Abstract
Vascular endothelial growth factor (VEGF) induces increased vessel permeability and formation of abnormal vessels. To investigate cerebral blood flow (CBF) during local overexpression of VEGF recombinant adenoviruses carrying the human VEGF165 complementary DNA (2.3 to 23 · 108 pfu/mL) were injected stereotactically into the caudate nucleus of anesthetized rats. Saline and adenoviruses carrying the β-galactosidase gene served as controls. Eleven days later (1) size and density of vessels were assessed in hematoxylin–eosin–stained sections, (2) vascular permeability was measured by intravenous Evans blue injections, and (3) local CBF (lCBF) was quantified using the iodo-[14C]antipyrine technique. Dose-dependent increases were found in (1) vessel density and size (only vessels >43 μm could be quantified morphologically), (2) Evans blue extravasation and brain edema formation, and (3) lCBF (up to eightfold). At medium doses, hyperemic areas and smaller areas of decreased lCBF were found. In low flow areas, vascular cross-sectional areas were increased 223-fold and vessel density up to 10-fold. In high flow areas, these parameters were increased 32-fold and up to 15-fold, respectively. Adenovirus mediated VEGF overexpression results in (1) increased vessel size and density, (2) areas of increased and of decreased flow, and (3) more and smaller vessels in high flow than in low flow areas. These results indicate a diverging flow pattern of newly formed vessels.
Positron emission tomography and functional magnetic resonance imaging studies have shown an increased anaerobic glycolysis caused by hypoxia in brain tissue surrounding cerebral infarcts (Weiller et al., 1993; Cramer et al., 1997). Thus, neoangiogenesis in the penumbra could improve the functional outcome after stroke. This idea is supported by postmortem analysis of brains of stroke patients indicating that survival is correlated with the density of newly formed blood vessels in the penumbra (Krupinski et al., 1994). In animal experiments, angiogenesis could also be demonstrated in the area surrounding the infarct a few days after focal cerebral ischemia (Marti et al., 2000). The effectiveness of this physiologic reaction could be enhanced by therapeutic induction of angiogenesis, which might be especially beneficial in patients with a diminished hemodynamic reserve and consequential lower penumbral blood flow (Friberg and Olsen, 1991; Witt et al., 1994).
Angiogenesis has been induced experimentally in different types of tissue, including the brain, by the application of high doses of the most potent angiogenic factor, vascular endothelial growth factor (VEGF). In these studies, the formation of abnormal, angioma-like new vessels has consistently been shown (Rosenstein et al., 1998; Lee et al., 2000; Pettersson et al., 2000). Such vessel malformations might result in spontaneous hemorrhage (Zhang et al., 2000). A second effect of experimental induction of angiogenesis by VEGF is an increased permeability of the blood–brain barrier (Zhang et al., 2000) resulting in brain edema. Because the formation of normal vessels seems to require a fine tuning of the expression of different pro- and antiangiogenic factors (Yancopoulos et al., 2000), it is not surprising that the exclusive overexpression of VEGF results in the formation of morphologically abnormal and leaky vessels (Carmeliet, 2000b; Carmeliet and Jain, 2000; Lee et al., 2000). A pathophysiologic example of such an effect is tumor angiogenesis (Nagy et al., 1995). Whereas the effects of an overexpression of VEGF on the formation of pathologic vessels are known, the functional consequences for tissue blood flow have not been investigated. Specifically, in the brain the relation between local vessel density and size and local blood flow after VEGF transfection is not known. Therefore, it was the aim of the present study to define the dose–response relation between the applied concentration of adenoviruses carrying the human VEGF165 complementary DNA (cDNA) and cerebral blood flow (CBF) and to relate the resulting vascular density and size of the newly formed vessels to local blood flow values. To this end, recombinant adenovirus particles containing the human VEGF165 cDNA were stereotactically injected into the caudate nucleus of rats. Although VEGF protein could be applied directly to the brain tissue (Kawakami et al., 1995; Tenjin et al., 1995), the transfection with the cDNA encoding VEGF (Muhlhauser et al., 1995; Isner et al., 1996a, b ; Byun et al., 2001) seems to be more effective when a single injection is performed because the local protein concentration is increased for a longer period. Adenovirus particles have a low target cell specificity (Rosenfeld et al., 1991; Akli et al., 1993; Engelhardt et al., 1993) and infect all cells near the injection site. Therefore, an adenovirus vector has been chosen for the present study. Eleven days after the intracerebral injections, vessel density, cross-sectional areas, and permeability were investigated and related to the local blood flow at the same tissue sites.
MATERIALS AND METHODS
Preparation of the adenovirus vector
The replication-deficient adenovirus Ad d1324, the packaging human embryonic kidney cell line 293 as well as the adenovirus carrying the β-galactosidase cDNA [previously subcloned into the adenovirus expression vector pAC.CMV.plpA(+)] were provided by Dr. J. Kreuzer, Heidelberg. The cDNA for human VEGF165 (a generous gift from Dr. G. Breier, Max-Planck Institute for Physiological and Clinical Research, Bad Nauheim, Germany) was subcloned into the adenovirus expression vector pAC.CMV.plpA(+). The linearized DNAs of the expression vector and the virus were cotransfected using a commercial transfection reagent (SuperFect; Qiagen, Hilden, Germany) according to the manufacturer's instructions at a ratio of 1:1 onto a 50% confluent monolayer of 293 packaging cells. After 2 weeks, the viral cytopathic effect was clearly visible. Chinese hamster ovary cells and human umbilical cord vein endothelial cells infected with recombinant virus, but not with a control virus, secreted copious amounts of VEGF into the medium (Quantikine-ELISA; R & D Systems, Wiesbaden, Germany). Having established the successful recombination of DNA in the virus, several monolayers of 293 cells were infected and the media were collected on maximum cytopathic effect. The 293 cells were lysed by freeze/thawing. The virus was enriched and purified using CsCl2 gradient centrifugation followed by dialysis. The virus stocks were stored in aliquots in liquid nitrogen after estimating the concentration of infectious particles by measuring the DNA concentration using ultraviolet spectrophotometry.
In vivo experiments
The experiments were performed in male Wistar rats in accordance with institutional guidelines. The animals were anesthetized using a gas mixture containing 1% to 1.5% halothane, 70% N20, and the remainder O2. Body temperature was maintained at 37°C using a temperature-controlled heating pad. The animals were placed in a stereotactic frame (custom made). After exposure of the skull, a burr hole (diameter 1 mm) was made in the angle between the coronary suture and the temporal crest. The virus suspension was injected into the caudate nucleus using a steel needle (diameter 0.8 mm) that was forwarded through the burr hole 5 mm deep into the brain under an angle of 20° to the sagittal plane. Using an infusion pump (Harvard, Holliston, MA, U.S.A.), 45 μL of the virus suspension was infused at a rate of 0.5 μL per minute. In rats treated with adenoviral particles containing the β-galactosidase gene, this slow infusion rate resulted in spheric volumes of transfected tissue of 52 ± 8 μL, which refers to a diameter of 4 to 5 mm (data not shown). Thereafter, the needle was withdrawn, the skin was sutured, and the animals were placed back in their cages to allow them to recover from the anesthesia. Then they were housed in the central animal facility of the University of Heidelberg under standard conditions for 11 days.
For each experiment, a fresh aliquot of virus solution (2.3 · 1010 plaque-forming units (pfu)/mL) was thawed on ice, gently mixed, and diluted with saline before the injection into the caudate nucleus. Seven different dilutions of the virus stock solution with saline were used (1:10, 1:20, 1:30, 1:40, 1:60, 1:80, and 1:100). In an additional group, the pure virus solution was tested. Saline and a suspension of an adenovirus containing the cDNA for β-galactosidase (1.9 · 1010 pfu/mL) served as controls. Each experimental group consisted of three to four rats.
Because in preliminary studies the expression peak using the adenovirus containing the cDNA for β-galactosidase with the same promotor was found 3 d after transfection and angiogenesis might take approximately 7 to 10 d (Pettersson et al., 2000), all rats were reanesthetized 11 d after the injections using the aforementioned gas mixture. Catheters were placed into the right femoral artery and vein to allow the infusion of iodo[14C]antipyrine (Biotrend, Cologne, Germany) for the measurement of the local CBF, the injection of Evans blue (Sigma, Deisenhofen, Germany), the measurement of the arterial input function of iodo[14C]antipyrine, and the measurement of blood acid base status. After surgery, the anesthesia was changed to intravenous etomidate (Etomodat Lipuro; Braun Melsungen, Melsungen, Germany) at a dose of 0.025 mg/kg body weight/min to minimize the effects of anesthesia on CBF (Janssen et al., 1975; Famewo and Odugbesan, 1978). One hour later, Evans blue (4% in saline, 2.5 mL/kg) was injected intravenously and was allowed to circulate for 10 minutes. Thereafter, CBF was measured according to the autoradiographic method of Sakurada et al. (1978). To this end, 125 μCi per kg body weight iodo[14C]antipyrine (Biotrend) dissolved in 1,000 μL saline were infused at an increasing infusion rate for 1 minute. Parallel to this, 12 to 16 timed arterial blood samples were taken for the determination of the time course of the arterial iodo[14C]antipyrine concentration. At the end of the infusion period, the animals were decapitated, and the brains removed as quickly as possible and frozen in 2-methylbutane chilled to −60°C. Then the brains were embedded in M-1 embedding matrix (Lipshaw, Detroit, MI, U.S.A.) and cut into 20-μm coronal sections at −20°C in a cryomicrotome. After drying them on a heating plate at +60°C, the sections were exposed together with a [14C] standard set on a Kodak MinR1 x-ray film for 21 d. From the optical densities of the autoradiograms, the local CBF was calculated using an image analyzing system (MCID; Imaging Research Inc., St. Catherines, Ontario, Canada). To quantify the changes of local CBF, the contralateral, untreated side was used as internal standard. On the autoradiograms in the treated hemisphere, the pixels were defined in which the CBF was higher compared with the same anatomic landmarks of the contralateral side. Then the volume and the average CBF of this brain tissue were determined. Thereafter, the volume of lowered CBF and the average CBF within this volume was determined in a similar way. Then the average CBF and size of each hemisphere were determined. The latter data were used to calculate an edema index (volume of treated side/volume of untreated side).
Sections adjacent to those used for autoradiography were used for histology. One set was used for hematoxylin–eosin staining. Because measurements of CBF using the iodo[14C]antipyrine technique were performed, it was impossible to perfuse or fixate the animals before decapitation. To get rid of blood inside the vessel lumina, the unfixed cryosections had been immersed in phosphate-buffered saline (two times for 5 minutes) before fixation of the section in 4% paraformaldehyde and consecutive hematoxylin–eosin staining. The blood was washed away using this procedure, whereas the tissue remained on the slide. For the measurements of the cross-sectional areas, six to eight sections of each animal were analyzed at a 60-fold magnification. Another set of cryosections was used for the investigation of the permeability of the newly formed vessels. After taking photographs for assessment of the Evans blue extravasation on the same sections, indirect immunofluorescence against fibronectin was performed (Gobel et al., 1990).
Statistics
All data measured in the different experimental groups were compared with both control groups using a two-tailed t-test with Bonferroni correction. Significance was assumed when the experimental group was different from both control groups. In this case, the lower level of significance is indicated in the tables and figures.
RESULTS
Table 1 shows the physiologic variables measured in the different experimental groups to evaluate different conditions of the experimental animals. The differences of physiologic parameters between the groups are minimal and significant differences are indicated in the table. No literature is available showing an influence of the differences in body weight on the parameters measured in the present study.
Physiologic variables of the different experimental groups
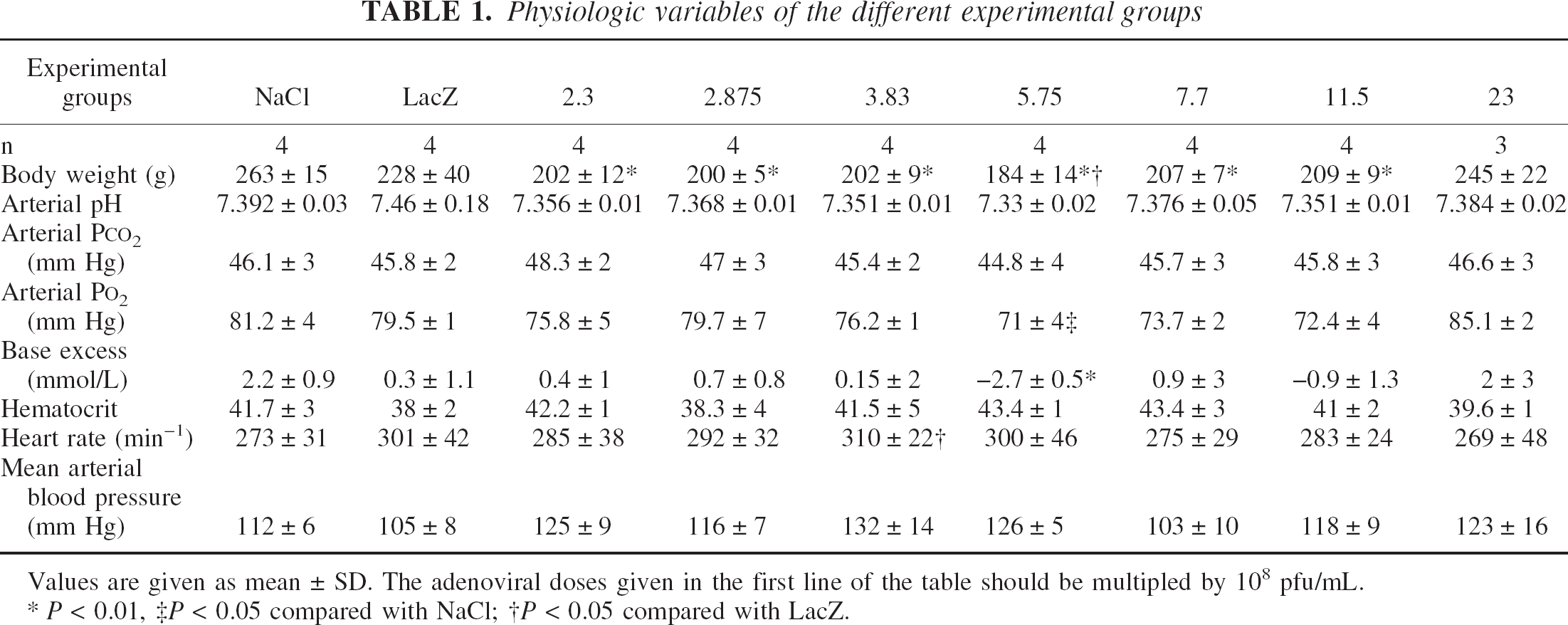
Values are given as mean ± SD. The adenoviral doses given in the first line of the table should be multipled by 108 pfu/mL.
P < 0.01
P < 0.05 compared with NaCl
P < 0.05 compared with LacZ.
Figure 1 shows the changes in volume of the injected hemisphere compared with the contralateral (control) side. All rats that received the undiluted virus solution (2.3 · 1010 pfu/mL) died, whereas all other animals showed no signs of discomfort or neurologic disorders. In both control groups and in animals infected with adenoviral doses up to 7.7 · 108 pfu/mL, no brain edema was detected. In contrast, at higher doses the volume of the treated hemisphere was increased by up to 30% compared with the contralateral side. As shown in Fig. 2, this effect on the development of brain edema was paralleled by a local Evans blue extravasation indicating an increased permeability of the blood–brain barrier that did not exist in rats treated by local injection of NaCl or virus carrying the cDNA for β-galactosidase. In addition, an increased tissue immunoreactivity of fibronectin could be found that, in contrast to normal conditions, was not associated with blood vessels (Fig. 2). Both effects of VEGF, Evans blue extravasation, as well as the increased fibronectin immunoreactivity made it impossible to determine the density of newly formed capillaries in the transfected brain area. Therefore, only the formation of larger vessels could be quantified. “Larger” was defined as vessels with a cross-sectional area >1,500 μm2, which corresponds to a diameter of about 43 μm. Within normal parenchyma, vessels of this size were found only occasionally. Figure 3A shows the hematoxylin–eosin staining of large vessels at the highest survival dose compared with a saline-treated control rat. Figure 3B shows the densities and cross-sectional areas of newly formed large vessels exceeding a cross-sectional area of 1,500 μm2. Density and cross-sectional area of such vessels increased significantly with increasing dose. At the highest survival dose, up to 11 new large vessels per square millimeter could be found (mean: 7/mm2) in the transfected hemisphere; the maximal cross-sectional area of these vessels was up to 2.3 mm2 (mean: 1.2 mm2) (Fig. 3B). Extremely large vessels, however, were rare. In all experimental groups, the cross-sectional area of more than half (at least 56%) of the newly formed vessels was smaller than 6,000 μm2. This cross-sectional area corresponds to a diameter less than 87 μm.
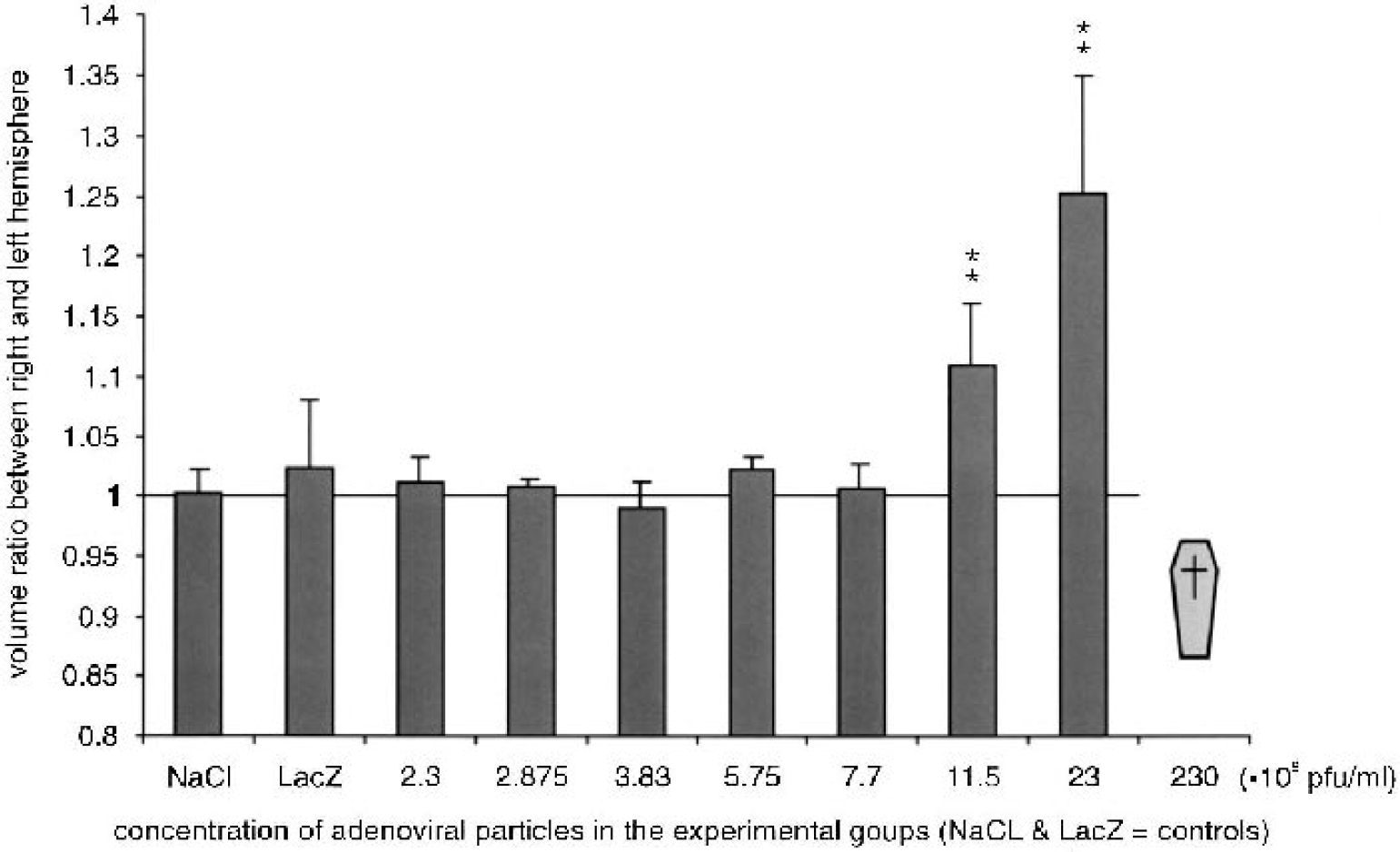
Occurrence of brain edema in response to vascular endothelial growth factor transfection in the experimental groups. An edema index (volume of transfected hemisphere/volume of contralateral side) was calculated for each dose of viral particles. The increase in tissue volume was significant at doses of 11.5 and 23 · 108 pfu/mL. Extrapolating from these data to the highest dose applied (230 · 108 pfu/mL) suggests that the death of all rats at this dose was caused by severe brain edema. **P < 0.01.
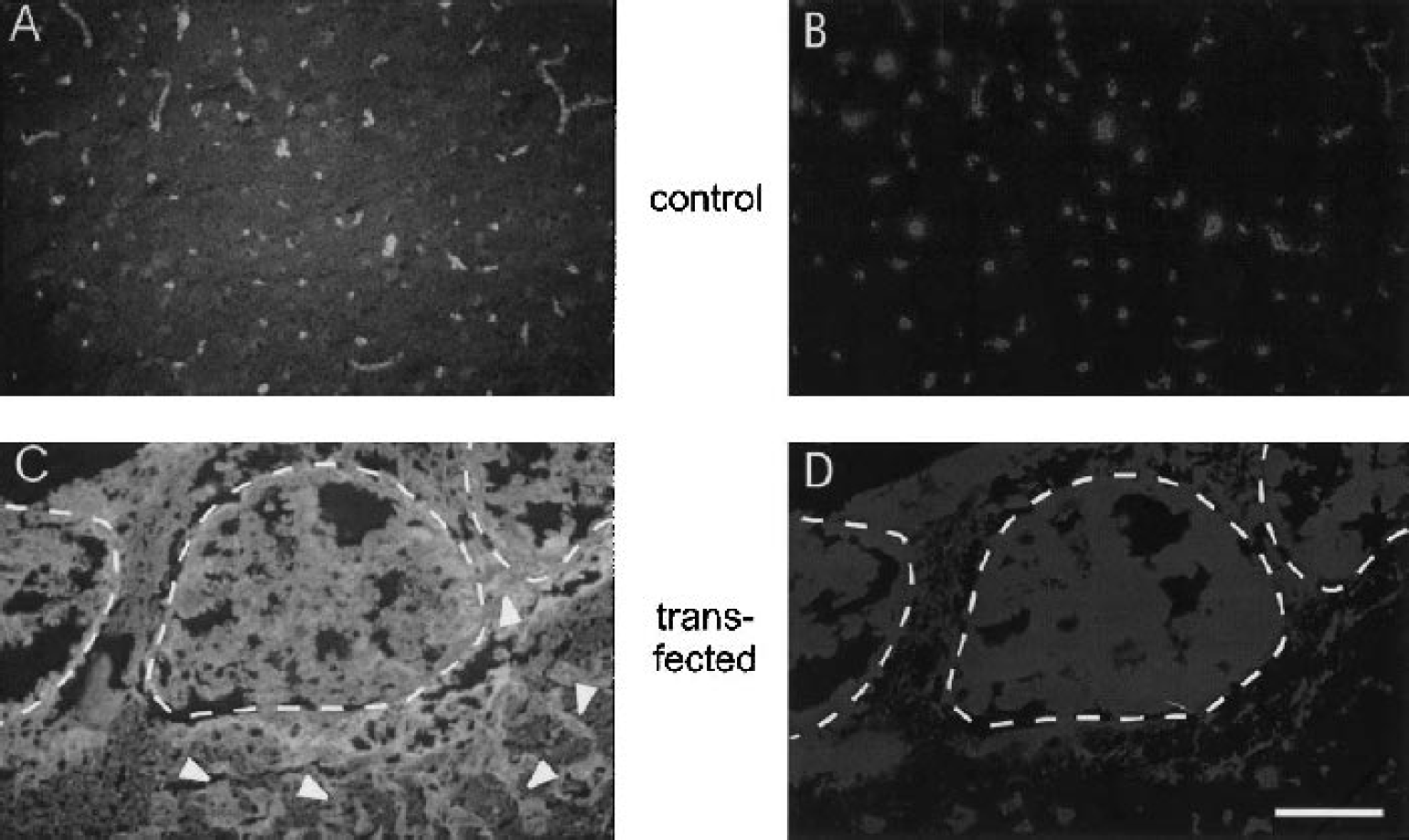
Fluorescent microscopic photographs of two representative brain sections stained for fibronectin using indirect immunofluorescence
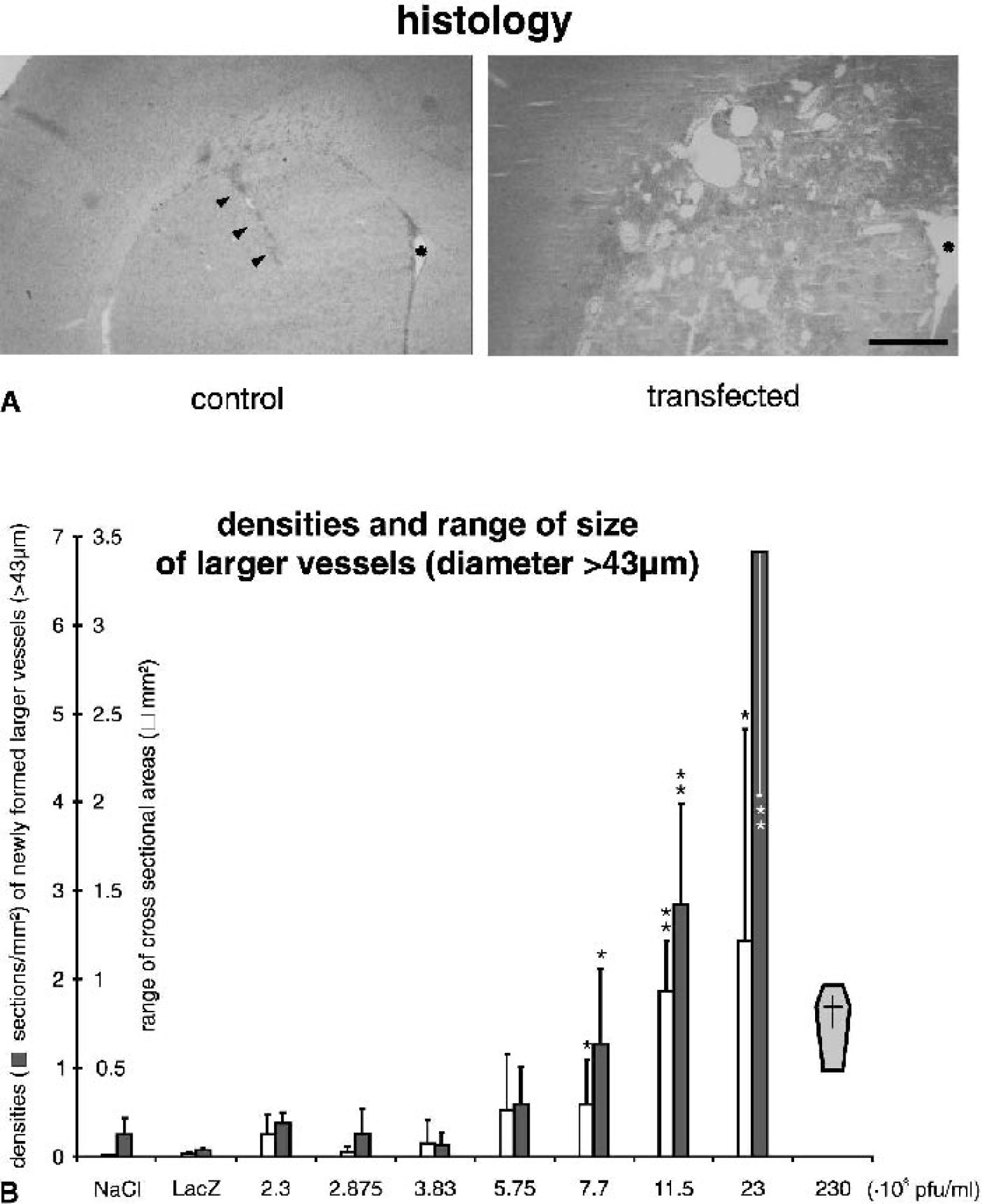
Vessel morphology
The values of changes in blood flow resulting from the newly formed vessels are given in Fig. 4. This analysis showed an unchanged global flow in the transfected hemisphere compared with the contralateral side. Near the injection site, however, the average local CBF (lCBF) was up to 178% higher than the flow in corresponding contralateral section areas at the highest survival dose. In some of these areas, lCBF was even increased up to eightfold compared with the contralateral side. Adjacent to regions of increased CBF, CBF was up to 50% lower than in corresponding contralateral section areas, and in some of these areas CBF was decreased to values as low as 15% compared with the contralateral side. Figure 4B depicts the tissue volumes in which CBF was either decreased or increased. In both control groups and up to a dose of 5.75 · 108 pfu/mL, the volume of the changed blood flow was negligible. At higher doses, the volume of the changed CBF (elevated + lowered CBF) increased significantly. At two doses (7.7 · 108 and 11.5 · 108 pfu/mL), the volume of increased CBF was more than double that of lowered CBF. This indicates that adenovirus mediated transfection of VEGF can result in a net increase of the local CBF. Because the tissue volumes in which CBF was changed were small compared with the total hemisphere, these changes did not result in a significant increase in blood flow of the total transfected hemisphere.
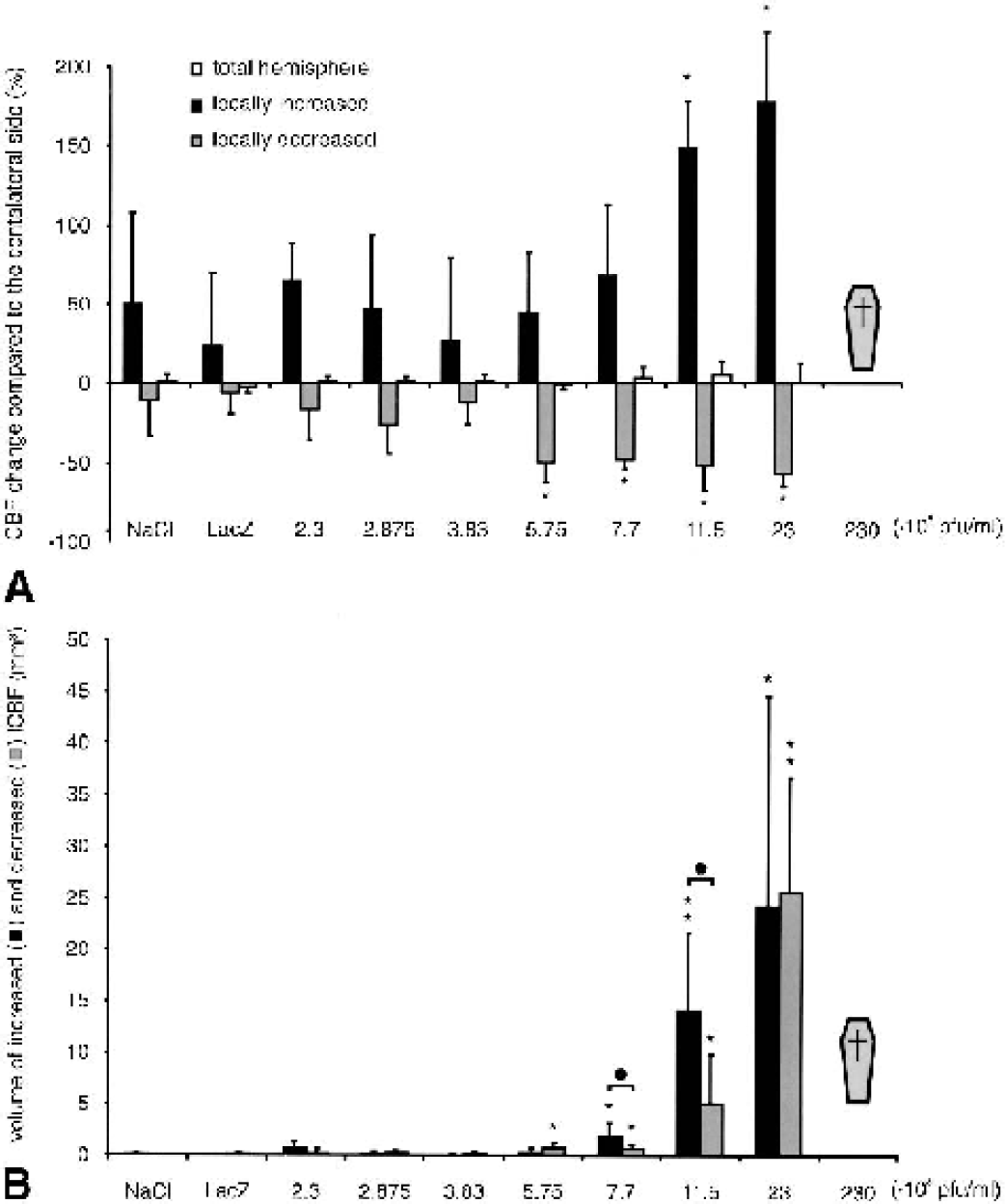
Changes in cerebral blood flow (CBF)
Because angiogenesis in the present experiments resulted in increased as well as decreased lCBF, it was of interest to relate the size and density of newly formed vessels to the lCBF found in the same tissue areas. It appeared that the largest vessels were associated with decreased lCBF. Therefore, the maximal cross-sectional areas found in areas of increased lCBF were compared with those found in areas of decreased lCBF. Figure 5A shows the relation between the maximal size of the cross-sectional areas of the newly formed vessels and the lCBF. In areas of decreased lCBF, the largest cross-sectional areas of the newly formed vessels were seven times larger than those found in areas of increased lCBF. Compared with the maximal vessel size in normal brain tissue, the newly formed vessels were up to 223 times larger in areas of decreased lCBF and up to 32 times larger in areas of increased lCBF. This finding was significant for 5.75, 11.5, and 23 · 108 pfu/mL. In addition, the density of newly formed vessels exceeding a diameter of 43 μm appeared to be higher in areas with increased lCBF. Figure 5B shows the densities of these larger newly formed vessels in relation to locally increased and locally decreased CBF. In areas with decreased lCBF, the density of the newly formed vessels was 30% to 0% lower than areas with increased lCBF. This observation was significant between 3.83 and 23 · 108 pfu/mL. Compared with normal tissue, the density of larger vessels (diameter > 43 μm) was 10-fold higher in areas with decreased lCBF and 15-fold higher in areas with increased lCBF at the highest survival dose. These findings indicate that functionally different patterns of new vessels had been formed: a high density of medium-sized vessels (up to 240-μm diameter) associated with increased lCBF and a less increased density of large vessels (up to 650 μm diameter) associated with decreased lCBF. For illustration, these findings are summarized in Fig. 6.
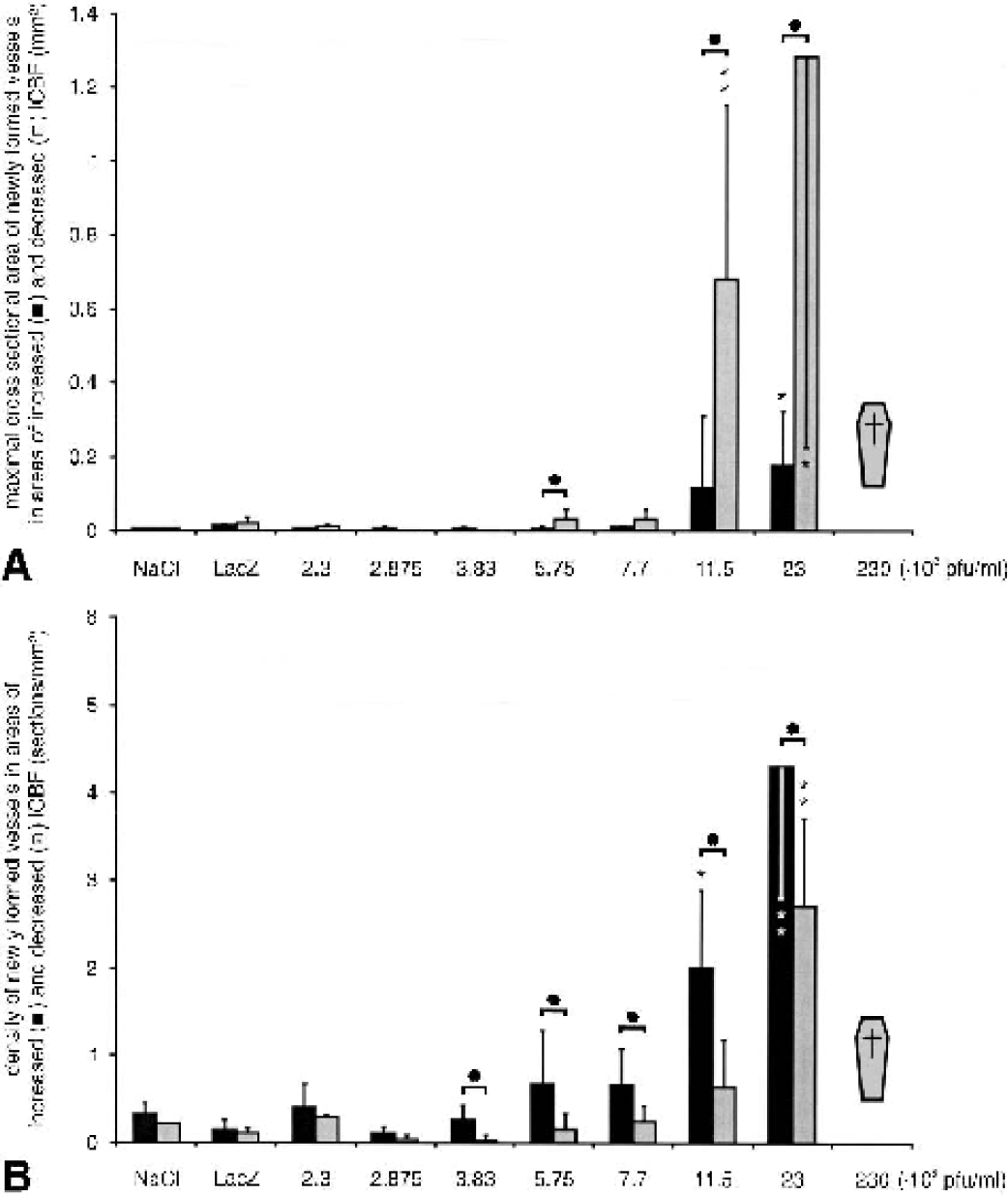
Maximal size and density of newly formed vessels larger than 43 μm in diameter related to local CBF (lCBF) in controls and transfected brains.
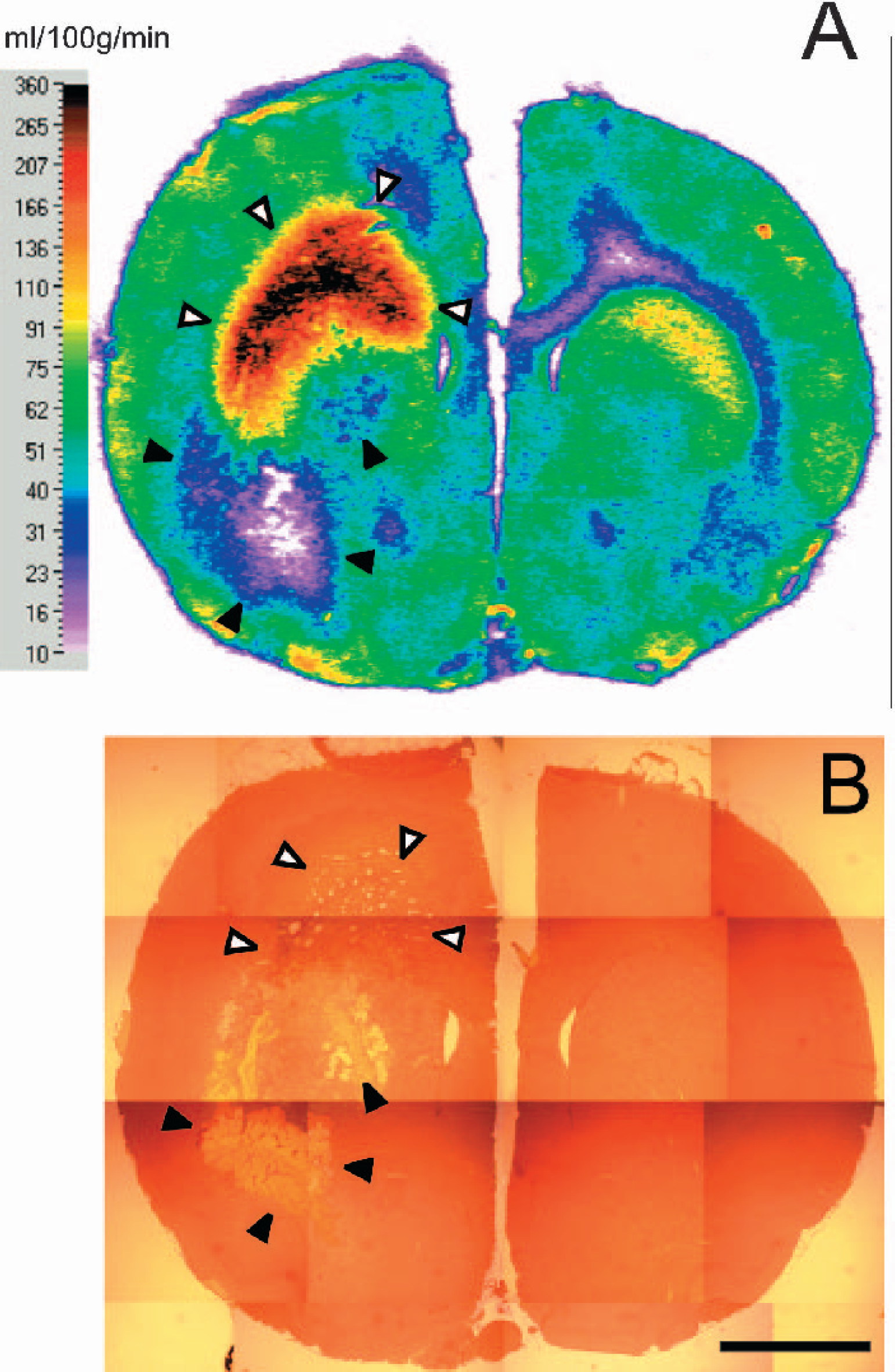
Representative color-coded autoradiogram
DISCUSSION
The present study shows the dose-dependent formation of large angioma-like blood vessels with disrupted blood–brain barrier as the result of an adenovirus-mediated transfection of the human VEGF165 cDNA into rat brains. Whether or not additional normal capillaries were formed could not be determined: the capillary marker, fibronectin (Suzuki and Choi, 1990; Hamann et al., 1995), was visible all over the brain tissue in the transfected areas and not confined to the capillaries. In addition, the intravascular marker, Evans blue, was leaking from the blood into brain tissue and prevented the detection of capillaries by an intravascular marker. Therefore, only vessels of a diameter >43 μm could be detected accurately. Compared with normal brain tissue, the maximal vessel size of the newly formed vessels was up to 223 times larger and the vessel density was up to 15-fold higher. Whereas vascular cross-sectional areas were increased in the transfected tissue, CBF showed various reactions. LCBF was increased up to eightfold in some areas and decreased down to 15% of normal values in others. At medium doses of the adenoviruses, tissue volumes of increased lCBF were more than double as large as volumes of decreased lCBF, resulting in a net increase of local CBF around the injection site. However, these local changes in CBF did not result in changes of the global flow within the transfected hemisphere. Although vessel diameters were increased in all transfected areas, the pattern of distribution of vessel diameters was related to lCBF: in the low flow areas, fewer and larger vessels were found than in the high flow areas.
Adenovirus-mediated transfection has been applied to tissues of several organs (Pettersson et al., 2000) and turned out to be an effective technique of gene transfer into adult brain tissue (Peltekian et al., 1997). This is probably caused by the small or missing target cell specificity of adenoviruses in nonpermissive hosts such as rats or mice as well as in permissive hosts such as humans (Rosenfeld et al., 1991; Engelhardt et al., 1993). Thus, all brain cells that gain contact with the virus can be transfected effectively. In the present study, an infusion rate of 0.5 μL/min was chosen for the infused virus suspension. This is the lowest infusion rate reported in the literature (Davidson et al., 1993) and was effective to transfect a volume of more than 50 μL of the rat brain tissue that could be detected by the reporter gene β-galactosidase (data not shown). Probably because of the low infusion rate of 0.5 μL/min that was kept for 90 minutes, a long-lasting contact between virus suspension and tissue could be established. In addition, a slow infusion rate prevents (or at least reduces) back flow of the virus suspension along the needle track, which would result in a loss of adenovirus particles into the subarachnoid space (Deinsberger et al., 1996). Therefore, the adenovirus transfection protocol chosen in the present study resulted—to the knowledge of the authors—in the largest volumes of transfected brain tissue ever reported for rats in the literature (52 ± 8 μL). Such large volumes of transfected tissue were the basis of a reliable analysis of the vessel morphology and CBF in the present study.
Because angiogenesis induces increased vessel density, an increase in blood flow near the injection site could be expected after transfection of VEGF. In the present study, this was found only in some areas of the transfected brain tissue. Areas of increased or decreased CBF did not show any systematic relation to the needle tract. It is surprising that an increased density of newly formed vessels was also associated with a severe decrease of lCBF in adjacent brain areas. This is an important finding of the present study because it contradicts the expectation that an increased vessel density should result in an increased blood flow. Angiogenesis might result in severely decreased flow. In principle this could have two reasons: (1) The flow capacity of the upstream vessels might not be sufficient to serve additional, newly formed vessels; or (2) functionally effective (in areas of increased CBF) parallel to functionally ineffective new vessel networks (in areas of decreased CBF) might have been formed. Because up to eightfold increased values of CBF could be found in some areas, a limited flow capacity of upstream vessels appears to be unlikely. This is in line with estimates that at least 50% of the upstream vascular resistance in the brain is located in arteries and arterioles larger than 160 μm in diameter (Faraci et al., 1987; Faraci and Heistad, 1990). However, it could be argued that different brain areas have a different vascularization, which might have a different flow capacity, e.g., some areas might have a limited flow capacity whereas others do not. If this were the only reason, a similar vascular pattern in areas of increased and areas of decreased CBF should be expected. This, however, was not the result of the present study because different vascular patterns were found that were associated with different CBF values: a high density of medium-sized vessels in which CBF is increased and a moderately increased density of large vessels in which CBF is decreased. The fact that in the present study the largest vessels were associated with a severe reduction of CBF indicates a specific reduction of the perfusion in the most abnormal angioma-like vessels. This might be explained by a reduction of flow velocity in these large vessels, resulting in a reduced wall shear stress as a consequence of an increased diameter as long as the blood flow (in milliliters per minute) is unchanged. Recent studies suggest that a reduction of the wall shear stress triggers vessel regression (Bongrazio et al., 2000; Pries et al., 2001). Because all measurements had been performed 11 d after VEGF transfection, such mechanisms might have started resulting in a reduced CBF as a sign of a beginning regression of the angioma-like vessels (Pettersson et al., 2000).
Effects of an unregulated overexpression of VEGF, for example, in the heart, have been reported in other studies. In accordance with the present study, abnormal vessels have been found (Carmeliet, 2000b; Lee et al., 2000). This could be explained by the interaction of several factors, which is necessary for the formation of morphologically normal and functional microvessels (Carmeliet, 2000a). Whereas the action of additional pro- and/or antiangiogenic factors is the prerequisite for a normal vessel formation, the situation is less well defined during pathophysiologic conditions. Upregulation of VEGF has been shown in parallel with the growth of new vessels in hypoxic myocardium (Marti and Risau, 1999) and in the penumbra around focal brain ischemia (Marti et al., 2000). In addition, it also has been demonstrated recently that angiopoietin is upregulated in penumbral tissue of cerebral infarcts (Lin et al., 2000, 2001). These findings indicate that at least angiopoietin in addition to VEGF is necessary to form normal vessels. It is possible to induce new vessel growth in the brain by transfection of VEGF. Doses of adenovirus vectors between 0.77 and 1.15 · 109 pfu/mL were effective to induce a net increase of lCBF. Lower doses had no effect, whereas at higher doses transfection was deleterious or even lethal. However, even at the doses with a net increase of lCBF, deleterious effects occurred such as increased blood-barrier permeability, reduced blood flow in neighboring brain areas, or abnormal vessel formation. These findings and those of Lin et al. (2000, 2001) concerning angiopoietin overexpression after cerebral stroke indicate that only a coadministration of different angiogenic factors might result in the formation of healthy vessels.
The present results show that adenovirus-mediated transfection of the human VEGF165 cDNA in rat brains results in areas of locally increased and decreased CBF. At a defined dose of adenoviral particles, the tissue volumes with locally increased CBF are more than two times larger than tissue volumes with decreased CBF. Areas with decreased lCBF were characterized by 30% to 40% fewer but sevenfold larger newly formed vessels compared with areas of increased lCBF. Transfection of the human VEGF165 cDNA into rat brains appears to result in two functionally different patterns of newly formed vessels: high density of moderate-sized vessels resulting in an increased lCBF and moderately increased density of large vessels resulting in a decrease of lCBF.
Footnotes
Acknowledgments:
The authors thank Dr. G. Breier (Max-Planck-Institute for Physiological and Clinical Research, Bad Nauheim, Germany) for the kind gift of the human VEGF165-cDNA and Dr. J. Kreuzer (Dept. of Internal Medicine, University of Heidelberg, Heidelberg, Germany) for providing the replication-deficient adenovirus Ad d1324, the packaging human embryonic kidney cell line 293, the adenovirus carrying the β-galactosidase cDNA and the adenovirus expression vector pAC.CMV.plpA(+). In addition, the authors thank Renate Bangert for assembling the adenovirus containing the VEGF165 cDNA.
