Abstract
Intracerebral cell therapy (CT) is emerging as a new therapeutic paradigm for stroke. However, the impact of physical therapy (PT) on implanted cells and their ability to promote recovery remains poorly understood. To address this translational issue, a clinical-grade neural stem cell (NSC) line was implanted into peri-infarct tissue using MRI-defined injection sites, two weeks after stroke. PT in the form of aerobic exercise (AE) was administered 5 × per week post-implantation using a paradigm commonly applied in patients with stroke. A combined AE and CT exerted sub-additive therapeutic effects on sensory neglect, whereas AE suppressed CT effects on motor integration and grip strength. Behavioral testing emerged as a potentially major component for task integration. It is expected that this study will guide and inform the incorporation of PT in the design of clinical trials evaluating intraparenchymal NSCs implantation for stroke.
Introduction
Cell therapy (CT) for stroke is an emerging therapeutic paradigm that is undergoing clinical translation.1,2 Intraparenchymal peri-infarct implantation of human neural stem cells (NSCs), such as the CTX0E03 cell line, has been shown to be efficacious in animal models3–6 and found to be safe in patients with stroke.7,8 CTX0E03 cells have been widely characterized in vitro and in vivo to elucidate its biological mechanisms, which include secretion of growth factors to enhance angiogenesis9–12 and neurogenesis,3,5 as well as differentiation into neurons, astrocytes and oligodendrocytes. 3 Integration of NSCs into neural circuitry regulates motor behavior and improves impairments.13,14 This NSC-induced neuroplasticity is evidenced by the formation of novel functional synaptic connections that support long-term potentiation and excitatory postsynaptic potentials. 15 Neuroplasticity is also a key mechanism exploited by physical therapy (PT) to promote behavioral improvements. 16
Multi-modal PT, including resistance training, aerobic exercise (AE) and task integration, is considered the standard of care for stroke patients. 17 In preclinical models of stroke, enriched environments aim to replicate this multi-modality and have demonstrated therapeutic benefits if combined with intravascular or intracerebral cell therapy in some studies,18–20 but not in others.21,22 As enriched environments typically vary between laboratories, it remains unclear what specific aspects of the animals’ experience impart therapeutic benefits. 23 Its translational validity hence remains uncertain. 23 In contrast, implementation of AE can be achieved using treadmill running in animal models,24,25 as well as patients with stroke.26,27 A majority (88%) of PT professionals indicated that AE (20–30 minutes, 3–5 days a week for 2–8 weeks) should be incorporated in the rehabilitation of stroke patients. 28 In conjunction with intravascular cell therapy in stroke, AE has shown added benefits,29,30 but did not improve efficacy of NSCs after intracortical implantation in a rat model of traumatic brain injury. 31 A major consideration for the clinical translation of intraparenchymal NSCs implantation for stroke therefore is whether and how these two potentially complimentary therapeutic paradigms interact.1,2,32
To determine how behavioral recovery after intraparenchymal NSCs implantation is impacted by PT, we contrasted the effects of AE (i.e. treadmill running) versus CTXOE03 cell implantation, as well as the combination of both approaches in a rat model of stroke. To determine the level of administration of AE, maximum capacity testing (MCT) was conducted pre-implantation, as well as weekly thereafter to ensure the appropriate delivery of AE. An 80% sub-maximal activity level was delivered in a single 30-minute session 5 days/week for 10 weeks. 24 Therapeutic efficacy of AE, NSCs, and AE + NSCs was established using a battery of behavioral tests, and the biological substrates of these effects were analyzed using in vivo magnetic resonance imaging (MRI) and post-mortem histology. It is expected that this study will guide and inform the incorporation of PT in the design of clinical trials evaluating intraparenchymal NSCs implantation for stroke.
Methods
Experimental design
Animal procedures complied with the US Animals Welfare Act (2010) and were approved by the University of Pittsburgh Institutional Animal Care and Use Committee (IACUC). Reporting of animal use follows the ARRIVE guidelines. 33 Sprague-Dawley rats (male, n = 100, 12 weeks of age, 260 ± 15 g, Taconic Labs, USA) were maintained on a 12-hour light/dark schedule (lights on 06:00), with food and water available ad libitum. Based on previous effect sizes,3,24 an a priori power analysis determined a total sample size of >50 animals (Figure S1). Animals were randomly assigned (using a random number sequence) into groups (Figure S2A): healthy control (n = 9), middle cerebral artery occlusion (MCAo, n = 13), MCAo + AE (n = 8), MCAo + cells (n = 9) and MCAo + cells + AE (combined, n = 9). Two sequential cohorts (N1 = 23; N2 = 25) established the consistency and reproducibility of experimental effects. 34 NSCs were implanted 2 weeks post-stroke with AE initiated 1-week post-implantation (Figure S2B). AE and behavioral testing occurred between 09:00-16:00 by blinded experimenters.
Middle cerebral artery occlusion (MCAo)
Sham or right transient MCAo (70 minutes) surgery was achieved by advancing a 5-0 silicon rubber-coated monofilament (diameter 0.12 mm, length 30 mm, tip coating at 0.37 mm for 5–6 mm, 503756PK10, Doccol, USA) to the ostium of the MCA under isoflurane (4% induction, 1.5% maintenance in 30% oxygen) anaesthesia. 35 After MCAo, animals were assessed for forelimb flexion and contralateral circling. Animals were sutured and given topical analgesic cream (2.5% Lidocaine and 2.5% Prilocaine, Sandoz). Buprenex (0.05 mg/kg i.p.; Henry Schein) was administered twice daily for 3 days. Daily post-operative care and neurological assessment occurred until they recovered pre-operative weight.35,36 This model causes ischemic damage to the striatum in 2/3 of cases, and 1/3 of cases have more extensive damage that also encompasses overlying cortical areas, 3 thereby modelling the tissue pathology observed in 2/3 of ischemic stroke in patients. 37
Magnetic resonance imaging (MRI)
To assess the presence, location and volume of tissue loss, rats were anesthetized with isoflurane (4% induction, 1% maintenance) and scanned using a T2-weighted (T2w) spin-echo MRI sequence (6000 ms TR, 8 ms TE, 8 Averages, FOV 40 × 40 mm, 128 × 128 matrix, 60 slices at 0.3 mm thickness, 25 minutes) on a horizontal bore 9.4 T Varian scanner at 10 days post-infarction and 10 weeks post-implantation. Rats with hyperintense T2-weighted lesion volumes >10 mm3 were randomly assigned to experimental conditions.
Diffusion tensor imaging (DTI, Spin Echo; 2500 ms TR; 19 ms TE; 4 Averages; 6 diffusion directions; 11 ms Δ; 2.5 ms δ; 700 s/mm2 b value, 60 slices, slice thickness = 0.3 mm, FOV = 30 × 30; 96 × 96 matrix; 85 minutes) was acquired after completion of the T2w scan. Using DSI studio (www.dsistudio.labsolver.org), an Eigenvector analysis calculated the tensor used for image reconstruction. 38 Mean (MD), axial (AD), radial diffusivity (RD) and fractional anisotropy (FA) were computed for measurement of microstructural changes in tissue. Fiber tract reconstruction was achieved by random sub-voxel positioning of the seeds (10 seeds/voxel) with a minimal tract length of 2.2 mm and a trilinear interpolation (0.15 mm step size), terminated at 60° angular threshold, 0.02 FA threshold or a 10 mm maximum length. 39
The following day, functional MRI (fMRI) and baseline cerebral blood volume (CBV) measurements were acquired. A single i.v. bolus of monocrystalline iron oxide nanoparticles (MION, 15 mg Fe/kg, ferumoxytol, AMAG Pharmaceuticals) was injected for CBV-weighted (CBVw) contrast, which has a higher sensitivity and neuronal specificity than BOLD.40,41 Subdermal needle electrodes were placed in the forepaws at digits two and four. A gradient-echo echo-planar imaging sequence (1.5 s TR, 15 ms TE, 30 × 30 mm2 FOV, 96 × 96 matrix, 12 slices, 0.9 mm slice thickness, 2 segments) acquired images when a single forepaw was stimulated (1.5–2.0 mA, 8 Hz, 1 ms pulse width) using an isolator (Isoflex, AMPI, Israel) equipped with an electrical pulse generator (Master 8, AMPI, Israel). A block design experiment (9 s duration, 60 s inter-stimulus interval/block; 5 blocks/run, 4-6 runs/forepaw) was analyzed in SPM12 (Wellcome Trust Centre for Neuroimaging, London, UK). Activation maps were calculated with a general linear model accounting for the hemodynamic response function. Group t-maps were calculated using a one-sample T-test on relative signal change maps.
In the absence of stimulation, intravascular MION changes the transverse relaxation rate (ΔR2*) of tissue and is indicative of baseline CBV. A gradient-recalled echo sequence was used to acquire images (2.23 s TR, 3.18 ms minimum TE, 30 × 30 mm FOV, 256 × 192 acquisition matrix reconstructed to 256 × 256, 12 slices, 0.9 mm slice thickness) before and after 15 Fe mg/kg MION injection. The post-MION image was co-registered to the pre-MION condition in SPM12 to calculate ΔR2* and fractional CBV (fCBV, mL blood/mL brain) maps. 40
Maximum capacity testing (MCT) and AE
MCT in rats was performed 24 akin to the Bruce Protocol used in human subjects. 42 Prior to stroke, animals were acclimatized to the 4-lanes motor-driven treadmill (AccuScan) for 10 min/day at a rate of up to 10 m/min for two consecutive days. The following day, MCT was determined, starting with a set speed of 5 m/min, and gradually increasing speed by 5 m/min every 3 minutes. Maximum capacity was reached when animals could no longer run. A single 30-minute running session/day, 5 times/week, at 80% of maximum capacity, delivered AE for 10 weeks post-implantation.
Behavioral assessment
The bilateral asymmetry test evaluates tactile extinction, probing sensory neglect.3,35 Two strips of brown tape of equal size (6 cm long, 0.5–0.8 cm wide) were applied to the saphenous part of the forepaws. Six trials (300 sec each) recorded the removal time for each paw. Sensorimotor bias was determined by subtracting the unaffected (right) from the affected (left) paw. The impact of the therapeutic intervention was calculated by percentage (%) change between the pre- and final time point post-transplantation.
The footfault test measures the integration of motor responses.3,43 Rats were placed in the Motorater (TSE systems) with horizontal bars to record the placement of both unaffected and affected forelimbs over 2 trials (>50 placements per time point). The percentage of footfaults was calculated for group comparisons.
Amphetamine-induced rotation (contra-/ipsilateral) was used as an index of striatal damage. 3 Lesioned and control animals were harnessed into jackets tethered to an automated rotameter (TSE Systems) and injected with amphetamine (2.5 mg/kg dissolved in 0.9% saline, i.p., Sigma) 30 minutes prior to assessment. The number of contra- and ipsilateral rotations were then measured over 30 minutes.
To investigate forelimb motor dysfunction, grip strength was measured on a grip strength meter (TSE Systems) using 5 trials per paw to calculate mean force exerted by averaging the 3 best trials. 44 Grip strength of the affected forepaw and its ratio with the unaffected paw was calculated.
Cell implantation
Derivation, culturing and characterization of the cmyc-ERTAM conditional immortal 12-weeks’ old fetal cortical anlage-derived CTX0E03 human NSC line has been described in detail.6,9,10 For implantation, cryopreserved CTX0E03 cells (P32-36) were thawed and seeded on laminin-coated T175 flasks (5 × 106 cells/35 mL of media) for 4 days. As these NSCs are developmentally primed to develop into cortical neurons, no further in vitro specification occurred. Cell suspensions were formulated in HypoThermosol (BioLife Solutions) at 50,000 cells/µL. 45 Pre-implantation viability was >90% and >85% post-implantation (Figure S3). Under isoflurane anesthesia, animals received 2 deposits of a 4.5 µL cell suspension (in total 450,000 cells/rat, 1 µL/min) at the following coordinates: 1. AP −1.3 mm; L −3.5 mm; V −6.5; 2. AP −1.8 mm; L −4.0 mm; V −.0 mm). Animals were sutured and given topical analgesic cream (2.5% Lidocaine and 2.5% Prilocaine, Sandoz) and Buprenex (0.05 mg/kg i.p.; Henry Schein). Methylprednisolone (20 mg/kg day 1–7; 10 mg/kg day 8–12; 5 mg/kg day 13–14, s.c., Solu-Medrol, Pfizer) was given daily, whereas cyclosporine A (10 mg/kg in saline, s.c., Sandimmune, Novartis) was given on alternate days for 14 days. 3
Harvesting of blood and brain
To evaluate the biological effects of cell and physical therapy, blood and brains were collected. Animals were terminally anesthetized using an overdose of FatalPlus (VorTech) and perfused transcardially with ice-cold 0.9% saline followed by 4% paraformaldehyde (in 0.2 M PBS). 5 mL of whole blood was collected from the femoral artery during the saline flush prior to perfusion with paraformaldehyde. Whole blood was allowed to coagulate overnight at 4 °C. The excised brain was post-fixed overnight before being transferred to 30% sucrose (with sodium azide) for storage at 4 °C.
Blood serum isolation and enzyme-linked immunosorbent assay (ELISA)
After centrifugation of coagulated blood at 1000 g for 20 minutes, supernatant blood serum was collected in a clean polypropylene tube (ThermoFisher) and stored at −80°C. Enzyme-linked immunosorbent assays (ELISA) quantified growth factors (Table S1). Standards and diluted serum samples were incubated for 2 hours. 24 Assay diluent served as a negative control. An automated microplate reader (PowerWave 340, Bio-Tek) set to 450 nm (correction wavelength set to 540 nm) measured signal intensity within 15 minutes of adding the stop solution.
Histological assessments
Brains sections (50 µm thickness) were washed 3 × 5 minutes with PBS prior to application of the primary antibodies (Table S2) for overnight incubation at 4 °C. After 17 hours of incubation, primary antibodies were washed off and appropriate secondary fluorescent antibodies (Alexa dyes, Molecular Probes) were applied for 1 h at 21 °C. After counterstaining with the nuclear marker DAPI, slides were coverslipped with Vectashield for fluorescence. All imaging was performed on a M2 microscope (Zeiss) interfaced with a motorized stage and StereoInvestigator (MBF Bioscience).
For stereology, the area occupied by grafted SC101+ cells was manually delineated at 2.5 × before a sampling grid of 100 μm × 80 μm was applied with a counting frame of 50 μm × 30 μm. The optical fractionator method 46 was applied to estimate total cell number (coefficient of error ≤0.5) on every 5th section. The phenotypic fate of implanted cells was established by immunohistochemistry for neurons (NeuN, see Table S2 for detailed antibody information), and oligodendrocytes (SOX10) co-labeling with the human cell markers STEM101 (nuclear marker) or STEM121 (cytoplasmic marker). Human astrocytes were counted based on the human-specific astrocyte antibody STEM123. To determine synaptic (synaptophysin) and axonal (neurofilament) density in the graft, tiled whole graft images were outlined and thresholded in Fiji (imagej.net). Percent coverage was calculated across 5 different slices for the entire hemisphere. Additionally, measurements in tiled images covering only the graft area were taken to determine if cells exerted a more localized effect, as compared to AE, which could exert more widespread effects. Blood vessel (RECA) coverage and microglia (Iba1) activation were calculated for the whole ipsilateral hemisphere, as well as the area containing grafted cells.
Statistics
An a priori power analysis was performed in G*Power (www.gpower.hhu.de) to determine the appropriate sample size for β-1 = 0.8, p < 0.05 and f(V)=0.7, based on previous behavioral effects. 3 Graphing and statistical analyses were performed in Prism version 8 (GraphPad) with the arithmetic average and errors bars, representing the standard error of the mean (SEM), depicting descriptive statistics of the data. To perform parametric statistical inference testing, the normal distribution of data was evaluated using the Shapiro-Wilk test. Grubb’s test was used to define and exclude any statistical outliers (>2 standard deviations from mean) prior to statistical analyses. Two-way analyses of variance (ANOVA) were used to evaluate the impact of two independent variables (time and experimental group), whereas 1-way ANOVAs compared experimental groups followed by Tukey post-hoc testing.
Results
Maximum capacity testing verifies the therapeutic delivery of AE
To ensure an appropriate delivery of AE, treadmill running was evaluated using the MCT and training was adjusted based on their performance. At baseline prior to MCAo surgery, treadmill running was equivalent for all groups (Figure 1(a)). Stroke decreased this performance and without AE no improvements in running were observed. Groups receiving AE demonstrated a gradual improvement in treadmill running, indicating an appropriate delivery of PT. This AE effect was consistent between the two experimental cohorts (Figure S4). Stroke significantly reduced body weight post-surgery, but the rate of weight increase post-MCAo was equivalent for all groups (Figure 1(b)). Corticosterone levels in blood, a surrogate marker of stress, did not reveal a significant difference between groups at 10 weeks (Figure S5). These results indicate that AE was delivered without impacting weight gain or stress levels.
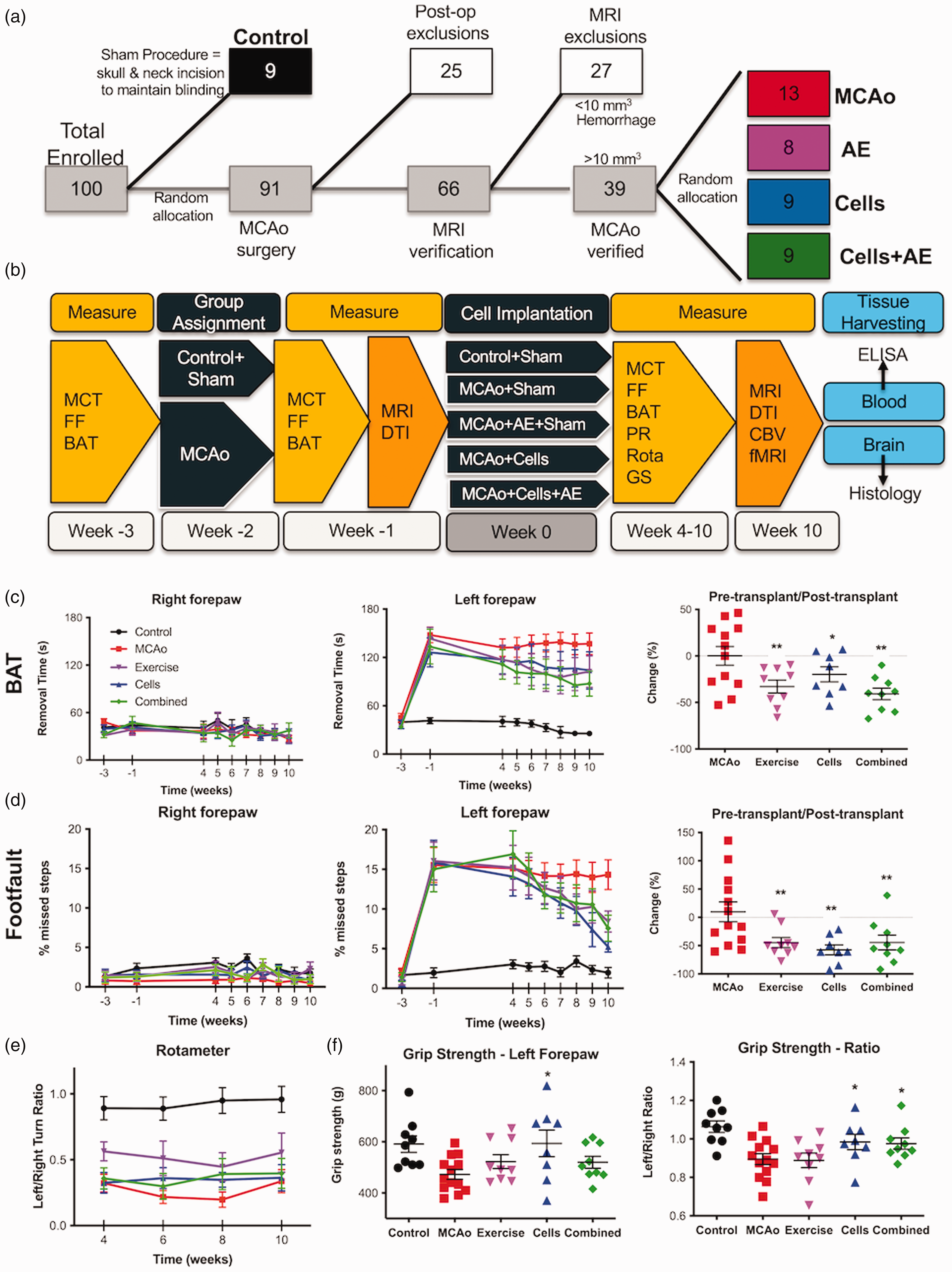
Experimental outline and behavioral analyses. (a) Sprague-Dawley rats (n = 100) were randomly allocated to experimental groups after inclusion and exclusion criteria were applied. (b) Behavioral measures, consisting of maximal capacity testing (MCT), footfault (FF) and bilateral asymmetry test (BAT) were acquired prior to middle cerebral artery occlusion (MCAo), as well as post-stroke. Two weeks post-MCAo, rats underwent T2-weighted magnetic resonance imaging (MRI) and diffusion tensor imaging (DTI) before being randomly assigned to experimental groups. Four weeks after therapeutic interventions were initiated, behavioral testing, including rotameter (Rota) and grip strength (GS), were evaluated weekly for the impact of exercise, cells and their combination onContinued.behavioral impairments. After all behavioral testing was completed 10 weeks post-implantation, a final MRI acquisition included measurement of baseline cerebral blood volume (CBV) and forepaw-stimulated functional MRI (fMRI). Animals were perfusion fixed with blood and brain being harvested for ex vivo analyses. (c) The bilateral asymmetry test (BAT) indicated an equivalent performance between all groups for tape removal of the unaffected paw. In contrast, the affected paw revealed a significant level of sensory neglect in all animals with stroke. Evaluating the experimental effect size (i.e. % change between pre- and final time point post-transplantation) revealed that Exercise (−33.67% SEM 8.77), Cells (−19.45%, SEM 14.11) and their Combination (−41.67%, SEM 7.47) reduced the impact of sensory neglect compared to the no treatment MCAo only condition. (d) Performance on motor integration, as assessed by the footfault test, revealed similar effects with Exercise (−44.65%, SEM 8.85), Cells (−57.37%, SEM 8.72) and a combined treatment (−44.43%, SEM 13.06) alleviating the impact of stroke. (e) However, amphetamine-induced asymmetry on the rotameter was not affected by the therapeutic interventions. A rotation bias occurs due to ischemic damage in the right striatum, with rats preferentially turning rightwards (clockwise) in response to a presynaptic dopamine receptor-mediated motor activity increase. (f) Grip strength measurement of the affected paw at the final time point indicated a performance of the Cell group equivalent to controls and significantly improved compared to MCAo only animals, but non-significantly different from the Exercise and Combined groups. Considering the ratio between both paws, however, indicated that the Cells and Combined group had similar grip strength that was significantly different from controls, as well as the MCAo only and the Exercise groups. (p < 0.05, p < 0.01 in comparison to MCAo).
A combination of NSCs and AE promote sub-additive behavioral benefits
Sensory neglect after stroke only affected the left forepaw (Figure 1(c)). The 3 treatments (AE, Cells, Combined) improved sensory neglect compared to MCAo (p < 0.05), but were not significantly different from each other. The combined treatment achieved a 41.67% (SEM 7.41) reduction in neglect, followed by AE (33.67%, (SEM 8.77), and cells (19.45%, SEM 14.11). No improvement in function was evident between the initiation of treatments and 4 weeks post-implantation. Gradual improvements in behavioral functions were only observed when behavioral testing started at 4 weeks. Motor integration of forepaws also only affected the left forepaw after stroke (Figure 1(d)). All treatments reduced footfaults compared to MCAo only (p < 0.01), but there was no significant difference between treatment groups. NSCs implantation achieved the largest effect with a 57.37% (SEM 8.721) reduction in footfaults, with both AE (44.65%, SEM 8.85) and the combined treatment groups (44.43%, SEM 13.06) also achieving a therapeutic improvement. Amphetamine-induced rotations in all MCAo animals was significantly biased compared to controls, with no improvement over time for any of the treatment modalities (Figure 1(e)). At 10 weeks post-implantation, grip strength in the left “affected” forepaw was significantly reduced in MCAo animals (p < 0.01) with implantation of NSCs significantly (p < 0.05) improving this deficit to control levels (Figure 1(f)). The grip strength of the combined group was equivalent to AE, 13% lower than the cell group. However, while accounting for grip strength of the right unaffected paw (i.e. left/right ratio), the combined group performed equivalent to the cell group.
NSCs and AE affect tissue microstructure and connectivity
MR images measured the stroke lesion before and after therapeutic interventions (Figure 2(a)). Quantification of whole brain, lesion, parenchyma and ventricular volume revealed a significant impact of stroke, but none of the treatments affected these measurements (Figure 2(b)). DTI probed tissue microstructure and afforded the visualization of major fiber tracts (Figure 2(c)). MD, an index of cellularity, was reduced in controls at 10 weeks, but significantly (p < 0.05) increased in the striatum of stroke animals (Figure 2(d)). CT significantly increased MD in the striatum and motor cortex compared to MCAo animals. Cells and AE increased (p < 0.05) FA in the striatum and primary somatosensory (S1) cortex, indicating an improvement in tissue structure, but these effects were not evident in the combined treatment group (Figure 2(e)). There was no change in connectivity in the striatum for any of the groups (Figure 2(f)). An increase in connectivity (p < 0.05) was, however, observed in both the control (79.87%, SEM 43.94) and cell group (39.38%, SEM 19.36) in the motor cortex. Although AE revealed increased connectivity in the thalamus (36.96%, SEM 13.68) and somatosensory cortex (61.69%, SEM 31.18), these did not reach statistical significance. Connectivity between the left and right motor cortex was increased for controls, whereas AE increased connectivity of the motor and somatosensory cortex with the striatum (Figure 2(g)). These changes in connectivity did not reach statistical significance.
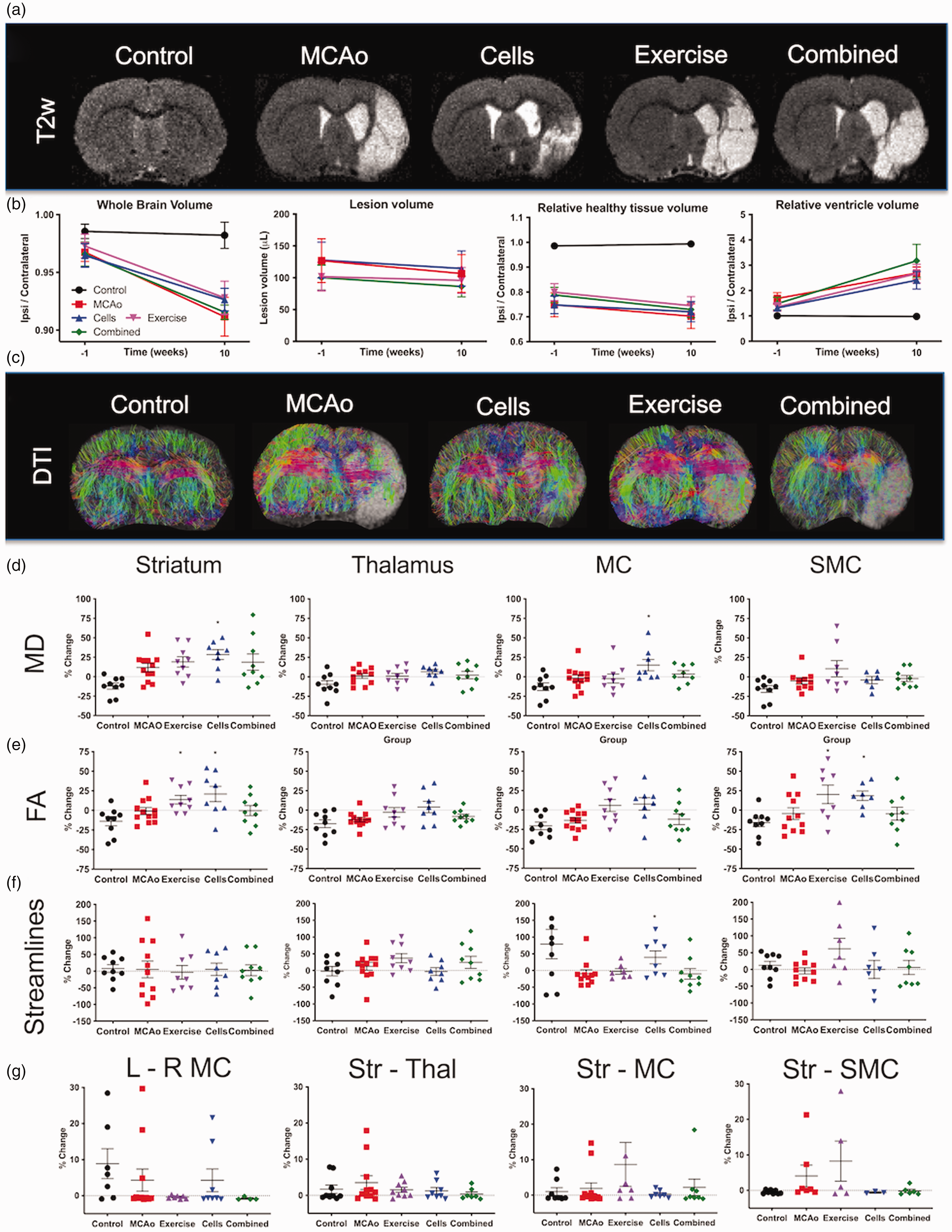
MRI-based volumetry and connectivity. a. T2-weighted (T2w) MR images reveal an extensive hyperintense region in the right hemisphere caused by stroke (10-weeks time point shown here). b. Volumetric analyses revealed a significant decrease in the ipsilateral hemisphere. Neither lesion volume, nor ipsilateral parenchymal volume changed significantly between the pre-implantation and final time point. Ventricular volume in contrast increased significantly over time. However, treatments did not affect these volumetric measurements. c. Diffusion tensor imaging (DTI) visualized the impact of stroke on brain connectivity. d. Measurements of mean diffusion (MD) revealed a significant change in the striatum due to stroke, but no effect of treatment was observed. No significant effects on MD were evident in the thalamus, motor cortex (MC) or somatosensory cortex (SMC). e. Fractional anisotropy (FA) was significantly increased after AE and Cells in the striatum and SMC compared to stroke only animals. FA in the control groups was consistently reduced at 10 weeks compared to the pre-implantation time point. f. No change in streamlines in the striatum were evident, with minor significant changes evident in the thalamus in groups receiving exercise. g. Region-to-region connectivity revealed no significant difference between groups, although exercise non-significantly increased the number of streamlines connecting the striatum with the motor (+9%) and somatosensory cortex (+15%). (p < 0.05 in comparison to MCAo).
The combination of NSCs and AEs affect tissue function, but not baseline CBV
To probe functional tissue changes using fMRI, electrical stimulation of the forepaws revealed a robust activation of the unaffected left S1 for all groups, but a lack of activation in the stroke-affected right S1 for MCAo and AE animals (Figure S6). Implantation of NSCs (p < 0.05) and the combined treatment (p < 0.01) revealed a significant activation of right S1 (Figure 3(a)), including an increased area of activity in both the unaffected and affected S1 regions (Figure 3(b)). Baseline fCBV maps revealed a dramatic loss of blood volume in the lesion cavity (Figure 3(c)) and quantification of fCBV in peri-infarct tissue indicated small increases in the striatum, thalamus, motor cortex, and S1 in MCAo, AE, and combined groups compared to the control (Figure 3(d)). No treatment effects were evident on fCBV measures, except for the thalamus of the MCAo group (p < 0.05).
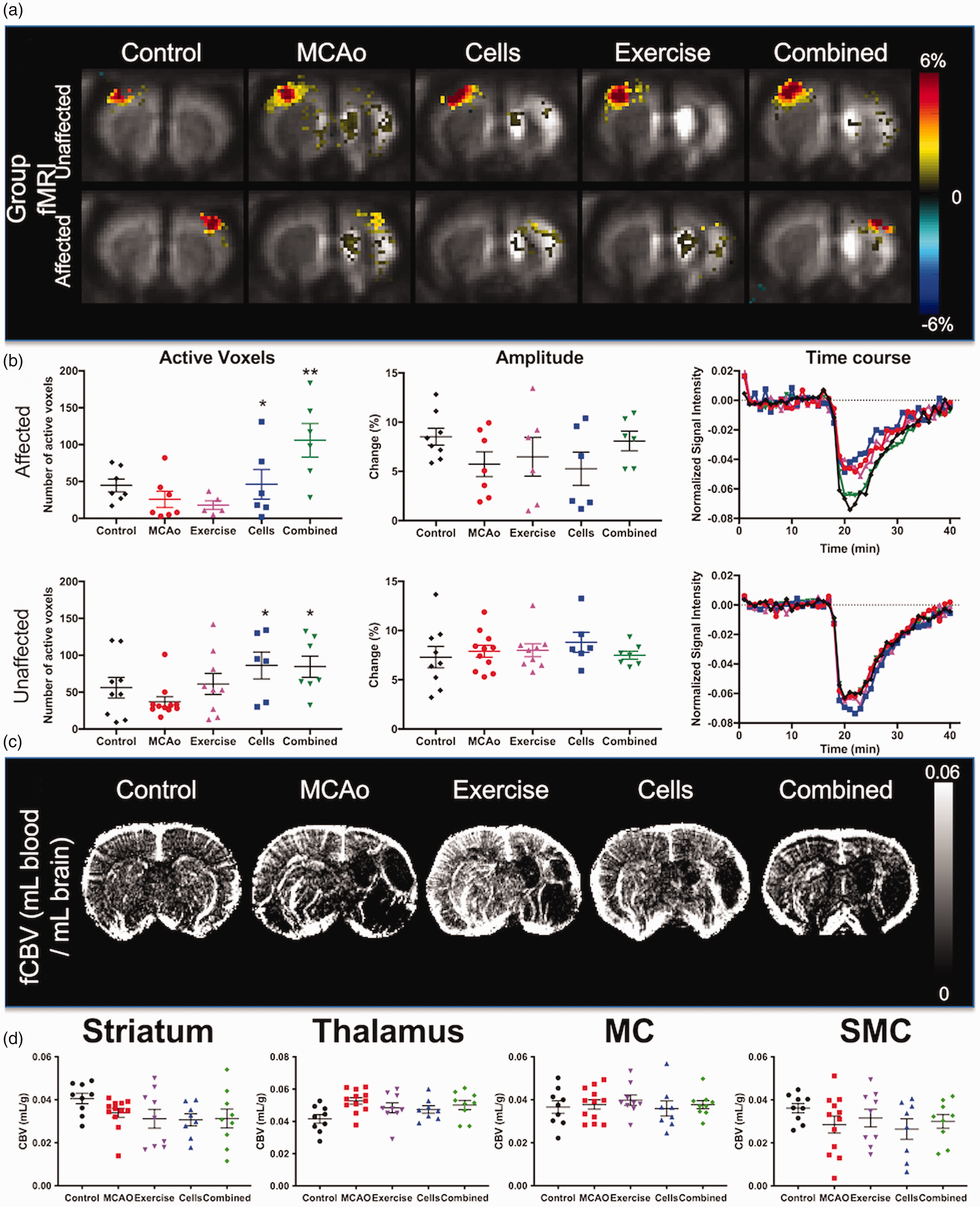
Functional MRI (fMRI) and baseline cerebral blood volume (CBV). a. Group fMRI activation maps reveal brain responses to left (unaffected) or right (affected) S1 activation to forepaw stimulation (p < 0.05 voxel-wise, >80 voxels in a cluster for family-wise error correction). b. Mean activation sizes (i.e. number of active voxels) in bilateral S1 from individual rats show a significant increase in the area of activation for the cells and combined treatment group (p < 0.05 voxel-wise, >9 voxels in a cluster for family-wise correction). The response magnitudes (i.e. % fMRI signal change) and mean signal traces in affected S1 further show response recovery in the combined group. Only animals with significant activation in S1 were included in this analysis (# included/total rats); affected right S1: 8/9 control, 7/12 MCAo, 6/9 exercise, 6/8 cells, 6/9 combined; unaffected left S1: 9/9 control, 11/12 MCAo, 9/9 exercise, 6/8 cells, 7/9 combined. Threshold in A-B: p < 0.05 voxel-wise, >9 voxels in a cluster for family-wise error correction. c. Baseline fractional CBV (fCBV) maps revealed a dramatic loss of blood volume in the area of stroke. d. Quantification of fCBV in different regions of interest revealed an impact of stroke on blood volume in striatum, thalamus, motor cortex (MC), and somatosensory cortex (SMC), but no effect of treatment was evident. (p < 0.05, p < 0.01 in comparison to MCAo).
NSCs, AE and their combination promote the release of different circulating growth factors
It has been suggested that the beneficial effect of exercise on behavioral outcomes may be mediated, at least in part, by circulating myokines. Therefore, ELISAs of blood evaluated how stroke and treatments impacted systemically circulating growth factors (Figure S7). BDNF was reduced in animals with stroke (p < 0.05), but was restored to control levels after implantation with NSCs (p < 0.05). In contrast, no significant effect of stroke or treatment was observed on VEGF-A levels at 10 weeks post-implantation. IGF-1 was significantly (p < 0.05) increased in the cell and combined groups. Klotho was also upregulated after NSCs implantation (p < 0.01) and combined treatment (p < 0.05) compared to MCAo animals. Although Klotho was decreased in the stroke and AE treatment group, these were not significantly different from controls. GDF-10 was increased in all treatment groups compared to the MCAo only group, but only reached statistical significance for the AE and cell group (p < 0.05). IL-6 was increased in the stroke group compared to controls (p < 0.01). All treatments significantly (p < 0.01) reduced IL-6 levels to control levels.
Combination of AE with NSCs does not improve graft survival or differentiation
Implanted NSCs (SC101+ cells) densely packed damaged peri-infarct tissue (Figure 4(a)). No migration out of the deposit area was evident. Implanted cells distributed throughout damaged tissue with an almost complete loss of neurons (Figure 4(b)). Quantification of human cells in rat tissue indicated the presence of an average of 18,874 cells (range 13,790–39,390) in the cell only group and 11,252 cells (41% lower, range 5,410–24,175) in the combined treatment group (Figure 4(c)). In each group, two animals had no human cells surviving by 10 weeks and were excluded from analysis. There was no statistically significant difference in graft survival, graft volume (0.6 μL) or density (26,484 cells/μL, SEM 7099 for cells; 24,916 cells/μL SEM 5747 for combined).
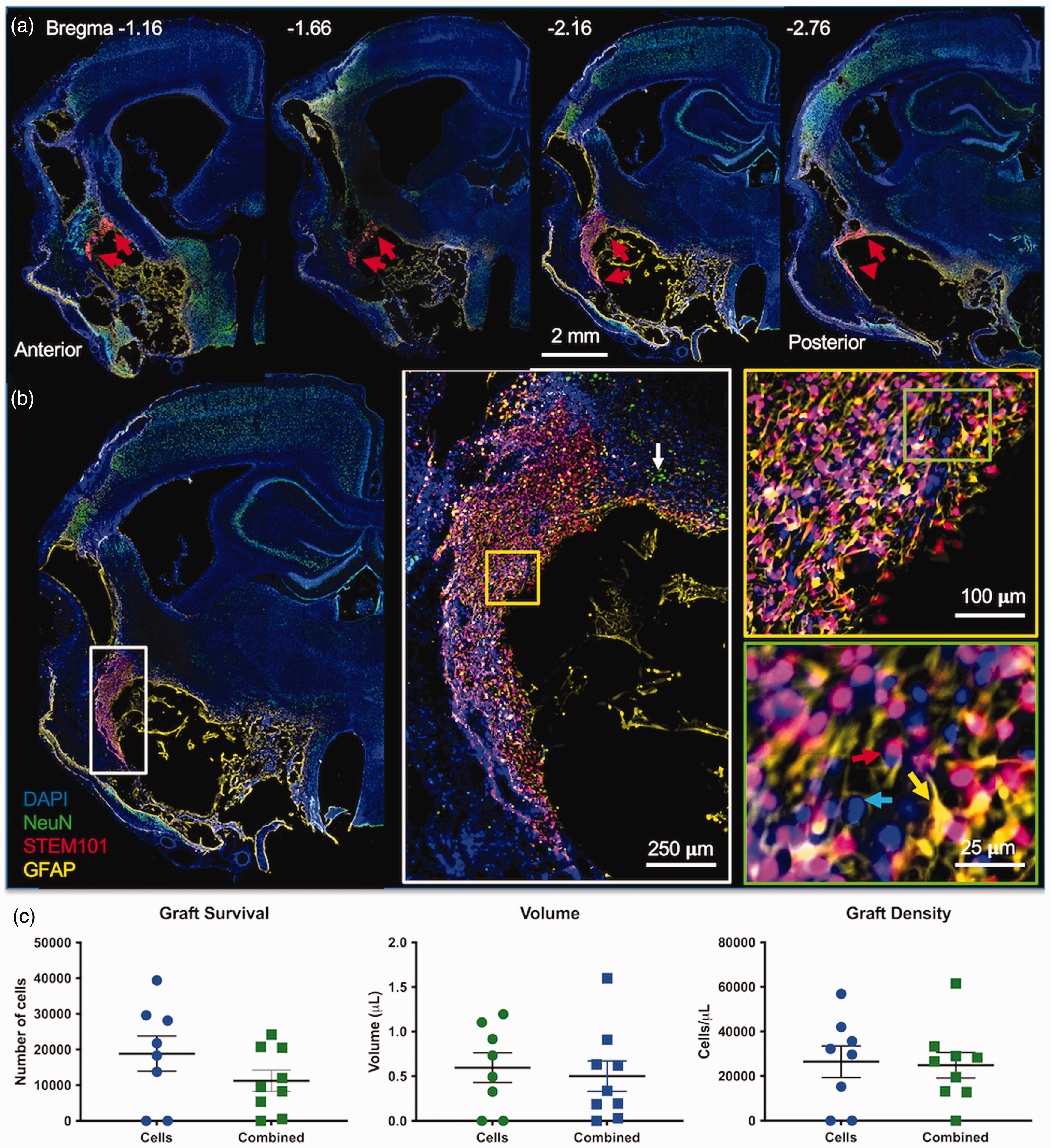
Graft survival. a. Neural stem cells (NSCs, STEM101+, red arrows) were implanted in the damaged peri-infarct tissue and distributed throughout this tissue, but no widespread migration was evident. The anterior-posterior image series of a rat implanted only with NSCs here illustrates the location and distribution of the graft in relation to the stroke damage. b. Nevertheless, implanted NSCs spread through several damaged tissues with very few host cells remaining. These tissues were mostly lacking host neurons (white arrow) with some implanted NSCs (red arrow) differentiating into both neurons (NeuN+/STEM101+) and astrocytes (GFAP, yellow arrow) to repopulate these regions. Very few host cells (blue arrow) were present in the graft core. c. Cell survival in the combined treatment group was reduced by 41%, but this difference did not reach statistical significance. There was no significant difference in graft volume (i.e. distribution of cells) or density of implanted cells in the tissue.
Implanted cells differentiated into neurons (Figure 5(a)), astrocytes (Figure 5(b)), and oligodendrocytes (Figure 5(c)). Neuronal differentiation (Neun+/SC101+) was low (0.81% SEM 0.14 for cells; 0.76% SEM 0.19 for combined) for both groups with most implanted cells differentiating into astrocytes (SC123+, 20.33% SEM 6.94 for the combined group; 29.82% SEM 2.1 for the cells only group) and oligodendrocytes (SOX10+/SC101+, 21.5% SEM 1.05 for cells only; 20.17% SEM 4.88 for combined, Figure 6(d)). SOX10 can also be expressed in some undifferentiated neural stem cells, but almost half of implanted cells here did not exhibit a marker for neurons, astrocytes or oligodendrocytes (including SOX10) at 10 weeks post-implantation, suggesting that cell maturation at this time point might be incomplete.
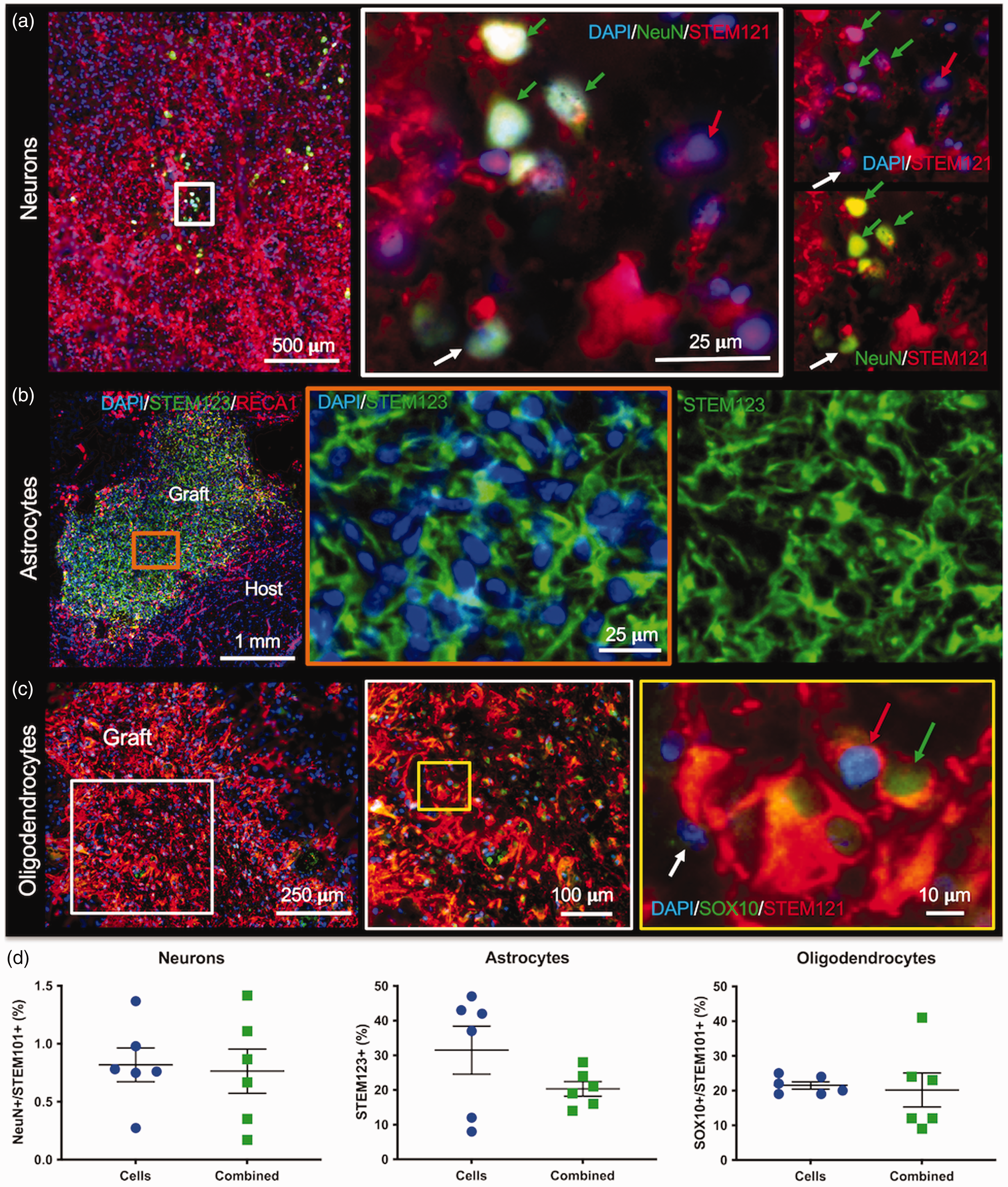
Phenotypic differentiation of implanted cells. a. Neuronal differentiation (NeuN+ cells) of CTXOE03 (STEM121+cells) was rarely observed and mostly commonly occurred in small clusters (green arrows) in the proximity of some host neurons (white arrow). b. The human-specific astrocytes marker SC123 defined the area of the grafted cells against the host background. A tight web of NSC-derived astroctytes was evident with the characteristic processes of reactive astrocytes. c. A considerable proportion of implanted human NSCs (STEM121+) co-localized with the oligodendrocyte marker SOX10 throughout the graft. A fairly homogenous distribution of SOX10 was observed, although in some instances small clusters of SOX10+ cells (green arrow) were observed in densely packed areas of the graft. d. Quantitation of differentiation indicated that <1% of grafted cells become neurons (NeuN+/STEM121+) and approximately 20% oligodendrocytes (SOX10+/STEM121+). Astrocyte (STEM123+) differentiation in the cells only group was almost 10% higher compared to the combined group.
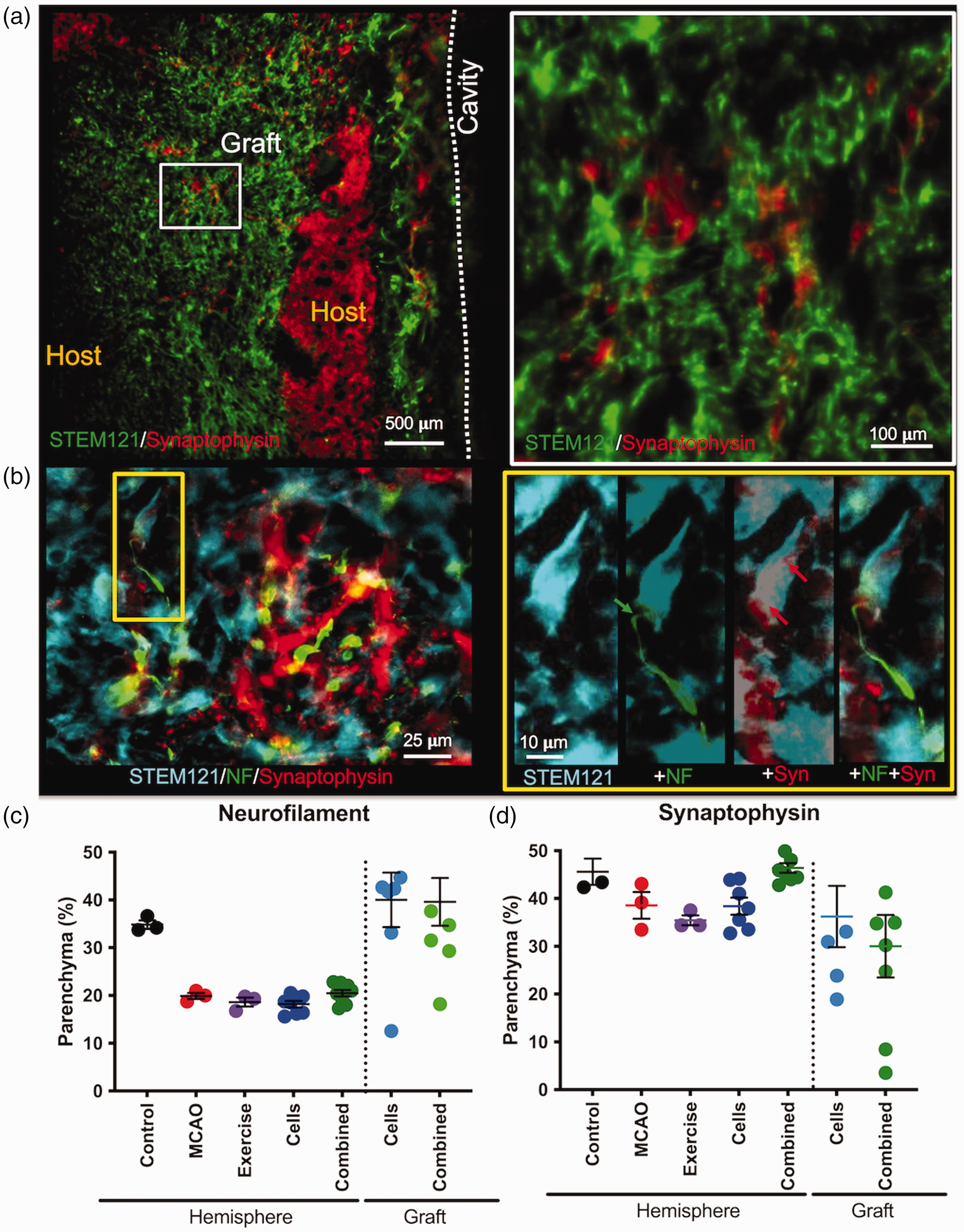
Synaptogenesis and Axons. a. Synaptic density in tissues occupied by grafted cells was low and contrasted with regions containing host neurons. Nevertheless, some grafted cells were surrounded by synaptophysin (Syn)+ synapses. b. In some cases synaptophysin was evident on the cell body of a grafted cell that extended a neurofilament (NF) containing axon. c. Quantification of neurofilament revealed a significant decrease in the ipsilateral hemisphere compared to controls, but axonal density within the graft was increased in both the cell and combined groups. d. Synaptophysin was also reduced in the ischemic hemisphere compared to controls. A combined cell and exercise treatment significantly increased synapse density to control levels. Synapse density in the graft area for both the cells and combined groups was equivalent to that observed in MCAo only animals.
Although there was a very low neuronal differentiation, there was evidence of connectivity with synaptophysin+synapses present within the graft area (Figure 6(a)). Neurofilament+ axons extended from implanted cells and these neurons also were associated with synapses on their cell body (Figure 6(b)). Although neurofilament revealed a decrease in the stroke-damaged tissue, in the graft area there was a significant increase in axonal density in both groups (Figure 6(c)). Nevertheless, the level of synaptic density was decreased in the stroke-damaged hemisphere, as well as the graft area (Figure 6(d)).
A combined AE and NSCs enhanced angiogenesis
Blood vessel density in peri-infarct tissues was increased compared to unaffected tissues, and was associated with an increased microglia activity (Figure 7(a)). In the graft area, blood vessel density was further increased compared to surrounding tissue (Figure 7(b)). Quantification of blood vessel density further indicated that there was a significant increase (p < 0.01) within the graft area of both experimental cell groups compared to the levels found in untreated peri-infarct tissues (Figure 7(c)). Exercise, cells and their combined treatment also increased (p < 0.05) blood vessel density in peri-infarct tissues compared to MCAo only. The graft area was densely packed with microglia, which were also associated closely with blood vessels in this area (Figure 7(d)). A quantitative comparison revealed a significant (p < 0.05) increase in microglia across the ischemic hemisphere compared to controls, but no effect of treatment modality (Figure 7(e)). Nevertheless, microglia in the grafted area were very significantly (p < 0.001) upregulated compared to the peri-infarct tissue. These results indicate that implantation of NSCs exerts potential therapeutic “bystander” effects on host brain tissue.
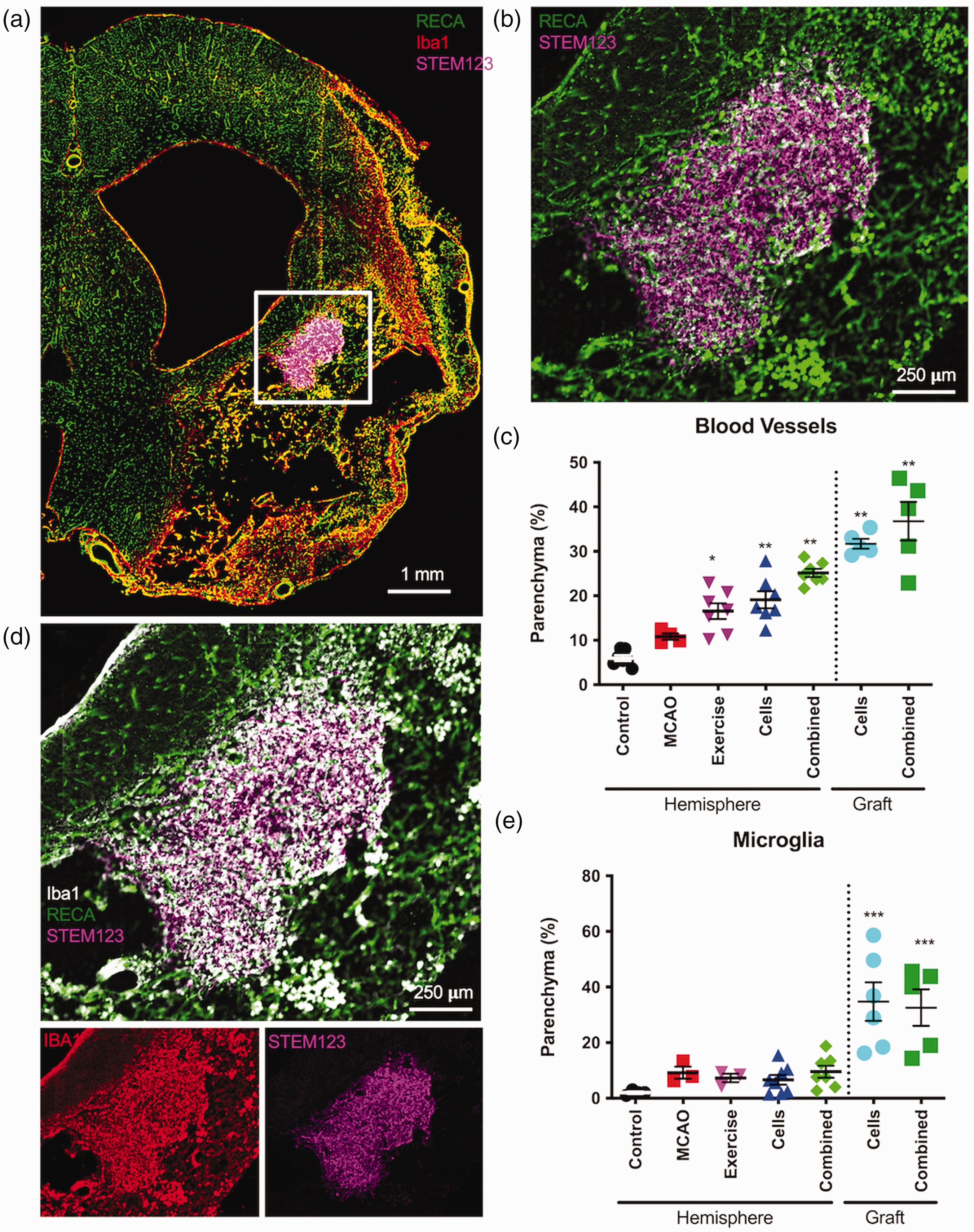
Angiogenesis and microglia response. a. Whole hemisphere images visualizing blood vessels (rat endothelial cell antigen, RECA-1), as well as microglia (Iba1) activity. b. In the graft area (SC123 marking human cells), an increased density of blood vessels was evident. c. Quantification of the area of blood vessels in the parenchyma and in the grafted area revealed a significant increase in blood vessel density in the cells and combined groups. In the graft area, the density of blood vessels was higher than in the ipsilateral hemisphere. d. Microglia density in the graft area was also increased compared to surrounding damaged tissue. e. Quantification of microglia density was increased due to MCAO, but only a localized increase in the grafted area was evident. (p < 0.05, p < 0.01, p < 0.001 in comparison to MCAo).
Discussion
A key outstanding question for the clinical translation of the intraparenchymal implantation of NSCs in stroke patients is how PT can be applied to augment therapeutic efficacy. Here, we demonstrated that AE, which is a common component of rehabilitation programs for patients with stroke, can provide sub-additive improvements on some behavioral tasks, but also potentially attenuates the beneficial effects of cell therapy on others tasks.
Sub-additive and potentially suppressive behavioral effects of AE on NSC efficacy
Complex neurological disorders, such as stroke, affect many brain regions involved in a variety of behavioral tasks. The use of a test battery efficiently probes these different deficits, but also highlights the challenge to achieve improvements on all these disparate impairments. 36 Applying multiple complimentary treatments could hence be essential to achieve the best outcome for patients with stroke. 47 As PT is routinely given to patients with stroke, a key question addressed here was how this intervention interacts with intraparenchymal NSCs implantation.1,2 This question is particularly relevant given that PT, like CT, is aimed at enhancing neuroplasticity. 48 Therapeutic interactions can be additive (i.e. each therapeutic targets separate anatomical substrates and the effect size is the sum of both individually), synergistic (i.e. overall therapeutic effect is greater than their sum), suppressive (i.e. one therapy reduces the effect of the other) or antagonistic (i.e. one therapeutic abrogates the effect of the other).49,50 Here, we observed both sub-additive (i.e. combined effect lower than the sum of individual effects), as well as suppressive, effects of combining AE with NSCs implantation.
Intraparenchymal NSC or AE here exerted similar therapeutic effects on somatosensory and motor functions. The combination of both produced a sub-additive effect on somatosensory dysfunction (i.e. BAT), but AE suppressed the therapeutic effects of NSCs on motor functions (i.e. footfault and grip strength). MCAo only animals revealed a consistent deficit for 10 weeks indicating that the behavioral testing here was resistant to spontaneous recovery, as has been previously observed for up to 1 year after stroke. 51 No therapeutic effects were evident for the first 4 weeks during which no behavioral testing was performed. The extensive behavioral testing in combination with AE and NSCs, however, could have exerted a task specificity and integration of function that is often considered essential for multi-modal PT to harness the benefits of neuroplasticity.52,53 It is noteworthy that at 10 weeks, the recovery curves had not yet plateaued, suggesting that longer behavioral assessment could produce a more extensive recovery. We previously reported that NSCs implanted into ischemic lesions only involving the striatum exhibited a more pronounced therapeutic effect. 3 However, it remains unclear here if sub-group effects occur. A larger sample size (>20 rats/group) would be required to contrast striatal versus striatal+cortical stroke effects in all treatment groups.
Only a single dose of AE and “task integration” was delivered here, which could limit the generalizability of the results. As with different enriched environments, a different combination of behavioral tests could lead to a different outcome, if recovery is task-specific. As grip strength was only administered at 10 weeks post-implantation and exhibited the same pattern of deficits and recovery observed on the footfault test, it suggests that some of these effects are not highly task-specific. The suppressive effect of AE on NSC-mediated motor recovery raises the question of therapeutic dosing. AE intensity (80%) here was based on normal rather than stroke animals. 24 Overdosing of AE or induction of stress in animals undergoing PT could potentially suppress the effects of NSCs and abrogate a combined effect by reducing BDNF levels.54,55 BDNF levels for the combined treatment group here were lower than the cell group, even though no differential effect of stress levels between groups was evident. Future experiments will need to investigate how AE and task integration combine to exert rehabilitative effects and provide a better understanding of how these can be exploited to enhance the recovery observed after NSCs implantation.
Biological underpinnings of PT and NSC therapy
Understanding of the individual and interactive effects of PT and CT is dependent on the biological foundations of their therapeutic targets. For NSC implantation, the number of surviving cells provides the mode of action. NSCs’ survival was robust in 80% of animals, consistent with previous reports. 3 No significant impact of AE on graft volume, cell survival, density, differentiation, axonogenesis or synaptogenesis was evident. Although grafted cells increased microglia density in the area of implantation, AE did not affect this increase. In contrast, AE did increase blood vessel density and exerted an added effect to the angiogenesis observed after implantation of NSCs in the ischemic hemisphere, as well as a more pronounced local effect in the graft area. Blood vessel density and improvement in vascularization therefore appear to be crucial to the observed therapeutic effects of AE, NSCs and their combination. Promotion of angiogenesis and the resulting increase in vascularization have previously been demonstrated to support the efficacy of NSCs.3,56 Nevertheless, no effect on CBV was evident on MRI scans.
The impact of AE, NSCs and their combination did not affect lesion volume or other volumetric MRI measures, consistent with previous observations. 3 However, there were microstructural changes in the striatum after cell implantation that indicate an improvement of tissue cellularity and structure, as indicated by MD, as well as FA. Changes in connectivity were limited to the AE group with improved connectivity between the striatum and the motor/somatosensory cortex. An improved connectivity could partially be due to a restoration of anisotropic tissue structure in the striatum that affords better tractography. However, an increased axon density in the graft area, as indicated by neurofilament, could also indicate new connectivity targets for fibers from cortical tissues, as also demonstrated by others. 57 Synaptic inputs from the stroke-injured brain to implanted NSCs, as shown here and previously, 58 potentially provides Hebbian synapses required for a functional integration of NSCs into a neuronal circuit. Indeed, cortical areas exhibited changes in functional response to forepaw stimulation after cell transplantation and combined treatment. As these areas are remote from the graft site, it is tantalizing to speculate that these functional changes were downstream from the restoration of striatal connectivity or improved signal transmission in peri-infarct tissues. Optogenetic investigations of transplanted NSCs further support these downstream functional effects that are remote from the area of implantation.59–61
In addition to axonal connections exerting remote effects in the brain, AE and implanted cells also can affect other brain regions through growth factors that circulate in the blood. It is well established that AE releases growth factors in the blood that induce changes in the brain.24,62 In part, the therapeutic effects of NSCs are exerted through the release of growth factors, such as BDNF and VEGF-A, and the release of these from NSCs is dramatically increased in the presence of endothelial cells.9,10 Achieving an increase in angiogenesis and blood vessels through AE could hence potentiate the growth factor release by NSCs in situ and provide the basis for a sub-additive therapeutic effect.
Translational considerations
Considering that AE, NSCs and their combination only resulted in behavioral improvement after initiation of behavioral testing potentially suggests that some task integration is crucial to harness the neuroplasticity induced by these therapeutic approaches. It remains unclear if or at what point the task integration will cease to yield a benefit, as our study was terminated 10 weeks post-implantation. Longer-term studies will therefore be important to address this issue, ideally with the inclusion of a group that does not receive any weekly behavioral testing. We propose that AE and NSCs alone or their combination are insufficient to promote a significant improvement of behavioral impairments. We further stipulate that both AE and NSCs implantation have a significant overlap in their therapeutic effects and therefore only a sub-additive effect was evident in the combined therapy group.
Two crucial differences need to be considered that potentially limit the translational impact of these results. First, young rats were included in this study, which are known to recover more efficiently than older subjects. The demographics of stroke, however indicate that older subjects have a much higher incidence of stroke. 63 The impact of AE and NSCs on an aged brain could potentially have a more pronounced effect by inducing neuroplasticity in an environment that typically is less adept at repairing itself. 64 Second, implantation of NSCs was performed 2 weeks post-stroke. At this time point, a considerable spontaneous recovery due to neuroplasticity still occurs. In a clinical setting, patients would typically receive PT during the sub-acute phase of stroke to improve functional outcome. In contrast, clinical trials for NSCs implantation to date have only enrolled patients with a stable deficit that was no longer improved using PT. 65 Implantation of NSCs combined with AE in these patients could re-open a neuroplasticity window to be exploited by task integration with effect sizes larger than observed here.
Conclusion
As NSCs implantation is undergoing clinical translation, key issues such as the interaction with neurorehabilitation protocols remain poorly addressed.1,2 The main focus here has been on PT and specifically AE to determine its impact on NSCs implantation. Both sub-additive and suppressive effects of AE on CT have been observed, as well as an indication that behavioral testing, which is typically part of a multi-modal PT paradigm, might exert a task integration function. Although both therapies exerted beneficial effects, it remains unclear if different levels of PT or CT are required to promote a maximum combined recovery. Our results here highlight the need to conduct controlled preclinical studies to inform clinical trial designs. This bench-to-bedside-to-bench effort will be an essential process to ensure that intraparenchymal NSCs implantation will be as efficacious as possible for a wide-ranging population of patients with stroke.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X211062955 - Supplemental material for Physical therapy exerts sub-additive and suppressive effects on intracerebral neural stem cell implantation in a rat model of stroke
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X211062955 for Physical therapy exerts sub-additive and suppressive effects on intracerebral neural stem cell implantation in a rat model of stroke by Harmanvir Ghuman, Nikhita Perry, Lauren Grice, Madeline Gerwig, Jeffrey Moorhead Jr.Franziska Nitzsche, Alexander John Poplawsky, Fabrisia Ambrosio and Michel Modo in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding from the Alliance for Regenerative Rehabilitation Research & Training (AR3T), which is supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), National Institute of Neurological Disorders and Stroke (NINDS), and National Institute of Biomedical Imaging and Bioengineering (NIBIB) of the National Institutes of Health under Award Number P2CHD086843 and R56AG052978. AJP was supported by R01EB003324.
Acknowledgements
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The authors thank Ms. Jessie Liu for blinding of the experimental conditions and Mr Brendon Wahlberg for technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Harmanvir Ghuman: Collection and/or assembly of data, data analysis and interpretation
Nikhita Perry: Collection and/or assembly of data, data analysis and interpretation
Lauren Grice: Collection and/or assembly of data, data analysis and interpretation
Madeline Gerwig: Collection and/or assembly of data, data analysis and interpretation
Jeffrey Moorhead: Collection and/or assembly of data, data analysis and interpretation
Franziska Nitzsche: Collection and/or assembly of data, data analysis and interpretation
Alexander J. Poplawsky: Collection and/or assembly of data, data analysis and interpretation,
Fabrisia Ambrosio: Conception and Design, Financial Support, Data analysis and interpretation
Michel Modo: Conception and Design, Financial Support, Data analysis and interpretation, Manuscript Writing, Final Approval of Manuscript
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
