Abstract
To explore the role of neutrophil-derived matrix metalloproteinases (MMPs) during angiogenesis in the brain, we hypothesized that transient neutrophil depletion attenuates the angiogenic response to focal hyperstimulation with vascular endothelial growth factor (VEGF). Brain focal angiogenesis was achieved using an adeno-associated virus delivered VEGF (AAV-VEGF) gene transfer in the mature mouse. Four groups of mice underwent AAV vector injection in the brain parenchyma: (1) AAV-LacZ; (2) AAV-VEGF; (3) AAV-VEGF plus anti-polymorphonuclear (PMN) antibody; and (4) AAV-VEGF plus serum. Animals in groups 3 and 4 underwent 4 days of PMN antibody or serum treatment before transfection; treatment was sustained for an additional 14 days. Anti-PMN treatment decreased circulating neutrophils to 9% of baseline (P < 0.001). Microvessels in the AAV-VEGF-group increased 25% compared with the AAV-lacZ-transduced group (256 ± 15 versus 208 ± 16; P < 0.05). Anti-PMN treatment attenuated the increase to 10% compared with control serum treatment (234 ± 16 versus 255 ± 22; P < 0.05). Similarly, compared with control serum treatment, anti-PMN treatment also reduced MMP-9 by 50% (2 ± 0.9 versus 4 ± 1.4; P < 0.05) and MPO expression by 25% (2 ± 0.8 versus 3 ± 0.9; P < 0.05); MMP-9 activity correlated with MPO expression (R2 = 0.8, P < 0.05). Our study demonstrated that transient depletion of neutrophils suppressed VEGF-induced angiogenesis, indicating that circulating neutrophils contribute to VEGF-induced focal angiogenesis. In addition, brain MMP-9 activity was attenuated after neutrophil depletion, suggesting that neutrophil is an important source of MMP-9.
Introduction
Recent studies have demonstrated that angiogenesis plays important roles in the tissue response to brain injury, including cerebral ischemia (Zhang et al, 2002), and may participate in the pathogenesis of other vascular disorders, such as brain arteriovenous malformations (Chen et al, 2006c). Vascular endothelial growth factor (VEGF) and matrix metalloproteinases (MMPs) are two major angiogenic factors in activating angiogenesis. Numerous studies have shown that VEGF hyperstimulation induces angiogenesis both in vivo and in vitro (Alitalo and Carmeliet, 2002; Chang and Werb, 2001; Fraser and Lunn, 2000; Hofman et al, 2001; Springer et al, 1998). Using adeno-associated viral vector-delivered human VEGF165 cDNA (AAV-VEGF), our previous study demonstrated that it induced reproducible focal angiogenesis in the mature mouse brain (Yang et al, 2003). This brain angiogenesis model provides a unique tool to study the mechanisms of angiogenesis in the brain.
Matrix metalloproteinases are a family of zincdependent endopeptidases that are responsible for the degradation of extracellular matrix component and are necessary during induction of angiogenesis models in vivo and in vitro (Haas and Madri, 1999). We have demonstrated previously that MMP-9 activity is greatly increased after VEGF transduction in the adult mouse brain, which is accompanied by increased angiogenesis. Further, MMP-9 inhibitor attenuated the VEGF-induced angiogenesis (Lee et al, 2004; Zhu et al, 2005). Recent studies show that neutrophils are important sites of MMP storage and activation (Opdenakker et al, 2001; Schwesinger et al, 2001). They may also contribute to MMP-9 expression in focal cerebral ischemia (Asahi et al, 2001; Planas et al, 2002; Romanic et al, 1998). Depletion of neutrophils reduced MMP-9 upregulation in middle cerebral artery occlusion/perfusion model (Justicia et al, 2003), reduced MMP-2 and MMP-9 upregulation in experimental hindlimb ischemia (Muhs et al, 2004), diminished fibroblast growth factor-2-induced angiogenesis in mouse cornea (Shaw et al, 2003), correlated with reduced choroidal neovascularization in adult mice (Zhou et al, 2005), and inhibited experimental abdominal aortic aneurysm formation (Eliason et al, 2005). Myeloperoxidase (MPO) expression was highly correlated with MMP-9 activity in brain arteriovenous malformation tissue (Chen et al, 2006b). These findings suggest that neutrophils associate with MMPs during activating angiogenesis.
Although angiogenesis has been extensively studied in vivo and in vitro, the extent to which circulating neutrophils contribute to activation of angiogenesis in the brain is not clear. We hypothesized that neutrophils play an important role in facilitating focal angiogenesis in the brain, and that they provide a major source of MMPs. To test this hypothesis, we investigated the effect of neutrophil depletion on MMP-2 and −9 changes and focal angiogenesis in the mouse brain after AAV-VEGF gene transfer.
Materials and methods
Experimental Groups
All experimental procedures for using laboratory animals were approved by the Institutional Animal Care and Use Committee of the University of California, San Francisco. Forty-eight adult C57BL/6 male mice (Charles River, Wilmington, MA, USA) weighing 25 to 30 g were used for the experiments. The mice were divided into four groups (n = 12 per group): (1) AAV-lacZ-transduced group; (2) AAV-VEGF-transduced group; (3) AAV-VEGF-transduced mice with rabbit anti-mouse PMN polyclonal antibody treatment; and (4) AAV-VEGF-transduced mice with normal rabbit serum treatment group. We chose injecting the same volume of normal rabbit serum at the same time point because it would exclude the effect of normal serum. Animals underwent AAV-VEGF (2 μL viral suspensions, containing 2 × 109 genome copies of virus) or AAV-lacZ transduction. We have established AAV-VEGF and AAV-lacZ construction (Su et al, 2002, 2000) and in vivo brain transduction in our laboratory, as described previously (Shen et al, 2006a, b; Yang et al, 2003).
Generation of Neutropenia
Neutrophils were depleted by intraperitoneal administration of 1 mL of saline-diluted (1:10) rabbit anti-mouse PMN polyclonal antibody (Accurate Chemical & Scientific, Westbury, NY, USA) daily for 4 days, which induced neutropenia in the mice on the day of the gene transfer (Figure 1). An additional dose was given every other day after AAV-VEGF injection to maintain neutropenia. A similar study demonstrated that anti-mouse PMN polyclonal antibody could effectively inhibit neutrophil for a period of time (Eliason et al, 2005). The control group received the same amount of normal rabbit serum (Accurate Chemical & Scientific, Westbury, NY, USA). Peripheral blood was collected from all mice before treatment, on the day of gene transfer, and thereafter every 2 to 4 days in EDTA-coated microtainer brand tube (Becton-Dickinson, Franklin Lakes, NJ, USA). Neutrophils were analyzed with a HEMAVET 950 multispecies hematology instrument (DREW Scientific Inc, Oxford, CT, USA).
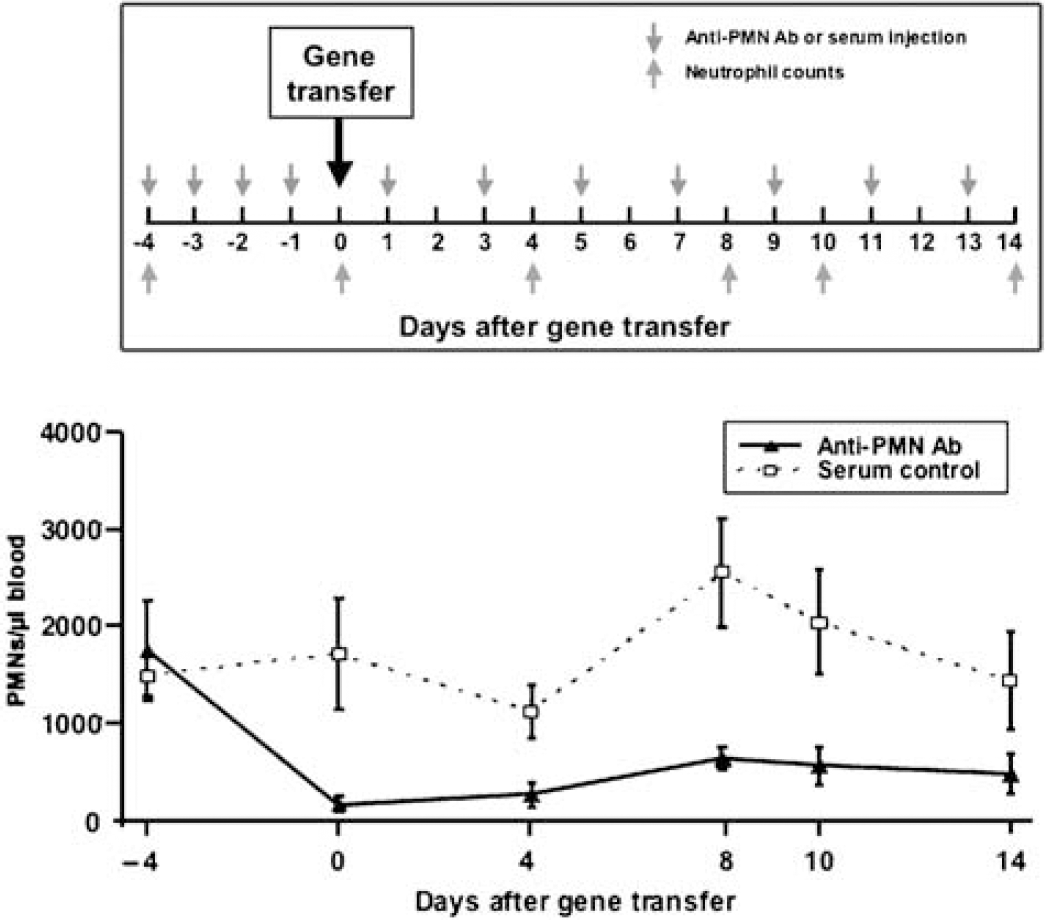
Experiment design. Upper panel: the time course of neutrophil depletion and neutrophil counts. Red arrows indicate the time points of anti-PMN antibody injection; purple arrows indicate the time points of neutrophil counts. Day 0 indicates AAV-VEGF injection. Bottom panel: line graph shows the average circulating neutrophils in mice treated with anti-PMN antibody versus normal serum control. Peripheral blood was collected every 2 to 4 days in an EDTA-coated microtainer brand tube and was analyzed with a HEMAVET 950 multi-species hematology instrument. Animals received 500 μg of intraperitoneal anti-PMN antibody or the same amount of normal serum injection daily for 4 days before AAV-VEGF gene transfer, and once every other day thereafter. Data are mean ± s.d. N = 6 in each group. P < 0.05, anti-PMN antibody-treated group versus normal serum control group.
AAV-VEGF and AAV-lacZ Gene Transfer in the Brain
The mice were anesthetized using ketamine/xylazine (100/10 mg/kg body weight) intraperitoneal before the operation. They were then placed in a stereotactic frame with a mouth holder (David Kopf Instruments, Tujunga, CA, USA). A burr hole was drilled in the pericranium 2 mm lateral to the sagittal suture and 1 mm posterior to the coronal suture. A 10 μL Hamilton syringe was inserted into the parenchyma approximately 3.0 mm under the cortex. Two microliters of viral suspension containing 2 × 109 genome copies of AAV-VEGF or AAV-lacZ were stereotactically injected into the left lateral ventricle and caudate putamen at a rate of 0.2 μL/min.
Lectin Staining and Microvessel Counts
The mice were reanesthetized and cardioventricularly perfused with phosphate-buffered saline at day 14 after gene transfer. Brains were removed and stored at −80°C. Coronal sections, from 1 mm anterior and 1 mm posterior to the needle track, were collected for protein extraction used for zymography and Western-blot analysis. Coronal cryostat sections of 20 μm were prepared for microvessel counts and immunostaining. Fluorescein-lycopersicin esculentum lectin (Vector Laboratories, Burlingame, CA, USA) staining was used to identify microvessels. Briefly, sections were fixed and incubated with lectin 2 μg/mL at 4°C overnight. Three adjacent areas of microvessels (left, right, and bottom within approximately ≈ 0.5 mm from the needle track) were chosen in two separate sections, and pictures were taken at low magnification (× 10). Microvessel numbers were counted in the images using NIH Image 1.63 software. The number of microvessels was calculated as the mean of the numbers obtained from the six images, as described previously (Xu et al, 2004).
Gelatin Zymography
Mice were killed 2 weeks after AAV-VEGF gene transfer. The brain tissue of the portion of caudate putaman of the viral transduction side was collected and homogenized on ice. After being centrifuged at 13,000 r.p.m. for 30 mins, the supernatants were collected and analyzed by gelatin zymography to characterize gelatinase activity. After electrophoresis, gels were washed and incubated overnight at 37°C in a developing buffer. The gels were stained with Coomassie Blue R-250 (Bio-Rad Laboratories, Hercules, CA, USA). Standard recombinant MMP-9 and MMP-2 (Chemicon, Temecula, CA, USA) were used on the gels for identification. Gelatinolytic bands were quantified by scanning densitometry and analyzed using NIH Image 1.63 software.
Western-blot Analysis
Aliquots from the tissue samples used for gelatin zymograpy were used for Western-blot analysis. Proteins in the gel were electrotransferred onto a nitrocellulose membrane (Bio-Rad Laboratories) in transfer buffer (Invitrogen, Inc., Carlsbad, CA, USA) by semidry blotting. After blocking in 5% milk, the membrane was immunoprobed with MPO (1:4000 dilution, Upstate, Chicago, IL, USA) overnight at 4°C. After washing, the membrane was incubated with horseradish peroxidase conjugated anti-rabbit secondary antibody (Amersham, Buckinghamshire, UK) diluted 1:10,000, and then reacted with FEMO detection reagent (Pierce, Rockford, IL, USA).
Immunohistochemistry
Two weeks after gene transfer, the mouse brains were collected and coronal sections (20 μm) were cut, air-dried, and fixed with cold acetone. After blocking endogenous peroxidase with 0.3% hydrogen peroxide in 100% methanol, incubation followed with 5% normal goat serum to block nonspecific binding. The sections were incubated with anti-MPO (Lab vision, Fremont, CA, USA) or anti-MMP-9 (a gift from Dr Zena Werb, UCSF) polyclonal antibodies overnight at 4°C, and then with biotinylated anti-rabbit IgG for 1 h and with avidin—biotin enzyme reagent (ABC kit, Vector Laboratories) for 30 mins. Chromogenic staining was developed using DAB kit (Vector Laboratories) for MPO and VectorRed (Vector Laboratories) for MMP-9, followed by counter staining with hematoxylin.
Statistical Analysis
Parametric data in different groups were compared using a one-way analysis of variance, followed by Fisher's protected least significant difference test. All data are presented as mean ± standard deviation. A probability value of < 5% was considered to be statistically significant.
Results
Anti-Polymorphonuclear Antibody Results in Systemic Neutropenia
Injections of anti-PMN antibody significantly diminished the number of circulating neutrophils compared with the control rabbit serum-treated animals. The number of neutrophils in the anti-PMN-treated mice decreased after treatment with 1750 ± 520 to 160 ± 80 neutrophils/μL blood (P < 0.001) before VEGF hyperstimulation, representing a 91% decrease in the absolute neutrophil number (Figure 1). The anti-PMN-treated group remained neutropenic during the course of the experiment. This result indicates that anti-PMN antibody can efficiently inhibit circulating PMN, which provides a reproducible systemic neutropenia model for the study of neutrophil function. At day 8, there was an increase in blood neutrophils compared with day 0 in the serum control group (P = 0.04), which could have been the result of VEGF inducing mobilization of bone marrow cells into the bloodstream, thus reflecting a significant increase in white blood cell counts (Grunewald et al, 2006; Hattori et al, 2001).
VEGF-Induced Angiogenesis is Suppressed by Neutrophil Depletion
We analyzed the effect of neutropenia on microvessel counts 2 weeks after AAV-VEGF gene transfer (Figure 2A). The number of microvessels in the AAV-VEGF-transduced mice increased compared with the AAV-lacZ-transduced group (Figure 2B, 256 ± 15 versus 208 ± 16 microvessel/field, P < 0.05). It was interesting to note that microvessels did not increase after neutrophil depletion compared with the rabbit serum-treated control animals (Figure 2B, 234 ± 16 versus 255 ± 22 microvessel/field, P < 0.05).
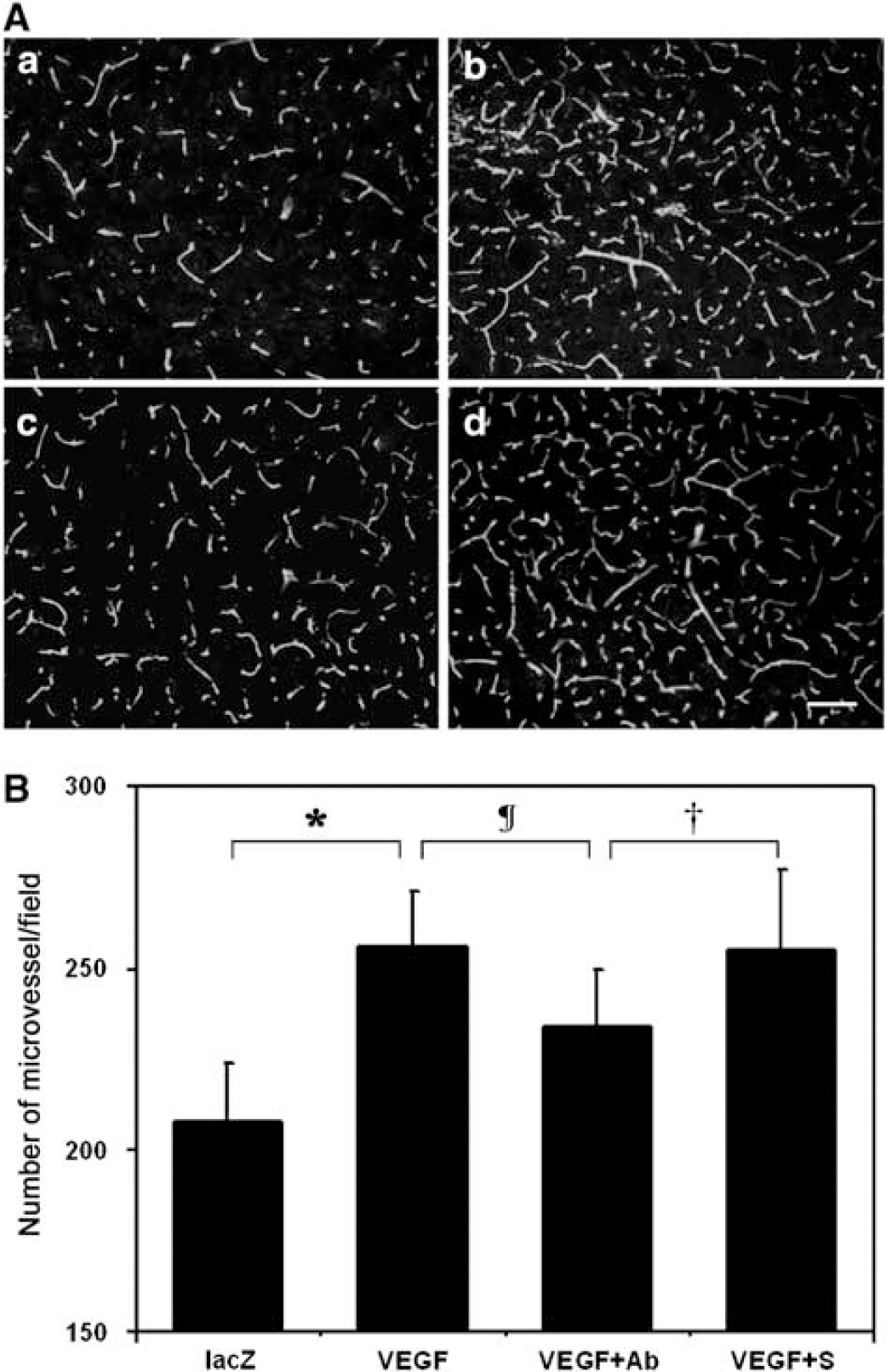
Effect of neutropenia on microvessel counts in the AAV-VEGF-transduced mouse brain. (
MPO is Attenuated in the Neutropenic Mice
Western-blot analysis showed that MPO expression increased in the AAV-VEGF-transduced mice compared with the AAV-lacZ-transduced mice (Figure 3A, 3 ± 0.9 versus 1 ± 0.4, P < 0.05). MPO expression in the anti-PMN-treated mice decreased compared with the serum-treated group (Figure 3A, 2 ± 0.8 versus 3 ± 0.9, P < 0.05). Immunostaining showed that MPO-positive staining was mostly located in the adjacent area of the needle track in the AAV-VEGF-transduced mice (Figure 3B-b). Parallel to the Western-blot results, MPO staining was reduced after anti-PMN treatment (Figure 3B-c), but not after control serum treatment (Figure 3B-d). There were no MPO-positive cells in the contralateral hemisphere (data not shown).
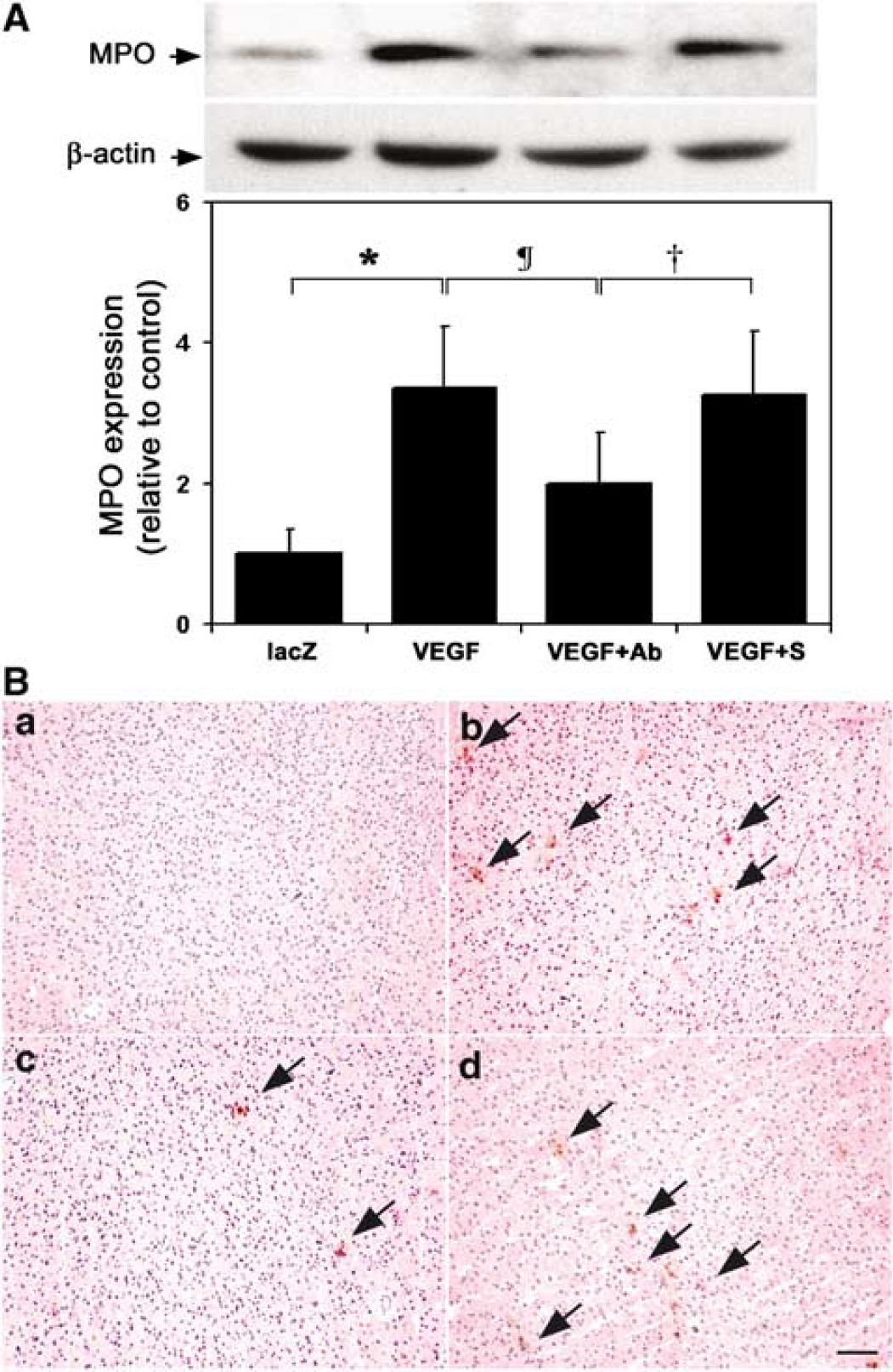
Effect of neutropenia on MPO expression in AAV-VEGF-transduced mice. (
Angiogenesis Suppression is Associated with Decreased Matrix Metalloproteinase-9 but not Matrix Metalloproteinase-2
Zymography showed that MMP-9 activity is highly upregulated in the AAV-VEGF-transduced mice compared with the AAV-lacZ-transduced mice (Figure 4, 4 ± 1.1 versus 1 ± 0.3, P < 0.05). Anti-PMN treatment greatly reduced the increased MMP-9 activity compared with the rabbit serum treatment (2 ± 0.9 versus 4 ± 1.4, P < 0.05). Unlike MMP-9, there was no statistical difference in MMP-2 activity in the different treatment groups (Figure 4C, P > 0.05).
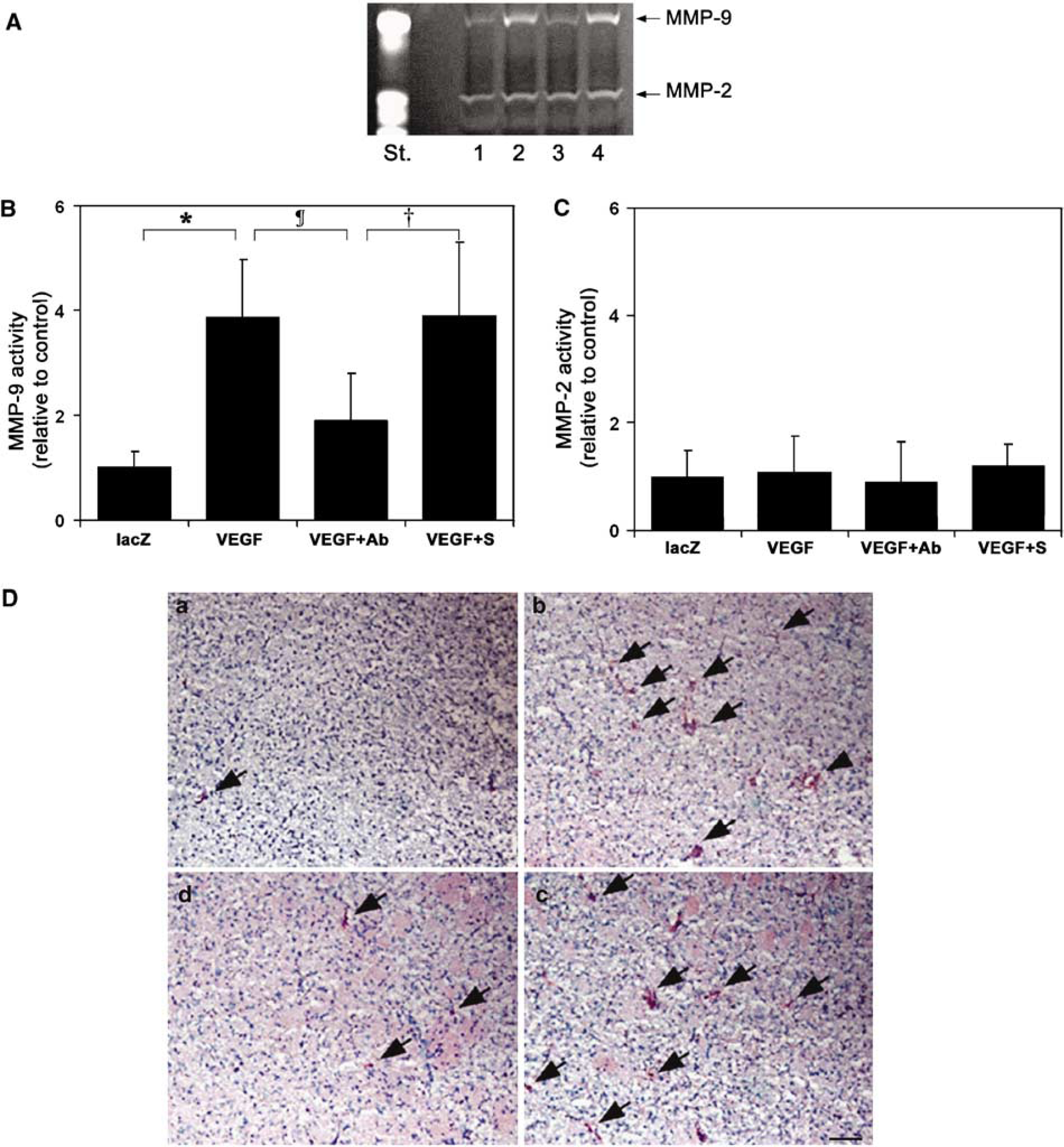
Effect of neutropenia on MMP-9 and MMP-2 activity in AAV-VEGF-transduced mice. (
Cellular MMP-9 distribution was detected by immunohistochemistry. Few MMP-9-positive cells were detected in the AAV-lacZ-transduced mouse brain (Figure 4D-a). The MMP-9-positive cells were greatly increased in the AAV-VEGF-injected hemisphere of the brain (Figure 4D-b). We demonstrated that MMP-9-positive staining was greatly reduced after the depletion of neutrophils (Figure 4D-c), but there was no change in MMP-9 expression in the serum-treated control animals (Figure 4D-d). There were no MMP-9-positive cells in the contralateral hemisphere (data not shown).
Active Matrix Metalloproteinase-9 Increase is Associated with Brain Tissue MPO Level in the Brain
Double-labeled immunostaining identified infiltrated neutrophils appearing in MMP-9-positive staining in the activating angiogenesis area in the AAV-VEGF-treated mouse brain (Figure 5A). A linear regression across all groups showed that semiquantitative counting of increased MMP-9 activity was closely correlated to the increase of MPO in the brain tissue (Figure 5B, R2 = 0.8, P < 0.05).
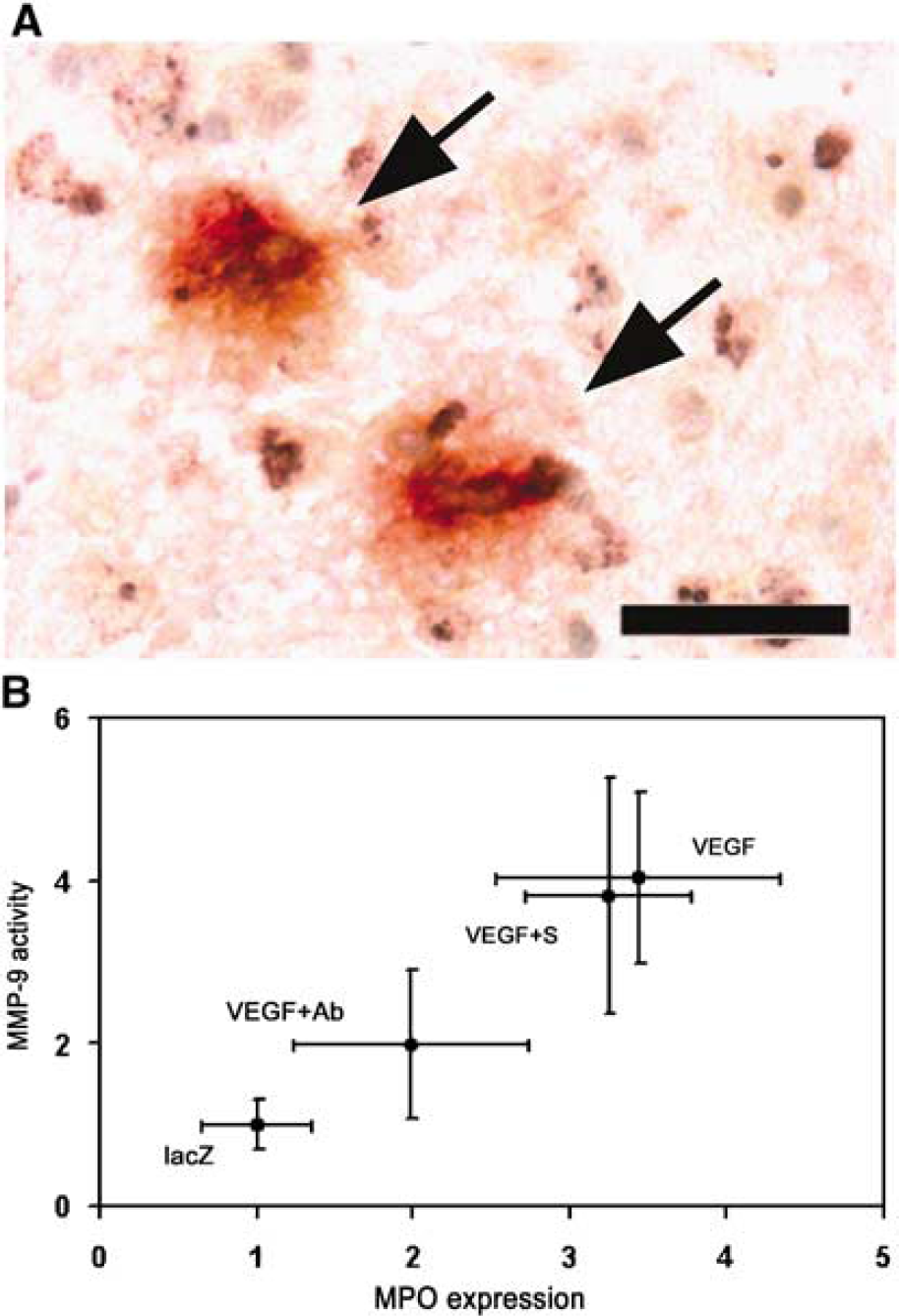
Relationship between MPO expression and MMP-9 activity. (
Discussion
In this study, using a well-established VEGF hyperstimulation focal angiogenesis model in the mouse brain (Chen et al, 2006a; Yang et al, 2003), we demonstrated for the first time that (1) neutrophil depletion attenuated AAV-VEGF-induced brain angiogenesis; (2) MMP-9 activity, but not MMP-2, increased in the AAV-VEGF-transduced mice, and that neutrophil depletion downregulated the increase of MMP-9; (3) MPO expression increased in the AAV-VEGF-transduced mice—this MPO increase was attenuated after neutrophil depletion; and (4) increased MMP-9 activity was closely associated with elevated MPO expression. Our results suggest that circulating neutrophils promote VEGF-induced focal angiogenesis in the mouse brain through upregulation of MMP-9 activity, and that they may be an important source of MMP-9 in the angiogenesis region.
Studies have shown that MMP-9 plays a crucial role in vascular remodeling (Bergers et al, 2000; Haas and Madri, 1999; Itoh et al, 1998). We have also reported that MMP-9 activity is significantly elevated after VEGF transduction in the adult mouse brain, which is accompanied by increased angiogenesis (Lee et al, 2004; Zhu et al, 2005). In addition, MMP-9 inhibitor was found to reduce potently VEGF-induced angiogenesis (Lee et al, 2004).
Our previous studies demonstrated that AAV-VEGF could greatly increase VEGF protein level in the transduced brain tissue and active focal angiogenesis (Shen et al, 2006a, b). In this study, we used this well-established brain focal angiogenesis model to explore the function of neutrophils during AAV-VEGF-induced angiogenesis. We found that many MPO staining cells were detected surrounding the needle track in AAV-VEGF transduced mice, indicating that neutrophils could infiltrate into the mouse brain parenchyma. MPO protein level and MPO positive staining were reduced in the same angiogenesis region after the depletion of neutrophils, suggesting that VEGF overexpression could transiently recruit circulating neutrophils into the brain tissue where angiogenesis was actively developed.
We found that neutrophil depletion attenuated VEGF-induced focal angiogenesis in the brain, providing additional evidence that neutrophils participate in activating angiogenesis in the mature mouse brain. The function of neutrophils during focal brain angiogenesis, however, is unclear. Neutrophils have been recognized as an important site for storing and activating MMPs. (Opdenakker et al, 2001; Schwesinger et al, 2001) Circulating neutrophils are also responsible for upregulating MMP-9 (Asahi et al, 2001; Muhs et al, 2004; Planas et al, 2002). A study by Schwesinger et al (2001) has demonstrated that neutrophils could secrete MMP-9. Our results showed that increased MMP-9 activity after AAV-VEGF transduction was attenuated by the depletion of neutrophils. Double-labeled immunostaining showed that MPO and MMP-9 were colocalized, and furthermore, correlation analysis confirmed that increased MMP-9 activity is closely related to the elevated expression of MPO. The present data are consistent with our previous report that MMP-9 activity highly correlates with MPO expression in brain AVM (arteriovenous malformation) tissues (Chen et al, 2006b). All of these data indicate that neutrophils are responsible for a major portion of increased MMP-9 activity after VEGF hyperstimulation, and that infiltrated neutrophils may be the source of the MMP-9. In addition to VEGF and MMP-9, white blood cells, including neutrophils, may be a rich source of several other angiogenic factors, such as MMP-12, IL-8, and basic fibroblast growth factor, which may also be involved in angiogenesis.
Neutrophils are the most abundant leukocyte in the blood circulation that can rapidly infiltrate an activating site of angiogenesis. Administration of anti-PMN antibody significantly inhibited neutrophil function during angiogenesis. Involvement of other circulating cells, such as monocytes or lymphocytes, after neutrophil depletion is unclear. Macrophages have been reported to be a source of angiogenic growth factors. However, we did not investigate their role in our experimental model, but they are an important consideration for future studies.
VEFG hyperstimulation induces angiogenesis, which is accompanied by neutrophil recruitment. Before infiltration, circulating neutrophils need to adhere to the vascular wall in a process mediated by adhesion molecules. Vascular endothelial growth factor has been reported to enhance ICAM-1 (intercellular adhesion molecule-1) expression and neutrophil recruitment under inflammatory conditions (Goebel et al, 2006). However, VEGF, in contrast to basic fibroblast growth factor, does not increase the chemotactic activity for neutrophil recruitment and ICAM-1 expression during acute inflammation (Zittermann and Issekutz, 2006). This may be because of different experimental models. Recent studies show that focal increased VEGF appears to be one molecule attracting bone marrow-derived circulating cells to the place where VEGF is produced through SDF-1/CXCR-4 signaling pathway (Grunewald et al, 2006). In our experiment, we found that circulating neutrophils move toward the place where VEGF is overexpressed, suggesting that VEGF plays an important role in the recruitment of circulating blood cells. Infiltrated neutrophils may release MMP-9 in the activating angiogenesis region. MMP-9 then causes brain—blood barrier disruption and facilitates tissue-bound VEGF (Bergers et al, 2000; Engsig et al, 2000), which in turn facilitates circulating neutrophils to move into and infiltrate the brain parenchyma and promote focal angiogenesis. How neutrophils are recruited into the activating angiogenesis region of the mature mouse brain in our experiment model needs to be studied further.
Unlike MMP-9, there was no change in MMP-2 activity after VEGF transduction or after neutrophil depletion. This suggests that MMP-2 was not involved in angiogenesis in this VEGF transduction model, which is consistent with our previous study (Lee et al, 2004). Because MMP-8 is a neutrophil collagenase, examining the effect of MMP-8 during brain angiogenesis needs to be considered in future studies.
For some disease processes such as cerebral ischemia, early neutrophil infiltration may induce focal inflammatory response, which may damage surrounding brain tissue. VEGF hyperstimulation may disrupt blood—brain barrier and induce focal brain edema. These negative effects should be considered when stimulated angiogenesis is utilized as a therapeutic approach.
In summary, neutrophil infiltration was part of the inflammatory responses during AAV-VEGF-induced angiogenesis. Neutrophil depletion correlated with reduced microvessel counts and decreased MMP-9 protein activity, suggesting that neutrophils promoted the early development of focal angiogenesis partially through secretion of MMPs. This effect was dependent on neutrophils.
