Abstract
Functional neuroimaging and normal brain function rely on the robust coupling between neural activity and cerebral blood flow (CBF), that is neurovascular coupling. We examined neurovascular coupling in rat sensory cortex in response to direct stimulation of transcallosal pathways, which allows examination of brain regions inaccessible to peripheral stimulation techniques. Using laser-Doppler flowmetry to record CBF and electrophysiologic recordings of local field potentials (LFPs), we show an exponential relation between CBF responses and summed LFP amplitudes. Hemodynamic responses were dependent on glutamate receptor activation. CNQX, an AMPA receptor blocker, strongly attenuated evoked CBF responses and LFP amplitudes at all stimulation frequencies. In comparison, N-methyl
Keywords
Introduction
Functional brain imaging techniques, such as functional magnetic resonance imaging (fMRI), are based on the robust coupling between changes of neuronal activity and cerebral metabolism or blood flow (Raichle, 2003). A major limitation in the interpretation of, for example, fMRI data is a shortage of knowledge and understanding of the quantitative relation between local nerve cell activity and increases in cerebral blood flow (CBF) or oxygen and glucose consumption. Ideally, one should be able to define a ‘neurovascular’ or ‘hemodynamic’ transfer function that could provide a basis for predicting the level and type of local neuronal activity given a defined change in the vascular imaging or BOLD signal, and vice versa (Lauritzen, 2001; Devor et al, 2003; Jones et al, 2004). Not unexpectedly, the definition of such a transfer function has proven to be exceedingly complex. The spiking activity of activated neurons do correlate to the amplitude of the fMRI BOLD signal (Mukamel et al, 2005), but it is the Ca2+ dependent mechanisms in neurons and possibly astrocytes activated by synaptic neurotransmission that trigger and control the synthesis of vasodilators and in turn the amplitude of the hemodynamic responses and the BOLD contrast signal (Irikura et al, 1994; Lindauer et al, 1999; Ureshi et al, 2004; Iadecola, 2004; Lauritzen, 2005; Takano et al, 2006).
The majority of rodent studies of neurovascular coupling published so far have used stimulation of somatosensory afferents to evoke synaptic and vascular responses in the contralateral sensory cortex, but this limits the cortical regions that can be stimulated and studied (Austin et al, 2003; Enager et al, 2004). In comparison, we know very little about neurovascular coupling in relation to activity in neuronal networks intrinsic to the brain. In this study, we explored the characteristics of neurovascular coupling in a cortical region activated by stimulating the homologous contralateral cortex. We report that activity-dependent changes in synaptic activity and CBF evoked by stimulation of transcallosal pathways depend on interaction of glutamate with both AMPA and N-methyl
Materials and methods
Seventeen male Wistar rats (280 to 350 g) were anesthetized with Isofluorane (induction: 5%, surgery: 2%) while ventilated with 30% O2 and 70% N2O. The head was placed in a stereotactic head holder. Lidocaine (5 mg/mL) was used at the contact spots for the ear pins. A femoral artery catheter was inserted for measuring arterial blood pressure and taking blood-gas samples. A femoral vein catheter was inserted for drug infusion. After surgery, isofluorane was discontinued and anesthesia was maintained with α-chloralose (bolus: 45 mg/kg i.v., continuous infusion: 45 mg/kg/h i.v.). An intra-peritoneal catheter was used for injection of 7-nitroindazole (7-NI). The trachea was cannulated and the rats were ventilated and maintained throughout the experiment at pH 7.35 to 7.45, PaCO2 35 to 43 mm Hg, and PaO2 95 to 130 mm Hg (ABL 715, Radiometer). Body temperature was maintained at 36.7°C using a custom-made heating pad. Craniotomies were performed over homologous regions of the sensory cortex (1 to 5 mm posterior to bregma and 3 to 6 mm lateral to the midline) with a dental drill under continuous saline superfusion. Two wells were made around each craniotomy using 5% agar in Ringer solution. The dura was carefully removed under a microscope. Any visible sign of brain damage or bleeding excluded the rat from the protocols. During and after removal of the dura the craniotomy sites were continuously superfused with aCSF (composition in mmol/L: 2.0 glucose, 126.0 NaCl, 2.8 KCl, 22.0 NaHCO3, 1.45 CaCl2, 1.0 Na2HPO4, and 0.876 MgCl2). At the end of the experiment, the rats were killed by an i.v. injection of air. All experiments were in full compliance with the guidelines set forth in the European Council's Convention for the Protection of Vertebrate Animals Used for Experimental and other Scientific Purposes and were approved by the Danish National Ethics Committee.
Experimental Protocol
The experimental setup is shown in Figure 1. According to previous anatomic studies of the somatosensory cortex, transcallosal fibers are axons originating from medium to large-sized pyramidal cells located in layer III and terminating predominantly on dendritic spines of pyramidal cells in layer II, III, and V in the homologous contralateral cortex (White and Czeiger, 1991; Carr and Sesack, 1998). The transcallosal terminals form asymmetric synapses (Jacobson, 1966; Jones and Powell, 1968), which are generally accepted as being excitatory. Evidence of transcallosal monosynaptic inhibition is sparse. Postsynaptic intracellular recordings of cortical pyramidal cells activated by a callosal input have shown that the typical transcallosal response consists of an excitatory postsynaptic potential (EPSP) followed by a prolonged inhibitory postsynaptic potential (IPSP) (Innocenti, 1986). The source and the exact location of this inhibition is not entirely clear, but functional studies of transcallosal connectivity strongly suggest the existence of local inhibitory interneurons (Netz et al, 1997; Holloway et al, 2000; Dijkhuizen et al, 2001).
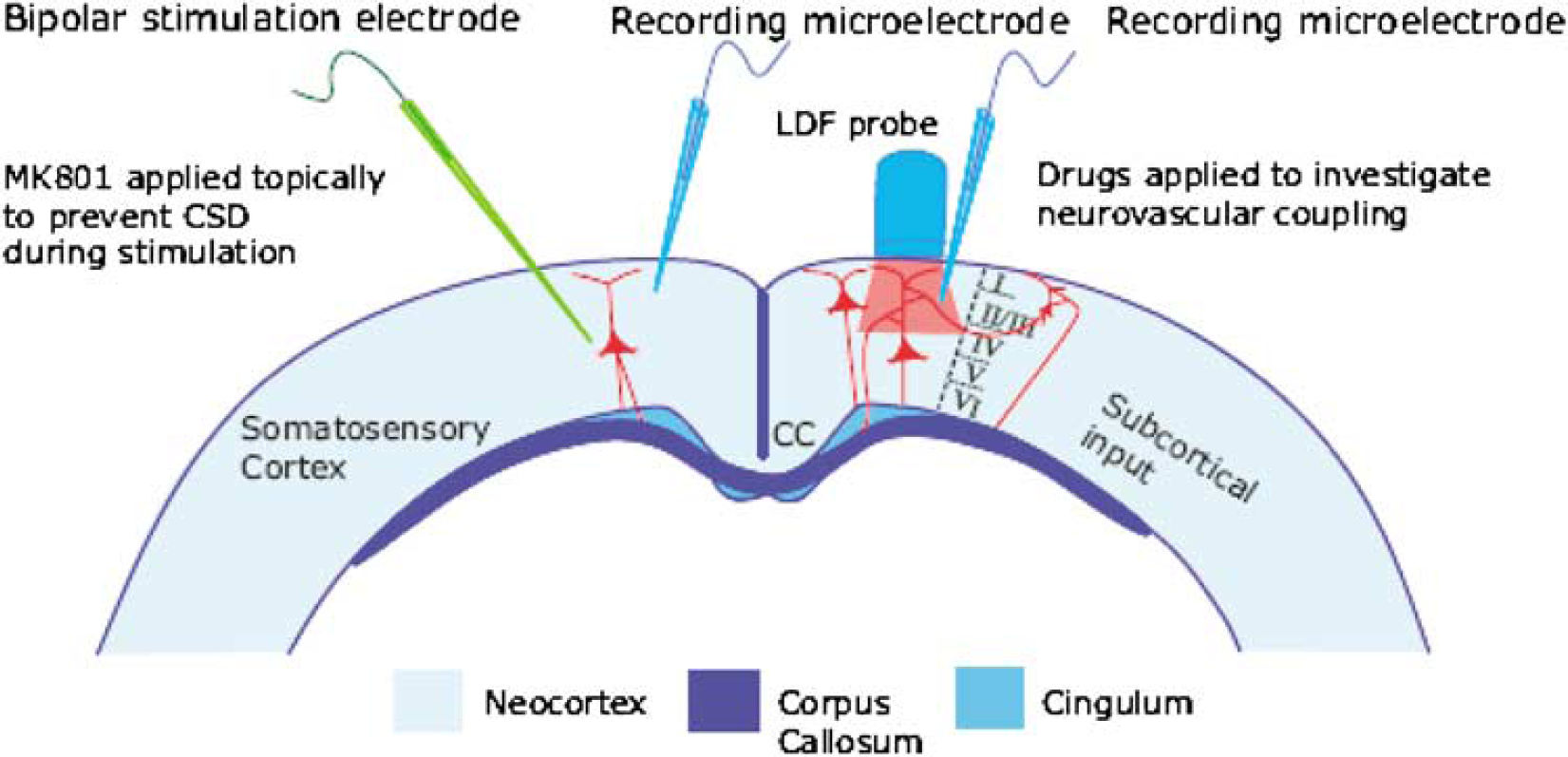
Experimental setup of transcallosal stimulation. Schematic two-dimensional drawing of experimental setup, including neurons of interest and placement of LDF probe, and of stimulating- and recording electrodes. A bipolar stimulating electrode was positioned in the region that was to be stimulated. The cortical surface was superfused with the NMDAR blocker MK801 to avoid generation of seizure activity and cortical spreading depression that would otherwise jeopardize the experiments. The protection of the stimulated side was essential for obtaining reproducible results, and monitored by a microelectrode positioned close to the stimulation electrode. On the contralateral side, in the homologous cortical region, LFPs were recorded by a glass microelectrode. Cerebral blood flow was recorded by LDF using a probe that was positioned at 0.3 to 0.5 mm above the pial surface. Drugs were applied on the activated side to study mechanisms of neurovascular coupling. Figure adapted from Figure 1 by Kumar and Huguenard (2001).
The present study was designed to investigate the coupling between neuronal activity and CBF in response to stimulation of transcallosal pathways. Four groups of rats were used (Tables 1 and 2). In the first experimental group (n=4), cortical AMPARs were blocked using CNQX for two reasons. First, to verify that the evoked CBF response was indeed due to local synaptic neurotransmission, and second, to carry out a correlation analysis of CBF responses and summed LFP amplitudes during partial blockade of synaptic excitation. In three animals, CNQX was subsequently washed out and the neurovascular responses regained.
Physiological variables before and after MK801 and CNQX application
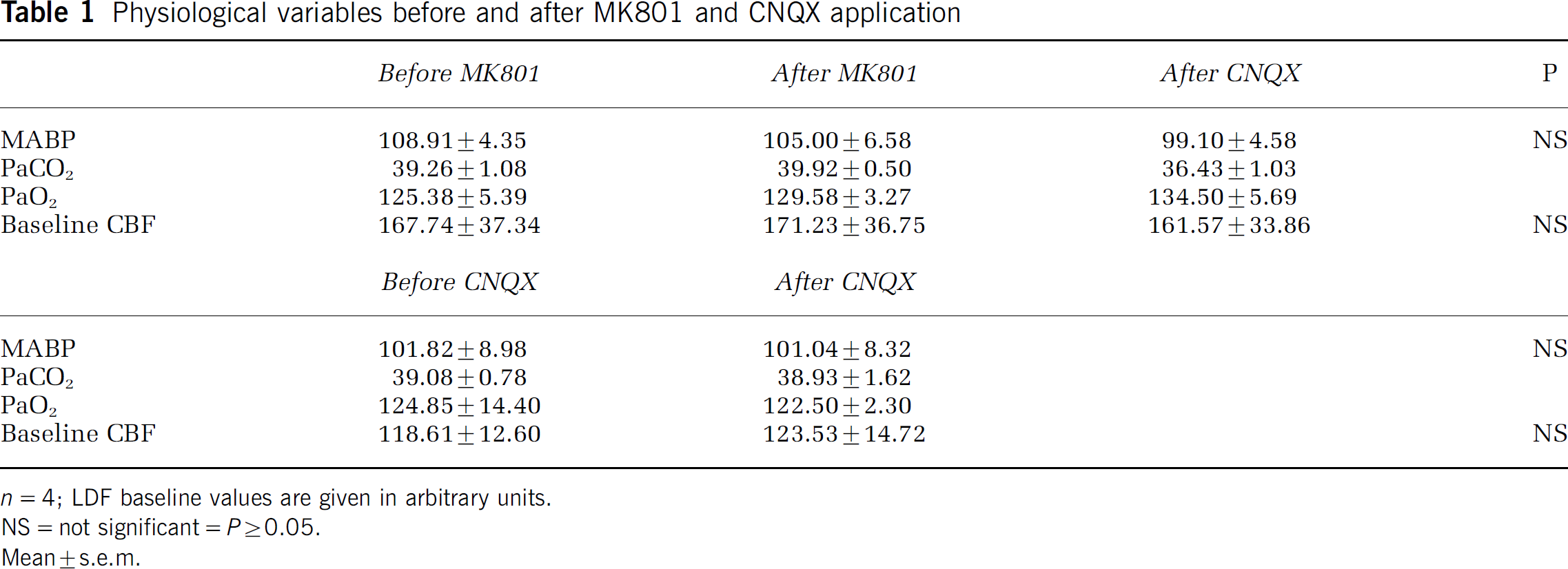
n=4; LDF baseline values are given in arbitrary units.
NS=not significant=P≥0.05.
Mean±s.e.m.
Physiological variables before and after 7-NI and IMC application
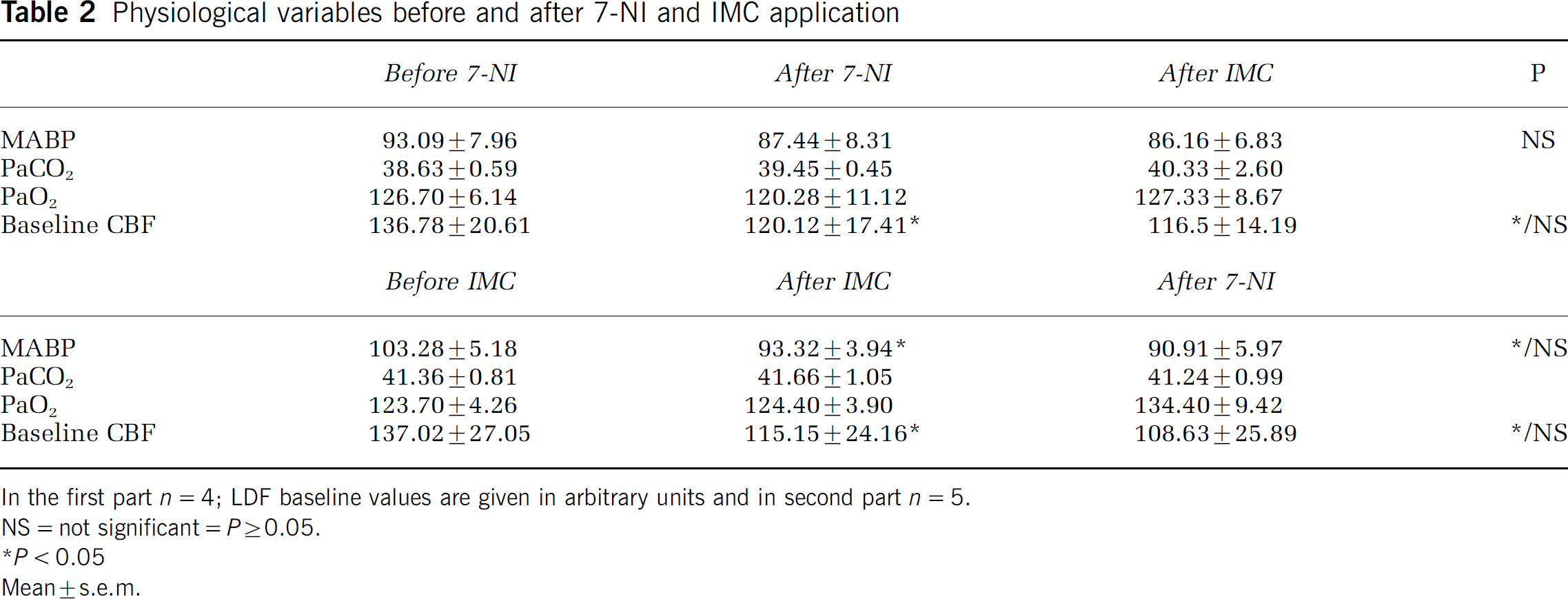
In the first part n=4; LDF baseline values are given in arbitrary units and in second part n=5.
NS=not significant=P≥0.05.
P<0.05
Mean±s.e.m.
In the second experimental group (n=4), we examined the dependence of the intrinsic neurovascular response on NMDAR activity by applying MK801, an NMDAR blocker. Subsequently, CNQX was applied to evaluate the combined effect of AMPAR and NMDAR blockade.
In the third and fourth experimental groups, we aimed to identify the nature of the vasodilators involved in neurovascular coupling of this neuronal circuit. In group 3 (n=4), we used 7-NI, a relatively specific blocker of neuronal NOS activity, and indomethacin (IMC), a nonspecific blocker of cyclooxygenase (COX), to attenuate activity-dependent CBF responses. The order of application of 7-NI and IMC was reversed in group 4 (n=5).
Stimulation of Transcallosal Fibers
A coated bipolar stainless-steel electrode (SNEX 200, contact separation: 0.25 mm; RMI, Woodland Hills, CA, USA) was lowered 0.5 mm into the right sensory cortex ∼3.5 mm lateral to the midline and ∼3.5 mm posterior to bregma using stereotactic instruments. Direct current stimulation (ISO-flex; A.M.P.I., Israel) was given as square-wave pulses (100 μs, 200 μA) at 1, 3, 5, 7, 10, 15 or 20 Hz in trains lasting 4 secs. Each frequency was run three times with interstimulus intervals of 1.5 mins to allow the re-establishment of a stable baseline between stimulations. In pilot studies, we observed that cortical stimulation often elicited episodes of cortical spreading depression (CSD) in the sensory cortex on the stimulated (right) side. To circumvent this problem, we applied the NMDAR antagonist MK801 to the stimulated cortex, which prevented the occurrence of CSD (Lauritzen and Hansen, 1992) and ensured reproducible LFPs and CBF responses measured in the activated (left) cortex.
Laser-Doppler Flowmetry
Cerebral blood flow was measured using a laser-Doppler flowmetry (LDF)-probe (PF 403: wave length 780 nm, fiber separation 250 μm; Perimed, Järfälla, Sweden) at a fixed position 0.3 to 0.5 mm above the cortical surface in an area devoid of large vessels. The geometry of the LDF probe allowed the measurement of CBF down to a depth of ∼1000 μm (Fabricius et al, 1997). Laser Doppler flowmetry measures CBF in arbitrary units (a.u.) and is therefore used for measuring relative changes in CBF. All signals were A/D-converted, smoothed with a time constant of 0.2 secs and digitally sampled at 200 Hz using the 1401-plus hardware (Cambridge Electronic Design, Cambridge, UK) connected to a pc running the SPIKE 2.3 software.
Electrophysiologic Recordings
Neuronal signals from the sensory cortex were recorded using single-barrel glass microelectrodes (pulled from capillary glass tubes) filled with 2 mol/L NaCl (electrical impedance: 2 to 3 MΩ, tip diameter: ∼2 μm). One microelectrode was inserted into the activated (left) sensory cortex at a depth of 300 to 400 μm and used to record local field potentials (LFPs) produced by transcallosal stimulation. A second glass microelectrode was inserted into the stimulated (right) sensory cortex to continuously monitor the electrocorticogram (ECoG; Figure 1). An Ag/AgCl ground electrode was implanted in the neck muscle and used as a reference for both of these recordings. The signals were A/D-converted, amplified, band pass filtered at 0.5 to 1000 Hz, and digitized using the 1401-plus hardware (Cambridge Electronic Design, Cambridge, UK) connected to a PC running the SPIKE 2.3 software. Digital sampling rates for LFPs and ECoGs were 10 kHz.
Data Analysis
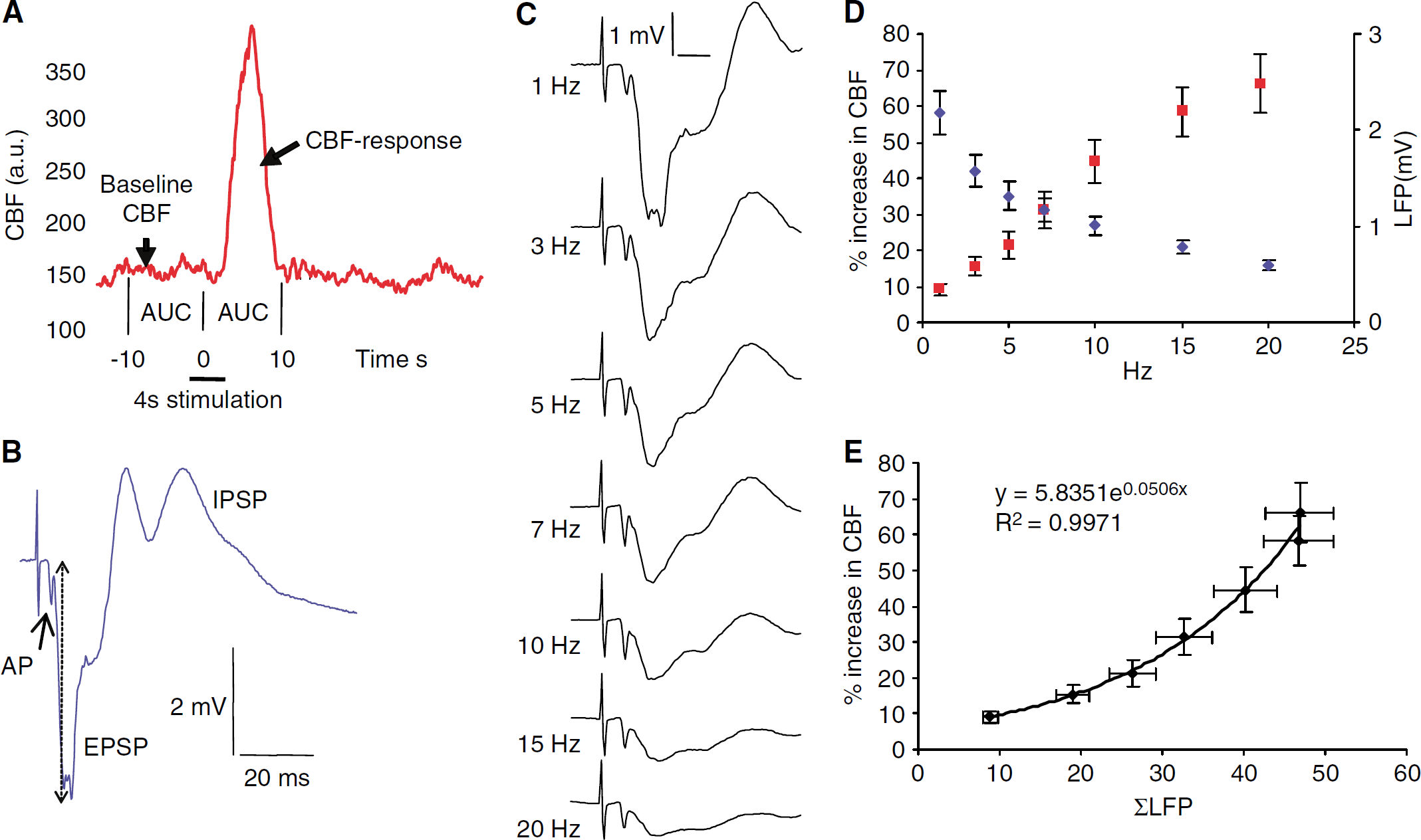
Local field potential and CBF responses were averaged for each stimulation frequency and treatment. The averaged responses were analyzed as described below and used for statistical calculations. Cerebral blood flow responses were calculated as area under curve (AUC) for 10 secs from the start of the stimulation train and expressed as percent of baseline, defined as AUC for the 10 secs immediately preceding stimulation (Figure 2A). The time limits of 10 secs always contained the full CBF response.
Nonlinear relation between ΣLFP and CBF responses evoked by transcallosal stimulation. (
Local field potential amplitudes were calculated as the difference between peak and baseline. Local field potential baseline was defined as the average potential measured during the 15 ms interval before a stimulation impulse was given. We examined the coupling between CBF and synaptic activity by correlating the increase in CBF to the sum of LFP amplitudes (ΣLFP=LFP amplitude (mV) × stimulation frequency (Hz) × stimulation duration (4 secs)).
Drugs
The selective and noncompetitive NMDAR antagonist (5R,10S)-(5-methyl-10,11-dihydro-5H-dibenzo[a,d]cyclohepten 5,10-imine/dizocilpine (MK801, Sigma) was dissolved in aCSF, kept at 37°C and bubbled with 95% air/5% CO2 until pH reached 7.3 to 7.45. A half an hour before the stimulation electrode was inserted into the right cortex, MK801 (1 mmol/L) was applied topically to this side and allowed to work for 1 h before any stimulation was begun. MK801 was also applied to the left cortex in those experiments in which the contribution of NMDAR to the evoked synaptic and CBF responses was examined. In these experiments, MK801 was allowed to work for 1 h before data collection.
The AMPAR antagonist 6-cyano-7-nitroquinoxaline-2.3-dione (CNQX, Sigma) was also dissolved in aCSF (0.5 mmol/L), bubbled with 95% air/5% CO2 and applied topically to the left (activated) cortex to examine the effect of AMPAR antagonism on the evoked responses. The effect was evaluated continuously and was maximal after 15 mins.
The NO synthase inhibitor 7-NI was dissolved in 3 ml 5% Cremophor (polyoxyethylene glycol; BASF) in saline, ultrasonicated for 20 min and applied slowly through an intraperitoneal catheter (40 mg/kg body weight). Indomethacin was dissolved in saline and administered as an i.v. bolus (5 mg/kg body weight) followed 30 mins later by a continuous infusion (6.25 mg/kg body weight/h; 1.25 mL/h). After injection of each substance, we used repeated transcallosal stimulation at 10 Hz to identify the time at which the attenuation of CBF responses was maximal and stable (for 7-NI: 10 to 15 mins, for IMC: ∼10 mins).
Statistics
Three-way ANOVA was used to compare CBF, LFP- and ΣLFP changes before and after drug application. Statistics were performed on log-transformed data. A Mixed Model analysis (SAS software, SAS Institute Inc., Cary, NC, USA) was used to compare additional effects of MK801 on CBF when applied together with CNQX versus CNQX alone. The slopes of the linear relationships between LFP amplitude and stimulus frequency before and in the presence of CNQX were compared using student's paired t-test. Changes in physiologic variables were also evaluated by Student's paired t-test. Values were considered statistically significant at P<0.05. All values are given as mean±s.e.m.; n is the number of rats in each experiment.
Results
Effect of MK801, CNQX, 7-Nitroindazole, and Indomethacin on Physiologic Variables
MK801 and CNQX had no significant effects on MABP, PaCO2, PaO2, and baseline CBF (Tables 1 and 2), while injection of both 7-NI and IMC significantly lowered baseline CBF by 12.2% and 16.0%, respectively. In addition, IMC had a significant effect on MABP, lowering it from 103.28±5.18 to 93.32±3.94 mm Hg. Injection of IMC after 7-NI application had no further effect on MABP. The shifts in CBF baseline because of 7-NI and IMC were uncorrelated to PaCO2, PaO2 and MABP.
Local field potential and Cerebral Blood Flow Responses Evoked by Stimulation of Callosal Fibers
Electrical stimulation of the right sensory cortex evoked reproducible frequency-dependent LFPs and hemodynamic responses in the contralateral homologous somatosensory cortex. The evoked LFP consisted of an action potential and a postsynaptic component. The action potential indicated the arrival of nerve impulses via the callosal fibers within 4 ms of stimulation of the right sensory cortex (Figures 2B and 2C). This response was clearly discernible in half of the animals. In the rest, it was observed as a notch in the downward slope of the postsynaptic LFP or not visible at all. The postsynaptic component was biphasic, consisting of an initial negative potential indicating synaptic excitation (EPSP) and a longer-lasting positive potential indicating synaptic inhibition (IPSP). The amplitude of the EPSP was maximal 9 to 11 ms after cortical stimulation and decreased with increasing stimulus frequencies (Figure 2C), which is ascribed to the shorter recovery times between stimulation impulses at higher frequencies (Creutzfeldt, 1995). In comparison, the amplitude of the presynaptic component was constant at all stimulation frequencies. In the following, LFP refers only to the EPSP. Stimulation evoked increases in CBF with a time latency of 1 to 1.5 secs and after cessation of stimulation, CBF decreased with a similar delay, suggesting a time constant of approximately 1 sec (Figure 2A). Cerebral blood flow responses increased as a function of stimulus frequency while LFP amplitudes decreased (Figure 2D (n=17)). To explore the relation between synaptic activity and CBF, we correlated the summation of LFP amplitudes, ΣLFP, to CBF responses (ΔCBF) as described previously (Mathiesen et al, 1998). The results shown in Figure 2E, indicated an exponential relation between ΣLFP and ΔCBF (n=17).
Effect of CNQX and MK801 on Cerebral Blood Flow and Local Field Potential Responses
The neural origin of the CBF responses was verified using local glutamate receptor blockade. Topical application of 0.5 mmol/L CNQX onto the activated left cortex attenuated both CBF and LFP responses (Figure 3). This attenuation was maximal after 15 mins, remaining so until washout. The effect of CNQX was fully reversible, and during the subsequent recovery period, LFP amplitudes and CBF responses returned to normal concurrently (Figures 3A and 3B (n=3)). CNQX abolished LFP amplitudes at all stimulation frequencies—the maximal attenuation of LFP amplitudes and CBF responses was 96.5%±0.8% and 77.5%±1.3%, respectively (both P<0.01; Figure 3E, n=4).
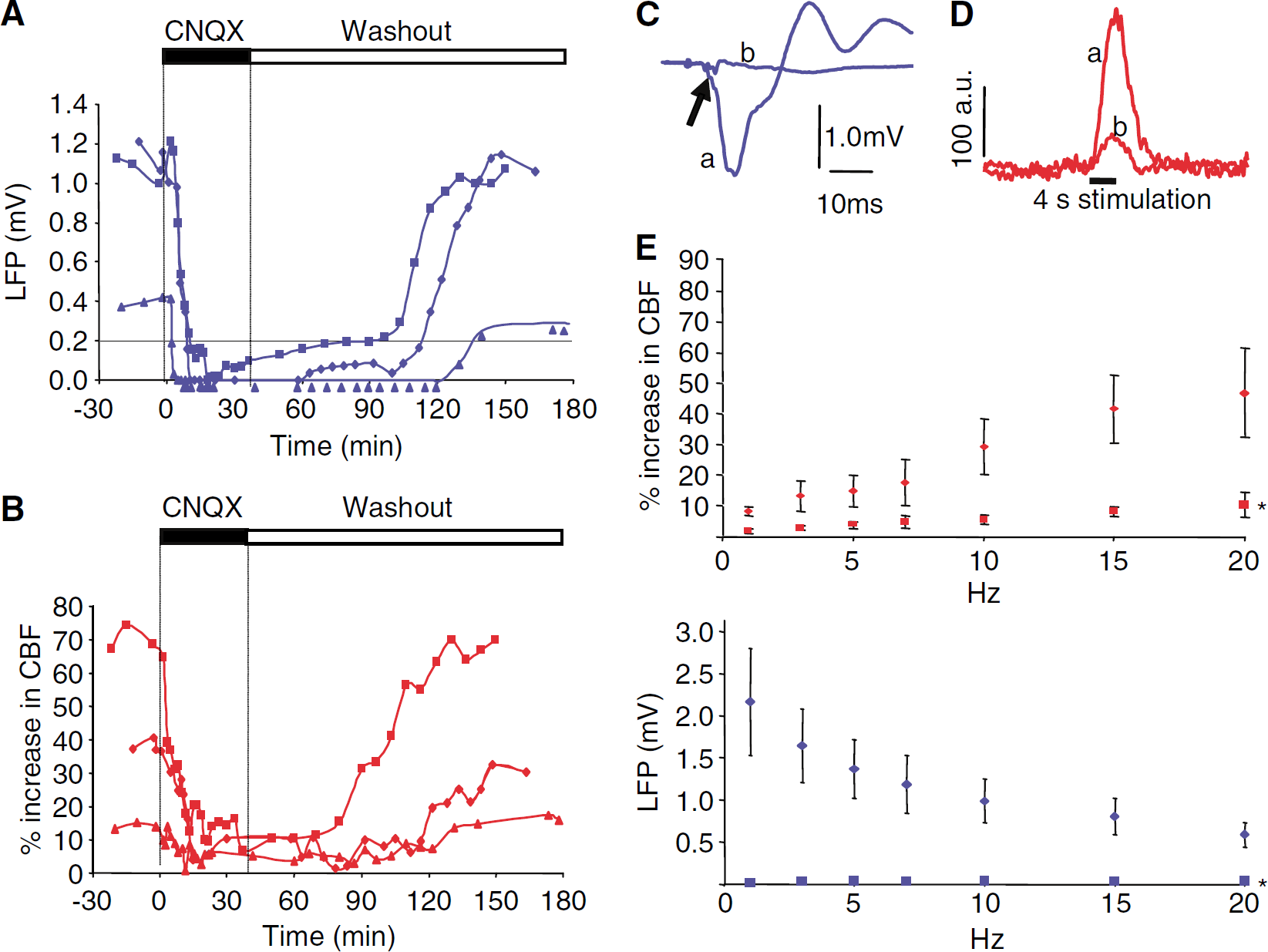
Proportional attenuation of LFP and CBF response amplitudes by AMPAR blockade (
Topical application of 1 mol/L MK801 significantly (P<0.01) reduced CBF responses to transcallosal stimulation at frequencies above 7 Hz, that is, 10, 15 and 20 Hz (Figure 4C). In contrast, LFP amplitudes remained constant after application of MK801 at all stimulation frequencies (Figures 4A and 4B). The time latency to the onset and the profile of the LFP recordings were unchanged after MK801 (Figure 4B), suggesting that the transmembrane ionic fluxes associated with activation of NMDARs are too small to be detectable by extracellular microelectrode recordings.
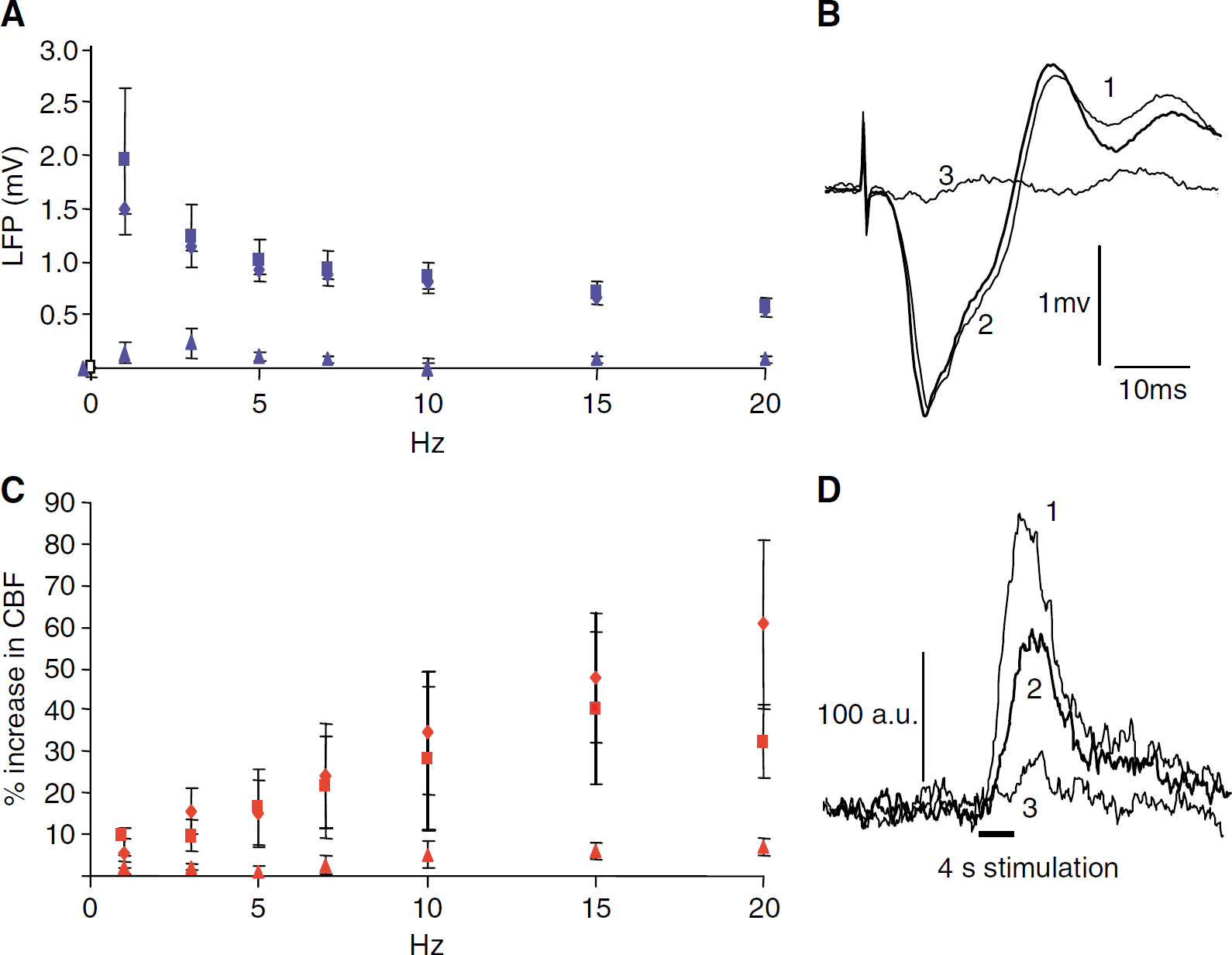
Attenuation of CBF, but not LFP response amplitudes by NMDAR blockade. (
The results from experiments using glutamate receptor blockers are summarized in Figure 5, which shows an exponential correlation between ΣLFP and CBF under control conditions (n=8). After MK801 application, the slope was reduced while the exponential correlation between ΣLFP and CBF was preserved (see figure). In comparison, CBF and ΣLFP responses were reduced proportionally by CNQX, both when applied alone (Figure 3E) and in the presence of MK801 (Figures 4A and 4C).
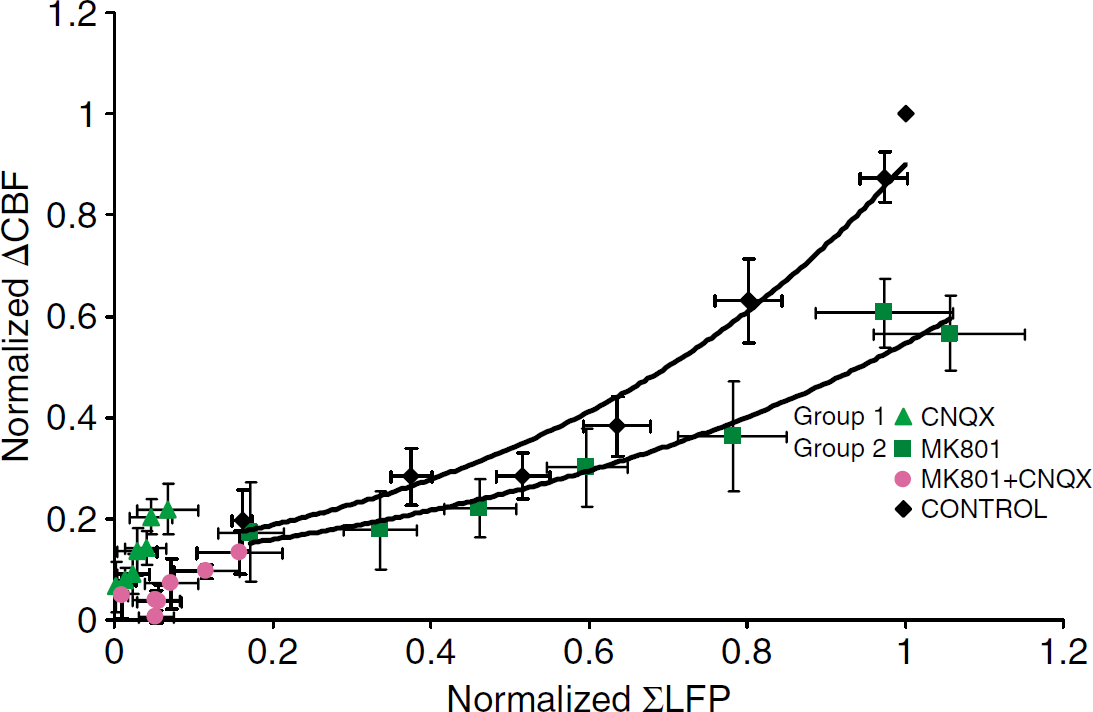
Summary of experiments using CNQX, MK801, and CNQX and MK801 combined on correlation between ΣLFP and CBF. The figure illustrates the effect of MK801 and CNQX on neurovascular coupling. Data were normalized to values obtained at 15 Hz stimulation. Each dot represents the mean averaged changes in ΣLFP and CBF responses for each stimulation frequency. The figure shows that neurovascular coupling for the control data (CNQX or MK801, n=8). An exponential function gave the best fit for the control group; Y=0.1266e1.9623X, R2=0.9604. MK801 reduced the CBF responses without a corresponding decrease in LFP amplitude. The attenuation of CBF responses was significant at 10, 15 and 20 Hz. The best fit was exponential; MK801: Y=0.1167e1.5423X, R2=0.961. CNQX strongly attenuated both CBF and LFP responses. There was no additional effect of MK801 in rats treated with CNQX. Black indicates control condition, dark green indicates responses after MK801, light green indicates responses after CNQX, pink indicates responses after MK801+CNQX.
Effect of 7-Nitroindazole and Indomethacin on Cerebral Blood Flow Responses and Local Field Potentials
Nitric oxide and COX products are powerful vasodilators and important for neurovascular coupling (Dirnagl et al, 1993; Akgoren et al, 1994; Cholet et al, 1996; Niwa et al, 2000, 2001). Separately, 7-NI and IMC significantly reduced stimulation-evoked CBF responses by 49.6%±1.4% and 47.9%±1.4% at all frequencies (both: P<0.01; Figure 6A). Latency time and profile of the CBF response were unchanged after both 7-NI and IMC application (data not shown). A further reduction in CBF response of ∼30% was seen after application of 7-NI and IMC together (P<0.01) (Figure 6A). Local field potential amplitudes were unchanged by IMC and 7-NI (Figure 6B; IMC: P=0.402; 7NI: P=0.3057). Figures 6C and 6D shows that the neurovascular coupling remained nonlinear after drug injection, and that IMC and 7-NI reduced CBF without affecting ΣLFP. The additive effect of the drugs suggests that NO and arachidonic acid derivatives induced by stimulation exert their vasodilator effects partly via separate pathways.
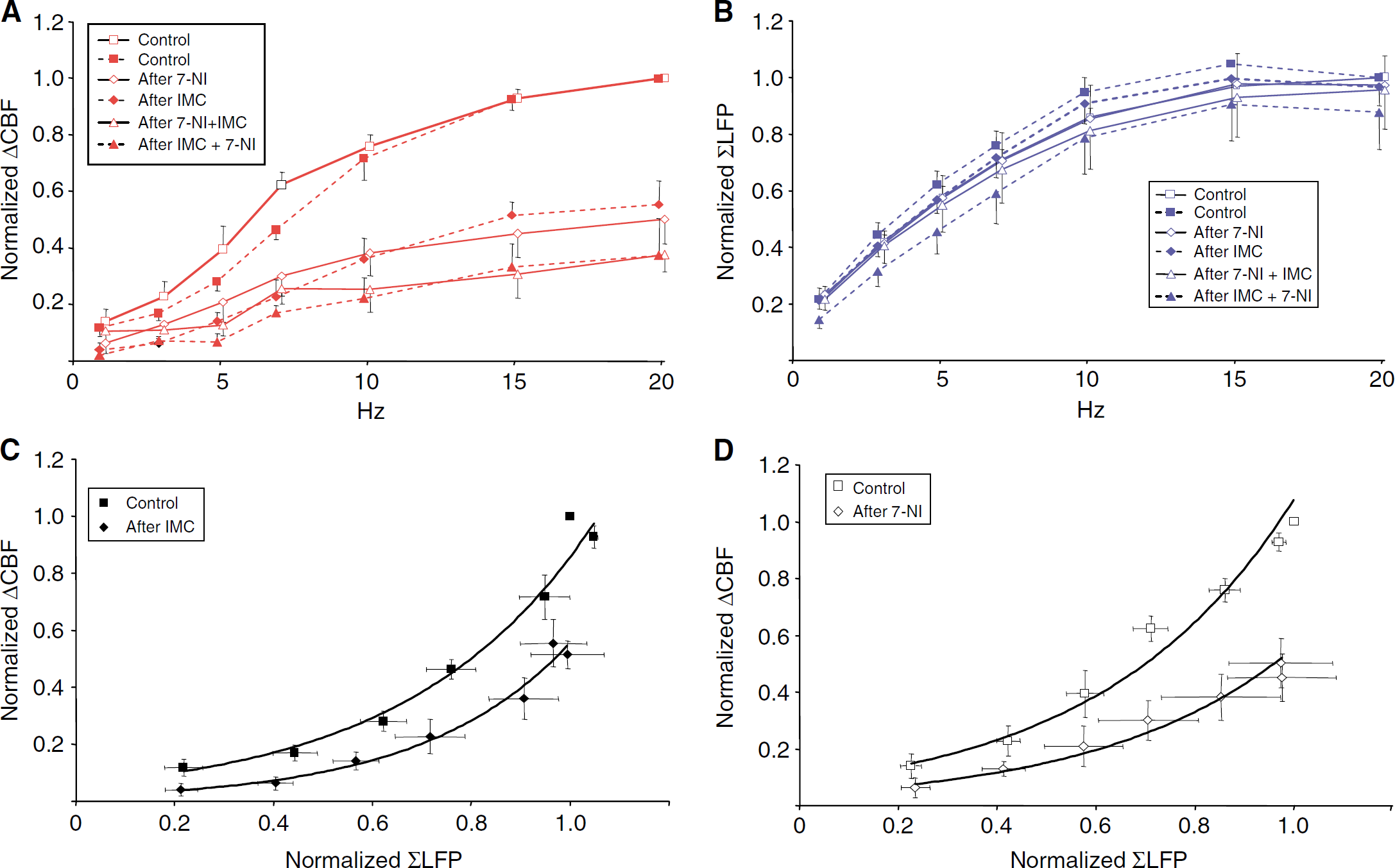
Mechanisms of rises in CBF evoked by transcallosal stimulation: effect of NOS- and COX-inhibition. (
Discussion
Stimulation of transcallosal fibers in the cerebral cortex of one hemisphere caused a frequency dependent postsynaptic response with a 4 ms transmission delay in the homologous brain region on the opposite side and a corresponding rise in CBF. This compares favorably with previously described interhemispheric conduction delays through corticocortical networks (Innocenti et al, 1995), and a previous study using direct cortical stimulation and fMRI in rats (Austin et al, 2003). Our pilot series of experiments were disturbed by elicitation of episodes of CSD on the stimulated side, which made it difficult to obtain reliable synaptic and vascular responses in the activated cortex. Application of NMDAR antagonists to the site of stimulation prevented the occurrence of CSD (Lauritzen and Hansen, 1992), and after this regimen was instituted, we had no difficulties in producing robust and reliable signals in the activated cortex.
Characteristics of Synaptic and Vascular Responses in Relation to Glutamate Receptor Activity
The evoked LFP consisted of an EPSP followed by a prolonged biphasic IPSP (Figure 2B). The LFP and CBF responses were greatly attenuated by CNQX (Figures 3A and 3B) indicating dependency on AMPAR activation (Kawaguchi, 1992). Both responses returned to normal after a washout period (Figures 3A and 3B). The decrease in LFP and CBF amplitudes by 95% and 75%, respectively, after CNQX corresponds well with our previous study of neurovascular coupling in the sensory cortex by stimulation of somatosensory afferents in vivo (Nielsen and Lauritzen, 2001).
NMDAR became increasingly involved in CBF responses at stimulation frequencies at or above 7 Hz. We speculate that depolarization by AMPAR activation may have to reach a certain threshold before the voltage-dependent Mg2+-block of NMDAR can be released and activation of NMDAR can occur. This condition could theoretically be met by stimulation at high frequencies. This view is supported by the observation that single pulse stimulation of callosal fibers in vitro activates NMDAR in the presence of picrotoxin, which blocks the effect of GABAA receptor activation (Kumar and Huguenard, 2001). It is also possible that NMDAR on aspiny inhibitory interneurons open in response to high-frequency stimulation and that it is the Ca2+ influx into this cell type that triggers the synthesis of vasoactive substances (Kawaguchi, 1992; Kumar and Huguenard, 2001; Vogt and Gorman, 1982; Netz et al, 1997; Carr and Sesack, 1998). Nitric oxide synthase and COX are both present in pyramidal cells (Kaufmann et al, 1996; Kaufmann et al, 1997; Wang et al, 2005), while interneurons are rich in NOS (Cauli et al, 2004). Therefore, a Ca2+ influx in either cell type would lead to synthesis of active vasodilators and an increase in CBF (Lauritzen, 2005).
The data are consistent with previously published in vivo experiments obtained in rat sensory cortex, which showed preserved cortical LFP amplitudes by stimulation of the infraorbital nerve in face of NMDAR blockade (Nielsen and Lauritzen, 2001). The biphasic IPSP that follows the initial EPSP is produced by activity of local inhibitory interneurons and not by release of GABA from callosal fibers (Vogt and Gorman, 1982; Carr and Sesack, 1998), since both the EPSP and the IPSP responses disappeared after local treatment with CNQX. We did not attain electrophysiologic evidence of NMDAR activity, which is difficult to record because the response is small and delayed, compared with the early and large AMPAR response. GABAA receptor antagonists may facilitate the recording of NMDAR responses, but this approach was not tested here because abolition of GABAergic tone in the cerebral cortex creates other problems such as epilepsy (Kawaguchi, 1992; Kumar and Huguenard, 2001). Both the NMDAR- and the GABA receptor-signal (Kumar and Huguenard, 2001) closely followed the EPSP, their responses are temporally and quantitatively mixed. This effectively masks the late depolarizing NMDAR signal. As there was no effect of NMDAR block on the amplitude of the EPSP at any stimulation frequency, we conclude that the short-latency EPSP evoked by transcallosal stimulation is mediated via AMPAR. In the following, we use the amplitude of the EPSP as indication of synaptic activity well aware that this only reflects activity at AMPARs.
The Neurovascular Coupling: Linearity Versus Nonlinearity
Activity-dependent rises in CBF have been shown to correlate tightly to changes in the activity of postsynaptic cellular elements as reflected in LFP amplitudes. The majority of studies suggest that the relation between LFP and hemodynamic response amplitudes is nonlinear (Mathiesen et al, 1998, 2000; Ngai et al, 1999; Logothetis et al, 2001; Miller et al, 2001; Sheth et al, 2004; Nemoto et al, 2004; Jones et al, 2004; Kayser et al, 2004). Our results showed that neurovascular coupling in the somatosensory cortex in response to transcallosal stimulation was better described by an exponential than by a linear function (Figures 5 and 6C and 6D). Our study was well suited to detect any nonlinearity because of the use of a wide range of stimulation frequencies coupled to fixed stimulation intensity that was expected to cover the entire dynamic range for both neuronal activation and CBF responses. That the entire dynamic range was covered with the stimulation frequencies used can be illustrated by the fact that the applied stimulation frequencies evoked both low and maximal ΣLFP amplitudes, and CBF responses both near threshold and at high levels. Our results seem to highlight the limitations of LFP amplitude measurements in the analysis of neurovascular coupling. Local field potentials are selective indicators of neural activity, requiring parallel generators, synchronous activity, spatial homogeneity of the synchronous parallel generators, large populations of active units and transmembrane ionic fluxes of sufficient magnitude to give rise to a measurable extracellular potential difference. We suggest that activation of NMDAR or other receptors that do not give rise to a measurable LFP, such as metabotropic glutamate receptors on glial cells or neurons, may explain part of the nonlinearity observed in this and other studies that correlate ΣLFP and hemodynamic variables.
Nitric Oxide and Cyclooxygenase-products Uncouple Evoked Cerebral Blood Flow Responses Through Different, but Related Pathways
Our study provides evidence that NO contributes both to the basal cerebrovascular tone and to CBF responses evoked by stimulation. Nitric oxide synthase inhibition reduced baseline CBF by ∼12%, and CBF responses to all stimulation frequencies by ∼50%. Cerebral blood flow reductions of this magnitude compare well with other studies using nNOS inhibition (Dirnagl et al, 1993). Neuronal NO synthesis may be associated with NMDAR activation (Faraci and Breese, 1993; Pelligrino et al, 1996; Bhardwaj et al, 2000). The NMDAR has been linked physically to neuronal NOS (nNOS) through specific protein structures (PSD-95) at the postsynaptic excitatory synapse, facilitating nNOS activation by NMDAR mediated calcium influx (Brenman et al, 1996; Sattler et al, 1999). It has been shown that separation of nNOS from PSD-95 inhibited the NMDAR dependent NO synthesis without affecting the general nNOS function (Sattler et al, 1999). Our experiments suggested that NOS inhibition reduced CBF responses equally at all stimulation frequencies (Figure 6A), while NMDAR blockade reduced the CBF responses only at high frequencies (Figure 5). This suggests that nNOS activity may be stimulated by mechanisms other than Ca2+ influx via NMDAR. This could involve gating of Ca2+-permeable AMPARs or opening of voltage-sensitive Ca2+ channels in dendrites of activated pyramidal cells (Tsay and Yuste, 2004) or interneurons (Goldberg et al, 2003a, 2003b; Goldberg and Yuste, 2005) that contain both NOS and COX activity (Kaufmann et al, 1996, 1997; Cauli et al, 2004; Wang et al, 2005). 7-Nitroindazole had no influence on LFP amplitude in accordance with previous experiments on somatosensory evoked potentials (Lindauer et al, 1996) and cerebral glucose consumption (Cholet et al, 1997), suggesting that neural activity is unaffected by 7-NI.
An arachidonic acid derivative contributes to the regulation of both basal blood flow and CBF responses to stimulation. We observed that IMC reduced baseline CBF by approximately 16%, while activity-dependent increases in CBF at all stimulation frequencies decreased by 48%. This is consistent with studies of other neuronal circuits (Dahlgren et al, 1984; Niwa et al, 2000; Bakalova et al, 2002a, 2002b). Indomethacin is a nonselective COX-inhibitor. Two distinct isoforms of COX have been identified (Hawkey, 1999). Cyclooxygenase-1 is constitutively expressed as a ‘housekeeping’ enzyme in most tissues while COX-2 is inducible. In the brain, both COX-1 and COX-2 are constitutively expressed. COX-1 inhibition has been shown to selectively reduce resting CBF and the rise in CBF associated with hypercapnia, bradykinin and the application of a calcium ionophore, but not activity-dependent rises in CBF. Thus, the importance of COX-1 is to maintain the basal cerebrovascular tone (Niwa et al, 2001). Cyclooxygenase-2 is localized, to certain populations of interneurons, and to excitatory neurons, such as glutamatergic pyramidal cells where dense concentrations are found in dendritic aborizations and spines especially in cortical layers II and III (Kaufmann et al, 1996; Wang et al, 2005), and to astrocytes (Zonta et al, 2003a). Selective inhibition of COX-2 attenuates CBF responses to somatosensory stimulation (Niwa et al, 2000; Bakalova et al, 2002a). We found that the CBF responses to stimulation were attenuated before baseline CBF was reduced (data not shown), which is consistent with distinct roles for COX1 and COX-2 in the cerebral circulation. Alternatively, one might consider an astrocytic pathway (Zonta et al, 2003b; Nedergaard et al, 2003) as glutamate and GABA released from neurons may produce calcium waves in astrocytes that travel to the perivascular end-feet. The rise in astrocytic Ca2+ may be transformed into the synthesis of an arachidonic acid derivative, leading to vasodilatation in brain slices (Zonta et al, 2003b) and in vivo (Takano et al, 2006), which can be blocked by COX inhibitors. The fact that the combined blockade of the ionotropic glutamate receptors virtually abolished the CBF response speaks however against this idea, since activation of metabotropic glutamate receptors that are believed to mediate the activity-dependent blood flow response mediated by astrocytes (Zonta et al, 2003a) is expected to be preserved and unaffected by AMPAR and NMDAR antagonists, and it was not.
Combined, 7-NI and IMC attenuated CBF responses by approximately 65% (P<0.01) (Figure 6A) indicating that no singular pathway is totally dependent on activity of the other to exert its effect during functional hyperemia. Other dual drug application paradigms have reached comparable values of CBF attenuation (Golanov and Reis, 1994). Interestingly, we observed no further drop in baseline CBF or MABP as a consequence of injection of the second drug (Table 2), indicating either one common pathway or two separate pathways that depend on the activity of the other to exert further vasodilatation.
In conclusion, we found that stimulation of transcallosal fibers produce robust electrophysiologic and hemodynamic responses in contralateral cortical regions provided that elicitation of CSD on the stimulated side is blocked. We suggest that this stimulation technique can be used to study local functional properties of the cerebral cortex or of other projecting pathways at examiners own choice, that is, independent of accessibility of peripheral stimulation techniques, as suggested by others as well (Austin et al, 2003). Stimulation of transcallosal fibers activated both glutamate AMPAR and NMDAR in the contralateral cortex, and activity of both types of glutamate receptors was necessary to obtain the full CBF response. Blocking NMDARs decreased the CBF responses, but were unaccompanied by changes in the amplitude of the LFP amplitude responses, while blockade of AMPA receptors caused a proportional decrease in LFP and CBF response amplitudes. The observation suggests putative limitations in using LFP as input function for a hemodynamic transfer function. Studies using correlation analysis of LFP versus vascular/hemodynamic response amplitudes will need to take this shortcoming into account. The observation highlights the theory that it is not the extracellular ionic current flow produced by synaptic excitation that produces the vascular response in brain cortex, but Ca2+ signaling in postsynaptic cellular elements.
Footnotes
Acknowledgements
The authors wish to thank Ms Lillian Groendahl for expert laboratory assistance.
