Abstract
Recent studies suggest that proliferation in the adult forebrain subventricular zone increases in response to a forebrain stroke and intraventricular infusions of growth factors enhance this response. The potential for growth factor infusions to regenerate the damaged motor cortex and promote recovery of motor function after stroke has not been examined. Here, we report that intraventricular infusions of epidermal growth factor and erythropoietin together, but not individually, promote substantial regeneration of the damaged cerebral cortex and reverse impairments in spontaneous and skilled motor tasks, in a rat model of stroke. Cortical regeneration and functional recovery occurred even when growth factor administration was delayed for up to 7 days after the stroke-induced lesion. Cell tracking demonstrated the contribution of neural precursors originating in the forebrain subventricular zone to the regenerated cortex. Strikingly, removal of the regenerated cortical tissue reversed the growth factor-induced functional recovery. These findings reveal that specific combinations of growth factors can mobilize endogenous adult neural stem cells to promote cortical tissue re-growth and functional recovery after stroke.
Introduction
The isolation and identification of multipotent neural stem cells (NSCs) from the adult forebrain (Reynolds and Weiss, 1992) and their localization to the subventricular zone (SVZ) of the lateral ventricles in both rodents and humans (Doetsch et al, 1999; Morshead et al, 1994; Sanai et al, 2004) has spurred significant interest in the biology and therapeutic potential of rostral forebrain neurogenesis (Alvarez-Buylla and Lim, 2004; Lie et al, 2004; Lindvall et al, 2004). In rodents and non-human primates, the normal biologic function of SVZ NSCs is the generation of new olfactory interneurons (Doetsch and Hen, 2005). Recently, a series of studies have provided evidence that a middle cerebral artery occlusion model of stroke results in enhanced proliferation within the SVZ, neuronal migration towards the ischemic cortex and striatum and subsequent differentiation into a few new neurons (Arvidsson et al, 2002; Jin et al, 2003; Parent et al, 2002; Zhang et al, 2004). Clearly, this phenomenon is insufficient to repair significant brain damage; however, a select combination of signals might enhance this process such that substantial and functional tissue regeneration can occur in major structures, such as the cerebral cortex.
We have previously found that epidermal growth factor (EGF) is a powerful mitogen for adult forebrain SVZ NSCs, both in vitro and in vivo (Craig et al, 1999; Morshead et al, 1994; Reynolds and Weiss, 1992) and may be particularly supportive of precursor cell re-direction through the generation of radial glial cells that support neuronal migration into the adult forebrain parenchyma (Gregg and Weiss, 2003). We have further reported that erythropoietin (EPO) can promote the differentiation of neurons from adult NSC progeny (Shingo et al, 2001). This led us to hypothesize that enhanced NSC proliferation, migration and neuronal differentiation, through the combined actions of EGF + EPO, might contribute to tissue regeneration and functional recovery after stroke. To test this hypothesis, we have developed a battery of behavioral tests that are sensitive to forebrain cerebral stroke induced by devascularization or middle cerebral artery occlusion (Gonzalez and Kolb, 2003), and have previously shown that intracerebroventricular infusions of inosine (Chen et al, 2002) or poststroke subcutaneous injections of nicotine (Gonzalez et al, 2006) enhanced performance in some of these tests, although functional improvement was incomplete. Here we report that intraventricular infusions of EGF + EPO, together but not alone, stimulate tissue re-growth and recovery of motor function after a focal, permanent devascularization (stroke) to the motor cortex. This motor recovery was greater than that seen by any previous treatments on animals with similar functional loss. Reversal of functional recovery after the removal of the newly generated tissue provides evidence for a direct link between growth factor activation of NSCs, cortical tissue re-growth and functional recovery after stroke.
Materials and methods
Animals
Male Long-Evans rats (N = 126), 90 to 110 days old at the beginning of the studies, were used as subjects. The rats were housed and maintained according to the regulations of the Canadian Council on Animal Care. Rats with motor cortex stroke were treated with artificial cerebrospinal fluid (aCSF) (n = 15), EGF (n = 10), EPO (n = 6), or EGF + EPO (n = 11) and compared with normal controls (n = 10) on the forepaw asymmetry and inhibition tests. A subset of these was trained on skilled reaching: aCSF (n = 10), EGF (n = 5), EPO (n = 6), EGF + EPO (n = 6) and control (n = 10). A second set of animals received EGF + EPO or aCSF at different time points after stroke (n's = 5/group). A third set of animals had a second surgery 4 weeks after EGF + EPO infusions (n's = 6/group). The brains of a final subset were used for anatomic studies.
Surgery
Under sodium pentobarbital anesthesia (60 mg/kg), a rectangular hole was drilled into the frontal and parietal bones running from + 3 to −4 mm anterior/posterior to the Bregma and running laterally from 1.5 to 4.5 mm from midline. The focal, permanent devascularization (stroke) has been described in detail elsewhere (Gonzalez and Kolb, 2003). Briefly, the dura was removed and a sterile saline-soaked cotton swab was used to wipe the pia and attached blood vessels from the cortical surface. For behavioral studies, surgery was always performed on the hemisphere contralateral to the preferred paw, which was determined in presurgery reach training. Infusion cannulae (23 gauge) were implanted in the lateral ventricle (LV) ipsilateral to the preferred paw using skull coordinates −0.8 anterior—posterior from the Bregma, 1.4 lateral from the midline, and 3.5 ventral from the skull surface. The cannulae were connected to filled, pretested, s.c. placed osmotic minipumps (Alzet, Cupertino, CA; model # 2001) via coiled flexible polyethylene tubing. These were filled with either vehicle (artificial cerebral spinal fluid plus 0.1% bovine serum albumin) or with the aCSF and EGF (10 μg/mL) or with EPO (1365 IU/mL). There was no mortality as a consequence of the surgeries; however, for 24 h after the devascularization animals were lethargic and sensitive to touch, after which they returned to their presurgical behavior.
Behavioral Training and Testing
Rats with focal stroke to the motor cortex, due to a devascularizing lesion, show chronic, severe deficits on a variety of motor tasks (Gonzalez and Kolb, 2003). These behavioral deficits include an asymmetry in forelimb use (favoring the ipsilateral forelimb) during vertical exploration of a small cylinder (Gharbawie et al, 2004), a failure to inhibit forelimb use during swimming, leading to paddling with the contralateral forelimb while the ipsilateral forelimb remains motionless under the chin (Kolb and Whishaw, 1983), and an impairment in skilled reaching to retrieve bits of food (Whishaw et al, 1991). To assess the effect of intraventricular infusions of growth factors on these behaviors after a devascularizing lesion, rats were pretrained on the asymmetry, forelimb inhibition and reaching tasks and then tested weekly for 4 weeks and again on week 6 after the lesion.
Subjects were maintained on a restricted food regime in which each animal obtained 20 g of food per day (normal daily consumption ranges from 18 to 25 g), but only an hour after the testing session was completed. Their body weight was maintained at approximately 95% to 98% until the completion of the behavioral testing. Animals were trained on all tests before surgery and the animals were assigned to groups to ensure that behavioral scores were balanced across groups before surgical treatments.
Cylinder Test (Forelimb Asymmetry)
Forelimb use for weight support during explorative activity was examined by placing the rats in a transparent cylinder 20 cm in diameter and 30 cm high for 3 mins (Schallert, 2000). The animals were individually placed in the cylinder during the 3 mins of each testing session. A mirror was placed underneath the cylinder at an angle to allow the experimenter to videotape the animal's activity from a ventral view. The cylindrical shape encouraged vertical exploration of the walls with the forelimbs, but the walls were high enough so that animals could not reach the top. Forelimb use was measured during vertical exploration. Each forepaw contact with the cylinder wall was counted. The asymmetry score of forelimb use in wall exploration was calculated as the percent preference for the paw ipsilateral to the lesion, or for the control rats, the paw that they used for reaching: preference = ipsilateral paw/(ipsilateral paw + contralateral paw) × 100.
Swimming Task (Forepaw Inhibition)
Rats were trained for 2 days before the lesions to swim to a visible platform located at the end of a rectangular aquarium (120 × 43 × 50 cm3). The water in the pool was maintained at 25°C at all times. During the training phase, animals were released from the opposite end of the tank and consecutive trials were given until they swam directly to the platform without touching the walls of the aquarium (approximately 10 trials for each animal). Kolb and Whishaw (1983) described that normal animals held their forelimbs immobile under their chins while using their hind limbs to propel through the water. By the end of the second day, animals were quite familiar with the tank and the task; therefore, inhibition of the forepaws was observed. On the third day, three trials were videotaped for each animal as they swam directly to the platform.
Disruption to the normal swim pattern (swim score) was quantified by counting the number of strokes made by each forelimb on each trial. The swim score of forelimb inhibition was calculated for each group by subtracting the number of contralateral forelimb strokes minus the ipsilateral number of forelimb strokes (i.e., contralateral forelimb strokes minus ipsilateral forelimb strokes).
Tray Reaching Boxes and Training
Training boxes were made of plexiglass with dimensions 26 cm high, 28 cm deep and 19 cm wide. The front of the boxes was constructed of 2 mm bars separated from each other by a 9 mm gap. Clear plexiglass tops allowed access to the inside of the box. A 4 cm wide and 0.5 cm deep tray was mounted in front of the bars. The tray contained food fragments weighing approximately 30 mg each. Animals had to reach between the bars, grasp the food and retract it where they were able to freely eat. Subjects were trained for a total of 15 days before the lesions to obtain a stable base line before the surgeries.
Reaching success was calculated as follows: If the rat made a reaching movement (forepaw inserted through the bars, but no food was grasped or the food was dropped), the movement was scored as a ‘reach’, whereas if the rat obtained the food and consumed it, the movement was scored as a ‘reach’ and a ‘hit’. Success was calculated then as success percent = (‘hit’/‘reach-hit’ × 100).
Single Pellet Reaching
This test was used only in the final behavioral study in which the growth factor-induced cells were removed. Boxes were made of clear Plexiglas, with the dimensions 45 cm deep by 14 cm wide by 35 cm high. In the center of each front wall was a 1-cm-wide slit that extended from 2 cm above the floor to a height of 15 cm. On the outside of the wall, in front of the slit, mounted 3 cm above the floor, was a 2 cm-wide shelf. Two indentations on the floor of the shelf were located 2 cm from the inside of the wall and were centered on the edges of the slit where rats could reach. Food pellets (45 mg Rodent Chow food pellets, Bioserve Inc.) were placed in the indentation contralateral to the limb with which the rat reached (Whishaw et al, 1991). After each reach, a short pause preceded the presentation of the next pellet, and an additional pellet could be dropped in the back of the box. This would encourage animals to return to the back of the box after each reach, which forced them to reposition themselves and prepare for the next reach. Success scores were computed as follows: percent success = (number of pellets retreived/20) × 100.
Retroviral Labeling
A replication-deficient retrovirus (RV) (a kind gift from Dr Alaine Garnier) engineered to introduce β-galactosidase (β-gal) was utilized to label proliferating SVZ cells in vivo as a non-diluting marker. Stereotaxic unilateral injections of 6 μL of RV (viral concentrate titer of approximately 106 active virions per mL) were made directly into the LV ipsilateral to the stroke lesion before stroke at a rate of 0.2 μL/min using a Hamilton syringe at the following coordinates about bregma at a tooth bar setting of −2: anteroposterior (A/P), −0.5 mm; mediolateral (M/L), −1.5 mm; and dorsoventral (D/V), −5.0 mm. At various times poststroke, rats were killed with an overdose of sodium pentobarbital and transcardially perfused with 4% paraformaldehyde in 1 × Dulbecco's phosphate-buffered saline, pH 7.3. Serial 30 μm coronal cryostat brain sections were processed for 5-bromo-4-chloro-3-indolyl-β-
β-gal Polymerase Chain Reaction
The newly generated cortical tissue from day 40 animals was removed and divided into two regions comprising the core (center) of the newly generated cortical tissue and the outer surrounding region using a disposable razor blade. A region of cortex similar in dimension to the newly generated cortical tissue was also dissected out of the contralateral hemisphere. Each sample of tissue was digested and its DNA purified using the DNeasy Tissue Kit (Qiagen). Subsequently, PCR was performed on equal amounts of purified sample DNA. Each final reaction mixture (50 μL) contained sample DNA (1 μg), primers (0.2 μmol/L each), 2.5 mmol/L MgCl2, 0.15 mmol/L dNTP, dimethylsulfoxide (0.6 mol/L), 10 × PCR buffer, Taq DNA polymerase (Fermentas) and sterile deionized water. The sense and antisense primers used for PCR amplification were as follows: LacZ sense 5′-GAATGGTGCAGCGC GATCG, and LacZ antisense 5′-AGGAGCTCGTTATCGC TATGA. Cycling conditions were 94°C for 5 mins, 40 cycles of 94°C for 30 secs, 60°C for 1 min, 72°C for 3 mins and a final extension at 72°C for 5 mins. LacZ Primers generated a final PCR product of 700 bp. PCR products were analyzed by electrophoresis on a 2% agarose gel and visualized with ethidium bromide.
5-bromo-2′-deoxyuridine and Immunocytochemistry
Rats were injected with 5-bromo-2′-deoxyuridine (BrdU) (Sigma-Aldrich, Oakville, ON, Canada) (60 mg/kg, intraperitoneally) 1 day before stoke, once every 3 h for 15 h and killed 1 h after the final injection (day −1) or at various times during the recovery period (days 3, 11, 18, and 42 after stroke). Brains were removed and every tenth serial 15 μm coronal cryostat section was mounted and processed for immunofluorescent double-labeling with neuronal antibodies. The sections were initially permeabilized with 0.3% Triton X-100 in Stockholm phosphate-buffered saline for 20 mins, and blocked for 1 h using 10% normal goat serum in Stockholm phosphate-buffered saline. The sections were then incubated with monoclonal mouse antineuronal nuclei (NeuN) primary antibody (1:100; Chemicon) or polyclonal rabbit anti-glial fibrillary acidic protein primary antibody (1:400; Biomedical Technologies Inc., Stoughton, MA, USA) followed by (TRITC)-conjugated goat anti-mouse (1:200; Jackson ImmunoResearch) or fluorescein isothiocyanate -conjugated goat anti-mouse (1:200; Jackson ImmunoResearch). Rat monoclonal anti-BrdU primary antibody (1:100; Abcam) and fluorescein isothiocyanate-conjugated donkey anti-rat secondary antibody (1:200, Jackson ImmunoResearch) was used for double labeling. Sections were incubated with Hoechst (Sigma, Oakville, ON, Canada) (0.725 μg/mL). Confocal imaging system Zeiss LSM 510 was used to confirm the co-localization of fluorescent immunostaining within cells.
To quantify the numbers of labeled cells within the SVZ, the striatum and the contralateral cortex, the total numbers of cells within one 0.96 mm2 region were counted from two to four sections between the rostral genus of the corpus callosum and the crossing of the anterior commissure, which was at about anterior—posterior coordinates +1 to 0 and thus well within the devascularized region. Numbers were averaged per animal and expressed per unit area. Within the cortical lesion area and in the regenerated cortical tissue after growth factor treatment, the numbers of cells within one 0.96 mm2 ocular grid unit area in four regions (superior and inferior central core regions of the cortical lesion or newly generated cortical tissue and a medial and lateral portion of the cortical lesion or newly generated cortical tissue) from four sections per animal. The number of Hoechst and NeuN labeled cells was counted using the fractionator sampling method (West et al, 1991) to avoid possible bias associated with profile counting. Sampled sections were viewed with a × 40 objective using an Olympus Microsuite™-B3SV tissue analysis software. The localization of a cell nucleus within the z axis was determined and the position of each cell nucleus in the dissector volume probe was determined by focusing in one direction from the upper to the lower section surface. For example, a neuron was counted if the nucleus associated with that neuron first came into focus within the optical dissector counting frame (i.e., a neuron was not counted if the nucleus associated with that neuron came into focus within the guard zone or if the nucleus was touching the left or bottom side of the dissector frame). The guard zone is an area at the top and bottom of the histologic section in which neurons are not counted to avoid errors in estimation because of cutting artifact.
Statistical Analysis
Statistical analyses for the behavioral tests were performed using analyses of variance. Homogeneity of variance was established using the F-max test. Sample sizes were based on our previous work. The sample sizes in the EGF, EPO, and EGF + EPO behavioral study were larger reflecting our reporting of the original study and its replication as a single experiment. To reduce the risk of type I errors, the post hoc comparisons (Fisher's LSD, P < 0.05) were restricted to those comparisons described.
Results
EGF + EPO Infusions Lead to Tissue Regeneration in The Motor Cortex after Focal Stroke
A permanent, focal stroke induced by pial blood vessel devascularization (Kolb et al, 1997) leads to the death of cortical cells and underlying white matter and, subsequently, in the formation of a lesion cavity that extends down to the corpus callosum after 7 to 14 days (Gonzalez and Kolb, 2003). After such a stroke lesion to the motor cortex, we asked whether intraventricular infusions of EGF for 7 days, followed by EPO for 7 days, into the contralateral ventricle, would result in new cortical tissue (Figures 1A and 1B), when compared with intraventricular infusions of the carrier—aCSF. The hypothesis was that EGF would induce the proliferation and migration of SVZ neural precursor cells and the EPO infusion would promote their differentiation into neurons. Six weeks after the stroke, control animals that received infusions of aCSF had a characteristic cavity, underlying the lesioned cortical area (Figures 1C and 1E). Remarkably, however, the cortex of rats that received EGF + EPO displayed no cavity formation (Figures 1D and 1F). Examination of the tissue underlying the cortical lesion in growth factor-infused animals, using cresyl violet staining, revealed a cellular structure that lacked normal cortical organization (Figure 1H), for example, lack of lamination, when compared with non-lesioned cortex in the same brain (Figure 1G). The region was identified by the lesion coordinates, which were + 3 to −4 anterior—posterior from the bregma and lateral 1.5 to 4.5 (see Materials and methods for details). There were two possible explanations for the effect of EGF + EPO infusion after stroke; (1) EGF + EPO infusion maintained the lesioned cortical tissue and no cavity was formed or (2) the tissue was generated de novo as a result of the growth factor infusion. We double labeled brain sections of rats that received a stroke followed by EGF + EPO, with the nuclear marker Hoechst and the neuronal marker NeuN, respectively (Figure 1I—L). In the cortical tissue that was immediately below the area of devascularization, total cell number was unchanged 3 days poststroke (n = 3, P = 0.157) although the cell nuclei are less condensed, likely reflecting the presence of activated cells such as microglia as well as the infiltration of blood borne cells. Markedly, NeuN staining revealed the complete absence of neurons in the lesion site 3 days after stroke (Figure 1J). Cresyl violet staining confirmed the lack of neuronal profiles immediately underlying the area of devascularization (Supplementary Figure 1). At days 11 (n = 11, P = 0.009), 15 (n = 3, P = 0.002), 18 (n = 13, P = 0.00004), and 42 (n = 4, P = 0.007) poststroke (Figures 1I and 1K), the total numbers of cells in the lesioned cortex was significantly elevated relative to the unlesioned cortex. Interestingly, neuron numbers increased over time (Figure 1L) and by day 42 the neuronal density in the lesioned cortex was not different (P = 0.062) than prestroke values. The fact that neurons are completely absent by day 3 poststroke and then reappear in the tissue over time with EGF + EPO infusions, suggested that the EGF + EPO infusions were not acting to maintain neuronal survival but instead, were leading to the generation of new neurons. This evidence for newly generated cortical tissue led us to ask whether this tissue might contribute to recovery of motor deficits after stroke.
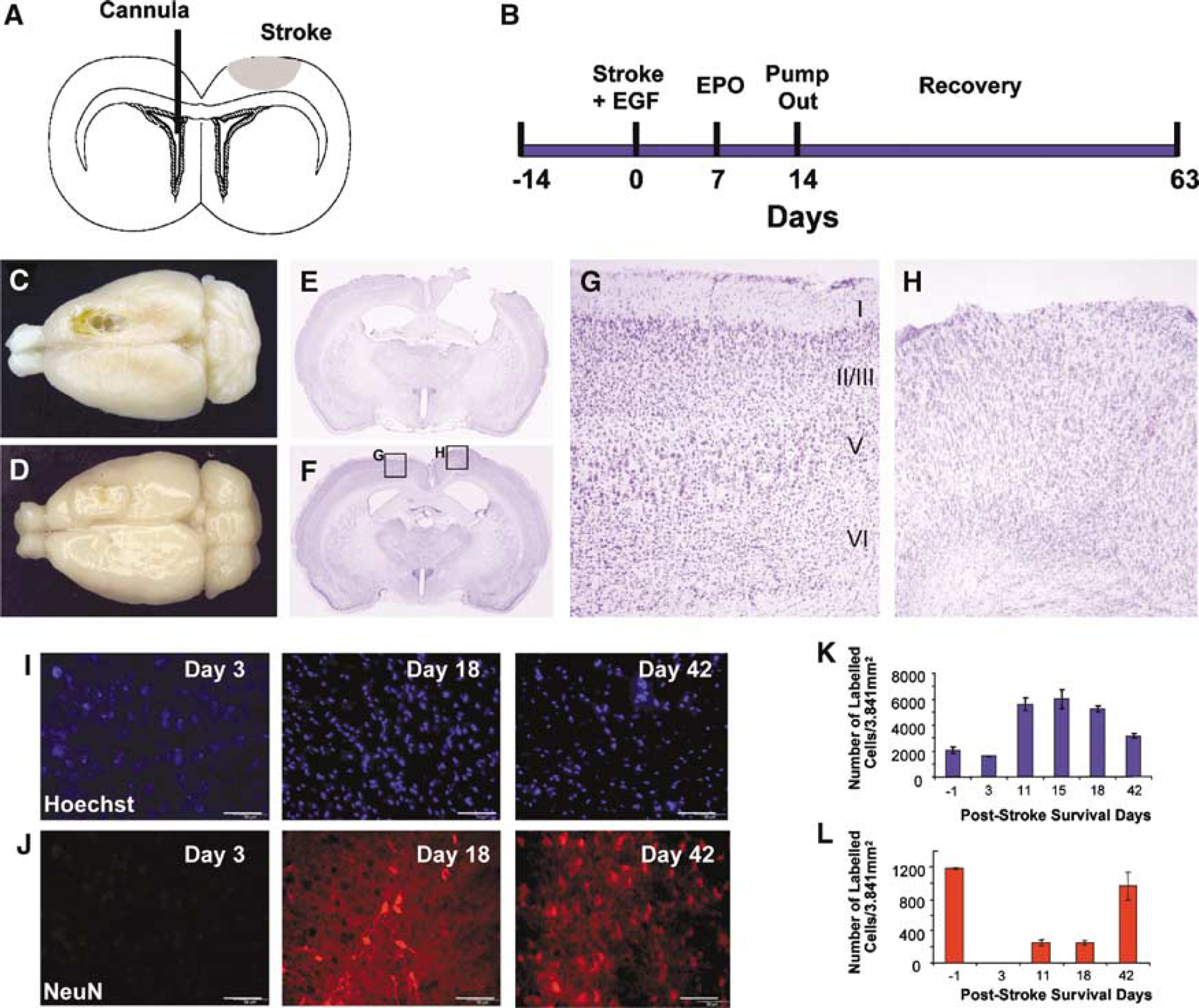
EGF + EPO infusions lead to tissue regeneration in the motor cortex after focal stroke. (
Stroke-Induced Motor Deficits Recover after EGF + EPO Infusions
After focal devascularization of the motor cortex, all rats showed a strong asymmetry in paw use in vertical exploration (from ~50:50 in unlesioned rats (control) to ~80:20 in lesioned rats), an absence of forelimb inhibition in swimming (from ~1 stroke to ~3 strokes), and a drop in reaching success to approximately 25% of preoperative levels (from ~80% success to ~20% success) (Figure 2; Supplementary Movies 1–3). Six weeks after stroke, rats treated with the carrier aCSF, EGF alone, or EPO alone, showed little improvement in either of forelimb asymmetry (Figure 2A) or in forelimb inhibition during swimming (Figure 2B). In contrast, the EGF + EPO group showed gradual improvement over the first 4 weeks and did not differ from controls by week 6. All groups showed spontaneous recovery to ~75% of preoperative levels in skilled reaching by week 3 (Figure 2C), although only rats treated with EGF + EPO approached control levels at 6 weeks.
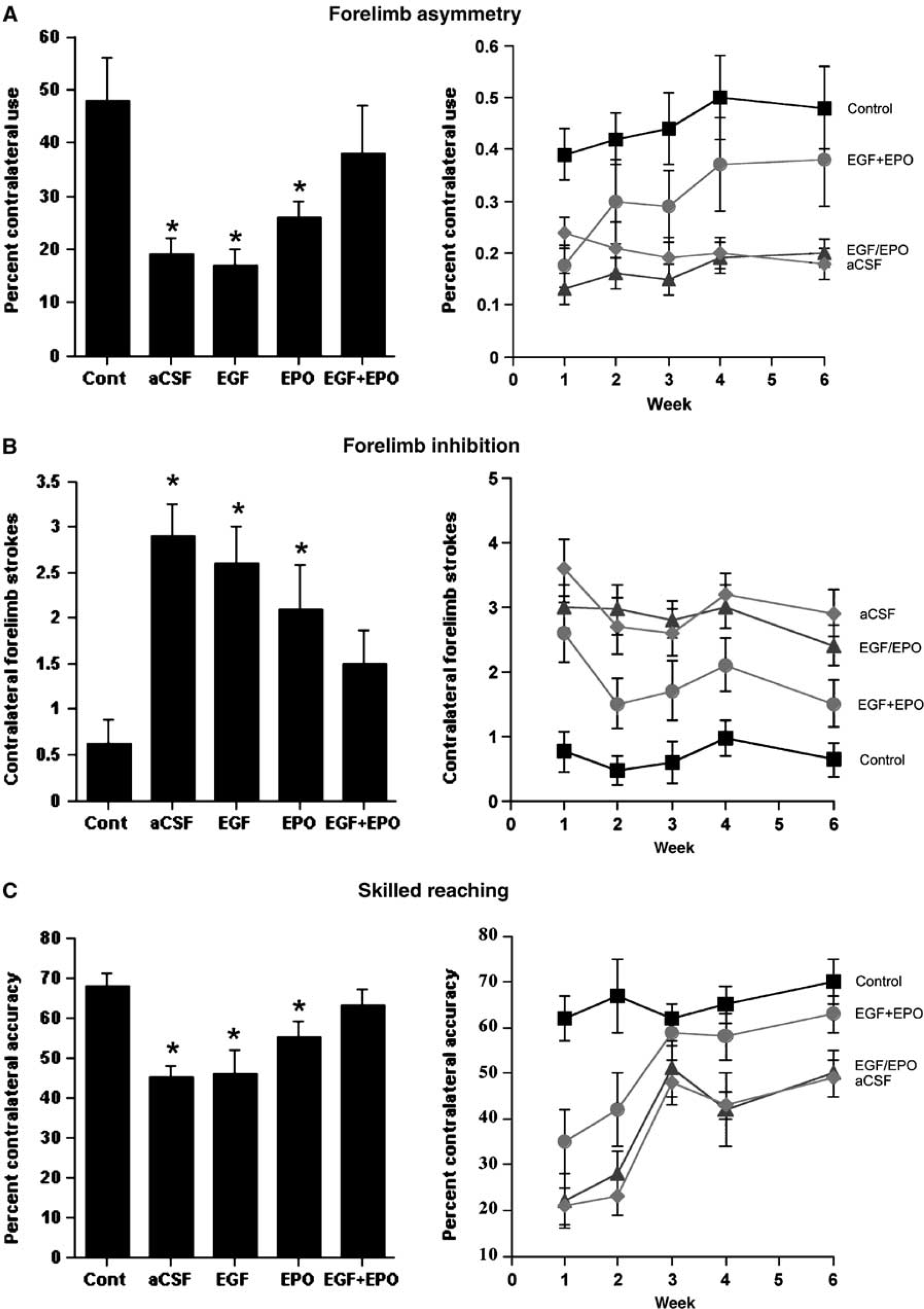
Stroke-induced motor deficits recover after EGF + EPO infusions. (Left panels) Summaries of the behavioral effects after 6 weeks of postinjury recovery. Rats treated with EGF + EPO showed better function on forelimb asymmetry (
Two-way analysis of variance (ANOVAs) (Group-Week) on each behavioral measure (cylinder, swimming, reaching, respectively) showed main effects of Group (F(4,47) = 6.92, P = 0.0002; F(4,47) = 10.59, P < 0.0001; F(4,22) = 9.35, P = 0.0001) and Week F(4,188) = 2.93, P = 0.02; F(4,188) = 6.93, P < 0.0001; F(4,88) = 3.08, P = 0.0004). In addition, there were significant Group × Week interactions for the cylinder and reaching tasks (F(16,188) = 2.24, P = 0.005; F(16,88) = 3.08, P = 0.0004). Post hoc tests on week 6 revealed that only the EGF + EPO group failed to differ from control (P‘s < 0.05).
Functional Recovery after Stroke with Delayed Growth Factor Infusions
To determine the effective window of treatment for promoting functional recovery, EGF was administered starting on days 0, 3, 7, or 14 poststroke and EPO infusions followed for 7 days after completion of EGF infusions (n = 5 for all groups) (Figure 3). As in the previous experiment, all lesion rats that received aCSF infusions showed clear impairments after 6 weeks of behavioral testing (Figure 3). Because comparing groups at different time points after surgery is meaningless given that the infusions started at different times, we elected only to do analyses once behavior had asymptoted at 6 weeks. ANOVAs on the cylinder and swimming on week 6 showed main effects of group (F(5,24) = 3.54, P = 0.015; F(5,24) = 3.75, P = 0.01) whereas the reaching scores were not significant (F(5,24) = 2.39, P = 0.068). Post hoc tests showed that rats that received EGF + EPO, beginning immediately (day 0) or day 3 after ischemic injury, did not differ from unlesioned (control) rats (asymmetry (P = 0.2617, day 0; P = 0.6597, day 3); inhibition (P = 0.372, day 0; P = 0.143, day 3); reaching (P = 0.3891, day 0; P = 0.5929, day 3)). The delayed infusion of EGF + EPO 7 days postischemic injury resulted in improvement in two of the three tests (not different than control, asymmetry, P = 0.0923; inhibition, P = 0.842), whereas the 14 days delayed infusion group was only marginally improved in the forelimb asymmetry test (not different than control, P = 0.0717), revealing a window of efficacy for EGF + EPO. The lack of improvement in the 14 days delayed infusions group in forelimb inhibition and skilled reaching was not simply due to more time needed for recovery, as the 4 week and 6 week performances were virtually identical (data not shown). As in the previous experiment, the functional improvements were gradual although there was a clear trend at the end of EPO infusion.
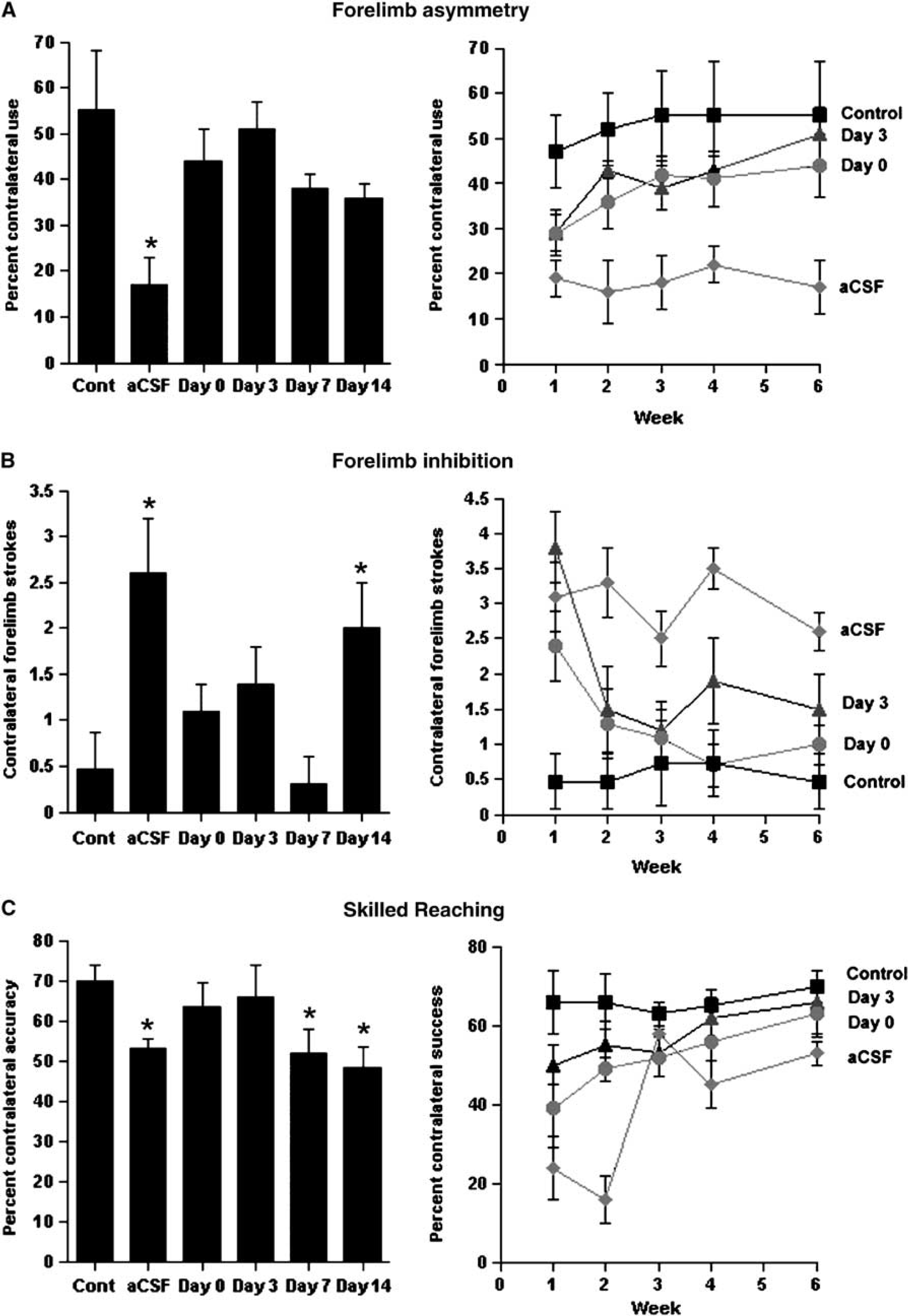
Infusions of EGF + EPO immediately or up to 7 days after stroke results in a gradual recovery of motor function in three behavioral tests. (Left panels) Summaries of the behavioral effects after 6 weeks of postinjury recovery in forelimb asymmetry (
In sum, these results confirm that EGF + EPO-induced functional recovery does not depend on the survival of pre-existing cortical neurons and show that EGF + EPO infusions are effective in promoting functional recovery when administered up to 7 days poststroke.
EGF + EPO-Stimulated Cells Migrate from the Subventricular Zone to Stroke Damaged Area of the Cortex
The cells generated in the animals that had received a stroke followed by EGF + EPO infusions could have originated from the SVZ, which contains a population of stem and progenitor cells (precursor cells), or alternatively, from cells residing in the brain parenchyma surrounding the lesion site. To test the hypothesis that SVZ precursors were the source of the newly generated cortical tissue, we infused a replication deficient RV expressing β-gal into the ipsilateral LV, before stroke, to selectively label proliferating SVZ precursor cells (Figure 4). Brains were examined at various survival times for the number and location of RV-labeled cells (Figure 4A, n = 3 to 4 rats per survival time). In rats with stroke and aCSF infusions, the RV labeled cells remained confined to the SVZ (Figure 4C (i and ii)) at all times examined and the numbers of cells decreased within the SVZ over time (Figure 4B). This is consistent with previous work showing that under baseline conditions (i.e., in the absence of a lesion), precursor cells are continually lost from the SVZ due to migration to the olfactory bulb and/or cell death (Craig et al, 1999; Morshead et al, 1998). However, in animals that received 7 days of EGF and were killed on day 11 poststroke (before the infusion of EPO, n = 4 animals), 55.3% ± 22.6% of the RV labeled cells were found to be migrating towards the lesion site with 10.0% ± 7.1% seen within the devascularized area and 37.1% ± 14.1% remained confined to the SVZ (Figures 4B and 4C(iii)). Many of the migrating cells were found within the white matter of the corpus callosum. This ‘directed’ migration of RV labeled cells towards the lesion site is distinct from that observed after growth factor infusion into non-lesioned animals (Craig et al, 1996) where SVZ cells are observed migrating in a radial fashion in all directions away from the ventricle. Interestingly, the initial decline (between days 4 and 11 poststroke) was not seen in growth factor treated animals where the number of RV labeled cells observed at day 11 poststroke (102.4 cells) was not significantly different from the starting population observed on day 4 poststroke (108.5 cells, P > 0.05) (Figure 4B). This could be the result of increased cell survival and/or proliferation of the surviving cells as a result of the growth factor treatment. Nonetheless, the complete lack of migration in aCSF infused animals indicates that the ischemic insult alone is not sufficient to induce SVZ precursor cell migration. However, in the presence of EGF, the lesion site is clearly able to attract the SVZ cells as seen with the ‘targeted’ migration.
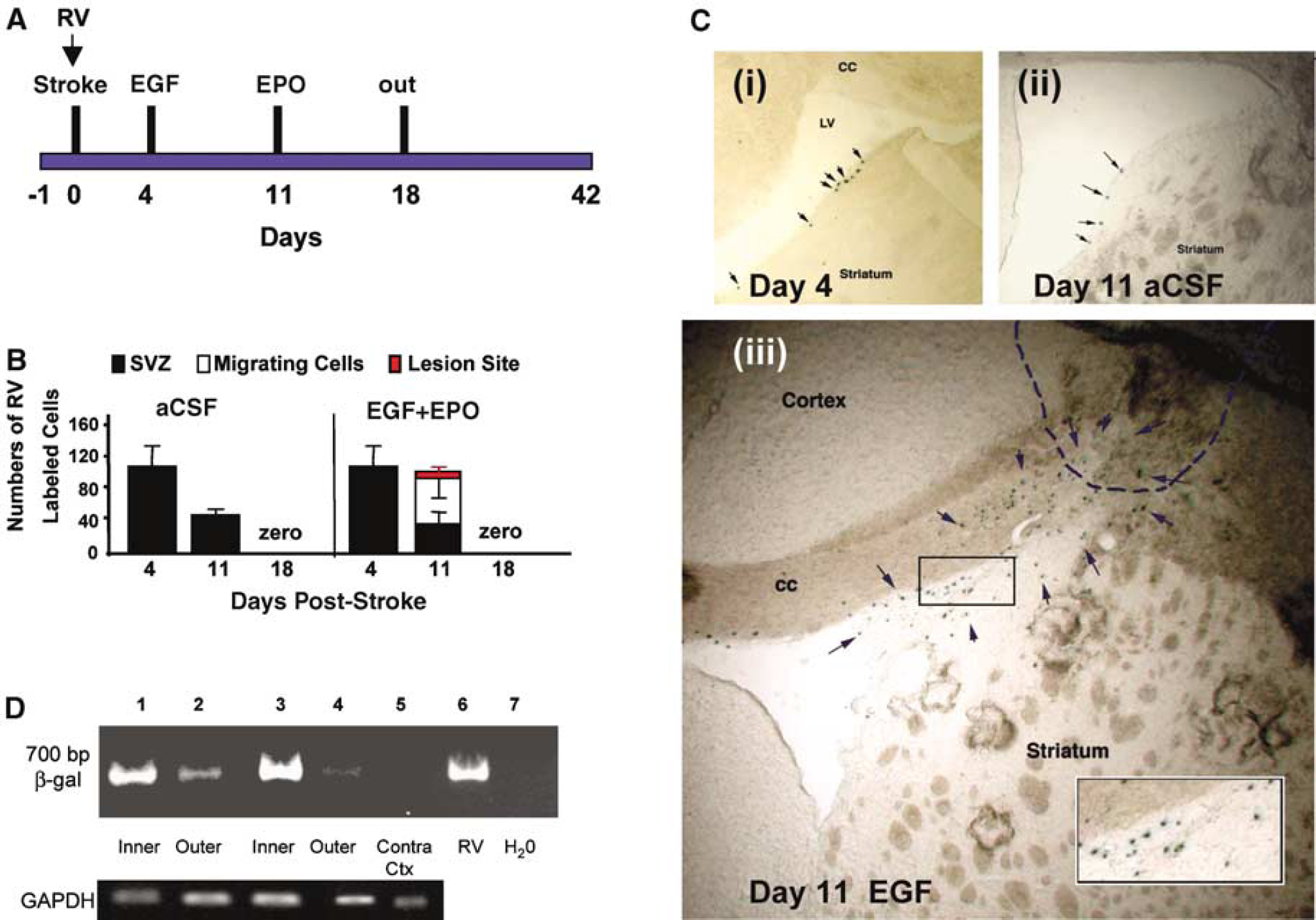
EGF + EPO-stimulated cells migrate from the SVZ to ischemic damaged area of the cortex. (
In both growth factor treated (n = 5) and saline infused (n = 4) animals killed on day 18 poststroke, no RV-labeled cells were detected within the SVZ, the lesion site, or en route. The inability to detect RV labeled cells within the parenchyma at 18 days poststroke could be explained by: (1) SVZ cells that migrate to the site of the injury contribute to tissue formation indirectly through the release of factors that activate/support cells but they do not themselves survive and contribute to the tissue formation at later survival times or (2) the RV labeled cells are present within the newly generated tissue however they have downregulated the expression of β-gal, and hence we are unable to detect them with histochemistry. To test these hypotheses, we performed PCR on uninjured cortical tissue and the newly generated cortical tissue from day 40 brains that received growth factor infusions and found the presence of β-gal within the newly generated cortical tissue (Figure 4D). Thus, the loss of β-gal positive cells was due to a downregulation of β-gal expression by RV labeled cells over time. Similar observations have been made in the olfactory bulb where retroviral expression is downregulated (as determined using PCR) leading to an underestimation of the numbers of migrating neuroblasts that reach the olfactory bulb at longer survival times (Craig et al, 1999). The findings suggest that cells within the newly generated cortical tissue are derived, in part, from forebrain SVZ-derived cells.
To further confirm the presence of SVZ-derived cells in the newly generated cortical tissue and to enable the phenotypic characterization of these cells, rats were given a series of injections of BrdU one day before the stroke to label proliferating SVZ precursors (Figure 5A). Rats then received EGF starting on day 3/4 and EPO on day 11. Brains were harvested on day −1 (i.e., before stroke; n = 3), or on day 11 (n = 4), 18 (n = 3), or 42 (n = 4) poststroke. The number of BrdU labeled cells was counted in the SVZ and lesion site (Figure 5B). On day −1, BrdU labeled cells were confined to the SVZ. There was a continual loss of BrdU labeling in the SVZ over time and a concomitant increase in BrdU-labeled cells observed in the regenerating cortical tissue at the lesion site (Figures 5C and 5D). Taken together, the RV and BrdU cell tracking data definitively show that SVZ precursor cells are contributing to the newly generated cortical tissue after focal, forebrain stroke.
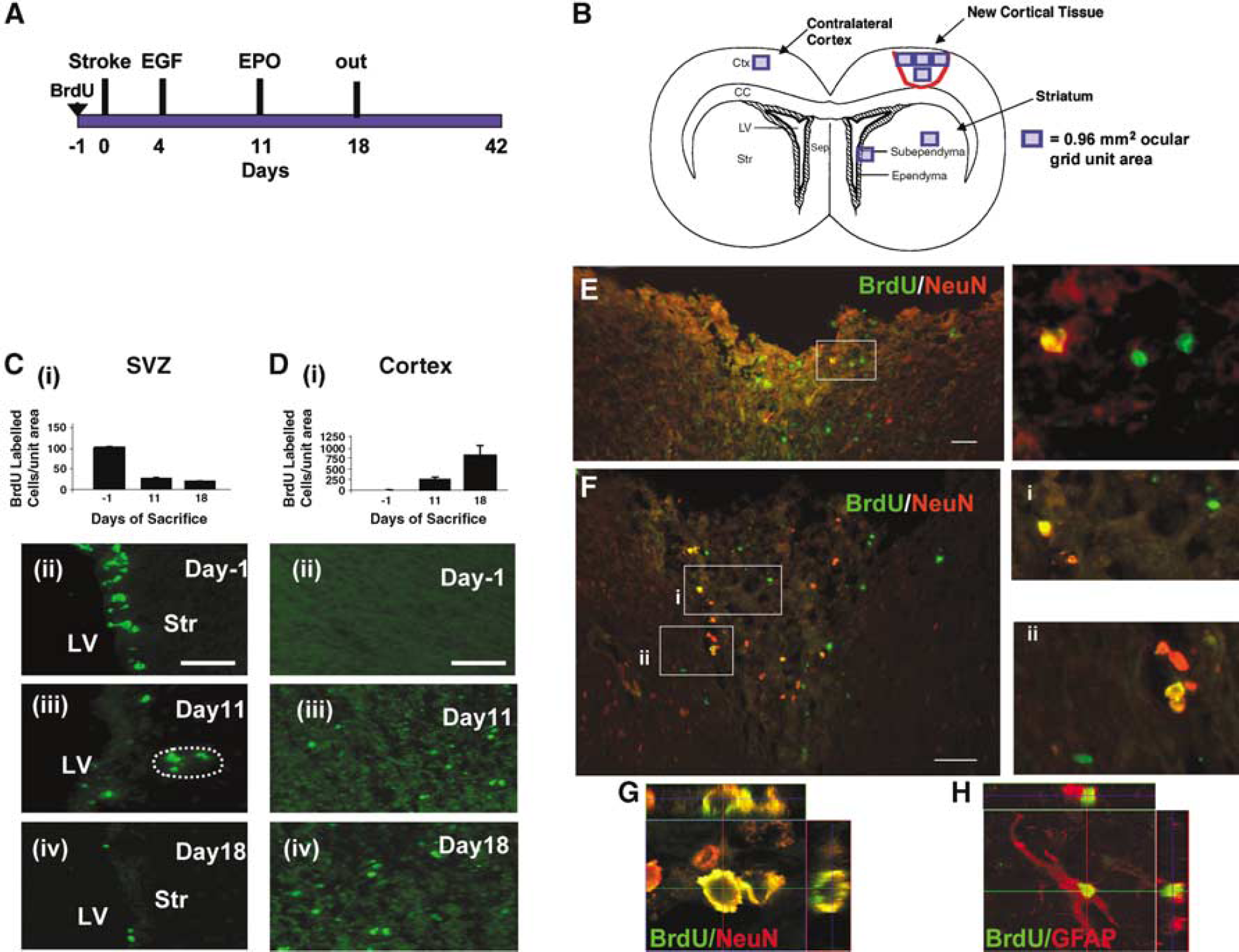
Newly generated cells from the SVZ, induced by EGF + EPO infusions, differentiate into neurons and glia in the regenerating cortex. (
The phenotype of the SVZ derived cells within the regenerated cortical tissue was determined by double labeling with BrdU and NeuN or glial fibrillary acidic protein (an astrocytic marker) in tissue from rats killed at various times poststroke. On days 11 (n = 4), 18 (n = 3), and 42 (n = 4), many cells within the newly generated tissue were clearly labeled with BrdU, NeuN, and glial fibrillary acidic protein. On day 18, 11.3% ± 5.3% and 12.3% ± 2.4% of BrdU labeled cells were double labeled for NeuN and glial fibrillary acidic protein, respectively (Figures 5E and 5F), indicating that SVZ-derived cells differentiated into neurons and astrocytes within the newly generated cortical tissue. Confocal reconstruction confirmed the double labeling (Figures 5G and 5H). Taken together, these findings suggest that after stroke, in the presence of EGF + EPO, SVZ-derived precursor cells migrate to the site of injury and differentiate into both neurons and glia.
Removal of Newly Generated Cortical Tissue Reverses Functional Recovery after Stroke
Our results suggest that there is a correlation between the generation of new cortical tissue and functional recovery after focal forebrain stroke. However, our observed functional recovery after stroke could be due to the presence of the newly generated cortical tissue or to the combined actions of EGF + EPO on the rest of the brain, or both. To determine if the newly generated cortical tissue plays a significant role in functional recovery, we prepared rats with focal forebrain stroke and administered EGF + EPO beginning on day 3 poststroke (Figure 6A). Rats were tested weekly for forelimb asymmetry and single pellet reaching. Forty-two days after the stroke rats were anesthetized and the newly (EGF + EPO) generated cortical tissue was carefully removed by dissection and suction (Figures 6B and 6C). When tested 1 week later (day 49), the performance of rats whose newly generated cortical tissue had been removed was not significantly different. However, 2 weeks later (day 63), the rats with tissue removal showed a virtually complete reversal of functional recovery in both forelimb asymmetry.
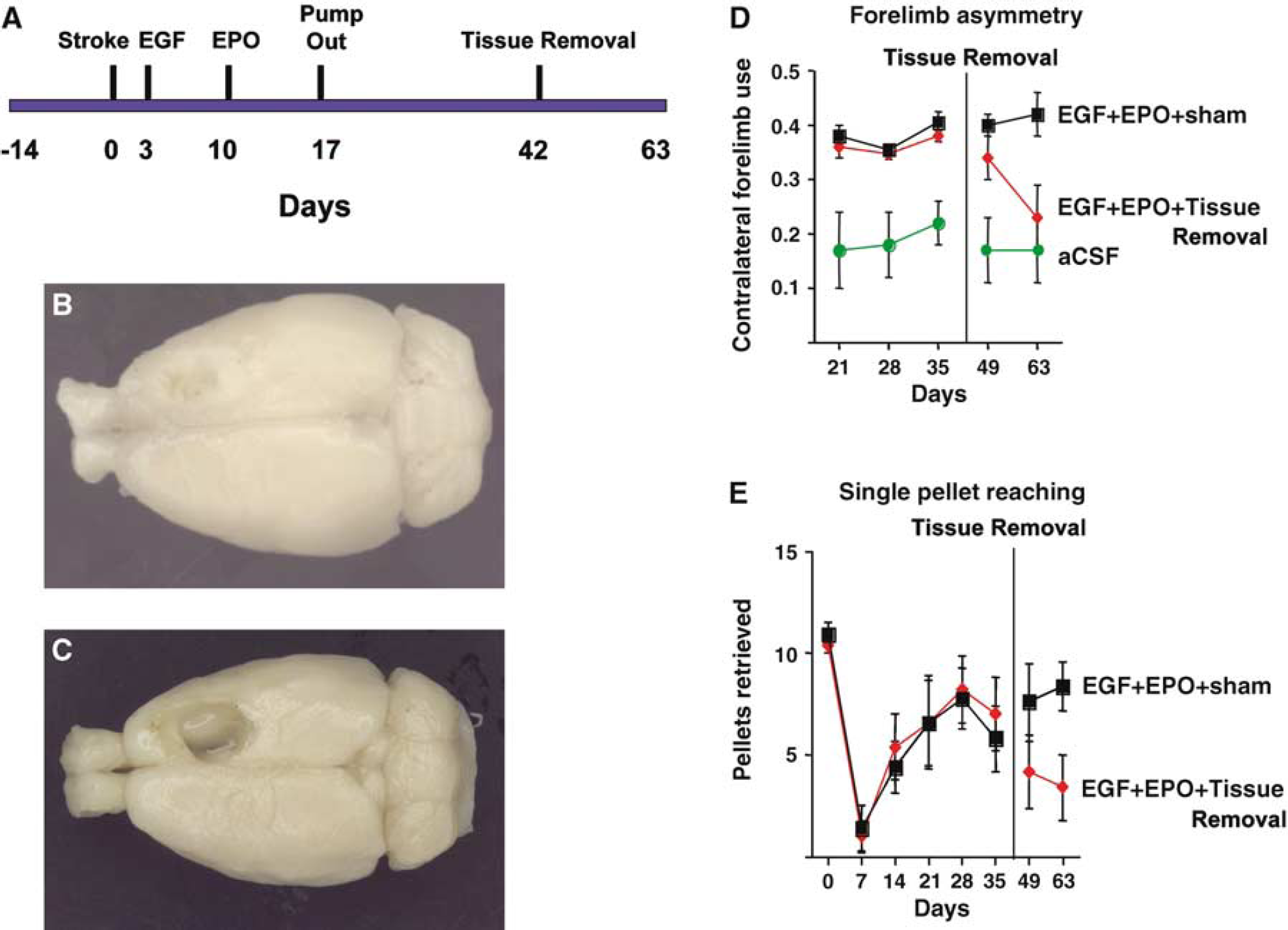
Removal of newly generated cortical tissue reverses functional recovery after stroke. (
ANOVA (Group × Week) on weeks 5, 7, and 9 found a main effect of Group (F(2,13) = 10.5, P = 0.002), but no effect of week (F(2,26) = 2.76, P = 0.08) or the interaction (F(4,26) = 1.97, P = 0.13). Post hoc tests showed that the EGF + EPO + LX group differed from the EGF + EPO group only on week 9 (P < 0.05). Post hoc tests showed that, whereas the two groups did not differ on weeks 5 or 7, they differed on both tests on week 9 (P's < 0.02).
Because it was possible that the removal of the newly generated tissue produced an injury that was larger in size than the original lesion, we estimated lesion volume by using NIH Image to measure lesion size from dorsal photographs of the post-mortem brains. The lesion size was virtually identical in the stroke + EGF + EPO + Tissue removal and stroke + aCSF groups (means = 15.3% ± 1.1% and 14.9% ± 1.1% of total hemispheric cortical tissue, respectively; F(1,12) = 0.05, P = 0.83).
In sum, these findings suggest that, after stroke and EGF + EPO infusions, the newly generated cortical tissue is necessary and sufficient for the observed recovery of motor function.
Discussion
In this study, we hypothesized that after a devascularizing insult that produces a chronic stroke lesion of the motor cortex, a combination of EGF + EPO would enhance SVZ proliferation, re-direct migration, and promote neuronal differentiation, and thus contribute towards tissue regeneration and functional recovery. Our results show that this specific growth factor combination can regenerate cortical tissue, which, in turn, is necessary and sufficient for recovery of motor function.
A combination of EGF + EPO, but neither one alone, was necessary for tissue regeneration and functional recovery after the motor cortex stroke lesion. Surprisingly, in the absence of these growth factors little if any enhanced proliferation, migration, or neural differentiation was observed. This is contrary to previous findings (Arvidsson et al, 2002; Jin et al, 2003; Parent et al, 2002; Zhang et al, 2004), which found that middle cerebral artery occlusion in rats stimulated all those processes. It is reasonable to propose that the more generalized (cortical and striatal) and larger lesion that develops after middle cerebral artery occlusion, as opposed to the more focal cortical lesion that develops after the devascularizing lesion, underlies these differences. However, after EGF + EPO infusions the newly generated SVZ cells did migrate exclusively to the lesion area of the cortex. This directed migration is very different from the non-directional, radial migration of SVZ precursors observed after EGF infusion in unlesioned animals (Craig et al, 1996). It is tempting to speculate that chemokine secretion is the common element underlying directed migration of endogenous cells to ischemic lesions, as has recently been suggested to be critical in attracting transplanted human NSCs to migrate to the infarcted area of the forebrain (Imitola et al, 2004). While previous studies suggest that EGF might contribute to regeneration by enhancing NSC proliferation (Craig et al, 1996; Reynolds and Weiss, 1992) and precursor cell migration (Gregg and Weiss, 2003), a precise role or mechanism for EPO, which is postulated to be a therapeutically relevant neuroprotectant after stroke in rats and humans (Brines et al, 2000; Ehrenreich et al, 2002; Siren et al, 2001), is less clear. EGF + EPO are both needed for functional recovery and EPO is delivered late enough to suggest that it is not involved in neuroprotection of the ischemic tissue. More likely, EPO is acting to promote neuronal differentiation (Shingo et al, 2001) and viability of the newly generated cortical tissue, through direct antiapoptotic effects (Digicaylioglu and Lipton, 2001) and/or enhanced angiogenesis (Wang et al, 2004).
Our study has made great strides in establishing the origin of stroke-induced cell genesis. The power of our cell tracking assays is that we prelabeled proliferating cells with RV or BrdU before stroke, when the SVZ cells are the only significant population of cells that will be labeled in unlesioned brains. This is especially the case for RV, which is administered directly into the LV. Indeed, labeling cells before stroke is the only way to definitively show that SVZ precursors are migrating and contributing to the regenerated tissue. Previous studies suggesting that SVZ cells contribute new neurons to the lesion sites involved injecting BrdU at various times poststroke and therefore all proliferating cells, including those in the parenchyma, were labeled making it impossible to determine their SVZ origin (Arvidsson et al, 2002; Parent et al, 2002; Jin et al, 2003). One must consider, however, that our prelabeling strategies very likely underestimate the contribution of SVZ derived precursor cells to the regenerated tissue for two reasons. First, we were only examining the contribution of a small cohort of cells and therefore any cells that were activated to migrate and contribute to the new tissue formation after the lesion were not examined. This was exacerbated in the case of the RV labeling. RV infection of cells was used as a means to introduce a non-diluting marker of cells, however, since we were unable to detect the RV with histochemistry because of downregulation of the marker gene, we were unable to determine the absolute numbers of cells that reach the regenerating tissue and contribute to its formation. The second reason for underestimating the SVZ contribution in our model is because of the fact that BrdU is diluted from cells as they continue to proliferate en route to, and within, the regenerating tissue. For both of these reasons, we are underestimating the contribution of SVZ derived cells. Highlighting this point is the recent evidence suggesting that cells are continually added to the lesion site for up to 4 months postinjury (Thored et al, 2005).
An important aspect of our study is the unambiguous demonstration that growth factor-generated brain tissue is necessary and sufficient for functional recovery after stroke. Nakatomi et al (2002) reported that EGF + FGF-2 augmented the regeneration of the CA1 pyramidal layer after global ischemia and this contributed to the recovery of hippocampal-dependent spatial learning. Because those authors could not reverse the recovery by eliminating the newly generated neurons, they could not be certain that enhanced recovery of function was not due to other actions of the growth factors. In fact, a recent study found that spontaneous regeneration of the hippocampus CA1 layer occurs after global ischemia, in the absence of growth factor infusions (Bendel et al, 2005). No spontaneous cortical regeneration was observed in this study, in the absence of growth factors. However, the question of which newly generated cells (i.e., neurons or glia) within the regenerated cortical tissue are participating in recovery, and the mechanism of recovery, is not clear. The new cortical tissue showed no evidence of laminar organization reminiscent of normal cortical tissue. Furthermore, some of the behavioral recovery was evident before the majority of mature, newly generated neurons were present in the lesion site. Finally, the behavioral gains did not completely reverse after removal of the newly generated tissue, but rather progressively worsened. Thus, it seems reasonable to postulate that the newly generated cortical tissue may participate in functional recovery by promoting the re-organization and modification of pre-existing cortical circuitry.
The analysis of behavioral change after motor cortex injury is a relatively new science. Although it has long been known that cortical injury in rats can produce motor impairments (e.g., Bures and Bracha, 1990), it has only recently been realized how skillful rats are with digit and paw movements and sophisticated analyses of these movements are still being developed (e.g., Whishaw, 2005). The current paper represents the first attempt to examine the effect of endogenous mobilization of NSCs on the recovery of complex motor behaviors on a battery of motor tests that are now known to be selectively sensitive to motor cortex injury. Although there is clear functional improvement in the EGF + EPO-treated animals in the current study, the enhanced recovery of skilled reaching overspontaneous improvement is smaller than the recovery on the other tasks. It seems likely that the control of paw and digit movements requires the contribution of the direct corticospinal connections from the pyramidal cells of the motor cortex and these are not likely to be replaced either by the mobilization of endogenous NSCs or the transplantation of exogenous cells. Rather, the functional improvements must be related to compensatory modifications in movement controlled by remaining motor regions, likely either in the anterior cingulate cortex, more lateral motor cortex, or striatum. The role of these areas in the compensatory process is unclear, although it is known, for example, that nicotine stimulates improvements in motor performance on the tests used in the current study and does so by facilitating postural adjustments that appear to act to enhance function (Gonzalez et al, 2006). The behavioral analyses in the current study do not permit a firm conclusion regarding the basis of the motor improvements, but they are likely related to changes in the neuronal circuitry in the perilesional cortex. Analyses of Golgi-stained tissue (data not shown) failed to show any changes similar to those seen in the nicotine studies, which suggest that the growth factors have produced a different type of change in circuitry.
Although we are unaware of previous studies showing that growth-factor stimulated neurogenesis can provide functional benefit after cortical lesions in adults, a series of recent studies has shown that postinjury infusion of FGF-2 into rat pups with motor cortex lesions on postnatal day 10 also leads to the generation of a new cortical tissue without normal lamination and functional recovery similar to that observed here (e.g., Monfils et al, 2005). Removal of the newly generated tissue in these animals also reversed functional recovery but in contrast to the current study, the reversal was immediate. FGF-2 was ineffective in stimulating either the formation of new tissue or functional recovery in rats with adult lesions, however. The Monfils et al results thus confirm that the stimulation of endogenous stem cells can be beneficial after cortical injury but also show that the SVZ may respond differentially to growth factors at different points in development. Furthermore, the fact that the functional deficits were present the day after removal of the newly generated tissue suggests that the mechanisms underlying the functional recovery may be different at different ages. This may be important in searching for treatments for ischemic injuries during development.
The ability to mobilize NSCs and significantly regenerate damaged or diseased brain tissue may have therapeutic implications for many neurologic disorders. Stroke, in particular, is the leading cause of chronic disability worldwide and new approaches to stimulating functional recovery are clearly needed (Ward and Cohen, 2004). Encouragingly, EGF + EPO treatment remains effective when delivered up to 7 days postlesion, as opposed to the current clinical poststroke therapy, TPA, which has a time window of efficacy of just a few hours (Schellinger and Warach, 2004). However, while stem cell therapies are being highly touted as having great promise for cell replacement (Lindvall et al, 2004), some element of caution in developing and transferring these putative therapies to the clinic is imperative. A tremendous human and fiscal investment was made in the preclinical and clinical testing of putative neuroprotectants for stroke, all of which subsequently failed to show clinical efficacy (Hoyte et al, 2004; Wahlgren and Ahmed, 2004). Similar promise followed by uncertain outcomes has plagued the development of cell replacement therapy for Parkinson's disease (Winkler et al, 2005). A common element might be the need to more carefully consider what are the most suitable assays for recovery of function—neuronal cell number and phenotype versus behavioral outcomes? We would argue that sophisticated batteries of behavioral assays that are clearly and unambiguously linked to the brain lesion being studied, in a number of animal models, are paramount. In our study, using such a battery of tests that are linked to motor cortex lesions, we observed recovery of function in the absence of anatomically faithful recapitulation of the cortex. This underscores the need to focus on behavioral outcomes, first and foremost when testing novel therapeutic approaches, and to use these as the foundation for understanding the cellular and physiologic mechanisms of functional recovery.
Footnotes
Acknowledgements
Thanks to Gloria Mak, Greg Silasi, and Anna Erlandsson for critical reading of an earlier version of this manuscript, to Trudi Stickland for reading the manuscript and assistance in preparation of the supplementary movies, and to Amy Hoyles and Angela Kam for their technical support and help with statistics. Samuel Weiss is an AHFMR Scientist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
