Abstract
Cerebral stroke induces proliferation of subventricular zone (SVZ) neural progenitor cells in adult rodent brain. Tumor necrosis factor-α-converting enzyme (TACE) proteolysis sheds the nonamyloidogenic soluble ectodomain of the amyloid precursor protein (APP) and is a convertase for tumor necrosis factor-α (TNFα). The resulting soluble peptides of APP and TNFα are mitogenic for neural progenitor cells of the SVZ. Therefore, we hypothesized a role for TACE proteolysis in stroke-induced neurogenesis. Using laser-capture microdissection, we found TACE transcription was increased in SVZ cells of ischemic brain. Immunohistochemistry revealed TACE protein was upregulated in SVZ neuroblasts. Intraventricular infusion of tumor necrosis factor-α protease inhibitor-2 (TAPI-2) decreased bromodeoxyuridine incorporation in SVZ cells of rats subjected to middle cerebral artery occlusion. Furthermore, primary culture SVZ neurospheres from ischemic brain overexpress TACE and its substrates APP and TNF-α. These cells proliferated more rapidly, possessed increased TACE protease-dependent α-secretase activity, and released more soluble APP and TNFα compared with nonischemic control. In addition, TAPI-2 reduced SVZ neuroblast migration out of SVZ explants in vitro. These findings indicate TACE proteolysis as a promoter of stroke-induced SVZ progenitor cell neurogenesis, and suggest this protease activity may represent an attractive therapeutic target for stroke recovery.
Keywords
Introduction
Stroke induces proliferation of subventricular zone (SVZ) neural progenitors of the adult brain, and treatments that increase SVZ neurogenesis are correlated with enhanced functional recovery (Arvidsson et al, 2002; Zhang et al, 2001, 2005). Tumor necrosis factor-α-converting enzyme/a disintegrin and metalloproteinase-17 (TACE/ADAM17) proteolysis releases many soluble SVZ cell-binding proteins, including soluble amyloid precursor protein-α (sAPPα), soluble tumor necrosis factor-α (sTNFα), and several epidermal growth factor receptor (EGFR/ErbB1)-activating proligands (Blobel, 2005; Romera et al, 2004; Sahin et al, 2004).
TACE proteolysis cleaves APP within the amyloid beta (Aβ) peptide sequence (Wang et al, 2004). This α-cleavage precludes Aβ formation and alternately produces the sAPPα peptide (Wang et al, 2004). The sAPPα ectodomain contains the 17-mer sequence APP328–332, which enhances neurite extension, increases learning and memory in rats, and promotes neuron survival (Jin et al, 1994; Roch et al, 1994; Yamamoto et al, 1994). Soluble amyloid precursor protein-α is present in normal brain and circulates in the cerebrospinal fluid (Palmert et al, 1989). Adult SVZ astrocytes express APP, and binding sites for sAPPα are found on neuroblasts and epidermal growth factor-(EGF)-responsive progenitor cells of the SVZ (Caille et al, 2004; Yasuoka et al, 2004). Infusion of sAPPα promotes SVZ progenitor cell proliferation, and ectopic EGF promotes release of sAPPα from SVZ neurospheres in vitro (Caille et al, 2004).
Like sAPPα, the ectodomain of TNFα is shed by TACE proteolysis, and sTNFα is found in cerebrospinal fluid of normal brain as well (Zaremba and Losy, 2001). Injection of TNFα into brain increases bromodeoxyuridine (BrdU) incorporation in SVZ cells, and intraventricular infusion of an antibody to TNF-α into rats after stroke impairs survival of SVZ neuroblasts (Heldmann et al, 2005; Wu et al, 2000). Additionally, TACE protease activity confers glutamate-transport-mediated ischemic tolerance on primary mixed cortical cells via TNFα release (Romera et al, 2004).
In addition to cleavage of TNFα and APP, TACE proteolysis sheds the ectodomains of several EGFR proligands including transforming growth factor-α, amphiregulin, epiregulin, heparin-binding epidermal growth factor (HB-EGF), betacellulin, EGF, and ErbB4 (Ghashghaei et al, 2006; Sahin et al, 2004). Although much interest has been generated concerning a role for TACE protease activity in EGF-responsive tumors, a similar role in the maintenance of EGF-responsive SVZ progenitor cells has yet to be described.
As multiple TACE-activated ligands are mitogenic for adult SVZ progenitor cells, we surmised TACE proteolysis might be a positive regulator of SVZ neurogenesis. Here, we show TACE protein expression increases in the SVZ ipsilateral to infarct after cerebral ischemic stroke. Intraventricular infusion of the TACE proteolytic inhibitor N-(R)-[2-(hydroxy-aminocarbonyl) methyl]-4-methylpentanoyl-
Subventricular zone cells express several substrates for TACE proteolysis in addition to APP and TNFα (Doetsch et al, 2002; Ghashghaei et al, 2006; Stump et al, 2002). In fact, several TACE-cleaved EGFR proligands represent attractive candidates for TACE-mediated SVZ neurogenesis as well (Doetsch et al, 2002; Jin et al, 2004; Stump et al, 2002). Additional studies are necessary to elucidate the contributions of possible SVZ cell-binding TACE substrates. However, in addition to revealing a role for TACE proteolysis in stroke-induced SVZ proliferation, our data suggest TACE protease-dependent production of subventricular sAPPα may represent a novel nonpathogenic function for APP within the adult brain. Taken together, our findings indicate TACE proteolysis promotes stroke-induced SVZ neurogenesis and thus, may contribute to stroke recovery.
Materials and methods
Reagents
To inhibit proteolytic activity of TACE, the TACE/ADAM17 inhibitor TAPI-2 (Peptides International, KY, USA) was employed. To inhibit β-secretase activity, the β-secretase inhibitor (AG Scientific, CA, USA) product #S1118 (S1118) was used. To inhibit matrix metalloproteinasess, tissue inhibitor of metalloproteinase 1 (TIMP-1, R&D Systems, MN, USA) was employed. Preliminary experiments lead us to select a concentration of 10 μmol/L for TAPI-2 and S1118 and 50 ng/mL TIMP-1, for all in vitro experiments.
Middle Cerebral Artery Occlusion Model
Adult male Wistar rats weighing 270 to 300 g were employed in all experiments. Permanent middle cerebral artery occlusion was induced by a method of intraluminal vascular occlusion as previously described (Zhang et al, 2001). Briefly, rats were anesthetized with 3.5% halothane and then maintained with 1% to 2% halothane in N2O and 30% O2 using a face mask. A length of 4-0 monofilament nylon suture (18.5 to 21.0 mm), determined by animal weight, was advanced from the external carotid artery into the lumen of the internal carotid artery until it blocked the origin of the middle cerebral artery.
Subventricular Zone Neurosphere Culture
Subventricular zone cells were dissociated from male adult Wistar rat (270 to 300 g) SVZ tissue and cultured, as previously reported (Katakowski et al, 2005). The generated neurospheres were passed by mechanical dissociation and reseeded as single cells at a density of 20 cells per microliter. Subventricular zone cells used from normal brain in experiments were from passages 2 to 5. Subventricular zone cells used from ischemic brain were extracted from rats 7 days post-MCAo, from the SVZ ipsilateral to infarct, and were employed between passages 2 to 4. For all experiments, SVZ cells were used 7 to 10 days after the preceding passage.
Immunohistochemistry
Coronal paraffin sections (8 μm) were obtained from normal rats, or those killed 1, 7, 14, and 30 days post-MCAo (n = 4 per group) or rats killed 8 days post-MCAo (n = 9) for BrdU labeling experiments. These were immunostained as previously described (Zhang et al, 2001). The following antibodies: mouse anti-β-tubulin-III (Covance, NJ, USA) as a marker for neuroblasts, rabbit anti-GFAP (glial fibrillary acidic protein) (Dako, CA, USA) as a marker for astrocytes, rabbit anti-APP (Cell Signaling Technologies, MA, USA), rabbit anti-TACE, rabbit anti-BACE1 (beta-site amyloid precursor protein-cleaving enzyme-1), and mouse anti-β-actin (Abcam, MA, USA), goat anti-TACE (Santa Cruz, CA, USA), and mouse-anti-BrdU (Roche Diagnostics, IN, USA) were employed. For BrdU staining, DNA was first denatured by incubating sections in 50% formamide 2 × SSC at 65° for 2 h and then 2 N HCl at 37° for 30 mins. Specimens were analyzed using fluorescent or phase contrast microscopy (Nikon) with Metamorph Imaging software.
Laser Capture Microdissection
Frozen tissue slides from normal rat brains or those killed 7 days post-MCAo (n = 3 each) were dehydrated in ethanol solutions (75%, 95%, 100%, each once for 30 secs) and cleared in xylene for 5 mins. After air-drying for 10 mins, SVZ cells were captured onto a thermoplastic film mounted on optically transparent LCM caps using the PixCell II LCM System (Arcturus Engineering, CA, USA). The following parameters were used during LCM: 7.5 μm laser spot size, 60 mW power, 750 ms duration. The transfer film was examined under the microscope to ensure cell adherence. Caps with cells were immediately placed into Eppendorf tubes containing 350 μL of lysis buffer and stored in −80°C before RNA isolation. Approximately 2000 cells were isolated from the SVZ of each rat.
Real-Time Reverse Transcriptase-Polymerase Chain Reaction
Quantitative polymerase chain reaction (PCR) was performed using SYBR Green real-time PCR system. Total RNA was extracted using an Absolutely RNA Miniprep kit (Stratagene, CA, USA). cDNA was prepared from total RNA using oligo(dT), dNTP mix, First-Strand Buffer, DTT, RNaseOUT, and Superscipt III (Invitrogen, CA, USA). Real-time reverse transcriptase (RT)-PCR was performed on an ABI 7000 PCR instrument (Applied Biosystems, CA, USA). Amplicon sizes were confirmed using RT-PCR as previously described (Katakowski et al, 2005). Primers employed were:β-actin: 5′-CCA TCA TGA Agt Gtg Acg Ttg-3′ (fwd), 5′-CAA TGA TCT TGA TCT TCA TGG TG-3′ (rev). APP: 5′-TGC AGC AGA ACG GAT ATG AG-3′ (fwd), 5′-ACA CCG ATG GGT AGT GAA GC-3′ (rev). TACE: 5′-ACT CTG AGG ACA GTT AAC CAA ACC-3′ (fwd), 5′-AGT AAA AGG AGC CAA TAC CAC AAG-3′ (rev). ADAM10: 5′-ACA GAC TTG GCT CTC GAT AAA CTT-3′ (fwd), 5′-GGT ATG TAC ATT GGC AAG TGA TGT-3′ (rev). BACE1; 5′-ATA CAC TGG CAG TCT CTG GTA CAC-3′ (fwd), 5′-CAC GAT GCT CTT GTC ATA GTT GTA-3′ (rev). β-tubulin-III: 5′-CTC CCA GGT TAA AGT CCT TCA GTA-3′ (fwd), 5′-GCA ACA TAA ATA CAG AGG TGG CTA-3′ (rev). GFAP: 5′-ACC ATT CCT GTA CAG ACT TTC TCC-3′ (fwd), 5′-AGT CTT TAC CAC GAT GTT CCT CTT-3′ (rev). Each sample was tested in triplicate and relative gene expression was determined using the 2−ΔΔCT method.
Western Blot
Western blotting was used to detect TACE, BACE1 (Abcam, MA, USA), and APPα (Cell Signaling, MA, USA). SuperSignal West Pico Chemiluminescent Substrate (Pierce, IL, USA) and Kodak X-omat film (Kodak, NY, USA) exposure were used for visualization. Concentration of soluble proteins in culture media was achieved using a Millipore centrifugal filter tube (Millipore, MA, USA). Protein concentrations were quantified using a BCA protein assay kit (Pierce, IL, USA). To analyze Western blot data, densitometric measurements of three or more blots per group were averaged and compared using Student's t-test.
Secretase Activity Enzyme-Linked Immunosorbent Assay
To measure α- and β-secretase enzymatic activity, 2 × 105 cultured SVZ cells derived from normal or ischemic rats were lysed and processed with either an α or β-secretase Activity Kit (R&D Systems, MN, USA) as per the manufacturer's instructions. Fluorescence was read using a Fusion Universal Multiplate reader (Packard Bioscience, CT, USA).
In Vivo Bromodeoxyuridine Incorporation
At 24 h after MCAo, an Alzet micro-osmotic pump was implanted into each rat. A measure of 10 mmol/L TAPI-2 or saline vehicle was delivered at rate of 1 μL/h into the lateral ventricle ipsilateral to ischemic injury for 7 days. After implant, rats were with administered doses of BrdU daily (100 mg/kg i.p.). Rats were killed 8 days post-MCAo, 3 h after final BrdU injection. Brains were fixed in paraformaldehyde, and embedded in paraffin. For statistical analysis, DAB+ cells were counted in three fields of view from three coronal sections between anterior posterior (AP) + 10.6 mm, AP 9.6 mm, and AP +8.74 mm (Paxinos and Watson, 1986), and the average BrdU positive cells/field was determined for each animal. Mean cell/field numbers for treatment (n = 5 rats) and saline control (n = 4 rats) group were averaged and compared using Student's t-test.
In Vitro Bromodeoxyuridine Incorporation Assay
Subventricular zone neurospheres derived from normal or ischemic rats were dissociated and 4000 cells were added to each well of a flat bottom 96-well plate (n = 4 wells per group). At 4 h after seeding the cells, TAPI-2 or S1118 were added at 10 μmol/L. After 24 h, BrdU at 10 μmol/L was added to each well and cells were cultured for an additional 14 h. BrdU incorporation was then quantified using the BrdU Labeling and Detection Kit III (Roche, Germany) as directed.
Subventricular Zone Explant Culture
Male Wistar rats (270 to 300 g), normal and 7 days post-MCAo were killed and SVZ tissue (from the ipsilateral hemisphere of ischemic rats) was quickly excised under sterile conditions. The SVZ was dissected from a frontal slice extending between the crossing of the anterior commissure and the rostral opening of the third ventricle. Subventricular zone tissue was cut into ~0.5 mm3 blocks and embedded in 40 μL of Matrigel in a 24-well culture plate. After 10 mins, 0.6 ml of Neurobasal-A medium containing 2% B27 was added to each well. Radial migration of cells out of the SVZ explants was measured for 7 days. Ara-c was added to cultures at 1 μmol/L. Cell migration was quantified by calculating the average distance between the explant perimeter and the perimeter formed by the leading edge of migrating cells. Experimental groups consisted of at least six explants.
Statistical Analysis
Student's two-tailed independent t-test was employed to compare the means of BrdU reactive cell numbers in vivo (degrees of freedom (d.f.) = 7). P-value <0.05 was considered statistically significant. Repeated measure analysis of variance was used to reveal the effect of stroke and TAPI-2 or S1118 treatment on in vitro BrdU incorporation (d.f. = 6), secretase activity (d.f. = 4), and cell migration (d.f. ≥ 10). Statistical significance was set at P < 0.05.
Results
Tumor Necrosis Factor-α-Converting Enzyme mRNA Transcription is Upregulated in the Adult Subventricular Zone Following Cerebral Stroke
To measure transcriptional changes of TACE within the SVZ, we employed laser-capture tissue microdissection of frozen tissue followed by real-time RT-PCR. Quantification of mRNA extracted from SVZ cells revealed that gene expression of TACE was substantially upregulated by ischemia (Figure 1C). In addition to TACE mRNA, we found APP mRNA was also increased within the SVZ 7 days post-MCAo. This prompted us to measure transcription of the APP β-secretase, BACE1, and ADAM10, another primary α-secretase for APP in brain (Blobel, 2005). ADAM10 mRNA was several-fold higher than nonischemic SVZ control, whereas BACE1 transcription was highly upregulated 7 days post-MCAo in all samples measured (Figure 1C).
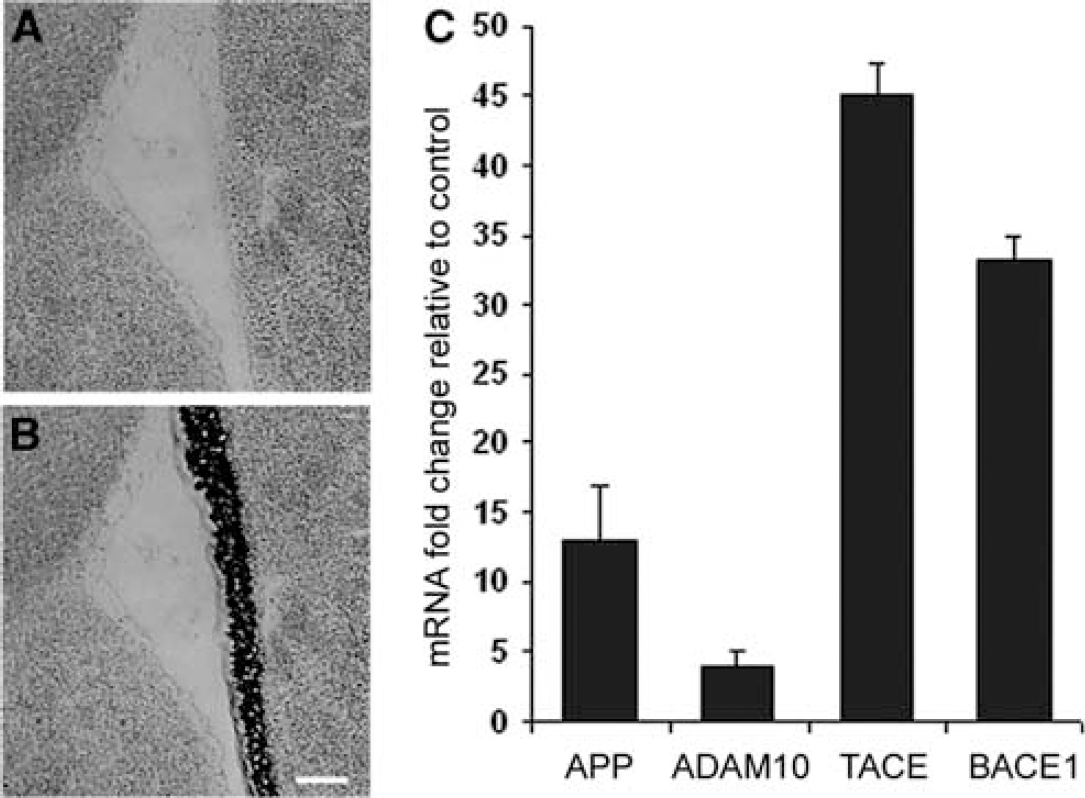
Real-time RT-PCR analysis of laser-capture microdissection SVZ tissue indicates TACE transcription is increased by MCAo. (
Tumor Necrosis Factor-α-Converting Enzyme Protein is Upregulated in the Adult Subventricular Zone After Cerebral Stroke
To determine if protein expression of TACE, a primary protease for TNF-α, APP, and several EGFR proligands was altered in the SVZ of ischemic brain, we immunostained paraffin-embedded tissue obtained from normal rat brain, and those obtained from rats 1, 7, 14, and 30 days post-MCAo. Immunohistochemistry revealed that MCAo caused an increase in subventricular TACE expression in a time course that was highest at 7 days post-MCAo among the time points we investigated (Figures 2A–2C). The stroke-induced expression of TACE was expressed on isolated cells or cell clusters, as well as in ependymal cells, or those in contact with the lateral ventricle (Figures 2B–2D). Notably, TACE expression was high in the walls of subventricular vasculature in both normal and ischemic brain. Double immunolabeling for TACE and immature neural marker β-tubulin-III or the astrocytic marker GFAP indicated that TACE was expressed by SVZ neuroblasts whereas little or no TACE expression was found in SVZ astrocytes (Figures 2E–2L). This upregulation of TACE was specific to the SVZ ipsilateral to infarct as expression within the contralateral SVZ remained near basal levels at all time points (not shown).
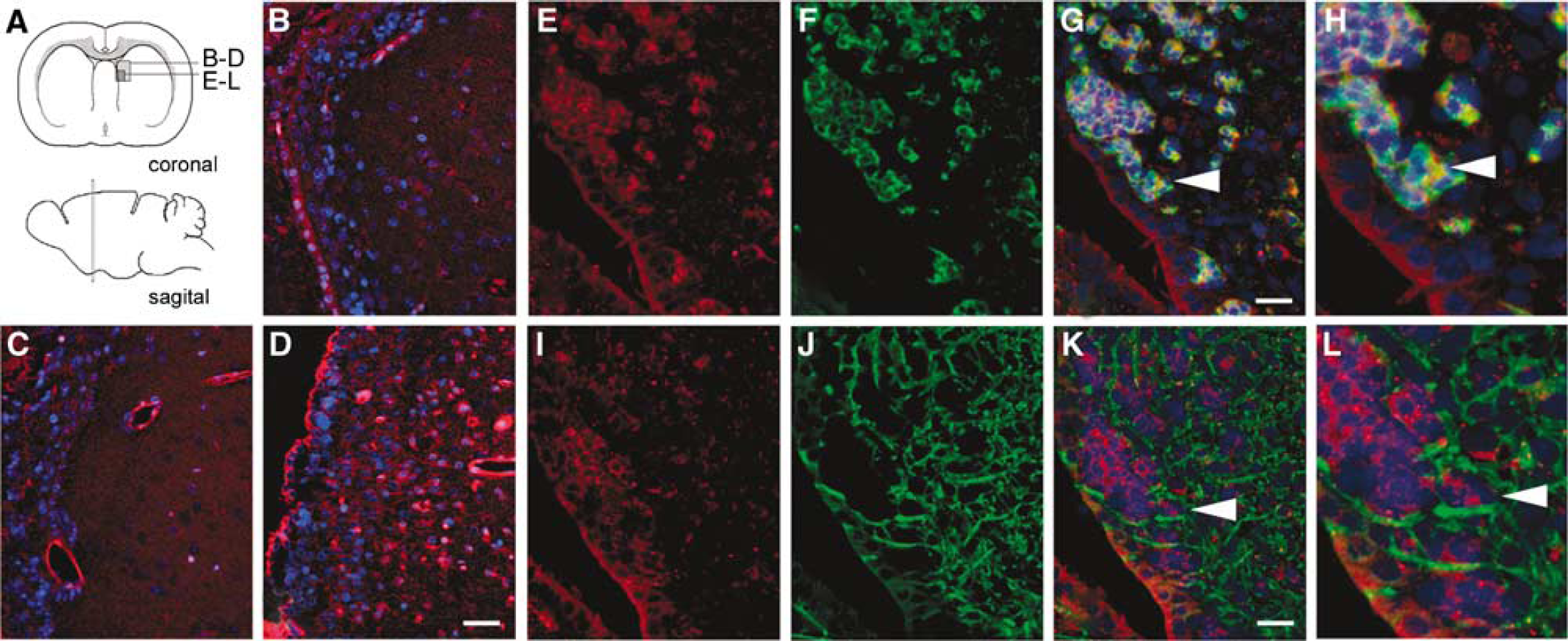
ADAM17 is upregulated in the adult SVZ following cerebral stroke. (
Amyloid Precursor Protein is Upregulated in the Adult Subventricular Zone Following Cerebral Stroke
As APP gene expression was increased by stroke, we measured APP protein expression in the SVZ of rats 1, 7, 14, and 30 days post-MCAo. Immunohistochemistry indicated MCAo caused an increase in subventricular APP protein expression that was highest at 7 days post-MCAo in the time points we investigated (Figures 3B–3D). By 4 weeks, SVZ APP immunoreactivity had returned to near basal levels (not shown). This upregulation was specific to the SVZ ipsilateral to infarct as expression within the contralateral SVZ remained unchanged. Double immunolabeling for the astrocytic marker GFAP and the immature neural marker β-III-tubulin (Figures 2G–2N) revealed that converse to TACE protein, APP expression co-localized strongly with SVZ astrocytes and not β-III-tubulin reactive neuroblasts.
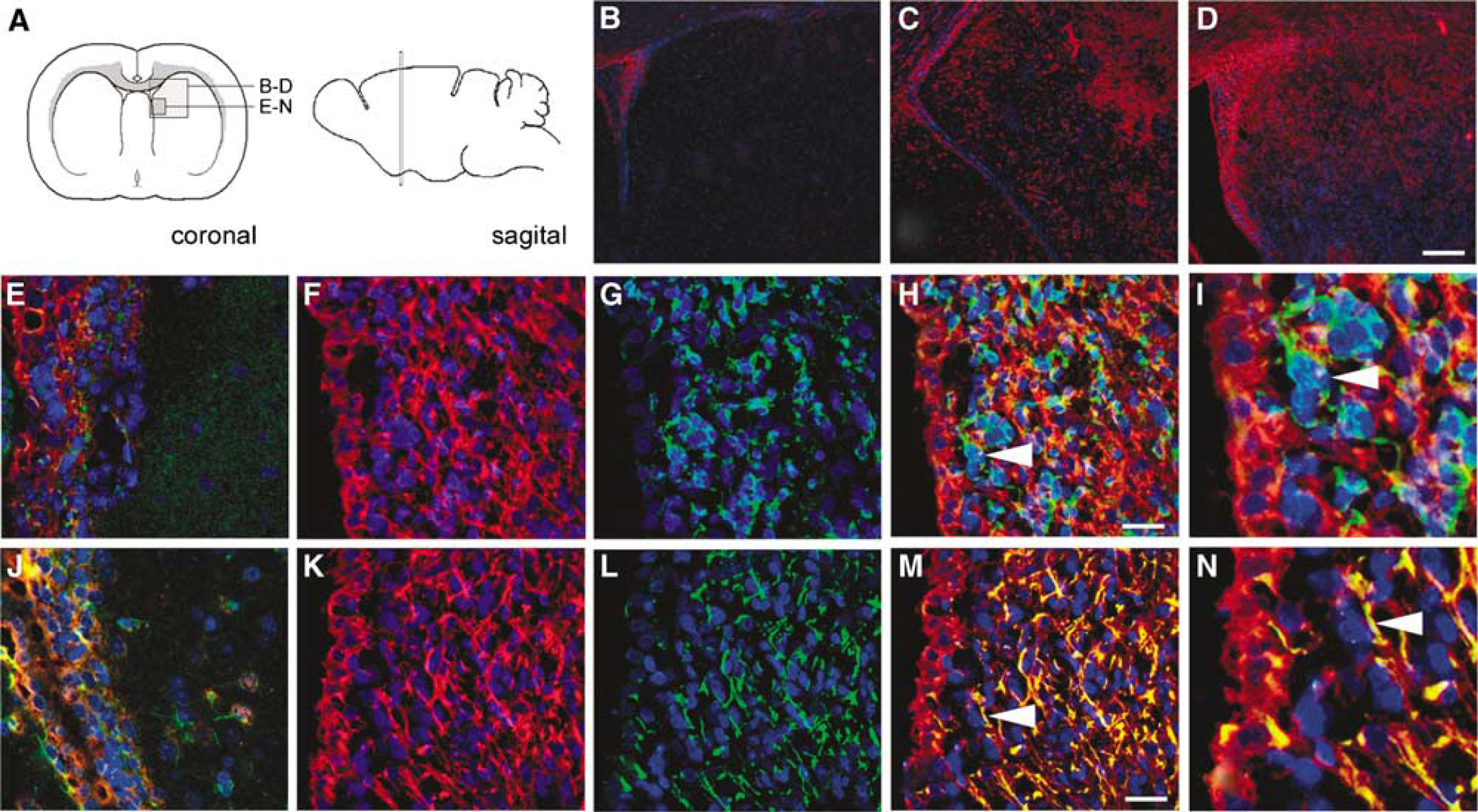
APP is upregulated by cerebral ischemic stroke. (
Stroke Brain Subventricular Zone Neurospheres Possess Increased Tumor Necrosis Factor-α-Converting Enzyme Protease-Dependent α-Secretase Activity
To determine if cerebral stroke altered α. or β-secretase activity within the adult SVZ, α and β proteolysis was measured in primary cultured SVZ neurospheres by enzymatic-specific fluorometric reaction. Subventricular zone neurospheres derived from brain 7 days post-MCAo possessed significantly enhanced α and β-secretase activity (P's < 0.01; Figures 4A and 4B). Treatment of SVZ neurospheres with 10 μmol/L TAPI-2 indicated stroke-increased α-secretase activity was largely dependent on TACE proteolysis (P < 0.01; Figure 4A).
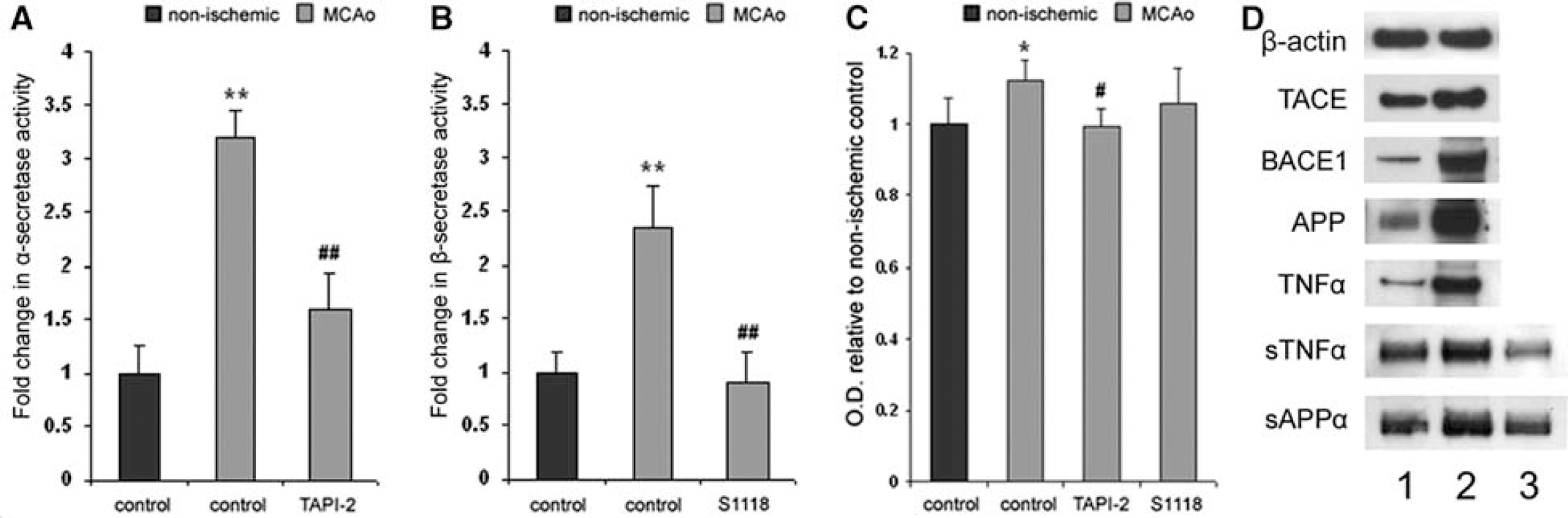
MCAo increases SVZ neurosphere α-secretase activity, TACE protease-dependent proliferation and TACE protein expression. (
Tumor Necrosis Factor-α-Converting Enzyme Proteolysis Promotes Stroke-Induced Subventricular Zone Progenitor Cell Proliferation In Vitro
To determine if TACE proteolytic activity regulated SVZ cell proliferation, normal and ischemic brain-derived SVZ neurospheres were cultured for 14 h in media containing 10 μmol/L BrdU and TAPI-2. Furthermore, to test whether β-secretase activity altered SVZ cell proliferation, we employed the β-secretase inhibitor S1118. Neither TAPI-2 nor S1118 significantly altered DNA replication in SVZ cells derived from nonischemic brain (not shown). However, densitometric measurement of BrdU incorporation indicated MCAo increased SVZ progenitor cell DNA replication (P < 0.05 compared with control), and that 10 μmol/L TAPI-2 was sufficient to block this increase (P < 0.05 compared with MCAo, P > 0.05 compared with control; Figure 4C). Inhibition of β-secretase activity had no significant effect on BrdU incorporation in either normal or ischemic-derived SVZ cells (Figure 4C).
Stroke Brain Subventricular Zone Neurospheres Over-Express Tumor Necrosis Factor-α-Converting Enzyme, Amyloid Precursor Protein and Tumor Necrosis Factor-α Protein, and Produce Soluble Amyloid Precursor Protein-α and Soluble Tumor Necrosis Factor-α via Tumor Necrosis Factor-α-Converting Enzyme Proteolysis
Western blot was employed to investigate whether ischemia altered SVZ neurospheres expression of TACE and its substrates. Immunoblotting revealed that SVZ neurospheres retained significantly increased expression of TACE, BACE1, APP, and TNF-α (Figure 4D, lanes 1 and 2). Furthermore, increased levels of sAPPα and sTNF-α were found in media conditioned by SVZ neurospheres. This production of sAPPα and sTNF-α by cultured SVZ neurospheres was upregulated by MCAo. Importantly, addition of TAPI-2 decreased levels of sAPPα and sTNF-α within MCAo-derived SVZ neurosphere media, indicating a role for TACE proteolysis in their release (Figure 4D, lane 3). Densitometric analysis of bands indicated that immunodetection levels were significantly different for all measured proteins (P's < 0.05, MCAo compared with normal, densitometric data not shown).
Inhibition of Tumor Necrosis Factor-α-Converting Enzyme Proteolysis Decreases Stroke-Induced Subventricular Zone Proliferation In Vivo
To determine if inhibition of TACE proteolysis could alter stroke-induced SVZ proliferation in vivo, 10 mmol/L TAPI-2, or saline vehicle was administered (1 μL/h) into the lateral ventricle ipsilateral to ischemic infarct beginning 24 h post-MCAo and continuing for 7 days. Concomitantly, animals received daily injections of BrdU (100 mg/kg, i.p.) to label mitotic cells. Immunohistochemical staining for BrdU after killing indicated that treatment with TAPI-2 significantly reduced the number of SVZ cells that had incorporated BrdU (P < 0.05 compared with saline control; Figure 5C).
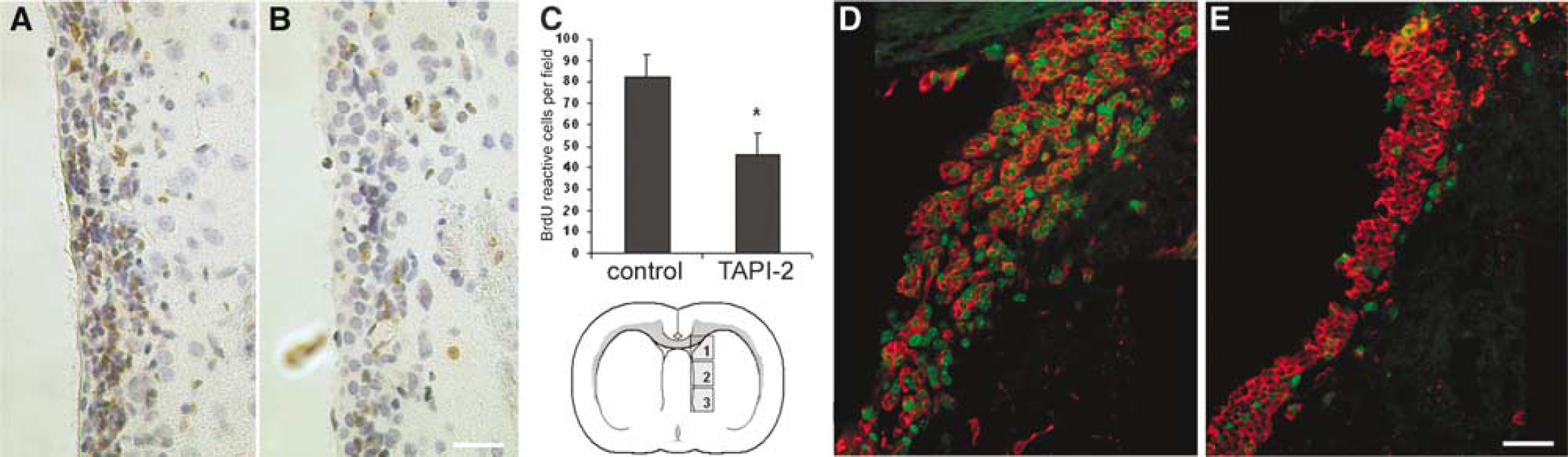
7-day intraventricular infusion of 10 mmol/L TAPI-2 beginning 24 h after stroke reduces SVZ proliferation. (
Immunostaining adjacent tissue sections for the proliferation marker Ki67 revealed a similar reduction of Ki67 reactive cells in SVZ of TAPI-2 treated animals (qualitative analysis). Doublestaining for doublecortin (Dcx), a marker for immature migratory neuroblasts, revealed Ki67/Dcx+ Ki67/Dcx− cells in the SVZ of both treated and untreated animals (Figures 5D and 5E). However, no clear difference in the total number of subventricular Dcx immunoreactive cells was observed between groups.
Stroke Increases Subventricular Zone Cell Migration and Tumor Necrosis Factor-α Protease Inhibitor-2 Decreases Migration Independent of Proliferation
To determine if TACE proteolysis altered SVZ progenitor cell motility, we cultured SVZ tissue explants from normal and 7 days post-MCAo adult rats embedded in Matrigel and measured radial migration of cells out of the explants over 5 days. Our assay revealed stroke significantly increased the rate of radial migration of SVZ progenitor cells into the matrix as compared with non-ischemic brain (P < 0.01; Figures 6A, 6B, and 6D). Addition of TAPI-2 or the broad-based MMP inhibitor TIMP-1 significantly reduced migration of MCAo-derived SVZ progenitor cells (P < 0.05; Figure 6A), but did not significantly alter normal SVZ progenitor cell migration (not shown). Treatment of SVZ explants with the β-secretase inhibitor S1118 decreased migration of MCAo-derived SVZ progenitor cells as well (Figure 6A; P < 0.05).
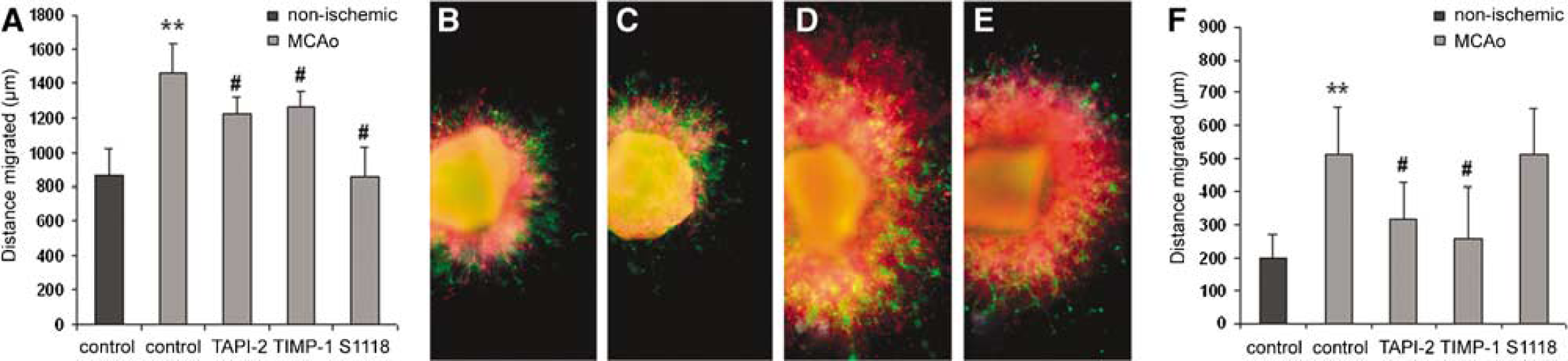
TAPI-2 inhibits stroke-increased SVZ neuroblast migration independent of proliferation. (
Our previous experiments indicated TACE inhibition could decrease SVZ cell proliferation. Therefore, to distinguish migration from proliferation, we repeated the same experiment with the addition of the mitotic antagonist Ara-C to all groups. Ara-C treatment reduced the rate of SVZ progenitor cell radial migration in all groups. However, addition of Ara-C revealed that MCAo significantly increased SVZ cell motility independent of proliferation (P < 0.01; Figure 6F). Furthermore, in the presence of Ara-C, TAPI-2 and TIMP-1 significantly reduced ischemic-brain SVZ progenitor cell migration whereas S1118 did not.
Immunolabeling SVZ explants for the neuroblast marker β-III-tubulin and the astrocytic marker GFAP indicated that both SVZ neuroblasts and astrocytes migrated into the Matrigel matrix (Figures 6B–6E). MCAo increased the neuroblast/astrocyte ratio migrating out of the explants compared with normal, as previously described (Katakowski et al, 2003); however, no significant changes in the neuroblast/astrocyte ratio resulting from TAPI-2, TIMP-1, or S1118 were evident in this experiment.
Discussion
Ischemic cerebral stroke induces SVZ neurogenesis in the adult rodent brain (Arvidsson et al, 2002; Zhang et al, 2001). The SVZ contains multipotent neural progenitor cells that, in response to stroke, exit the rostral migratory complex, and migrate laterally to the infarct boundary (Arvidsson et al, 2002; Zhang et al, 2001, 2004). As some SVZ-derived neuroblasts express mature neuronal markers and form somatodendritic junctions with adjacent striatal neurons, it has been hypothesized they contribute to restorative plasticity (Jin et al, 2003; Zhang et al, 2005).
In this study, we show TACE protease activity contributes stroke-induced SVZ cell proliferation. In adult rat, TACE protein expression is increased in β-tubulin-III reactive SVZ neuroblasts after MCAo, which was highest 7 days after onset. Subventricular zone neurospheres derived from SVZ tissue 7 days post-MCAo overexpress TACE and retain increased α-secretase activity. We also report the TACE substrate APP is highly upregulated in GFAP-reactive astrocytes of ischemic SVZ. Stroke brain-derived SVZ cells release more sAPPα and sTNFα compared with nonischemic control via a TACE protease-dependent mechanism. Furthermore, inhibition of TACE proteolysis reduced SVZ neuroblast migration in culture.
We suggest proteolytic shedding of APP, TNFα, and/or activation of EGFR proligands as a possible mechanism by which TACE proteolysis promotes SVZ proliferation. Tumor necrosis factor-α-converting enzyme is a primary protease for APP and TNFα in brain, and of the EGFR ligand-activating ADAMs 9, 10, 12, 15, 17, and 19, TACE is the major convertase of epiregulin, transforming growth factor-α, amphiregulin, and heparin-binding EGF-like growth factor (Blobel, 2005; Sahin et al, 2004). TAPI-2 has been employed as a potent inhibitor of TACE proteolysis in vivo and in vitro (Kaup et al, 2002; Lomniczi et al, 2006). However, ADAM10 is capable of analogous α-cleavage of APP and TNFα, and like TACE, activates many EGFR proligands as well (Blobel, 2005; Kaup et al, 2002; Sahin et al, 2004). Here, we found ADAM10 transcription was increased several-fold in the SVZ following stroke. Thus, it is possible ADAM10 or comparable proteases contributed to TAPI-2-responsive stroke-induced SVZ proliferation.
Subventricular zone cells can be subdivided into three primary cell types: type-A proliferative neuroblasts, type-B astrocytic self-renewing neural stem cells, and type-C transit-amplifying secondary precursors (Alvarez-Buylla and Garcia-Verdugo, 2002). Soluble amyloid precursor protein-α increases SVZ cell proliferation, by stimulating the transit-amplifying type-C cells or the type-A SVZ neuroblasts (Caille et al, 2004). We found APP and TACE proteins were not co-localized within SVZ cells in vivo. Thus, TACE shedding of sAPPα may occur largely within the adjacent ependyma where both proteins are present. Still, TACE protease-dependent production of sAPPα occurred in passaged SVZ neurospheres where ependymal cells are not thought to persist (Doetsch et al, 1999). In lieu of unreported trans cleavage of APP by TACE, it is possible sAPPα was released by another TAPI-2-susceptible protease. Further investigation of TACE shedding of APP within SVZ cells is thus warranted.
Stroke is a known cause of dementia, and ischemic events are known to contribute to Alzheimer's disease progression, as Aβ deposits are associated with ischemic injury (Koistinaho and Koistinaho, 2005). Specifically, evidence suggests that accumulation of C- and N-terminal fragments from β-secretase cleavage of APP interfere with plasticity-related factors such as MAP1B (Badan et al, 2004). It has therefore been surmised that overexpression of APP combined with enhanced amyloidogenic processing may be a factor in neurodegeneration after stroke (Koistinaho and Koistinaho, 2005). As TACE proteolysis of APP precludes Aβ production, this proteolytic point of divergence may represent an attractive target for stroke recovery.
Tumor necrosis factor-α is another substrate for TACE, which increased in SVZ cells after MCAo. TNF-α infusion increases the number of mitotic SVZ neural progenitors in the adult rat brain (Wu et al, 2000). Intraventricular infusion of an antibody to TNF-α reduces stroke-induced neurogenesis (Heldmann et al, 2005). In this investigation, we found that SVZ progenitor cells isolated from ischemic brain overexpressed TNF-α compared with uninjured control. These cells released more sTNF-α via a TACE protease-dependent mechanism. The neuroprotective effects of TACE-produced sTNF-α may therefore contribute to stroke-induced SVZ expansion.
Epidermal growth factor receptor activation is a potent regulator of SVZ cell proliferation, and EGF and its receptor EGFR are upregulated in the stroke brain (Craig et al, 1996; Doetsch et al, 2002; Zhang et al, 2005). Tumor necrosis factor-α-converting enzyme is a primary protease for activation of the EGF and HB-EGF ligands (Blobel, 2005). Infusion of HB-EGF into the stroked brain increases SVZ neurogenesis (Jin et al, 2004). In this study, we did not investigate expression of the multiple EGFR-binding ligands. However, it is possible TACE proteolysis of these proteins may contribute to stroke-induced SVZ proliferation.
It has recently been reported that Neuregulin-2 binding to ErbB4 increases the number of Sox2 and GFAP+ progenitors in the SVZ, and increases the number of migrating neuroblasts in the rostral migratory stream (Ghashghaei et al, 2006). Furthermore, TACE proteolysis sheds the ErbB4 ectodomain, and can produce an active intracellular fragment that stimulates proliferation of breast cancer cells independent of ligand binding (Maatta et al, 2006). It is therefore also possible processing of ErbB4 could be involved in TACE protease-dependent SVZ proliferation after stroke.
Little is known about the role for ADAMs in neural progenitor migration. Here, we found TAPI-2 significantly reduced SVZ cell proliferation. However, TAPI-2 has been reported to inhibit cleavage of matrix proteins in addition to membrane-bound ligands. As recombinant TIMP-1, which can inhibit several matrix metalloproteinases and ADAM10, but not TACE proteolysis, similarly reduced SVZ cell migration, it is possible that TAPI-2 inhibition of matrix protein cleavage may have attenuated SVZ migration as well.
Tumor necrosis factor-α-converting enzyme is a protease required for the activation of a variety of growth factors, cytokines, and receptors, some of which have function in maintenance of SVZ neural progenitor cells (Blobel, 2005; Caille et al, 2004; Craig et al, 1996; Wu et al, 2000). Our findings suggest that TACE may be involved in the maintenance of SVZ expansion via an autocrine neurotrophic mechanism. This hypothesis is supported by the recent finding that SVZ cells produce diffusible heat-labile molecules that promote SVZ progenitor cell proliferation (Agasse et al, 2004). Although we report increased release of sAPP-α and sTNF-α, further studies are required to evaluate the contributions of TACE protease substrates to stroke-induced SVZ cell proliferation.
TACE upregulation in the SVZ was transient after MCAo. Although we examined a limited number of time points, our data suggest TACE upregulation coincided with timing of stroke-induced SVZ neurogenesis (Zhang et al, 2001). As inhibition of TACE proteolysis reduced stroke-induced SVZ proliferation, it would be interesting to test whether augmentation of TACE expression and/or activity could increase or prolong stroke-induced SVZ neurogenesis. Conversely, as treatments that increase SVZ neurogenesis are correlated with enhanced functional recovery (Zhang et al, 2005), experiments designed to test whether TACE inhibitors reduce neurological outcome after stroke are warranted.
We show TACE is upregulated within the SVZ ipsilateral to infarct after MCAo, and TACE proteolytic activity promotes stroke-induced SVZ progenitor cell proliferation in vitro and in vivo. In addition, the TACE proteolytic substrate APP is overexpressed in the ischemic SVZ. Subventricular zone neurospheres derived from ischemic brain proliferate more rapidly, and produce more sAPP-α and sTNF-α via a TACE protease-dependent mechanism compared with nonischemic control. Taken together, these data suggest TACE proteolysis contributes to stroke-induced adult SVZ neurogenesis. As our findings indicate TACE proteolysis as a positive modulator of stroke-induced proliferation and thus, may represent a novel therapeutic target for stroke recovery.
Footnotes
Acknowledgements
The authors would like to thank Cindi Roberts and Debbie Jewell for technical assistance.
