Abstract
Hypertension is a leading risk factor for death and dependency after ischaemic stroke. However, administering anti-hypertensive medications post-stroke remains contentious with concerns regarding deleterious effects on cerebral blood flow and infarct expansion. This study sought to determine the effect of glyceryl trinitrate (GTN) treatment in both lissencephalic and gyrencephalic pre-clinical stroke models. Merino sheep underwent middle cerebral artery occlusion (MCAO) followed by GTN or control patch administration (0.2 mg/h). Monitoring of numerous physiologically relevant measures over 24 h showed that GTN administration was associated with decreased intracranial pressure, infarct volume, cerebral oedema and midline shift compared to vehicle treatment (p < 0.05). No significant changes in blood pressure or cerebral perfusion pressure were observed. Using optical imaging spectroscopy and laser speckle imaging, the effect of varying doses of GTN (0.69–50 µg/h) on cerebral blood flow and tissue oxygenation was examined in mice. No consistent effect was found. Additional mice undergoing MCAO followed by GTN administration (doses varying from 0–60 µg/h) also showed no improvement in infarct volume or neurological score within 24 h post-stroke. GTN administration significantly improved numerous stroke-related physiological outcomes in sheep but was ineffective in mice. This suggests that, whilst GTN administration could potentially benefit patients, further research into mechanisms of action are required.
Introduction
Glyceryl trinitrate (GTN) is an organic nitric oxide (NO) donor widely used for the treatment of angina, myocardial infarction and heart congestion. 1 NO metabolites are potent vasodilators via actions on smooth muscle cells, and GTN reduces blood pressure due to increased arterial compliance and decreased venous return. 2 Treatment is easy to administer via transdermal patches, exhibits a moderate half-life, and has a low risk of serious side effects. GTN is therefore an attractive drug to re-purpose for the acute treatment of ischemic stroke. Preliminary evidence from a small ambulance-based trial (Rapid Intervention with Glyceryl trinitrate in Hypertensive stroke Trial (RIGHT)) found that transdermal administration of GTN to probable stroke patients significantly lowered blood pressure (∼18 mmHg) within 2 h of patch administration. 3 GTN also improved functional outcome at 90 days. The subsequent large multi-centre Efficacy of Nitric Oxide in Stroke (ENOS) trial that administered GTN within 48 hrs of symptom onset for 7 days failed to observe functional benefit at 90 days. 4 However, a prospective analysis of a sub-group of ENOS patients treated within 6 h of stroke onset suggested that GTN improved functional outcome. 5 The multi-centre ambulance-based RIGHT-2 trial was therefore designed to determine whether early (<4 h), ambulance based administration of GTN could improve functional outcome, but the results were surprisingly neutral, possibly because treatment was started too early. 6
Successful re-purposing of GTN may require greater understanding of the risks and benefits of anti-hypertensive treatment in the acute stroke setting, as reviewed by Appleton et al. 7 If mean arterial blood pressure (MABP) decreases, cerebral perfusion pressure (CPP) and cerebral blood flow (CBF) may also decrease. Therefore, aggressive reductions in blood pressure at the time of stroke may compromise collateral blood flow. 8 On the other hand, hypertension is an independent risk factor for death and dependency at 6 months post-stroke, 9 and can contribute to dangerous increases in intracranial pressure (ICP). Interestingly, despite GTN’s effect on MABP and vasodilatory capacity, it did not reduce CBF or CPP in stroke patients. 10 This may be because the cerebral vessels are already maximally dilated, but if this was the case, it is expected that ICP would be increased. Measuring ICP in patients is difficult due to the requirement of intracranial probes. As such, we have developed a method to measure ICP in an ovine model of stroke.11 –15 We sought to measure ICP, MABP, and CPP in the ovine stroke model following a clinically relevant, transdermal dose of GTN.
Rodent models also offer the opportunity to obtain further insight into the pleiotropic mechanisms by which GTN may or may not offer benefit in stroke. For example, a single intra-peritoneal, or intra-arterial, dose of GTN has been reported to reduce neuronal death and infarct volume when given before or near to the time of transient middle cerebral artery occlusion (MCAO) in rats 16 and mice. 17 Additionally, there was evidence of decreased production of the anti-apoptotic factor Bcl-2 as a result of GTN administration. 16 GTN did not influence cerebral perfusion, implying additional mechanisms. The dosing regimens in both studies, however, were quite high compared with the clinical scenario. Therefore, we sought to establish a dose response for a more prolonged dose of GTN in a mouse model of MCAO. This was combined with measures of CBF and brain oxygenation as well as functional outcome.
Materials and methods
Ethical approval
All applicable institutional and national guidelines for the care and use of animals were followed, and all procedures were performed in accordance with local laws. Ovine experiments were approved by the Animal Ethics Committee of South Australian Health and Medical Research Institute (SAHMRI; SAM238) and conducted according to guidelines established for the use of animals in experimental research as outlined by the Australian National Health and Medical Research Council code of practice for the care and use of animals for scientific purposes (8th edition, 2013). All rodent experiments were performed in accordance with the Animal (Scientific Procedures) Act, 1986, under licences approved by the UK home office. Studies are reported in accordance with the ARRIVE guidelines. 18 , 19
Ovine studies
Animals and GTN administration
Purebred female Merino sheep (Ovis aries, n = 18) aged 18–24 months and weighing 60.71 ± 5.58 kg were sourced from a single property in Penneshaw, South Australia. Animals were transported to the SAHMRI Large Animal Research and Imaging Facility (LARIF) in South Australia, where all experimental procedures were carried out. Statistical power calculations were performed (GPower v 3.1) to determine the required sample size to reach a power of 80%, alpha 0.05, and an effect size of 0.85 (based on findings from previous studies 14 ) with the minimum number of animals required n = 7/group. Historical sham (n = 4) and stroke with no treatment (n = 7) data was used in this study. An additional group received subdermal GTN (n = 7; Nitro-Dur 0.2 mg/h, Merck Sharp & Dohme Ltd, UK), applied at 1 h following MCAO and left in place for the duration of the experiment. The GTN dose administered was equivalent to human trials. 6 Unfortunately, male animals were unable to be used in this study due to the difficulty in catheterising the highly convoluted male urethra, as urinary catheterisation is a physiological necessity with the prolonged general anaesthesia (>24-h) in our experiment. An individual trained surgeon carried out all intracranial procedures, and as such, surgery and treatment processes were unable to be blinded. However, all analyses were conducted in a blinded fashion, including calculation of ICP, oedema, midline shift, physiological variables, MABP and infarct volume. Exclusion criterion included premature death and iatrogenic injury (e.g. significant bleeding or cerebral contusion) during the MCAO surgery.
Anaesthesia, blood pressure and intracranial pressure monitoring
Anaesthesia was induced with a combination of intravenous ketamine (100 mg/kg, CEVA, Australia) and diazepam (5 mg/ml, Pamlin, CEVA, Australia). Animals were intubated and anaesthesia maintained via intravenous ketamine infusion (4 mg/kg/h, Parnell Australia Pty Ltd) in combination with 1.5–2% isoflurane (Veterinary companies of Australia Pty Ltd) 20 , 21 with medical air or 100% oxygen. An arterial femoral catheter continuously monitored blood pressure, and blood gas samples were acquired at hourly intervals to maintain physiological pO2, pCO2 and pH. Sodium chloride was used to ensure arterial catheter patency. A femoral venous catheter was used for ketamine and crystalloid fluid administration (sodium lactate, Baxter Health, Australia).
A burr hole was drilled in the right parietal bone posterior to the coronal suture and approximately 20 mm from the sagittal suture, the dura perforated, and skull bolts secured. A Codman microsensor ICP probe (Codman & Shurtleff Inc., MA) was inserted into the cerebral parenchyma (approximately 15 mm in depth) to measure ICP within the supratentorial compartment. LabChart Reader (v 8.1.1) continuously recorded ICP and MABP throughout the 24 h monitoring period. CPP was calculated by subtracting MABP from ICP. ICP/CPP/MABP data for one animal was unfortunately lost, therefore for these analyses an n = 6 is reported for vehicle treated animals.
Surgical approach
Briefly, a 50 mm vertical incision was made posterior to the right eye. A small craniotomy was performed at the junction of the parietal and squamous temporal bones, followed by a durotomy. 11 , 12 The anterior temporal lobe was gently retracted, and the proximal middle cerebral artery (MCA) located and dissected. In the permanent MCAO group, the MCA was occluded proximally via electrocautery with Malis bipolar diathermy forceps (Valleylab Inc., CO). In both MCAO and sham animals, the brain was irrigated with saline, synthetic dura (Durepair, Medtronic) interleaved to reinforce the dural matrix, and sealed with ethyl cyanoacrylate (Bostik, Australia) to reduce cerebrospinal fluid (CSF) leakage. Sham animals underwent all surgical procedures, including dural opening, CSF aspiration and MCA dissection, with the exception of direct electrocautery and occlusion of the MCA. 11 In all animals, a cranioplasty was performed with dental acrylic cement (Lang Dental, IL) to maintain the shape of the cranial cavity and homeostasis of ICP dynamics, and the wound closed. The head was returned to a neutral position for monitoring under general anaesthesia for 24 h.
Magnetic resonance imaging
At 24 h MRI was performed under 3% isoflurane on a 3 T scanner (Siemens Skyra; Siemens AG, Munich, Germany). Fluid attenuated inversion recovery (FLAIR), T2 weighted (T2WI), echo planar (EPI), magnetic resonance angiography (MRA) and diffusion weighted (DWI) images were acquired. Apparent diffusion coefficient (ADC) maps were automatically calculated using inbuilt Siemens software (Syngo.via vB10) where a log-linear regression model was used to calculate the ADC for each voxel acquired in the DWI (ADC reported as 10−3 mm2/s).
The extent of the midline shift was assessed from the T2WI at the level of the septum pellucidum (Horos DICOM image viewer v3.1.1). Cerebral oedema was calculated from the FLAIR images via computer-aided manual delineation of the hyperintense lesion by a blinded assessor using Horos DICOM image viewer (v3.1.1). Semi-automated segmentation of DWI data was performed with ITK-SNAP (v3.7) 22 to estimate infarct volume.
Histological examination
Following MRI, animals were maintained on inhalational isoflurane, intravenous heparin (5000 I.U./5 ml; Pfizer, NY) was administered and animals euthanised via exsanguination and perfusion with TRIS-buffered saline. The brains were removed and sliced into 10 mm coronal sections and immersed in 3% 2,3,5-Triphenyltetrazolium chloride solution (TTC, Sigma-Aldrich, Australia) at 37 °C in the dark for 10 min. Slices were photographed and lesion volume calculated as a percentage of the ipsilateral hemisphere and averaged for both posterior and anterior surfaces using ImageJ (v1.49) by an individual blinded to the treatment allocation.
Rodent studies
Animals and GTN administration
Seventy-four male C57BL/6 mice (Charles River, UK, approx. 25–30 g, 10–12 wks old) were housed under standard conditions (21–23 °C, 12 h light-dark cycle) in individually ventilated cages, with standard bedding and a tube, and fed ad libitum on standard rodent chow.
A GTN dose of 0.84 µg/h was calculated (based on body surface area) to be equivalent to 0.2 mg/h in humans. 4 Nevertheless, a range of doses were tested. To examine the effect of GTN on CBF, 16 mice were randomised to receive a transdermal patch that was adjusted for size (50 µg/h = 1.6 cm2, 12.5 µg/h = 0.8 cm2, 3.12 µg/h = 0.4 cm2, and 0.69 µg/h = 0.2 cm2), to compare to baseline measurements. An additional 12 mice were similarly randomised for tissue oxygenation measurements. This number of mice was chosen for these exploratory studies to see if any strong effect on cerebral blood flow was evident, and therefore the data is presented with descriptive statistics only.
As there were limitations with using transdermal patches for recovery experiments, the effect of GTN on stroke outcome in 46 mice was assessed using pre-primed osmotic mini-pumps (8.0 µl/h for >24 h; ALZET, US; model: 2001 D), which would deliver GTN for the duration of the study. An independent investigator used adaptive randomisation (4.1, University of Texas M.D. Anderson Cancer Centre) based on infarct volume data from the previous day, which assigns more animals to doses that appear to be effective. Power calculations based on lesion volumes from the same model produced in our group indicated for a power of 80% and alpha 0.05, to see a 50% reduction in lesion volume (37.5 mm3 to 18.75 mm3, 13.7 SD) 8 per group was required. Therefore, 46 mice entered the adaptive randomisation (with the additional 6 mice being included to account for any attrition). All experimenters performing surgery, carrying out behaviour and analysing infarct volume and NOx levels were blind to group allocation. Treatment group was random across time of day and cage of animal to control for bias. GTN (Hospira, UK, 5 mg/ml GTN in 67% ethanol and 4.3% PEG) was diluted further with PEG to deliver the following doses: 0.078, 0.78, 7.8 and 60 µg/h. Due to a limit on the alcohol concentration that could be delivered by the pumps (15%), the highest dose was prepared from a 5% stock solution (50 mg/ml GTN in 70% ethanol (UNIKEM, Switzerland) donated by Professor Eaton)). Control mice received the vehicle (15% ethanol in PEG). The pumps were inserted under the skin of the flank 20 min after stroke. A priori exclusion criteria included complications during surgery (failure to advance filament or excessive bleeding) prior to treatment. Meeting humane endpoints (for example, 20–30% weight loss, laboured breathing, or seizures) after treatment, resulted in euthanasia, but maximal scores were included in analysis of neuroscore and infarct volume (two animals in the 0.078 µg/h group). Final group sizes were: 0 (n = 10), 0.078 (n = 7), 0.78 (n = 9), 7.8 (n = 12) and 60 µg/h (n = 8).
Cerebral blood flow and oxygenation measurements
Mice underwent CBF measurements with laser speckle flow imaging (moorFLPI-2, Moor Instruments, UK). Animals were anaesthetised with isoflurane anaesthesia in room air and artificially ventilated (Zoovent Jetsys ventilator, Universal Lung Ventilators Ltd, Milton Keynes, UK). Animals were fixed in a stereotactic frame (Narishige, Tokyo, Japan) and body temperature maintained at 36.9 ± 0.2 °C via a heating blanket controlled with a rectal probe (Harvard Apparatus, Kent, UK). Heart rate was monitored via ECG throughout the experiment. A midline incision in the scalp was made and the skull exposed. CBF was recorded at baseline (timepoint 1), during (timepoint 2), and after (timepoint 3) the application of GTN via the transdermal patches. Data from one mouse was of insufficient quality to include in analysis (0.69 µg/h group).
Two-dimensional optical imaging spectroscopy (Abbott, Berkshire, UK) was performed to measure tissue oxygenation. Animals were prepared as per above, and then a soft paraffin (Vaseline) well was constructed over the left somatosensory cortex and filled with 37 °C saline. The well was sealed with a glass coverslip to create a stable imaging window. 23 Imaging data were collected using a high signal to noise charged coupled device (CCD) camera (Pantera 1M30, DALSA, Munich, Germany). The region of interest was illuminated sequentially by four different wavelengths of light (550 ± 10,560 ± 10,577 ± 10, and 700 ± 10 nm) using a Lambda DG-4 high-speed filter changer (Sutter Instruments, Novato, CA, USA). Camera data collection was synchronised with the filter changer so that each image frame was recorded with one of the four wavelengths in a sequential manner at a rate of 28 Hz. A modified Beer Lambert law was used for the calculation of µM changes in oxyhaemoglobin (HbO2) concentration at all timepoints (baseline, 30 and 60 min after patch administration, and 30 and 60 min after patch removal), 4 data points could not be obtained (30 and 60 min at 0.69 and 12.5 µg/h).
Transient MCAO
Briefly, animals were anaesthetised using isoflurane and a silicone coated nylon filament (diameter: 0.19 mm, Doccol, US) was advanced up the right common carotid artery and secured. 24 20 min after occlusion preloaded minipumps (see above) were inserted under the skin of the flank. The filament was removed after 30 min, the common carotid was ligated and the wounds sutured. Body temperature was maintained with a homoeothermic blanket system (Harvard Apparatus, Kent, UK) and mice received subcutaneous saline. A line block of Ropivacaine Hydrochloride (2 mg/kg; AstraZeneca, UK) was carried out at both surgical sites prior to surgery, and on two further occasions at 8 h intervals. Moist diet was provided the day before and after surgery, animals were monitored at least hourly once recovered from anaesthesia, until deemed well enough to be left overnight, then at least 4 times per 24 hrs.
Neurological score
At 24 h following MCAO, neurological deficits were determined using the Extended Neurological Score (general and focal) as described previously.25 –27 Prematurely euthanised animals were given maximum neuroscore values and included in the analysis.
Histological examination
Mice from the MCAO study were euthanised using an intraperitoneal injection of Euthatal (0.1 mL; 200 mg/mL, Merial Animal Health Ltd, UK). A blood sample was taken by cardiac puncture into tubes coated in K3 EDTA (1.3 mL; Greiner Bio-one, UK) and the brain was removed. The cerebellum was flash frozen for NOx analysis, and the forebrain sectioned into 1 mm slices and stained with 2% TTC (>95%; Sigma, US), to assess infarct volume. Sections were photographed with a camera (DS3100; Nikon, UK). Measurements of the infarcted and intact hemispheres were determined using ImageJ (v 1.49) and non-direct lesion volume was obtained to correct for oedema. 28 Prematurely euthanised animals were given maximum infarct volumes (110mm3) and included in the analysis.
Nitrate/nitrite analysis
Cerebellar tissue was homogenised in a lysis buffer (20 mM Tris and 10 mM EDTA, 1 mL to 100 mg of tissue) and spun at 3800 g in a cooled centrifuge for 20 min. The supernatant was collected and 5 µL of nitrate reductase and 5 µl of co-factor (Nitrite/Nitrate Assay Kit Sigma-Aldrich, UK) was added to a 40 µL sample and incubated for 2 h at 25 °C to reduce nitrate to nitrite and then to NO by acetic acid. The same process was repeated for the blood plasma. NO concentration was quantified using the Sievers Nitric Oxide Analyser (NOA 280i; GE Instruments, UK). Prematurely euthanised animals were not used in this analysis and 3 plasma and 2 brain samples were not of sufficient quality to obtain a reading.
Statistical analysis
All data are expressed as mean ± SD, the experimental unit is an individual animal, and analysis was carried out in GraphPad prism (v 8.0.1). Data was tested for normality using the Shapiro-Wilk normality test, or by assessing Q-Q plots of residuals. A p-value <0.05 was considered significant throughout. The primary outcome for both ovine and murine studies was infarct volume.
In the ovine studies, physiological data (pH, pO2, pCO2) were averaged for each treatment group across all time points and analysed using one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison test. MABP and ICP data underwent a logarithmic exponential transformation, 12 , 29 and along with CPP, were analysed by repeated measures two-way ANOVA followed by Tukey’s post-hoc multiple comparison test. MRI infarct volumes, cerebral oedema, and midline shift were analysed by t-test. TTC infarct volume was analysed by Mann-Whitney U test. Linear regression analyses were run to assess the relationship between cerebral oedema, midline shift and ICP, where an r value >0.5 was considered significant. In the rodent model, NOx levels were analysed via one-way ANOVA. Infarct volume and neuroscore were analysed via Kruskal-Wallis.
Results
Ovine studies
GTN significantly lowers intracranial pressure without affecting cerebral perfusion pressure
All experimental procedures were carried out without complication. There was no statistically significant difference in pCO2 (F2,15 = 3.521, p = 0.055) or pH (F2,15 = 0.410, p = 0.67) between any of the groups (Supplemental Table 1). There was, however, a significant increase in pO2 (Interaction: F2,15 = 29.78, p < 0.0001) in GTN treated animals (pO2 602 ± 21 mmHg) when compared to sham (p < 0.0001; pO2 113 ± 14 mmHg) and vehicle (p < 0.0001; pO2192 ± 186 mmHg).
MABP remained within normal physiological limits in sham animals throughout the course of the experiment (Interaction F8,56 = 0.098, p = 0.999), but was significantly elevated in vehicle treated animals following stroke at 9 (p = 0.038), 14 (p = 0.011) and 19 h (p = 0.028) compared to sham. In GTN treated animals, however, there was no significant difference in blood pressure when compared to vehicle or sham at any of the time points (p > 0.05) (Figure 1(a)).
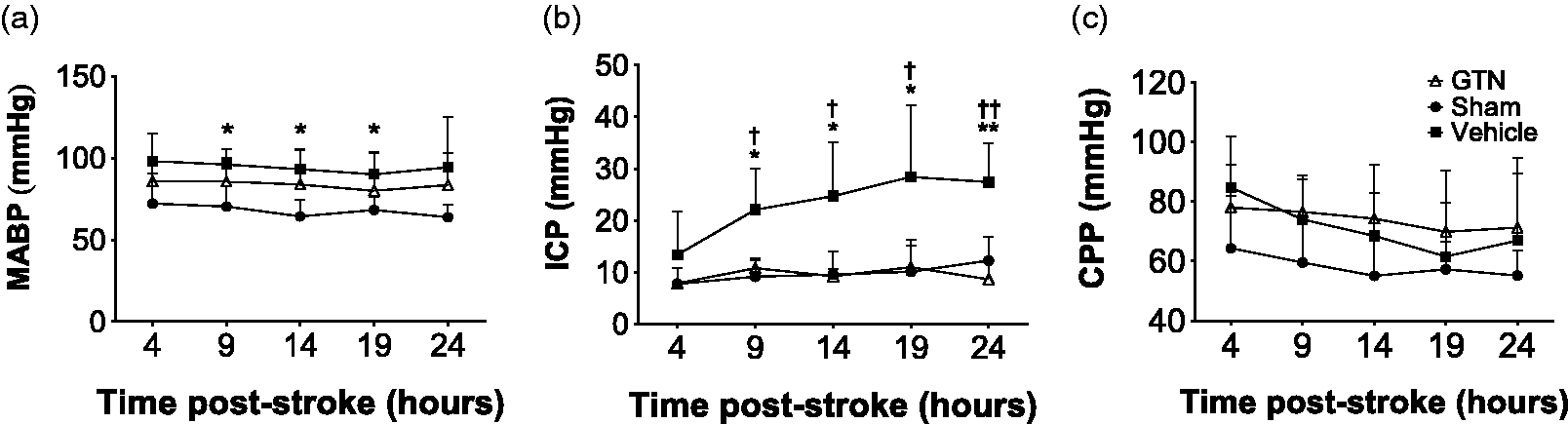
MABP, ICP and CPP over 24 hours following sham or MCAO surgery in sheep. Mean ± SD of MABP (a), ICP (b) and CPP (c) for all groups throughout the 24h monitoring period. *p < 0.05, **p < 0.01, vehicle cf. sham; †p < 0.05, ††p < 0.01 vehicle cf. GTN. Sham (n = 4), GTN (n = 7), Vehicle (n = 6).
At 24 h post-stoke, GTN prevented a rise in ICP seen in vehicle treated animals, maintaining pressure comparable to sham (Figure 1(b); Interaction F8,56 = 2.371, p = 0.028). There was no significant difference in ICP between any of the groups in the initial 4 h post-stroke (p > 0.05). By 9 h, however, the vehicle treated MCAO group exhibited significantly higher ICP compared to shams (p = 0.036) and GTN treated animals (p = 0.019). ICP continued to rise in vehicle treated stroke animals at 14 h (cf. sham, p = 0.033; cf. GTN, p = 0.034), 19 h (cf. GTN, p = 0.05) and 24 h (cf. sham, p = 0.002; cf. GTN, p = 0.006) (Figure 1(b)). ICP remained within normal physiological limits across all time points in both sham and GTN treated MCAO animals. CPP, however, was unchanged in any of the groups at any timepoint (Figure 1(c); Interaction F8,56 = 0.412, p = 0.901).
GTN reduces infarct volume and cerebral oedema
MRI was unremarkable in sham animals with no evidence of infarction or midline shift (Figure 2). All stroke animals exhibited hyperintensities on DWI and reduced ADC values within the MCA territory at 24 h. There was also strong hyperintensity observed in the same region with T2-weighted images.
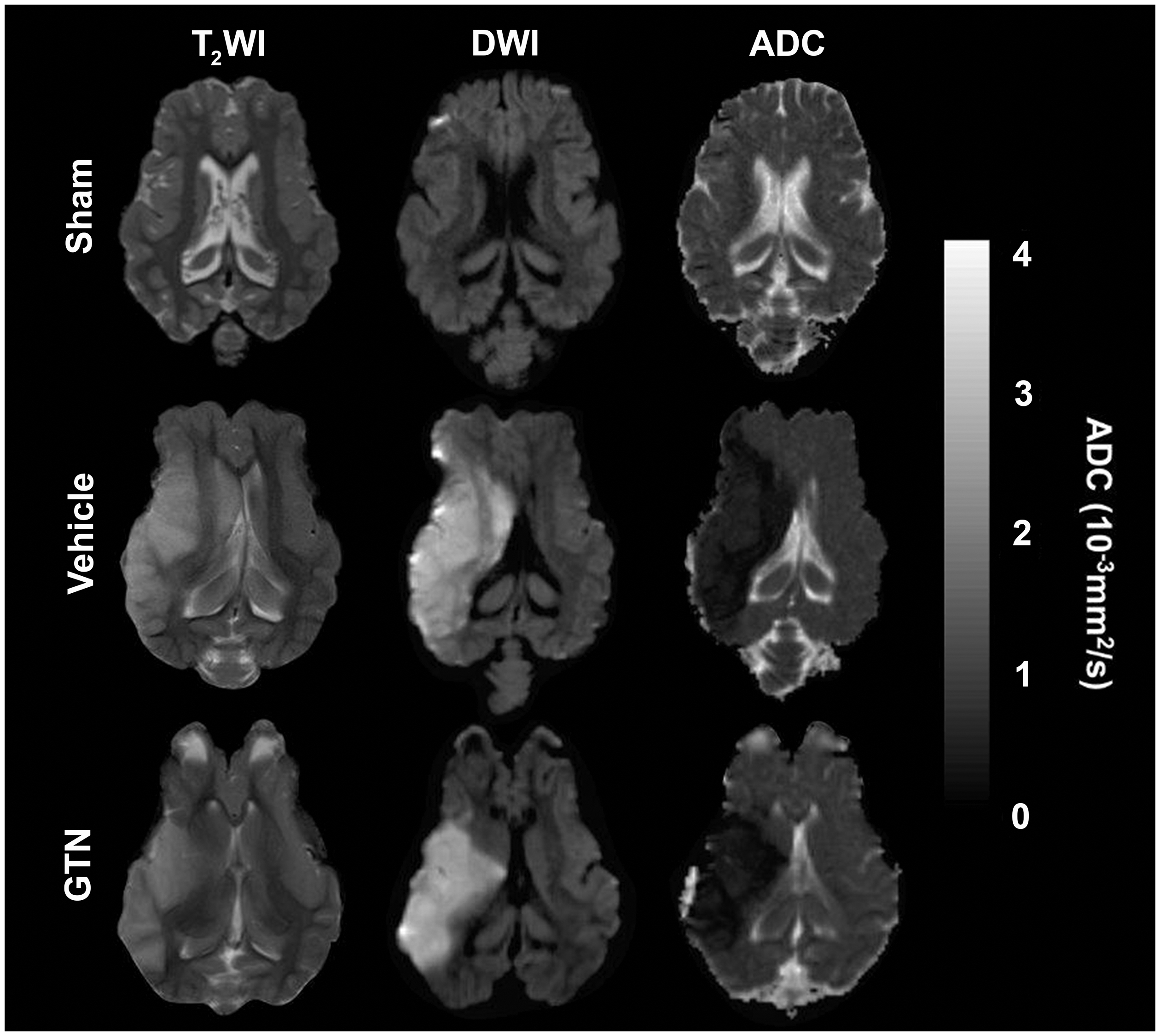
MRI findings 24 hours post sham or MCAO surgery in sheep. There was no evidence of hyperintensity or tissue deformation in sham animals. All stroke animals exhibited hyperintensity to T2 and DWI and lower ADC values (10−3 mm2/s). GTN (n = 7), Vehicle (n = 7).
On MRI, infarct volume was reduced in GTN compared to vehicle treated animals (GTN 19.32 ± 9.51 cf. vehicle 36.13 ± 17.02; t12 = 2.80, p = 0.042, effect size (Cohen) = 0.99 ± 0.08–2.36) (Figure 3(a)). The same reduction was also observed for cerebral oedema (t12 = 3.362, p = 0.0057) (Figure 3(b)). Midline shift was present in all stroke animals. However, the extent was markedly reduced in GTN animals compared to vehicle (t12 =2.920, p = 0.013) (Figure 3(c)). Infarct volume from TTC stained brain slices was also significantly reduced following GTN treatment when compared to vehicle (GTN 24.81 ± 12.58 cf. vehicle 46.23 ± 18.08; U = 6, p = 0.018, Figure 1 supplement). There was a strong positive correlation between infarct volume and cerebral oedema (Figure 3(d), r = 0.749, p < 0.001), and cerebral oedema and midline shift (Figure 3(e), r = 0.629, p < 0.001). There was also a positive relationship between ICP and cerebral oedema (Figure 3(f), r = 0.468, p < 0.01).
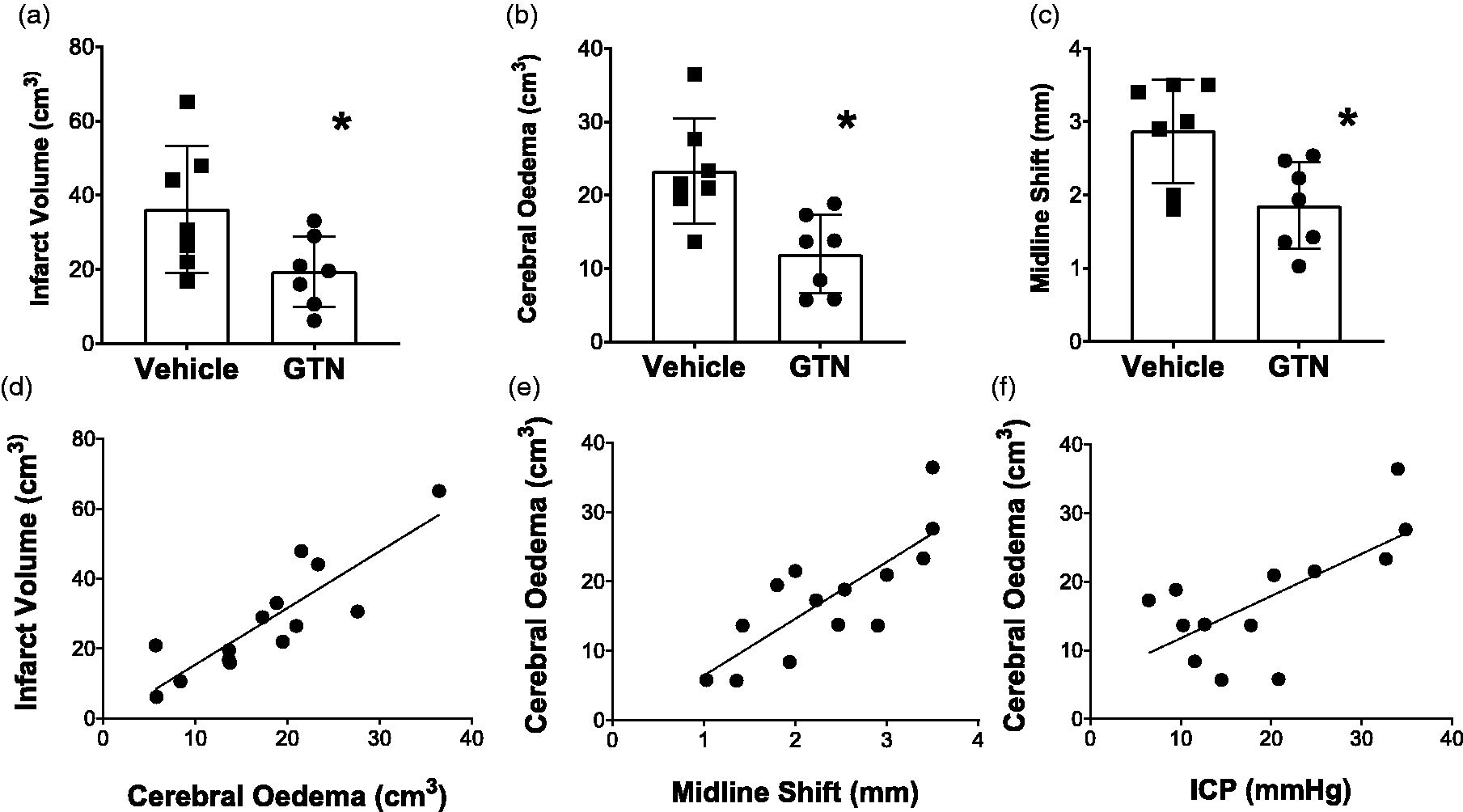
Infarct volume, oedema and the relationship to ICP in sheep. Mean ± SD of infarct volume (a), cerebral oedema (b) and midline shift (c) was all lower in the GTN compared to vehicle treated animals. (d) There was a strong positive relationship between cerebral oedema and infarct volume (r > 0.5) and (e) cerebral oedema and midline shift (r > 0.5). GTN (n = 7), Vehicle (n = 7 (except for ICP n = 6)).
Rodent studies
GTN does not affect cerebral blood flow or tissue oxygenation
Changes in CBF were observed following all GTN doses (Figure 4(a)). However, there was a high variability between subjects – some exhibited reductions and others increases in CBF. Even after the patch had been removed for 1 h, CBF did not consistently return to baseline.
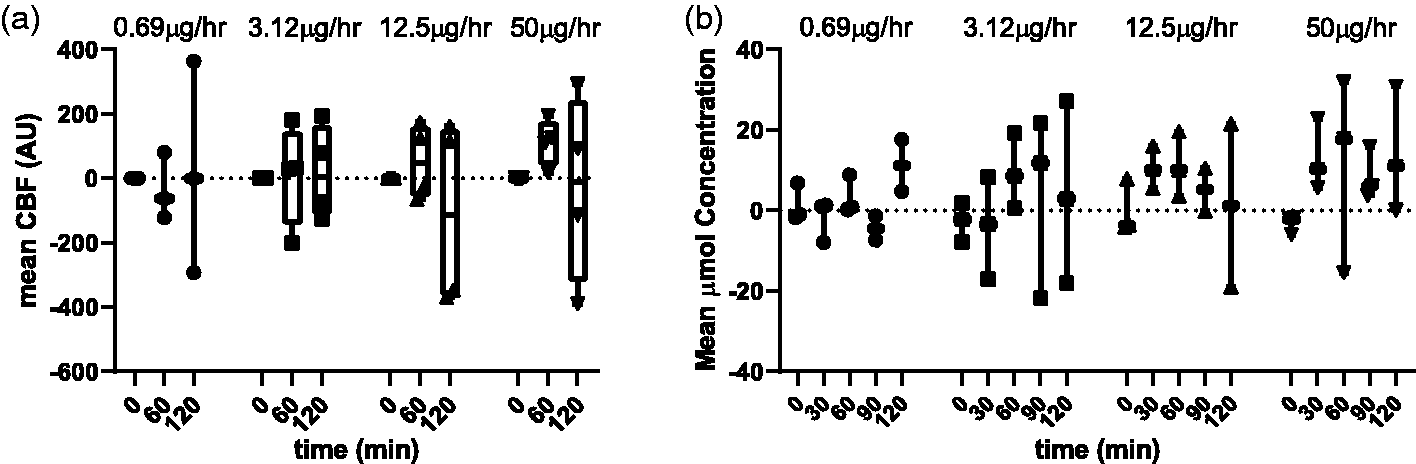
CBF and brain oxyhaemoglobin measurements with different doses of GTN in mice. (a) Mean CBF in arbitrary units for 4 different doses of GTN: 0.69 (n = 3), 3.12 (n = 4), 12.5 (n = 4) and 50µg/h (n = 4). ICP CBF changes were normalised against baseline at time 0. GTN was administered for 60 min, then removed and final measurements taken an additional 60min later (time: 120 min). Data: median, max and min and interquartile range. (b) Mean oxyhaemoglobin concentrations for 4 different doses of GTN: 0.69 (n = 3), 3.12 (n = 3), 12.5 (n = 3) and 50 µg/h (n = 3). GTN was administered for 60 min, then removed and measurements taken an additional 30 and 60 min later (time: 90 and 120 min). Data: median, max and min. 4 data points could not be obtained (brain oxyhaemoglobin, 90 and 120 min at 0.69 and 12.5 µg/h).
The HbO2 concentration at baseline was stable, but there was a large variation in values after GTN application. Each individual took up to 1 h to reach the peak change in oxygenation compared to baseline and few had returned to baseline 1 h after the patch was removed. The application of the different doses of GTN did not influence brain HbO2 concentrations (Figure 4(b)).
GTN has no effect on infarct volume and functional outcome after stroke
None of the GTN doses influenced infarct volume at 24 h (KW = 2.53, p = 0.639) (Figure 5(a)), and neurological score outcomes were also comparable across all groups (KW = 6.03, p = 0.197) (Figure 5(b)).
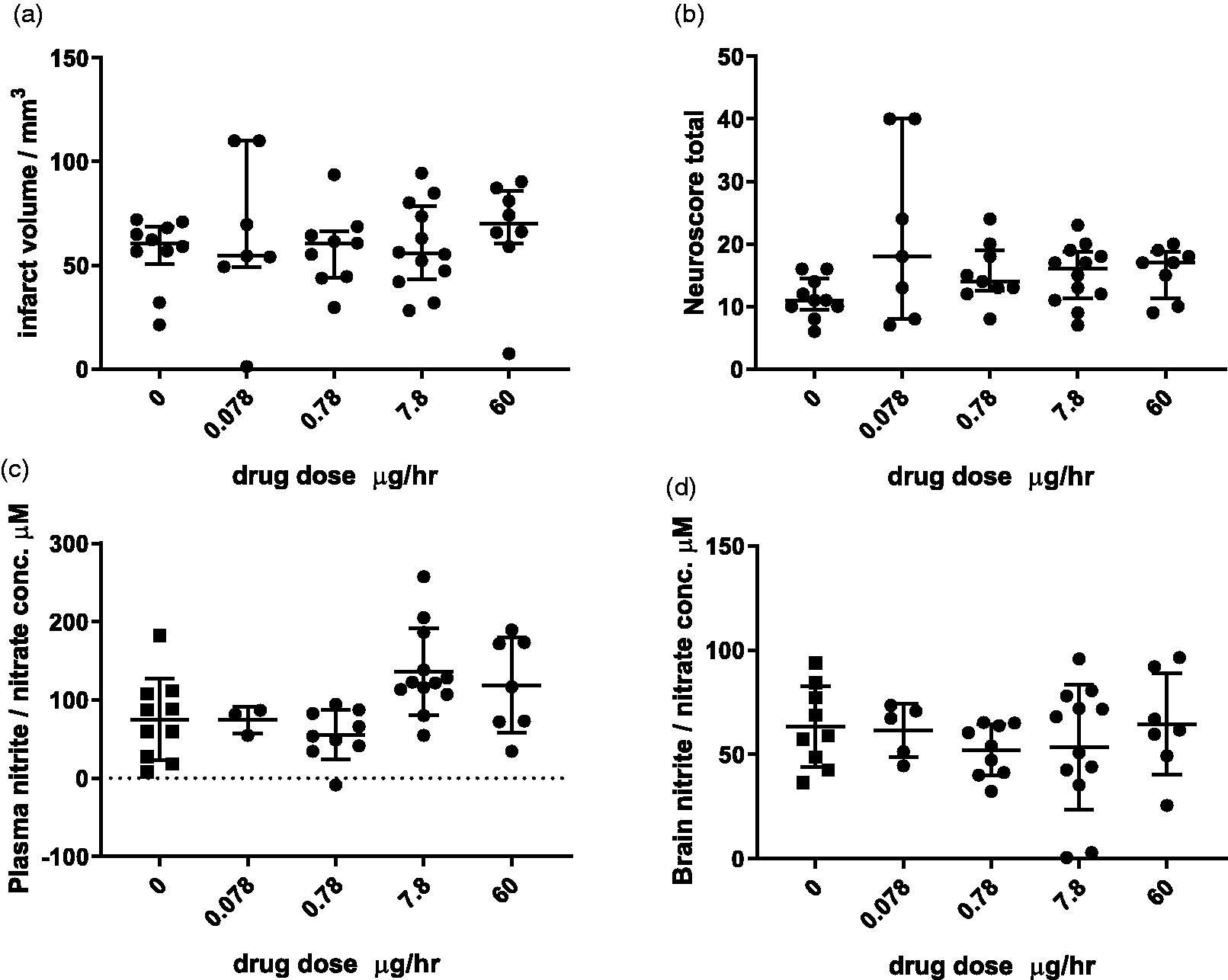
Infarct volume, neurological score and NOx levels following GTN administration in mice. Median ± interquartile range of infarct volume (a) and of neurological score (b). (c) Plasma and (d) brain concentrations of NOx (Mean ± SD). 0 (n = 9–10), 0.078 (n = 3–7), 0.78 (n = 9), 7.8 (n = 12) and 60 µg/h (n = 7–8). For NOx levels 3 plasma and 2 brain samples were not of sufficient quality to obtain a reading and were excluded.
Administration of 7.8 µg/h of GTN increased NOx concentration in the plasma (F4,36 =4.327, p = 0.005, post-hoc: 0 vs 7.8 µg/h: p = 0.025) (Figure 5(c)). However, NOx levels in the brain were not altered by GTN administration (F4,37 = 0.5752, p = 0.682) (Figure 5(d)). An additional study was performed with patch administration at 0.2 mg/h, which was also neutral (Supplemental Figures 2 and 3).
Discussion
This study demonstrates that sub-dermal GTN modestly reduced MABP and prevented dangerous elevations in ICP, without impacting CPP, in a permanent ovine model of stroke. GTN treatment also significantly reduced cerebral oedema and infarct size when compared to the vehicle group. However, the complementary rodent studies suggest that GTN appears to have no consistent effect on CBF and brain oxygenation, which is in line with clinical data, and was unable to influence outcome in a transient mouse model of stroke.
A clinically relevant dose of GTN, provided at 2 hrs post permanent MCAO in the ovine model via a subdermal patch that is used transdermally in humans, only marginally reduced MABP when compared to vehicle (approximately 10 mmHg). Whilst this reduction was not significant, vehicle treated sheep with stroke exhibited significantly higher MABP than the sham surgery group. This suggests that GTN was able to slightly reduce MABP, a reduction that is within the range of systolic blood pressure reduction of 5–20 mmHg in clinical studies. 3 , 4 , 6 , 10 The most interesting finding from this study is that GTN was extremely effective at preventing a rise in ICP without affecting CPP. While CPP was slightly higher in both vehicle and GTN-treated stroke groups compared to sham, there were no significant differences among any of the three groups. These findings are consistent with a recent report in which cerebral perfusion was shown to be unaffected by pharmacological reduction of MABP (by approximately 12 mmHg) in patients with ischaemic stroke. 30 The authors also observed no increase in the volume of the infarct core, penumbral tissue nor the total region of hypoperfused tissue following administration of antihypertensive treatment with beta-blockers or nitrates at >24 hrs post-stroke onset. The effect of long duration antihypertensive therapy, however, remains elusive. In stroke patients treated with GTN, despite a reduction in MABP, no significant overall effect on CPP or CBF measurements were seen, and on the contrary, some patients showed increased CBF in and around the infarcted region. 10 While the present study did not look at mechanisms by which GTN may be lowering ICP, it could be by directly combating cerebral oedema. There are many molecular mechanisms proposed to be involved in the formation of cerebral oedema in response to ischemia, such as increases in sodium influx and calcium signalling, break-down of blood-brain barrier (BBB) tight junctions, enzymatic activity and inflammation, reviewed by Simard et al. 31
Using the same ovine model of stroke, we have previously shown that blocking neurogenic inflammation with an NK1 tachykinin receptor antagonist was able to substantively decrease ICP and prevent BBB breakdown, potentially through the upregulation of transcellular mediator calveolin-1. 14 BBB integrity is well known to be differentially mediated by NO depending on the NO donor, with inducible and neuronal nitric oxide synthase (NOS) generally considered detrimental, whereas endothelial NOS is considered beneficial, reviewed by Thiel & Audus. 32 Vascular NO is reduced in stroke patients, 33 and GTN may be acting via restoring this. Within our study the effect of GTN on blood pressure suggests endothelial availability, although more research could be done to understand cerebral absorption and biodistribution, particularly since we did not measure any brain NOx changes in mice across the wide range of doses. Although we observed a correlation between oedema and ICP, there might be additional mechanisms beyond oedema that have not been considered. Indeed, increases in ICP in rodent models of stroke have been reported that were not associated with oedema 34 , 35 suggesting previously unknown mechanisms of ICP regulation that require additional investigation. One limitation of the ovine data is that there were unexpected facility changes that meant the GTN treated animals had to be maintained with pure oxygen rather than room air. This led to significantly higher pO2 levels in the GTN group compared to sham or vehicle. Breathing 100% oxygen (hyperoxia) is well established to cause cerebral vasoconstriction and subsequent reduction in CBF, 36 , 37 albeit without impacting pressure autoregulation dynamics. 38 Following a cerebral insult such as traumatic brain injury, hyperoxia also results in cerebral vasoconstriction, 39 along with significantly impaired pressure autoregulation. 40 , 41 However, it is generally assumed that the increased oxygen-carrying capacity under hyperoxic conditions more than outweighs the small reduction in CBF. We cannot exclude that hyperoxia may have impacted our MABP results and potentially reduced CBF. However, given that CPP and pCO2 (a much more potent cerebral vasodilator) levels were preserved, this is unlikely. Indeed, in a recent ovine stroke study utilising 100% oxygen, no significant differences in the sham animals with respect to ICP or MABP were observed. 15 A recent systematic review of the literature surrounding hyperbaric oxygen therapy in stroke suggested evidence from clinical trials is inconclusive with negative, positive and neutral results all reported. 42
While CBF was not directly measured in the ovine experiments, the murine results suggest that GTN did not influence CBF in a robust fashion. This is very similar to findings with intraarterial administration in the transient mouse MCAO, 17 as well as in healthy volunteers treated with intravenous GTN in which global CBF remained unchanged along with internal carotid artery flow velocity, though there was a dose dependent decrease in flow velocity of the MCA. 43 Stroke patients treated with GTN patches also exhibited reduced MCA flow velocity 44 and another report indicated no effect of GTN on CBF in stroke patients. 10 Despite no overall effect, high variability among patients was reported, and we observed the same in the mice. It could be that GTN has differential effects on CBF depending on differences in intra-individual absorption rates due to sub-cutaneous fat or even patch adhesion problems in the mice. There could also be potential issues with bioavailability. Indeed, plasma NOx levels were only noticeably higher than vehicle at the two highest doses (7.8 and 60 µg/h) and even then, at 60 µg/h, were too variable to achieve significance. However, when plasma NOx was measured in stroke patients treated with GTN at a range of doses (5–10 mg), there was no change compared to control. 44
The results of the murine experiments also suggest that GTN was unable to reduce infarct size across a wide range of doses, nor provide functional benefit. This is in direct contrast to the study by Maniskas et al. 17 in which a reduction in infarct volume was observed with the two highest doses of GTN (6–12µg/µL) along with reduced motor impairments on the rotarod. This study administered GTN intra-arterially, so it is possible that there are differences in distribution and metabolism of GTN between species, and a greater potency can be achieved via this route compared to sub-cutaneous absorption from our mini-pumps. A comparable rat study administered high dose (10 mg) GTN intra-peritoneally before MCAO and also showed a reduction in infarct volume and reduced levels of anti-apoptotic Bcl-2 expression. 16 This high dose could have resulted in better availability than our rodent study, however, it is not a clinically relevant treatment window. In addition, in the ovine studies ketamine was required for anaesthesia, and this may have had a synergistic interaction with GTN to improve CBF, improving the efficacy of the administered GTN. Timing of treatment across the studies is one factor that may account for the differing results. For our mouse studies we chose 20 min post occlusion for GTN administration as this was the most acute timepoint possible within the range of the confines of the surgical procedure. Timing was a key consideration in the clinical studies, with early evidence indicating ambulance-based delivery within 2 h of symptom onset was ideal. 3 A subset of the ENOS patients treated early (within 6 h) also showed improved recovery, 5 but the RIGHT-2 patients treated within 4 h did not have a more favourable outcome. 6
Overall, it is difficult to compare absorption and bioavailability in rodents to large animals and humans, even though our plasma analysis in rodents is in line with the clinical literature. The ovine model offered the advantage that the exact same patch available for humans could be used along with a comparable body mass, whereas the small body size of the rodent resulted in adherence issues that made mini-pumps preferable for recovery experiments. In addition to potential NO availability, the ovine model was also permanent, and it is feasible that GTN may be more beneficial in conditions of ongoing cerebral ischaemia. The transient model has a slightly delayed penumbra by comparison and can be accompanied by reperfusion injury in which GTN may be unable to offer benefit. The permanent model is also characterised by faster lesion evolution and considerable mortality due to development of space occupying oedema, elevated intracranial pressure and resultant tonsillar herniation. 12 Consequently, studying time-points beyond 24 hrs in the permanent MCAO model is not feasible practically or ethically. Nevertheless, assessment of GTN treatment in the permanent ovine stroke model is warranted to determine the durability of the treatment effect. Once established, treatment could then be assessed in a transient model 15 which permits the recovery of animals following stroke induction, thereby enabling the investigation of longer time-points and additional outcome measures such as function. Indeed, examination in both permanent and transient large animal stroke models, in addition to multiple time-points, is essential when assessing the efficacy of any therapy or intervention pre-clinically. However, all of these aspects are not achievable in a single study and must be performed in a stepwise manner. First by starting with non-survival permanent MCAO studies with acute time-points before extending to survival transient MCAO studies allowing for longer and more comprehensive assessment of outcomes. Additional limitations that should be noted are a lack of randomisation in the ovine studies due to use of historical vehicle and sham animals, and that neither ovine nor murine models were aged or had co-morbidities.
Together these results cannot indicate which model has greater translational relevance, but it does highlight the benefits of using multiple models to enhance translational work. Our results suggest that GTN’s ability to reduce ICP in the sheep might have implications for patients with malignant infarction or a substantive midline shift that would normally be treated with decompressive craniectomy or osmotherapy. 31 A greater understanding about the mechanisms of GTN action and its interaction with the pathophysiological time course of stroke may reveal subsets of patients that would benefit from treatment.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211018901 - Supplemental material for Glyceryl trinitrate for the treatment of ischaemic stroke: Determining efficacy in rodent and ovine species for enhanced clinical translation
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211018901 for Glyceryl trinitrate for the treatment of ischaemic stroke: Determining efficacy in rodent and ovine species for enhanced clinical translation by Annabel J Sorby-Adams, Annastazia E Learoyd, Philip M Bath, Fiona Burrows, Tracy D Farr, Anna V Leonard, Ingo Schiessl, Stuart M Allan, Renée J Turner and Rebecca C Trueman in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the FP7-HEALTH-2013-INNOVATION-1 [603043-2]; Biotechnology and Biological Sciences Research Council [BB/F016956/1]; and the Wellcome Trust [204843/z/16/z]. Supporting FB (awarded to SA), AEL and RCT respectively. PMB is Stroke Association Professor of Stroke Medicine and is a NIHR Senior Investigator. This research was funded in whole, or in part, by the Wellcome Trust [204843/z/16/z].
Acknowledgments
The authors would like to thank Sunthara Rajan Perumal for the acquisition of the ovine MRI and Shenyi Peng, Tony Nguyen and Martina Kontos for their contribution to the MRI analysis. Professor Philip Eaton, Queens Mary University London for the 50mg/ml GTN in 70% ethanol. We are also grateful for technical assistance provided by Michelle Hammett during some of the rodent studies. We would also like to acknowledge the facilities, scientific and technical assistance of the National Imaging Facility, a National Collaborative Research Infrastructure Strategy (NCRIS) capability, at Large Animal Research and Imaging Facility, South Australian Health and Medical Research Institute (SAHMRI).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PMB has received grants from the British Heart Foundation, BUPA Foundation, Hypertension Trust and UK Medical Research Council supporting trials of GTN in clinical stroke. The other authors declare that there is no conflict of interest.
Authors’ contributions
PMB, RCT, RJT, SMA and IS conceived the studies. AJSA, AVL and RJT completed the ovine studies. AEL, PMB, TDF, IS, FB, SMA and RCT completed the rodent studies. AJSA, AEL, TDF, RJT and RCT wrote the manuscript which was revised and approved by all authors.
ORCID iDs
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
