Abstract
Circulating bone marrow-derived immature cells, including endothelial progenitor cells, have been implicated in homeostasis of the microvasculature. Decreased levels of circulating endothelial progenitor cells, associated with aging and/or cardiovascular risk factors, correlate with poor clinical outcomes in a range of cardiovascular diseases. Herein, we transplanted bone marrow cells from young stroke-prone spontaneously hypertensive rats (SHR-SP) into aged SHR-SP, the latter not exposed to radiation or chemotherapy. Analysis of recipient peripheral blood 28 days after transplantation revealed that 5% of circulating blood cells were of donor origin. Cerebral infarction was induced on day 30 posttransplantation. Animals transplanted with bone marrow from young SHR-SP displayed an increase in density of the microvasculature in the periinfarction zone, reduced ischemic brain damage and improved neurologic function. In vitro analysis revealed enhanced activation of endothelial nitric oxide synthase and reduced activation p38 microtubule-associated protein (MAP) kinase, the latter associated with endothelial apoptosis, in cultures exposed to bone marrow-derived mononuclear cells from young animals versus cells from aged counterparts. Our findings indicate that partial rejuvenation of bone marrow from aged rats with cells from young animals enhances the response to ischemic injury, potentially at the level of endothelial/vascular activation, providing insight into a novel approach ameliorate chronic vascular diseases.
Introduction
Homeostasis of the microcirculation involves a delicate balance between injurious and reparative processes. Repair of the microvasculature has traditionally been considered to result from outgrowth of preexisting vessels to injured areas. More recently, an important contribution of circulating bone marrow-derived immature cells, including endothelial progenitor cells, has been recognized to have a role in the maintenance of the microvasculature, both as a source of endothelial cells (Asahara et al, 1997) and growth/angiogenesis factors (Majka et al, 2001). Decreased levels of bone marrow-derived circulating endothelial progenitor cells have been demonstrated in patients with cardiovascular risk factors (Hill et al, 2003) and correlate with vascular dysfunction (Hill et al, 2003) and poor cardiovascular outcomes (Schmidt-Lucke et al, 2005; Werner et al, 2005). Similarly, we have shown that patients with cerebrovascular disease have decreased circulating bone marrow-derived immature cells, the latter associated with impaired cerebrovascular function (Taguchi et al, 2004a ) and reduced cognition (Taguchi et al, 2008, 2009). In contrast, increased levels of bone marrow-derived immature cells are associated with neovascularization of the ischemic brain (Yoshihara et al, 2008), and transplanted bone marrow cells improved microcirculatory function in a variety of ischemic organs (Taguchi et al, 2003; Tateishi-Yuyama et al, 2002). In addition to these clinical observations, studies in experimental ischemia demonstrated improved microcirculatory function after transplantation of bone marrow-derived immature cells (Kamihata et al, 2001; Taguchi et al, 2004b ).
These clinical and experimental results lead us to hypothesize that ‘exhaustion’ and/or ‘aging’ of bone marrow cells in patients with cardiovascular and cerebrovascular diseases may be associated with microvascular dysfunction (Taguchi, 2009). In this study, we used aged stroke-prone spontaneously hypertensive rats (SHR-SP) (Zhang et al, 2006) and investigated the effect of partial rejuvenation of their bone marrow with strain-matched bone marrow mononuclear cells from young animals in the setting of experimental stroke.
Materials and methods
All experiments were approved by the Ethics Committee of Ehime University Graduate School of Medicine and the National Cardiovascular Center. All procedures were performed in accordance with the guidelines of the Animal Care Committee of Ehime University Graduate School of Medicine and the National Cardiovascular Center. Quantitative analyses were conducted by investigators who were masked to the experimental protocol and identity of animal, tissue, and experimental conditions pertaining to the animals under study.
Partial Replacement/Rejuvenation of Bone Marrow Cells in Stroke-Prone Spontaneously Hypertensive Rats
Male SHR-SP (SHRSP/Izm; Japan SLC, Hamamatsu, Japan) (Zhang et al, 2006), aged 4 to 5 weeks, were used as donors for bone marrow transplantation. Bone marrow cells were obtained from both thigh bones, and mononuclear cells were isolated by density gradient centrifugation using Ficoll (GE Healthcare, Uppsala, Sweden) at 400g for 40 minutes according to the manufacturer's protocol. Female SHR-SP, aged around 55 weeks (55±3 weeks) maintained on a high salt diet (OA-2, Japan Clea, Tokyo, Japan) for >40 weeks were recipients of bone marrow cell transplantation. To avoid injury to the microvasculature likely to occur with standard pretreatment for bone marrow transplantation, recipient animals received no radiation or chemotherapy before bone marrow transplantation. Instead, donor bone marrow cells were transplanted by intravenous infusion and direct intrabone marrow injection, the latter to increase transplantation efficiency. Intrabone marrow injection of bone marrow cells has been shown to result in a high seeding efficiency (Inaba et al, 2007), and this approach has been used in a clinical trial (Li et al, 2007). To obtain the highest seeding efficiency without pretreatment, we used both intravenous and intrabone marrow injection in this study. A dental drill was used to make one hole at each end of the left shin bone, and donor bone marrow mononuclear cells (0.5 mL; 1 × 106 cells/mL suspended in phosphate-buffered saline (PBS)) were injected into one hole whereas suction was applied to the other hole (at the other end of the bone) to remove host bone marrow (Figure 1A). Subsequently, intravenous infusion of the same volume of bone marrow mononuclear cells (0.5 mL; 1 × 106 cells/mL suspended in PBS) was performed using the tail vein. The viability of transplanted cells was 99.2%, evaluated with Trypan Blue staining (Sigma-Aldrich, St Louis, MO, USA). As a control, the same volume of PBS was injected into the bone marrow and tail vein. Animals were housed in an animal room with a temperature range of 21°C to 23°C and a 12-hour light/dark cycle (light on: 0700 to 1900 hours) for 30 days. The mean arterial blood pressure in each animal was measured using a rat tail manometer—tachometer system (MK-1030, Muromachi Co., Tokyo, Japan) on days 15, 30, and 45 after injection of bone marrow or PBS. No significant difference in blood pressure was observed between groups (data not shown).
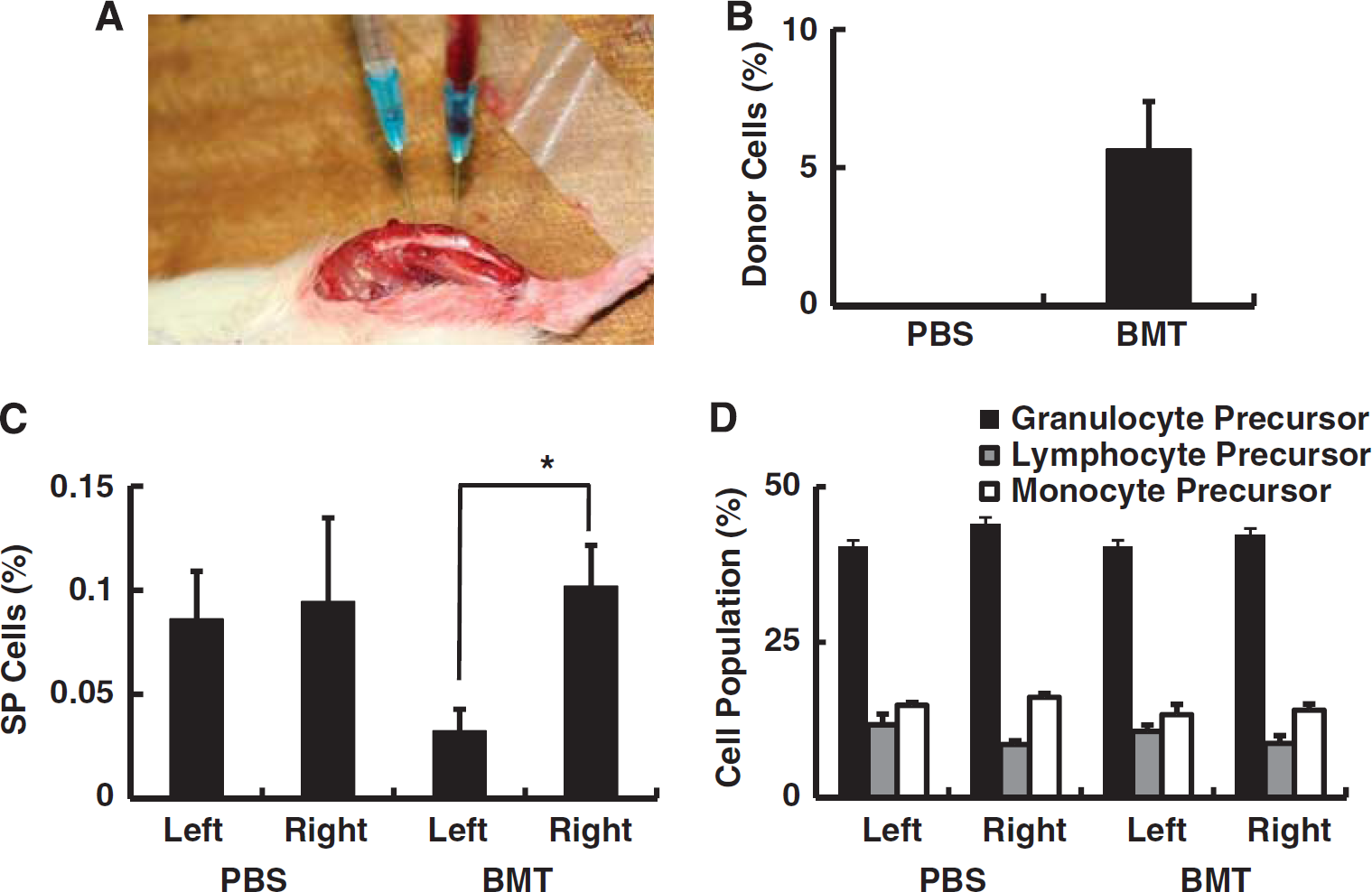
Transplantation of bone marrow from young rats into aged rats. (
Induction of Focal Cerebral Ischemia
On day 28 after transplantation of bone marrow mononuclear cells or PBS injection, focal cerebral ischemia was induced in SHR-SP as described previously (Zhang et al, 2006). Briefly, rats were anesthetized with 1.5% halothane in a 4:3 mixture of nitrous oxide and oxygen, and brain temperature was maintained at 37°C±0.5°C during the surgery. The left middle cerebral artery above the rhinal fissure and distal to the striate branches was coagulated and cut. After recovery from anesthesia, animals were maintained in an air-conditioned room at ∼22°C. On day 30 after induction of stroke, whole brain images were captured with a digital camera system (Olympus, Tokyo, Japan), and the area of intact cortex was measured by NIH image. Sham-operated rats were also studied to evaluate the influence of bone marrow manipulation and induction of stroke on animal survival. The sham group was subjected to an operative procedure on the middle cerebral artery, but no hole was drilled in shin bone to avoid a manipulation that might mobilize bone marrow cells to the systemic circulation.
Assessment of Neurologic Function
To assess cortical function, rats were subjected to behavioral testing using the open field task at 14 days after stroke (Taguchi et al, 2004b ). In this behavioral paradigm, animals were allowed to search freely in a square acrylic box (60 × 60 cm2) for 60 minutes. A light source on the ceiling of the enclosure was on during the first 30 minutes (light period) and was turned off during a subsequent 30-minute period. On the X- and Y-banks of the open field, two infrared beams were mounted 2 cm above the floor, spaced at 10 cm intervals, forming a flip-flop circuit between them. The total number of beam crossings by the animal was counted and scored as traveling behavior. Twelve infrared beams were set 5 cm above the floor, spaced at 3 cm intervals, on the X-bank and the total number of beam crossings was counted and scored as rearing behavior. The total count of traveling and rearing behavior was calculated as total locomotion.
To assess spatial learning and memory, rats were subjected to sequential Morris water maze tests at 2 weeks after induction of stroke, as described previously (Zhang et al, 2006). Briefly, each test included three trials per day for 4 consecutive days. Rats were allowed to swim until they reached a submerged platform. Then, animals were allowed to remain on the platform for at least 10 seconds. In the event that rats could not find the platform within 90 seconds, they were placed by hand on the platform for 15 seconds and their escape latency was recorded as 90 seconds. The mean latency of finding the invisible platform was measured for individual animals on each day.
Analysis of Peripheral Blood and Bone Marrow Cells
On day 30 after bone marrow transplantation, peripheral blood was analyzed by fluorescence-activated cell sorter (FACS), using anti-CD4, CD8, CD25, CD45RA, NKT, and granulocyte antibodies (all of these antibodies were from BD Bioscience, San Jose, CA, USA), according to the manufacturer's protocol. Peripheral blood was also analyzed to assess the chimera ratio of transplanted (donor):recipient rat blood cells on day 28 after bone marrow transplantation by fluorescence in situ hybridization analysis. Briefly, rat-chromosome12-FITC chromosome paint probe was used as a fluorescence in situ hybridization control and ratY-Cy3 chromosome paint probe was used to identify donor-derived chromosomal DNA (each probe; Cambio, Cambridge, UK). Fluorescence in situ hybridization analysis with Cambio probes was performed according to the manufacturer's instructions as described in http://www.cambio.co.uk. The level of immature cells, side-population (SP) cells (Pearce et al, 2007), in bone marrow was evaluated by FACS using Hoechst 33342 (Molecular Probes, Eugene, OR, USA). Briefly, rat bone marrow was obtained from the femurs and tibias of transplanted recipient rats, and single cell suspensions were made by passage of bone marrow through an 18-gauge needle. Bone marrow cells were resuspended at 106 cells/mL in prewarmed DMEM containing 2% fetal calf serum, 1 mmol/L Hepes, 100 units/mL penicillin, 100 μg/mL streptomycin, and 5 μg/mL Hoechst 33342. Cell suspensions were then incubated for 90 minutes at 37°C. Resolution of Hoechst populations is highly sensitive to the staining time and Hoechst dye concentration (Watson et al, 1985). After Hoechst staining, cells were pelleted and maintained at 4°C before FACS analysis (Becton Dickinson & Co., Mountain View, CA, USA). The level of circulating SP cells was also evaluated, but the number of SP cells in peripheral blood was too small to obtain reproducible data (data not shown).
Analysis of Cytokine Level by Enzyme-Linked Immunosorbent Assay
On day 3 after induction of stroke, serum and tissue samples were obtained from peripheral blood as well as infarcted and periinfarcted cortex, as described in Figure 3A. Proteins were extracted with lysis buffer, containing 1% NP-40 (Sigma-Aldrich), 1% Triton-X (Sigma-Aldrich), and 1 × protease inhibitor cocktail (Sigma-Aldrich). Levels of interleukin (IL)-1β, IL-6, tumor necrosis factor-α, and monocyte chemoattractant protein-1 (MCP-1) were evaluated by enzyme-linked immunosorbent assay (R&D, Minneapolis, MN, USA) according to the manufacturer's protocol.
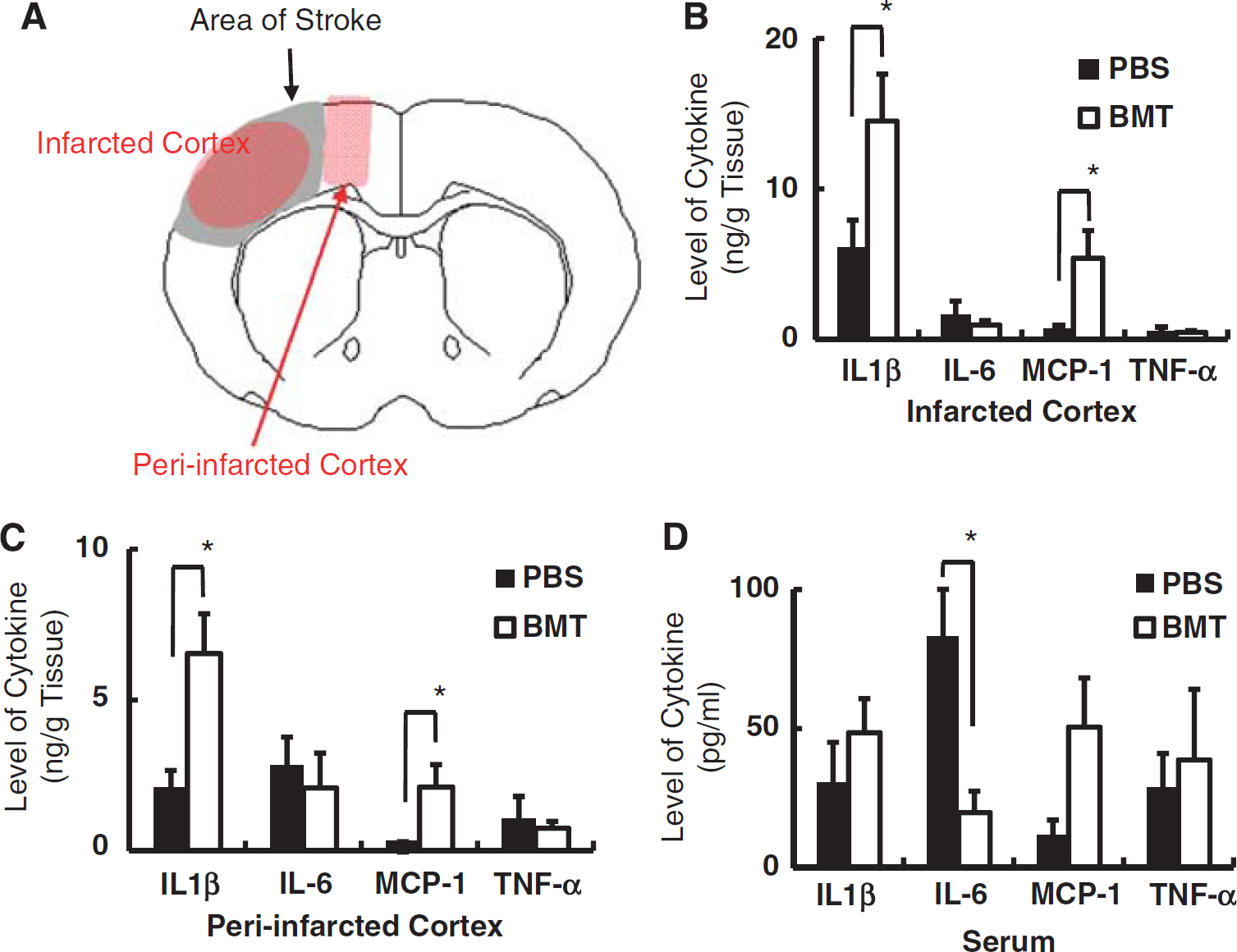
Profile of inflammatory cytokines in poststroke brain. (
Immunohistochemistry
Under deep anesthesia with a lethal dose of sodium pentobarbital (0.1 g/kg), the rats were perfused with 4% paraformaldehyde in 0.1 mol/L phosphate buffer (pH 7.4). Then, the brains were dissected out and postfixed in the same fixative for 1 day. Coronal sections (20 μm) were prepared using a vibratome (Leica, Wetzlar, Germany) and immunostained according to the standard procedures with antibodies to microtubule-associated protein-2 (MAP-2; Chemicon, Temecula, CA, USA; 1:200), Iba-1 (Wako Pure Chemical Industries, Osaka, Japan; 1:300), Lectin (Invitrogen, Carlsbad, CA, USA; 1:50) and Ki67 (BD Pharmingen, San Jose, CA, USA; dilution 1:20). Infarct volume was evaluated at 30 days after induction of ischemia as described previously (Kasahara et al, 2010). Briefly, forebrain samples were sectioned coronally, and a section at each 2 mm interval was stained with anti-MAP-2 antibody. The area staining positively for MAP-2 in each section was measured using a microscopic digital camera system (Keyence, Osaka, Japan). The MAP-2-positive volume of each hemisphere was calculated and percent stroke volume was evaluated by [(contralateral hemisphere volume)−(infarcted hemisphere volume)]/[(contralateral hemisphere volume) × 2] × 100%. Activation of microglia was quantified using anti-Iba-1 antibody as described previously (Taguchi et al, 2007) with the following modification. Briefly, the number of Iba-1-positive cells in the anterior cerebral artery area (∼0.5 mm from the border of infarction) and contralateral cortex at the exact center of the forebrain section was counted by investigators masked to the experimental protocol (three random fields in each section and the area of each field was 0.12 mm2). Cerebral vascular density was quantified using anti-Lectin antibody as described previously (Yamahara et al, 2008) with the following modification. Briefly, the number of Lectin-positive vascular structures in the anterior cerebral artery area (∼0.5 mm from the border of infarction) and contralateral cortex at the exact center of the forebrain section was counted by investigators masked to the experimental protocol (three random fields in each section and the area of each field was 0.12 mm2). Proliferation of endothelial cells was evaluated with anti-Ki67 antibody, labeled with red fluorescence (Alexa Fluor 555; dilution 1:500) and anti-Lectin antibody, labeled with green fluorescence (Alexa Fluor 488; dilution 1:500). Nuclei were stained with 4′,6-diamino-2-phenylindole (Kirkegaard & Perry Laboratories, Gaithersburg, MD, USA).
Wound Healing Model
Backs of SHR-SP were shaved 1 day before creating skin wounds. Skin wounds were placed at the center of back 4 cm caudal from neck. Under deep ketamine anesthesia, wounds were created by performing a full-thickness skin biopsy using an 8-mm punch. Back wounds were left uncovered and no local treatment was applied. To evaluate the healing process, wound diameter was measured 10 days after skin injury. For evaluation of microvasculature, semiquantitative analysis used an angiographic score, according to a modification of a previously described method (Taguchi et al, 2003, 2004b , 2007). Briefly, pictures of wounds were taken 30 seconds after creation of the wound. Horizontal and vertical lines crossing the exact center of wound were drawn, and the number of vascular structures that crossed each line was counted and the sum of the counts was defined as a semiquantitative angiographic score.
Coculture of Endothelial Cells with Bone Marrow Mononuclear Cells In Vitro
Human umbilical vein endothelial cells (HUVECs; Kurabo, Osaka, Japan) were cocultured with aged (>50 weeks old) or young (4 weeks old) SHR-SP bone marrow-derived mononuclear cells. Briefly, 24 hours before coculture, 1 × 106 of HUVEC was plated in six-well plate in HuMedia medium (Kurabo) with 10% fetal bovine serum. One hour before coculture, medium was replaced to HuMedia medium without fetal bovine serum, and 1 × 104 rat-derived bone marrow cells were plated onto the HUVEC. At 2 hours after coculture, plated bone marrow cells were removed, and HUVECs were washed twice with PBS. Activation of HUVEC by bone marrow cells was evaluated based on the level of phosphorylation of endothelial nitric oxide synthase (eNOS), extracellular signal-regulated kinase 1/2 (ERK1/2), p38 and c-Jun NH2-terminal kinase 1/2 (JNK1/2), as described by the manufacturer's protocol (Phospho eNOS (S1177), ERK1/2 (T202 and Y204), p38 (T180 and Y182) and JNK1/2 (T183 and Y185) Flex Set; BD Bioscience). Briefly, total protein was obtained from HUVEC in denaturing buffer containing protease and phosphatase inhibitors, and phosphorylation of eNOS, ERK1/2, p38 and JNK1/2 was evaluated by bead array flow cytometric analysis.
Statistical Analysis
Statistical comparisons among groups were made using the χ2 test (Figure 2A) or one-way analysis of variance followed by Dunnett test for post hoc analysis (Figures 4E and 5K). Individual comparisons were performed using Students’ t-test. Results are reported as the mean±standard error. Significance was assumed when P<0.05.
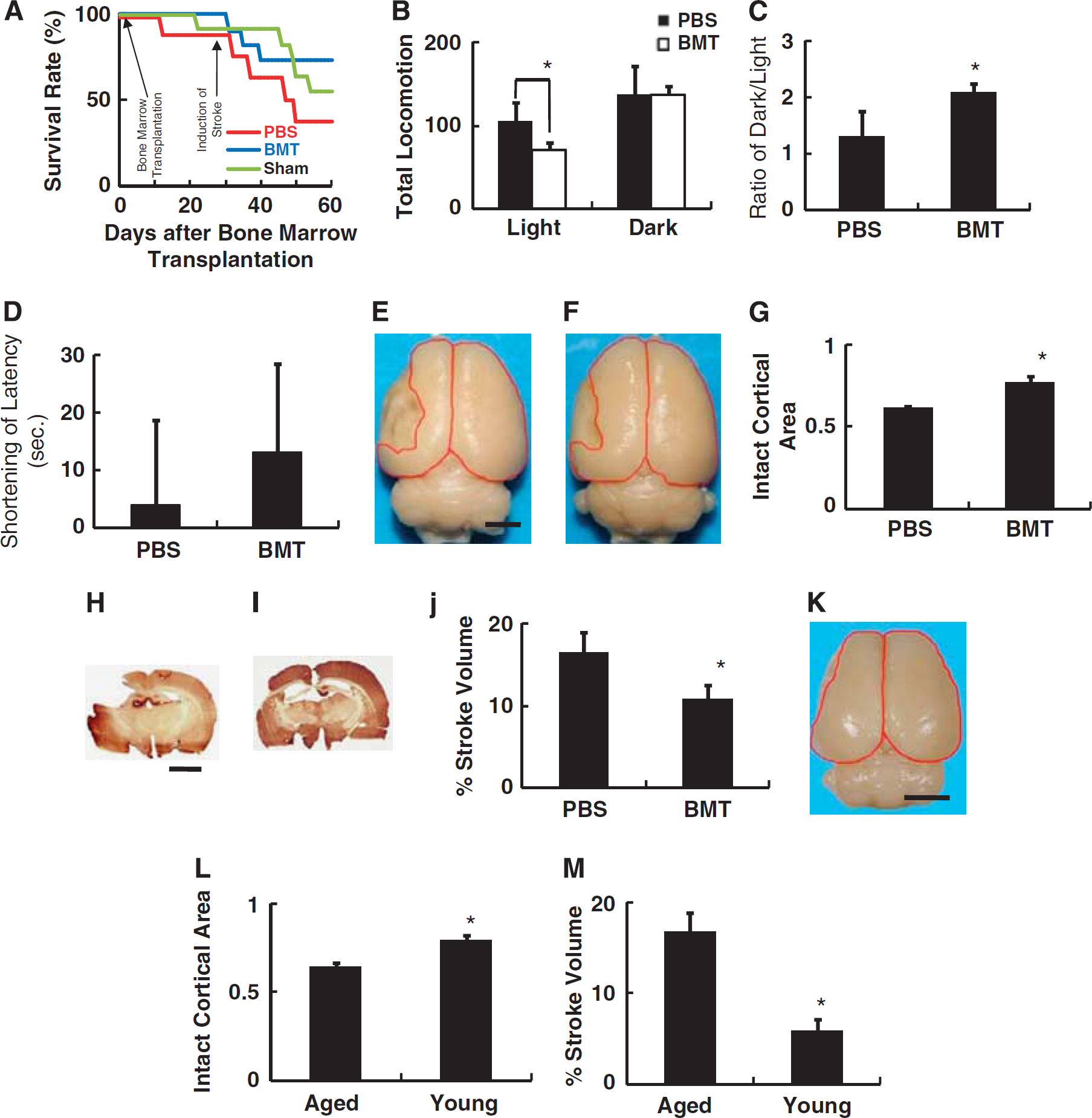
Transplantation of bone marrow from young rats into aged animals reduced ischemic damage in the poststroke period. (
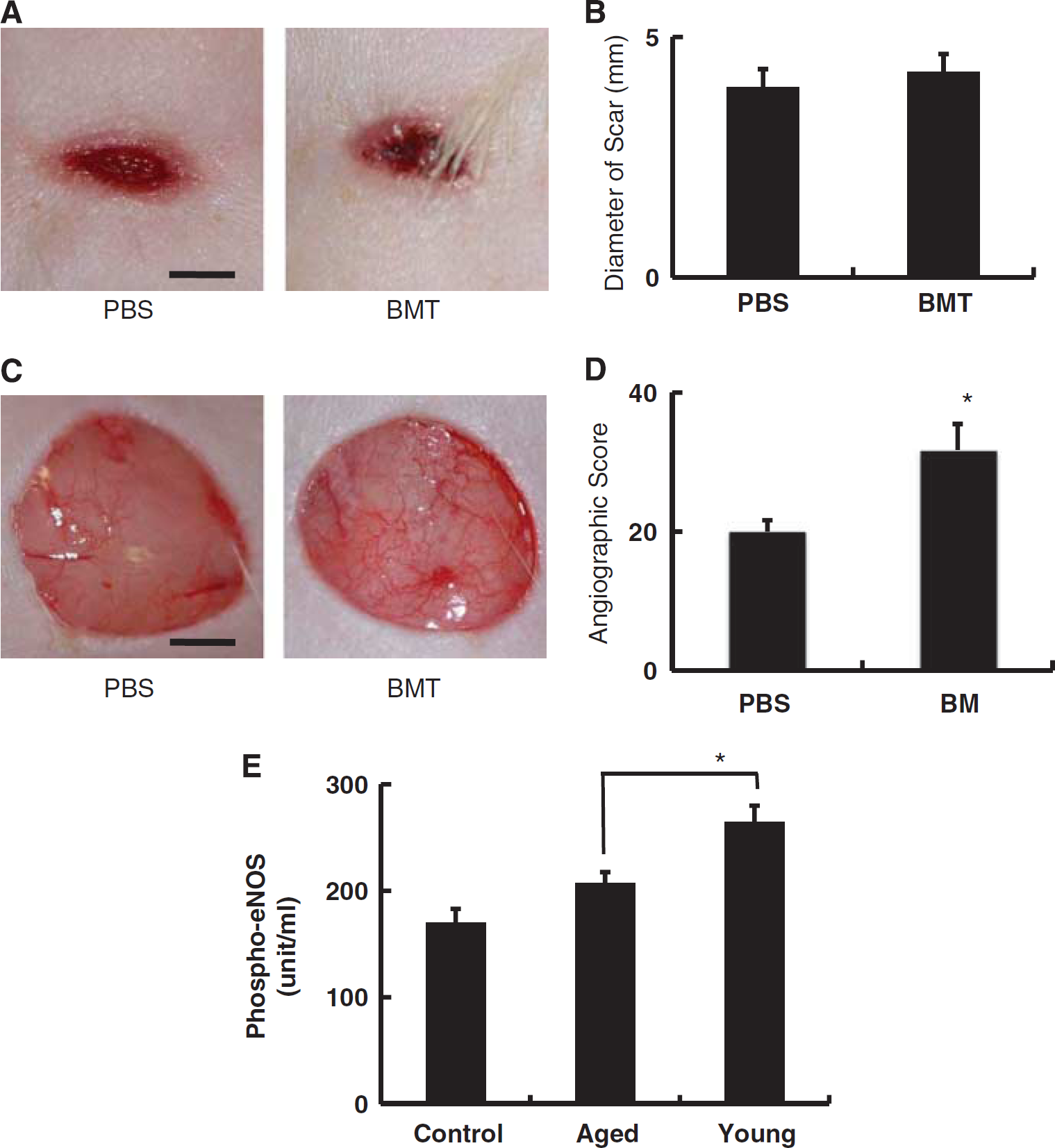
Endothelial cell activation: effect of bone marrow cells in vivo and in vitro. (
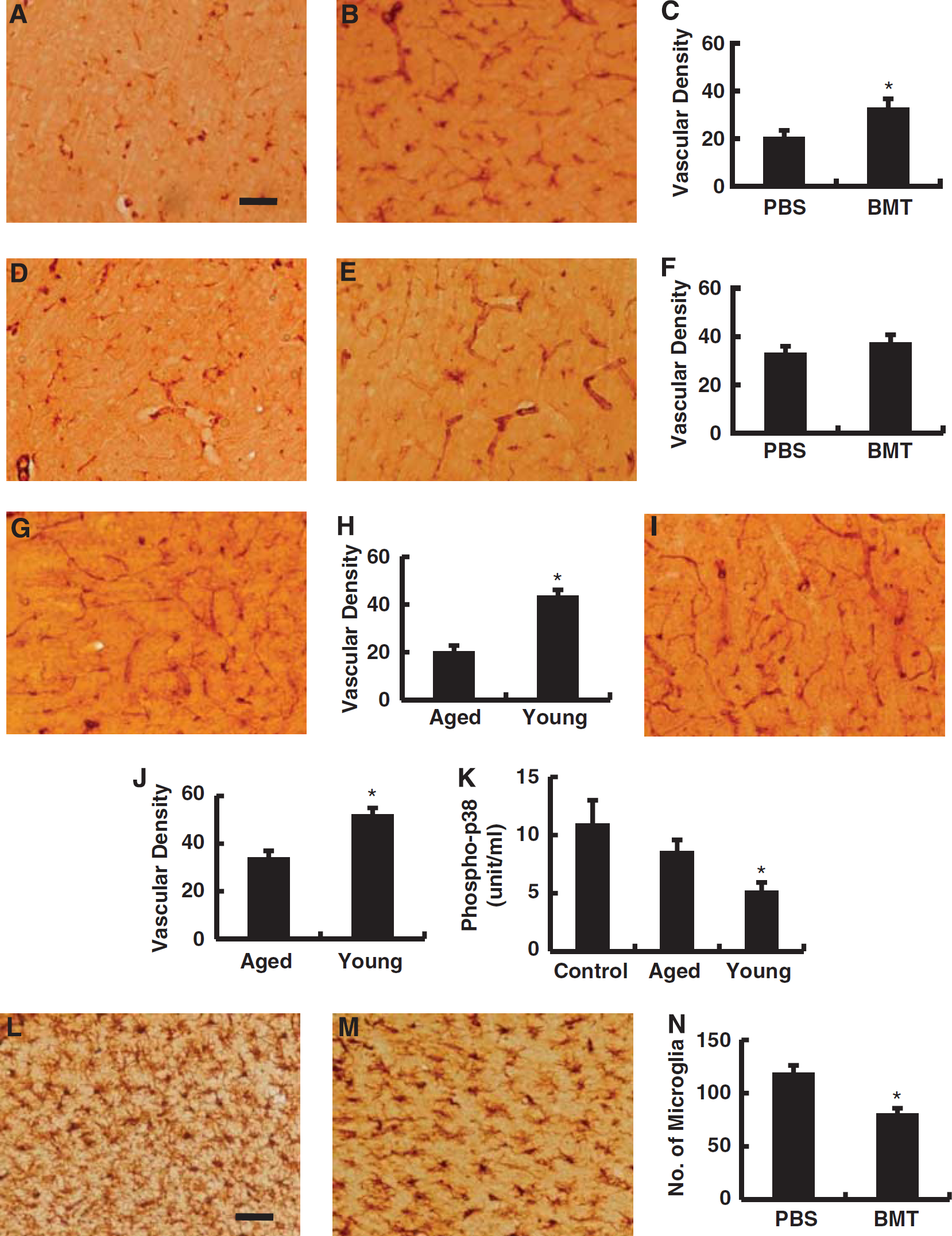
Effect of bone marrow transplanted (BMT) using bone marrow from young animals into aged animals on the density of vascular structures in the periinfarction area during the poststroke period. (
Results
Fluorescence In Situ Hybridization Analysis After Intrabone Marrow Transplantation
To evaluate transplantation efficiency of intrabone marrow plus intravenous bone marrow transplantation, the chimera ratio of circulating nuclear cells was evaluated by fluorescence in situ hybridization analysis on day 28 after cell injection. The number of Y- and X-chromosome positive donor-derived cells and Y-negative and X-positive recipient-derived cells was counted and the ratio of Y-positive donor-derived cells was evaluated. The results indicated that about 5% of circulating cells were Y-chromosome positive in female rats after intrabone marrow plus intravenous transplantation, though no Y-chromosome positive cells were observed in PBS-injected control female rats (Figure 1B). To investigate the mature cell population in blood, peripheral blood samples were analyzed to assess a series of parameters including the number of red blood cells, platelets, total white blood cells, CD4-positive T-lymphocytes, CD8-positive T-lymphocytes, NK cells, B cells, and granulocytes. No statistically significant changes were observed comparing peripheral blood of recipients who were transplanted with young bone marrow cells compared with PBS controls (Table 1).
Peripheral blood analysis after bone marrow cell transplantation
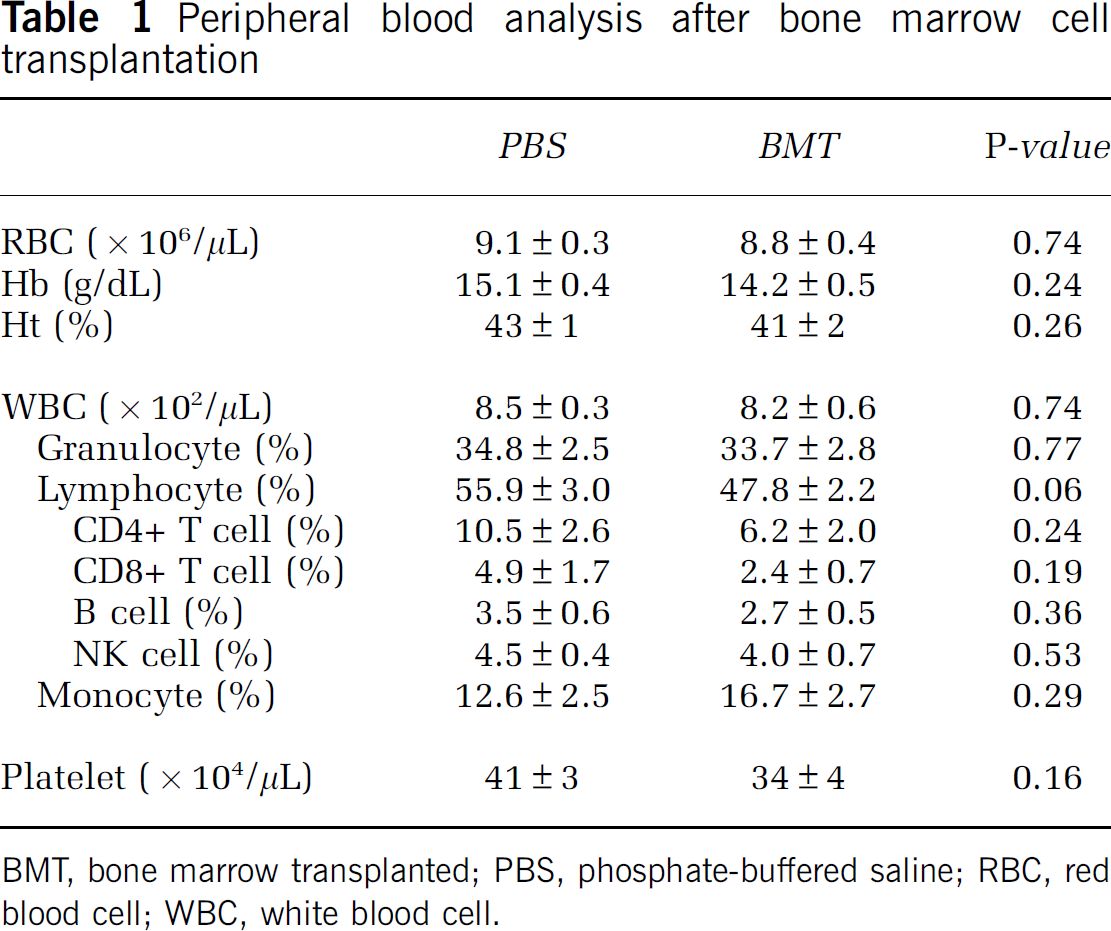
BMT, bone marrow transplanted; PBS, phosphate-buffered saline; RBC, red blood cell; WBC, white blood cell.
To analyze the cell population at the site of intrabone marrow injection, the number of SP cells in the shin bone was evaluated. The SP cells are known to include a population of immature cells of hematopoietic lineage that significantly increase with aging in bone marrow (Pearce et al, 2007). Consistent with the latter, a significant reduction in the percent of SP cells was observed in shin bone after transplantation of young bone marrow (left) compared with the nontransplanted side (right) (Figure 1C). In contrast, there was no significant difference observed between the two sides in animals subject to PBS injection (Figure 1C). The population of mature hematopoietic cells in bone marrow was also investigated, but no significant differences were observed between the left and right shin bones in rats subject to intrabone marrow plus intravenous injection of young bone marrow cells versus PBS (Figure 1D).
Intrabone Marrow Injection of Young Bone Marrow and the Survival Rate of Aged Rats
The mean lifespan of SHR-SP has been shown to be significantly shorter than that of their wild-type counterparts (Brandle et al, 1997). Consistent with this observation, by day 30 after bone marrow or PBS transplantation, 13% and 10% of rats in PBS-treated and nontreated sham-operated group had expired, respectively. In contrast, no deaths were observed in rats transplanted with young bone marrow cells. On day 30 after transplantation of young bone marrow cells or injection of PBS, cerebral ischemia was induced. By day 60, a total of 63%, 27%, and 45% of rats had expired in the PBS, bone marrow transplanted and sham-operated groups, respectively (Figure 2A). There was no significant difference in survival between groups (P=0.31). Individual comparisons between each group using the χ2 test also did not show statistically significant differences, including PBS versus bone marrow transplanted groups (P=0.12).
Intrabone Marrow Injection of Young Bone Marrow Reduced Cortical Brain Damage After Stroke
Cerebral ischemia in a rodent model induced by ligation of the middle cerebral artery at a site distal to the striatal branches mainly causes infarction of the cerebral cortex (Zhang et al, 2006). Dysfunction of the cortex is closely linked to disinhibition of behavior (Farkas et al, 2003). On day 14 after induction of stroke, cortical function was evaluated using the open field task. Compared with PBS-injected rats, significant improvement of behavior, as characterized by suppression of locomotion in the presence of light, was observed in rats after transplantation with bone marrow from young animals (Figure 2B). Analysis of the response of animals placed in the dark also revealed a significant improvement in rats after bone marrow transplantation (Figure 2C).
Next, spatial learning/memory was evaluated using a water maze assay. The change in mean time to escape the submerged platform was evaluated; only a nonsignificant trend favoring improvement was observed in the group subjected to bone marrow transplantation (Figure 2D). The apparent discrepancy between functional recovery (locomotion) and cortical function/learning (water maze) might reflect variations in learning capacity in aged rats compared with a younger group of SHR-SP animals (Meaney et al, 1995).
On day 30 after induction of stroke, images of whole brain were captured to evaluate the extent of cortical damage/recovery morphologically. Compared with PBS controls (Figure 2E), significant reduction of ischemic cortical damage was observed in animals subject to bone marrow transplants (Figures 2F and 2G). To confirm the effect of transplanting young bone marrow cells into aged rats on reduction of brain damage poststroke, sequential brain sections were prepared and stroke volume was evaluated with MAP-2 staining. Compared with PBS controls (Figure 2H), significant reduction of stroke volume was observed in animals subject to bone marrow transplantation (Figures 2I and 2J). It is notable that stroke area was limited at the level of the cortex in rats who had received bone marrow transplants, in contrast to controls in which ischemic damage expanded to reach the lateral ventricle.
To evaluate the effect of aging and rejuvenation of the bone marrow with cells from young animals on the size of cerebral infarcts, cerebral infarction was induced in 8-week-old SHR-SP (Figure 2K). Quantitative analysis revealed a significant reduction in the area of infarction in young rats, compared with aged (>50 weeks; Figure 2L). The reduction in stroke volume was confirmed by sequential sections with MAP-2 staining (Figure 2M).
Changes in Expression of Proinflammatory Cytokines After Intrabone Marrow Injection of Bone Marrow Cells from Young Animals into Old Stroke-Prone Spontaneously Hypertensive Rats
To investigate possible causes of apparent reduction in cortical damage observed in rats transplanted with bone marrow cells from young animals, the level of inflammatory cytokines, including IL-1β, IL-6, tumor necrosis factor-α, and MCP-1, was evaluated 3 days after stroke. The areas from which tissue samples were harvested are shown in Figure 3A. Although functions of each inflammatory cytokine are multiple, increased levels of IL-1β after stroke have been associated with mainly negative effects, including inflammation, apoptosis, and edema (Holmin and Mathiesen, 2000). Similarly, MCP-1 has been ascribed a principally negative function (Chen et al, 2003). In contrast, tumor necrosis factor-α has been shown to have both neuroprotective (Sullivan et al, 1999) and neurotoxic effects (Yang et al, 1998). The IL-6 has been suggested to display principally positive effects (Loddick et al, 1998). In the infarcted cortex, a significant increase in levels of IL-1β and MCP-1 was observed in rats subject to bone marrow transplantation compared with PBS-treated controls (Figure 3B). In contrast, there was no significant change observed in levels of IL-6 and tumor necrosis factor-α. Similarly, increased levels of IL-1β and MCP-1 were observed in rats subjected to bone marrow transplantation when the periinfarcted cortex was studied (Figure 3C). The increase in levels of the latter cytokines in the infarcted and periinfarcted cortex and serum, contrast with a decrease of IL-6 levels in serum of rats subject to transplantation (Figure 3D; there was no significant change in IL-6 levels in the infarcted and periinfarcted cortex). These results suggest that modulation in expression of inflammatory cytokines (i.e., favoring recovery/decreased inflammatory profile) did not occur, and, thus, is not likely to explain the beneficial effect of the response to cerebral ischemia observed in older SHR-SP transplanted with bone marrow cells from young SHR-SP.
Intrabone Marrow Injection of Bone Marrow Cells from Young Rats into Old Stroke-Prone Spontaneously Hypertensive Rats Animals Does Not Affect the Host Response to Cutaneous Wounding
To more generally address the issue of the host reparative response in old SHR-SP subject to transplantation of bone marrow cells from young animals, we used a model of cutaneous wound repair. We monitored repair of full-thickness skin wounds made on the backs of animals using a biopsy punch. On day 10 after injury, the size of the scar was measured. No significant difference in the size of wounds was observed between animals transplanted with young bone marrow cells or PBS controls (Figures 4A and 4B). However, low power micrographs soon after injury (30 seconds) displayed many more visually apparent vascular structures in wounds from animals receiving transplanted cells from young animals compared with PBS controls (Figures 4C and 4D). As the latter occurred quickly after injury, it could be considered an acute vascular response to the skin wound (i.e., activation of microvasculature involving recruitment of additional channels or dilation of channels already subject to blood flow) rather than formation of new vascular channels, which would, presumably, require additional time.
The above observation suggested the possibility that activation of endothelial cells might occur more readily in the presence of bone marrow cells. To assess this possibility in vitro, HUVECs were cultured in the presence of bone marrow mononuclear cells derived from young (4 weeks old) or aged (50 weeks old) SHR-SP. As an index of endothelial activation, phosphorylation of eNOS in HUVECs was monitored. Endothelial cells cocultured with bone marrow-derived mononuclear cells from young animals displayed a higher level of phospho-eNOS, compared with HUVECs exposed to bone marrow cells from aged rats (Figure 4E).
Transplantation of Bone Marrow from Young Animals Impacts on the Cerebral Microvasculature Poststroke
To investigate the possible mechanisms underlying reduced brain damage observed poststroke in aged rats subject to transplantation with bone marrow cells from young animals, cerebrovascular density and proliferation were studied 30 days after induction of stroke. Compared with PBS-injected rats (Figure 5A), vascular density in the peristroke area of transplanted animals showed a significant increase in vascular density (Figures 5B and 5C). In contrast, there was no significant difference in the density of vasculature in the contralateral cortex comparing animals treated with PBS (Figure 5D) or bone marrow transplantation (Figures 5E and 5F). To evaluate the effect of aging on vascular density, cerebral infarction was induced in 8-week-old SHR-SP, and the density of vasculature was evaluated. The results demonstrate a significantly higher vascular density in the peristroke area (Figures 5G and 5H) and contralateral cortex (Figures 5I and 5J) in young rats compared with aged rats. These results are consistent with previous reports that cerebrovascular density decreases with aging (Hutchins et al, 1996; Lynch et al, 1999; Sonntag et al, 1997). To investigate proliferation of endothelium in the cerebrovasculature of the cerebral cortex, cells coexpressing Ki67 and Lectin were investigated. However, the number of cells expressing both markers was too small (mean number of double-positive cells was less than one cell per section in both group) to perform quantitative analysis and obtain meaningful data.
To evaluate the effects of bone marrow cells on endothelial cells, activation of MAP kinases was evaluated in vitro. The MAP kinases are serine/threonine-specific protein kinases that regulate critical cellular activities, such as mitosis, differentiation, and cell survival/apoptosis (Kant et al, 2006). Exposure of cultured endothelial cells to bone marrow mononuclear cells from young animals resulted in decreased levels of phospo-p38 compared with endothelial cells exposed to bone marrow from older rats (Figure 5K). As the latter is consistent with decreased activation of p38 (activation of p38 in endothelium induces apoptotic death through a mitochondrial pathway) (Mehta et al, 2007), it would suggest a basis for increased survival of endothelial cells in the presence of bone marrow mononuclear cells from young animals. In contrast, there were no significant differences in the phosphorylation of two other MAP kinases, ERK1/2 and JNK1/2, comparing the two groups (data not shown). These results are consistent with our in vivo observation that transplantation of young bone marrow into aged mice has little impact on endothelial proliferation, but enhances the density of microvasculature in the peristroke area.
To investigate the effect of transplantation of bone marrow from young mice into aged mice on microglia, sections of poststroke brain were stained with anti-Iba-1 antibody. Compared with PBS-treated rats (Figure 5L), a significant decrease in microglia was observed in the peristroke area in bone marrow transplanted rats versus nontransplanted animals (Figures 5M and 5N). In contrast, no significant difference was observed in the number of microglia in the contralateral cortex comparing PBS-treated rats and bone marrow transplanted rats (29.1±1.6 and 27.2±1.7/field, respectively; P=0.45).
Discussion
We have demonstrated that partial rejuvenation of bone marrow from aged rats with bone marrow-derived mononuclear cells from young animals reduces damage after experimental cerebral ischemia in the SHR-SP rat strain.
Intravenous infusion of bone marrow-derived cells without pretreatment for transplantation (such as irradiation) is associated with a very low seeding efficiency of transplanted cells into host bone marrow. However, pretransplant radiation of individuals not requiring complete repopulation of bone marrow with transplanted cells (as would be true for patients with cerebral ischemia, especially during the immediate period of ischemic stress) is not likely to be clinically acceptable, due to short- and long-term complications. Thus, use of intrabone marrow administration of bone marrow-derived mononuclear cells, in addition to intravenous administration of bone marrow cells, was used for our studies. Using our protocol (i.e., no pretreatment for bone marrow transplantation, followed by intravenous and intrabone marrow administration of bone marrow cells), the level of engraftment resulted in ≈5% chimera of transplanted versus host cells in peripheral blood. Furthermore, analysis of peripheral blood from transplanted animals showed no significant difference in mature cell populations between the two groups. Although the overall change in health status of the aged rats because of transplantation of bone marrow cells is difficult to evaluate, these results indicate that the beneficial effect of transplanting bone marrow-derived mononuclear cells from young SHR-SP into old animals, in terms of limiting brain injury following ischemia, is not simply due to optimization of the level of circulating mature cells.
Initially, we suspected that our bone marrow transplant protocol would result in a change in the cytokine response to cerebral ischemia consistent with diminished inflammation and enhanced repair. However, levels of IL-1β and MCP-1, considered deleteriously inflammatory (Chen et al, 2003; Holmin and Mathiesen, 2000), increased (Figure 3), and levels of IL-6, considered protective (Loddick et al, 1998), was not increased (Figure 3), following bone marrow transplantation. These findings indicate that beneficial effects of transplanting young bone marrow into aged animals are not likely due to modulating the profile of inflammatory cytokines at the site of cerebral infarction or in the serum.
Vascular endothelial dysfunction in aging has been shown to be correlate with impaired vasodilatation (LeBlanc et al, 2008) and reduced expression of NOS (Smith and Hagen, 2003). Although some aspects of cell senescence are likely to be irreversible, certain endothelial properties can be maintained/restored with antioxidants (Smith and Hagen, 2003). Furthermore, exposure to a ‘young systemic environment’ has been shown to rejuvenate aged progenitor cells in rats (Conboy et al, 2005). Taken together with these previous observations, our results indicate that partial reconstitution of bone marrow from aged rats with cells from young animals improves the host response to cerebral ischemia, probably in part through a beneficial effect on endothelial function. Although bone marrow cells have the potential to differentiate into endothelial cells (Asahara et al, 1997), the half-life of endothelial cells is relatively long and differentiation of circulating cells into endothelium is relatively rare (Taguchi et al, 2004b ). Thus, the principal effect of young progenitors in the bone marrow may be to promote an environment in the systemic circulation conducive to vascular activation, as evidenced by increased levels of phospho-eNOS (i.e., the results of our in vitro studies with cultured HUVEC exposed to mononuclear bone marrow cells from young versus old rats; Figure 4E). In addition to eNOS, young bone marrow cells were shown to decrease activation of p38 MAP kinase in cultured endothelium, a pathway leading to cell death (Mehta et al, 2007). In contrast, no significant change in activation/inactivation of other MAP kinases, ERK1/2 or JNK, was observed in endothelial cells exposed to bone marrow cells from young animals in vitro. In a previous report, inactivation of p38, but not JNK, was shown to suppress endothelial cell death after hypoxia/reoxygenation injury (Lee and Lo, 2003). Taken together, our data thus far suggests that the beneficial effect of bone marrow cells from young animals may be due, at least in part, to effects on the vascular endothelium, especially at the level of cerebral microvasculature. This concept is consistent with our observation of an increased density of vascular structures in the periinfarct area of poststroke brain from aged rats transplanted with bone marrow cells from young animals. However, the precise mechanisms linking bone marrow-derived immature cells to enhanced function of the cerebral microcirculation remains to be determined (Pearson, 2009). In view of the likely impact of inflammatory mechanisms on reparative mechanisms in stroke, another important observation might be the decrease of microglial invasion/activation observed in poststroke aged animals transplanted with bone marrow from young animals.
Our results suggest a novel strategy for enhancing the host response to ischemia in aged patients, provided these observations in a rodent model can be translated to humans. If such an extrapolation to man is possible, the source of ‘young’ bone marrow is relatively easily obtained through collection and preservation of autologous cord blood or bone marrow cells harvested earlier in life. In addition, an alternative approach might be induction of hematopoietic stem cells using autologous induced pluripotent stem (iPS) cells (Hanna et al, 2007). Although the fundamental mechanisms underlying senescence of mammalian cells (and senescence of the vasculature) remains to be elucidated, our findings indicate that impairment of the vascular response in aged individuals may be partially restored through transplantation with bone marrow from young animals. In addition to possible therapeutic application in patients with an evolving stroke, rejuvenation of bone marrow may also have a preventive role in those at high risk for stroke, such as individuals with recurrent transient ischemic attacks.
