Abstract
The functions of the epidermal growth factor (EGF) family members in the adult brain are not known. This study investigated the changes in the expression of members of the EGF family following global ischemia employing in situ hybridization and immunohistochemical techniques to elucidate their roles in pathological conditions. EGF mRNA was not detected in either the control or the postischemic rat brain. Although transforming growth factor-α (TGF-α) mRNA was widely expressed in the normal brain, its expression did not change appreciably following ischemia. By contrast, heparin-binding EGF-like growth factor (HB-EGF) mRNA expression was rapidly increased in the CA3 sector and the dentate gyrus of the hippocampus, cortex, thalamus, and cerebellar granule and Purkinje cell layers. EGF receptor mRNA, which was widely expressed, also showed an increase in the CA3 sector and dentate gyrus. Conversely, HB-EGF mRNA did not show any increase prior to ischemic neuronal injury in the CA1 sector, the region most vulnerable to ischemia. Immunohistochemical detection of HB-EGF in the postischemic brain suggested a slight increase of immunostaining in the dentate gyrus of the hippocampus and the cortex. These findings showed that the gene encoding HB-EGF is stress-inducible, indicating the like-lihood that HB-EGF is a neuroprotective factor in cerebral ischemia.
Keywords
Accumulating evidence suggests that neurotrophic factors play an important role, not only during development but also in adulthood, for neuronal survival and protection against various insults such as trauma, excitotoxicity, and ischemia. One line of evidence stems from the observation that a wide variety of insults up-regulate various neurotrophic factors. For instance, mRNA for brain-derived neurotrophic factor (BDNF), a member of the neurotrophin family, is up-regulated following seizures (Zafra et al., 1990), trauma (Ballarín et al., 1991), glutamate toxicity (Gwag and Springer, 1993), hypoglycemia, ischemia (Lindvall et al., 1992), and spreading depression (Kawahara et al., 1997). Similar up-regulation was observed for basic fibroblast growth factor mRNA (Endo et al., 1994; Gall et al., 1994). These findings suggest that neurotrophic factors act as endogenous neuroprotectants against noxious stimuli. In fact, exogenous administration of some growth factors protects neurons against ischemic damage. Protective effect against either focal or global ischemia in vivo has been shown for nerve growth factor (Shigeno et al., 1991), BDNF (Beck et al., 1994), basic fibroblast growth factor (Koketsu et al., 1994), and insulin-like growth factor 1 (IGF-1) (Gluckman et al., 1992).
Among these growth factors, epidermal growth factor (EGF) is one of the prototypes and has been shown to be expressed in the adult rodent brain by Fallon and colleagues (1984). Prompted by this finding, several laboratories have demonstrated its neurotrophic activities in vitro, such as survival-promoting and neurite outgrowth effects (Morrison et al., 1987) and neuroprotective effects against anoxia and nitric oxide toxicity (Maiese et al., 1993). Subsequent studies, however, have failed to confirm the presence of EGF in the adult rodent brain (Probstmeier and Schachner, 1986; Kaser et al., 1992; Yeh et al., 1992) and suggest a more important role for transforming growth factor-α (TGF-α), another member of the EGF family, as an endogenous ligand for EGF receptor (EGFR) in the adult brain (Kaser et al., 1992).
Recently, we have reported that heparin-binding EGF-like growth factor (HB-EGF), a recently identified member of the EGF family, first isolated from macrophages as a potent mitogen for vascular smooth muscle cells (Higashiyama et al., 1991), is abundantly expressed in the adult rat brain (Mishima et al., 1996). In view of the fact that HB-EGF exerts its biological activity by binding to EGFR (Higashiyama et al., 1991, 1995), this observation suggested a role for this factor in the maintenance and survival of neurons in the mature brain.
In spite of these studies, the functions of members of the EGF family in the mature brain have not been well characterized to date. One of the approaches to elucidate their functions is to examine their expression in response to various physiological or pathological stimuli. In the present study, therefore, we employed cerebral ischemia as a pathological insult and examined changes in the expression of the EGF family members (EGF, TGF-α, and HB-EGF) in conjunction with EGFR. Our data have shown that HB-EGF mRNA is selectively up-regulated in the postischemic brain and suggested a role in cerebral ischemia as a neuroprotective factor.
MATERIALS AND METHODS
Experimental groups
In the present study, a total of 50 male Sprague-Dawley rats weighing 280 to 350 g (Charles River Japan, Yokohama, Japan) were used. The rats were maintained on a 12:12-hour light/dark cycle with food and water made available ad libitum. For in situ hybridization study, the animals were divided into the following groups: In the ischemia group (n = 18), rats were subjected to global ischemia and killed at 2, 6, 12, and 24 hours and 3 and 7 days following recirculation (n = 3 at each time point); in the sham group (n = 12), rats were subjected to the sham operation and killed at the same time intervals (n = 2 at each time point); in the control group (n = 5), rats were killed immediately following the sham operation. For immunohisto-chemical study, the animals were killed at 2, 6, and 24 hours and 3 days following recirculation (n = 3 at each time point). As control served animals killed immediately following the sham operation (n = 3). All animal-related procedures were conducted in accordance with the guidelines of the National Institutes of Health (Guide for the Care and Use of Laboratory Animals).
Induction of global ischemia
Transient global cerebral ischemia was induced by cardiac arrest as described previously (Kawai et al., 1992; Kawahara et al., 1995), with minor modifications. After induction of anesthesia, the rats were intubated and mechanically ventilated with 1.0% halothane in a 30% O2/70% N2O mixture. The femoral artery and vein were cannulated for monitoring blood pressure and intravenous injection. Following midsternal skin incision, a bent hook was inserted into the right thoracic cavity and rotated underneath the heart. Then, mechanical ventilation was discontinued and the major cardiac vessels were occluded by pulling the hook and simultaneously applying finger pressure from outside. Resuscitation was initiated at 7 minutes with external cardiac massage and mechanical ventilation. The duration of ischemia was defined as the time from the onset of vascular compression to restoration of blood pressure to 50 mm Hg. Acid-base balance was corrected by intravenous injection of 7% sodium bicarbonate. Mechanical ventilation and blood pressure monitoring were discontinued 30 minutes after recirculation, and the animals were returned to their cages after skin closure.
Rectal temperature was controlled by a heating blanket (Animal Blanket Controller model ATB-1100; Nihon Kohden), and temporal muscle temperature was monitored with a thermometer (model BAT-12; Physitemp Instruments, Clifton, NJ, U.S.A.) via a needle microprobe (MT-23/5; Physitemp Instruments). The rectal temperature was controlled before and after ischemia at ∼37.5°C. Arterial blood samples were analyzed by a blood gas analyzer (model 248; Chiron Diagnostics, Essex, U.K.) and a glucose analyzer (Glu-1; TOA Electronics, Tokyo, Japan).
For the sham operation, animals were subjected to the same procedure as described above, but the hook was withdrawn without occlusion of the vessels.
Probe preparation
The probes for mRNA in situ hybridization were prepared by in vitro transcription of complementary DNA (cDNA). For EGF, a partial cDNA (794 bp) of rat prepro-EGF corresponding to nucleotides 2,783 to 3,576 (Saggi et al., 1992) was amplified by polymerase chain reaction following reverse transcription of total RNA prepared from the rat liver. The primers used were 5′-AGT AAA GAG GTG GCA TCG TTG G (sense primer) and 5′-CGA GTC CTG TAG TAG TAA GTC C (antisense primer). Polymerase chain reaction was conducted with 35 cycles at an annealing temperature of 55°C. For TGF-α, a partial cDNA (905 bp) of rat TGF-α corresponding to nucleotides 179 to 1,083 (Blasband et al., 1990) was prepared by reverse transcription polymerase chain reaction as described above. The primers used were 5′-CGC TGG GTA TCC TGG TAG CTG T (sense primer) and 5′-AGA CAC ATC ATC CTG GCA ACC G (antisense primer). The sequences of these partial cDNAs were confirmed by restriction enzyme analyses. A partial cDNA of rat HB-EGF corresponding to nucleotides 20 to 613 (Abraham et al., 1993) was obtained as described previously (Mishima et al., 1996). The 2.2-kbp EcoRI fragment of rat EGFR cDNA (Petch et al., 1990) was kindly provided by Dr. H. S. Earp (Chapel Hill, NC, U.S.A.).
Antisense and sense RNA probes were prepared from each cDNA subcloned into a plasmid [pBluescript SK(+)] using an RNA-labeling kit (Riboprobe Gemini System; Promega, Madison, WI, U.S.A.) with T7 and T3 RNA polymerases. The probes were labeled by [α-35S]UTP (SJ603; Amersham, Buckinghamshire, U.K.) to a specific activity of ∼1 to 2 × 106 cpm/ng.
In situ hybridization study
The animals were killed at various time intervals as described above in each group under deep anesthesia by intraperitoneal injection of sodium pentobarbital. The brains were immediately removed, frozen in powdered dry ice, and cut into 12-μm-thick sections at −20°C using a cryostat. The forebrains were cut coronally to obtain two different sections corresponding to approximately −3.0 and −4.16 mm from the bregma (Paxinos and Watson, 1986). For the cerebellum, midsagittal sections were obtained similarly. The sections were stored at −70°C for later use.
On the day of in situ hybridization study, the sections were fixed in 4% paraformaldehyde in phosphate-buffered saline (pH 7.4) and acetylated for 10 minutes with 0.25% acetic anhydride in 0.1 mmol/L triethanolamine-HCl (pH 8.0). Following dehydration in ascending graded alcohols, the sections were prehybridized at 55°C with the hybridization solution (0.3 mol/L NaCl, 5 mmol/L ethylenediaminetetraacetate, 50% formamide, 1× Denhardt's solution, 10 mmol/L Tris-HCl, pH 7.0, 10 mmol/L dithiothreitol, 0.1 mg/ml yeast tRNA, 10% dextran sulfate) for 3 hours. The sections were then washed in the wash buffer (0.3 mol/L NaCl, 5 mmol/L ethylenediaminetetraacetate, 50% formamide, 10 mmol/L Tris-HCl, pH 7.0, 10 mmol/L dithiothreitol) for 10 minutes, dehydrated in ascending graded alcohols, and hybridized with each cRNA probe at 55°C for 16 hours at the concentration of 1.0 × 106 cpm/50 μL in the hybridization solution. On the next day, high-stringency washes were then carried out at 55°C for EGF, TGF-α, and HB-EGF and at 60°C for EGFR in the wash buffer for 30 minutes. The remaining nonhybridized cRNA probes were degraded using RNase A (20 μg/mL) in RNase buffer (0.5 mol/L NaCl, 10 mmol/L Tris-HCl, pH 7.5, 0.1 mol/L ethylenediaminetetraacetate) at 37°C for 30 minutes. The sections were again washed in the wash buffer for 30 minutes, then in 2× saline sodium citrate and 0.1× saline sodium citrate at 45°C for 30 minutes each, and dehydrated in ascending graded alcohols. The dehydrated sections were exposed to BioMax MR film (Eastman Kodak, Rochester, NY, U.S.A.) for 3 to 7 days along with a radioactive standard strip. For emulsion autoradiography, the hybridized sections were dipped in liquid emulsion (NTB-2; Eastman Kodak), developed 3 to 5 weeks later, and lightly stained with hematoxylin.
Densitometric analysis
To make comparisons at different time intervals, one set of sections from each time point was exposed to the same film along with a corresponding section from a control rat. Optical density was calibrated with the radioactive standard, using the public domain program NIH Image 1.59 (W. Rasband, NIH, Bethesda, MD, U.S.A.). The mean density of each area was compared with that in the control sections and expressed as a percentage change from the control. The brain regions examined were the striatum, the pyramidal layers of the CA1 (medial/lateral) and CA3 sectors, the granule cell layer of the dentate gyrus of the hippocampus, the ventral part of the thalamus, the outer layer of the cortex (middle third), and the granule cell layer of the cerebellum.
Statistical analysis
The mean values at different time points within each group were analyzed by one-way analysis of variance using Bonferroni's post hoc test for multiple comparisons. The differences between the sham and control groups were compared using the unpaired t test.
Immunohistochemical study for HB-EGF
The animals were reanesthetized with halothane and immediately killed at various time intervals following recirculation as described above by thoracotomy and transcardiac perfusion with 4% paraformaldehyde in 0.1 mol/L phosphate buffer (pH 7.4). The brains were removed, postfixed overnight at 4°C, and subsequently embedded in paraffin. Tissue sections (4 μm) were deparaffinized, treated with 0.3% H2O2 in methanol for 20 minutes, and microwaved in 10 mmol/L citrate buffer. After incubation in 5% normal goat serum in Tris-buffered saline (50 mmol/L Tris, 150 mmol/L NaCl, pH 7.6) for 20 minutes, the sections were kept in the rabbit anti-HB-EGF antibody (H1) at a dilution of 1:100 in Tris-buffered saline with 1% bovine serum albumin at 4°C overnight. This antibody specifically recognizes the HB-EGF precursor COOH-terminal residues 185 to 208 (intracellular domain) and does not cross-react with the mature secreted form of HB-EGF and other members of EGF family (Miyagawa et al., 1995). The following day, the sections were incubated in biotinylated goat anti-rabbit immunoglobulins for 30 minutes at a dilution of 1:500 in Tris-buffered saline and in avidin-biotin-peroxidase (Vectastain ABC Kit; Vector Laboratories, Burlingame, CA, U.S.A.). Then the tissue sections were visualized with 3,3′-diaminobenzidine in the presence of hydrogen peroxide (DAB Peroxidase Substrate Kit; Vector Laboratories), dehydrated in graded alcohols and xylene, and coverslipped. As a negative control, the primary antibody (H1) was omitted or replaced by normal rabbit immunoglobulins.
RESULTS
Physiologic variables for each group
In the present study, a constant recirculation time (ischemia time) of 10.0 ± 0.6 minutes (mean ± SD) was obtained in the ischemia group with no statistically significant differences among the groups in terms of time, as determined by analysis of variance (Table 1). In regard to physiological variables, the comparison of the mean values, as shown in Table 1, did not disclose any statistically significant differences among the various groups.
Physiologic variables

As within-group analysis with regard to time using ANOVA did not reveal any significant differences, the values were pooled in each group. Furthermore, values from the control and sham groups were pooled as “control group” because no significant differences were noted between these groups. For each variable before ischemia, the unpaired t-test did not reveal any significant differences between the pooled control and ischemia groups. MABP, mean arterial blood pressure; NA, not available.
Expression of EGF mRNA
In situ hybridization for EGF mRNA did not reveal any specific hybridization signal under our experimental conditions in the control rats (Fig. 1A and B). Following either ischemia or the sham operation, no increase in the signals for EGF mRNA was noted up to 7 days (data not shown).
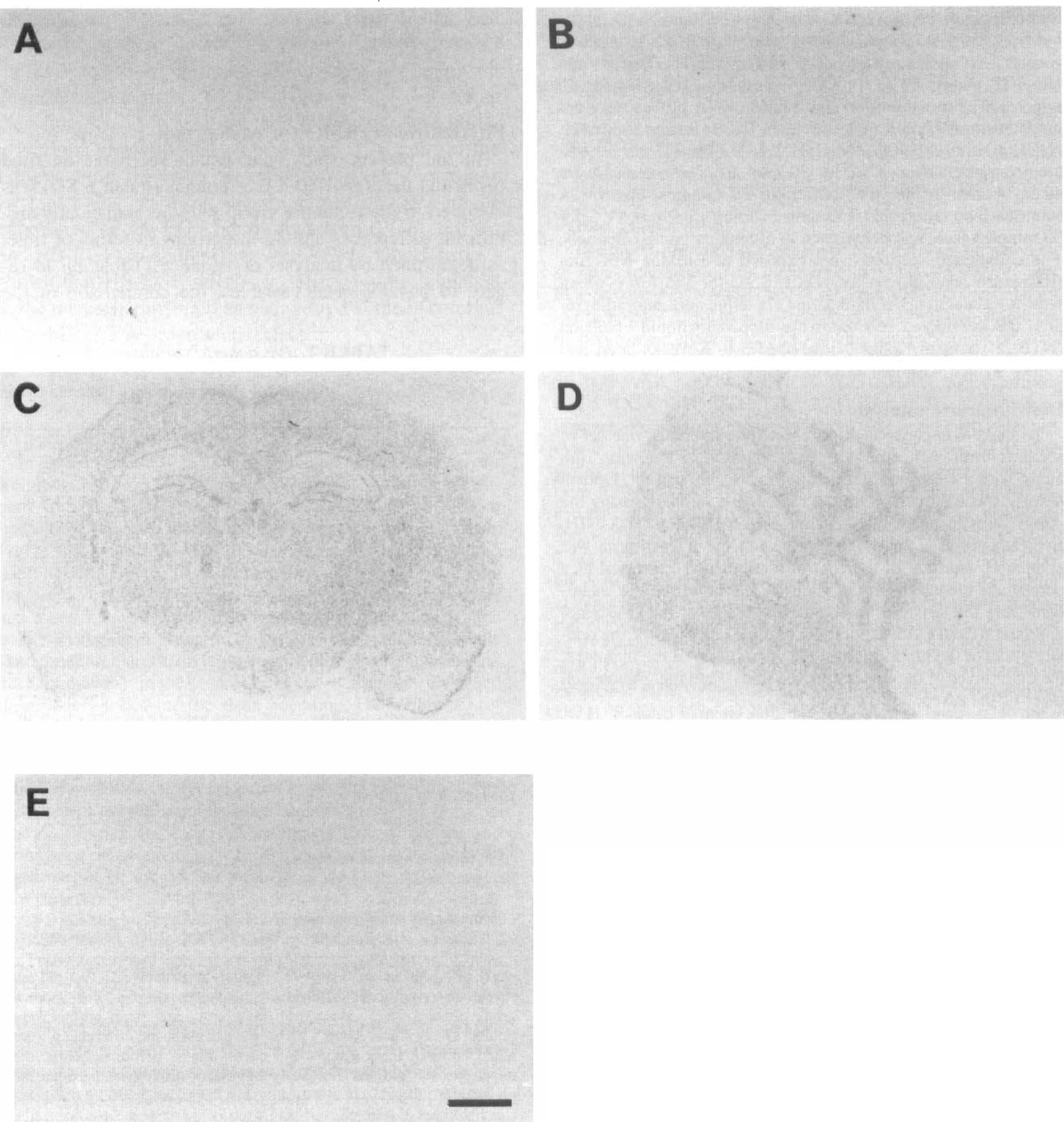
Autoradiographs of EGF and TGF-α mRNA expression. No specific hybridization signals for EGF mRNA were noted in the control animals using antisense probes
Expression of TGF-α mRNA
In the control animals, moderate hybridization signals specific for TGF-α mRNA were detected in the white matter of the forebrain, cerebellum, and brainstem (Fig. 1C to E). Relatively diffuse signals were also noted throughout the cortex, thalamus, and striatum. In the hippocampus, the granule cell layer of the dentate gyrus and the pyramidal layer of CA1 to CA3 sectors exhibited moderate hybridization signals. The expression and distribution of the hybridization signals did not show any change following either the sham operation or ischemia, except in the CA1 sector of the hippocampus at 7 days after ischemia, where the hybridization signals were lost simultaneously with neuronal death (data not shown).
Alteration of HB-EGF mRNA
In contrast to TGF-α, specific hybridization signals for HB-EGF mRNA were mainly detected in the superficial cortex and hippocampus of the forebrain and in the granule and Purkinje cell layers of the cerebellum (Fig. 2A and G). The pattern of expression was similar after the sham operation at all time points examined.
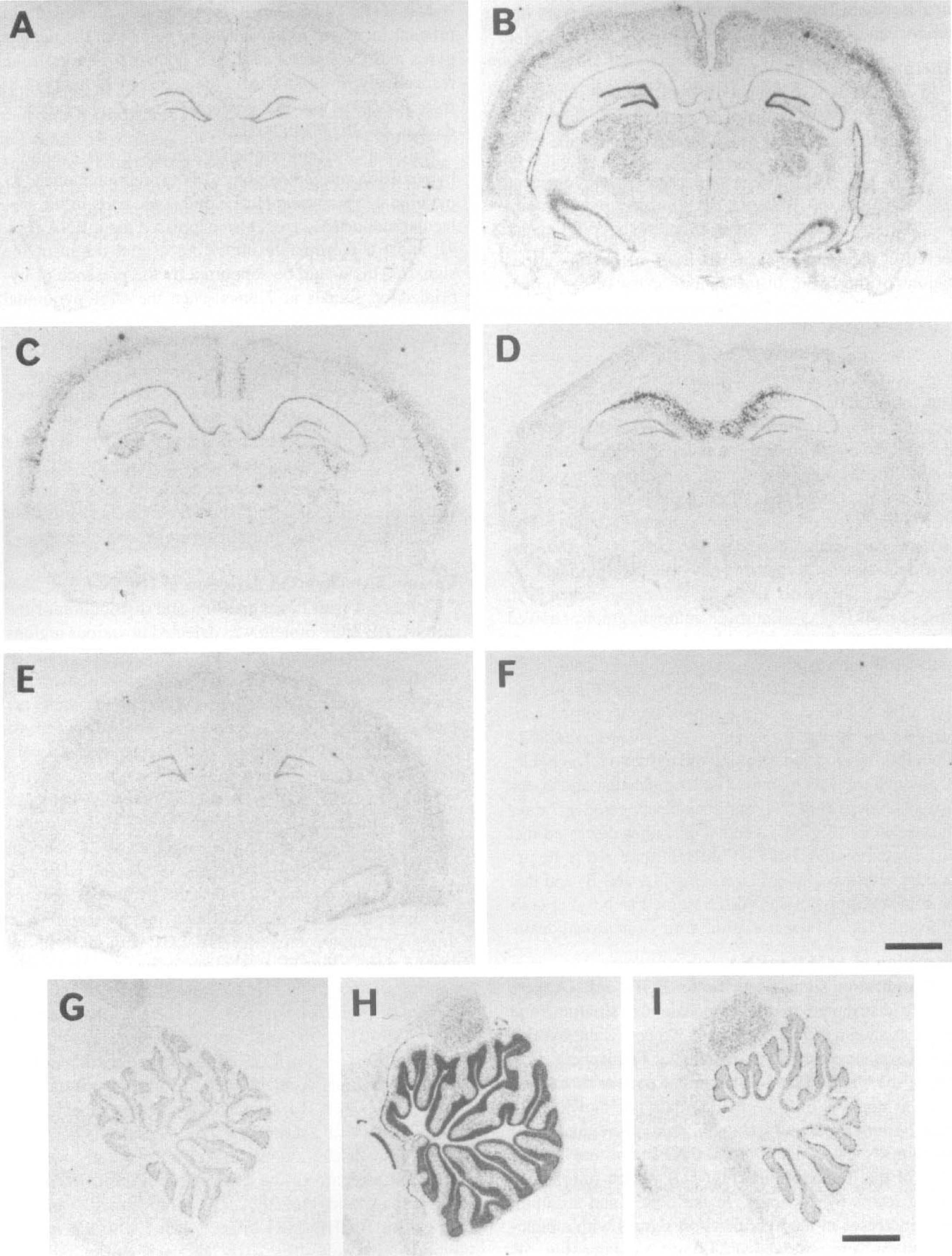
Autoradiographs of HB-EGF mRNA expression following ischemia.
Following ischemia, remarkable changes were observed in the expression of HB-EGF mRNA in various regions of the brain. In the dentate gyrus of the hippocampus and the granule cell layer of the cerebellum, HB-EGF mRNA expression was dramatically increased at 2 hours and then immediately returned to basal level (Fig. 2B and H). Quantitative analysis revealed significant three- and fivefold increases in these regions, respectively (Fig. 3). The cells expressing HB-EGF mRNA appeared to be neurons by emulsion studies (data not shown). In the outer cortex, thalamus, lateral geniculate body, and the CA3 sector of the hippocampus, HB-EGF mRNA expression was rapidly increased at 2 hours (Fig. 2B) and then gradually returned to basal level. Densitometric analysis demonstrated statistically significant increases, at least up to 12 hours, following ischemia in these regions (Fig. 3). Emulsion autoradiography showed that most cells bearing hybridization grains had a moderate-sized nucleus, probably suggesting neuronal localization of HB-EGF mRNA in these regions (Fig. 4C and D). In the CA1 sector of the hippocampus, on the contrary, increased signal intensity was first observed at 24 hours following ischemia along the pyramidal layer (Fig. 2C), which robustly extended to the molecular and oriens layers at 3 days (Fig. 2D) and then disappeared at 7 days after ischemia (Fig. 2E). Emulsion studies disclosed that the cells expressing HB-EGF mRNA appeared to be pyramidal neurons up to 24 hours (Fig. 4A and B) and that the robust expression at 3 days seemed to be glial cells infiltrating the CA1 sector at the time of neuronal death.
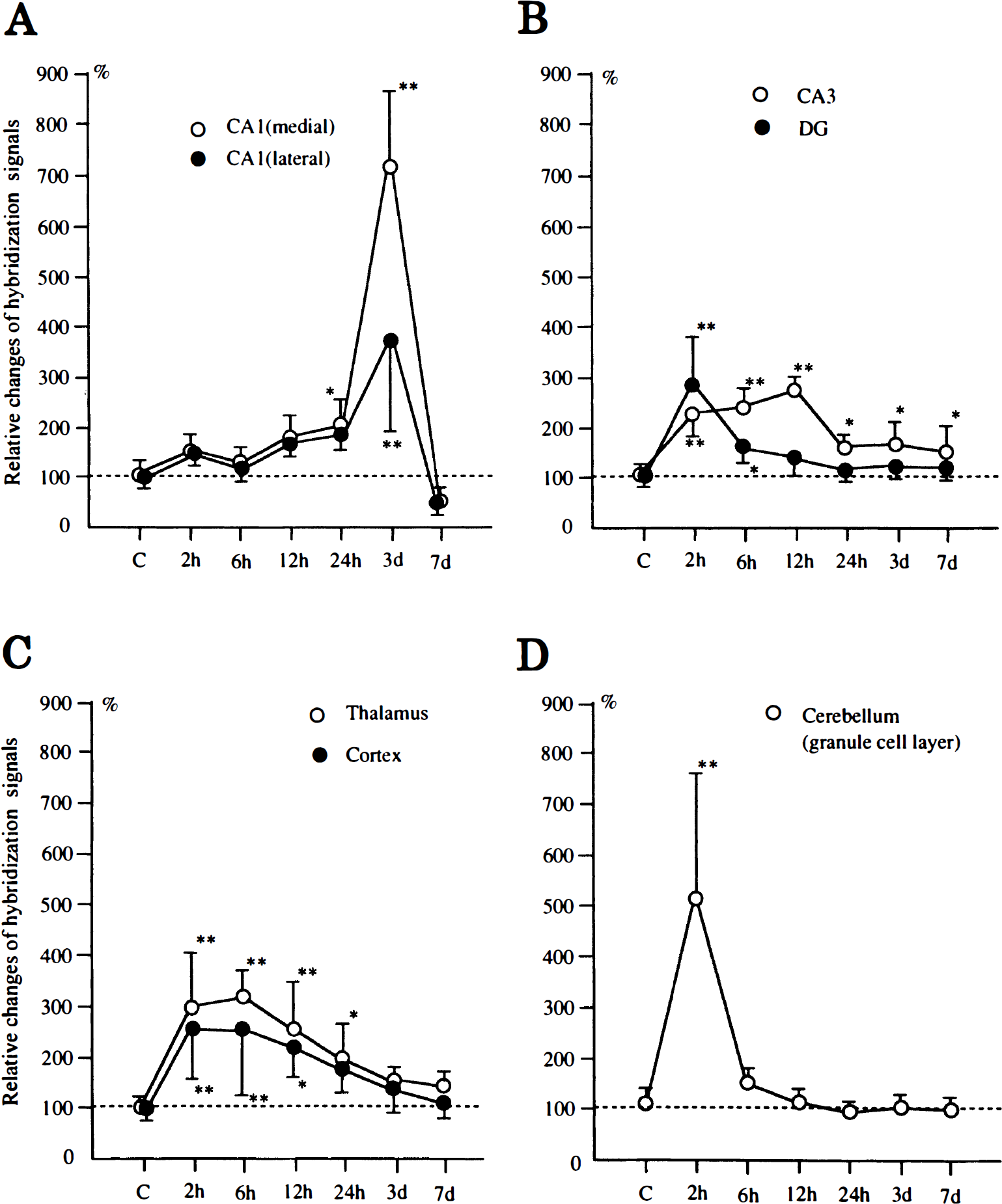
Relative change of HB-EGF mRNA expression in the hippocampus
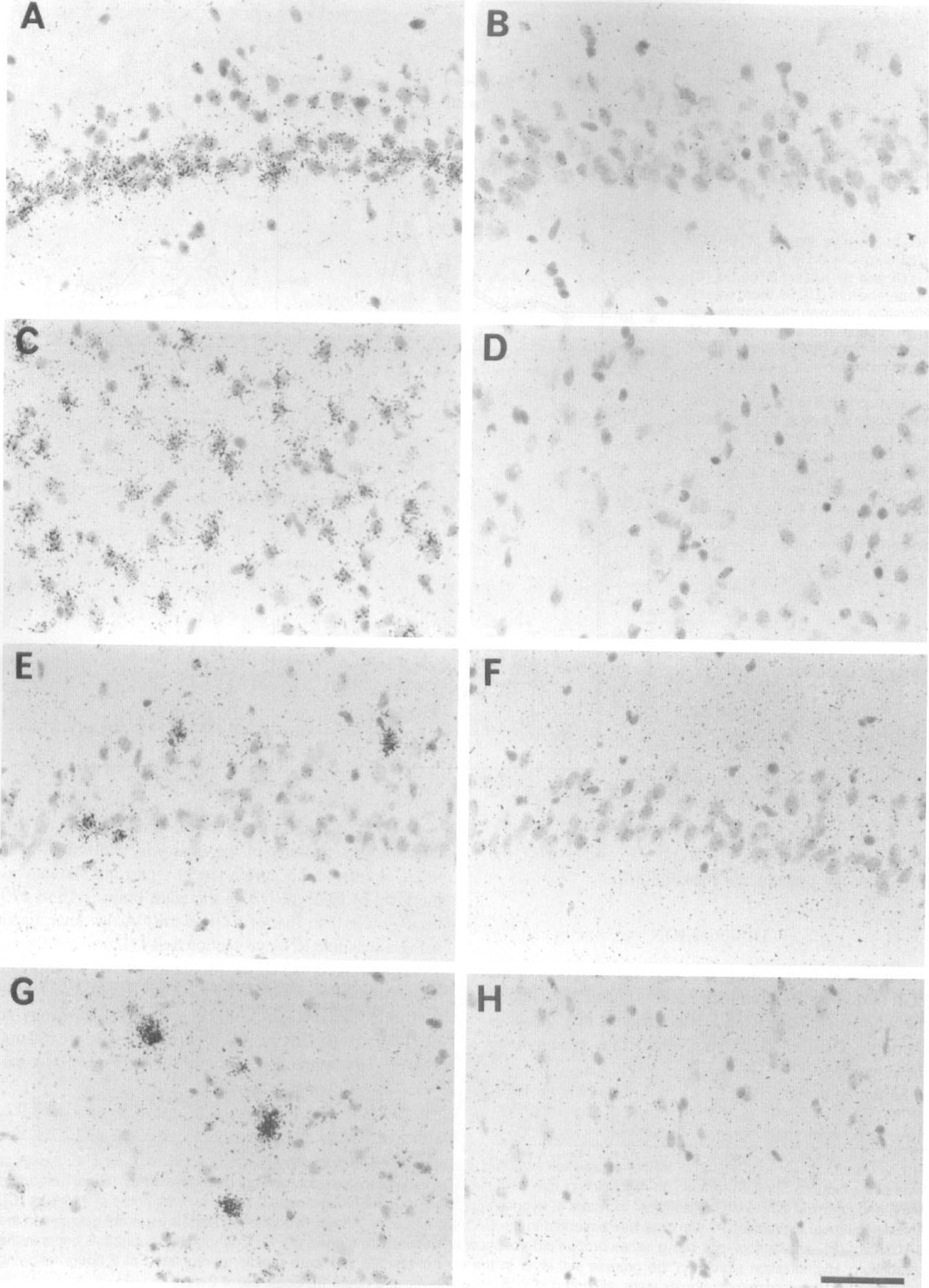
Emulsion autoradiographs for HB-EGF
Alteration of EGFR mRNA
Hybridization signals specific for EGFR mRNA were broadly distributed in the hippocampus, striatum, and cortex as shown in Fig. 5A and C. No consistent hybridization was observed in the cerebellar hemisphere. Following the sham operation, a similar pattern of expression was noted at all time points (data not shown).
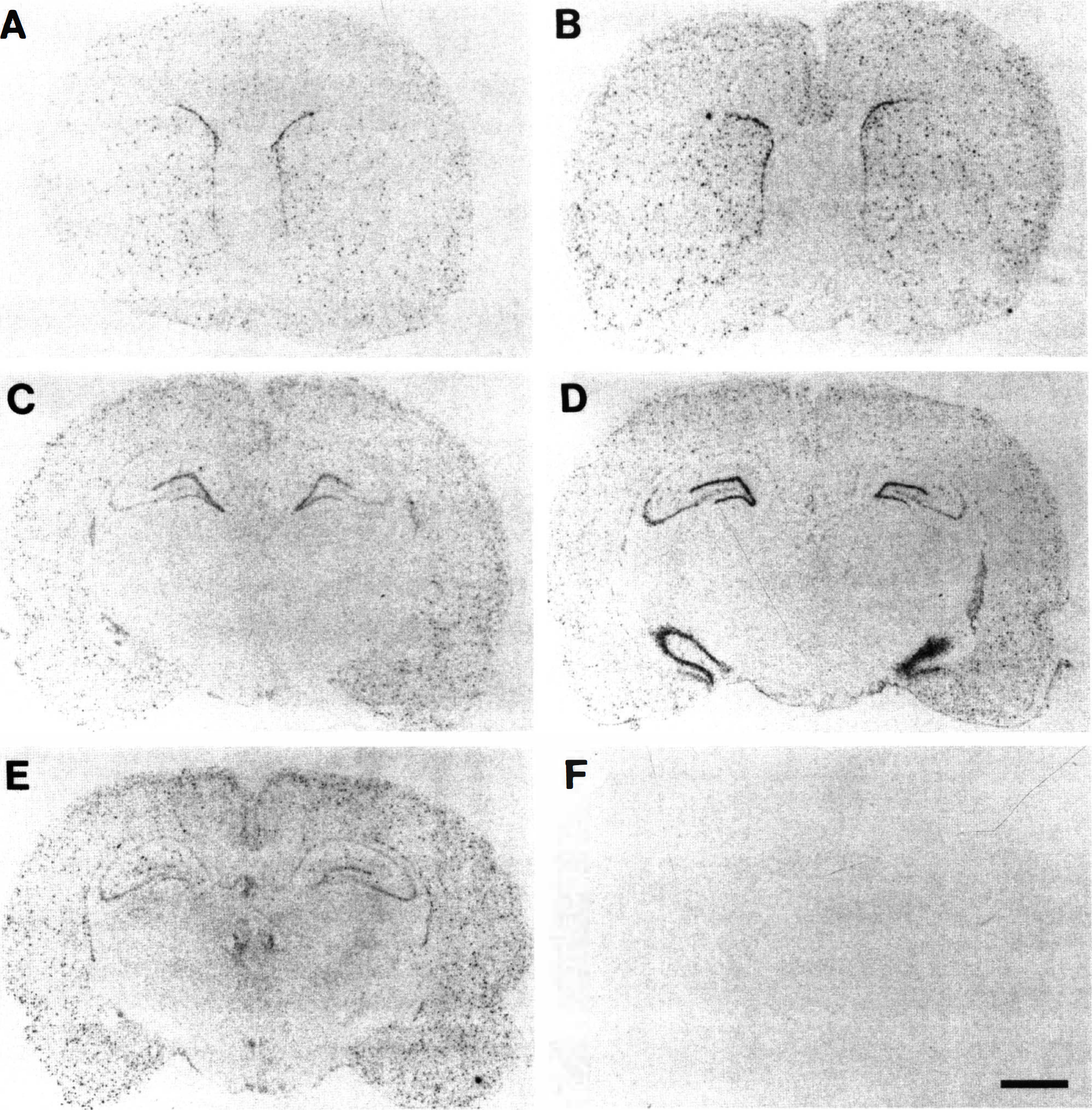
Autoradiographs of EGFR mRNA expression following ischemia.
At 2 hours following ischemia, rapid up-regulation of EGFR mRNA was noted in the CA3 sector and dentate gyrus of the hippocampus (Fig. 5D), which returned to normal levels by 24 hours. In the cortex and striatum, slight increases in the hybridization signals with a punctate pattern were observed at 24 hours and later (Fig. 5B and E). In the CA1 sector of the hippocampus, a minimal increase in hybridization was noted at 7 days following ischemia (Fig. 4 E). Densitometric analysis revealed significant increases in hybridization signals in the dentate gyrus and CA3 sector at 2 to 6 hours (2- and 2.5-fold, respectively; P < 0.0001), in the cortex at 24 hours (1.8-fold; P < 0.01), and in the lateral part of CA1 sector at 7 days (1.6-fold; P < 0.001).
Cellular localization of EGFR mRNA was examined by emulsion autoradiography. The expression in the CA1 pyramidal layer was punctate and some cells in the molecular and oriens layers also expressed the mRNA (Fig. 4E and F), presumably indicating a glial origin of the signals. This would be supported by the presence of hybridization signals at 7 days when the CA1 pyramidal neurons had died. Hybridization grains in the dentate gyrus and CA3 sector of the hippocampus, however, appeared to be located in the neuronal layer. In the cortex, intense hybridization signals were localized in scattered cells without any noticeable layer pattern in either the control or the postischemic brain, and those cells with a relatively large nucleus were devoid of the signals (Fig. 4G and H). Furthermore, some scattered cells in layer I also expressed EGFR mRNA. These findings probably suggest glial expression in the cortex.
Immunohistochemical detection of HB-EGF
Consistent with the expression and distribution of the mRNA, HB-EGF protein was detected in various regions of the adult brain (Figs. 6 and 7). The pattern of HB-EGF immunostaining and its changes following ischemia in the CA1 sector and dentate gyrus of the hippocampus are shown in Fig. 6. In the CA1 sector, pyramidal neurons, particularly their nuclei and perinuclear zones, were moderately immunostained (Fig. 6A) compared with the negative control (Fig. 6H). Scattered glial cells were also immunostained. Following ischemia, this pattern of expression did not show major changes up to 24 hours in the CA1 sector (Fig. 6B and C). At 3 days after ischemia, neuronal staining was lost when the pyramidal neurons had died, and glial cells migrating in this sector were strongly immunopositive for HB-EGF (Fig. 6D). In the dentate gyrus (Fig. 6E to G), the intensity of immunostaining of the granule cells seemed to be slightly increased at 6 and 24 hours after ischemia. In the cortex (Fig. 7A to D), cells with various-sized nuclei were immunopositive for HB-EGF throughout the cortex. Following ischemia, a slight increase of immunostaining was noted in layer II at 6 hours (Fig. 7B), which extended up to layer IV at 3 days (Fig. 7D). These cells had relatively large nuclei, suggesting an increase in neuronal cells. With regard to the expression in the cerebellum, the granule and Purkinje cells were strongly immunopositive for HB-EGF in accordance with the in situ hybridization study (Fig. 7E). However, no remarkable changes of immunostaining were noted in the postischemic brain (Fig. 7F to H) despite an increase of the mRNA in the granule and Purkinje cells.
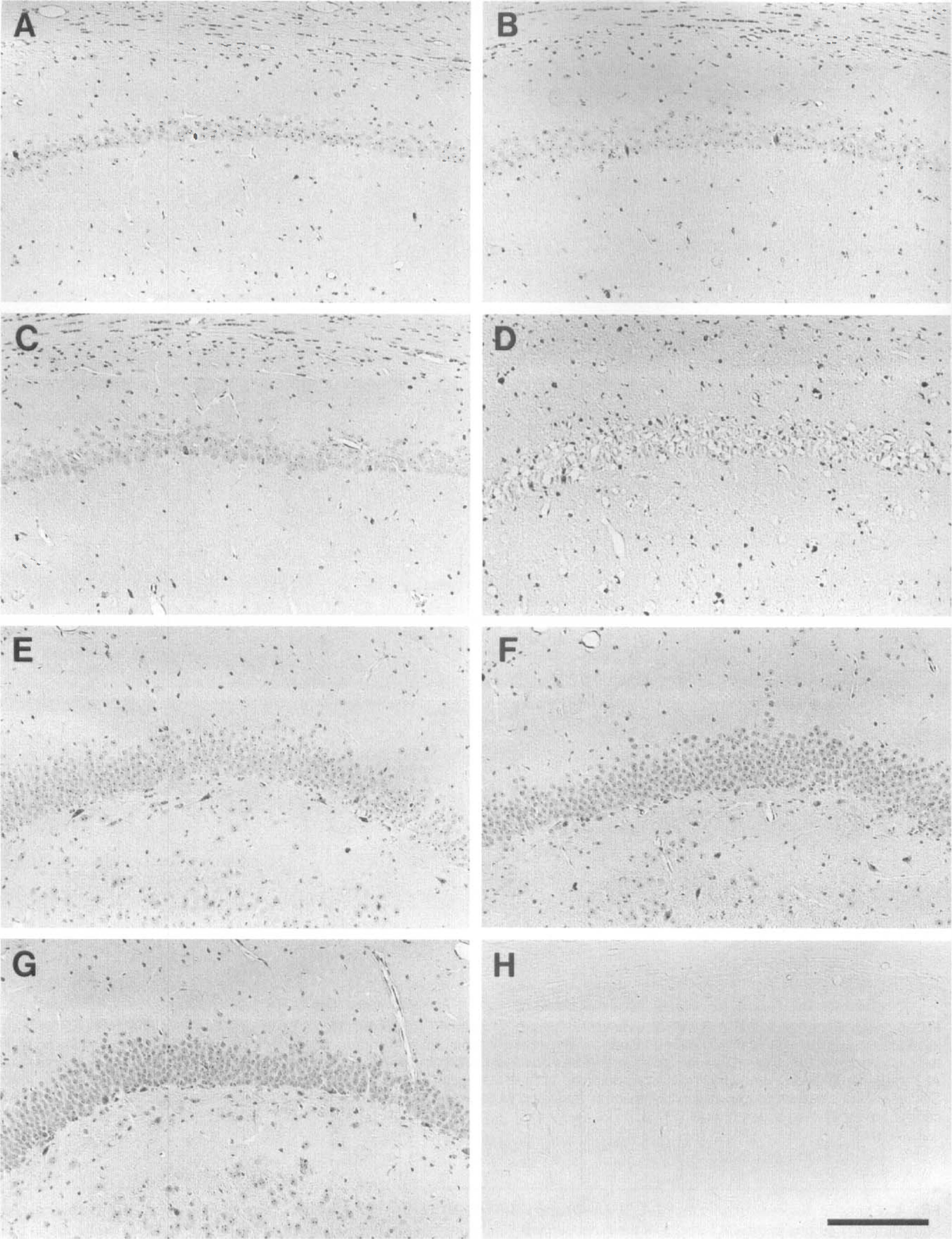
Immunohistochemical detection of HB-EGF in the hippocampal CA1 sector (A-D) and dentate gyrus (E-G).
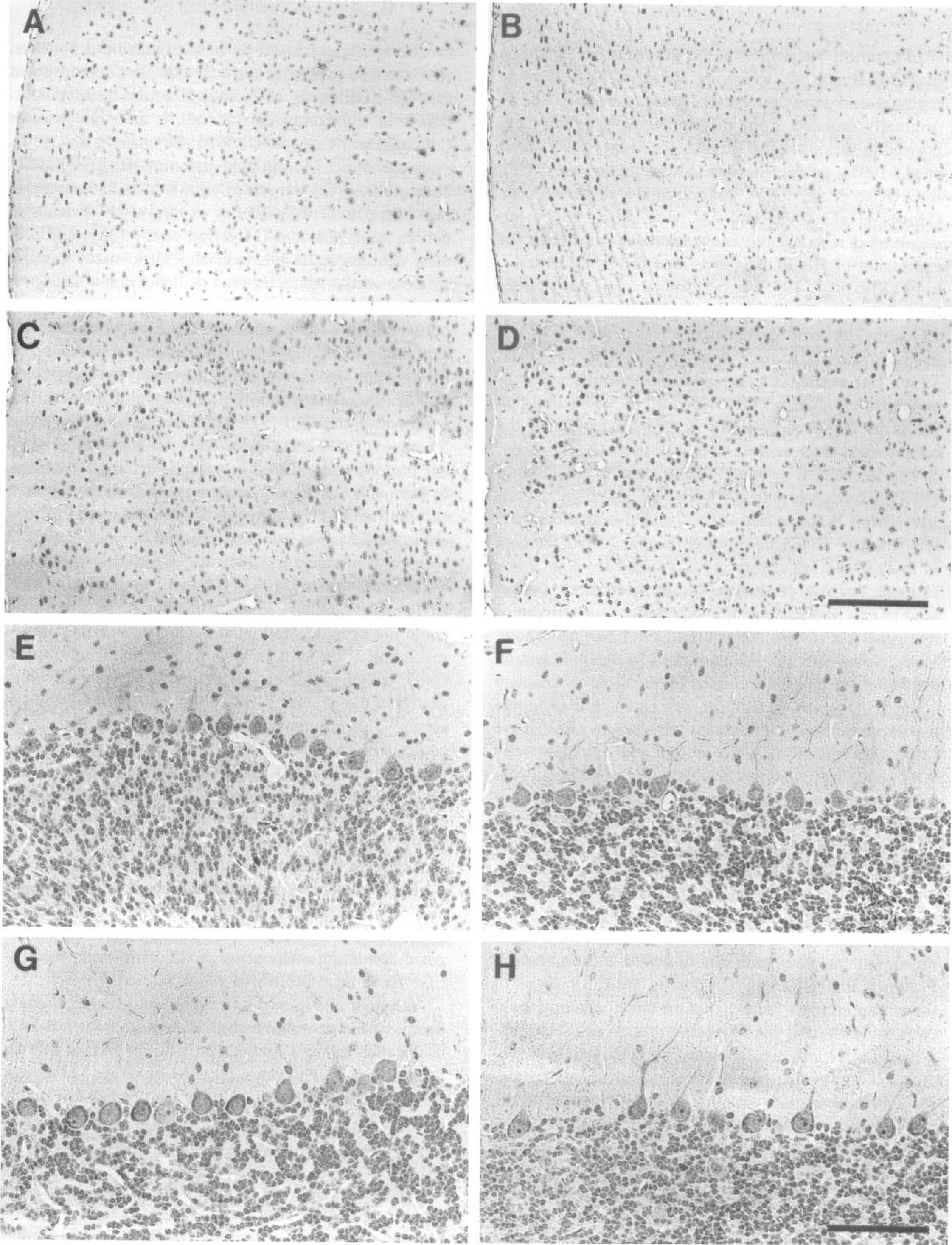
Immunohistochemical detection of HB-EGF in the cortex (A-D) and cerebellum (E-H).
DISCUSSION
The results of this study revealed that members of the EGF family are differentially expressed and regulated in the adult rodent brain and demonstrated that the gene encoding HB-EGF is a stress-inducible gene among these members.
Failure to detect EGF mRNA in this study is probably due to low sensitivity of the in situ hybridization technique employed, because a recent report has shown that the level of EGF mRNA in the adult brain is 15 to 170 times lower than that of TGF-α mRNA, as determined using a sensitive RNA protection assay (Lazar and Blum, 1992). In regard to TGF-α, we demonstrated widespread expression of its mRNA in the adult brain, consistent with previous reports (Kudlow et al., 1989; Seroogy et al., 1991, 1993; Kaser et al., 1992). However, we did not observe any appreciable increase in TGF-α mRNA expression following ischemia.
In contrast, we demonstrated dramatic up-regulation of HB-EGF mRNA following ischemia, with discrete regional and temporal pattern of changes. HB-EGF, originally isolated from macrophages, is widely expressed in various tissues such as the kidney (Nakagawa et al., 1997), gastric mucosa (Murayama et al., 1995), liver (Kiso et al., 1995), and vascular endothelial cells (Morita et al., 1993) and has been shown to be up-regulated in response to various insults. For instance, the expression of HB-EGF is increased following hepatectomy (Kiso et al., 1995), hyperosmolar and shear stresses to endothelial cells (Morita et al., 1993; Asakawa et al., 1996), and ischemic or oxidative stress in the kidney and gastric mucosa (Homma et al., 1995; Miyazaki et al., 1996). Our results first demonstrated that cerebral ischemia also up-regulates HB-EGF mRNA, indicating that the gene encoding HB-EGF is stress-inducible in the adult rodent brain as well.
The mechanism by which stresses lead to induction of HB-EGF mRNA is not known. One stress-inducible gene that is well characterized is the 70-kDa heat shock protein (hsp70). This gene has a defined sequence element in its upstream regulatory regions called the heat shock element. In case of thermal stress, heat shock factors bind to heat shock element and promote transcription of this gene (for review see Schlesinger, 1990; Morimoto, 1993; Nowak, 1993). Whether hsp70 mRNA induction following ischemia is mediated by the same mechanism is not yet resolved. In fact, the well characterized human hsp70 promoter region includes sequence motifs that are responsive individually to serum and calcium/cyclic AMP and heavy metals (Schlesinger, 1990; Nowak, 1993), which might contribute to its transcriptional activation following diverse environmental stresses. On the other hand, the murine HB-EGF promoter region lacks heat shock element but contains putative transcription factor binding sites for nuclear factor-κB, AP-1, Ets, E-box binding factors, Pit-1, and SP-1 (McCarthy et al., 1997), suggesting a different mode of transcriptional regulation compared with that of hsp70. Indeed, there is evidence that ischemia/reperfusion activates some of these transcription factors. For instance, cerebral ischemia induces AP-1 binding protein, such as c-Fos (Onodera et al., 1989), and activates mitogen-activated protein kinases, particularly p44 and p42 extracellular signal-regulated kinases (Kindy, 1993; Hu and Wieloch, 1994), which may lead to activation of Ets-2 (McCarthy et al., 1997). Likewise, nuclear factor-κB has been shown to be activated following cerebral ischemia (Clemens et al., 1997). How these changes in transcription factor activity are causally related to induction of HB-EGF in vivo following cerebral ischemia remains to be resolved.
As the expression of HB-EGF was only recently identified in the adult rodent brain (Mishima et al., 1996), its precise biological functions in the brain have not been characterized to date. In vitro studies have demonstrated that HB-EGF exerts its biological action by binding to EGFR in a paracrine, juxtacrine, or autocrine manner (Higashiyama et al., 1991, 1995; Freeman et al., 1997). Although direct neurotrophic action of HB-EGF has not been reported yet, several in vitro studies have revealed that stimulation of EGFR by exogenous EGF leads to neurotrophic activities (Morrison et al., 1987) and protects neurons against anoxia and nitric oxide toxicity (Maiese et al., 1993). In vivo evidence for a neuroprotective role of EGF against ischemia has also been reported recently (Peng et al., 1997). In view of the wide expression of EGFR in the adult brain, as evidenced by observation in this study and other investigations (Gómez-Pinnila et al., 1988; Seroogy et al., 1995), it is conceivable that HB-EGF functions as an endogenous ligand for EGFR and contributes to neuronal survival and protection against various insults in the mature brain. In line with this hypothesis, we demonstrated that both HB-EGF and EGFR mRNAs were up-regulated following ischemia in the ischemia-resistant hippocampal CA3 sector and dentate gyrus, whereas in the most vulnerable CA1 sector (Kirino, 1982; Pulsinelli et al., 1982), HB-EGF mRNA failed to be up-regulated. However, we could not provide substantial evidence for HB-EGF up-regulation at the protein level by immunohistochemical study, although increased immunoreactivity was suggested in the dentate gyrus of the hippocampus and the cortex. This discrepancy presumably could have been caused by translational inhibition in the postischemic brain (for review see Krause and Tiffany, 1993). Another limitation of our study is that H1 antibody recognizes only the intracellular domain of HB-EGF and does not reflect the dynamic changes of the secreted active form. The changes of HB-EGF at the protein level should be further evaluated by more precise quantitative methods, and future studies employing strategies such as exogenous administration of recombinant HB-EGF or targeted disruption of its gene would clarify whether HB-EGF has a neuroprotective action in vivo.
Although the present immunohistochemical study demonstrated neuronal and glial expression of HB-EGF in the hippocampus and cerebellum, its precise cellular localization in the cortex remains unknown. Likewise, we could not identify the cellular source of EGFR in this study. Future study using co-labeling methods would clarify these issues. Nevertheless, the expression of EGFR was limited compared with that of HB-EGF and seemed to be localized in glial cells, particularly in the hippocampal CA1 sector, thus raising doubts about the direct neurotrophic actions of HB-EGF in this region. Another important finding is that HB-EGF was robustly up-regulated in glia in the CA1 sector at 3 days following ischemia. Although we speculate that glial HB-EGF may serve as a stimulating and proliferative signal for microglia and astrocytes after injury in view of its potential role as a mitogen (Higashiyama et al., 1991), glial cells expressing HB-EGF in the CA1 sector at 3 days after ischemia did not express EGFR, thus again suggesting a discrepancy between HB-EGF and EGFR expressions. There is evidence, however, that HB-EGF also binds to other members of the Erb-B receptor family, including EGFR (also known as Erb-B1) (Beerli and Hynes, 1996). Furthermore, a recent study (Francoeur et al., 1995) has revealed that Erb-B2, -B3, and -B4 are expressed in the chick brain and suggested that Erb-B2 and -B4, in particular, are expressed in neurons. Thus, the precise biological actions of HB-EGF in various regions of the brain under physiological and pathological conditions need to be further evaluated by studies on regional and cellular expression of these receptors.
In summary, we have demonstrated that the gene encoding HB-EGF is a stress-inducible gene in the adult rodent brain and have suggested a role for this factor in cerebral ischemia. However, we could not conclude from our study whether HB-EGF has any neuroprotective role in cerebral ischemia. Nonetheless, we believe that these data offer promise for HB-EGF as a neurotrophic and neuroprotective factor, warranting further studies on its role in cerebral ischemia.
Footnotes
Acknowledgements
The authors thank Dr. H. Shelton Earp (University of North Carolina at Chapel Hill, NC, U.S.A.) for kindly providing us with rat EGFR cDNA and Ms. Reiko Matsuura for technical assistance in in situ hybridization.
