Abstract
Circulating blood endothelial progenitor cells (EPCs) contribute to postnatal vasculogenesis, providing a novel therapeutic target for vascular diseases. However, the molecular mechanism of EPC-induced vasculogenesis is unknown. Interleukin-6 plays multiple functions in angiogenesis and vascular remodeling. Our previous study demonstrated that the polymorphism (174G > C) in IL-6 gene promoter was associated with brain vascular disease. In this study, we investigated if IL-6 receptor is expressed in human EPCs derived from circulating mononuclear cells, and if interleukin-6 (IL-6) stimulates EPC angiogenesis in vitro. First, we isolated and cultured mononuclear cells from adult human circulating blood. We obtained EPC clones that were further cultured and expended for the angiogenesis study. We found that the EPCs possessed human mature endothelial cell phenotypes; however, they proliferated much faster than mature endothelial cells (P <0.05). We then found that IL-6 receptor (gp-80) was expressed in the EPCs, and that administration of IL-6 could activate receptor gp80/gp130 signaling pathways including downstream extracellular signal-regulated kinase 1/2 and STAT3 phosphorylation in EPCs. Furthermore, IL-6 stimulated EPC proliferation, migration, and matrigel tube formation in a dose-dependent manner (P <0.05); anti-IL-6 antibodies or IL-6 receptor could abolish these effects (P <0.05). These results suggest that IL-6 plays a crucial role in the biologic behavior of blood-derived EPCs, which may help clarify the mechanism of IL-6 inflammatory-related diseases.
Introduction
Interleukin-6, a multifunctional cytokine, is a critical factor in various physiologic conditions including immune regulation, hematopoiesis, and inflammation by modulating a variety of events, such as cell proliferation, differentiation, and apoptosis (Akira et al, 1993; Hibi et al, 1996; Jones et al, 1996; Kamimura et al, 2003; Taga and Kishimoto, 1997). Interleukin-6 receptor consists of a heterodimeric complex made up of two Ig-like containing proteins: interleukin-6 (IL-6) receptor (gp80 or CD126) and gp130. When IL-6 binds non-signal-transducing IL-6 receptor gp80, the signal-transducing receptor gp130 is subsequently activated. IL-6, IL-6 receptor gp80, and gp130 form a binding complex, which activates downstream signaling molecules such as signal transducer and activator of transcription-3 (STAT3) and ERK1/2 phosphorylation (Hirano et al, 2000; Kamimura et al, 2003; Oberg et al, 2006; Tebbutt et al, 2002; Yang et al, 2003; Zhang et al, 2006). Interleukin-6 participates in many kinds of diseases. Our previous studies demonstrated that IL-6 174C > G polymorphism is associated with brain arteriovenous malformation (AVM) patients (Pawlikowska et al, 2004). Brain AVM patients with IL-6 174GG polymorphism have higher IL-6 protein in both blood samples and surgical tissue (Chen et al, 2006). Recent studies show that IL-6 is a potent proangiogenic cytokine which stimulates cerebral endothelial cell and smooth muscle cell proliferation and migration in vitro (Nilsson et al, 2005; Wang and Newman, 2003; Yao et al, 2006), and promotes neovascularization in the cerebellum in IL-6 transgenic mice in vivo (Campbell et al, 1993).
Endothelial progenitor cells, first reported by Asahara et al (1997), were initially identified from human peripheral blood, which expresses CD34 or CD133. Both CD34+ CD133+ cell populations were differentiated from endothelial cells in vitro under appropriate endothelial differentiation-promoting conditions (Gehling et al, 2000). According to the initial discovery, EPCs were defined as cells that were positive for both stem cell markers such as CD34 or CD133, and endothelial cell markers such as KDR, vWF, VE-cadherin, Tie2, and CD31-positive staining (Miraglia et al, 1997; Quirici et al, 2001). Studies have shown that EPCs contribute to postnatal vasculogenesis, namely the de novo vessel formation, during wound healing, limb ischemia, or postmyocardial infarction (Khakoo and Finkel, 2005; Liew et al, 2006). Other studies have identified two types of EPCs from peripheral blood: spindle-like cells with limited proliferation capacity, and cobblestone-shaped cells with high expansion capacity (Hur et al, 2004). Although EPCs respond to growth factors and cytokines such as vascular endothelial growth factor (VEGF), thrombin, granulocyte-macrophage colony stimulating factor, and β-fibroblast growth factor (Liew et al, 2006), the effect of IL-6 on EPC function is still unknown.
In this study, we isolated, cultured, and characterized EPCs from adult human circulating blood. We examined whether IL-6 receptor existed in the EPCs, and if so, whether IL-6 stimulated EPC proliferation, migration, and tube formation on matrigel through gp80/gp130 signaling pathway.
Materials and methods
Endothelial Progenitor Cells Culture
All procedures were approved by the Institutional Review Board Service at the University of California, San Francisco. Circulating mononuclear cells were isolated from adult healthy human volunteers by using reagent accuspin system-Histopaque 1077 (Sigma-Aldrich, St Louis, MO, USA). Briefly, 20 mL blood was collected using an anticoagulant tube. The blood was mixed with Histopaque 1077 (10 mL) and phosphate-buffered saline (PBS, 10 mL), then centrifuged at 400 g for 30 mins at room temperature. Mononuclear cell layer was collected and the isolated cells were washed three times with PBS plus 2% fetal bovine serum and resuspended in EBM-2 MV Bullet kit system (Cambrex, Cottonwood, AZ, USA) consisting of 5% fetal bovine serum, hEGF, VEGF, hFGF-B, IGF-1, ascorbic acid, and heparin. About 1 × 107 cells per well were seeded in a six-well plate coated with human fibronectin (Sigma, St Louis, MO, USA) and cultured in a 5% CO2 incubator at 37°C. The first medium change was performed after 5 days of culture and then changed every 3 days. After the EPC colonies grew, cells were trypsinized and seeded onto a plate coated with 0.25% gelatin, named passage 1. All the experiments were performed using EPCs within 3 to 5 passages. Human cerebral endothelial cells were purchased from Cell Systems (Cell Systems, St Katharinen, Germany) as a control.
1,1′-Dioctadecyl-3,3,3′,3′-Tetramethylindocarbocyanine-Labeled Acetylated Low-Density Lipoprotein Uptake and Ulex Europaeus Agglutinin-1-Binding Assay
Endothelial progenitor cells were incubated in a medium containing 2.5 μg/mL of 1,1′-dioctadecyl-3, 3,3′,3′-tetramethylindocarbocyanine-labeled acetylated low-density lipoprotein (Molecular Probes, Eugene, OR, USA) for 1 h at 37°C, and then fixed with 2% paraformaldehyde for 10 mins. After washing with PBS, the cells were blocked using 2% goat serum for 1 h, and reacted with fluorescein isothiocyanate (FITC)-labeled Ulex europaeus agglutinin-1 (UEA-1; 10 μg/mL, Sigma) at room temperature for 1 h. Cells were washed to remove free UEA-1, counter stained with 4′,6-diamidino-2-phenylindole, and examined under a fluorescence microscope (Olympus, Japan).
Western Blot Analysis
Endothelial progenitor cells were scratched from plates for protein extraction using RIPA buffer, and protein concentration was determined by using a bicinchoninic acid assay (Pierce, Rockford, IL, USA). An equal amount of protein (40 μg) was loaded onto an electrophoresed 10% gel. Subsequently, proteins were electroblotted onto a polyvinylidene difluoride membrane (Bio-Rad, Richmond, CA, USA). After being blocked in 5% fat-free milk, the membrane was immunoprobed with primary antibodies (Table 1) at 4°C overnight. The interest proteins were detected by horseradish peroxidase-conjugated anti-rabbit antibody (1:10000, Pierce Biotech, Milwaukee, WI, USA), followed by development using SuperSignal West Femto kit (Pierce). The membrane was plastic-wrapped and exposed to Kodak film (Kodak Co., Rochester, NY, USA). Target band density was analyzed by using the software NIH image J.
List of antibodies used
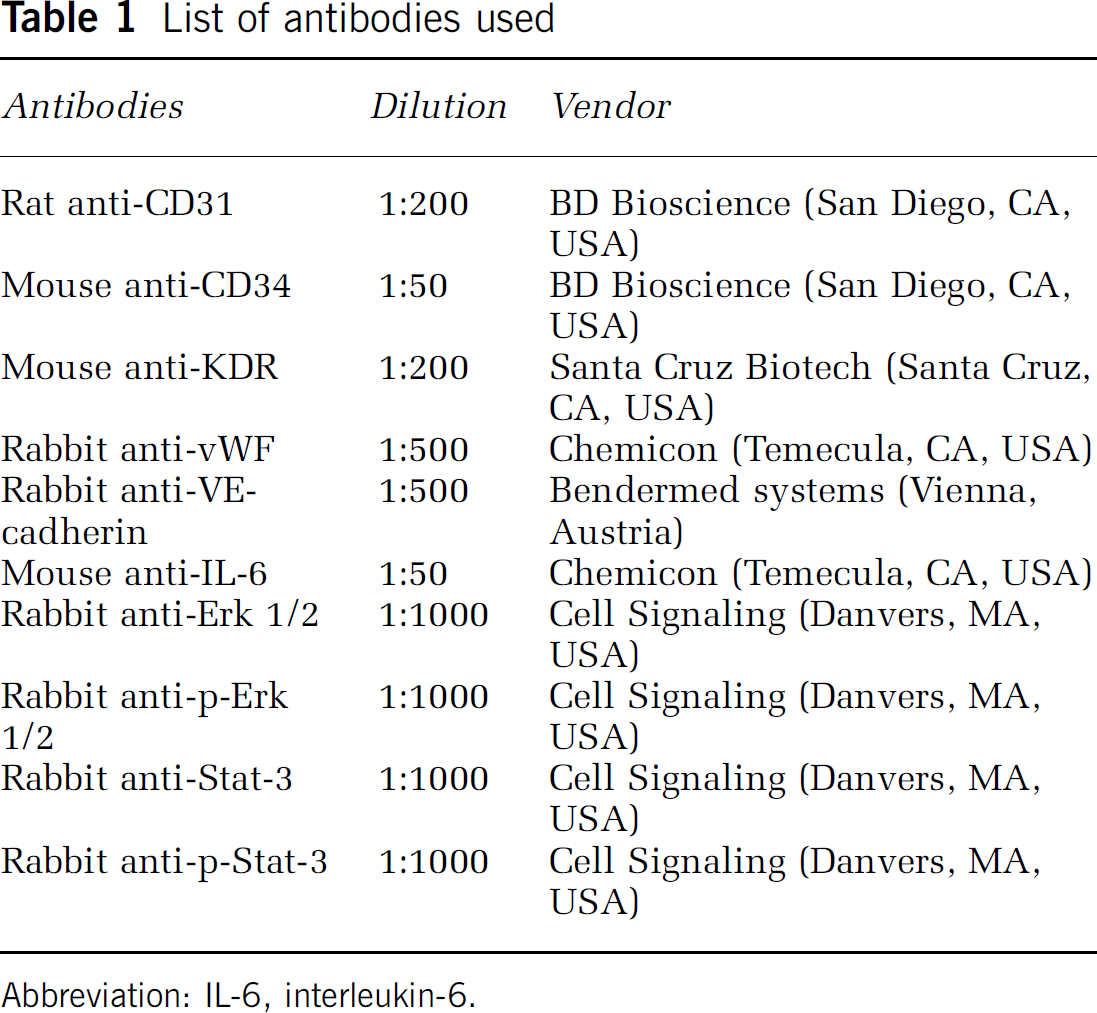
Abbreviation: IL-6, interleukin-6.
Immunostaining
Endothelial progenitor cells up to 75% confluence on the chamber slides were fixed using 4% paraformaldehyde and blocked in a blocking buffer (0.01 mol/L PBS with 2% goat serum, 1% bovine serum albumin (BSA), 0.1% gelatin, 0.1% Triton X-100, and 0.05% Tween-20). The cells were incubated in primary antibodies (Table 1) diluted in 0.01 mol/L PBS with 1% BSA, 0.1% gelatin at 4°C overnight. Second antibodies conjugated with fluorescence were applied (Molecular Probes). Fluorescent images were taken through a fluorescence microscope (Olympus, Japan). 4′,6-Diamidino-2-phenylindole was used for counterstaining and negative controls were performed by omitting the primary antibodies.
Endothelial Progenitor Cell Proliferation Assay
Bromodeoxyuridine incorporation was detected to evaluate the cell proliferation. Endothelial progenitor cells were seeded onto 96-well gelatin-coated plate (1 × 104) in EBM-2/Single Quote medium and cultured up to 75% confluence. After starvation overnight in EBM-2 with 0.1% BSA, recombinant human IL-6 (R&D Systems, Minneapolis, MN, USA) was added to the medium (EBM containing 0.1% BSA) at the indicated concentration. After 48 h of treatment, EPCs were labeled with bromodeoxyuridine (BrdU) overnight (BrdU Kit, Roche, Mannheim, Germany), and then fixed and denatured after application of alkaline phosphatase-conjugated anti-BrdU antibody for 90 mins. Absorbance at 450 nm was measured using an ELISA reader (E max, Molecular Devices, Sunnyvale, CA, USA) after 10 mins of incubation in subject buffer. The results were expressed as the mean percentage of OD450 over control groups.
Endothelial Progenitor Cell Migration Assay
Cell migration assay was performed using a 24-well tanswell cell culture chamber with 8.0 μm pore polycarbonate filter inserts (Costar, San Diego, CA, USA). Serum-starved EPCs or anti-IL-6 receptor antibody preheated EPCs were suspended in medium with 0.1% BSA at a concentration of 2.5 × 105/mL. About 2.5 × 104 cells were applied to precoat insert filters. Test factors were added in serum-free medium and placed in the lower chamber. After incubation at 37°C for 18 h, cells on the upper side of the filter were scraped and the migrated cells on the lower side were fixed and stained with hematoxylin. The membrane was mounted on a slide and examined under a microscope. Migrating cells were quantified by measuring the stained cells in five random areas per membrane, and the results were expressed as the mean percentage of migrating cells over control groups.
Tube Formation Assay
A matrigel tube formation assay was performed as described by Lee et al (1999). A 96-well culture plate was coated with 50 μl of growth factor-reduced matrigel (BD Bioscience, San Diego, CA, USA) per well and then allowed to polymerize for 30 mins at 37°C. Total 2 × 104 cells/well of starved EPCs or anti-IL-6 receptor antibody pretreated EPCs were seeded on the matrigel in 0.1% BSA/EBM-2 medium with testing factors and incubated for 18 h at 37°C. After being washed, images were captured with a light microscope. Tube formation ability was quantified by counting the total number of cell cluster and branching under three × 4 magnified fields per well. The results were expressed as mean folds of branching compared with the control groups.
For IL-6 neutralizing experiments, serum-free medium with IL-6 (25 ng/mL for proliferation assay and 50 ng/mL for migration and tube formation assays) plus anti-IL-6 antibody (1 μg/mL for proliferation assay and 2 μg/mL for migration and tube formation assays, R&D Systems) were first incubated for 1 h at 37°C, and then the medium was used to treat EPCs; for IL-6 receptor blocking experiments. First, EPCs were pretreated with anti-IL-6 receptor α (0.5 μg/mL, Chemicon, Temecula, CA, USA) at 37°C for 1 h to block IL-6 receptor α, and then were treated with 50 ng/mL of IL-6. Treatment with VEGF (20 ng/mL) was used as positive control.
Statistical Analysis
All experiments were performed at least three times, with the data being represented as mean ± standard deviation (s.d.). Results of the dose-dependent proliferation and inhibition experiments were analyzed using a one-way analysis of variance with Fisher's comparison tests. A random probability value < 0.05 is considered statistically significant for the comparisons.
Results
Endothelial Progenitor Cell Culture and Characterization
When cultured in specific endothelial growth factors (EGM-2 MV medium plus Single Quote), seeded mononuclear cells from human circulating blood were initially suspended with a round shape (Figure 1A). Partial cells were attached to the plates during the culture. After 5 days, the attached cells revealed a spindle shape (Figure 1B), called early EPCs (Hur et al, 2004). The early EPCs gradually disappeared within 2 weeks while several small cell colonies (or extended EPCs) formed (Figure 1C). At confluence, these EPCs were firmly attached to the plate, exhibiting the cobblestone-like morphology and monolayer growth pattern typical of the endothelial lineage (Figure 1D). The EPCs were characterized by 1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine-labeled acetylated low-density lipoprotein uptaking and FITC-UEA-1-binding experiments (Figure 2A). The endothelial cell phenotypes of EPCs were further confirmed using endothelial cell markers such as CD34, vWF, KDR, and VE-cadherin (Figure 2B). The results demonstrated that the EPCs revealed the endothelial cell's phenotype. Interestingly, the growth curve analysis showed that the EPCs proliferated much faster than mature human cerebral endothelial cells (Figure 2C, P <0.05).
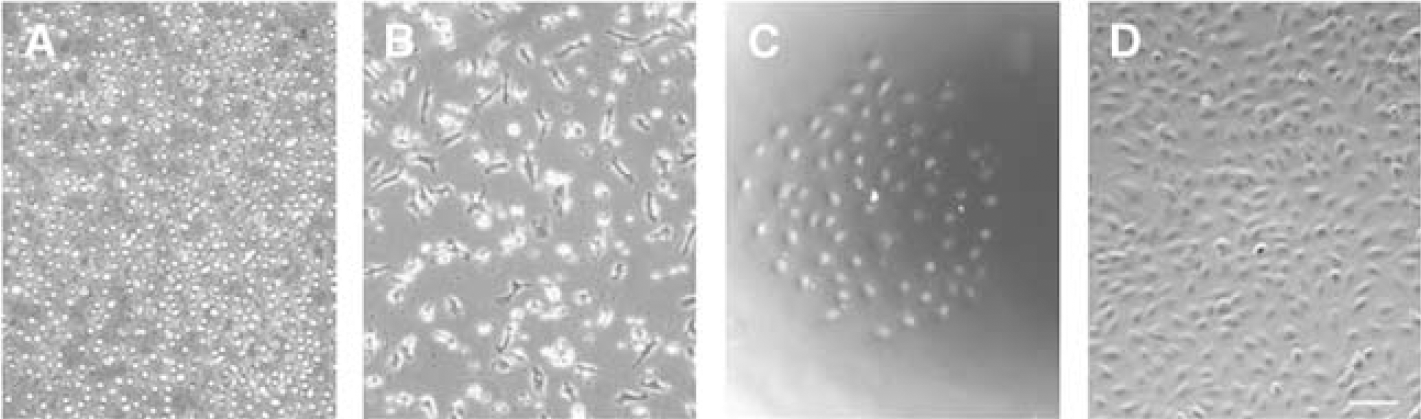
Endothelial progenitor cell culture. Phase-contrast figures show the morphology of EPCs. (
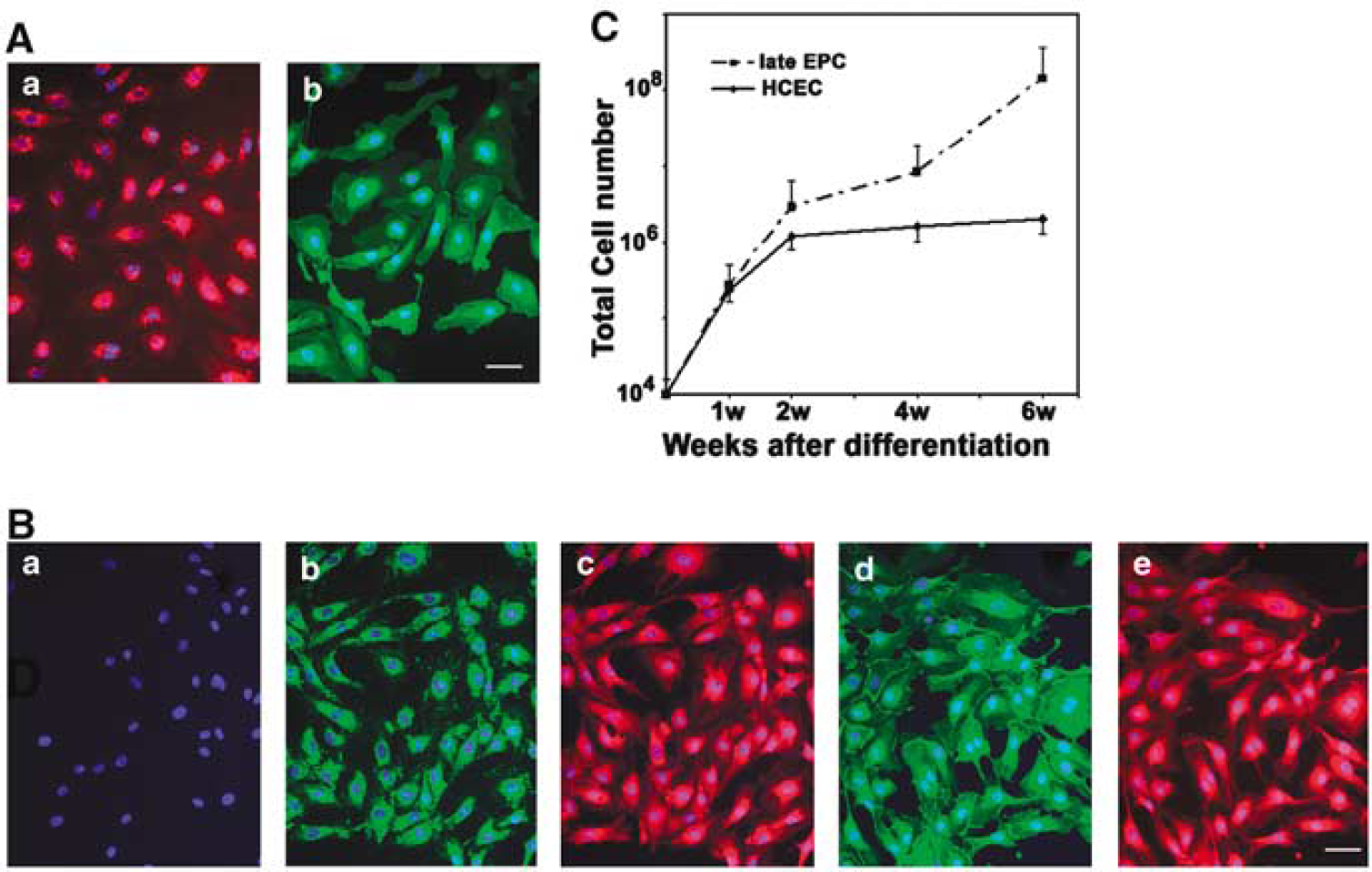
Extended EPC characterization. (
Endothelial Progenitor Cell Expresses Interleukin-6 Receptor and Activates Phosphorylation of Extracellular Signal-Regulated Kinase 1/2 and STAT3
To determine whether IL-6 receptor expressed in the EPCs, we performed immunohistochemistry. We demonstrated that the EPCs were gp80 positively stained, well colocalizing with endothelial cell marker VE-cadherin and CD31 (Figure 3A). To determine whether IL-6 activated gp130 signal pathway in the EPCs, we treated EPCs with IL-6 and then examined the downstream signaling molecules of IL-6/gp130. Phospho-ERK1/2 was detected as early as 10 mins, peaked at 30 mins, maintained up to 60 mins, and reduced at 120 mins after treatment of IL-6. Phospho-ERK1/2 was induced in a dose-dependent manner with optimal effects observed at 50 ng/mL (Figure 3B). Neutralizing antibodies against IL-6 or IL-6 receptor abolished IL-6-induced ERK1/2 phosphorylation (Figure 3C). We further examined whether IL-6/gp130 activated STAT3 signaling pathway. We found that phospho-STAT3 increased at 5 mins, peaked at 10 mins and disappeared at 120 mins after IL-6 treatment (Figure 3D). Interleukin-6/gp130 activated phospho-STAT3 in a dose-dependent manner, with optimal effects observed at 100 ng/mL. Neutralizing IL-6 antibody or IL-6 receptor showed abolished STS-3 phosphoralytion (Figure 3E).
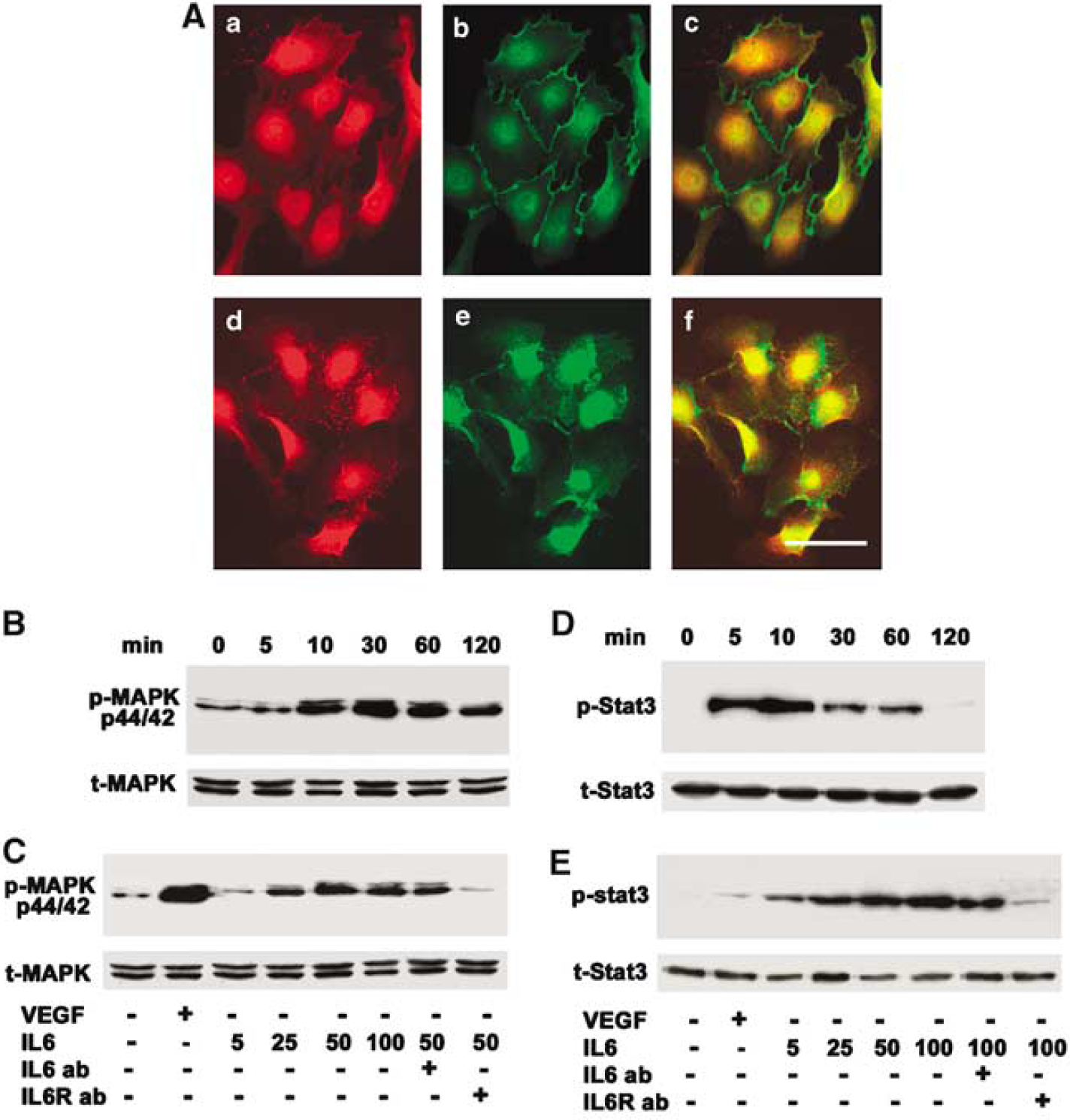
Identification of IL-6 receptor in EPCs. (
IL-6 Stimulates EPC Proliferation
To determine whether IL-6 stimulated EPC proliferation, we performed a BrdU incorporation assay. EPCs were starved overnight and were incubated with different doses of IL-6 for 48 h. We demonstrated that IL-6 induced BrdU incorporation in a dose-dependent manner with a maximal effect at 25 ng/mL (Figure 4A). BrdU incorporation in IL-6-treated EPCs increased more than 2 fold compared to the non-treated control, as VEGF achieved (Figure 4B, P <0.01). Furthermore, when pretreated with neutralizing antibody against IL-6 receptor, BrdU incorporation in EPCs was abolished (Figure 4B, P <0.01). This result indicates that IL-6 stimulates EPC proliferation.
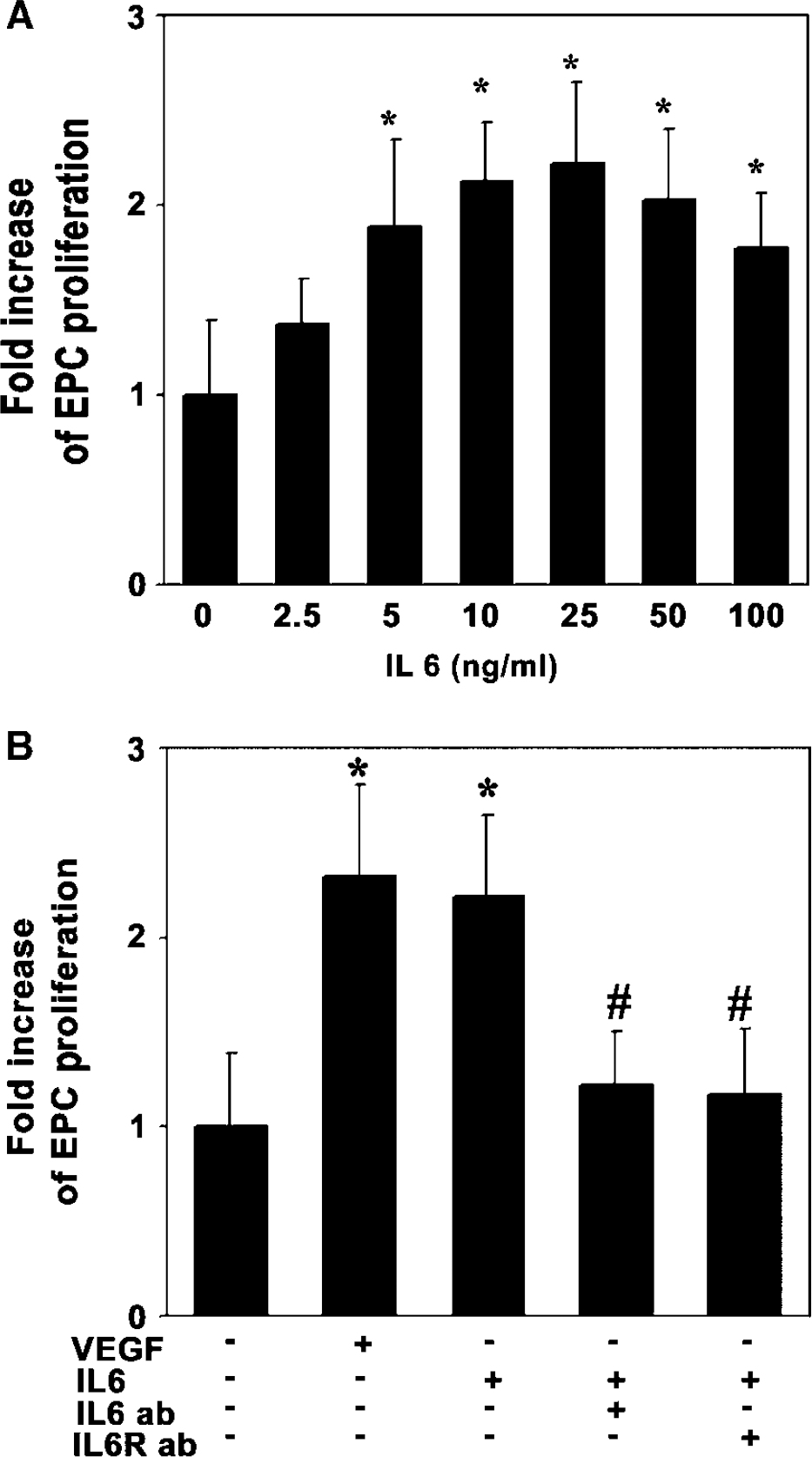
Interleukin-6 stimulates EPC proliferation. (
IL-6 Triggers EPC Migration
To determine whether IL-6 enhanced EPC migration, we counted cells moving through an insert filter in a chamber filled with BSA-free EBM2 containing testing factors after 18 h of migration. We demonstrated that IL-6 enhanced EPC migration in a dose-dependent manner, with an optimal effect at 50 ng/mL (Figure 5A). We further demonstrated that IL-6 significantly increased EPC migration compared to the control group (Figure 5B, P <0.01). Similarly, pretreatment with the neutralizing antibody against IL-6 or treatment with anti-IL-6 receptor significantly reduced EPCs migration compared to the IL-6 treatment (Figure 5B, P <0.05). This result demonstrates that IL-6 triggers EPC migration.
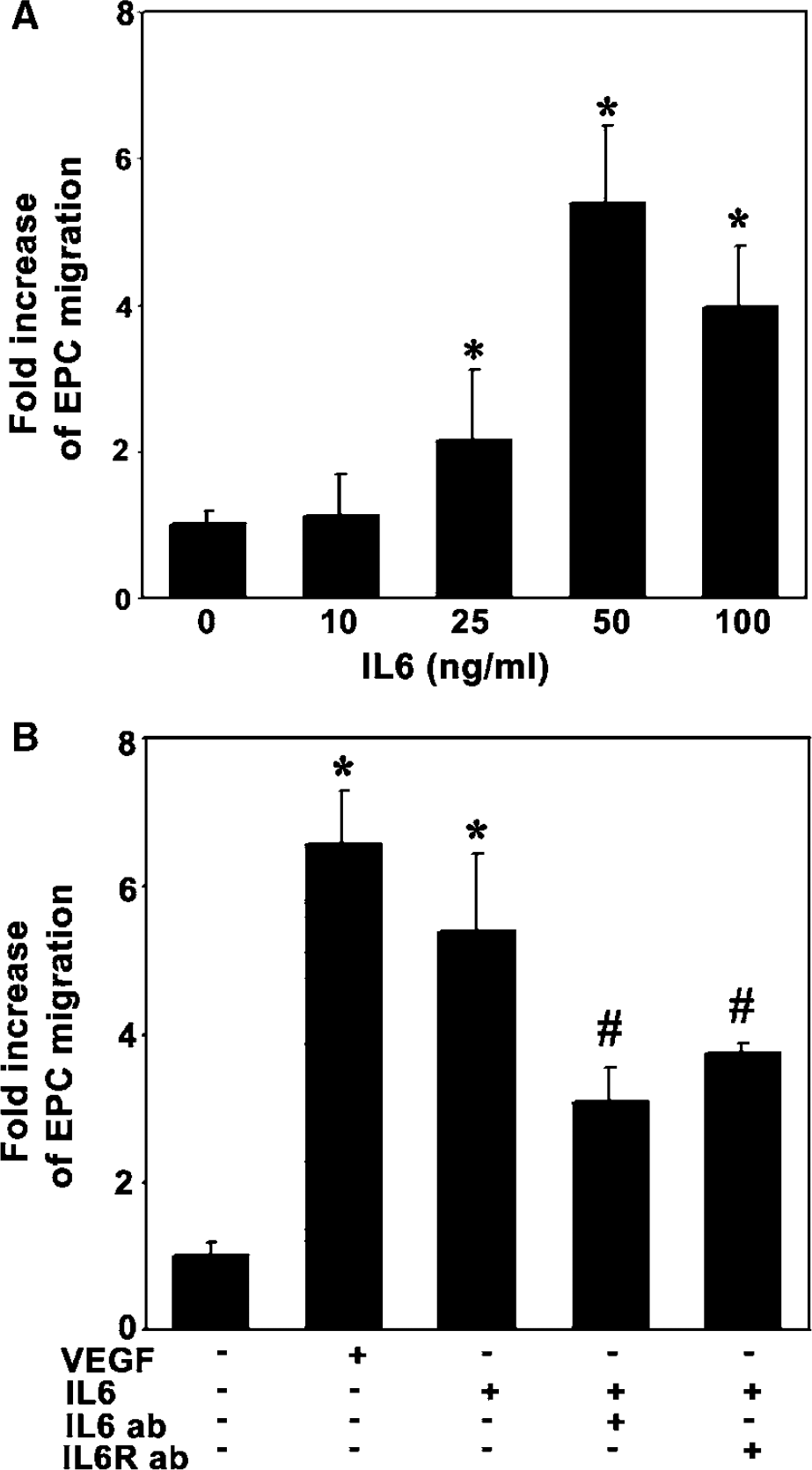
lnterleukin-6 attracts EPC migration. (
IL-6 Promotes EPC Tube Formation on Matrigel
We used a matrigel model to examine whether IL-6 would induce EPCs to differentiate to capillary-like structures. The EPCs formed few capillary-like structures when cultured for 18 h in EBM2 without serum. IL-6 significantly promoted EPC organization into branched structures and pseudotubes with enclosed areas on the matrigel (Figure 6A). This angiogenic effect occurred 8 h after IL-6 treatment, and persisted for up to 18 h. This effect revealed a dose-dependent manner, and the optimal dose of IL-6 for stimulating tube formation is 50 ng/mL (Figure 6B, P <0.01). Treatment with IL-6 neutralizing antibody or anti-IL-6 receptor could attenuate tube formation (Figure 6C, P <0.01), suggesting that IL-6 promotes EPC tube formation.
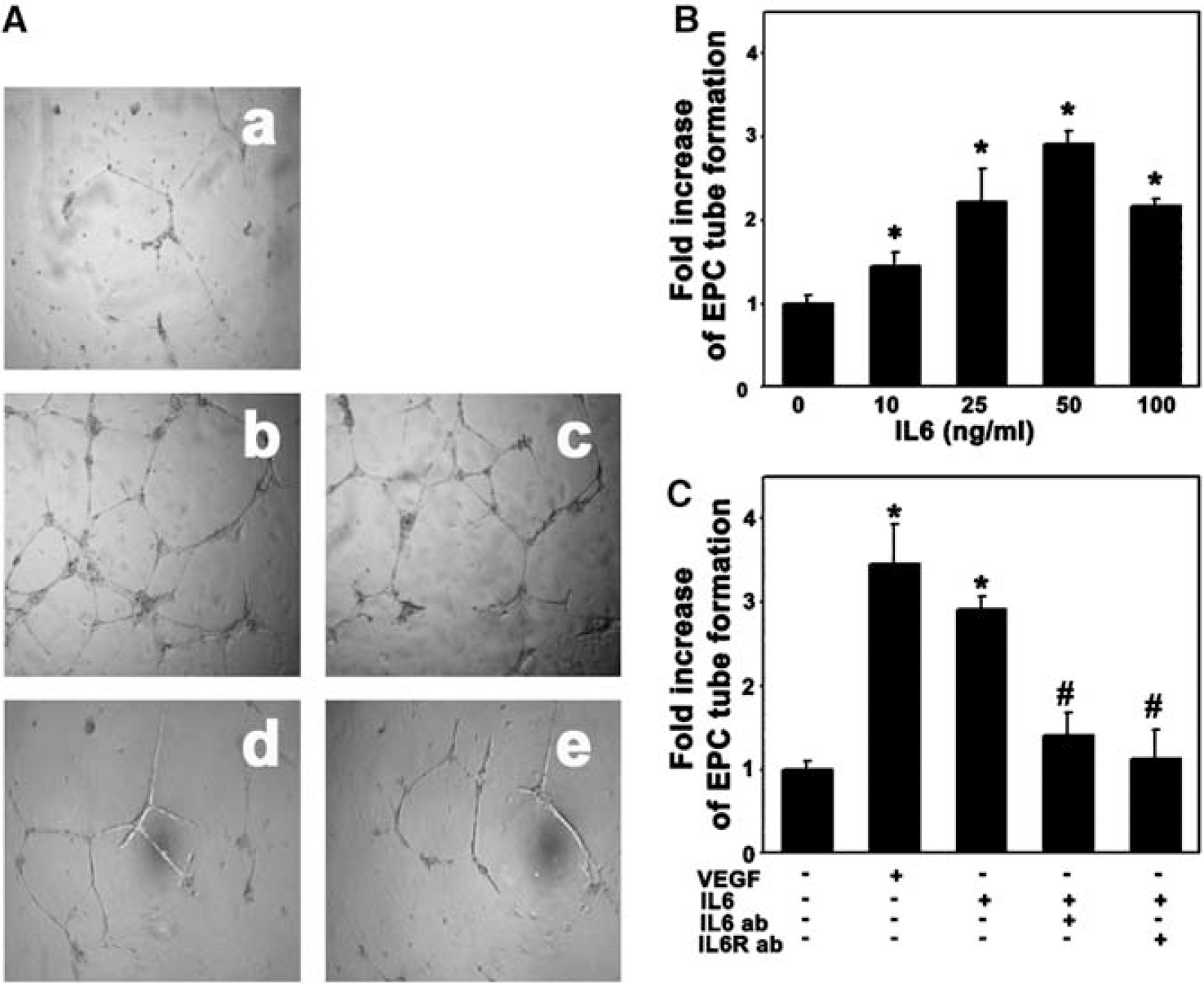
Interleukin-6 promoted EPC tube formation. To assess the ability of IL-6 to promote EPC to form microvessel-like tubes, we performed EPC tube formation assay. Vascular endothelial growth factor (20 ng/mL) acted as a positive control and PBS as a negative control. (
Discussion
Our previous studies demonstrated that IL-6 could stimulate human cerebral smooth muscle cell and endothelial cell proliferation and migration (Jee et al, 2004; Yao et al, 2006), which function as an angiogenic factor (Huang, 2004 #18412; Yao et al, 2007). In the present study, we provide new evidence regarding the role of IL-6 on angiogenesis in EPCs in vitro. We successfully isolated, characterized and cultured EPCs from human circulating blood. We demonstrated for the first time that: (1) EPCs expressed the functional IL-6 receptor; (2) IL-6 could activate gp80/gp130 signaling pathways on EPCs; and (3) IL-6 could stimulate EPC proliferation, migration and enhanced tube formation on matrigel in vitro.
Endothelial progenitor cells were initially identified through the isolation of CD34-positive cells from human peripheral blood. Later studies demonstrated that the source of EPCs included adult bone marrow (BM) (Reyes et al, 2002), peripheral blood (PB) (Lin et al, 2000; Peichev et al, 2000), cord blood (Murohara et al, 2000), and tissue resident cells (HSC, MSC, cKit+, neuronal progenitor cells). We have chosen to study EPCs from peripheral blood because circulating EPCs appear to be extremely important in that they directly contribute to endothelial recovery or rapidly migrate into the region of activating angiogenesis. In addition, it is relatively easy to obtain blood EPCs compared to bone marrow EPCs, which would be convenient for future clinical application. In this study, we particularly chose a homogeneous population of EPCs that directly incorporated into the neovascularization (Hur et al, 2004). We found that EPCs could rapidly replicate from several cells to a colony and grow to a monolayer with almost full confluence. To identify the purification of these EPCs, we checked cell phonotype and used progenitor cell and endothelial cell markers. We also performed 1, 1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine-labeled acetylated low-density lipoprotein uptake assay and FITC-UEA-1-binding experiments. The results demonstrated that EPC in peripheral blood could be cultured to a uniform population with high proliferation potential without senescence.
Interleukin-6 has been shown to be a potent proangiogenic cytokine, because IL-6 participates in angiogenesis during tumor progression, wound healing, and brain vascular system development (Fee et al, 2000). Interleukin-6 promotes matrix metalloproteinase-9 (MMP-9) activation and induces release of VEGF from cultured endothelial cells and tumor cells (Cohen et al, 1996; Wei et al, 2003; Yao et al, 2006). Interleukin-6 receptor is expressed in human endothelial cells within normal ovary and carcinoma specimens (Nilsson et al, 2005). Although endothelial cell is differentiated from EPCs, they have different phenotype and functioning. Using immunofluorescent technique, we first demonstrated that IL-6 receptor also existed in EPCs. Although the main function of IL-6 receptor is related to focal inflammation, it could mediate angiogenesis. Our study in vitro indicates that IL-6 mediated angiogenesis partially by activating blood EPCs through IL-6 receptor.
In addition, when treating EPCs with different IL-6 concentrations, we found that phospho-ERK1/2 and STAT3 were significantly increased in a dose-dependent manner. It occurred as early as 5 or 10 mins after treatment and lasted for at least 60 mins. The results further showed that IL-6 receptor was functionally expressed in EPCs, and that IL-6 regulated EPCs’ biological behaviors through activation of both REK1/2 and STAT3 pathways. The signal pathways widely exist in many cell types including endothelial cells. Activation of ERK1/2 and STAT3 pathways plays a crucial role in endothelial cell proliferation, migration and microvascular tube formation (Bartoli et al, 2003; Deo et al, 2002; Yahata et al, 2003; Yao et al, 2006), and mediates pathogenesis of vasculization (Seino et al, 1994). Interestingly, ERK and STAT activation are significantly inhibited by IL-6 receptor blockade and only partially inhibited by the IL-6 neutralizing antibody. However, inhibiting either IL-6 or IL-6 receptor produces similar results in IL-6-induced proliferation, migration, and tube formation function in EPCs. This result suggests that IL-6-induced angiogenic effect has a high threshold, and that only a high level of IL-6 can induce angiogenic function.
Circulating EPCs can home to ischemic tissue and participate in activating vasculogenesis, thereby increasing blood flow and alleviating tissue injury (Asahara et al, 2000). Endothelial progenitor cells also participate in tumor neovascularization, either by incorporating into vascular endothelium of the tumor or by secreting proangiogenic growth factors (Davidoff et al, 2001; Hammerling and Ganss, 2006; Lyden et al, 2001). The mechanisms by which circulating EPCs home to the activating angiogenesis region are unknown; it might rely on the integrin α4β1 (VLA-4), which promotes the EPCs to home to the VLA-4 ligands vascular cell adhesion molecule-1 (VCAM) and cellular fibronectin (Jin et al, 2006). Growth factor and cytokines, such as VEGF, granulocyte-macrophage colony-stimulating factor, and stromal cell-derived factor-1 (SDF-1), can mediate EPC mobilization to peripheral circulation and homing to sites of activating angiogenesis (Moore et al, 2001; Takahashi et al, 1999). We demonstrated that IL-6 receptor existed in EPC. Therefore, focal increased IL-6, whether caused by inflammation or ischemia, can be an attractant to recruit EPC to this area and produce its function.
