Abstract
Adult ependymal cells are postmitotic and highly differentiated. Radial glial cells are neurogenic precursors. Here, we show that stroke acutely stimulated adult ependymal cell proliferation, and dividing ependymal cells of the lateral ventricle had genotype, phenotype, and morphology of radial glial cells in the rat. The majority of radial glial cells exhibited symmetrical division about the cell cleavage plane, and a radial fiber was maintained throughout each stage of cell mitosis. Increases of radial glial cells parallel expansion of neural progenitors in the subventricular zone (SVZ). Furthermore, after stroke radial glial cells derived from the SVZ supported neuron migration. These results indicate that adult ependymal cells divide and transform into radial glial cells after stroke, which could function as neural progenitor cells to generate new neurons and act as scaffolds to support neuroblast migration towards the ischemic boundary region.
Introduction
The embryonic ventricular zone (VZ) contains two cell types: neuroepithelial progenitors (NEPs) and radial glial cells, which are distinct both morphologically and functionally. Neuroepithelial progenitors are present before neurogenesis and are considered cycling progenitor cells and the source of neurons (Takahashi et al, 1993). Radial glial cells containing radial processes and astro-glial properties function as migratory scaffolds and glial progenitors during development (Chanas-Sacre et al, 2000). This concept has been changed by the observation that most or all VZ cells possess radial glia characteristics and many radial glial cells are dividing progenitors capable of generating neurons, indicating that radial glial cells can act as neural stem cells (Miyata et al, 2001; Noctor et al, 2001; Weissman et al, 2003; Anthony et al, 2004). In the adult mammalian brain, radial glial cells differentiate into astrocytes and are no longer present (Voigt, 1989) However, others have shown that radial glial cells give rise to adult neural stem cells, suggesting a role of radial glial cells in the postnatal period and adulthood (Doetsch et al, 1999; Gregg and Weiss, 2003; Merkle et al, 2004).
Ependymal cells lining the lateral ventricular wall have cuboidal morphology with cilia facing the ventricular lumen (Bruni, 1998; Spassky et al, 2005) and they play a critical role in brain homeostasis (Bruni, 1998; Spassky et al, 2005). Ependymal cells are descendants of the embryonic VZ cells, which share several markers with VZ cells. A recent study shows that ependymal cells are derived from radial glial cells during embryogenesis and adult ependymal cells are postmitotic and highly differentiated (Spassky et al, 2005). Ependymal cells are not neural stem cells under normal conditions (Chiasson et al, 1999; Johansson et al, 1999; Rietze et al, 2001).
The subventricular zone (SVZ) of adult rodent brain contains SVZ astrocytes (type B cells), rapidly dividing transient amplifying cells (type C cells) and migratory neuroblasts (type A cells). Type B cells are neural stem cells with characteristics of astrocytes (Doetsch et al, 1999; Gregg and Weiss, 2003; Merkle et al, 2004; Mori et al, 2005). Ischemic stroke induces neurogenesis in the SVZ and neuroblasts in the SVZ migrate towards the ischemic boundary region (Zhang et al, 2001; Arvidsson et al, 2002; Parent et al, 2002; Jin et al, 2003; Zhang et al, 2004b). Depleting type A and C cells in the SVZ results in a more rapid repopulation of neuroblasts after stroke than repopulation in the non-stroke SVZ, suggesting that stroke may increase recruitment of neural stem cells (Zhang et al, 2004b). Adult SVZ proliferating cells exhibit interkinetic nuclear migration, one of characteristics of radial glial cells. Transient increases of SVZ cell division from asymmetric to symmetric contribute to stroke-induced neurogenesis (Zhang et al, 2004a). In addition, stroke induces proliferation of ependymal cells (Zhang et al, 2001). Infusion of epidermal growth factor (EGF) into the lateral ventricle induces ependymal cells to adopt radial glial morphology in the adult mouse (Gregg and Weiss, 2003). Collectively, these studies raise the possibility that after stroke ependymal cells differentiate into radial glial cells. Based on their functions as neuronal precursors and as guides of neuronal migration, radial glial cells might be involved in stroke-induced neurogenesis and in supporting redirection of neuroblasts oriented to the ischemic boundary region. In the present study, we hypothesized that stroke induces generation of functional radial glial cells from ependymal cells. Using an array of in vivo and in vitro experiments, we tested this hypothesis in adult rats subjected to embolic middle cerebral artery occlusion.
Materials and methods
All experimental procedures have been approved by Institutional Animal Care and Use Committee of Henry Ford Hospital.
Animal Model of Stroke
Male Wistar rats (3 to 4 months old) were employed. The right middle cerebral artery was occluded by placement of an embolus at the origin of the middle cerebral artery (Zhang et al, 1997). Animals were killed at 1, 2, 4, and 7 days after middle cerebral artery occlusion.
5-Bromo-2′-deoxyuridine Labeling
Single pulse labeling: Stroke rats receiving a single dose of 5-bromo-2′-deoxyuridine (BrdU) (50 mg/kg, i.p) were killed 1 h (n = 4 per stroke time point) after the injection (Takahashi et al, 1993).
Cumulative labeling protocol: Experimental stroke rats were i.p injected. 5-Bromo-2′-deoxyuridine (50 mg/kg) at 2 h intervals over a total period of 18.5 h, and rats were killed 30 mins after the last injection (Takahashi et al, 1993). Four to six animals per stroke time point were used for this study.
Histology and Immunohistochemistry
Animals were transcardially perfused with heparinized saline followed by 4% paraformaldehyde. Brains were fixed in 4% paraformaldehyde and embedded in paraffin. Coronal sections (6 μm) of the lateral ventricles were cut serially at the level of between AP + 10.6 mm and AP + 9.2 mm, which cover entire the SVZ (Zhang et al, 2001, 2004a).
Immunohistochemistry was performed, as previously described (Zhang et al, 2001). The following antibodies were used in the present study: mouse anti-BrdU (1:1000, Boehringer Mannheim, Indianapolis, IN, USA); mouse anti-phosphorylated form of vimentin (4A4, 1:1600, Medical & Biological laboratories Co., Ltd, Naka-kuNagoya, Japan); mouse anti-nestin (1:100, BD Bioscience, Franklin, NJ, USA); mouse anti-vimentin (1:1000, Santa Cruz Biotechnology, Santa Cruz, CA, USA); guinea-pig anti-GLAST (1:4000, Chemicon, Temecula, CA, USA); mouse anti-3CB2 (1:50, DSHB, Iowa city, IA, USA); rabbit anti-S-100β (1:1000, DAKO, Caspenteria, CA, USA); rabbit anti-phospho-Histone H3 (HH3), (1:1000, Upstate Biotech, Lake Placid, NY, USA), rabbit anti-Ki67 (1:300, Abcam, Cambridge, MA, USA), and goat anti-doublecortin (DCX) (1:200, Santa Cruz Biotechnology, Santa Cruz, CA, USA).
For BrdU immunostaining, DNA was first denatured by incubating coronal sections in 50% formamide 2 × SSC at 65°C for 2 h and then in 2 N HCl at 37°C for 30 mins (Zhang et al, 2001). Sections were incubated with the anti-BrdU antibody (1:1000, Boehringer Mannheim, Indianapolis, IN, USA) overnight and then incubated with biotinylated secondary antibody (1:200, Vector, Burlingame, CA, USA) for 1 h. Reaction product was detected using 3′3′-diaminobenzidine-tetrahydrochloride (DAB, Sigma, St. Louis, MO, USA).
Image Acquisition, Quantification of Immunoreactive Cells and Measurement of Ischemic Lesion Area
For analysis of immunoreactive cells, every 30th section and the adjacent coronal sections that stained with the above described antibodies at the level of AP + 10.6 mm and AP + 9.2 mm were digitized under a × 60 objective (Olympus BX40) via the MCID computer imaging analysis system (Imaging Research, St Catharines, Canada). Total density for the four sections per rat was obtained for each brain according to the published methods (Zhang et al, 2001, 2004a). Three-dimensional images were acquired using Zeiss confocal microscopy (Zeiss LSM 510 NLO).
To examine whether occlusion of the middle cerebral artery induces an ischemic lesion, a coronal section at the level of AP + 9.70 mm from each rat was stained with hematoxylin and eosin, and this section was at the center of the territory supplied by the middle cerebral artery (Zhang et al, 1997). The ischemic lesion area was measured using MCID system as previously described (Zhang et al, 1997). To reduce errors associated with processing of tissue for histologic analysis, the lesion area was presented as the percentage of the lesion to the area of the contralateral hemisphere (Zhang et al, 1997).
Neurosphere Cultures
Subventricular zone cells of the lateral ventricle were dissociated from non-stroke (n = 4) and 2 day stroke rats (n = 6) (Zhang et al, 2004b). The cells were cultured in medium containing 20 ng/mL bFGF and EGF. The generated neurospheres (primary sphere) were passaged by mechanical dissociation and reseeded as single cells at a density of 20 cells per microliter in EGF-containing media (passage 1 cells). Passage 1 cells were employed in the present studies.
In Vitro Migration Assay
A neurosphere (~200 μm) was embedded in Matrigel (30 μL) and cultured in medium containing bFGF and EGF. Cultured neurospheres were incubated in an environmental chamber with 5% CO2 and 37°C (LiveCell Control Unit), which was placed on the stage of the Nikon TE2000-U Inverted Microscope equipped with a motorized Z stage. Cellular processes and cell motility were recorded by a CCD camera by acquiring a stack of images (30 images with a 5-μm step in Z axes) at 30 mins intervals for a total of 20 h. Cell migration speed was analyzed by tracing individual cells at different levels and times using Universal Imaging MetaMorph Software.
Laser Capture Microdissection
Subventricular zone cells were captured according to our published methods (Liu et al, 2006). Briefly, cryostat sections were stained with propidium iodide dye (1:3000 dilution, Sigma) for 5 mins and sections were then air-dried under laminar flow for 10 mins. Propidium iodide positive SVZ cells were captured onto a thermoplastic film mounted on optically transparent LCM caps using the PixCell II LCM System (Arcturus Bioscience Inc., Mountain View, CA, USA). Caps with cells were immediately placed into Eppendorf tubes containing 350 μL of lysis buffer and stored in -80°C before RNA isolation. Approximately 3000 cells were isolated in the SVZ from each animal.
Isolation of Single Ependymal Cells
Single ependymal cell was isolated according to the published methods with some modifications (Palma et al, 2005). Briefly, brains from non-stroke and stroke rats were quickly removed and placed in an oxygenated storage chamber containing ice-cold physiologic rat artificial cerebral spinal fluid (95% O2 and 5% CO2) for 3 mins. Coronal slices containing the lateral ventricles (400 μm) were cut with a vibratome and incubated in the oxygenated artificial cerebral spinal fluid at 32°C for 30 mins. Thereafter, a coronal slice was mounted on a custom-made perfusion chamber, which was continuously perfused with the oxygenated artificial cerebral spinal fluid at 3.0 mL/mins. The brain slice was viewed under Nikon Eclipse E600-FN microscope (Nikon Corporation, Tokyo, Japan). A cell with cilia along the lateral wall of the lateral ventricle was approached with a patch clamp pipette (7 to 10 MΩ) filled with the intracellular solution. After immersion of the pipette into the perfusion solution, a positive pressure was applied to the pipette to prevent obstruction. Using a three-dimensional micromanipulator, the patch pipette was advanced to touch the cell (Figure 4C). The cell was then harvested from the tissue by applying negative pressure to the pipette and the pipette was carefully moved out of the solution. The cell and pipette content were then expelled into a reaction tube.
Multiplex Single-cell Reverse Transcription-Polymerase Chain Reaction and Real-time Reverse Transcription-Polymerase Chain Reaction
For synthesis of the first-strand cDNA, cell content was incubated at 65°C for 5 mins in a total volume of 13 μL containing 1 μL oligo(dt)20 primer and 1 μL 10 mmol/L dNTPs (Invitrogen Inc., Carlsbad, CA, USA). After that, 4 μL 5 × first-strand buffer, 1 μL 0.1 M DTT, 1 μL of RNaseOUT recombinant RNase inhibitor, and 200 U of reverse transcriptase (Superscript™ III; Gibco-BRL, Carlsbad, CA, USA) were added and incubated at 50°C for 60 mins. The single-cell cDNAs were kept at -80°C until PCR amplification.
Single-cell reverse transcription-polymerase chain reaction (RT-PCR) was performed based on methods described previously (Palma et al, 2005). Multiplex-PCR was performed as hot start in a final volume of 30 μL containing 10 μL RT products, 0.1 μmol/L of each primer, HotStarTaq DNA polymerase, and 15 μL Qiagen Mutiplex mix in a Perkin Elmer Thermal Cycler 48°C with the following cycling protocol. The first amplification consisted of 10 mins hot start at 95°C followed by 28 cycles of (40 secs 94°C, 40 secs at 56°C, 1 min at 72°C), 10 mins at 72°C. The second-PCR amplications were performed in 12 individual reactions, which consisted of 3 mins at 95°C followed by 35 cycles of (40 secs at 95°C, 40 secs at 56°C, 1 min at 72°C), 10 mins at 72°C. The amplification was performed in a total volume of 20 μL containing water, 2 μL first PCR product, 0.2 mmol/L dNTPs, 1.5 mmol/L MgCl2, 1 μmol/L of corresponding primer pair, 1 × PCR buffer, and 1 U Platinum Taq DNA polymerase. After amplification, the PCR products were analyzed on 1.5% agarose gels.
Total RNA from captured cells was isolated using RNeasy Micro Kit (Qiagen Incorporation, Valencia, CA, USA) and was followed by reverse-transcription. Quantitative RT-PCR was performed on an ABI 7000 PCR instrument (Applied Biosystems, Foster City, CA, USA) using three-stage program parameters provided by the manufacturer. Primers (Table 1) used in the present study were designed using OligoPerfect™ Designer software. Sequence specificity was examined using a nucleotide database. Each sample was tested in triplicate and data obtained from three independent experiments were expressed as a subtraction of the quantity of specific transcripts from the quantity of the control gene β-actin in mean arbitrary units. CT values were quantified by the 2−ΔΔCt method (Liu et al, 2006).
Primers used for mutiplex single cell RT-PCR

Results
Stroke Stimulates Adult Ependymal Cell Proliferation
Adult ependymal cells are postmitotic and their proliferative capacity remains controversial (Johansson et al, 1999; Spassky et al, 2005). To examine whether stroke induces ependymal cell proliferation, we injected BrdU at 2 h intervals over a total period of 18.5 h, and rats were killed 30 mins after the last injection. Double immunostaining with antibodies against S100β and BrdU was performed to identify ependymal and proliferating cells, respectively. In non-stroke rats, BrdU+ SVZ cells and S100β+ ependymal cells were detected, which were not co-localized each other (Figure 1A–Figure 1C). This result is consistent with previous findings that ependymal cells do not proliferate in adult brain (Spassky et al, 2005). However, 1 to 2 days after stroke, BrdU+ and S100β+ ependymal cells were found in the ipsilateral lateral ventricle (Figure 1D–Figure 1F). BrdU+ mitotic ependymal cells were also detected (Figure 1G, arrow). These BrdU+ ependymal cells had fewer cilia than BrdU− ependymal cells (Figure 1D–Figure 1F and Figure 1H, arrowhead) and exhibited a short process (Figure 1D and Figure 1E, arrows). BrdU+ and S100β+ ependymal cells were not detected in the ipsilateral ventricle 7 days after stroke. All rats subjected to middle cerebral artery occlusion exhibited the ischemic lesion, with an ischemic area of 41% ± 5.6% of the contralateral hemisphere at 2 days, 43% ± 7.4% at 4 days, and 44% ± 5.4% at 7 days after stroke, which are consistent with our published data (Zhang et al, 1997, 2001). These data indicate that stroke transiently stimulates ependymal cell proliferation.
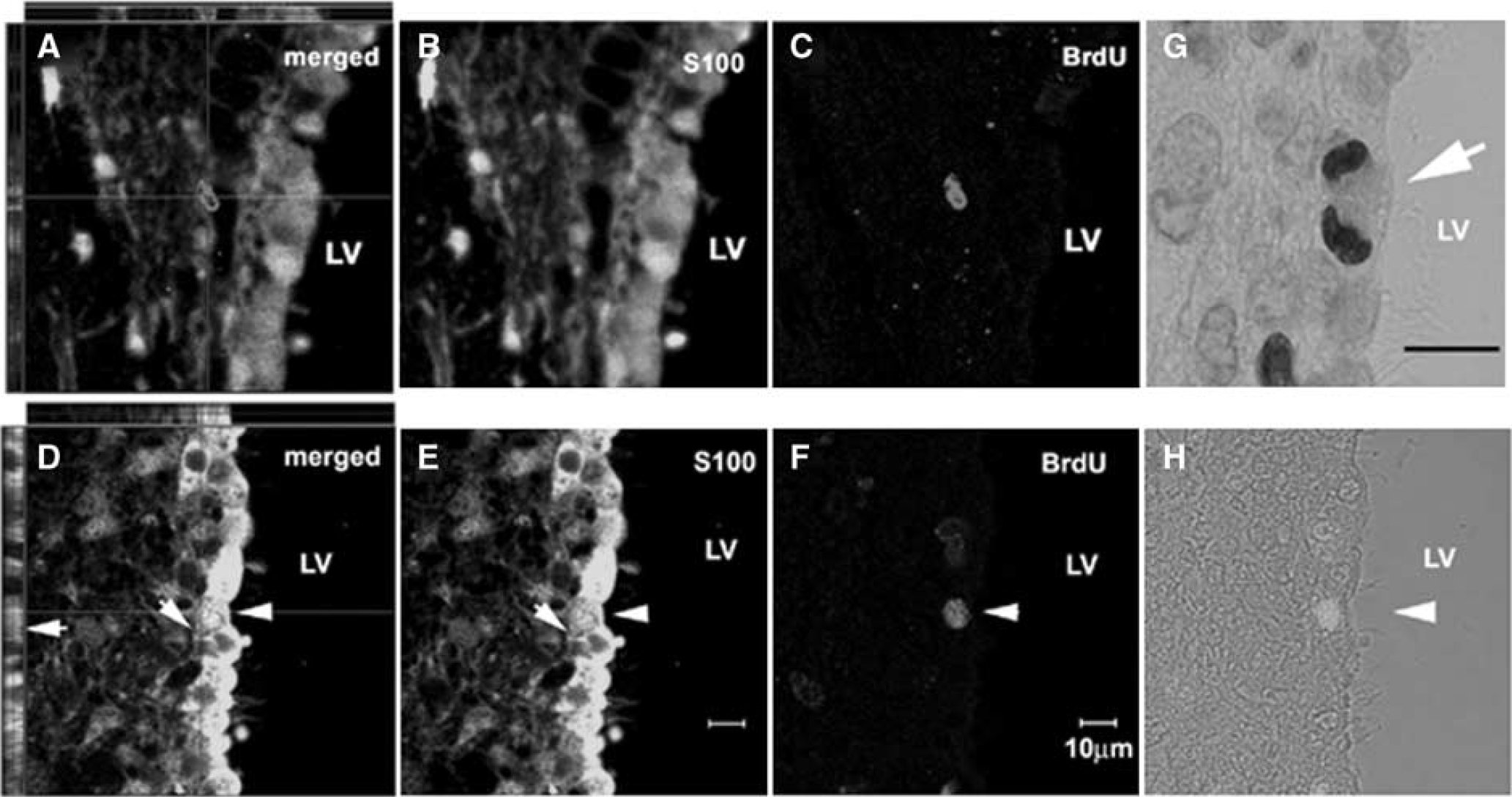
Ependymal cells divide after stroke. Confocal images of double immunofluorescent staining show S100β+ (
Dividing Ependymal Cells Exhibit Phenotype and Morphology of Radial Glia
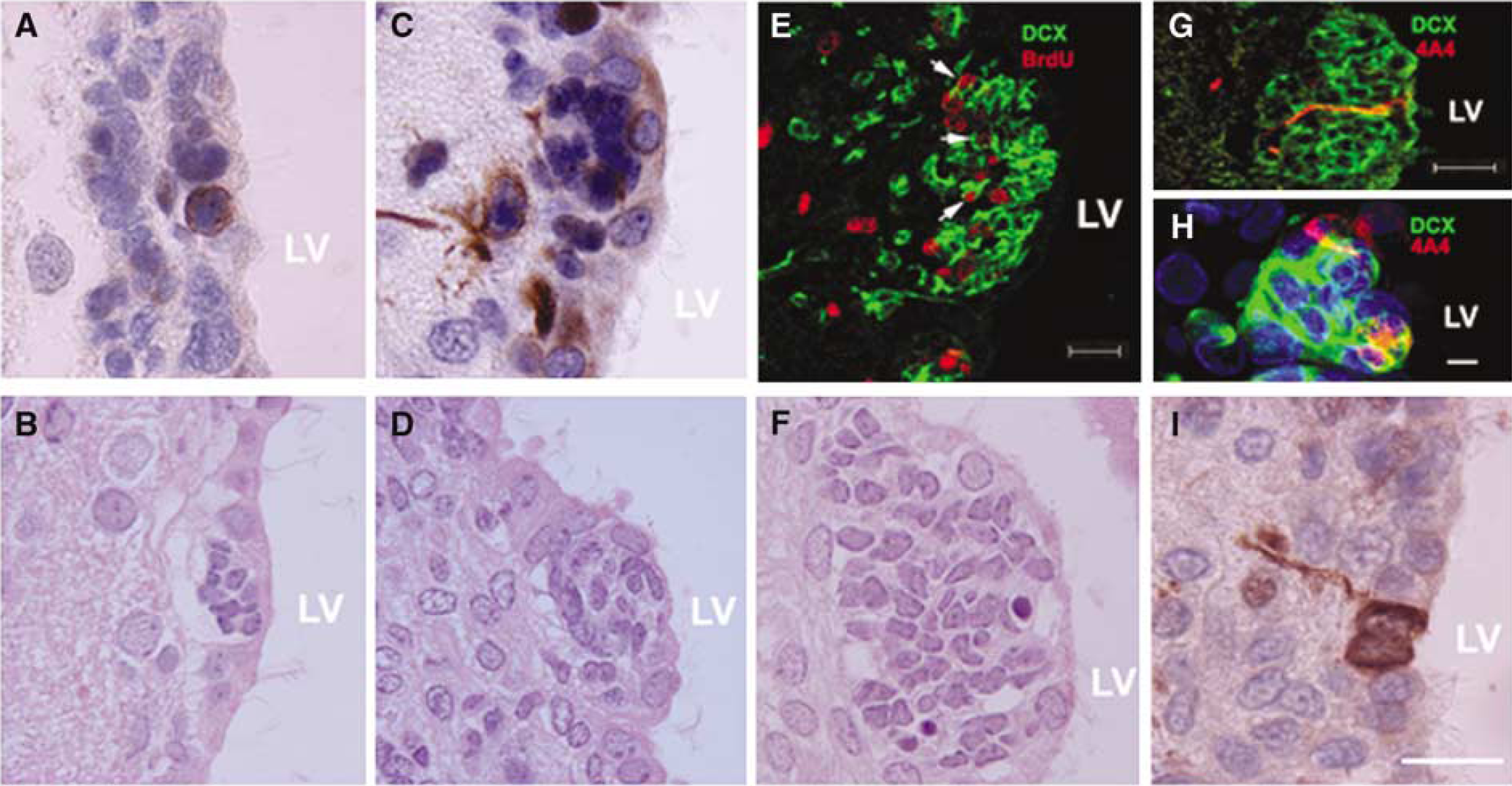
Adult ependymal cells are derived from radial glia during embryogenesis (Spassky et al, 2005). Infusion of EGF into the lateral ventricle induces ependymal cells to adopt radial glial morphology in adult mice (Gregg and Weiss, 2003). These data raise the possibility that proliferating ependymal cells in stroke brain can transform to radial glial cells. To examine this, we performed immunostaining with a monoclonal antibody that specifically recognizes the cdc2-kinase phosphorylated form of vimentin (4A4), a radial glial-specific mitotic marker (Weissman et al, 2003). In non-stroke rats (n = 6), few 4A4+ cells (< 0.3/section, n = 6) were detected, whereas 1 to 2 days after stroke 4A4+ abundant cells were found (6 ± 0.3/section, n = 8) along the ipsilateral lateral ventricular surface with a basal process directed away from the ventricular surface and towards the striatum (Figure 2A and Figure 2B, star). To further assess the proportion of proliferating cells that exhibit radial glial phenotype, we performed double immunofluorescent staining of 4A4+ cells with antibodies against one of three cell cycle markers: BrdU, the thymidine analog that is incorporated into the DNA of dividing cells during S-phase, Ki67, that labels a nuclear transcription factor expression from S-phase through M-phase (Weissman et al, 2003) and HH3, which labels a histone phosphorylated from G2 to M-phase (Weissman et al, 2003; Figure 2E). Stroke rats killed 1 h after a single BrdU injection, which labels a cohort of cells that are in S-phase at the time of the injection (Takahashi et al, 1993; Zhang et al, 2006) did not exhibit co-labeling of 4A4 with BrdU (Figure 2F), which is consistent with fact that it takes 3 to 6 h for BrdU labeled cells to enter M-phase after a single BrdU injection (Takahashi et al, 1993). However, after the cumulative BrdU-labeling which labels cells in the cell cycle phases, 8% ± 4.9% (n = 8) of BrdU+ cells were 4A4+ (Figure 2F) (Takahashi et al, 1993; Zhang et al, 2006). We observed that 82% ± 10.4% (n = 8) of HH3+ cells were 4A4+ (Figures 2C and Figure 2F). The large overlap of these two markers are consistent with the fact that HH3 is present G2 to M-phase (Weissman et al, 2003), whereas 27% ± 9.5% (n = 8) of Ki67+ cells were 4A4+ (Figure 2D and Figure 2F), which reflects Ki-67 being present during a longer portion of the cell cycle (Weissman et al, 2003). These data suggest that most mitotically active cells along the lateral ventricular surface of the ipsilateral ventricle have radial glial phenotype.
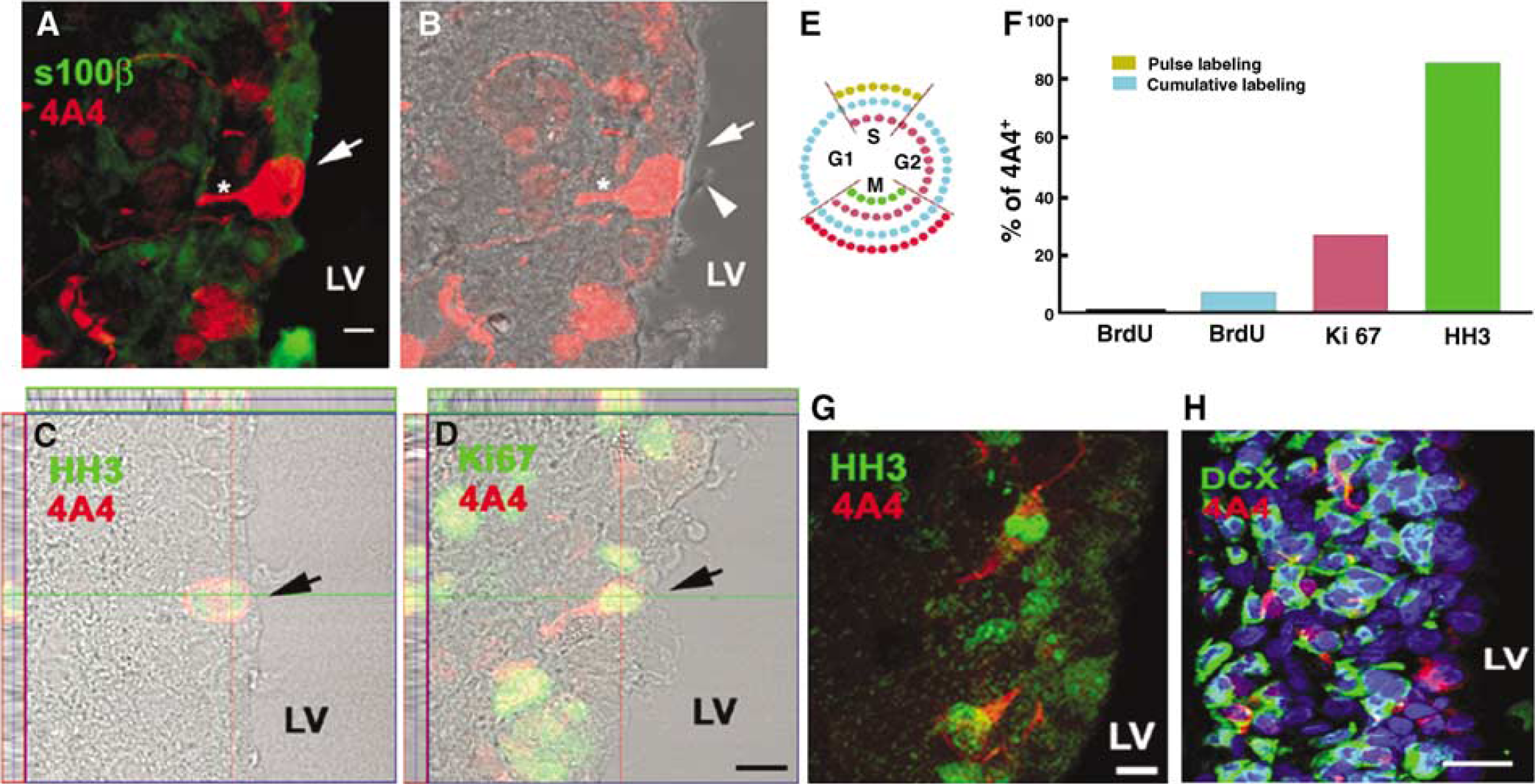
Dividing ependymal cells express radial glial cell antigen after stroke. Confocal images of double immunofluorescent staining show a S100β+ and 4A4+ cell in the lateral wall of the lateral ventricle (
Radial glial cells maintain radial glial fibers during mitosis (Miyata et al, 2001; Noctor et al, 2001; Weissman et al, 2003). We thus, examined morphology of 4A4+ and HH3+ cells during the mitotic stage from prophase to telophase. Two days after stroke, the majority of 4A4+ cells (60% ± 13.2%, n = 8) divided with a vertical cleavage plane (Figure 3F), as has been previously reported in VZ cells of rat E15 (Weissman et al, 2003). During prometaphase to anaphase, these cells exhibited a 4A4+ process stemming from the center of the basal surface (Figure 3C–Figure 3F). During telophase, a 4A4+ process was associated with one of the daughter cells (Figure 3G). In contrast, horizontally dividing 4A4+ cells (34.9% ± 12.3%, n = 8) exhibited a 4A4+ region between daughter cells (Figure 3H). These data indicate that in adult stroke brain, the majority of 4A4+ cells with a vertical cleavage plane lining the lateral ventricular surface of the ipsilateral ventricle maintain their radial processes throughout all stages of mitosis.
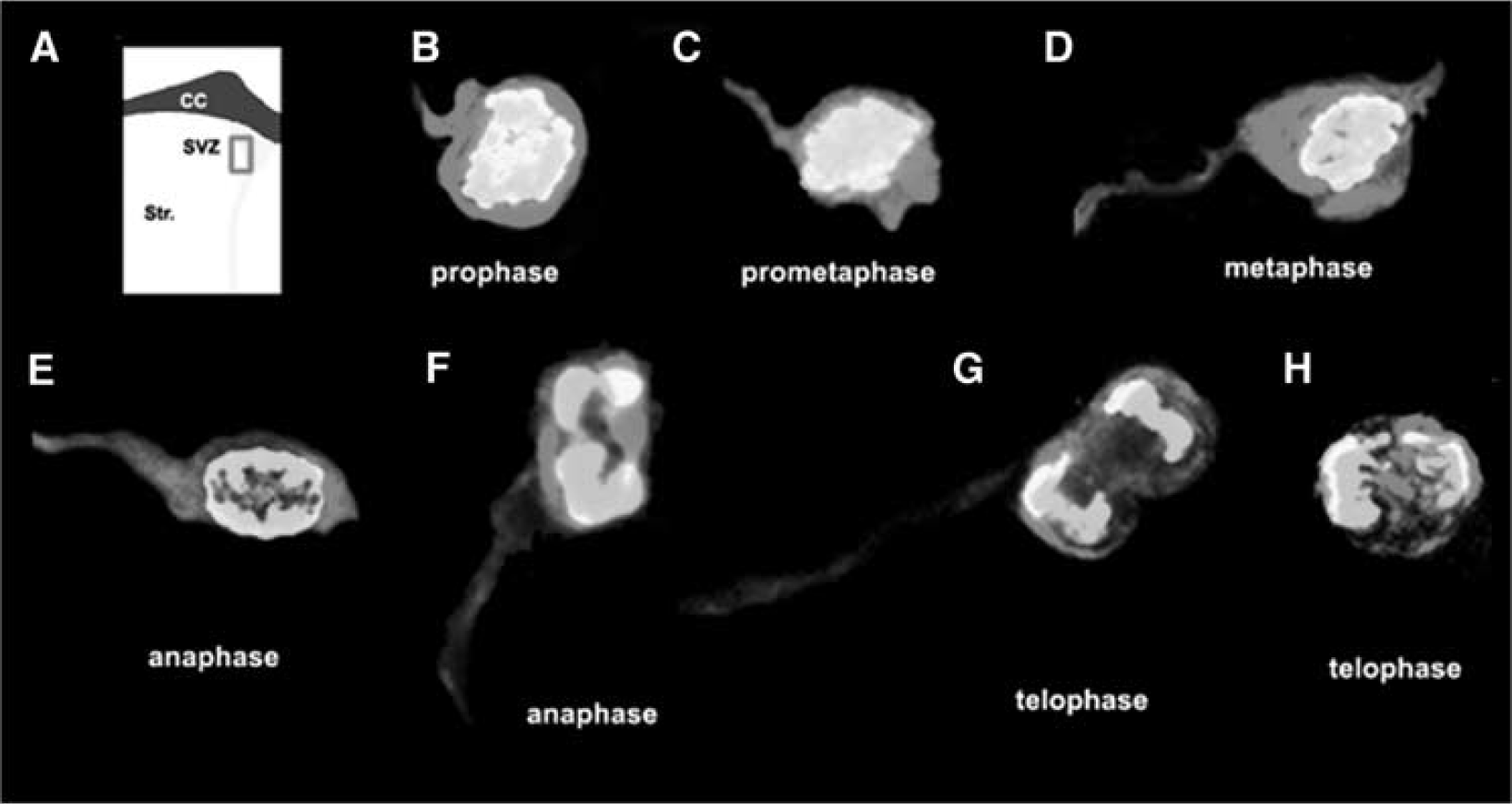
Radial glial cells retain their radial fiber throughout mitosis. (
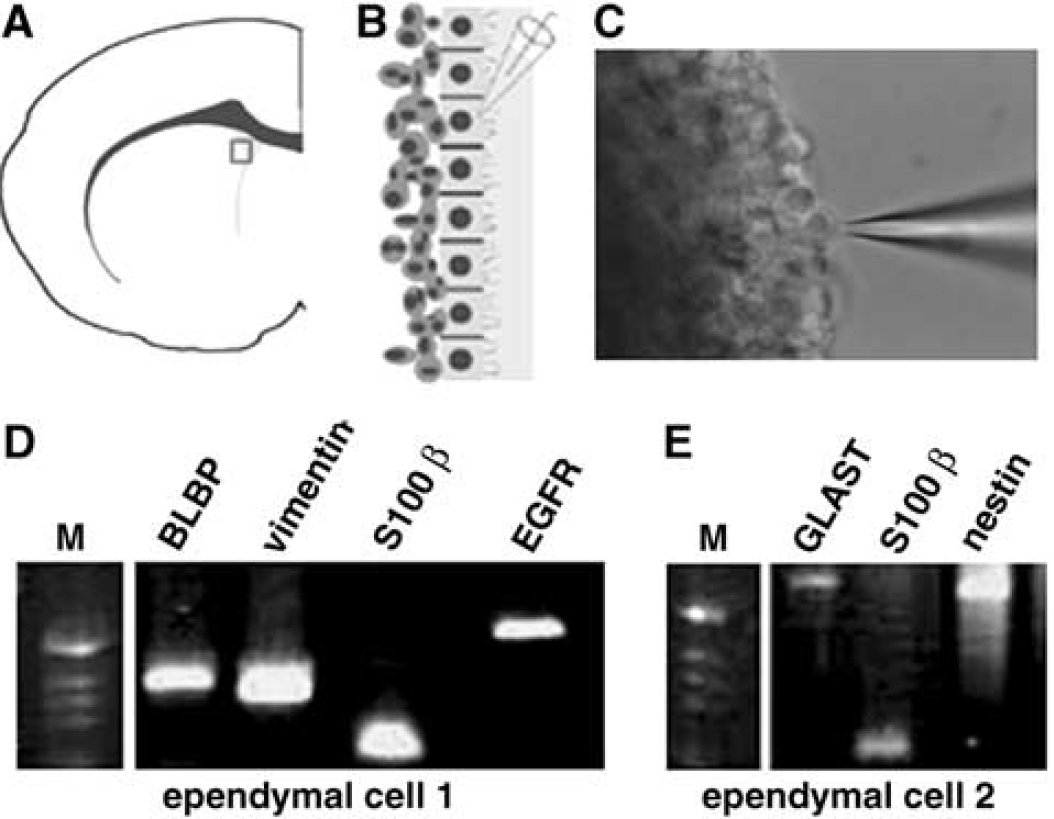
Gene expression in ependymal cells after stroke. (
We also detected 4A4+ cells in the SVZ 2 days after stroke (Figure 2C, arrowhead). Seven days after stroke, 4A4+ cells were detected in the ipsilateral SVZ, but not in the ependymal layer (Figure 2G and Figure 2H). These cells had a significantly lower percentage co-expression with HH3 (38.1% ± 11.5%, P < 0.01, n = 8; Figure 2G), than the population in the ependymal layer of 1 to 2 days after stroke. Morphologically, these 4A4+ cells in the ipsilateral SVZ exhibited a short or no basal process (Figure 2G and Figure 2H), suggesting that this population of 4A4+ cells may differ from 4A4+ cells detected in ependymal cells.
Stroke Induces Ependymal Cells Expressing Molecules with Characteristics of Radial Glial Cells
Radial glia express the astrocyte-specific glutamate transporter (GLAST) and brain-lipid-binding protein (BLBP) (Shibata et al, 1997; Hartfuss et al, 2001). Epidermal growth factor receptor signaling stimulates ependymal cells to exhibit radial glial phenotype (Gregg and Weiss, 2003). Given our observation that some ependymal cells proliferate and have radial glial cell phenotype (4A4) after stroke, we asked whether ependymal cells express GLAST, BLBP, and EGFR. Single-cell multiplex RT-PCR was performed from single randomly isolated ependymal cells, which permits analysis of the expression of several genes at a single-cell resolution (Palma et al, 2005). Using a patch-clamp pipette, we localized an ependymal cell based on its cuboidal morphology with cilia facing the ventricular lumen in a coronal brain slice and isolated RNA from single ependymal cell (Figure 4A–Figure 4C). All sampled ependymal cells expressed S100β, which is consistent with the fact that ependymal cells express S-100β (Bruni, 1998; Gregg and Weiss, 2003). In non-stroke rats, one of 13 (8%) sampled ependymal cells expressed GLAST, and nestin, 12 of the 13 cells (92%) expressed BLBP, and none of the cells expressed EGFR. However, in stroke rats, 2 of 8 (25%) sampled ependymal cells expressed GLAST (Figure 4E), 3 of the 8 cells (38%) expressed nestin (Figure 4E), 1 of the 8 cells (13%) expressed EGFR (Figure 4D), and 7 of the 8 cells (88%) expressed BLBP (Figure 4D). The frequency of BLBP expression was not substantially altered as expected, since ependymal cells express BLBP (Sundholm-Peters et al, 2004). To quantify BLBP expression, we isolated ependymal and SVZ cells from rats (n = 3) 2 days after stroke by means of laser capture microdissection (Liu et al, 2006). Real-time RT-PCR was performed to measure mRNA levels of BLBP. We found 2.3-fold increases of BLBP mRNA in the ipsilateral ependymal cells compared with mRNA levels in contralateral ependymal cells. In contrast, BLBP mRNA levels in the ipsilateral SVZ cells only increased 0.4-fold compared with levels in contralateral SVZ cells. These data suggest that stroke increases BLBP levels in ependymal cells. To further confirm our findings of mRNA in ependymal cells, immunostaining was performed. In non-stroke rats, we found that the S100β+ ependymal cell population was present as a single-cell layer on the surface of the lateral ventricle, with cuboidal morphology (Figure 1A and Figure 1B). After stroke, some ependymal cells extended a single basal process away from the ventricular surface (Figure 1D and Figure 1E). Nestin is an intermediate filament and is expressed in radial glial cells (Gotz and Barde, 2005). Nestin+ cells were detected in the SVZ but not in the ependymal layer of non-stroke rats (Figure 5A), which is consistent with data that SVZ cells express nestin (Ernst and Christie, 2005). However, stroke induced nestin+ ependymal cells (Figure 5B) and some of these cells extended a single, long thin basal process towards the striatum (Figure 5C and Figure 5E) and were also S100β+ (Figure 5D and Figure 5E), indicating that in addition to their morphology resembling to developmental radial glia, these ependymal cells express radial glial cell antigens. Furthermore, double immunostaining of vimentin, the main intermediate filament protein in radial glia (Alvarez-Buylla et al, 2001), and GFAP showed that vimentin+ ependymal cells had a long thin process directed away from the ipsilateral ventricular surface (Figure 5F, arrows, and Figure 5G), and these cells were not GFAP+ (Figure 5F and Figure 5G), which is consistent with fact that radial glia in rodent do not express GFAP. Some of vimentin+ ependymal cells were HH3+ (Figure 5H). Finally, stroke induced GLAST+ ependymal cells (Figure 5I and Figure 5J). Collectively, these data indicate that stroke induces adult ependymal cell expression of the radial glial antigen.
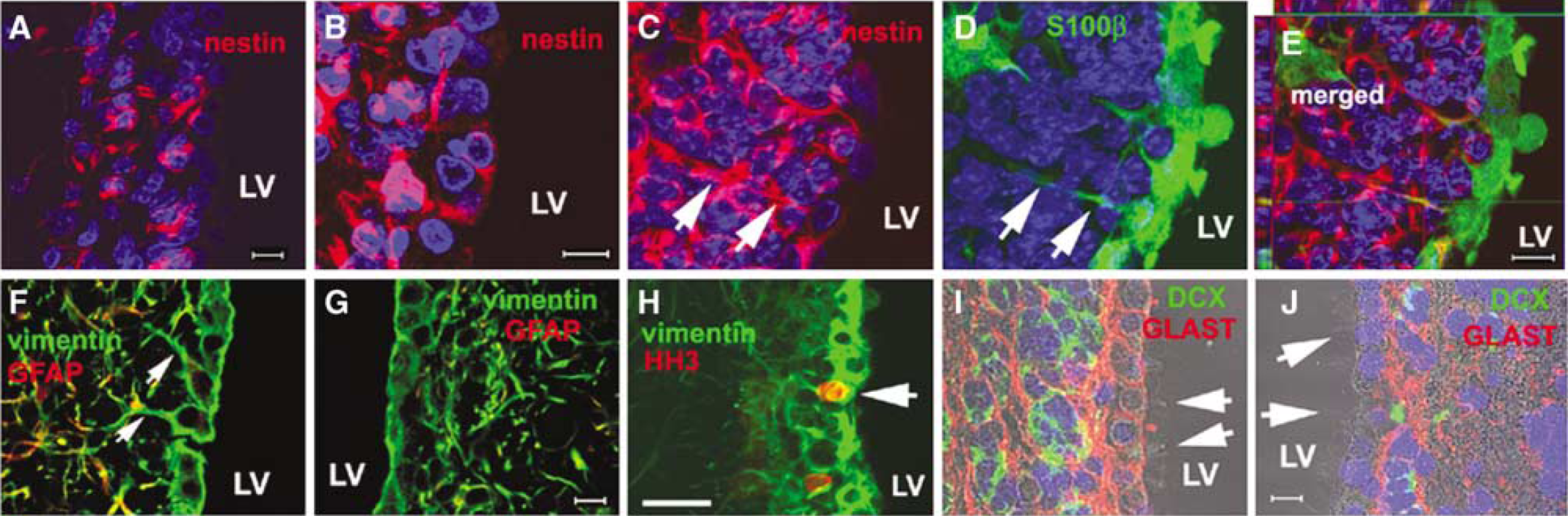
Ischemic stroke induces ependymal cell expression of GLAST, nestin, and vimentin. Confocal microscopic images show that nestin was expressed in ependymal cells after stroke (
Increases of Radial Glial Cells Parallel Expansion of Neuroblasts in the Subventricular Zone after Stroke
During cortical development, radial glial cells are the predominant form of neural progenitors cells, giving rise to both neurons and the secondary progenitors that populate the SVZ (Fishell and Kriegstein, 2005; Gotz and Barde, 2005). In the adult SVZ, radial glial cells produce neural stem cells (type B cells), transit-amplifying cells (type C cells), and neuroblasts (type A cells) (Doetsch et al, 1999). We found that 4A4+ cells were localized within a cluster filled by dark cells distributed near the surface of the lateral ventricle and some of these cells were mitotic 2 (Figure 6A) and 4 (Figure 6C) days after stroke, suggesting that proliferating cells are closely associated with radial glial cells. The number of cells within a cluster dramatically increased at 2 (11 ± 3.9 per cluster; Figure 6B), 4 (25 ± 6.7 per cluster; Figure 6D), and 7 (43 ± 11.1 per cluster; Figure 6F) days after stroke, which was concurrent with increases of the number of BrdU+ (Figure 6E, red). Many of BrdU+ cells were DCX+ 4 days after stroke (Figure 6E, arrows) and 4A4+ cells were detected within a cluster filled by many DCX+ cells (Figure 6G and Figure 6H). In addition, asymmetric inheritance of a radial glial fiber by one daughter of 4A4+ cells was detected along the lateral wall of the lateral ventricle (Figure 6I). These data underline the critical role of radial glial cells in neuronal production of adult brain after stroke.
Radial glial cells are closely localized to proliferating SVZ cells. 4A4+ cells (
Radial Glial Cells Support Neuroblast Migration towards the Ischemic Boundary
Four days after stroke, many 4A4+ cells were detected in the ischemic SVZ and striatum (Figure 2H and Figure 6C). We and others have shown that stroke induces neuroblasts in the SVZ to migrate towards the ischemic boundary region (Arvidsson et al, 2002; Parent et al, 2002; Jin et al, 2003; Zhang et al, 2004b). To examine whether these radial glia support migration of neuroblasts, double immunostaining with antibodies against DCX, a marker of migrating neuroblasts, and 3cb2, a marker of radial glia, was performed. Confocal images revealed that there were many 3cb2 positive fibers in the ischemic SVZ and striatum (Figure 7A) and some of the 3cb2 processes were closely associated with leading processes of DCX+ cells orientated towards the ischemic boundary region (Figure 7A–Figure 7C), indicating that radial glial process might support neuroblast migration towards the ischemic boundary. To further investigate function of radial glia in supporting neuroblast migration, we performed in vitro migration assay to measure radial glial processes and neuronal migration by means of time-lapse microscopy. Two neurospheres derived from SVZ cells were plated in a matrigel containing the growth medium. Processes extended from one neurosphere to reach the other one. Some cells from one neurosphere migrated along a thin process to reach the adjacent neurosphere (Figure 7D–Figure 7H; Supplementary movie 1). Immunofluorescent staining revealed that the thin processes were nestin+ (Figure 7I and Figure 7J, arrow) and GLAST+ (Figure 7K and Figure 7L, arrow), while cells that migrated along thin processes were TuJ1+ (Figure 7L, arrowhead). These in vitro findings are consistent with previous reports that radial glia guide neuronal migration (Edmondson and Hatten, 1987; Gregg and Weiss, 2003).
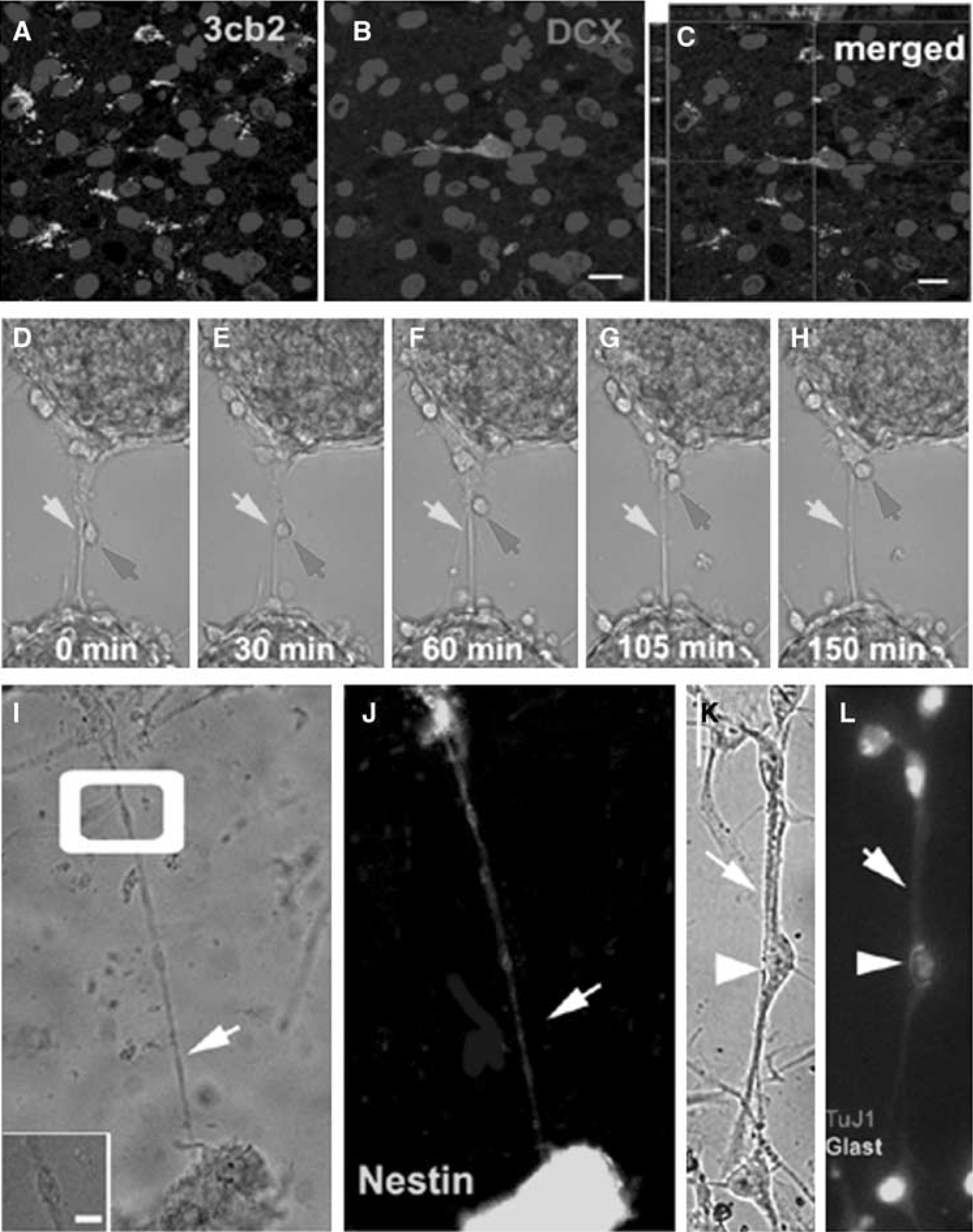
Radial glial cells support neuron migration. Confocal microscopic images (
Discussion
The present study shows that stroke acutely induced adult ependymal cell proliferation and that dividing ependymal cells of the lateral ventricle had genotype, phenotype, and morphology of radial glial cells. In addition, these radial glial cells were closely localized to actively proliferating type A (neuroblasts) and C cells in the ipsilateral SVZ. Furthermore, radial glial fibers in the ipsilateral SVZ and striatum were associated with leading processes of migrating neuroblasts oriented to the ischemic boundary, and time-lapse data revealed that radial glia supported neuronal migration. Together, these results suggest that in the ischemic brain of the adult rat, radial glial cells are induced and ependymal cells can proliferate and transform into radial glial cells.
Adult ependymal cells are postmitotic and their proliferative capacity remains controversial (Johansson et al, 1999; Spassky et al, 2005). Administration of BrdU over 2 to 6 weeks results in labeling dividing ependymal cells in the adult mouse (Johansson et al, 1999). However, after infusion of [3H]thymidine for 6 weeks, Spassky et al (2005) failed to detect adult ependymal cell division in the lateral wall of the lateral ventricles. Using a single pulse and cumulative BrdU labeling, we did not detect BrdU labeled ependymal cells of the lateral wall of the lateral ventricles in the non-stroke adult rats. In contrast, BrdU labeled ependymal cells in the lateral wall of the ipsilateral lateral ventricle were detected 1 to 2 days after stroke. Subventricular zone astrocytes (type B cells) are in contact with the ventricular surface and dividing ependymal cells have been identified on thick coronal sections as astrocytes (Spassky et al, 2005). Using confocal microscopy, we found that BrdU labeled cells in the lateral wall of the ipsilateral lateral ventricle were cuboidal, ciliated, S100β positive, but GFAP negative. Therefore, these results strongly indicate that adult ependymal cells proliferate in response to stroke.
The most intriguing finding of the present study is that stroke-induced dividing adult ependymal cells transform into radial glial cells on the basis of genotype, phenotype, and morphology. This finding can be placed into context with recent studies that showed the lineage relationship between radial glia and ependymal cells. Ependymal cells originate from radial glia between E 14 and E16 (Spassky et al, 2005). Intraventricular infusion of EGF promotes adult ependymal cells adapting radial glial cell morphology, although radial glial cells are not normally present in the adult brain (Gregg and Weiss, 2003). We found that dividing ependymal cells expressed cdc2-kinase phosphorylated form of vimentin (4A4), a radial glial-specific mitotic marker (Kamei et al, 1998), 1 to 2 days after stroke, and 82% of HH3 positive cells were 4A4 positive, which is comparable to observations in embryonic VZ of the human (92%) and mouse (99%). Moreover, these 4A4 positive cells had radial glial morphology and retained a radial fiber throughout mitotic phases, as has been previously reported in embryonic VZ of the rat (Weissman et al, 2003). During embryonic stages, ependymal cilia on the walls of the lateral ventricles gradually develop from E14 to P4, when radial glial cell transform to ependymal cells (Spassky et al, 2005). Interestingly, we observed that some of 4A4 positive ependymal cells exhibited fewer cilia than adjacent 4A4 negative ependymal cells, suggesting that these adult ependymal cells recapture morphology of embryonic ependymal cells. Tanycytes wedged between ependymal cells are unciliated and occupy approximately 0.6% cell population of the wall of the lateral ventricle (Doetsch et al, 1997). We found that many 4A4 positive cells were present on the lateral wall of the lateral ventricle after stroke and these cells had cilia. Although single isolated ependymal cells that express GLAST and nestin may not be those cells that are 4A4 positive cells, increases of GLAST and nestin immunoreactive ependymal cells and upregulation of BLBP in ischemic ependymal cells suggest that these specific radial glial molecules are associated with morphological transformation of adult ependymal cells to radial glial cells. Thus, 4A4 positive cells are unlikely to be tanycytes. Together, we show here that stroke stimulates adult ependymal cells to transform into radial glial cells, suggesting that the niche to generate radial glia during embryonic development is retained in the ependymal layer of the lateral ventricle of the adult rat.
In addition to the ependymal layer, we observed radial glial cells in the ipsilateral SVZ and striatum during 2 to 7 days of stroke and these 4A4 positive radial glial cells exhibited a short or no basal process. Moreover, many 3Cb2 positive cells with radial like fibers were detected in the SVZ and striatum. Infusion of EGF or TGFα induces radial glial cells in the SVZ and striatum of adult mouse and EGF stimulates neural stem cells to give rise radial glial cells in vitro (Gregg and Weiss, 2003). Thus, these findings suggest that radial glial cells detected in ipsilateral SVZ and striatum could be generated by neural stem cells. Further studies are warranted to dissect the various sources and the relative cell contributions to radial glial cells in adult brain after stroke.
Radial glial cells are a major source of neurons during development (Miyata et al, 2001; Noctor et al, 2001; Gotz and Barde, 2005). Our results suggest that radial glial cells transformed from ependymal cells may promote endogenous generation of new neurons to replenish ischemic damaged neurons in the adult brain. During cortical neurogenesis, clusters of neural progenitors in the VZ are organized around single radial cells (Bittman et al, 1997). In adult stroke brain, we found that single radial glial cells were surrounded by transit-amplifying (type C cells) and neuroblasts (type A cells), indicating that radial glial cells serve as neural progenitors to produce type C cells that differentiate into neuroblasts early after the onset of stroke. In addition, we show here that the majority of radial glial cells exhibited symmetric divisions according to the cleavage plane (vertical to the surface of the lateral ventricle), which is consistent with our previous findings that stroke-induced neurogenesis at least partially results from transient increases of symmetrical cell division of neural progenitor cells (Zhang et al, 2004a). However, by observing an asymmetric radial fiber associated with one daughter radial glial cell during vertical divisions in the rat E12 to E18, Weissman et al (2003) suggest that some of VZ cell divisions occurring in the vertical cleavage plane are asymmetric or result in neuronal production. In parallel, we detected that a radial glial fiber was selectively associated with one daughter cell during vertical cell divisions in the adult stroke brain. Therefore, according to the morphology of daughter cells, radial glial cells are considered to undergo asymmetric divisions.
Radial glial cells serve to guide neurons to their destination (Noctor et al, 2001). Neuroblasts generated in the SVZ migrate to the olfactory bulb via the rostral migratory stream (Luskin, 1993; Lois and Alvarez-Buylla, 1994). However, stroke redirects neuroblasts in the SVZ to migrate towards the ischemic boundary region (Arvidsson et al, 2002; Parent et al, 2002; Jin et al, 2003; Zhang et al, 2004a, b ). The present study shows that some migrating neuroblasts within the ischemic boundary were closely associated with 3cb2 positive radial fibers in the ipsilateral striatum. Time-lapse microscopy analysis revealed that processes generated from stroke neurospheres supported neuronal migration. Many factors, such as stromal cell-derived factor 1 α and matrix metalloproteinases promote neuroblast motility after stroke (Wang et al, 2003; Imitola et al, 2004; Robin et al, 2006). The present findings indicate that stroke-induced radial glial cells guide migrating neuroblasts to the ischemic boundary region.
In summary, our findings show that adult forebrain ependymal cells generate radial glial cells in response to stroke, which could function as neural progenitor cells and as guide-wires for migration. Therefore, strategies to promote endogenous regeneration of radial glial cells may foster brain repair during stroke recovery.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
