Abstract
Neuroimaging studies in right-handed patients with left hemisphere brain lesions have demonstrated a shift of language activity from left to right inferior frontal gyrus (IFG). This shift may be caused by greater right hemisphere dominance before the injury or by reduced inhibitory activity of the injured left hemisphere. We simulated a brain lesion applying transcranial-magnetic stimulation over left IFG in normal subjects, while simultaneously measuring language activity with positron — emission tomography. Interference with transcranial-magnetic stimulation decreased activity in left and increased it in right IFG in all subjects. We thus demonstrate for the first time that a rightward shift of language activity is caused by the brain lesion and not by greater right-hemisphere dominance, thus supporting the hypothesis of reduced transcallosal inhibition.
Introduction
In recent years, neuroimaging studies of language networks in patients with lesions of the left language-dominant hemisphere have been brought into focus of neuroplasticity research. A common finding of many studies was the recruitment of the right inferior frontal gyrus (IFG) while performing language-related tasks in cases where the left IFG (known as Broca's Area) has been affected by a brain lesion. This right-sided language-related brain activity has been observed in right-handed patients after stroke (Perani et al, 2003) as well as in patients with brain tumours (Thiel et al, 2001). Recent studies, combining functional neuroimaging and repetitive transcranial magnetic stimulation (rTMS) in these patients have demonstrated that this right-sided language-related activity is indeed essential for the observed performance in verbal fluency tasks. The extent of its contribution however, differs between acute brain lesions like stroke (Winhuisen et al, 2005), where it is rather limited, as compared with chronic brain lesions like brain tumours (Thiel et al, 2005a) where it makes near normal language performance possible at least in some patients.
It is however, still a matter of debate whether this right-sided language activity is because of a greater right-hemisphere language function before the onset of the brain lesion or whether it is caused by the brain lesion itself. In the latter case the phenomenon should be mediated by a decrease of inhibitory activity on the right hemisphere, a mechanism called transcallosal or interhemispheric inhibition (Netz et al, 1995; Netz, 1999).
Since the exact hemispheric dominance in these patients before the brain lesion is unknown, only indirect evidence can so far be referenced to answer this question. In a large screening study with transcranial Doppler sonography Knecht et al (2000) report right-hemisphere dominance in 7.5% of right-handed normal subjects and were able to show that the susceptibility to left hemisphere brain lesions depends on the extent of language lateralization to the left hemisphere (Knecht et al, 2002) thus underlining the importance of prelesional dominance. The incidence of right-sided language dominance in right-handed patients with brain lesions however ranges between 14% and 20% (Thiel et al, 2001, 2005a; Winhuisen et al, 2005) and is thus approximately twice as high as in the normal population, indicating that the lesion-effect might also be associated with right-sided language function.
We designed the present study to test this hypothesis. In six normal right-handed subjects images of relative cerebral blood flow (rCBF) were acquired using O-15-water positron emission tomography (PET) during rest and while performing a verbal fluency task. Half of the scans were performed while interfering with language function of the left IFG by rTMS, thus simulating a left hemisphere brain lesion. If the recruitment of the right IFG is caused more by the brain lesion than by language dominance, an increase in rCBF of the right IFG should be expected during rTMS, according to the hypothesis of reduced transcallosal inhibition.
Subjects and methods
Subjects
Six right-handed male subjects aged 25 to 34 years without any history of prior neurologic or psychiatric disease were included. Subjects underwent a standard clinical and neurologic examination before inclusion into the study and handedness was assessed using the Edinburgh handedness inventory. All subjects had normal structural T1- and T2-weighted magnetic resonance imaging (MRI) scans and a normal resting EEG-pattern. Written informed consent was obtained from all participants. The study was approved by the ethics committee of the University of Cologne.
Positron Emission Tomography and Magnetic Resonance Imaging Scans
Positron emission tomography imaging was performed on an ECAT EXACT scanner in three-dimensional (3D) mode after bolus injection of 370 MBq of O-15-water. Twelve consecutive scans were performed in each subject and reconstructed to 47 contiguous slice of 3.375 mm thickness and 2.57mm voxel size. For attenuation correction and for reconstruction of the TMS coil a transmission scan of 10 mins duration was performed before the emission scans. An additional mu-metal shielding (Paus et al, 1997), was not used in our study, since blank scans and phantom measurements did not show any effect of the TMS coil's magnetic field onto the photomultiplier tubes of our PET scanner.
Magnetic resonance imaging was performed on a 1.5T Philips Intera Master clinical MRI scanner. T1-weighted images were acquired as isotropic data sets of 1 mm voxel size.
Repetitive Transcranial Magnetic Stimulation and Activation Paradigm
For rTMS a Magstim 200 rapid stimulator with a 79 mm figure-of-eight coil was used. Before the stimulation procedure in the PET scanner, the resting motor-threshold was determined in each subject, the detailed description of the TMS procedure has recently been reported (Thiel et al, 2005b). One rTMS test session was performed outside the scanner to familiarise the subject with the procedure. TMS was performed with 4 Hz at resting motor threshold for 30 secs.
During the PET measurement, the TMS coil was positioned over the triangular part of the left IFG. The position on the subject's head was determined from the 3D-reconstructed MRI scan using surface-distance measurements as described previously (Thiel et al, 2005a, b)
In total, 12 PET-Scans were performed with three scans each during baseline rest with eyes closed, overt verb generation, rest with TMS and verb generation with TMS in a balanced sequence. The click noise of the coil was played from a loudspeaker to match for the auditory input during the scans without TMS. For verb generation, high frequency German nouns selected for similar association content (Bascheck et al, 1977) were presented at a fixed rate of one verb every 2.5 secs, thus 12 verbs were presented during the 30 secs TMS stimulation.
Positron emission tomography data acquisition was started simultaneously with tracer injection and presentation of verbs. Repetitive transcranial magnetic stimulation stimulation (or TMS click noise) began 15 secs after scan start to account for the circulation time of the activity bolus and lasted for 30 secs. Data acquisition was continued for further 15 secs after the end of stimulation. During the verb generation scans the subjects' responses were recorded and reaction time latencies were measured as described previously (Thiel et al, 2005b).
Image Analysis
Emission- and transmission images were coregistered to the T1-weighted MRI using a fully automated mutual information-based algorithm (Cizek et al, 2004). The three PET scans of each condition were averaged and ratio normalized. Activation images were calculated as difference images of rCBF changes for the contrasts verb generation versus rest, verb generation with TMS versus rest, rest with TMS versus rest and verb generation with TMS versus verb generation and smoothed with 12 mm Gaussian filter.
For analysis, activation PET images were rendered onto the 3D-reconstructed MR image and the TMS coil was reconstructed from the transmission scan (Figure 2, right). Regional CBF change was measured on the reconstructed cortical surface under the centre of the TMS coil with a cylindrical volume of interest of 10 mm diameter and 10 mm depth (von Stockhausen et al, 1998) and at the mirror region in the right hemisphere. Differences in regional CBF change across subjects and between patients were assessed by conventional non-parametric tests.
Results
Behavioural Data
As shown in Figure 1, there was a significant increase in reaction time latencies during the verb generation task when comparing the mean reaction time latency of the scans without TMS to the mean reaction time latency of the scans with TMS (signed rank test, P = 0.031). This increase was observed in each individual, demonstrating an effective interference with language function.
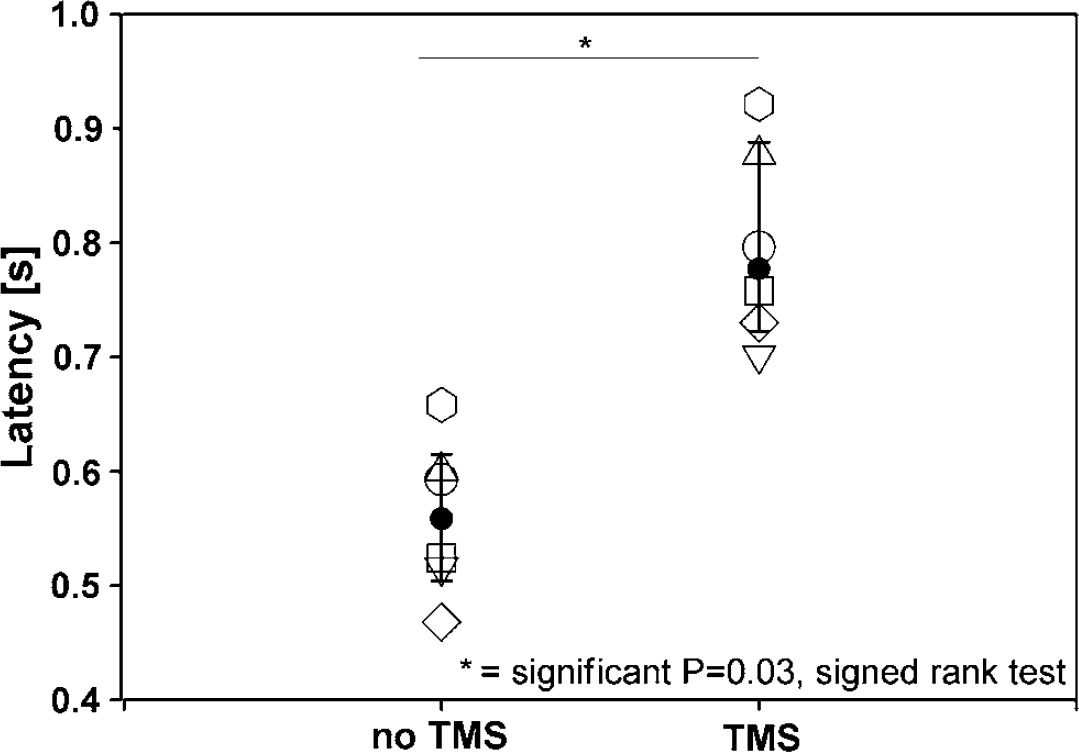
Reaction time latencies for the verbal fluency task. Mean latencies, averaged over three respective PET scans during verb generation with and without TMS are given. The symbols represent the subjects, median and 25th/75th percentiles are indicated.
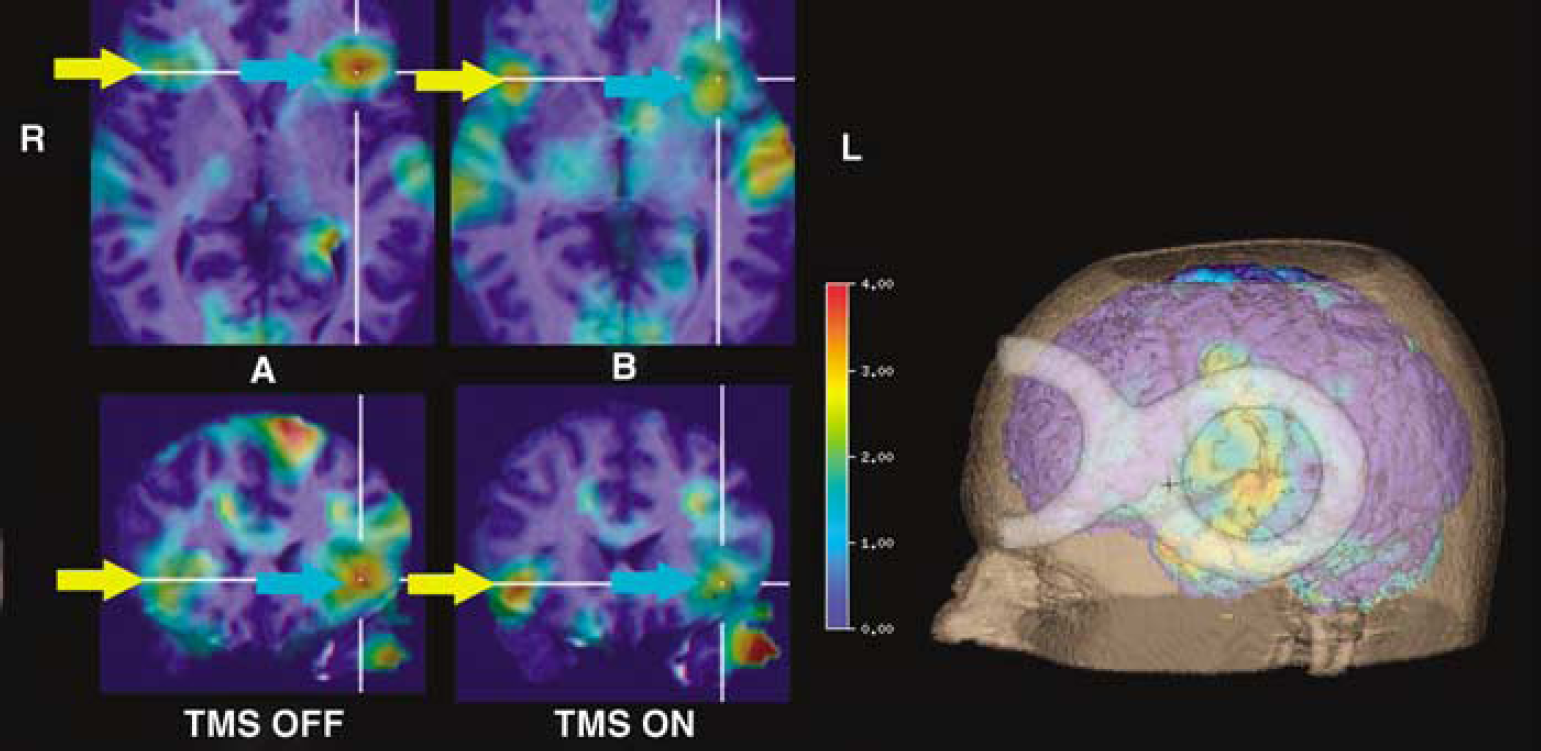
Illustration of TMS effect. The coil position is shown in the 3D rendering. Image
Imaging Data
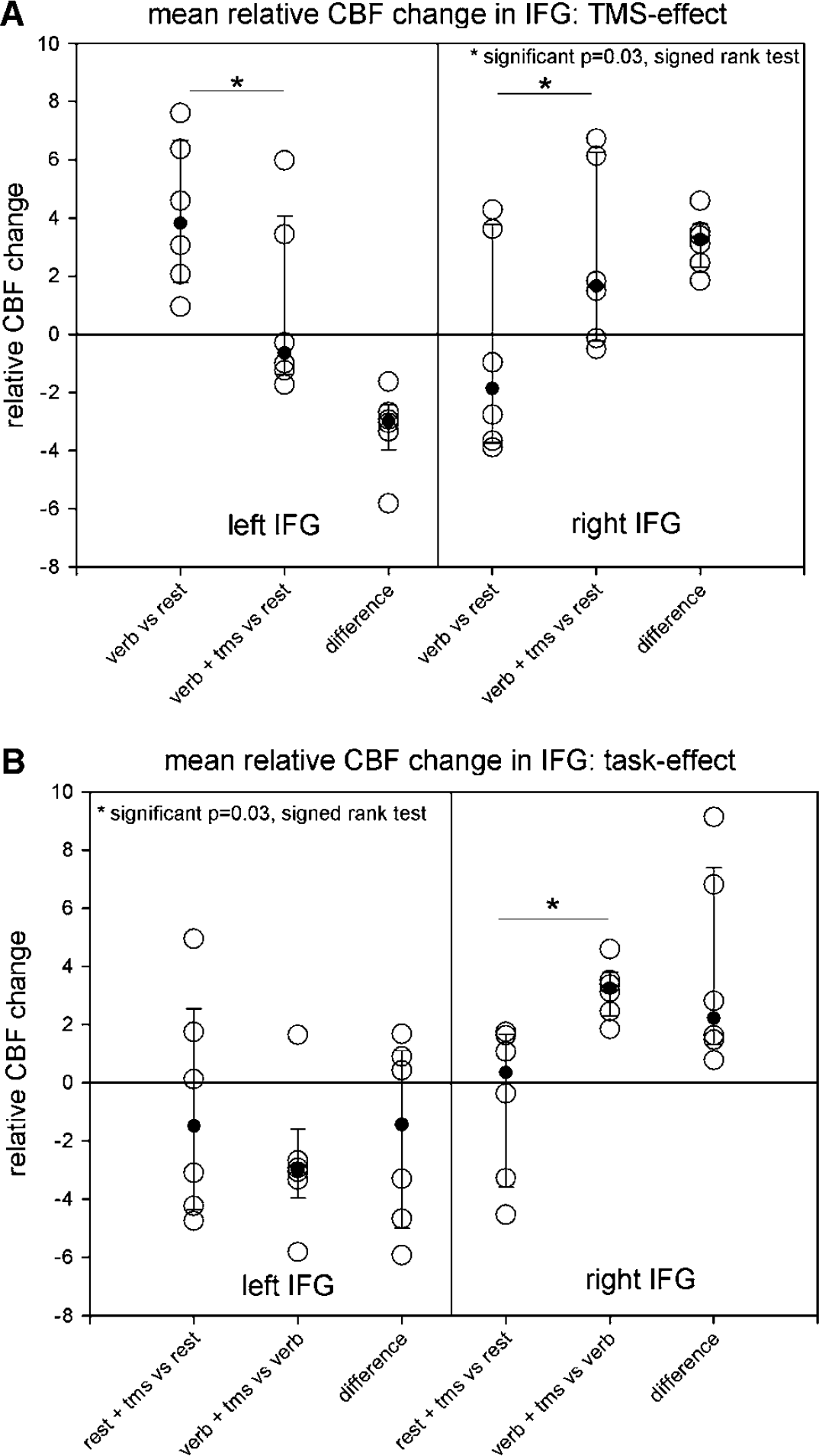
During verb generation, an increase in relative CBF change over the left IFG was observed, which decreased significantly (signed rank test P = 0.03) during interference with TMS over the left side. This decrease was observed in each individual (Figure 3A left). Conversely activity increased significantly (signed rank test, P = 0.03) in the right IFG (Figure 3A right). Again this increase was seen in each subject thus demonstrating a reproducible effect of left-sided TMS on the activation within the left and right IFG. Figure 2 gives an exemplary illustration of the effects in one subject.
In figure
To exclude that the observed effect is an unspecific effect of TMS interference rather than an effect on language function, we compared TMS-induced CBF-changes during rest with those during verb generation. Over the left side, no significant difference between rest and verb generation during TMS was observed suggesting that the left-sided TMS effect described above is not task specific (Figure 3B, left). On the right side, however, a significant TMS-induced increase in right IFG activity (signed rank test, P = 0.03) was only seen during the verb generation task (Figure 3B, right), demonstrating that this increase is specific for the task. As before, this increase occurred in each individual.
Discussion
The main finding of our study was a decrease of rCBF change in the left IFG during verb generation relative to rest caused by TMS interference and a simultaneous increase of rCBF change in the right IFG (Figure 3A). Thus, rTMS interference shows the same effect on activation patterns which has repeatedly been described in real brain lesions (Thiel et al, 2001, 2005a; Perani et al, 2003; Winhuisen et al, 2005). It was further shown that this right-sided increase in IFG activation is specific for the verb generation task and not an unspecific remote TMS effect, since it was only observed during verb generation and not during resting condition (Figure 3B, right). This increase was also observed in two subjects with some right-sided activity before the TMS interference underlining the independence of this finding from the extent of language dominance (Figure 3A).
Thus, our study for the first time provides data on rTMS interference with activity of language networks measured as language-associated cerebral blood flow changes. Previous studies have only investigated the effect of sub- and suprathreshold TMS stimuli on resting cerebral blood flow or metabolism in motor cortex and not the interference effect of TMS with brain activation as in our study.
Data from these studies where TMS was applied with intensities above the resting motor threshold report increases of cerebral blood flow (Fox et al, 1997), metabolism (Siebner et al, 2001) and f-MRI blood oxygen level-dependent (BOLD) signal (Bestmann et al, 2004) relative to no TMS at the stimulation site over primary motor cortex during rest. In the contralateral hemisphere a decrease of rCBF during stimulation of ipsilateral M1 has been observed (Fox et al, 1997).
For subthreshold stimuli increases as well as decreases of resting CBF (Paus et al, 1998), metabolism (Siebner et al, 2000) and BOLD signal (Bestmann et al, 2003) were reported depending on frequency of stimulation. In our study, a trend towards a decreased rCBF under the coil during suprathreshold stimuli under resting conditions was observed. This may be because of differences in excitability of primary motor cortex and IFG. Paus et al (1997, 1998), have shown that applying TMS with the same parameters to different brain regions may yield decreases or increases of resting CBF depending on the brain region. It is further unclear whether the resting motor threshold which is determined as the minimum stimulator output which leads to a discharge of pyramidal cells in the primary motor cortex is sufficient to yield a similar effect over the IFG. It may thus be that stimulating the IFG at resting motor threshold is in fact subthreshold and that these subthreshold stimuli cause an ipsilateral decrease in resting CBF.
Nevertheless the TMS parameters in our study have obviously been effective in interfering with language function as demonstrated by consistently increased reaction time latencies (Figure 1). Qualitative effects of TMS, over the triangular part of the left IFG, like speech arrest or verb generation disturbances have so far only been described in patients with left-hemisphere brain lesions (Winhuisen et al, 2005; Thiel et al, 2005a). The TMS interference in normal subjects is weaker than in patients and an increase in reaction time latency is the quantitative behavioural effect which can be expected. It has been shown in a previous study that the effect is caused by a specific interference with language function (Thiel et al, 2005b), thus a control stimulation site (e.g. vertex) was not included in this study because a repositioning of the coil during the PET-scanning procedure was not feasible.
Since it was shown that the right-sided increase in activation-related CBF change is caused by TMS only during verb generation, the question about the mechanisms mediating this task-specific shift in brain activity arises. Again no data on the language domain are available but investigations from the motor system may provide models which can be applied to the language system as well.
The unmasking of pre-existing but inactive neuronal connections, the so-called silent synapses by reduction of inhibitory activity (e.g. caused by a brain lesion) may be a possible mechanism for mediation of rapid changes in activation patterns (Jacobs and Donoghue, 1991). The existence of these mechanisms in vivo has been demonstrated by double pulse TMS techniques (Ferbert et al, 1992). The application of a conditioning TMS stimulus over the motor cortex and a test stimulus within 10 to 50 ms later over the contralateral motor cortex causes a decrease of the actionpotential amplitude elicited by the test-stimulus compared with the same test stimulus without preceding conditioning stimulus. This reduction of stimulus response caused by excitatory transcallosal input from the contralateral hemisphere is termed inter-hemispheric inhibition (IHI). It was shown that this IHI is mediated by inhibitory inter-neurons on the side where the test stimulus is applied (Daskalakis et al, 2002). It has further been hypothesized that IHI is crucial for maintenance of hemispheric asymmetry. In a double-puls TMS -study. Netz et al (1995) were able to show that in the motor cortex of right handers IHI of the left hemisphere onto the right hemisphere is larger than in the opposite direction, whereas in left handers left as well as right dominance regarding IHI was found.
In patients with focal brain lesions of the motor cortex a reduction in IHI has repeatedly been reported (Manganotti et al, 2002). The same reduction has been described for lesions in the corpus callosum (Meyer et al, 1998).
For the language domain, metabolic language activation studies in normal subjects have also shown that activation of left hemisphere language areas and homologous right hemisphere areas are negatively correlated with metabolic activity in the corpus callosum (Karbe et al, 1998).
These mechanisms may provide the physiologic basis for the observed rCBF changes in the contralateral hemisphere but they tell nothing about the functional relevance of the right-sided language-related activation. There is however evidence from TMS studies in patients with real brain lesions that in some patients (especially in tumor patients with long disease duration) the right hemisphere has become essential for language production (Thiel et al, 2006). These effects cannot be expected from transient brain lesions like the proposed TMS model which last for 20 or 30 secs. However, this is a unique advantage of TMS-lesion models because of its limited extent in space and time the ‘lesion’ is rather circumscribed and there is very little time for compensatory mechanisms to become effective. The interference effect with TMS is thus probably more related to the physiologic function of a certain brain region than the effect of long-lasting real brain lesions.
Conclusion
Taken together these data may serve as a reasonable model to explain the immediate increase of right IFG activity during interference with language function of the left hemisphere as a reduction of trancallosal inhibitory activity oft the language dominant left hemisphere caused by rTMS interference. Language dominance before the interference seems not to be sufficient to explain the observed phenomenon. These findings may offer new perspectives for the development of novel rehabilitation strategies for aphasic patients by influencing these mechanisms on a pharmacologic or electrophysiologic basis.
