Abstract
Endothelial progenitor cells (EPCs) are being investigated for advanced therapies, and matrix metalloproteinase 9 (MMP9) has an important role in stroke recovery. Our aim was to determine whether tissue MMP9 influences the EPC-induced angiogenesis after ischemia. Wild-type (WT) and MMP9-deficient mice (MMP9/KO) were subjected to cerebral ischemia and treated with vehicle or outgrowth EPCs. After 3 weeks, we observed an increase in the peri-infarct vessel density in WT animals but not in MMP9/KO mice; no differences were found in the vehicle-treated groups. Our data suggest that tissue MMP9 has a crucial role in EPC-induced vascular remodeling after stroke.
INTRODUCTION
Transplantation of endothelial progenitor cells (EPCs) has become a promising approach to enhance angio-vasculogenic responses after ischemia. EPCs can be mobilized in response to ischemia, can home to sites of neovascularization, and can differentiate into endothelial cells. 1 There are two subsets of EPCs: the so-called circulating angiogenic cells and the outgrowth endothelial cells (OECs). 2 Both types of EPCs have been demonstrated to increase brain angiogenesis in animal models of cerebral ischemia.3–5
The degradation and remodeling of the vascular basal membrane is required to allow endothelial cells to migrate, and matrix metalloproteinases (MMPs) have key roles in the initial steps of angio-vasculogenesis. Specifically, MMP9 has been shown to be essential for the invasion of endothelial cells and capillary branching, and
Our core hypothesis is that tissue MMP9 is a key protease required for an effective and successful cell-based therapy to potentiate vascular remodeling. In this study, we investigated the effects of brain MMP9 deficiency on the cerebral ischemia after EPC-based treatment to potentiate angio-vasculogenesis. To examine this hypothesis, we evaluated the infarct size in the acute phase as well as the long-term peri-infarct angiogenesis in wild-type (WT) and MMP9 knock-out (MMP9/KO) mice after EPC transplantation in a model of cortical stroke.
MATERIALS AND METHODS
Animals
A total of 50 male WT and MMP9/KO mice (8-to-12-weeks old, FVB background) were obtained from Jackson Laboratories (Sacramento, CA, USA). Both genotypes were bred in-house and the offspring were used for experimentation. All procedures were approved by the Ethics Committee of Animal Experimentation of the Vall d'Hebron Research Institute (58/08/10) and were conducted in accordance with the Spanish legislation and the Directives of the European Union. The ARRIVE guidelines were considered in the design and report of the study.
Permanent Focal Cerebral Ischemia
The left middle cerebral artery was permanently occluded (MCAO) by electrocauterization, affecting the cortex as described. 6
In Vivo Magnetic Resonance Imaging
Magnetic resonance studies were carried out 24 hours after MCAO using a 7-Tesla horizontal magnet (BioSpec 70/30, Bruker BioSpin, Rheinstetten, Germany). Sixteen coronal slices for T2-weighted fast spin-echo images (T2WI) and eight coronal slices for T2 map and diffusion tensor imaging were acquired. Maps of the trace of the diffusion tensor, also named the apparent diffusion coefficient (ADC), and the fractional anisotropy (FA) were derived using the Paravision software program, v5.0 (Bruker BioSpin). Regions of interest corresponding to the infarct area were traced on T2, ADC, and FA maps in all slices presenting infarcts and in matching contralateral hemispheres. The mean values are expressed as the ipsilateral/contralateral ratios.
Treatments
Growing OECs were obtained and expanded as previously described
6
from WT FVB mice. The cells were trypsinized, and 5 × 10
5
cells were prepared in 200 μL of endothelial basal medium (EBM, Lonza, Allendale, NJ, USA). Treatments were randomly administered intravenously (200 μL via retro-orbital sinus) at 30 to 32 hours postischemia to the following groups: WT mice receiving vehicle (EBM,
Measurement of the Infarct Lesion
The hyperintense area corresponding to the injured tissue and the total areas of both ipsilateral and contralateral hemispheres were traced manually on T2W images by investigators who were blinded to the genotype. The edema index and lesion volume were calculated as previously described. 4
Behavioral Testing
The corner test (used to assess sensorimotor asymmetries) and grip strength meter test (used to measure the maximum strength of the mouse forelimbs) were used to evaluate the functional outcomes. The tests were performed blindly before surgery and were repeated 1, 7, 14, and 21 days after MCAO as described elsewhere. 12
Evaluation of the Brain Vasculature
To assess the poststroke angio-vasculogenesis, mice were injected intravenously with 80 μg of Dylight 594-labeled tomato (
Doublecortin (DCX) and NeuN Immunohistochemistry
EPC-induced neurogenesis was assessed in contiguous coronal sections, which were immunostained with antibodies for mature neurons (NeuN; Millipore, Billerica, MA, USA) and neuroblasts (DCX; Abcam, Cambridge, UK). The presence of DCX-positive cells was analyzed in the dorsolateral and subventricular zones of the ipsilateral hemisphere, and mature neurons were measured in the peri-infarct cortical area (
Statistical Analysis
SPSS 15.0 software (IBM, Armonk, NY, USA) was used for the statistical analyses. All variables were normally distributed, and the data are expressed as the mean ± s.d. Statistical significance was assessed by Student's
After a pilot study including
RESULTS
MMP9 Deficiency Reduces the Cortical Ischemic Lesion The ischemic lesions were evaluated by magnetic resonance imaging 24 hours after permanent MCAO in 32 animals. MMP9 deficiency entailed significant neuroprotection, reducing the infarct volume by 25% in MMP9/KO animals (17 ± 1.5 mm3 in WT versus 12.8 ± 0.9 mm3 in MMP9/KO,
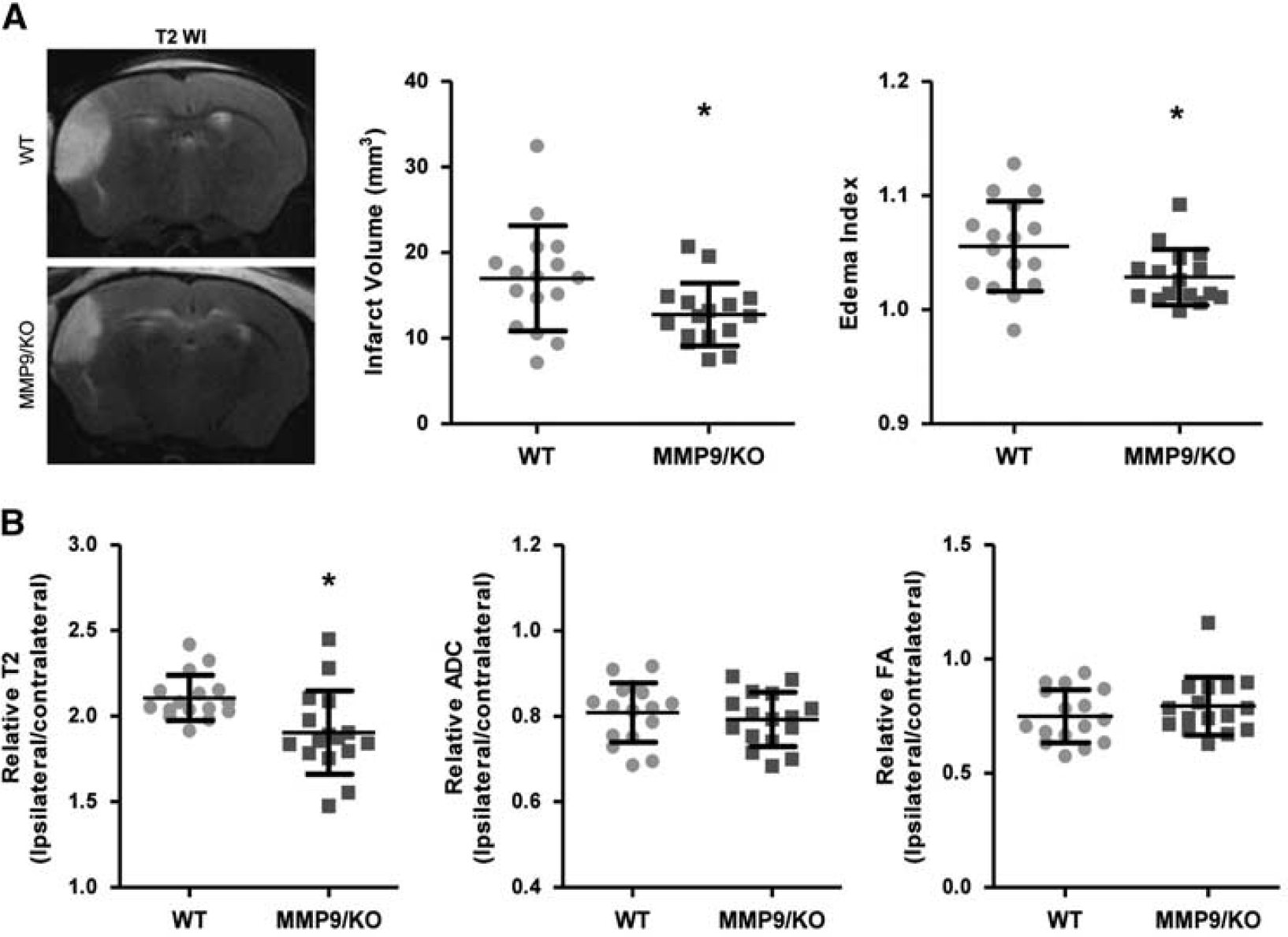
Magnetic resonance imaging 14 hours after cerebral ischemia. (
Neurorepair with OECs Enhanced the Cortical Angiogenesis Only in WT Mice
The cortical vessel density was analyzed 21 days after the induction of ischemia. One animal in the MMP9/KO vehicle group died 2 weeks after treatment after suffering severe aggression from the other mice housed in the same cage. Interestingly, no correlation existed between the vessel density and the magnetic resonance imaging parameters (infarct volume and ADC and FA values) in any of the groups. Green fluorescent cells were observed to be associated with the brain vessels in the peri-infarct areas in both WT and MMP9/KO mice receiving cell therapy (see Figure 2A), confirming the arrival of administered cells to the brain and its incorporation to the brain vasculature. When we compared the amount of functionally perfused microvessels in the peri-infarct area, there were no significant differences between the WT and MMP9/KO mice that received the vehicle (
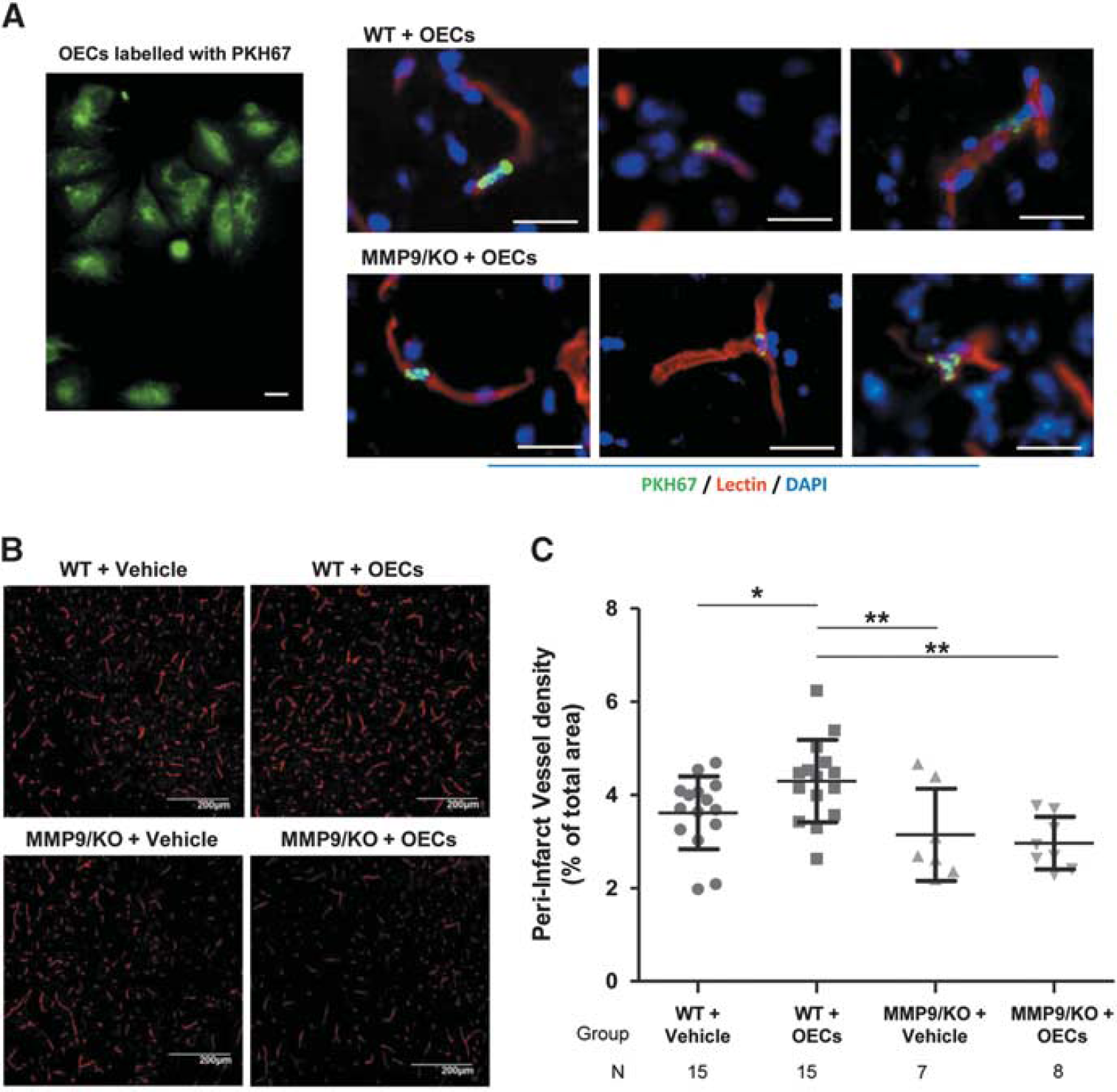
The brain peri-infarct vasculature in wild-type (WT) and matrix metalloproteinase-9 knockout (MMP9/KO) mice 21 days after cerebral ischemia. Functional blood vessels stained after lectin perfusion were quantified in the ipsilateral peri-infarct cortex 21 days after ischemia. (
Immunohistochemistry studies showed a non-significant decrease in DCX+ and NeuN+ cells in the MMP9/KO mice that received OECs compared with WT mice with the same treatment (0.51 ± 0.26% vs. 0.83 ± 0.42% for DCX+,
The corner test and the grip strength meter test failed to show any impairment after ischemia (pre- vs. 24-hour scores;
DISCUSSION
The current results suggest that MMP9 has a key role in the tissue vessel remodeling mediated by cell therapy with EPCs after cerebral ischemia. The upregulation of MMPs, including MMP9, after cerebral ischemia and the contribution of the MMPs to brain injury have been widely documented.7,8 Although pharmacological or immunological inhibition of MMP9 has proven to be neuroprotective, as evidenced by a decreased infarct size, discrepant results have been reported in MMP9-deficient mice.8,9,11 These discrepancies could be attributed to the use of different experimental models of cerebral ischemia (proximal and distal MCA occlusion in the presence or absence of reperfusion) and/or to the use of different techniques to assess the lesion extension (tissue staining for 2,3,5-triphenyl-tetrazolium-chloride). The present study includes the largest sample size of WT and MMP9/KO mice used to evaluate the infarct volume (
In addition to its detrimental effects in the acute phase, MMPs also participate in tissue remodeling after brain injury, and MMP9 has been suggested to participate in the unique tissue biobridge between neurogenic and non-neurogenic sites. 13 After a stroke, the pharmacological inhibition of MMPs with broad-spectrum drugs demonstrated that they have essential roles to ensure an effective neurorepair after cerebral ischemia and poststroke angiogenesis. 14 However, there have been no previous studies that have analyzed the specificity of MMP9 using genetically modified ischemic mice with poststroke EPC treatment to enhance angiogenesis.
We have recently described that there was an increase in the peri-infarct vessel density in WT ischemic mice compared with sham mice, which was not observed in ischemic MMP9/KO individuals. 6 In the present study, WT and MMP9/KO ischemic mice subjected to long-term brain ischemia showed a similar vessel density 21 days after cortical ischemia. The discrepancy between this finding and those of other studies analyzing the effects of MMP inhibitors in cortical remodeling after stroke 14 may be attributed to the small infarct volumes typical of the FVB strain in our model of cerebral ischemia, 12 the role of other MMPs, the longer time point for the tissue analysis in our study, or to differences in the neurorepair mechanisms in mice vs. rats. In this regard, a possible dual role of MMP9 in angiogenesis that depends on the endogenous tissue vascularization has been observed by other authors, as this protease was required for ischemia-induced angiogenesis after hindlimb ischemia, 15 whereas in cardiac tissue MMP9-deficiency facilitated angiogenesis after myocardial infarction. 16
The most important finding of our study, which we believe will be of great importance to the development of future cell-based neurorepair strategies after stroke, is the MMP9 tissue dependency after EPC therapy. To date, several different subsets of EPC populations have been demonstrated to have therapeutic potential in experimental models of cerebral ischemia.3–5 In the present study, we used OECs (a homogenous and highly proliferative cell population of endothelial-like cells) as a pro-angiogenic treatment for stroke, which was shown to be MMP9 dependent for tubulogenesis in an
In summary, MMP9 deficiency provides protection against the adverse effects of cortical cerebral ischemia in mice, although the protease is unequivocally required for the success of neurorepair therapies based on vascular remodeling, such as EPC transplantation.
Footnotes
AM, JM, and AR participated in the conception and design; AM, FM, and VB collected and assembled the data; AM, DG, JM, and AR analyzed and interpreted the data; and AM and AR wrote the paper. All authors corrected and approved the manuscript.
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
We are grateful to Silvia Lope-Piedrafita, from Servei RMN of Universitat Autònoma de Barcelona, for her technical assistance with the magnetic resonance imaging.
