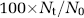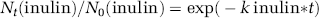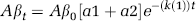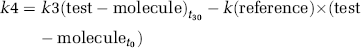Abstract
Amyloid β-peptide (Aβ) clearance from the central nervous system (CNS) maintains its low levels in brain. In Alzheimer's disease, Aβ accumulates in brain possibly because of its faulty CNS clearance and a deficient efflux across the blood—brain barrier (BBB). By using human-specific enzyme-linked immunosorbent assays, we measured a rapid 30 mins efflux at the BBB and transport via the interstitial fluid (ISF) bulk flow of human-unlabeled Aβ and of Aβ transport proteins, apolipoprotein E (apoE) and apoJ in mice. We show (i) Aβ40 is cleared rapidly across the BBB via low-density lipoprotein receptor-related protein (LRP)1 at a rate of 0.21 pmol/min g ISF or 6-fold faster than via the ISF flow; (ii) Aβ42 is removed across the BBB at a rate 1.9-fold slower compared with Aβ40; (iii) apoE, lipid-poor isoform 3, is cleared slowly via the ISF flow and across the BBB (0.03–0.04 pmol/min g ISF), and after lipidation its transport at the BBB becomes barely detectable within 30 mins; (iv) apoJ is eliminated rapidly across the BBB (0.16 pmol/ming ISF) via LRP2. Clearance rates of unlabeled and corresponding 125I-labeled Aβ and apolipoproteins were almost identical, but could not be measured at low physiologic levels by mass spectrometry. Amyloid β-peptide 40 binding to apoE3 reduced its efflux rate at the BBB by 5.7-fold, whereas Aβ42 binding to apoJ enhanced Aβ42 BBB clearance rate by 83%. Thus, Aβ, apoE, and apoJ are cleared from brain by different transport pathways, and apoE and apoJ may critically modify Aβ clearance at the BBB.
Introduction
Amyloid β-peptide (Aβ) accumulation in brain and its neuronal toxicity contribute to the pathogenesis and progression of Alzheimer's disease (AD) (Hardy and Selkoe, 2002; Zlokovic, 2005). Recent studies from our and other laboratories suggest a major role of Aβ clearance in determining Aβ concentration in the central nervous system (CNS) (Selkoe, 2001; Zlokovic, 2004, 2005; Tanzi et al, 2004; Holtzman and Zlokovic, 2006). In particular, bidirectional transport of soluble-free Aβ across the blood—brain barrier (BBB) via low-density lipoprotein receptor-related protein-1 (LRP1) (Shibata et al, 2000; Deane et al, 2004) and receptor for advanced glycation end products (Mackic et al, 1998a; Deane et al, 2003), binding of Aβ to apolipoprotein E (apoE) (Martel et al, 1997) and apoJ (Zlokovic et al, 1996), Aβ metabolism (Selkoe, 2001; Iwata et al, 2000, 2001), and degradation by astrocytes (Wyss-Coray et al, 2003; Koistinaho et al, 2004) may influence Aβ transport exchange at the BBB and/or its clearance from the CNS.
A microdialysis technique has been developed to measure transport exchange of an endogenous soluble pool of human Aβ in brain interstitial fluid (ISF) in different transgenic models of AD (Cirrito et al, 2003). However, the BBB bidirectional transport of exogenous soluble human Aβ (Zlokovic et al, 1993; Maness et al, 1994; Ghilardi et al, 1996; Ghersi-Egea et al, 1996; Poduslo et al, 1997; Mackic et al, 1998b; Deane et al, 2003, 2004, 2005; Banks et al, 2003; LaRue et al, 2004) and the BBB influx of its circulating binding transport proteins (Zlokovic et al, 1996; Martel et al, 1997) have been frequently studied with 125I-radiolabeled tracers. However, the clearance transport pathways of unlabeled exogenous Aβ and apolipoproteins from brain ISF have not been studied.
Here, we modified our well-established clearance technique in mice (Shibata et al, 2000; Deane et al, 2004, 2005) to determine transport routes and clearance rates across the BBB and via the ISF bulk flow of unlabeled synthetic human Aβ40 and Aβ42 and of Aβ transport proteins, human recombinant apoE (isoform 3) and native human plasma-derived apoJ. To determine clearance of unlabeled test-molecules from the CNS, we used human-specific enzyme-linked immunosorbent assays (ELISAs). We have also explored the feasibility of matrix-assisted laser desorption ionization time-of-flight (MALDI-TOF) mass spectrometry (MS) and liquid chromatography (LC) electrospray ionization mass spectrometry (MS/MS) to determine clearance of unlabeled Aβ and apolipoproteins from the CNS. Finally, we compared clearance rates of unlabeled and 125I-labeled Aβ and apolipoproteins. Our data indicate Aβ, apoE, and apoJ are cleared from brain by different transport routes, and binding of Aβ to apoE and apoJ may critically influence Aβ efflux at the BBB and its clearance from brain.
Materials and methods
Aβ and Apolipoproteins
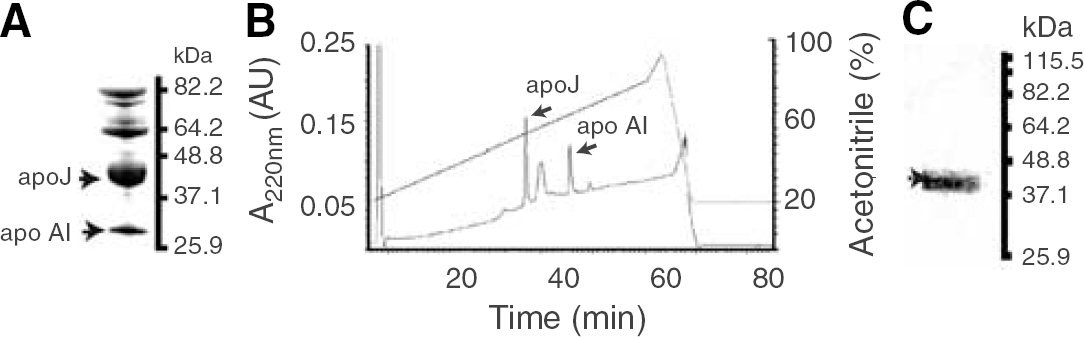
Human Aβ40 and Aβ42 were synthesized and characterized as we described (Deane et al, 2004). Affinity-purified crude extract of human plasma apoJ (Figure 1A) was purified by high-pressure LC (HPLC) (Figure 1B), as described (de Silva et al, 1990; Zlokovic et al, 1996). The Tris/Tricine sodium dodecyl sulfate-polyacrylamide gel electrophoresis (10%–20%) shows a single apoJ band consisting of α and β chain (Figure 1C), confirmed as apoJ by MALDI-TOF (not shown). Recombinant human apoE (isoform 3) was from baculovirus-transfected Sf9 cells purchased from Invitrogen (Carlsbad, CA, USA, Cat. No. P2003). Lipidated apoE (isoform 3) was prepared from conditioned media of immortalized mouse astrocytes, as we described (Morikawa et al, 2005). Lipid-poor apoE complex with synthetic human Aβ40 was prepared as we described (Martel et al, 1997) and purified by the size exclusion fast protein LC to remove excess Aβ. Amyloid β-peptide 42–apoJ complex was prepared as we described (Zlokovic et al, 1996) and purified over a fast protein LC column before use. Within the 30 mins of Aβ–apoE and Aβ–apoJ complex administration into brain ISF, Aβ in phosphate-buffered saline-soluble brain supernatant fraction co-immunoprecipitates with anti-apoE (3D12, Biodesign International, Sako, ME, USA, Cat. No. H61529M) or anti-apoJ (A241, Quidel, San Diego, CA, USA) antibody >98% and 99%, respectively, indicating that the peptide remains stable in the form of complexes with apolipoproteins over the period required for clearance measurements.
(
Clearance Technique
Mice were kept under standard housing conditions and feeding schedules until experimental procedures were performed. All studies were performed according to the National Institutes of Health guidelines using an approved institutional protocol. Briefly, a stainless-steel guide cannula was implanted stereotaxically into the right caudate—putamen of anesthetized C57B16 mice (100 mg/kg ketamine and 10 mg/kg xylazine intraperitoneally) with the cannula tip coordinates 0.9 mm anterior and 1.9 mm lateral to the Bregma and 2.9 mm below the surface of the brain. Animals were recovered after surgery before clearance studies. The experiments were performed before substantial chronic process occurred, as assessed by histologic analysis of tissue, that is, negative staining for astrocytes (glial fibrillar acidic protein) and activated microglia (anti-phosphotyrosine), but allowing time for the BBB repair for large molecules, as reported previously (Cirrito et al, 2003; Deane et al, 2004, 2005).
The test-molecules, unlabeled synthetic human Aβ40 and Aβ42, human apoE (isoform 3) non-lipidated and lipidated, and human native apoJ were administered into brain ISF simultaneously with 14C-inulin (reference molecule) over 5 mins using our clearance technique (Shibata et al, 2000). The test-molecules were administered in 0.5 μL mock ISF at equimolar concentrations of 40 fmol/μL corresponding to 0.173 and 0.181 ng/μL of Aβ40 and Aβ42, respectively, 1.40 ng/μL of apoE, nonlipidated or lipidated, and 3.20 ng/μL of apoJ. When the effects of anti-LRP1 (N20, polyclonal goat anti-human antibody that crossreacts with mouse LRP1; Santa Cruz Biotech. Inc., Santa Cruz, CA, USA), anti-LRP2 (LRP2; Rb 6286, from Dr S Argraves), and control non-immune immunoglobulin G (murine serum immunoglobulin G, Sigma, St Louis, MO, USA) on Aβ or apoJ clearance were studied, these were infused at concentrations of 60 μg/mL 10 mins before and then simultaneously with the test-molecules. Receptor-associated protein (RAP) (Oxford Biomed Research, Oxford, MI, USA), which blocks both LRP1 (Shibata et al, 2000; Deane et al, 2004) and LRP2 (Zlokovic et al, 1996), was infused at 5 μmol/L 10 mins before and then simultaneously with the test-molecules. Clearance of Aβ, apoE, and apoJ was determined focally in brain by using human-specific ELISAs (see below). At predetermined times, which in most experiments was within 30 mins of intracerebral administration, brain and blood were sampled and prepared for Aβ, apoE, and apoJ ELISAs. At the time of CNS administration, unlabeled Aβ40 and Aβ42 were negative for Aβ oligomers and Aβ aggregates, as shown by negative dot-blot analyses with Aβ-oligomer-specific (A11) and Aβ-aggregate-specific (OC) antibodies obtained from Dr Charles Glabe (data not shown).
In studies with 125I-labeled test-molecules, the amount of injected tracers was determined using a micrometer to measure the linear displacement of the syringe plunger in the precalibrated microsyringe. Tracer fluid (0.5 μL) containing 125-labeled test-molecules (e.g., Aβ, apolipoproteins) and 14C-inulin (reference molecule) was injected over 5 mins. To compare directly the clearance rates of 125I-labeled
Aβ and apolipoproteins with the corresponding unlabeled Aβ and apolipoproteins, we administered exactly the same amounts of 125I-labeled Aβ40 (0.173 ng/μL), Aβ42 (0.181 ng/μL), apoE (1.40 ng/μL) nonlipidated and lipidated isoform 3, and apoJ (3.20 ng/μL), as in the experiments with unlabeled Aβ and apolipoproteins. Brain and blood were sampled and prepared for radioactivity analysis as described (Shibata et al, 2000; Deane et al, 2005). Clearance of Aβ, apoE, and apoJ was determined by the radioactivity analysis (see below).
Detection of Clearance by Human-Specific Enzyme-Linked Immunosorbent Assays
Briefly, about 15 mg of brain tissue adjacent to the site of microinjection was homogenized with an extraction buffer containing 50 mmol/L NaCl, 0.2% diethylamine and complete protease inhibitor cocktail (Roche, Indianapolis, IN, USA). Diethylamine was used as it improves Aβ recovery from tissue homogenates, reduces background signal, and requires low dilution before ELISA determination (Schmidt et al, 2005). This allows for quantification of typically low levels of soluble Aβ found in tissues without plaques (Best et al, 2005; Deane et al, 2005). Samples were centrifuged for 1 h at 100,000 g, and supernatant (pH 8.0) diluted 1:1 before adding to ELISA plates. Amyloid β-peptide 1–40 and 1–42 colorimetric kits (Invitrogen, KHB3441 and KHB3544) were used to determine Aβ40 and Aβ42 levels in brain extract and plasma, respectively. These kits detect whole intact Aβ 1–40 and 1–42 molecules and do not recognize Aβ fragments. For apoE and apoJ, polysterene microtiter plates were coated with 10 μg/mL monoclonal anti-apoE antibody (3D12) or polyclonal anti-apoJ antibody (Chemicon, Temecula, CA, USA), respectively, overnight at 4°C. After blocking with 3% bovine serum albumin for 1 h at room temperature, standards and samples were added and incubated for 2 h. Bound apoE and apoJ were detected by affinity-purified biotinylated goat anti-apoE (Biodesign International, Cat. No. K74180B) and monoclonal anti-apoJ (Quidel, Cat. No. A241) antibodies, respectively, followed by streptavidin—horseradish peroxidase conjugate (Invitrogen, Cat. No. SNN2004) or anti-mouse-horseradish peroxidase conjugate (Sigma, Cat. No. A4789). The reaction was developed for 30 mins using tetramethyl benzidine substrate and stopped with 1
Detection of Clearance with Iodinated Ligands
Amyloid β-peptide 40 and Aβ42 were iodinated using a mild lactoperoxidase method (LaRue et al, 2004). Radiolabeled peptides were HPLC purified to eliminate free iodide, diiodinated Aβ, oxidized Aβ, or unlabeled Aβ, as we reported (Deane et al, 2003; LaRue et al, 2004). For clearance studies, we used only reduced monoiodinated Aβ peak (specific activity ~60 μCi/μg), as confirmed by MALDI-TOF MS analysis, as reported (LaRue et al, 2004). ApoJ and apoE were labeled by Iodo-Gen (Pierce, Rockford, IL, USA, Cat. No. 28600) to specific activity of 9–12 μCi/μg. Free iodide was removed from radiolabeled apoE and apoJ preparations by gel filtration. All 125I-labeled molecules were used within 24 h of labeling to avoid radiolysis, as we described (LaRue et al, 2004). The trichoracetic acid (TCA)-precipitable 125I-radioactivity and the non-TCA precipitable 125I-radioactivity (e.g., degraded Aβ) were determined, as we reported (Shibata et al, 2000; Deane et al, 2003, 2004). Our earlier studies with 125I-labeled Aβ have shown that both radiolabeled Aβ40 and Aβ42 remain mainly intact in brain ISF (>95%) within 30–90 mins of in vivo clearance studies (Shibata et al, 2000), as well as during rapid clearance studies in vitro on brain capillaries (Deane et al, 2004), as shown by the TCA analysis and confirmed by the HPLC and sodium dodecyl sulfate-polyacrylamide gel electrophoresis analyses. In the present study, the TCA, HPLC, and sodium dodecyl sulfate-polyacrylamide gel electrophoresis/immunoprecipitation analysis confirmed previous findings, indicating that molecular forms of transport of 125I-labeled Aβ and apolipoproteins within 30 mins of clearance studies remained mainly in their original form of intact molecules (>98%), as injected in the CNS.
Matrix-Assisted Laser Desorption Ionization Time-of-Flight and Liquid Chromatography Electrospray Ionization Mass Spectrometry/Mass Spectrometry
To determine the limit of quantification (LOQ) for human Aβ by MALDI-TOF MS, we spiked mouse CSF with various amounts of human synthetic Aβ40 (0.1–30 ng/spot) and a constant amount of metabolically labeled 15N-Aβ40 (internal standard; 30 ng/spot; R-peptide, Atlanta, GA, USA), as reported (Gelfanova et al, 2006). Samples were mixed with an equal volume of α-cyano-4-hydroxycinnamic acid in 50% acetonitrile and spotted onto the MALDI target using a MALDI-TOF MS (Voyager-DE STR BioSpectrometry Applied Bioscience, Foster City, CA, USA). Spectra were obtained from 15 separate areas of the spot with 200 laser shots per acquisition. All acquisitions were averaged for each spot.
For LC electrospray ionization MS/MS analysis, 15N-Aβ40 was digested into tryptic peptides and applied to a microcapillary LC system (Surveyor MS Pump Plus HPLC system Thermo Corporation, San Jose, CA, USA) coupled to the linear ion-trap mass spectrometer (Thermo Corporation, San Jose, CA, USA). An in-line analytical capillary column (75 μm i.d. 10 cm) was packed using C18 reversed-phase resin (5 μm, 200 Å Magic C18AG, Michrom BioR-esource, Auburn, CA, USA) and Picofrit capillary tubing (75 μm i.d. 10 cm, New Objective, Cambridge, MA, usa). Each sample was first concentrated and de-salted by loading in solvent A (0.1% acetic acid in solution of 5% acetonitrile and 95% water) for 20 mins.
Peptides were eluted using a linear gradient of 5–70% solvent B (to a final working concentration 0.1% acetic acid in solution of 95% acetonitrile and 5% water over 70 mins, followed by isocratic elution at 95% solvent B for 10 mins, to wash the column, with a flow rate of 0.200 μL/min across the column. Peptides eluting from the capillary column were automatically selected for collision-induced dissociation by the mass spectrometer using a data-dependent protocol that alternated between one MS scan and seven MS/MS scans for the seven most abundant precursor ions detected in the MS survey scan. Precursor m/z values, selected for collision-induced dissociation using a collision energy setting of 35%, were dynamically excluded for 30 secs after selection. The mass range for precursor ion detection was set from 400 to 2000 Da. The electrospray voltage was set to 2.1 kV. The operation of the mass spectrometer was controlled by Xcalibur LC/MS/MS software (Thermo Corporation, San Jose, CA, USA). The MS/MS spectra were sequence database searched using a database that had been indexed using 15N modifications via SEQUEST7 and Bioworks Browser (both from Thermo Corporation, San Jose, CA, USA). Default threshold cutoffs were made using the following parameters: normalized crosscorrelation score for +1, +2, and +3 charge peptides of 1.9, 2.7, and 3.5 respectively, and a DeltaCN value of 0.1. The MS/MS spectra were searched against a downloaded nonredundant human proteome sequence database from European Bioinformatics Institute (http://www.ebi.ac.uk/IPI/IPIhuman.html) to determine possible sequence correlations of known proteins for identification.
Mathematical Modeling
Transport clearance rates of unlabeled test-molecules, that is, Aβ, apoE, and apoJ, across the BBB and by the ISF bulk flow were determined from the respective ELISA measurements by using transport analysis similar to that as reported for radiolabeled test-molecules (Shibata et al, 2000; Deane et al, 2004). The concentrations of unlabeled test-molecules in the brain at time zero and at predetermined clearance times t were expressed in pmol/g ISF, assuming 1 g of brain contains 0.1 g of ISF (Zlokovic, 2005; Deane et al, 2005). Brain recovery of studied test-molecules and of simultaneously infused 14C-inulin (reference molecule) was calculated using
where Nt is the concentration of the test-molecule or the amount of inulin at the end of experiment at time t, and N0 is the concentration of a test-molecule or the amount of inulin injected in the brain ISF at time zero (Shibata et al, 2000). The fractional clearance rate constant of inulin (k, min−1) provides measure of the ISF bulk flow (Shibata et al, 2000), and is calculated as
In a case of Aβ multiple-time efflux series with departure of the later time points (> 30 mins) from the linear efflux phase, the concentration of unlabeled Aβ in brain ISF (pmol/g ISF) at time t (Aβt) is related to the concentration of Aβ at time zero (Aβ0) and the rates of Aβ total efflux from brain (k1) and its retention in brain (k2) by the biexponential equation,
where a1 = k2/(k1 + k2) and a2 = k1/(k1 + k2), and k1 and k2 are expressed in pmol Aβ/ming ISF, as reported for radiolabeled Aβ (Shibata et al, 2000). In a case of single-time point, rapid efflux series within the 30 mins of the linear efflux of test-molecules from the brain, the concentrations of unlabeled Aβ, apoE, and apoJ in the brain ISF (pmol/g ISF) at 30 mins (test-moleculet30) are related to the concentrations of the respective test-molecules at time zero (test-moleculet0) and the total rate of efflux (k3, pmol test-molecule/ming ISF) by the monoexponential equation,
as reported for radiolabeled Aβ (Deane et al, 2004). The clearance rates of Aβ, apoE, and apoJ across the BBB (pmol/ming ISF) were calculated from the single-time efflux series within 30 mins from the total efflux rates of test-molecules (k3, pmol/ming ISF; Equation (4)) and the fraction of test-molecules cleared via the ISF bulk flow determined by the clearance rate of inulin (reference molecule) given by
where test-moleculet0 is the initial concentration of the test-molecule injected into brain ISF at time zero, t0. The same equations were used to calculate clearance rates of radiolabeled test-tracers taking into account their respective concentrations, as we described (Shibata et al, 2000; Deane et al, 2004). In case of 125I-labeled Aβ, apoE, and apoJ, only 125I TCA-precipitable radioactivity was used to calculate the concentrations of intact tracers.
Statistical Analysis
Data are analyzed by multifactorial analysis of variance and Student's t-test. The differences were considered to be significant at P < 0.05. All values were mean ± s.e.m.
Results
Detection of Human Aβ, ApoE, and ApoJ Clearance by Human-Specific Enzyme-Linked Immunosorbent Assays
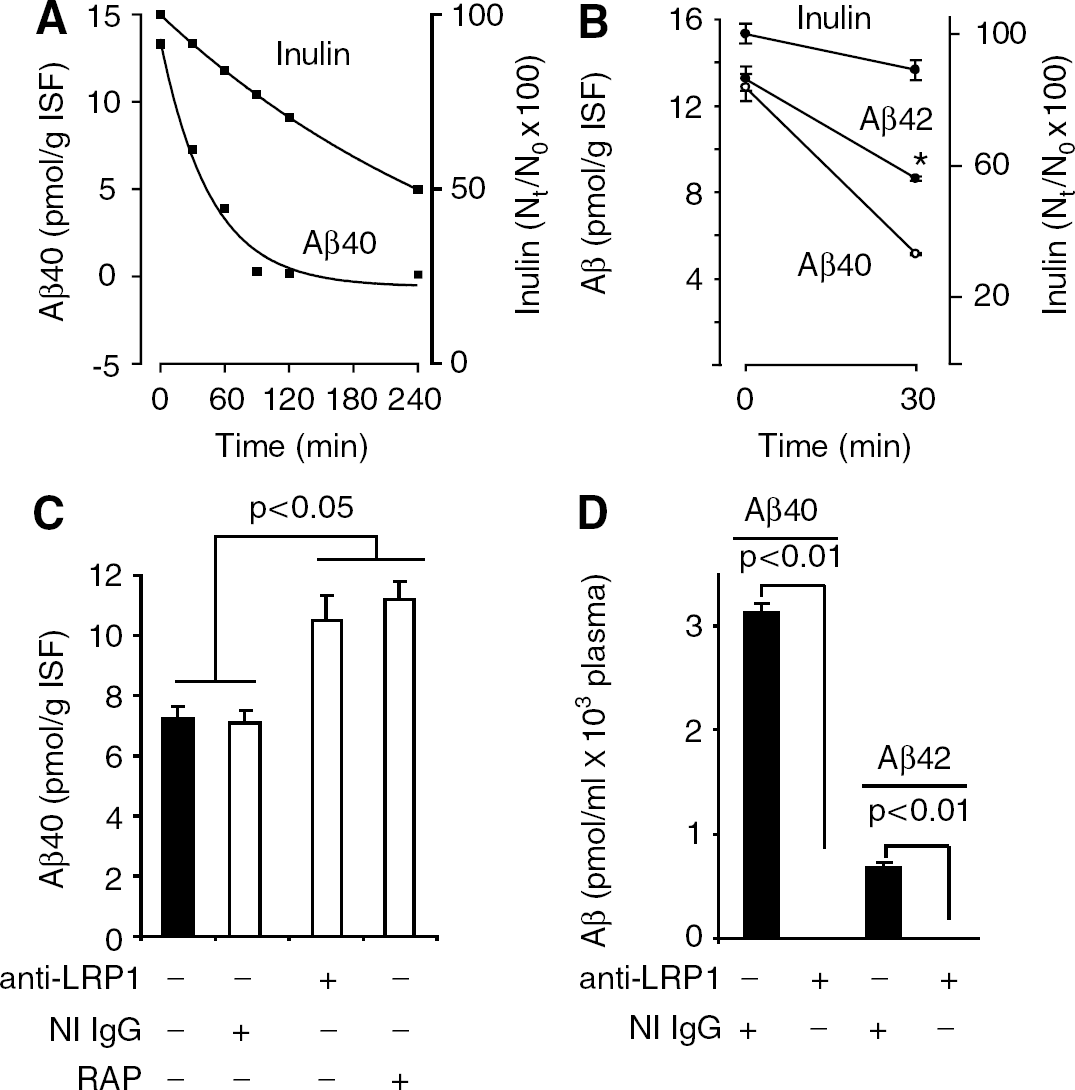
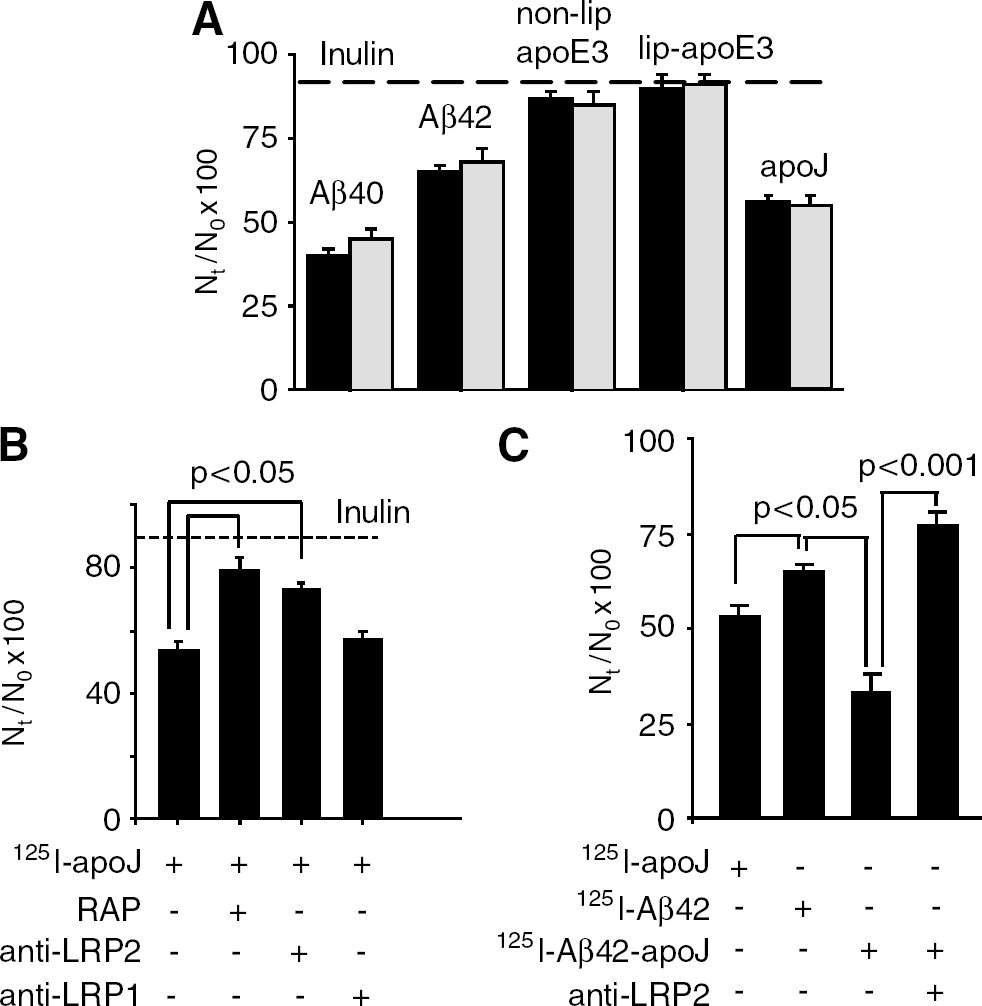
Figure 2A shows disappearance curve of human synthetic unlabeled Aβ40 from brain after its administration into brain ISF simultaneously with 14C-inulin over the studied period of time of 240 mins. There was time-dependent and rapid disappearance of Aβ40 from its initial levels at time zero of about 13 pmol/g ISF to almost undetectable levels at 90, 120, and 240 mins. In contrast to Aβ, 14C-inluin was cleared slowly from the brain via the ISF bulk flow (Shibata et al, 2000; Deane et al, 2004), as indicated by its high brain retention of 74% and 52% of the injected dose at 90 and 240 mins, respectively. The levels of human Aβ40 and Aβ42 at the beginning of the clearance study at time zero, Aβ0, were on average 12.8 and 13.2 pmol/g ISF, which was within the range of endogenous Aβ levels in the mouse brain, as reported (Deane et al, 2005) (Figure 2B) Both Aβ40 and Aβ42 levels decreased significantly to 5.16 and 8.64 pmol/g ISF within 30 mins, respectively, indicating clearance from the brain of both peptides, and significantly faster elimination of Aβ40. The decrease in inulin within 30 mins was ~10%, reflecting its passive transport via the ISF flow (Shibata et al, 2000; Deane et al, 2004). Figure 2C shows efflux of unlabeled Aβ at 30 mins is significantly inhibited (>70%) with centrally administered LRP1-specific antibody and RAP, but not with non-immune immunoglobulin G or anti-LRP2 (not shown). Moreover, intact human Aβ40 and Aβ42 were both present in mouse plasma within 30 mins of CNS administration, but were undetectable in plasma in the presence of centrally administered LRP1-specific antibody, as determined by the respective ELISAs (Figure 2D). These data confirm transcytosis of intact Aβ peptides across the BBB and indicate that Aβ efflux at the BBB is LRP1-dependent, as suggested by earlier work with radiolabeled Aβ (Shibata et al, 2000; Deane et al, 2004). It is of note that plasma levels of inulin were not significantly different form zero (not shown) consistent with its slow clearance via the ISF flow.
(
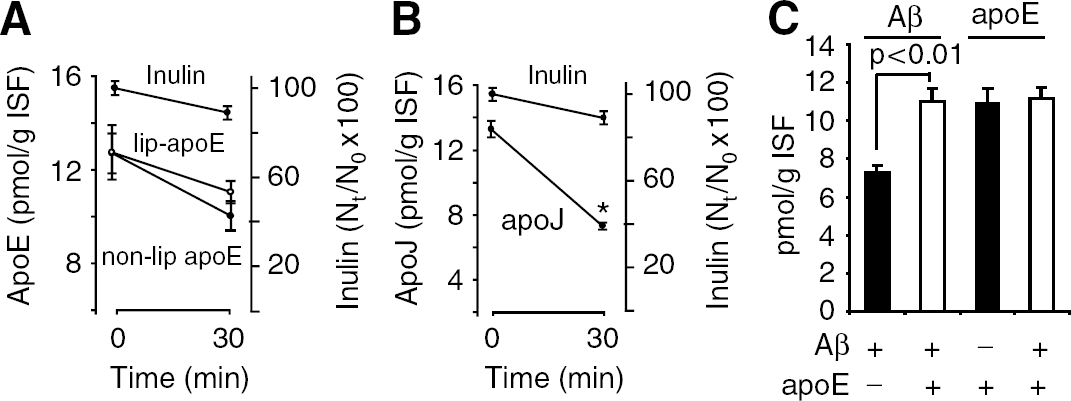
Figure 3A shows the levels of non-lipidated apoE (isoform 3) were slightly reduced from 13.1 pmol/g ISF at time zero to 10.9 pmol/g ISF at 30 mins. There was almost no difference in slope between inulin and lipidated apoE (isoform 3) within 30 mins of their simultaneous administration into brain ISF. In contrast, apoJ was cleared rapidly across the BBB as indicated by a steep decrease of its levels from 13.3 pmol/g ISF at zero time to 7.3 pmol/g ISF at 30 mins (Figure 3B). Figure 3C shows that Aβ40 clearance from its preformed complex with lipid-poor apoE is reduced substantially at 30 mins compared with clearance of free Aβ injected into brain ISF at equimolar concentration in the absence of apoE. In contrast to Aβ, the levels of apoE at 30 mins of the CNS administration were comparable regardless of whether apoE (isoform 3) was injected alone or in the form of a complex with Aβ.
(
The rates of Aβ, apoE, and apoJ efflux via BBB transport and the ISF flow were calculated from 30 mins series by using eqs. 2, 4, and 5 and a transport kinetic model similar to that we reported for radiolabeled Aβ (Shibata et al, 2000). Table 1 shows that transport via BBB represents a major efflux route for human Aβ40 from the CNS. The rate of BBB efflux of 0.21 pmol/ming ISF was >6-fold greater than the rate of its transport via the ISF flow consistent with the results obtained with 125I-labeled Aβ40 (Shibata et al, 2000). The rate of Aβ clearance across the BBB calculated from the multiple-time efflux series (Figure 2A) with eqs. 2, 3, and 4 was 0.22 pmol/min g ISF, which was almost identical as the rate of Aβ BBB efflux obtained from a single time-point series (Table 1). We also showed that the rate of Aβ42 BBB efflux was 1.9-fold lower than for Aβ40, although its elimination via the ISF flow was comparable with that of Aβ40. Nonlipidated apoE (isoform 3) at its physiologic CSF levels was transported via the ISF flow and across the BBB at comparable low rates, that is, from 0.03 to 0.04 pmol/min g ISF, respectively. There was no detectable rapid BBB efflux of lipidated apoE3 within 30 mins, which does not rule out the possibility that lipidated apoE may be cleared from brain over longer periods of time >30 mins. At its physiologic CSF levels of 40 fmol/μL, apoJ was cleared rapidly from brain ISF within 30 mins mainly via BBB transport at a rate of 0.16 pmol/min g ISF. Amyloid β-peptide 40–apoE complex was cleared at the BBB at a rate 5.7-fold lower than free Aβ40.
Clearance rates of unlabeled Aβ, apoE, and apoJ across the BBB and via the ISF flow
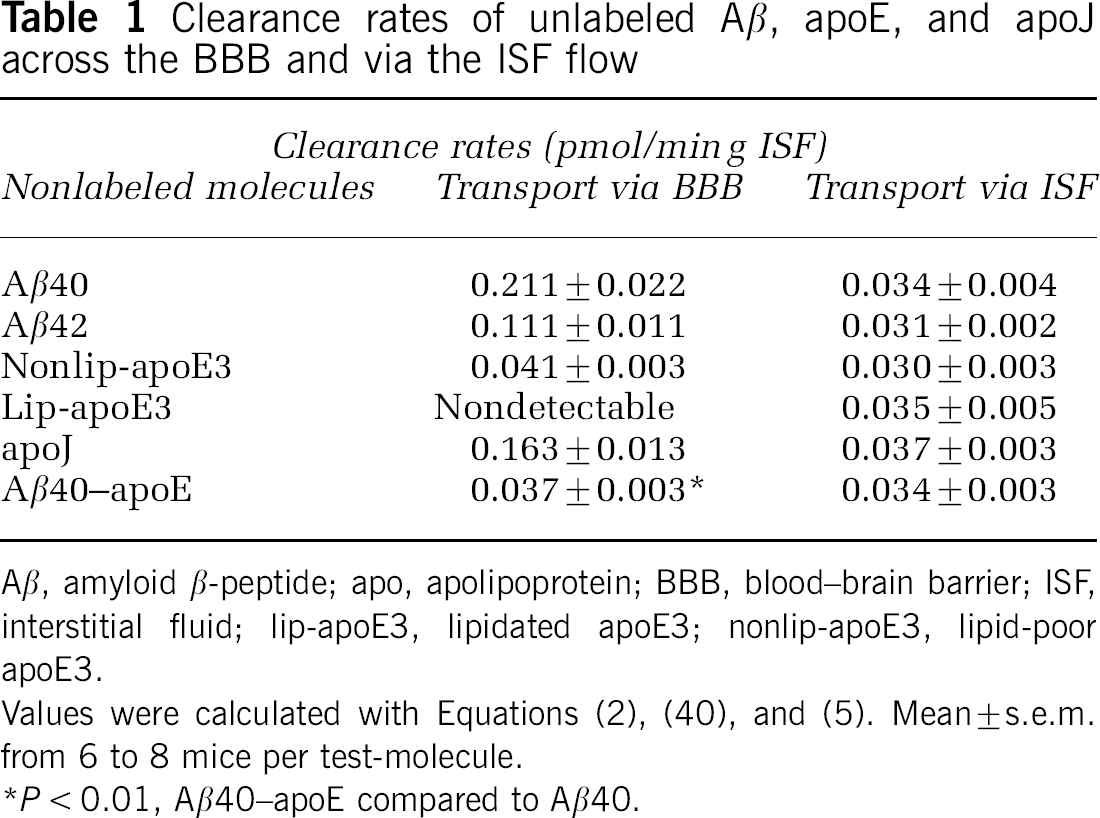
Aβ, amyloid β-peptide; apo, apolipoprotein; BBB, blood—brain barrier; ISF, interstitial fluid; lip-apoE3, lipidated apoE3; nonlip-apoE3, lipid-poor apoE3.
Values were calculated with Equations (2), (4), and (5). Mean ± s.e.m. from 6 to 8 mice per test-molecule.
P<0.01, Aβ40–apoE compared to Aβ40.
Detection of Human Aβ, ApoE, and ApoJ Clearance with Radiolabeled Tracers
After our earlier work on CNS transport of exogenous Aβ employed radiolabeled Aβ, we studied whether CNS clearance of 125I-labeled Aβ40 and Aβ42, as well as of 125I-labeled apoE and apoJ, corresponds to clearance of the respective unlabeled test-molecules, as measured above. To compare directly the clearance rates of 125I-labeled and unlabeled Aβ and apolipoproteins, we administered exactly the same amounts (ng/μL) of 125I-labeled Aβ40, Aβ42, apoE, and apoJ as in the experiments with unlabeled Aβ and apolipoproteins, as explained in Materials and Methods. Figure 4A shows that within 30 mins of the CNS administration, brain recovery of the corresponding unlabeled and 125I-labeled Aβ, apoE, and apoJ molecules were almost identical. Table 2 shows that the clearance rates via BBB transport and ISF flow of 125I-labeled Aβ40, Aβ42, apoE nonlipidated, and lipidated isoform 3 and of apoJ over the 30 mins period were not significantly different from the rates of their respective unlabeled molecules (see Table 1). These data suggest that iodination does not alter Aβ, apoE, and apoJ clearance from the CNS. Figure 4B shows that apoJ clearance from the brain was inhibited by RAP and anti-LRP2, but not by anti-LRP1, as indicated by significantly higher Nt/N0 brain retention values for apoJ in the presence of RAP and anti-LRP2. Appearance of TCA-precipitable 125I-radioactivity in plasma after administration of 125I-apoJ into brain ISF suggested apoJ transport from brain ISF to blood, which has been confirmed with native unlabeled apoJ by a human-specific apoJ ELISA (not shown). As apoJ is cleared faster from brain ISF than Aβ42, we tested whether binding of Aβ42 to apoJ may increase Aβ42 clearance from the CNS. 125I—Aβ42–apoJ complex was prepared as we described (Zlokovic et al, 1996). Figure 4C shows that Aβ42 bound to apoJ was cleared faster than Aβ42 alone, and clearance of Aβ–apoJ complex was blocked by anti-LRP2-specific antibody. The rate of Aβ42–apoJ clearance across the BBB was increased by 83% compared with Aβ42 alone.
Clearance rates of 125I-labeled Aβ, apoE, and apoJ across the BBB and via the ISF flow
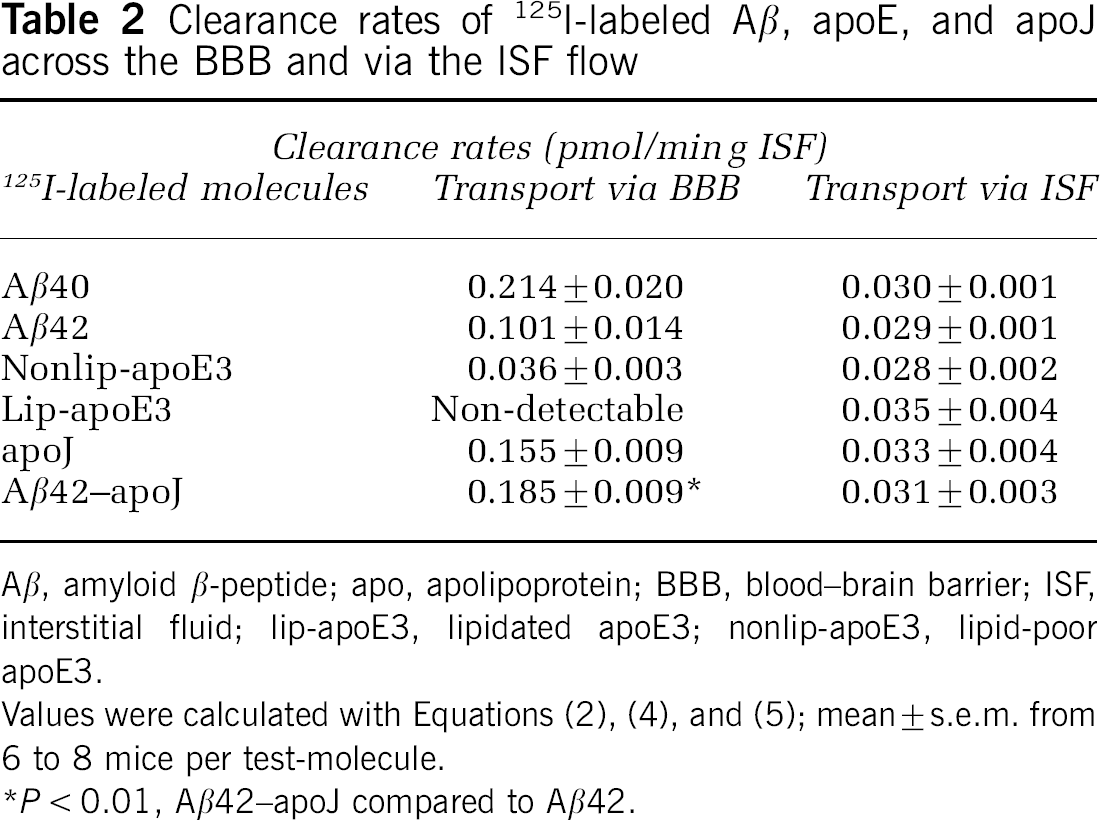
Aβ, amyloid β-peptide; apo, apolipoprotein; BBB, blood—brain barrier; ISF, interstitial fluid; lip-apoE3, lipidated apoE3; nonlip-apoE3, lipid-poor apoE3.
Values were calculated with Equations (2), (4), and (5); mean ± s.e.m. from 6 to 8 mice per test-molecule.
P < 0.01, Aβ42–apoJ compared to Aβ42.
(
Matrix-Assisted Laser Desorption Ionization Time-of-Flight and Electrospray Ionization Mass Spectrometry/Mass Spectrometry analysis
By using the MALDI-TOF MS analysis, we obtained an LOQ of 8 ng/μL for exogenous human Aβ40 in mouse CSF or artificial ISF. Since physiologic levels of exogenous Aβ in focal brain clearance studies are typically achieved with microinjections of 0.5 μL mock ISF containing 0.173 ng/μL or less of Aβ40 (Shibata et al, 2000; Deane et al, 2005), to achieve the LOQ it would require concentration of CSF and brain extracts from 920 and 231 mice, respectively. Using a more sensitive serial MALDI-TOF-TOF (Gelfanova et al, 2006), it would still require concentration of CSF from 12 mice and brain extracts from 3 mice to achieve the LOQ.
By using the LC electrospray ionization MS/MS analysis, the LOQ for15NAβ40 was 0.1732 ng/μL in mouse CSF or artificial ISF. Therefore, in a typical brain clearance study with physiologic focal levels of exogenous Aβ40, concentration of CSF from 20 mice and brain extracts from 5 mice would be required to achieve this LOQ. Regarding apoE, the LOQ was 1.4 ng/μL. Thus, in a typical clearance study with exogenous apoE at low physiologic levels, the CSF from 20 mice and brain extracts from 3 mice would need to be concentrated to achieve this LOQ. The purpose of determining the LOQ for Aβ and apolipoproteins was to evaluate the sensitivity of MALDI-TOF and Electrospray Ionization Mass Spectrometry/Mass Spectrometry to measure the clearance rates of Aβ and apolipoproteins from the mouse CNS.
Discussion
Our data suggest that one can measure accurately the clearance rates of unlabeled human Aβ and apolipoproteins across the BBB and via the ISF bulk flow in mice by using human-specific ELISAs. Matrix-assisted laser desorption ionization time-of-flight mass spectrometry and LC ESI MS/MS were less sensitive than the ELISA measurements in determining the CNS clearance rates in mice of unlabeled Aβ and apolipoproteins at low physiologic levels as found normally in the CSF and brain in mice and humans (DeMattos et al, 2003). The present study shows for the first time that iodination does not alter the CNS clearance measurements of Aβ 1–40 and 1–42, as long as transport of 125I-labeled Aβ is measured in its monoiodinated reduced form, as reported (Deane et al, 2003, 2004, 2005; LaRue et al, 2004). Moreover, we show for the first time that apoE and apoJ are cleared from the brain by different transport routes and at substantially different rates that the clearance of lipidated apoE is much slower than nonlipidated apoE, and that binding of Aβ to these apolipoproteins may critically alter its clearance from the CNS.
The present study shows unlabeled Aβ40, at the levels corresponding to those of mouse endogenous brain Aβ (Deane et al, 2005), is removed rapidly from brain ISF via transport across the BBB at a rate of 0.21 pmol/min g ISF. Transport of unlabeled Aβ40 at the BBB was mediated via LRP1, as found with 125I-labeled Aβ40 (Shibata et al, 2000; Deane et al, 2004). As the levels of soluble, rapidly exchangeable Aβ pool in brain ISF in AD mice are around 300 pmol/L ISF, as shown by microdialysis technique (DeMattos et al, 2003), given the Aβ BBB efflux rate as determined in the present study, it would take about 40 secs for LRP1 to clear this Aβ pool from brain ISF, assuming Aβ central production and re-entry of circulating Aβ into the brain (Deane et al, 2003) stop. In AD models and AD, the levels of soluble Aβ in the CSF and by extension brain ISF are around 3 nM, whereas total Aβ in brain including insoluble Aβ is in low micromolar range, for example around 6 μmol/kg brain (Cirrito et al, 2003; DeMattos et al, 2003). Making assumptions as above, it would take under pathologic conditions about 13 mins for LRP1 at the BBB to eliminate all soluble free Aβ from brain ISF, and about 18 days to remove total Aβ from brain in AD, provided all Aβ could be resolubilized into its free form. Although these numbers suggest high BBB efflux capacity for free Aβ, the present study indicates that the major pathogenic species of Aβ, Aβ42 (Selkoe, 2001), is cleared at the BBB at a rate 1.9-fold slower than Aβ40. Earlier work suggested LRP1 expression at the BBB may be substantially reduced in AD and in AD models (Deane et al, 2004). Thus, both of these factors may further modify elimination times of free Aβ under pathologic conditions.
We show lipid-poor apoE (isoform 3) is cleared slowly from brain compared with Aβ40 or Aβ42 mainly because of its low transport at the BBB, that is, 0.04 pmol/ming ISF. As a large part of the effect of apoE isoforms on AD and cerebral amyloid angiopathy risk is mediated by the interaction of apoE with Aβ (Holtzman and Zlokovic, 2006), and neither mouse apoE nor human apoE has an impact on the synthesis of brain Aβ in AD models (Bales et al, 1997; Holtzman and Zlokovic, 2006), we hypothesized apoE must affect clearance of Aβ within or from brain. Our data show that binding of Aβ40 to apoE (isoform 3) reduces by 5.7-fold its efflux rate at the BBB. The present study has been focused on apoE3 and used relatively short clearance times within the periods of 30 mins. There may be isoform-specific differences in apoE clearance from brain and in apoE-mediated retention of Aβ in the brain that could possibly be revealed over the longer times allowed for the clearance measurements (>30 mins) than in the present study. It is well recognized that the ∈4 allele has a gene—dose effect on the risk and age of onset of AD and the amount of deposited Aβ40 and vascular Aβ load, as reviewed by Holtzman and Zlokovic (2006). The isoform-specific effect of apoE on clearance of lipid-poor and lipidated apoE, the effects of longer efflux times on apoE clearance, and the receptors involved in mediating slow removal of apoE and apoE—Aβ complexes across the BBB remain to be determined by future studies. It is interesting to note that it has been recently suggested that low-density lipoprotein receptor may be involved in regulating the CNS levels of human and mouse endogenous apoE (Fryer et al, 2005) and amyloid pathology in AD mice (Cao et al, 2006).
Finally, we show that native apoJ is eliminated rapidly from brain ISF across the BBB at a rate lower than that of Aβ40, but significantly higher than the rate of Aβ42. ApoJ is the major carrier protein for Aβ in biologic fluids (Calero et al, 2000), and its receptor LRP2 is expressed at the BBB (Zlokovic et al, 1996; Chun et al, 1999). We hypothesized LRP2 may be involved in efflux of apoJ out of the CNS, and Aβ binding to apoJ may enhance clearance of highly pathogenic Aβ42. Our data show that both RAP and LRP2-specific antibody block apoJ clearance, indicating LRP2 is required for apoJ efflux at the BBB. We next show that binding of Aβ42 to apoJ accelerates Aβ42 clearance rate at the BBB by 83%, which again requires LRP2. It has been reported that LRP2 at the BBB is saturated from the blood side by physiologic levels of apoJ in plasma, which precludes brain influx of circulating Aβ bound to apoJ across the BBB (Zlokovic et al, 1996). In contrast, efflux of Aβ42–apoJ complex from brain ISF to blood is substantial at physiologic apoJ CSF levels as shown in the present study, suggesting the net transport of soluble Aβ via apoJ at the BBB favors its efflux from the brain. Consistent with the present study are findings that show lack of apoJ in AD mice may increase levels of soluble Aβ in the brain (DeMattos et al, 2004). As apoJ increases Aβ neurotoxicity in AD mice (DeMattos et al, 2002), clearance of Aβ–apoJ complexes from the brain could be neuroprotective.
In conclusion, the present study highlights the importance of Aβ clearance mechanisms in the CNS, suggesting that efflux of Aβ from brain is controlled by different transport pathways at the BBB. The lipoprotein receptors seem to play a major role in determining the rate of Aβ efflux at the BBB, either in its free form via LRP1 and/or in its bound form as a complex with apoJ via LRP2. Whether other lipoprotein receptors such as LDLR participate in slow clearance of Aβ–apoE complexes at the BBB remains to be explored. Future studies should characterize in greater detail possible role of apoE/LDLR transport interactions at the BBB and of apoJ/LRP2 interactions in regulating the levels of soluble, as well as of deposited Aβ in brain.