Abstract
Previously, water exchange across the blood–brain barrier (BBB) and blood–cerebrospinal barrier (BCSFB) was assessed by multi-delay, multi-echotime (TE) arterial spin labeling, albeit in separate acquisitions with different settings. In this study, we present a protocol for simultaneous measurement of BBB and BCSFB water exchange using a multi-TE 3D-GRASE acquisition combined with T2-preparation. We evaluate several modeling approaches, comparing two- and three-compartment models to estimate water exchange rates from blood to gray matter (
Keywords
Introduction
The blood–brain barrier (BBB) and blood–cerebrospinal fluid barrier (BCSFB) are vital structures of the human brain which protect neuronal tissue from harmful pathogens, regulate the transport of metabolic, immunologic, and hormonal molecules between fluids and tissues to preserve homeostasis, and facilitate the clearance of waste products.1–4 The BBB refers to the set of membranes and junctions which form the boundary between blood and tissue found throughout the brain, while the BCSFB refers to the boundary between blood and CSF. The main exchange between blood and CSF is traditionally understood to occur at the choroid plexus (CP), a small structure located within the ventricles. The BCSFB is also implicated in the process of brain waste clearance by regulating the exchange of water and waste products between the blood and CSF. Disruptions in the function of the BBB and BCSFB are involved in a wide variety of neurological disorders such as Alzheimer’s disease, stroke, small-vessel disease, hydrocephalus, as well as the brain’s aging process.4–12 Therefore, assessing the function of the BBB and BCSFB provides crucial information about brain health and progression of brain diseases and preferably one would like to assess both barriers at the same time, because of the close interactions between blood water, CSF, and interstitial fluid (ISF).
Traditionally, MRI measurements of BBB and BCSFB leakage are performed using gadolinium-based contrast agents (Gd), however there are limitations to these techniques. Firstly, they are invasive, measuring the leakage rate of intravenously injected Gd across the BBB 13 or the perivascular influx of Gd after intrathecal injection.14,15 For the latter, injections directly into the CSF may also disturb the equilibrium of the system we aim to characterize. 16 Secondly, Gd crosses the barriers at a very slow rate unless there is substantial disruption to the tight junctions of the endothelial cells at the BBB and the epithelial cells at the BCSFB, which regulate the paracellular movement of Gd.17,18 Whilst Gd has been found to cross the barriers at measurable levels in several neurodegenerative diseases as well as in the aging brain,13,19–25 using a smaller tracer may offer improved sensitivity to subtle or early-stage barrier impairment. Additionally, contrast-enhanced techniques cannot detect damage to other transport routes across the barrier, as Gd exclusively traverses via paracellular pathways. There is also a growing concern with the deposition of Gd in the brain over time from repeated contrast injections. Therefore, there has been increasing interest in using water as an endogenous contrast medium to measure BBB and BCSFB function as it is fully non-invasive, has the potential to provide measurements of more subtle disruptions in function, and represents healthy physiology, that is, existing processes of exchange which are naturally present in the brain.
Most of the proposed methods to assess water transport across the BBB and BCSFB employ arterial spin labeling (ASL) MRI,26–31 a technique which is generally used to measure cerebral perfusion. By optimizing ASL labeling parameters and readout strategies, blood water can be tracked as it traverses from large vessels into the tissue or CSF. Because of the different nature of the tissues and fluids involved, different techniques are used for the assessment of the integrity of the BBB and BCSFB. In all cases, multiple labeling durations (LD) and/or post-labeling delays (PLD) are employed to capture the dynamic process and either multiple echo times (TE) or different levels of flow-crushing are performed to determine in what compartment the label resides. When exploiting the difference in T2 between compartments, the technique of choice to characterize the BBB is the combination of time-encoding with a T2-preparation (T2prep) module.29,32–34 Time-encoding (or Hadamard encoding) is an efficient method for acquiring multiple LD/PLDs, especially short ones like those required for BBB imaging (~200–2000 ms). T2prep offers the advantage of allowing flexible effective echo times (eTEs), however it considerably increases scan time as only one eTE can be acquired per TR. For BBB water transport measurements, conventionally four short (<200 ms) eTEs are acquired. For BCSFB water exchange, we have previously proposed to use separate scans with different LD/PLDs in combination with a multi-TE gradient and spin-echo (GRASE) readout. 28 For this method, longer LD/PLDs (~500–4000 ms) are acquired as water exchange from blood to CSF is believed to occur more slowly and to a lesser extent than exchange from blood to GM. 28 Consequently, Hadamard encoding was found not very efficient and did not significantly reduce scan time. To isolate water signal in the CSF, extra-long TEs (>500 ms) must be acquired as CSF has a very long T2, which makes multi-TE GRASE an effective readout option. It has the advantage of acquiring multiple TEs simultaneously in one TR, albeit with longer inter-echo spacing. These long TE spacings (~260 ms in our previous work) do not allow proper separation of blood and GM signal because of their relatively short and similar T2s. In this work, we aim to reconcile the advantages and disadvantages of these two techniques (see Table 1) to measure blood water exchange to both GM and CSF in a single scan. In order for our protocol to contain a wide range of both TEs and PLDs, we combined T2prep and a multi-echo GRASE readout into one sequence and sequentially acquired multiple LD/PLDs.
Characteristics of the four compartmental models used in this work.
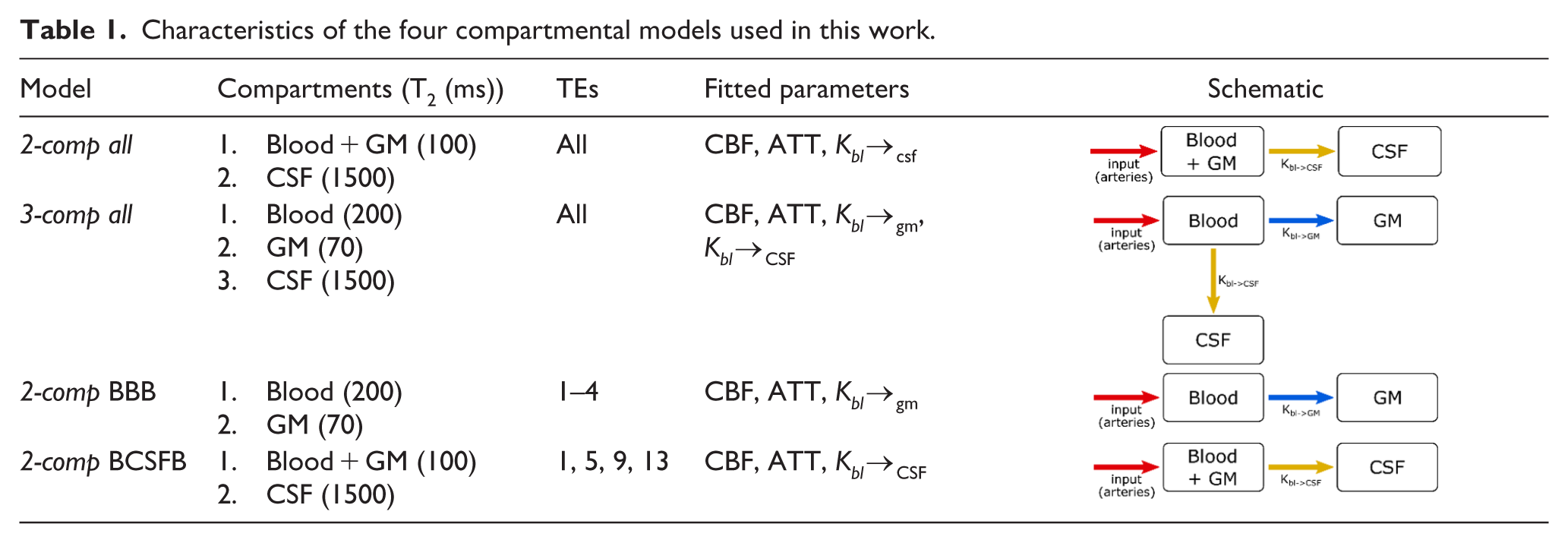
This study’s aims are therefore twofold:
• To investigate the use of a combination of T2prep and multi-TE acquisition for measurement of ASL signal at both intermediate and long TEs.
• To compare the use of two- and three- compartment models tailored to differentiate the contributions from several compartments (blood, GM, or CSF) to the ASL-signal for characterization of water transport both from blood-to-GM (BBB) as well as from blood-to-CSF (BCSFB).
Methods
MRI acquisition
Data was acquired from six healthy volunteers (1 M/5 F, age 21–63) on a Philips Achieva XT 3 T system (Philips, Best, the Netherlands) with a 32-channel head coil. This study was approved by the Medisch Etische Toetsingscommissie (METC), Leiden–Den Haag–Delft. The main imaging sequence in this protocol is a pseudo-continuous ASL (PCASL) scan with multi-echo 3D GRASE readout (as described in our previous work
28
), with the addition of a T2prep module before readout. Here, the multi-TE readout has been shortened to four TEs acquired sequentially (TE = 10 +
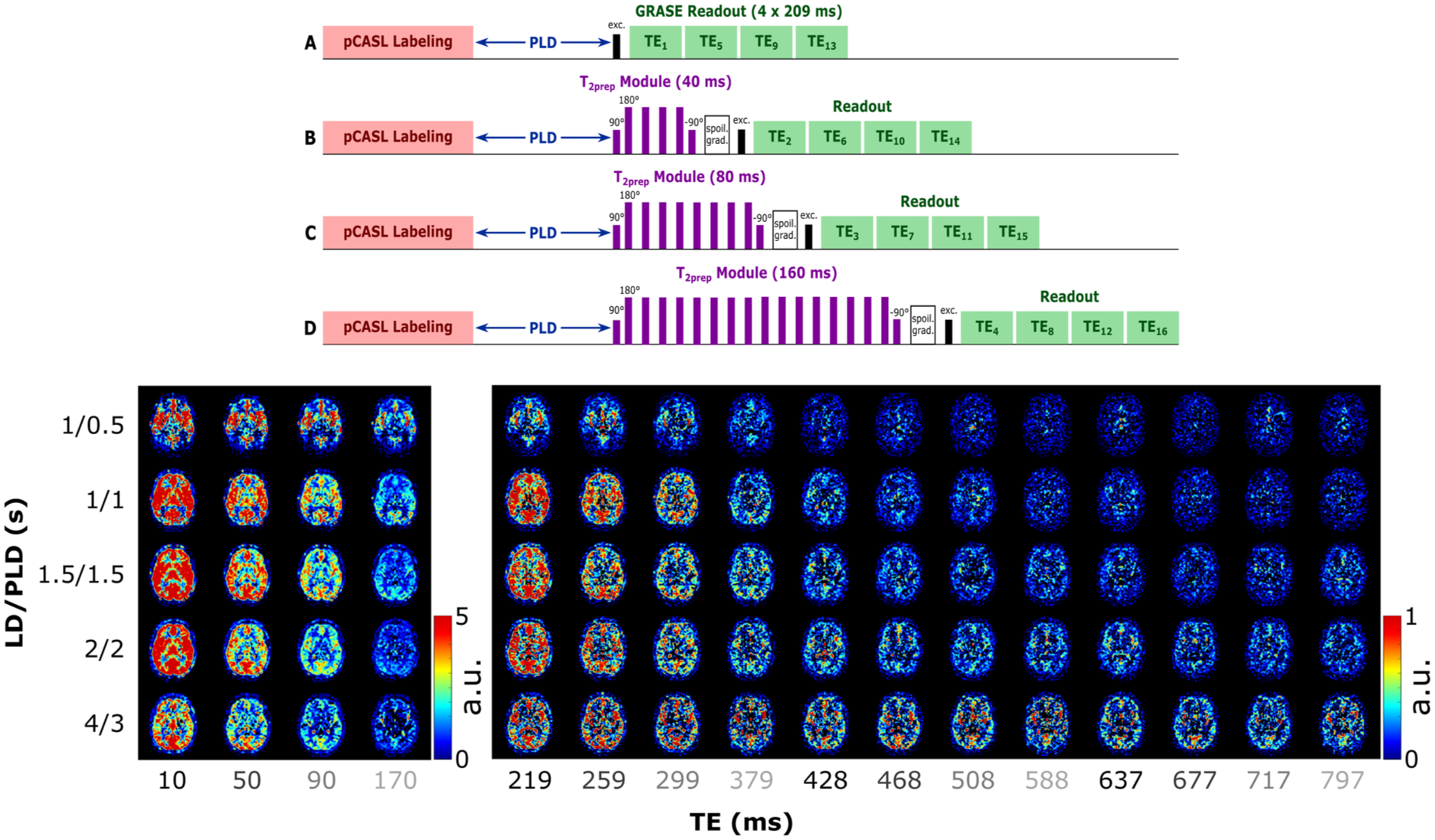
(Above) Schematic representation of the PCASL sequence used in this work. The image is not to scale (e.g. the length of the echoes in the multi-TE readout is longer than the longest T2prep module). Each of the T2prep module lengths (A–D) is acquired in a separate dynamic, resulting in an interleaved echo acquisition. (Below) ASL signal in a single slice for all LD/PLDs and TEs. Different scales allow the contrast to be preserved in the longer TEs. Echo times that are acquired sequentially within one readout are shown with the same grayscale intensity to highlight the interleaved acquisition.
This research was carried out in accordance with our institution’s IRB regulations (Medisch Etische Toetsingscommissie (METC), Leiden–Den Haag–Delft) and the Declaration of Helsinki.
Analysis pipeline
ASL control and label images were pair-wise subtracted to calculate ASL signal at all LD/PLDs and TEs. A CSF mask was created by thresholding the control image at the longest TE, and a GM mask was obtained by thresholding the ASL signal of the LD/PLD 2/2 s image (the closest to typical ASL perfusion parameters where there is maximal contrast between the GM and other tissues and fluids). The thresholds for these masks were adjusted for each subject by an experienced observer (first author who had 4 years of experience with ASL-data) to retain as much as possible the anatomical features such as the cortex gyri and CSF-filled sulci. The WM mask was isolated by removing the GM and CSF mask voxels from a whole brain mask. Finally, a CP mask was obtained by isolating the posterior part of the ventricles where the CP is predominantly located and using the same threshold as the GM mask to exclude the CSF surrounding the CP.
Two ROIs were created by superimposing an elliptical shape onto (1) a single slice in the middle of the brain (slice 15), comprising the majority of the left MCA territory and avoiding the ventricles (left MCA ROI) and (2) in a high slice of the brain (slice 22), comprising most of the left hemisphere in that slice (high slice ROI). The ASL signals from these ROIs were averaged, combining signal from all tissues and fluids within the elliptical regions, and four multi-compartmental models, detailed in the following section, were fitted to the resulting data. In addition, voxelwise fitting of these models was performed to generate whole-brain maps of the corresponding model parameters. Prior to fitting, the ASL signal was smoothed with a gaussian kernel (3 × 3 voxels, σ = 2 voxels) to reduce noise. To enable comparison of parameter values across brain regions, the estimated parameters were averaged within the following tissue region ROIs: GM (GM mask slice 15 and up), subarachnoid space (SAS; CSF mask slice 15 and up, excluding the ventricles), CP (CP mask), and WM (WM mask, used as a control region). Because of the small number of subjects (i.e. low statistical power), the parameters were compared qualitatively instead of performing formal statistical analyses.
Compartmental models
Four models were used to characterize the exchange of water between the blood, GM, and CSF compartments in this study. Detailed equations for the models can be found in the Supplementary Material. In brief, the first model (
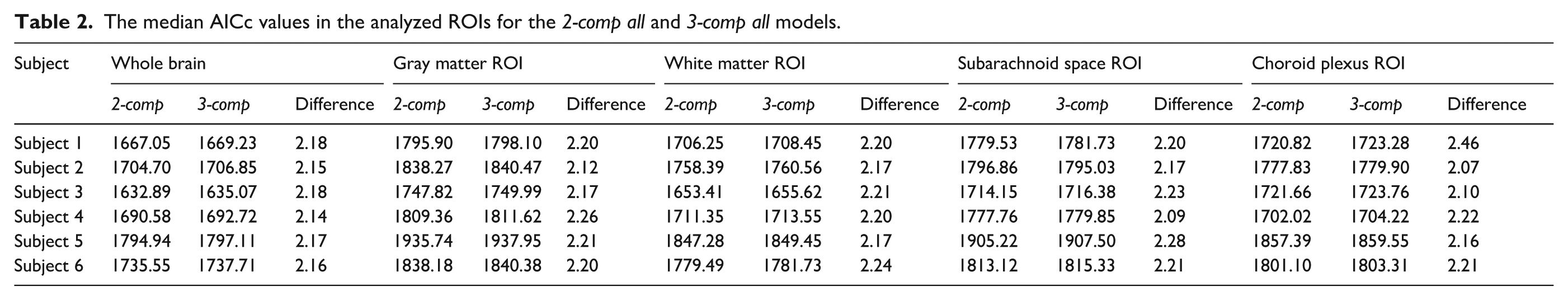
The median AICc values in the analyzed ROIs for the
Akaike Information Criterion (AIC) analysis
The
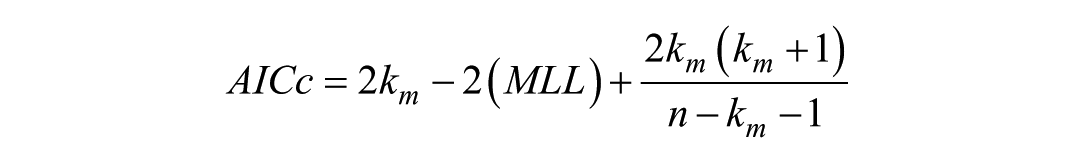
where
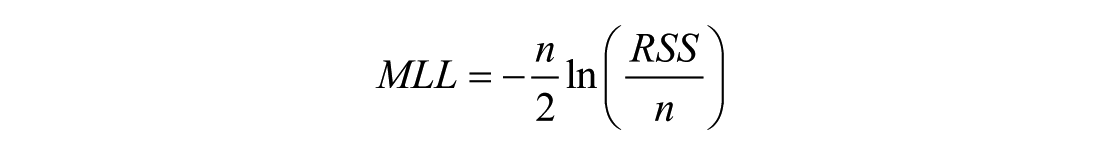
where RSS is the residual sum of squares. Voxelwise AIC values were evaluated across the whole brain, GM, CP, SAS, and WM ROIs.
Results
ASL signal
The measured ASL signal (Figure 1) is consistent with literature and previous work (note that two different scales are used for the first four echoes and the rest, to visualize contrast at long TEs where the signal is low). When focusing on the first TE (first column) we see the early arrival of blood in the vasculature in the first time point, then the signal appears in the GM as a perfusion pattern (LD/PLD 1/1–2/2 s) before fading away. In longer TEs (>500 ms), the CSF signal becomes apparent, with a slower inflow (it appears mostly from LD/PLD 2/2 s and longer) and slow decay through the TEs. The evolution of the signal through the TEs shows rapid decrease for blood and GM signals and slower decay in the CSF (CP and SAS). As seen in our previous work, 28 CSF signal from blood-to-CSF water exchange is present throughout the brain, and not exclusively in the choroid plexus.
Signal fractions
Using the parameter maps estimated from all compartmental models, the amount of signal arising from each compartment can be calculated from the model equations. These signal fractions are shown in Figure 2 for a single slice of subject 5 for all LD/PLD pairs of this study. Results of the
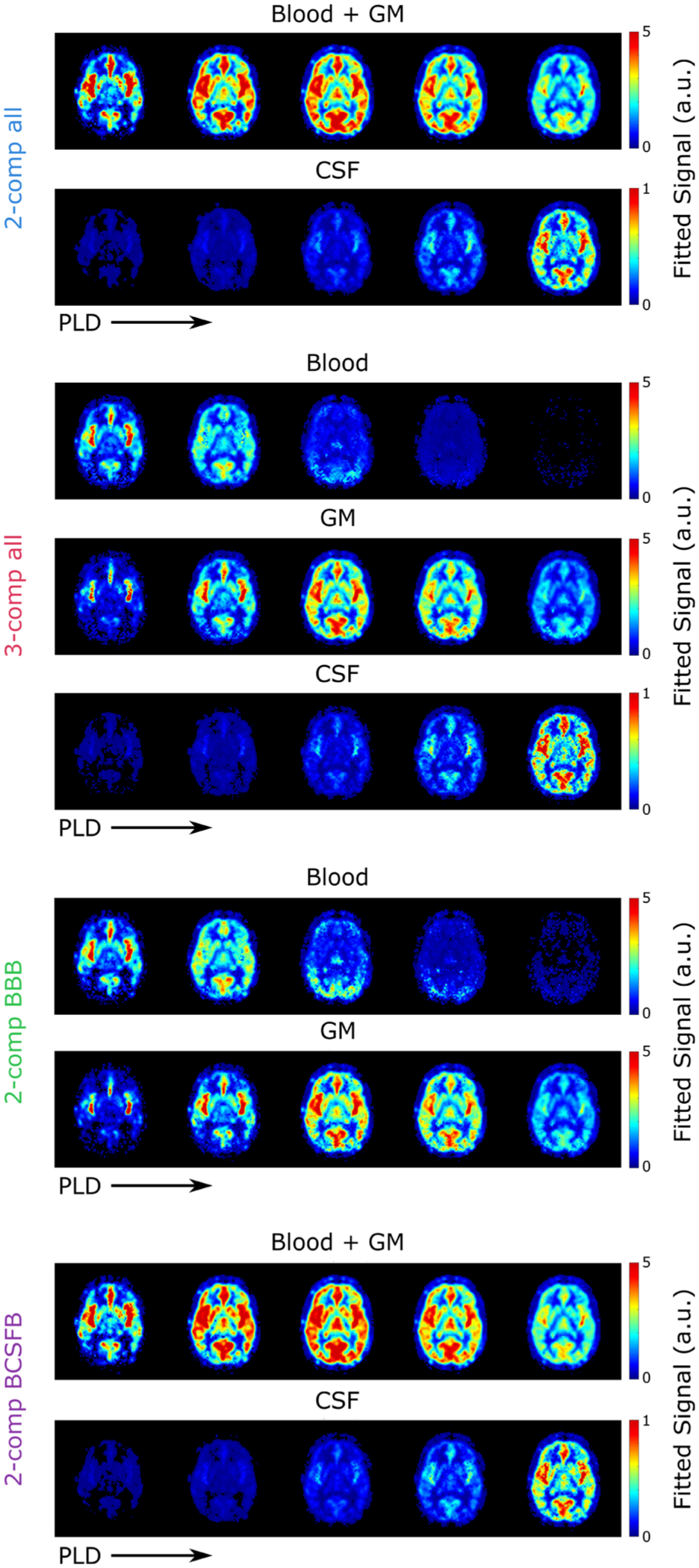
Relative amount of signal present in each compartment at TE = 10 ms and all LD/PLDs used in this experiment as fitted with the four models.
In our previous work, the blood and GM compartments were combined into a single compartment, because the large inter-echo spacing of the data did not allow their short T2s to be distinguished and hence resulted in a large amount of overlap between their signal maps when using a three-compartment model. Here, in the
Supplementary Figure S1 presents residual maps illustrating the differences between the total fitted signal and the measured signal at each TE/PLD for both the
Additionally, Supplementary Figure S2 shows a video of the signal obtained from the
AIC results
As shown in Table 2 and Supplementary Figure S3, the AIC values for the
ROI analyses: Left MCA and high slice ROIs
Figure 3 shows the fit of the four different models for the left MCA ROI and high slice ROI. While both ROIs combine a mixture of signal from blood, GM, and CSF, because of the geometry of acquisition the high slice ROI contains more SAS CSF than the MCA ROI. Based on the GM, WM, and CSF masks, 45% of the voxels in the left MCA ROI contained GM, 37% contained WM, and 22% CSF. For the high slice ROI, the proportions shift to 36% GM, 46% WM, and 27% CSF. Looking at the signal curves, we see earlier arrival of the signal in the MCA territory and generally higher signal intensity. In the MCA ROI, the CBF is higher, the ATT shorter,
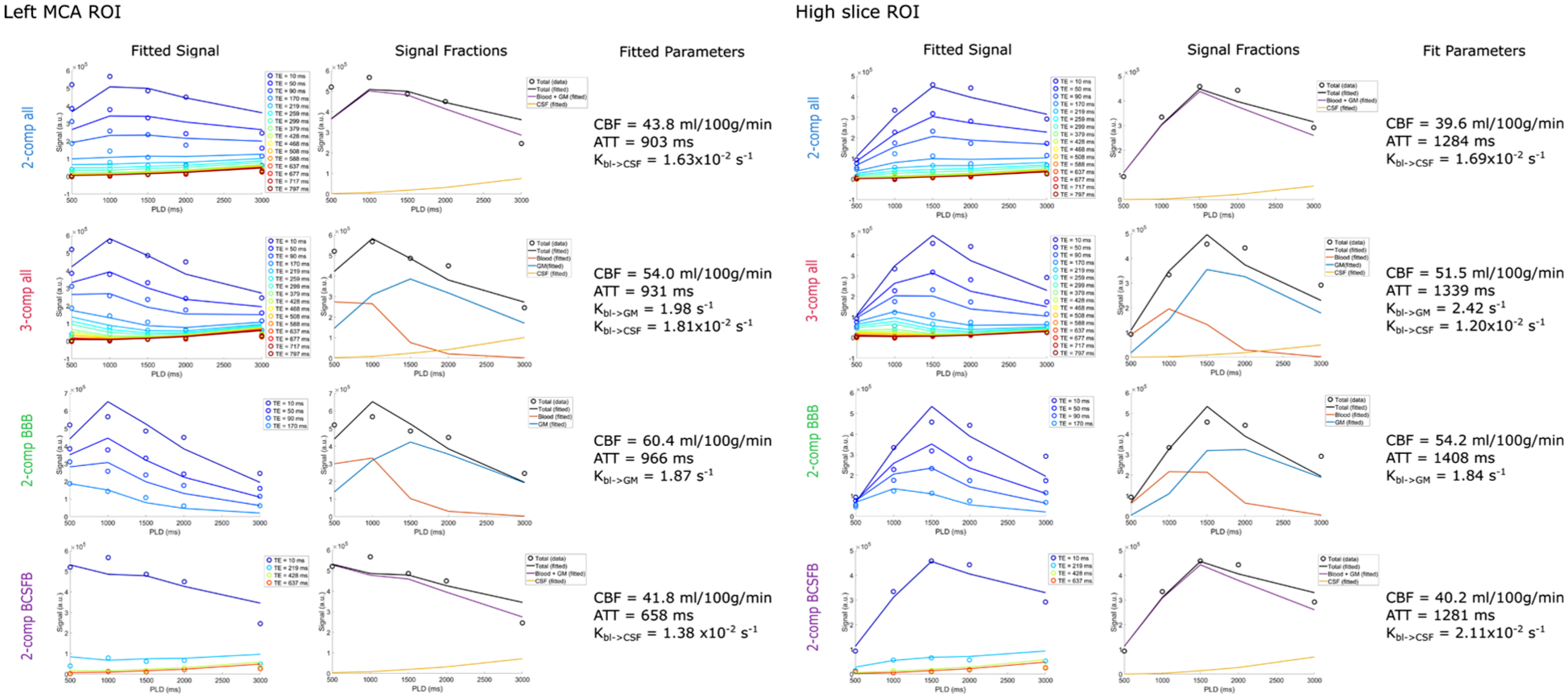
Model fitting of the signal in the left MCA territory ROI (single slice, containing 390 voxels) and the high slice ROI (single slice, containing 338 voxels) of subject 5 for all four models. The first column shows the signal (circles) and fitted curves (lines) for the different echo times. The second column shows the signal in each compartment based on the fit, and the third column shows the fitted parameters.
Parameter mapping: Whole brain, GM, WM, and CP ROIs
Parameter maps for a single slice created by all models (subject 5) are shown in Figure 4 (for full brain maps of two other subjects see Supplementary Figures S4 and S5). The CBF and ATT maps are consistent with literature. The CBF shows a typical perfusion pattern, while the ATT map highlights large arteries (the middle cerebral arteries and the anterior cerebral artery) and prolonged ATT in the posterior region of the brain and border zones.
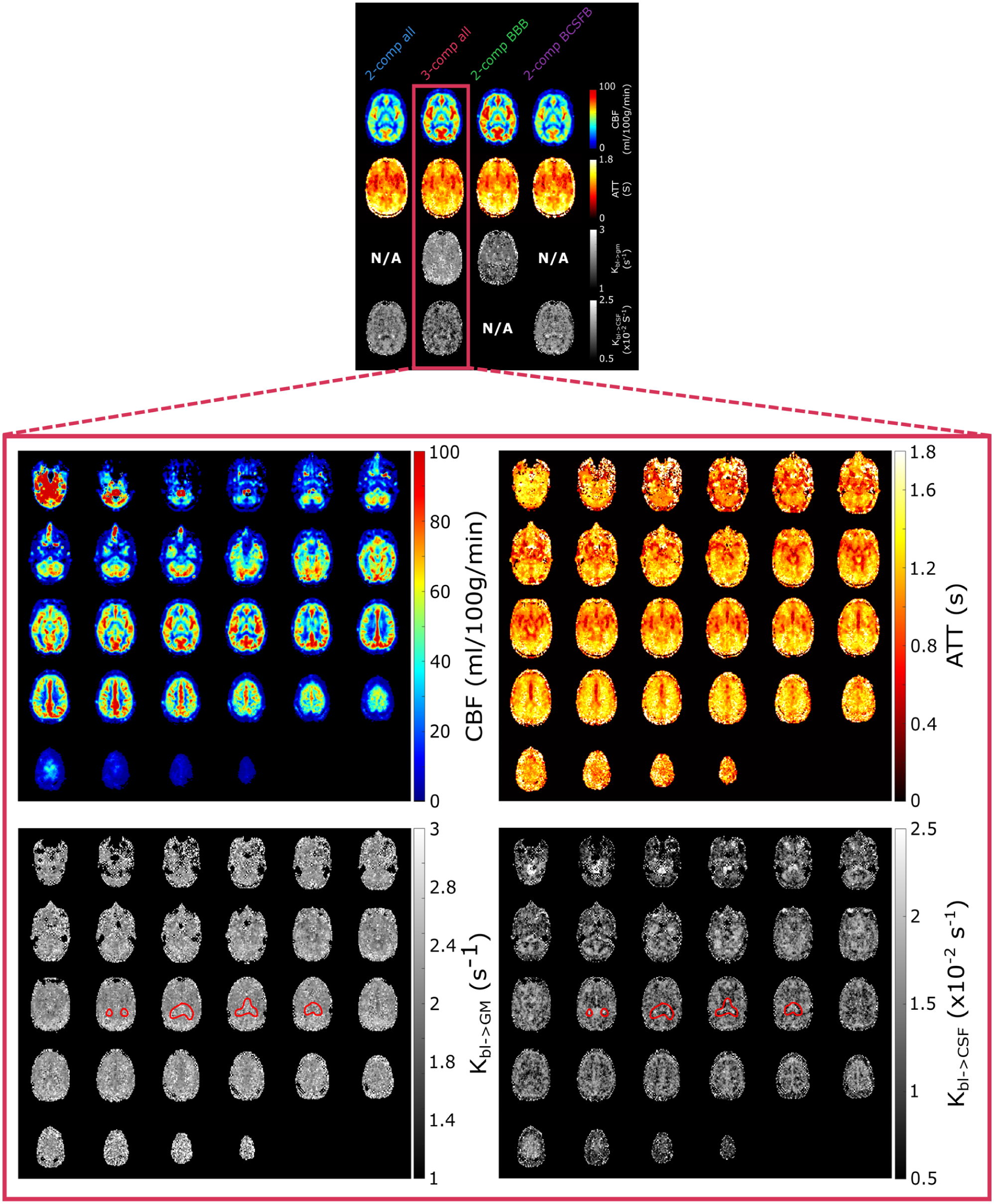
(Above) Single slice parameter maps for all models in subject 5. (Below) Whole-brain parameter maps in subject 5 for the
Whole-brain parameter maps are shown in Figure 4 for the
For a direct spatial comparison of these parameters across different tissue regions, the maps were averaged over the GM, SAS, CP, and WM ROIs. The averaged parameter values are shown in Figure 5 for all subjects and models. It is important to note that, although these ROIs were defined using tissue-specific masks and primarily represent regions dominated by the corresponding tissue types, they were not assumed to consist exclusively of a single tissue type. As a result, the ROIs are likely to include partial-volume contributions from surrounding tissues. For example, voxels within the GM ROI may contain partial-volume contributions from the SAS, such that at late TEs CSF signal is present within these voxels, allowing quantification of
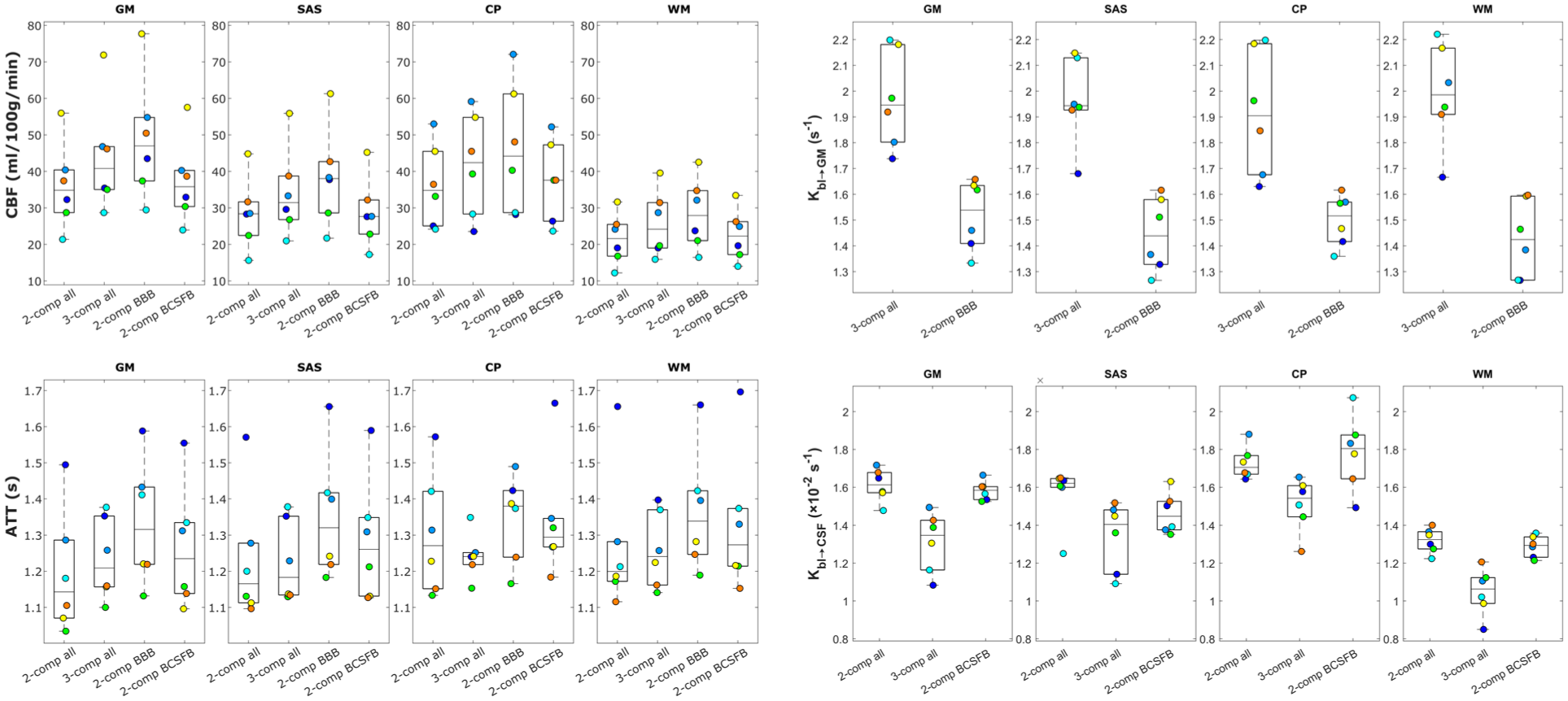
Average values of model parameters fitted with the four compartmental models for all subjects in GM, SAS, CP, and WM ROIs. Note that
Discussion
In this work, we aimed to simultaneously measure water exchange in the human brain from the blood to the GM and to the CSF. We also compared the CBF, ATT, and exchange parameters
The combination of T2prep and multi-echo readout was effective in measuring ASL signal for a large range of TEs with sufficient small inter-echo spacing. The ASL signal (Figure 1) is consistent with literature, and shows a relatively smooth transition from high signal in the short TEs to low signal in long TEs. The longest TEs (from ~500 ms) effectively isolate CSF signal as seen in the complete decay of blood and GM signal in the first two time points at longer TEs, followed by the gradual increase in long-TE signal consistent with the later arrival of labeled water in the CSF.
Furthermore, the proposed compartmental models provided good fits to the signal, with the
Turning to the brain-wide tissue region ROI analyses, the parameter maps shown in Figure 4 and the regional averages in Figure 5 demonstrate that the
Several studies using multi-echo and diffusion-prepared ASL report
In addition, variability in T2 of arterial blood due to its dependency on oxygenation might have influenced the results.
66
The similarity in values between brain regions could likewise be due to limitations of the fitting technique, or could indicate that the phenomenon which we aim to characterize, that is, the exchange of water from blood to tissue, is present throughout the brain at a comparable rate. The models employ literature values of GM for all brain tissues, that is, also for WM. This can be justified by their relative similar T2s compared to the much more different properties of CSF. Additionally, the
The AIC analysis didn’t favor a specific model as both models yielded similar AIC values across all brain regions, with the
There are a number of limitations to this study which should be noted. Firstly, the imaging sequences used are long, and a lot of optimization was needed to reach a satisfactory range of echo times and LD/PLDs which would fit into an hour of scanning. The timing was so limiting that the T1w image that would usually be acquired with such a protocol had to be forgone. A T1w scan is typically used to create GM, WM, and CSF masks which can subsequently be coregistered to the ASL data, but here we had to use more indirect masking techniques which may have resulted in less effective separation of tissue types. This might also explain the fact that the GM CBF values were lower in this study than typically observed in literature. The long scan times limit the clinical translatability of this method for which it may be more efficient to employ Hadamard time-encoding for BBB characterization, along with separate longer LD/PLD and TE T2-prepared acquisitions as implemented in this paper for the BCSFB. Reducing the number of sampled TEs and/or PLDs could also be beneficial however extensive simulations, such as leave-one-out cross-validation, would be required to ensure model sensitivity to exchange time and hemodynamic parameters is maintained.
Secondly, the ASL signal did show some instability through the TEs, with some small increases appearing at longer TEs. This is also observed when averaging signal over ROIs and plotting over TEs (shown in Supplementary Figure S6 for the two ROIs), where a slight saw-tooth effect was present. We however found that these variations were small in comparison to the natural signal decay along TEs, but these could still induce some errors in the analysis of this signal. This pattern could be caused simply by the interleaved nature of the acquisition of TEs with natural fluctuations in perfusion and physiological processes over the long scan time, or it could arise from an interaction between the T2prep module and the readout module (and BGS). However, this behavior was different in different subjects (i.e. signal peaks did not occur at the same TEs). These variations were deemed acceptable in the context of this work, but further improvements in sequence design could alleviate this issue, for example, by limiting the overall temporal footprint.
Additionally, these data are of course acquired in a small cohort of healthy individuals, and it therefore did not have statistical power to study the relationship between water exchange rates and, for example, age and sex. Testing this technique in larger groups of subjects as well as in patient populations is critical to reach a better understanding of the role of brain water exchange in health, aging, and disease.
Finally, the most relevant limitation of this work is the absence of a reliable reference standard for validating water-exchange measurements. As a result, particularly for
In conclusion, we find that the combination of a T2prep module with a multi-echo 3D GRASE readout allows the non-invasive measurement of ASL signal at a wide range of echo times necessary for the simultaneous characterization of the BBB and the BCSFB. The proposed compartmental models were effective in measuring perfusion and exchange parameters, and in particular the three-compartment model showed promise to provide a more complete picture of the dynamics of water exchange between blood, GM, and CSF.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X261429042 – Supplemental material for Simultaneous measurement of water transport across the blood–brain and blood–CSF barrier in the human brain with arterial spin labeling MRI
Supplemental material, sj-docx-1-jcb-10.1177_0271678X261429042 for Simultaneous measurement of water transport across the blood–brain and blood–CSF barrier in the human brain with arterial spin labeling MRI by Léonie Petitclerc, Helena Durrant, Lydiane Hirschler, Lena Václavů and Matthias J.P. van Osch in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
The authors thank Prof. Dr. Jelle J Goeman for statistical advice regarding Akaike Information Criterion analysis.
Abbreviations
ASL = Arterial spin labeling
AIC = Akaike information criterion
AP = Anterior-to-posterior
ATT = Arterial transit time
BBB = Blood–brain barrier
BCSFB = Blood–CSF barrier
BGS = Background suppression
CBF = Cerebral blood flow
CP = Choroid plexus
CSF = Cerebrospinal fluid
eTE = Effective echo time
GM = Gray matter
Gd = Gadolinium
GRASE = Gradient and spin-echo
ISF = Interstitial fluid
LD = Labeling duration
PCASL = Pseudo-continuous arterial spin labeling
PLD = Post-labeling delay
PVS = Perivascular space
RL = Right-to-left
SAS = Subarachnoid space
T2prep = T2 preparation module
TE = Echo time
TR = Repetition time
WM = White matter
Author contributions
Léonie Petitclerc: conceptualization, methodology, software, formal analysis, data curation, writing, visualization. Helena Durrant: software, formal analysis, data curation, writing, visualization. Lydiane Hirschler: conceptualization, methodology, writing. Lena Václavů: investigation, software, writing, supervision. Matthias J.P. van Osch: conceptualization, methodology, writing, supervision, funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is part of the research programme Innovational Research Incentives Scheme Vici with project number 016.160.351, which is financed by the Netherlands Organisation for Scientific Research. Lena Václavů is supported by the Dutch Research Council (NWO) Talent Programme Veni (Project No. 20212), which is partly financed by the NWO. Helena Durrant, Matthias J.P. van Osch, and Lena Václavů received funding from MODEM, a Dutch national consortium part of the Dementia Research Program of ZonMw (grant #10510032120006) supported by the National Dementia Strategy 2021–2030 of the Dutch Ministry of Health, Welfare, and Sport. Helena Durrant received funding from Alzheimer Nederland (#WE.30-2022-04) supporting MODEM research. Matthias J.P. van Osch and Lydiane Hirschler received support from a grant from the Leducq Foundation and the Leducq Foundation for Cardiovascular Research (23CVD03) and from the Joint Program for Neurodegenerative Diseases on Human Brain Clearance Imaging.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The research groups of Matthias J.P. van Osch and Lena Václavů receive research support from Philips.
Ethical considerations
This research was carried out in accordance with our institution’s IRB regulations (Medisch Etische Toetsingscommissie (METC), Leiden–Den Haag–Delft) and the Declaration of Helsinki.
Consent to participate
All participants gave written informed consent prior to participation.
Consent for publication
All participants gave written informed consent for non-identifiable images to be used for publication.
Data availability statement
Due to the restrictions of the informed consent, the data is not available publicly, but can be provided upon reasonable request to the corresponding or last author.
Supplemental material
Supplemental material for this article is available online.
