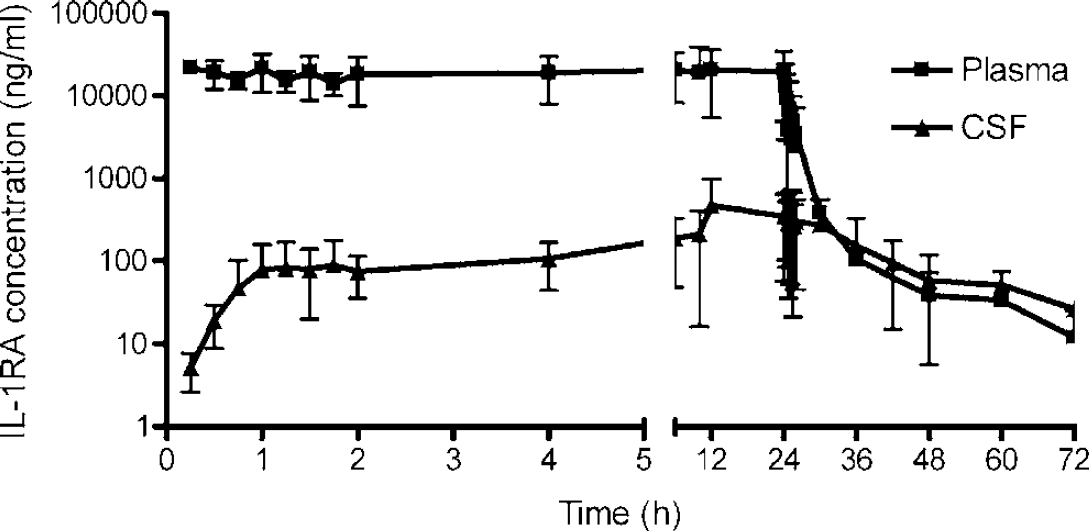
Concentration of IL-1RA in blood (plasma) and CSF after intravenous bolus (100 mg) and infusion of 2 mg/kg per h for 24 h in patients with SAH. Data are expressed as mean concentration of IL-1RA ± s.d. (n = 7).
Discussion
Numerous studies have reported neuroprotective effects of recombinant IL-1RA in many forms of brain injury in rodents (Relton and Rothwell, 1992; Toulmond and Rothwell, 1995; Garcia et al, 1995; Panegyres and Hughes, 1998; Mulcahy et al, 2003). However, most of these have administered IL-1RA intracerebroventicularly, where it can readily penetrate brain tissue. Some studies have shown efficacy of IL-1RA administered subcutaneously (Martin et al, 1994; Relton et al, 1996), but we believe this is the first report to describe the effects of intravenous administration of IL-1RA on neuronal injury in experimental animals. The reduction in ischaemic brain injury reported here (32%) is slightly less than that seen after intracerebroventicular injection, suggesting that brain penetration may be a limiting factor, or that a higher rate of infusion or bolus may be more effective. The protection was most evident in the cortical penumbra. The absence of effect in the subcortex is consistent with the fact that this is the primary site of injury in response to MCAo and is most resistant to neuroprotection. Interleukin-1 plays a central role in the cascade of inflammatory activity that may result in extending the area of ischaemia, after SAH or other types of stroke, and IL-1RA can block such actions. The only known action of IL-1RA is to block IL-1 via interaction with the IL-1 type 1 receptor (Rothwell et al, 1997b). The detrimental effects of IL-1 are not fully understood, but experimental studies would suggest indirect mechanisms including release of neurotoxin from glia (Thornton et al, 2006), immune cell invasion (Colotta et al, 1996), and induction of fever (Kozak et al, 1998). Our preliminary trial of IL-1RA in stroke patients supports this view in that it showed a reduction in plasma IL-6, as well as C-reactive protein and the leukocyte cell count (Emsley et al, 2005). Since the most likely source of increased plasma IL-6 after stroke is the central nervous system (Emsley et al, 2007), this suggests that IL-1RA has an impact on IL-6 induction in the brain. Although our trial in stroke hinted at clinical efficacy, it was too small to provide sufficient evidence of this. Our objective is therefore to conduct clinical trials in ischaemic stroke as well as SAH, but only after we have explored the potential for adverse outcomes and defined a suitable administration regime.
Interleukin-1 receptor antagonist was found in significant concentrations in rat CSF, indicating rapid brain penetration. It is unlikely that this was due to the surgical intervention, because no brain surgery was performed, although it is known that BBB breakdown occurs in cerebral ischaemia (Belayev et al, 1996). Interleukin-1 receptor antagonist reached mean plasma concentrations in humans similar to those reported previously for the same bolus and infusion rate in sepsis in humans (Fisher et al, 1994), and the observed plasma kinetics were identical to those observed in healthy volunteers (Granowitz et al, 1992). The proportion of IL-1RA that crossed the BBB was significantly higher in our series than in the studies described by Banks et al in mice (Gutierrez et al, 1994), where only a single dose was administered. This work and our unpublished data suggest that active transport of IL-1ra is the predominant mechanism for entry of IL-1ra into the CSF of normal animals. In addition to the continuous infusion regime used here, it is possible that enhanced permeability of the BBB, caused by the stroke or SAH, might have contributed to the increased transfer. Since patients with SAH have leakage of blood (and consequently plasma proteins such as albumin) into the subarachnoid space at the time of haemorrhage, estimating BBB integrity by monitoring entry of plasma proteins is problematic. However, in our series, the albumin concentrations did not increase during the infusion period, suggesting that there was no further significant compromise of the BBB after the initial bleed, and animal studies of SAH indicate that BBB function is restored approximately by day 3 (Germano et al, 2000). Although this potentially represents a limitation in extrapolating these results to those with a normal BBB, it presents less compromise in terms of considering therapeutic potential in patients with central nervous system pathology.
Pharmacokinetic modelling is important in the study of a putative neuroprotective agent in cerebral ischaemia. This study presents a pragmatic and empirical evaluation with respect to patients who might benefit from blockade of IL-1 effects and who may be expected to have a similar compromise in BBB function. It is the first study to compare neuroprotective concentrations of a therapeutic agent in an established model of stroke with those obtained in a related clinical scenario. Such evidence provides valuable data that will contribute to evaluation of whether progression to a phase III trial may be productive.
The intravenous bolus of 100 mg, followed by an intravenous infusion of 2 mg/kg per h, was chosen as the maximum dose of IL-1RA that has been administered previously to any patient (Fisher et al, 1994), and was that used in a phase II trial of IL-1RA in acute stroke (Emsley et al, 2005). The infusion time was fixed at 24 h, as results from the initial patients showed that this was sufficient for CSF concentrations to plateau, and many patients had EVDs removed soon after this. Although the dose of IL-1RA administered to rats was similar to that in humans (∼2mg/kg per h), the bolus was considerably higher and may have contributed to the slightly faster rise in CSF concentration. Preliminary modelling of the data suggests that a higher bolus dose decreases the time to steady state, whereas increasing the infusion rate provides a relatively modest increase to plateau concentrations. This is consistent with the difference between concentrations in rat and human. It may be appropriate to explore modification of these doses to optimize the therapeutic regimen, before conducting a large trial.
A potential limitation of the study is that the concentration of proteins in the CSF of patients may not accurately reflect the concentrations in the local brain environment (deLange et al, 1997). To address this issue, it will be necessary to measure concentrations in brain extracellular fluid and recent developments in microdialysis of proteins may allow this (Hutchinson et al, 2005). Endogenous IL-1RA is released from brain tissue into CSF and plasma after SAH (Gruber et al, 2000), so it is reasonable to expect that concentrations in brain extracellular fluid will also be increased. Although IL-1RA was present in baseline plasma and CSF, the relative contribution to overall concentration observed after infusion was low (estimated to be 0.004% in plasma and 0.2% in CSF).
Interleukin-1 receptor antagonist has showed an excellent safety profile in studies of sepsis and arthritis, although long-term administration of IL-1RA in a cohort of rheumatoid arthritis patients resulted in slightly increased incidence of infection (Fisher et al, 1994; Nuki et al, 2002; Fleischmann et al, 2003). This must remain a concern until data from larger trials in stroke or SAH are completed, particularly given evidence for immune suppression after head injury, including stroke (Prass et al, 2003). However, IL-1RA infusion had no significant impact on morbidity in trials of severe systemic sepsis (Fisher et al, 1994; Opal et al, 1997) or stroke (Emsley et al, 2005). Although we have no control subjects for comparison, and this was not primarily a safety study, the ventriculostomy-associated infection rates in our series (25%) were similar to those reported previously (39%) (Pfausler et al, 2003), and 50% of our patients developed chest infection, which is comparable to a recent study in which the incidence of respiratory infections was reported to be greater than 50% (O'Shea et al, 2004). One patient developed cardiac instability, and IL-1RA infusion was stopped. This clinical condition may be associated with SAH, although relatively uncommon, and evidence from a large trial of IL-1RA in sepsis suggests that IL-1RA was not implicated in the development of cardiac problems in that patient cohort (Fisher et al, 1994). The patients in this study were a selected subpopulation of all subarachnoid patients with clinical indications for insertion of EVDs, who generally reflected a more severely ill subgroup. Patients (50%) were of good grade at the end of the study as compared with 58% in a recent study (Ikawa et al, 2004), and the mortality of 12.5% in our small series was well within mortality rates reported elsewhere (Lozier et al, 2002).
There have now been numerous failures in stroke trials, most recently with NXY-059, which showed considerable promise in experimental studies and some suggestion of efficacy in the clinic (Lees et al, 2006). However, there has been little or no evidence that NXY-059 or other experimental drugs have been delivered to the central nervous system of patients at effective concentrations. This study indicated that peripherally administered IL-1RA reached concentrations in CSF that were similar to those in animal studies where neuroprotection was observed. Combined with the excellent preclinical efficacy data, and a reasonable indication that there are no major safety concerns, these data suggest that IL-1RA is a promising potential treatment for SAH and cerebral ischaemia. They may also have relevance for the evaluation of other therapeutic approaches.
Footnotes
Acknowledgements
We are grateful to Amgen for providing the IL-1RA as Kineret®, to D Horsfall, A Parry-Jones, J Selvarajah, and S Hulme for assistance with collection and preparation of clinical samples, to K Illingworth for establishing and undertaking the albumin assays, and to P Kirkpatrick and E Warburton (University of Cambridge) for acting as an independent data monitoring committee. Our thanks are also due to the patients who kindly participated in this study and the neurosurgical team at Hope Hospital for their support.
