Abstract
Transplantation of human mesenchymal stem cells (hMSCs) prepared from adult bone marrow has been reported to ameliorate functional deficits after cerebral artery occlusion in rats. Although several hypotheses to account for these therapeutic effects have been suggested, current thinking is that both neuroprotection and angiogenesis are primarily responsible. In this study, we compared the effects of hMSCs and angiopoietin-1 gene-modified hMSCs (Ang-hMSCs) intravenously infused into rats 6 h after permanent middle cerebral artery occlusion. Magnetic resonance imaging and histologic analyses revealed that rats receiving hMSCs or Ang-hMSCs exhibited comparable reduction in gross lesion volume as compared with the control group. Although both cell types indeed improved angiogenesis near the border of the ischemic lesions, neovascularization and regional cerebral blood flow were greater in some border areas in Ang-hMSC group. Both hMSC- and Ang-hMSC-treated rats showed greater improved functional recovery in the treadmill stress test than did control rats, but the Ang-hMSC group was greater. These results indicate the intravenous administration of genetically modified hMSCs to express angiopoietin has a similar effect on reducing lesion volume as hMSCs, but the Ang-hMSC group showed enhanced regions of increased angiogenesis at the lesion border, and modest additional improvement in functional outcome.
Introduction
Although transplantation of human mesenchymal stem cells (hMSCs) several hours after onset of ischemia can reduce infarction size and improve functional outcome in rodent cerebral ischemia models (Chen et al, 2001; Li et al, 2002; Iihoshi et al, 2004; Nomura et al, 2005; Honma et al, 2006; Horita et al, 2006; Liu et al, 2006), the detailed mechanisms by which MSCs promote functional recovery remain unclear. Angiogenesis is thought to be associated with improved neurologic recovery after ischemia (Krupinski et al, 1994; Chen et al, 2003).
Vascular endothelial growth factor has been reported to show strong angiogenetic effects in brain ischemia (Zhang and Chopp, 2002), limb ischemia (Baumgartner et al, 1998), and myocardial ischemia models (Kastrup, 2003). Vascular endothelial growth factor exerts its biologic functions via three related receptor tyrosine kinases, VEGFR-1, VEGFR-2, VEGFR-3 (Yancopoulos et al, 2000), and is required to initiate the formation of immature vessels by vasculogenesis or angiogenesis (Carmeliet and Collen, 1997). However, vascular endothelial growth factor (VEGF) is also a potent enhancer of vascular permeability to blood plasma proteins within minutes after ischemic insult, which often causes brain edema after the cerebral infarction (Wang et al, 1996; Bates et al, 2002).
Angiopoietin is involved in maturation, stabilization, and remodeling of vessels (Davis et al, 1996; Suri et al, 1996, 1998; Yancopoulos et al, 2000). Angiopoietin-1 is the best characterized member of this family and binds to Tie2, a receptor tyrosine kinase that is expressed on endothelial cells lining blood vessels and in the choroid plexus (Dumont et al, 1994; Nourhaghighi et al, 2003), and promotes angiogenesis in the brain (Ward and Lamanna, 2004). Furthermore, angiopoietin-1 (Ang-1) protects the peripheral vasculature from vascular leakage (Thurston et al, 1999), which may account for its anti-edematic effects after cerebral ischemia.
In the present study, hMSCs and Ang-1 gene-modified hMSCs (Ang-hMSCs), which had been transfected with the Ang-1 gene were intravenously delivered 6 h after induction of unilateral permanent cerebral ischemia to investigate if cellular delivery of Ang-1 by hMSCs could influence angiogenesis and functional outcome.
Materials and methods
Preparation of Human Mesenchymal Stem Cells
Human mesenchymal stem cells were processed for cell culture as described previously (Nomura et al, 2005; Honma et al, 2006; Liu et al, 2006). Briefly, human bone marrow from healthy adult volunteers was obtained by aspiration from the posterior iliac crest after informed consent was obtained; the subject's consent was obtained according to the Declaration of Helsinki Principles, and this study was approved by the Institutional Review Board at our university. Bone marrow mononuclear cells were isolated, and were plated in 150-cm2 plastic tissue culture flasks and incubated overnight. After washing away the free cells, the adherent cells were cultured in Mesenchymal Stem Cell Basal Medium (Cambrex, Walkersville, MD, USA) containing Mesenchymal Cell Growth Supplement (Cambrex), 4 mmol/L
Adenoviral Vectors
Adenoviral vectors carrying a human Ang-1 cDNA were constructed as described previously with minor modifications (Takahashi et al, 2003; Kurozumi et al, 2004). Briefly, human Ang-1 cDNA was cloned using the reverse transcription polymerase chain reaction method using the total RNA extracted from primary MSC as the template. The identity of Ang-1 cDNA obtained in this manner was confirmed by sequencing and comparing it with the GeneBank sequence U83508. The Ang-1 cDNA was inserted between the
Before being used, the above viral vectors were evaluated for their viral concentration and titer, and viral stocks were examined for potential contamination with replication-competent viruses. To determine viral concentration (particle unit (pu)/mL), the viral solution was incubated in 0.1% sodium dodecyl sulfate and
Adenovirus Infection
Adenovirus-mediated gene transfection was performed as described previously (Takahashi et al, 2003; Kurozumi et al, 2004). Briefly, the cells were seeded at a density of 2 × 106 cells per 15 cm plate. Human mesenchymal stem cells were exposed to the infectious viral particles in 7.5 mL Dulbecco's modified Eagle's medium at 37°C medium for 60 mins; cells were infected with AxCAhAng1-F/RGD or AxCALacZ-F/RGD at an multiplicity of infection (MOI) of 3.0 × 103 pu/cell. The medium was then removed, and the cells were washed once with Dulbecco's modified Eagle's medium and then recultured with normal medium for 24 h, after which transplantation was performed.
Phenotypic Characterization of the Human Mesenchymal Stem and Angiopoietin-1 Gene-Modified Human Mesenchymal Stem Cells
Flow cytometric analysis of primary hMSCs and Ang-hMSCs were performed as described previously (Honma et al, 2006; Liu et al, 2006). Briefly, cell suspensions were washed twice with phosphate-buffered saline containing 0.1% bovine serum albumin. For direct assays, aliquots of cells at a concentration of 1 × 106 cells/mL were immunolabeled at 4°C for 30 mins with the following anti-human antibodies: fluorescein isothiocyanate (FITC)-conjugated CD45, CD34, CD105 (Immunotech, Marseilles, France), and anti-human CD73 (SH-3; BD Biosciences Pharmingen, San Diego, CA, USA). As an isotype-matched control, mouse immunoglobulin G1-FITC (IgG1-FITC; Immunotech) was used. Labeled cells were analyzed by a FACSCalibur flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA) with the use of CellQuest software. Dead cells were gated out with forward- versus side-scatter window and propidium iodide staining.
Cerebral Ischemic Model
The rat middle cerebral artery occlusion (MCAO) model was used as a stroke model. We induced permanent MCAO by using a previously described method of intraluminal vascular occlusion (Longa et al, 1989). Adult male Sprague—Dawley rats weighing 250 to 300 g were anesthetized with an intraperitoneal injection of ketamine (75 mg/kg) and xylazine (10 mg/kg). A length of 20.0 to 22.0 mm 4-0 surgical dermalon suture with the tip rounded by heating near a flame was advanced from the external carotid artery into the lumen of the internal carotid artery until it blocked the origin of the MCA.
Transplantation Procedures
Experiments consisted of three groups: group 1, medium (Mesenchymal Stem Cell Basal Medium) alone (without donor cell administration) (
In some experiments, AxCALacZ-F/RGD adenovirus was used to transduce the LacZ gene into the hMSC (
Magnetic Resonance Imaging Studies and Measurement of Infarct Volume
Rats were anesthetized intraperitoneally with ketamine (75 mg/kg) and xylazine (10 mg/kg). Each rat was placed in an animal holder/magnetic resonance imaging (MRI) probe apparatus and positioned inside the magnet. The animal's head was held in place inside the imaging coil. All MRI measurements were performed using a 7-T, 18-cm-bore superconducting magnet (Oxford Magnet Technologies, Witney, Oxfordshire, UK) interfaced to a UNITYINOVA console (Oxford Instruments, UK and Varian, Palo Alto, CA, USA). T2-weighted images (T2WIs) were obtained from a 1.0-mm-thick coronal section with a 0.5 mm gap using a 30 mm × 30 mm field of view, TR= 3000 ms, TE = 37 ms,
The ischemic lesion area was calculated from both T2WI and DWI using imaging software (Scion Image, Version Beta 4.0.2, Scion Corporation, Frederick, MD, USA), based on the previously described method (Neumann-Haefelin et al, 2000). For each slice, the higher intensity lesions in both T2WI and DWI, where the signal intensity was 1.25 times higher than the counterpart in the contralateral brain lesion, were marked as the ischemic lesion area, and infarct volume was calculated taking slice thickness (1 mm/slice) into account.
Detection of Angiopoietin-1 In Vitro
In separate dishes, we cultured hMSCs (1.0 × 105) transfected
2,3,5-Triphenyltetrazolium Chloride Staining and Quantitative Analysis of Infarct Volume
One week after transplantation, the rats were deeply anesthetized intraperitoneally with ketamine (4.4 to 8 mg/kg) and xylazine (1.3 mg/100 g). The brains were removed carefully and dissected into coronal 1 mm sections using a vibratome. The fresh brain slices were immersed in a 2% solution of 2,3,5-triphenyltetrazolium chloride (TTC) in normal saline at 37°C for 30 mins.
The cross-sectional area of infarction in each brain slice was examined with a dissection microscope and was measured using an image analysis software, NIH image. The total infarct volume for each brain was calculated by summation of the infarcted area of all brain slices.
Detection of Donor Angiopoietin-1 Human Mesenchymal Stem Cells In Vivo
Immunohistochemistry
Two weeks after transplantation, the LacZ-transfected Ang-hMSCs
Three-Dimensional Image Acquisition for Angiogenesis
Seven and twenty-eight days after MCAO, rats were anesthetized and intravenously perfused with FITC-dextran (1 mL, Sigma). The brains were immediately removed, and were fixed in 4% paraformaldehyde at 4°C for 48 h. Coronal vibratome sections (100 μm) were analyzed with a laser-scanning confocal imaging system (LSM Pascal; Carl Zeiss, Jend, Germany). The sections were scanned in 512 × 512 pixel (651.5 μm × 651.5 μm) format in the
Dynamic Susceptibility Contrast-Enhanced Perfusion-Weighted Imaging
Perfusion-weighted imaging was acquired using T*2-weighted (TR = 13 ms, TE = 6.0 ms) gradient echo sequence (Ukai et al, 2007). A dynamic image series of 30 measurements resulted in a total scan time of 26 secs, with a fields of view of 30 mm and image acquisition matrix of 128 × 64, which was interpolated by zero-filling to 512 × 512. During the dynamic series, a triple-dose (0.6 mL/kg) bolus injection of Magnevist (Schering AG, Berlin, Germany) was started after the fifth acquired volume to ensure a sufficient precontrast baseline. Images were reconstructed by an Inova Vision. Perfusion-weighted imaging measurements were obtained 6 h, 3, and 7 days after MCAO. For the perfusion-weighted imaging (PWI) and PWI-derived parameter maps, only one representative slice (involving cortex and stria terminalis) with the maximum lesion involving both cortex and striatum was chosen for cerebral blood flow quantification. The readout of abnormal regional cerebral blood flow (rCBF) from the regions of perfusion deficiency as a percentage of that measured in the contralateral brain was generated using Perfusion Solver software. Regions of interest (ROI) consists of four groups, based on the results of DWI, T2WI, and PWI. ROI-1 is defined as abnormal in all images, ROI-2 as normal in only T2WI and abnormal in others, ROI-3 as abnormal in only PWI and normal in others, and ROI-4 as normal in all images.
Treadmill Stress Test
Rats were trained 20 mins/day for 2 days a week to run on a motor-driven treadmill at a speed of 20 m/min with a slope of 0°C. Rats were placed on a moving belt facing away from the electrified grid and induced to run in the direction opposite to the movement of the belt. Thus, to avoid foot shocks (with intensity in 1.0 mA), the rats had to move forward. Only the rats that had learned to avoid the mild electrical shock were included in this study. The maximum speed at which the rats could run on a motor-driven treadmill was recorded.
Statistical Analysis
Data are presented as mean values ± s.d., and were statistically analyzed. Differences among groups were assessed by ANOVA with Scheffes
Results
Characteristics of Primary and Angiopoietin-1 Gene-Transduced Human Mesenchymal Stem Cells
Primary hMSCs were cultured as plastic adherent cells to subconfluency, which took approximately 1 week (see Materials and methods). A characteristic feature of hMSCs is a CD34−, CD45−, SH2+ (CD105), and SH3+ (CD73) cell surface phenotype (Kobune et al, 2003). Both flattened and spindle-shaped cells can be recognized in the culture. Angiopoietin-1 gene-modified human mesenchymal stem cells showed similar flattened and spindle-shaped morphology. Flow cytometric analysis of the Ang-hMSCs (Figure 1B) was essentially identical to primary hMSCs (Figure 1A).
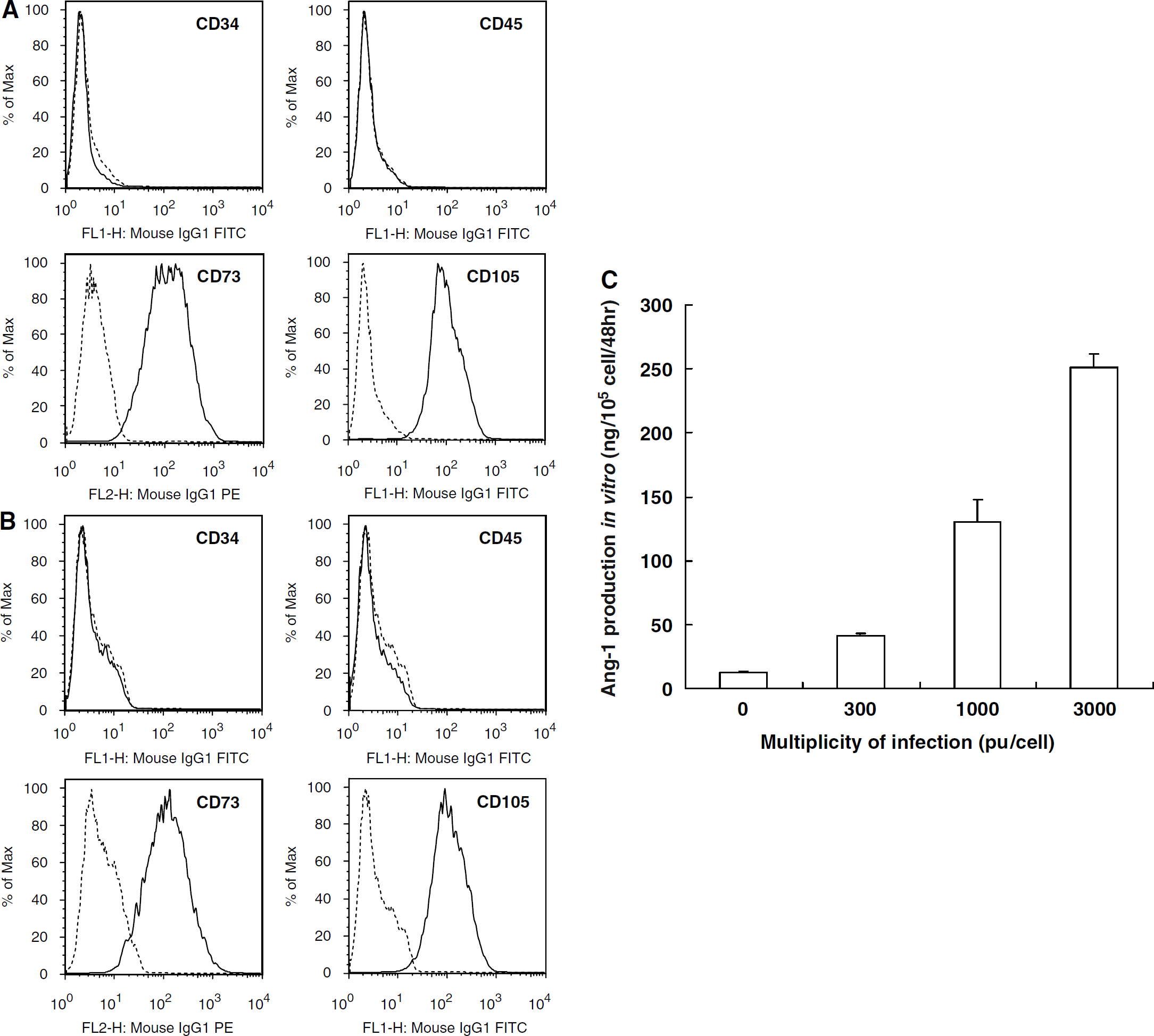
Flow cytometric analysis of surface antigen expression on primary hMSC (
Detection of Immunoreactive Human Angiopoietin-1 and Quantitative Analysis In Vitro
Levels of Ang-1 in the supernatant of cultured hMSCs and Ang-hMSCs with different MOI levels were studied. Human mesenchymal stem cell transfected with AxCAhAng1-F/RGD (Ang-hMSC) at an MOI of 300, 1000, and 3000 pu/cell secreted Ang-1 at a rate of 40.75 ± 1.94, 130.08 ± 17.78, and 251.37 ± 10.31 ng/105 cells/48 h, respectively (
Characterization of Ischemic Lesion Size by Magnetic Resonance Image Analysis
An estimate of lesion size was obtained using
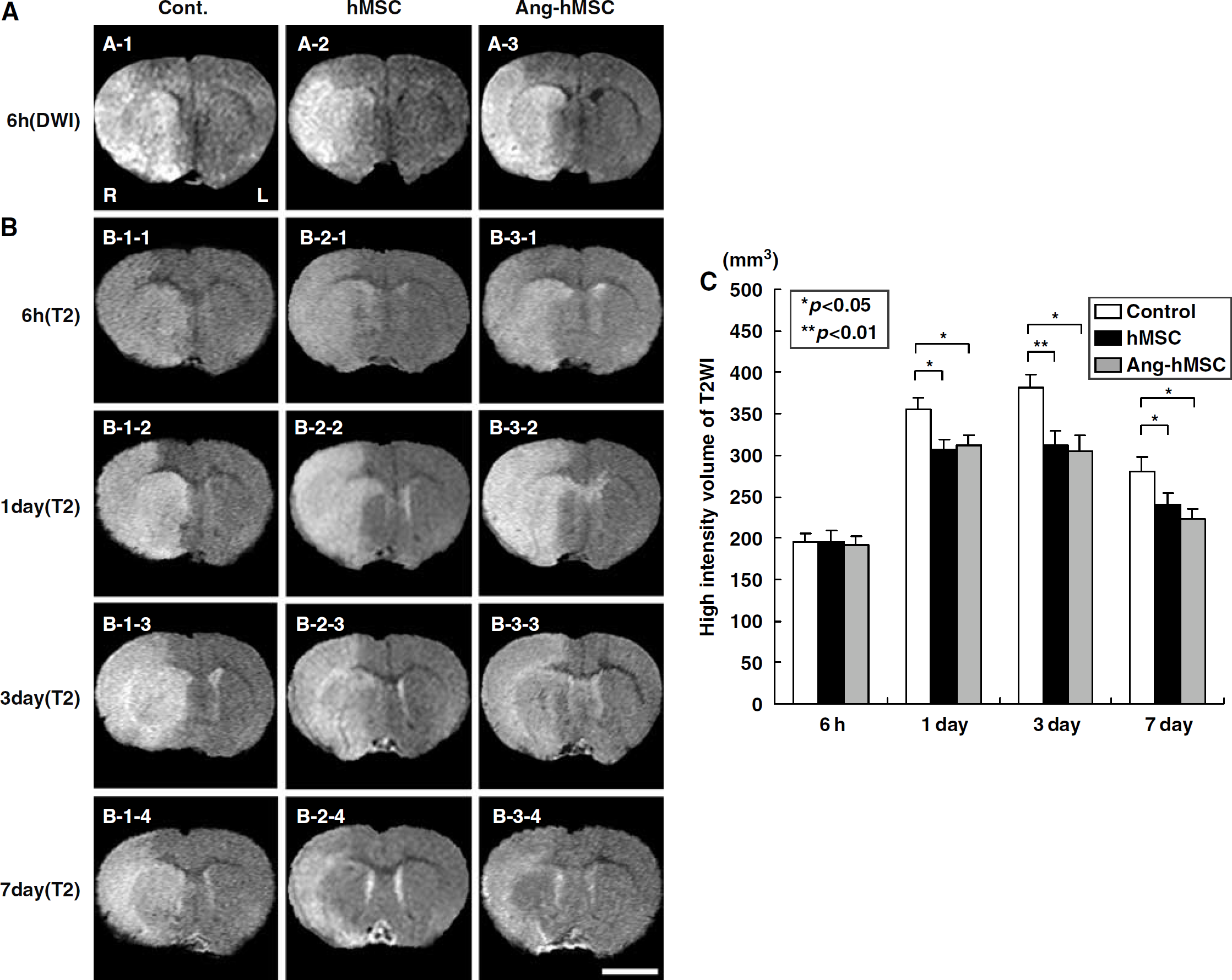
Evaluation of the ischemic lesion volume with DWIs and T2WIs. Human mesenchymal stem cells or Ang-hMSCs were intravenously injected immediately after the initial MRI scanning (6 h after MCAO). Diffusion-weighted image obtained 6 h MCAO in medium-injected (
To determine the efficacy of hMSCs or Ang-hMSCs transplantation in modulating the ischemic lesion volume, 1 × 106 cells were delivered intravenously at 6 h after MCAO, and animals were reimaged at 6, 24 h, 3, and 7 days later. In the sham control group, MRI-estimated lesion volume reached maximum at 3 days (Figure 2B-1–3) and gradually decreased at 7 days post-MCAO (Figure 2B-1–4). Although lesion volumes estimated 6 h after MCAO were the same among the three groups (Figures 2A and 2B) at 1, 3, and 7 days, lesion volume was less in both hMSC and Ang-hMSC groups as compared with control. The reduction in lesion volume showed no significant difference between the Ang-hMSC group and the hMSC group at 1, 3, and 7 days post-MCAO. These results are summarized in Figure 2C.
Histological Determination of Infarction Volume
A second independent measure of infarction volume was performed. The brains were stained with TTC 7 days after MCAO (Figure 3). Normal brain (gray matter) tissue typically stains with TTC, but infarcted lesions show no or reduced staining (Bederson et al, 1986). 2,3,5-Triphenyltetrazolium chloride staining obtained 7 days after MCAO without cell transplantation is shown in Figure 3A. Note the reduced staining on the lesion side. Lesion volume was calculated by measuring the area of reduced TTC staining in the forebrain (
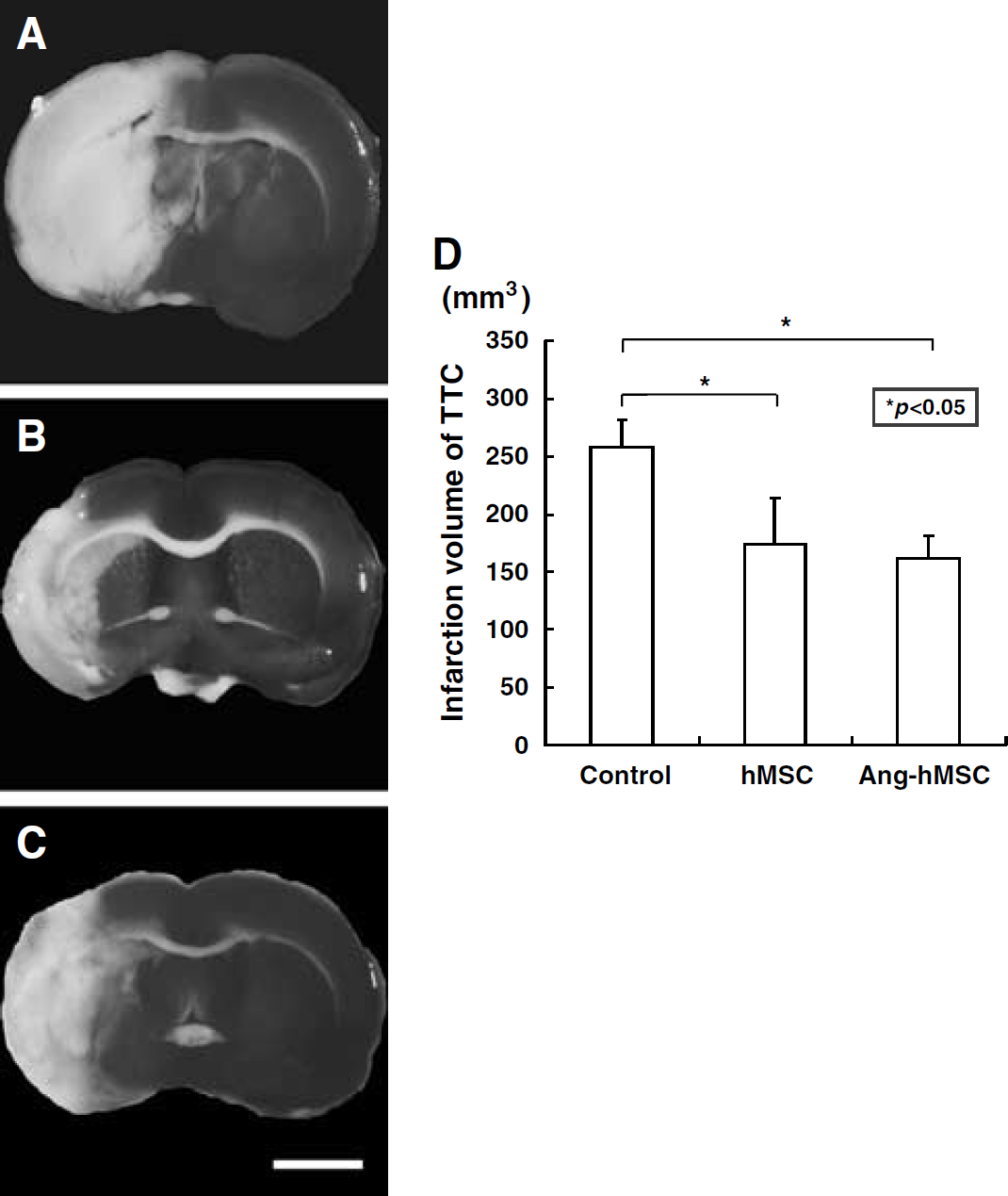
TTC Brain sections slices stained with TTC to visualize the ischemic lesions 7 days after MCAO. 2,3,5-Triphenyl tetrazolium chloride-stained brain slices from (
Angiogenesis Induced by the Transplantation
To examine whether the administration of hMSCs and Ang-hMSCs induces angiogenesis, three-dimensional analysis of capillary vessels in the lesion was performed using Zeiss LSM5 PASCAL software. Figure 4A-1 shows the three-dimensional capillary image in the normal rat brain (
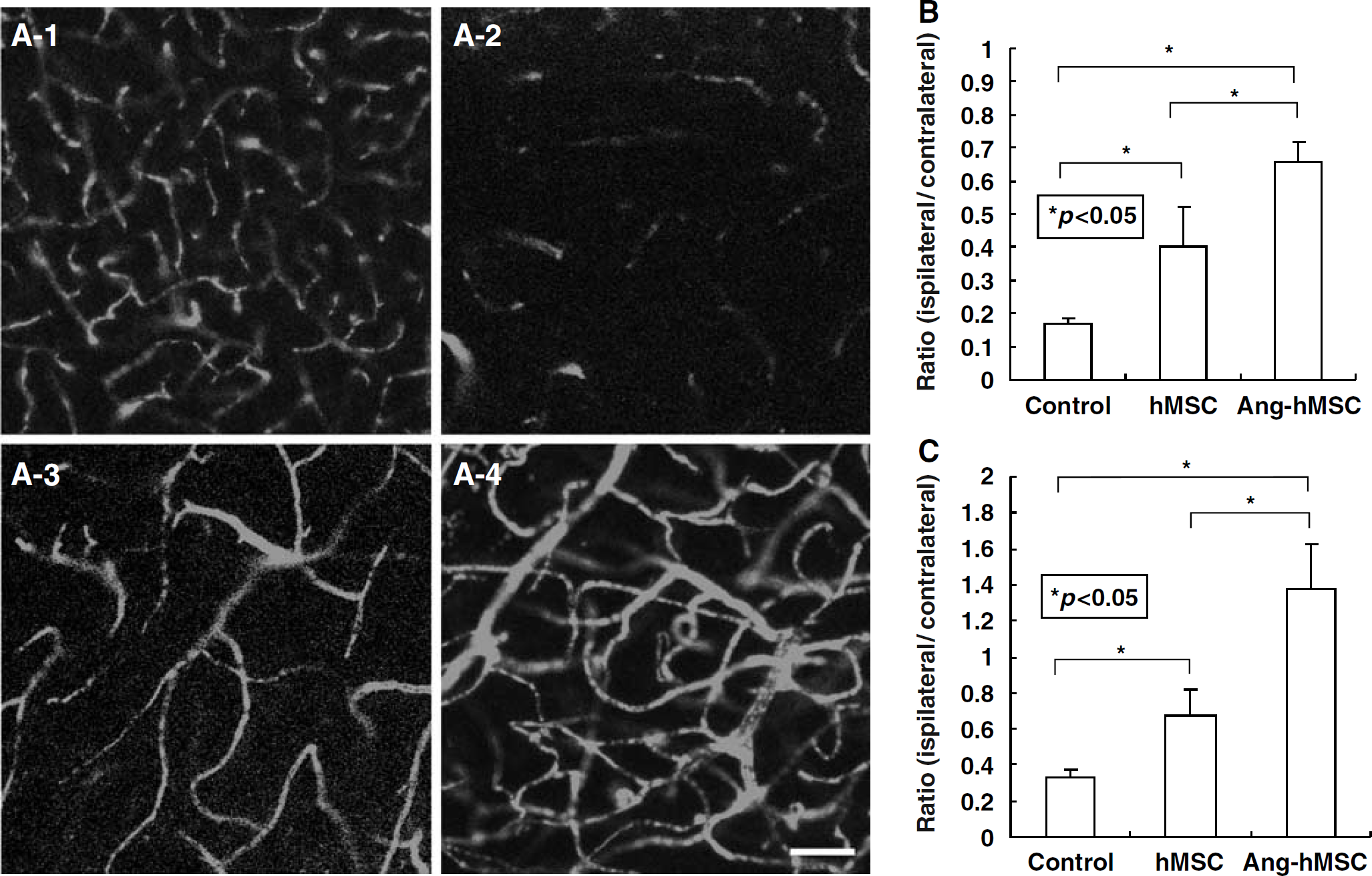
Seven and twenty-eight days after MCAO, the angiogenesis in boundary zone was analyzed using a three-dimensional analysis system. (
The capillary vascular volume was expressed as a ratio by dividing that obtained from the ischemic hemisphere by that of the contralateral control hemisphere. At 7 days after MCAO, the ratio (ipsilateral/contralateral) was significantly higher in both the hMSC (0.17 ± 0.07;
LacZ-transfected hMSCs and Ang-hMSCs that had been intravenously administered (1.0 × 106 cells) 6 h after MCAO were identified
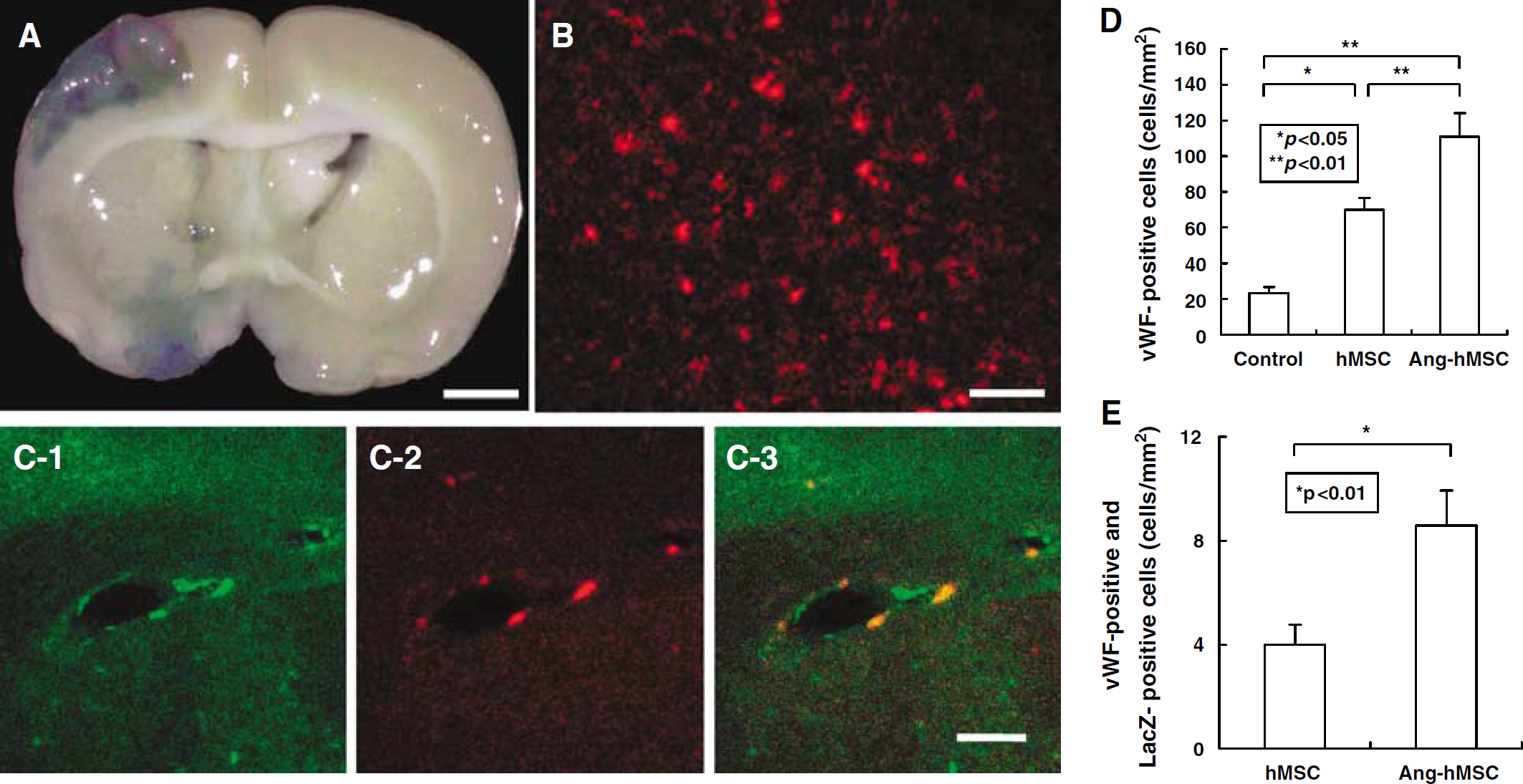
Intravenously—administered Ang-hMSCs accumulated in and around the ischemic lesion hemisphere. Angiopoietin-1 gene-modified human mesenchymal stem cells were transfected with the reporter gene LacZ. (
Immunohistochemical studies were performed to identify LacZ-positive cells in and around the lesion zone in animals transplanted with LacZ-transfected hMSCs. The photomicrographs of hMSCs and Ang-hMSCs demonstrated a large number of LacZ-positive cells in and around the lesion (Figure 5B). Double staining was performed to show endothelial cell differentiation of LacZ-positive cells in the lesion zone in animals transplanted with LacZ-transfected Ang-hMSCs (Figure 5C). The vWF-positive cells in the boundary region 7 days after MCAO were increased in the hMSC-treated group (
Dynamic Susceptibility Contrast-Enhanced Perfusion-Weighted Image
To assess regional cerebral blood flow, the PWI-derived parameter maps allowed further quantitative analysis for the hemodynamic changes of the lesions (see Materials and methods). Figures 6A—C shows images obtained at 6 h (row 1), 3 days (row 2), and 7 days (row 3). Control, hMSC- and Ang-hMSC-injected groups are in columns A, B, and C, respectively.
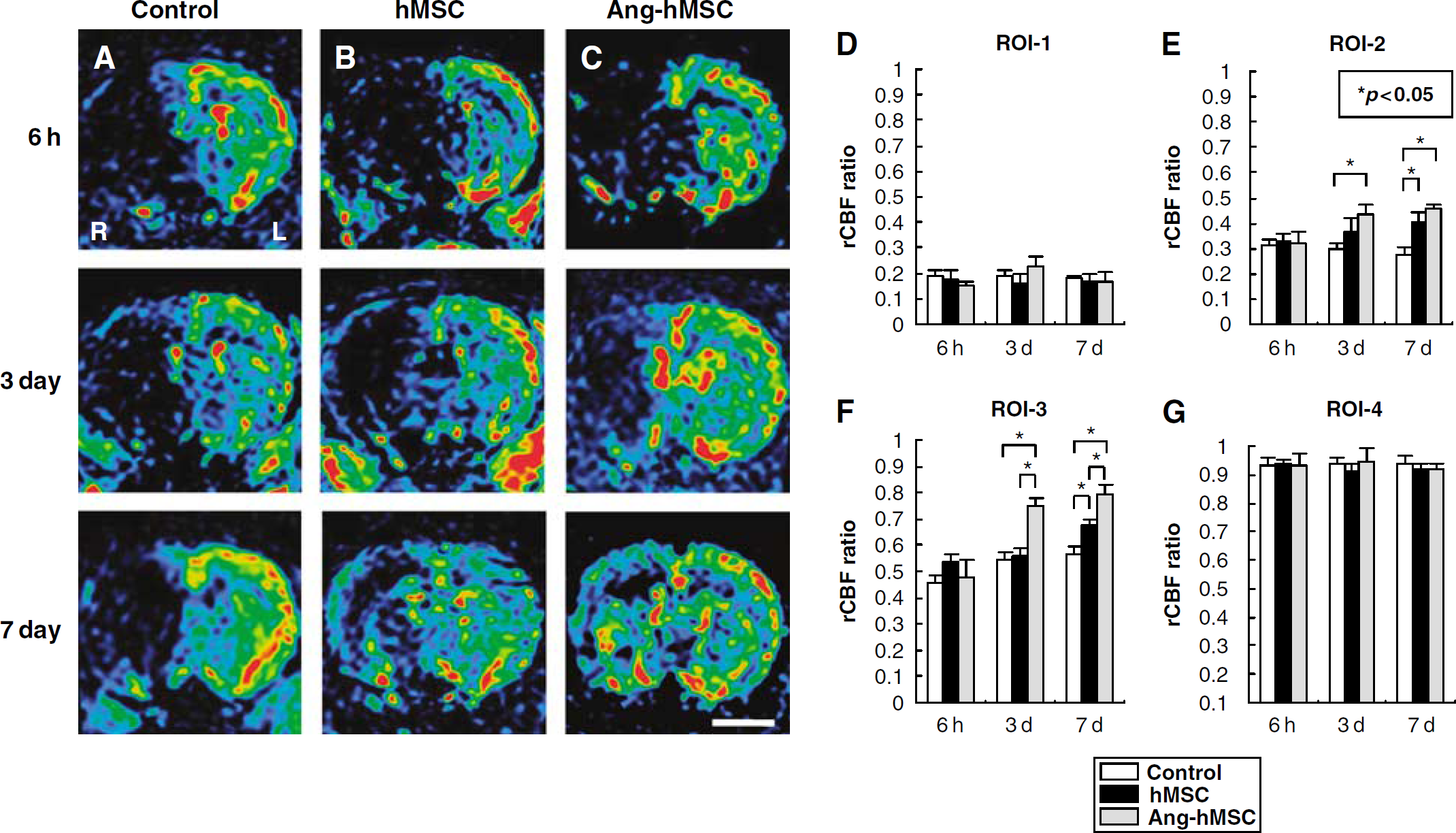
Evaluation of hemodynamic state (rCBF maps) with PWIs. Human mesenchymal stem cells or Ang-hMSCs were intravenously injected immediately after the initial MRI scanning (6 h after MCAO). Images obtained 6 h, 3, and 7 days MCAO in (
The four regions of interest (ROI) for the analysis are defined in Materials and methods. The severity of the lesion was greatest in ROI-1 and progressively less in ROI-2 through ROI-4. A rCBF ratio was calculated at each ROI from PWI obtained in the infarction hemisphere divided by that of the non-infarcted hemisphere. In ROI-1, the rCBF ratio of control, hMSC-treated, and Ang-hMSC-treated groups were similar and decreased to >25% at 6 h post-MCAO, and remained low at 3 and 7 days (Figure 6D). The rCBF ratio in ROI-2 of the three groups was similar at 6 h post MCAO. However, the rCBF ratio of both hMSC- and Ang-hMSC-treated groups was increased at 7 days after MCAO as compared with control (Figure 6E). The rCBF ratio in ROI-3 was similar for the three groups at 6 h, but again both hMSC- and Ang-hMSC-treated groups had a greater rCBF ratio at 3 and 7 days (Figure 6F). In ROI-4, the rCBF ratio slightly decreased in all groups at all time points, but not more than 10% (Figure 6G).
Functional Analysis
The maximum speed at which the rats could run on a motor-driven treadmill was recorded. BeforeMCAO, there was no significant difference among control (
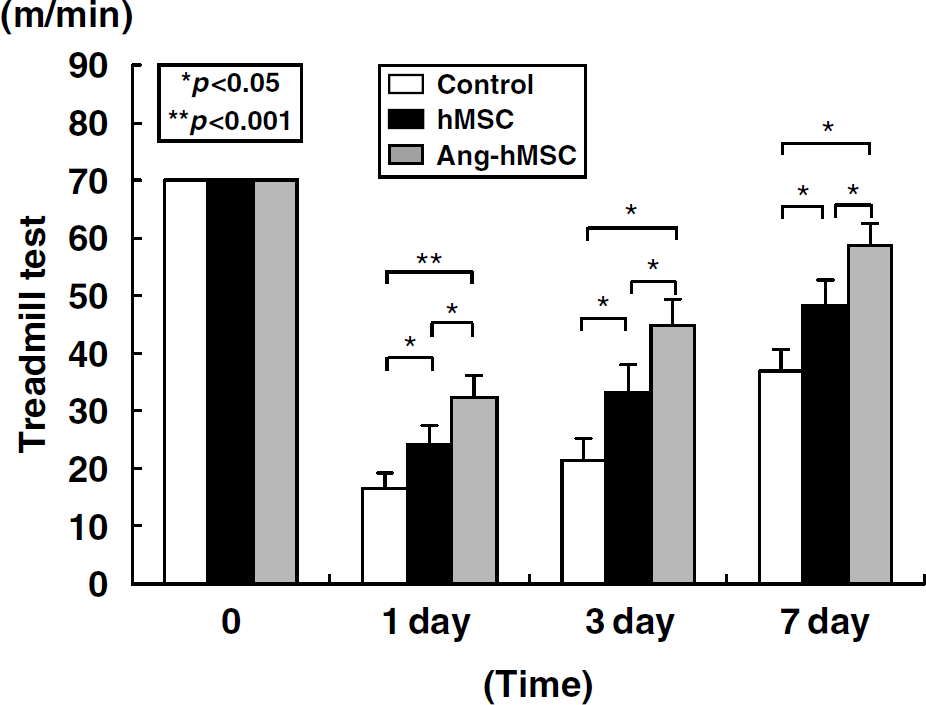
The treadmill stress test demonstrates that the maximum speed at which the rats could run on a motor-driven treadmill was faster in the hMSC- and the Ang-hMSC-treated rats than control. One day after MCAO, rats that received Ang-hMSC showed higher running speed compared with the medium alone and the hMSC group. From 3 days on after MCAO, the hMSC and Ang-hMSC groups attained higher velocities but the Ang-hMSC group was greater. The highest level of functional improvement was attained at 7 days.
Discussion
This study demonstrates that intravenous infusion of either hMSCs or Ang-hMSCs 6 h after permanent MCAO in the rat results in reduction in infarction volume, an induce in angiogenesis, and improvement in behavioral performance. These results are consistent with previous studies showing beneficial effects of bone marrow cell transplantation in experimental cerebral ischemic models (Chen et al, 2001; Iihoshi et al, 2004; Nomura et al, 2005; Honma et al, 2006; Horita et al, 2006; Kim et al, 2006; Liu et al, 2006; Ukai et al, 2007). Both hMSCs and Ang-hMSCs comparably reduced gross lesion volume as assayed with MRI and histology. Angiogenesis and rCBF were increased after infusion of either cell type, but were greater in small border areas after Ang-hMSC infusion. Functional recovery was also improved for both cell types, but modestly greater in the Ang-hMSC group. Thus, Ang-hMSCs do not effect lesion volume any more than hMSCs, but may facilitate small pockets of regional neovascularization and modest augumented functional recovery.
Both VEGF and Ang-1 play important roles in forming new vessels. Vascular endothelial growth factor has a role in enlarging small vessels (Pettersson et al, 2000). Although VEGF can initiate vessel formation, on its own it promotes formation of leaky, immature, and unstable vessels. Such destabilized vessels would be prone to regression in the absence of associated growth factors. In contrast, Ang-1 induces vessel sprouting and branching (Davis et al, 1996; Koblizek et al, 1998), and seemingly further stabilizes and protects the adult vasculature, makingit resistant to the damage and leak by VEGF or inflammatory challenges (Yancopoulos et al, 2000).
Vascular endothelial growth factor displays other effects such as an increase in the endothelium permeability (Bates et al, 2002). This effect could lead to vasogenic edema, particularly deleterious in cerebral ischemia. Indeed, early administration of VEGF after MCAO exacerbates blood—brain barrier leakage, causing massive brain edema (Zhang et al, 2000). Systemic delivery of Ang-1 by adenoviral gene delivery causes resistance to vascular leakage induced by VEGF in the brain (Zhang et al, 2002) and in the skin (Thurston et al, 1999).
Upregulation of endogenous Ang-1 in ischemic brain tissue was reported (Beck et al, 2000; Zhang et al, 2002), suggesting that Ang-1 might play an important role in endogenous repair in the ischemic lesion. However, therapeutic effects of exogenously applied Ang-1 on ischemic brain had not been studied. In this study, we applied Ang-1 by delivering hMSCs that hypersecrete Ang-1 to determine if Ang-1 would augment neovascularization in the post-ischemic brain and possibly improve functional outcome. Although in three-dimensional image analysis for angiogenesis, the Ang-hMSC-transplanted group showed small border areas of increased angiogenesis as compared with the hMSC transplanted and control groups, the effects were small and regional. Similarly, both hMSCs and Ang-hMSCs transplantation groups showed improvement of rCBF in the lesion, and only small areas of increased rCBF were observed in the Ang-hMSC transplantation group as compared with the hMSC group. No increase in cerebral edema was observed for either cell type as assayed by both
Cell-based therapeutic approaches are being considered for a number of neurologic diseases (Chopp et al, 2000; Pluchino et al, 2003; Iihoshi et al, 2004; Nomura et al, 2005; Pluchino et al, 2005; Zhang et al, 2005; Honma et al, 2006; Horita et al, 2006, Liu et al, 2006). Suggested mechanisms include reduction of inflammatory infiltration, elevation of trophic factors that may be neuroprotective, and structural repair such as remyelination (Radtke et al, 2007). A cell-based therapy may have the advantage of exerting multiple therapeutic effects at various sites and times within the lesion as the cells respond to a particular pathologic microenvironment.
Our results indicate that infusion of non-genetically modified hMSCs results in significant reduction in ischemic lesion volume and improved functional outcome, and Ang-hMSC had a modest additional influence on angiogenesis and functional outcome. Thus, hMSCs without genetic modification to induce additional neovascularization may be a useful source of cells for a cell-based therapeutic approach in clinical studies.
