Abstract
The mechanisms that are responsible for the remarkable potential for functional recovery from stroke in humans remain unclear, and functional tomographic neuroimaging techniques increasingly are being used to investigate this issue. Such studies confirmed that recovery of function is related to the volume of penumbra tissue that escapes infarction. For language, reactivation of the primary functional areas in the dominant hemisphere is associated with the best prognosis. Evidence for functional plasticity in the immediate vicinity of infarcts, as demonstrated under experimental conditions with invasive methods, is still limited after stroke in humans, probably because of the limitations of spatial resolution of most currently available methods. Often, functional changes in the large-scale networks that support motor (for example, supplementary and premotor cortex) and language functions (for example, prefrontal cortex) have been found, more extensively after lesions acquired during childhood than at adult age. A frequent finding is an increase in the cerebral blood flow response in corresponding regions of the healthy hemisphere during unilateral motor activation or language activation. It is, however, not yet clear whether that is related to functional recovery, and there are several observations indicating that it is often inefficient. Further systematic follow-up studies and therapeutic intervention trials are needed to clarify these issues.
Keywords
Clinical recovery of neurologic function after human stroke is frequently seen and has been described in many publications (Twitchell, 1951; Wade and Hewer, 1987; Bonita and Beaglehole, 1988; Mauritz et al., 1997; Jorgensen et al., 1999). In particular, language and motor functions often recover nearly completely from initial impairments of mild to moderate severity; yet, the physiologic correlates of that recovery are less clear. Most likely, edema and its resolution play a role, but there is little information regarding its influence on neuronal function (Rosenberg, 1999). Functional recruitment of additional neurons by appropriate training has been demonstrated, mainly by means of invasive electrophysiologic methods, in experimental animals (Nudo et al., 1996; Schallert et al., 2000) and is also likely to occur in humans. However, direct evidence is difficult to ascertain. There may also be rearrangements of large-scale neural networks involving ipsilateral or contralateral cortical areas, as suggested by electrophysiologic evidence. The motor network is reorganized with involvement of descending motor pathways contralateral to the lesion in patients after hemispherectomy and severe hemispheric lesions (Benecke et al., 1991). This may be because of the unmasking of existing but functionally inactive pathways, the sprouting of fibers from surviving neurons and formation of new synapses, neuronal group selection, or the redundancy of central nervous system circuitry allowing alternative pathways to take over functions (Bach-y-Rita, 1990; Lee and van Donkelaar, 1995; Buonomano and Merzenich, 1998).
Functional imaging techniques provide a means to study physiologic correlates of plasticity and recovery noninvasively in stroke patients. Early techniques, such as cerebral blood flow (CBF) measurements by single or multiple detectors or planar imaging devices, already demonstrated remote functional effects of focal ischemic lesions on resting CBF. Tomographic CBF techniques, such as single photon emission computed tomography (SPECT) and positron emission tomography (PET), demonstrated these effects with improved specificity and spatial resolution. In recent years, functional changes during execution of neuronal function have been elucidated by demonstration of an increase of CBF or cerebral metabolic rate of glucose (CMRGlc) in activated brain structures. Most recently, functional magnetic resonance imaging (fMRI) became available for such activation studies, providing further increases in spatial and temporal resolution (Wildgruber et al., 1997). Thus, these studies primarily address representational plasticity, that is, changes in the topographic representation of specific neural functions in brain. In principle, aspects of molecular and cellular plasticity (Buonomano and Merzenich, 1998; Johansson, 2000) could also be studied in vivo by use of specific radiotracers to assess changes of neurotransmitter synthesis and receptor binding capacity (Kegeles and Mann, 1997). Degeneration of white matter tracts that accompany neuronal reorganization can be studied with magnetic resonance imaging (MRI; Orita et al., 1994) and magnetic resonance spectroscopy (MRS; Pendlebury et al., 1999).
This review focuses on recent imaging findings that address the issue of plasticity and recovery with respect to motor and language function after stroke in humans.
MOTOR FUNCTION
Motor function may be impaired because of cortical damage or damage of fiber tracts. Fiber tracts may also be damaged by anterograde axonal injury after cortical lesions, which seems to be associated with the severity of contralateral motor deficit (Pendlebury et al., 1999). There is evidence that the somatotopic organization of the primary motor cortex consists of broad overlapping territories that may contribute to recovery after partial damage. It is possible that recovery of motor function may also be supported by primary motor neurons originating in premotor and supplementary motor areas (Schieber, 1995). The degree of motor impairment and the potential for recovery often depends on the exact site of the cortical lesion or a lesion to the associated fiber tracts. For example, Fries et al. (1993) found that lesions of the anterior or posterior limb of the internal capsule led to an initially severe motor impairment followed by excellent recovery, whereas lesions of the posterior limb of the internal capsule in combination with damage to lateral thalamus compromised motor outcome.
Recovery of neurons in the periinfarct zone
In acute infarcts, severe reduction of CBF may impair neuronal function in the penumbra, brain tissue that is still viable and has the potential for recovery. With functional imaging, tissue can be identified by CBF values that are lower than necessary for neuronal function but above the limit for structural integrity (Powers et al., 1985; Heiss et al., 1999b). Tissue viability, in spite of severe CBF reduction, can be demonstrated in vivo by preserved or even increased oxygen extraction (Wise et al., 1983; Marchal et al., 1993), intact GABA receptor binding (Heiss et al., 1998), and absence of major changes of proton diffusion (Warach et al., 1996; Jiang et al., 1998; Gonzalez et al., 1999). However, if local perfusion does not improve spontaneously or by successful therapeutic intervention, a significant part of the penumbra will deteriorate to infarction or suffer substantial diffuse neuronal loss (Heiss et al., 1992). It has been demonstrated that spontaneous neurologic recovery after stroke is significantly related to the volume of penumbra tissue that escapes infarction (Furlan et al., 1996; Heiss et al., 2000). After stroke demarcation, functional activation may occur in the immediate vicinity of a cortical infarct, as observed with fMRI and finger-tapping of the recovered hand in three patients (Cramer et al., 1997).
Recruitment of additional neurons adjacent to an ischemic lesion
Clinical evidence for neuronal plasticity in neurons adjacent to ischemic lesions mainly stems from studies with transcranial magnetic stimulation (TMS). Four months after stroke enlargement of the hand motor area on the affected hemisphere combined with an improvement of clinical scores was seen after an intensive neurorehabilitation program (Cicinelli et al., 1997). In a study of chronic arm paresis (average duration of 6 years) before and after successful treatment with a special type of movement therapy (constraint-induced movement therapy), TMS indicated enlargement and medial or lateral displacement of the cortical motor area that was sensitive to stimulation in the contralateral hemisphere (Liepert et al., 1998).
Motor cortex activity contralateral to paretic side in patients with lesions to fibers or peripheral nerves
Changes in activation after stroke may be compared with changes in which motor cortex is intact, but sensory input and movement is altered because of peripheral amputation. Kew et al. (1994) describe in patients with upper limb amputation that paced shoulder movements were associated with significant blood flow increases in the contralateral M1/S1 cortex. In traumatic amputees, these increases were present over a wider area and were of significantly greater magnitude in the partially deafferented cortex contralateral to the amputation. In congenital amputees, blood flow increases were also present over a wider area in the partially deafferented M1/S1 cortex, but their magnitude was not significantly different from that in the normally afferented M1/S1 cortex.
A common reason for lesions to motor fibers with contralateral hemiparesis are capsular infarcts or hematomas. Patients with such lesions usually show activation of sensorimotor and premotor cortex to the same extent as normal subjects (Weiller et al., 1992). Even the imagination of such movements may lead to substantial activation of motor cortex (Fig. 1). Tactile exploration of shapes with the paretic hand after subcortical infarction led to large activations in contralateral motor and sensory hand cortex (Weder et al., 1994). These activations were also similar to those observed in normal subjects with the same task. In a study of 6 patients with capsular or pontine infarcts, passive movements of the paretic arm also led to activation of contralateral sensorimotor cortex (Nelles et al., 1999). Thus, these functional imaging results currently do not provide evidence for neuronal plasticity in motor cortex after lesions to the efferent pathway, but sophistication of study design and resolution of imaging techniques may not yet be sufficient to detect subtle changes.
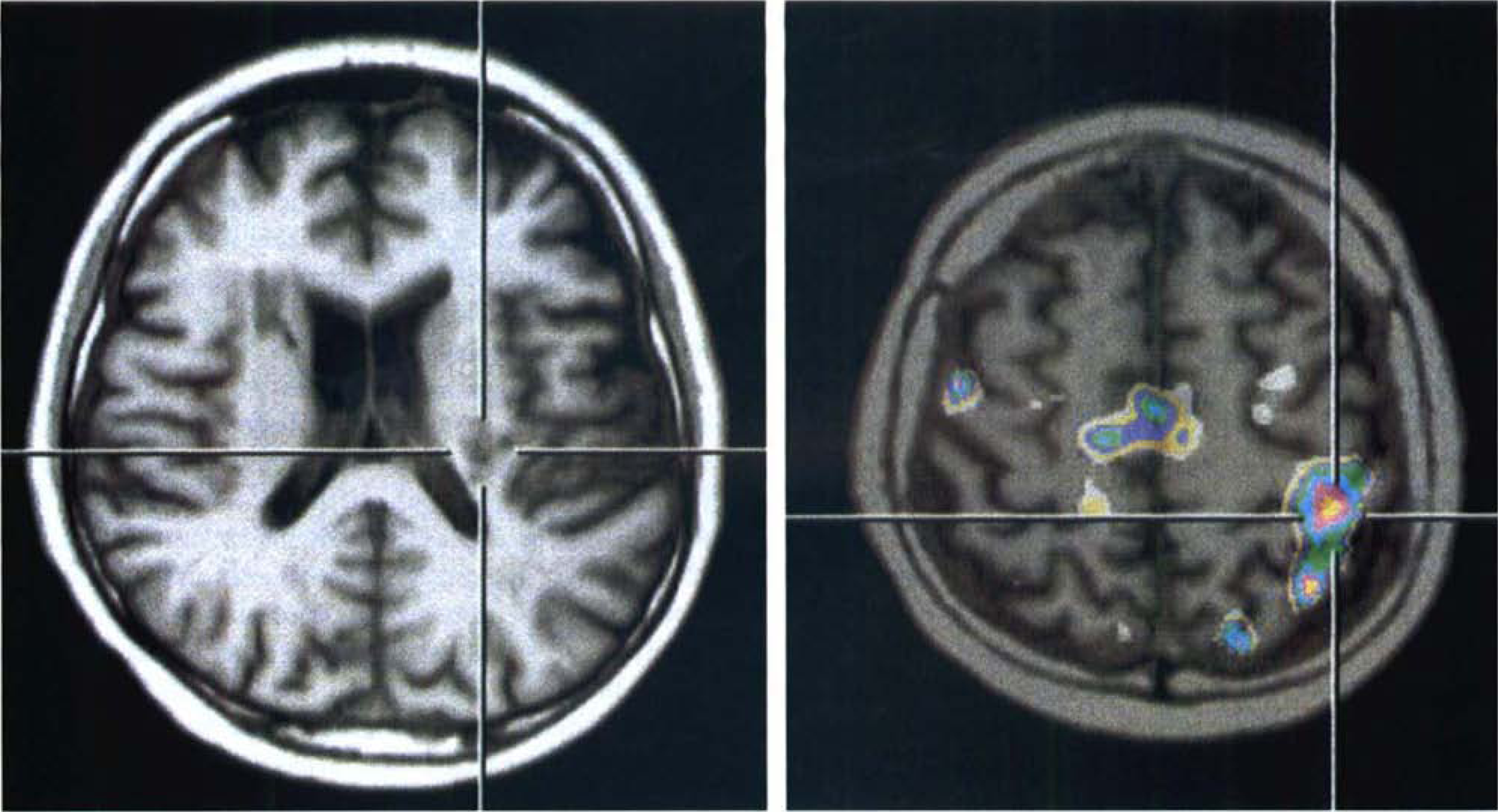
Activation of left motor and sensory motor cortex (positron emission tomography—magnetic resonance imaging fusion image on right side) in a patient with severe paresis of the right arm after left striatocapsular hemorrhage (see crosshairs on magnetic resonance imaging section on left side) by imagination of movement of the paretic hand.
Increased activity in intact motor cortex ipsilateral to paretic side
In normal subjects and for the unaffected hand in stroke patients, only little activation of ipsilateral motor cortex is seen during finger movement. It is a consistent finding in several studies that ipsilateral activation of motor cortex is stronger for movements of the paretic fingers after recovery from stroke (Chollet et al., 1991; Weder et al., 1994; Cramer et al., 1997; Honda et al., 1997; Cao et al., 1998). Chollet et al. reported that, in addition to stronger intensity, the spatial extent of activation in motor cortex was enlarged. Somewhat increased activation on the ipsilateral side was also seen in premotor and insular cortex. There was little difference between activation of paretic and normal fingers in supplementary motor cortex (SMA), putamen, and parietal cortex. These data were interpreted as evidence for stronger activity of the small noncrossing part of the corticospinal tract after lesion to the crossing fibers or neurons.
Weiller et al. (1993) suggested that voluntary mirror movements of the unaffected hand may contribute to the ipsilateral activations, because increased ipsilateral sensorimotor cortex activation was observed in only 4 of their 8 patients who had capsular infarcts. Severe mirror movements associated with ipsilateral motor cortex activation was also reported in a patient with unilateral perinatal pyramidal damage (Nirkko et al., 1997). Yet, such movements were rarely observed in another study (Cramer et al., 1997). In a PET study of two patients, increased activation of ipsilateral motor cortex was associated in movement-related cortical potentials with a lack of the contralateral predominance in amplitude of the late component of slow negative shift before the movement of the recovered hand (Honda et al., 1997).
Further confirmation of more intense ipsilateral activation was provided in a study by Cramer et al. (1997). However, the activation site in the ipsilateral hemisphere was located 1 to 2 cm more anteriorly, laterally, and ventrally than in the contralateral hemisphere (Cramer et al., 1999). In that respect, there was no major difference between controls and stroke patients, and the ipsilateral activation may be seen as an exaggeration of a normal event (Cramer, 1999). Ipsilateral activation was particularly intense in patients with perinatal damage (Cao et al., 1994). In particular, secondary motor and frontoparietal nonmotor cortices were more activated in patients with lesion onset before age 4 than after age 10, suggesting a greater potential for reorganization during early development than later in life (Müller et al., 1997) (Fig. 2).
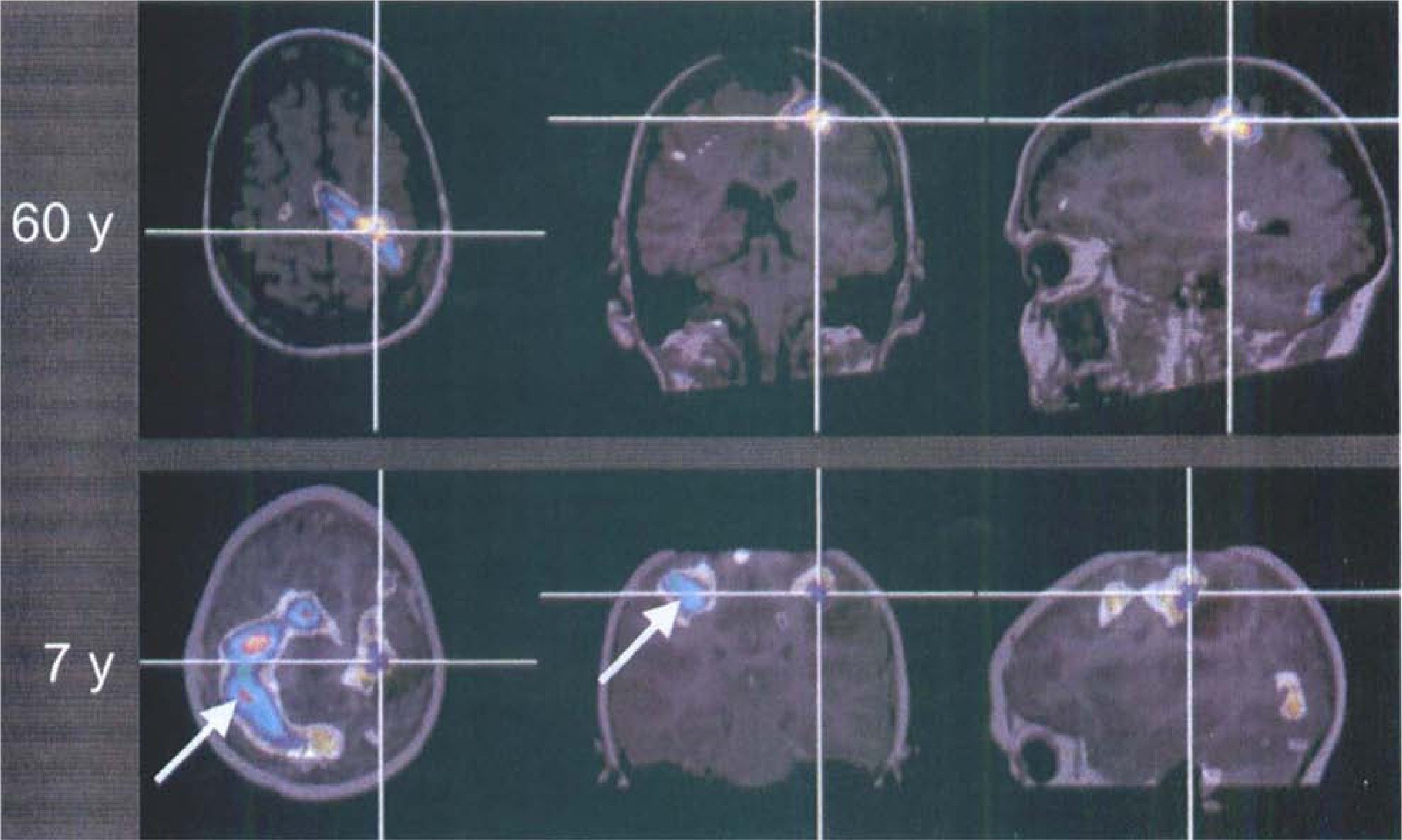
Age effect on activations induced by finger tapping of the right hand in two patients with tumor lesions of left motor cortex and good motor function. A 60-year-old patient shows normal activation left motor cortex with some dorsal displacement (top row), whereas a 7-year-old child with a dysembryoblasic tumor shows extensive activations in the right hemisphere (arrows, bottom row).
It is not clear whether increased activity in ipsilateral motor cortex contributes to functional recovery in adults. The extent of ipsilateral activation was not related to recovery of performance in a study by Cao et al. (1998). Nonrecovered hemiplegic patients showed stronger activations ipsilateral and contralateral during passive movements than normal controls (Nelles et al., 1999). In a small series, ipsilateral activation was only seen in the most severely impaired subject (Weder and Seitz, 1994). Cramer (1999) points out that TMS also does not support the hypothesis that noncrossing corticospinal fibers are responsible for recovery.
In a study of resting CMRO2, there was even a slight decline in the unaffected hemisphere during early recovery phase after stroke (Iglesias et al., 1996); it was not related to neurologic recovery and was interpreted as a possible consequence of transcallosal fiber degeneration. In an fMRI study of normal subjects, unilateral hand movements were associated with contralateral cerebral activation and ipsilateral cerebral deactivation, which was interpreted as resulting from transcallosal inhibition (Allison et al., 2000). Thus, increased activation of the intact ipsilateral motor cortex during movement may be because of disinhibition as a consequence of damage to predominantly transcallosal projections (Kujirai et al., 1993; Boroojerdi et al., 1996).
Rehabilitation of motor function after an ischemic stroke involves relearning of motor tasks. Thus, it is of interest to compare this process with motor learning in normals (Poldrack, 2000). It seems that the stronger activation of movement-related centers outside of primary motor cortex and in the contralateral hemisphere with simple finger tapping with the recovered hand in patients is similar to activations seen in normals with more complex movements (Rao et al., 1993; Kawashima et al., 1998). In normals, finger tapping with the nondominant hand seems to elicit slightly stronger and more bilateral activations than the same task performed with the dominant hand (Cramer et al., 1997; Cao et al., 1998), although the significance of that finding is not yet established.
Role of frontal premotor and supplementary motor cortex
Several authors described increased bilateral activation of premotor cortex and SMA, often associated with anterior cingulate activation. Bilateral activation of premotor areas was seen in seven patients with marked recovery of hand function after acute severe hemiparesis (Seitz et al., 1998). Weiller et al. (1992) described increased prefrontal and anterior cingulate activation in patients after recovery from striato-capsular infarction during sequential finger to thumb opposition. It was associated with increased activation of insula and inferior parietal cortex (Weiller et al., 1992). In their study, premotor cortex activation was increased on the ipsilateral side only, and they found increased activation in some of these areas also during movements of the unaffected hand. Abnormal activation of ipsilateral premotor cortex was found in a study with tactile exploration of geometric objects (Weder et al., 1994), which was not related to task performance.
Intensity and extent of premotor and SMA activation seems to be influenced considerably by details of the activation task. Self-paced tasks, requiring the initiative of the subject, may elicit stronger activations than externally paced tasks, even in normals (Wessel et al., 1997; Gerloff et al., 1998), but pacing with movement frequencies faster or slower than the subject's own pace may also increase SMA activation (Kawashima et al., 1999). Learning new movements also increases CBF in SMA and premotor cortex in normal subjects (Roland et al., 1980; Jenkins et al., 1994). Learning-associated activations also included sensorimotor and parietal cortex, basal ganglia, and cerebellum. Large interindividual variability has been noted (Schlaug et al., 1994), which may be because of different learning strategies (Rauch et al., 1995).
Changes in sensorimotor and parietal cortex
Plasticity in somatosensory cortex has been studied in humans much more extensively after peripheral lesions than after stroke. Consistent with similar findings in experimental studies (Schallert et al., 2000), marked intrusion of facial representations into the digit and hand area of somatosensory cortex has been observed by magnetoencephalography in humans after upper arm amputation (Yang et al., 1994; Elbert et al., 1994). Reorganization of the somatotopic organization in somatosensory cortex has also been observed after surgical separation of webbed fingers (syndactyly) (Mogilner et al., 1993). Reorganization of sensory pathways may occur very soon after amputation in humans, potentially because of the unmasking of ordinarily silent inputs, as demonstrated in a case report by Borsook et al. (1998). In that study, stimuli applied on the ipsilateral face were referred in a precise, topographically organized, modality-specific manner to distinct points on the phantom limb within 24 hours after arm amputation, and it was shown, albeit only after several weeks, that this corresponded to fMRI signal changes.
Some evidence of sensorimotor cortex plasticity after stroke was observed by Rossini et al. (1998) in a patient with an ischemic lesion in the left fronto-parietotemporal cortex. They found a posterior shift and asymmetrical enlargement of sensorimotor areas. Reduced activation of parietal, and of premotor and midfrontal association cortex, was seen with a tactile discrimination task after ischemic infarcts of thalamus or putamen with good motor recovery (Weder and Seitz, 1994). Weder and Seitz (1994) suggest that recovery of motor functions is not hindered by impairment of a fronto-parietal network that is involved in tactile discrimination. Complementary to this, Nelles et al. (1999) found increased activation in sensorimotor cortex and in the parietal lobes with passive arm movements in nonrecovered patients.
There seems to be less interhemispheric reorganization of the somato-sensory system than of the motor system, because representation of somatosensory area in ipsilateral cortex was observed only in one third of patients with perinatal unilateral brain lesion (Chu et al., 2000), whereas ipsilateral representation of motor cortex was much more common in these patients.
Role of subcortical structures and cerebellum
A SPECT study in the chronic phase several months after stroke found a positive correlation with contralateral resting CBF in thalamus, lentiform and caudate nuclei, and premotor cortex with the degree of motor improvement (Pantano et al., 1996). The thalamus is an important relay station for sensory afferences to the cortex, and it is also involved in the extrapyramidal motor system. A reduction of ipsilateral thalamic CMRGlc at rest is often seen after hemispheric stroke (Pawlik et al., 1985) and is associated with poor motor recovery (Binkofski et al., 1996). By multiple regression and discriminant analysis, Azari et al. (1996) found a close relation of CMRGlc between bilateral SMA, ipsilateral thalamus, and contralateral cerebellum in recovered patients, suggesting a stronger functional association of these structures than in normal subjects or nonrecovered patients. In a related study, activation of a similar network including bilateral occipital and bifrontal cortex and cerebellum, contralesional cingulate, hippocampus, and thalamus, was statistically associated with recovery of motor function (Seitz et al., 1999).
Cerebellar activation is seen in most activation studies. In patients after stroke, cerebellar activation was reported as normal (Weiller et al. 1992) or reduced, in particular, in ipsilateral cerebellum (Weder et al., 1994; Nirkko et al., 1997). There was less cerebellar activation in patients with lesions received in early childhood than later in life (Müller et al., 1997). Reduced activation may be a consequence of damage to cortico-ponto-cerebellar pathways by stroke, which is probably also the cause of the crossed cerebellar diaschisis that is seen already in resting state (Pantano et al., 1986).
Global factors influencing functional recovery
The potential for rehabilitation is also limited by several global factors, such as age and microvascular angiopathy, that affect basically the whole brain. These may be very strong factors, which are evident from many clinical studies, and they are also reflected in reduced glucose metabolism already at resting state (Heiss et al., 1993a).
With regard to the influence of age on clinical recovery, the potential for recovery is much greater for lesions acquired during early life, when even hemispherectomy is often followed by good recovery of motor and language function. This may be mediated by compensatory functional changes in corresponding cortical areas of the nonaffected hemisphere, including the ipsilateral corticospinal pathway (Benecke et al., 1991). Functional imaging studies suggest that metabolic recovery and functional plasticity in remaining subcortical structures of the operated hemisphere may also play an important role (Chugani and Jacobs, 1994; Müller et al., 1998a).
After childhood has passed, the potential for recovery is reduced. Yet, it remains unclear whether there is a continuing influence of age beyond the limitations imposed by age-related multimorbidity on functional recovery after stroke (Nakayama et al., 1994).
APHASIA
The contribution of various parts of the distributed network of speech function (Mesulam, 1990) to recovery from aphasia is a long-lasting controversy, in which the role of areas within the subdominant, usually right hemisphere is especially a matter of debate (Luria et al., 1969; Kertesz, 1989). Although the majority of authors stress the superior role of the dominant, usually left hemisphere (Benson, 1985), others argue for a compensatory function of the right hemisphere (Zaidel, 1985), or stress the importance of a bihemispheric network, in which the two hemispheres act as a unit with the right hemisphere aiding and sustaining speech processing and inhibiting unrelated activity (Sperry, 1986; Gazzaniga et al., 1984). Differences in the hemispheric representation of language (Ojemann and Whitaker, 1978) contribute to the ongoing debate on the role of undamaged regions in the left hemisphere and of homotopic contralateral regions for recovery of aphasia after brain damage, for example, by hemispheric stroke. Most clinical and anatomic data indicate that transfer of language function cannot be achieved in adults with clear speech dominance (Benson, 1985), but improvement of aphasia was repeatedly reported in single cases after removal or destruction of commonly recognized speech centers in the left hemisphere (Cappa and Vallar, 1992). Various factors for recovery from aphasia—for example, age, gender, education, and handedness—are important, but location and size of the lesion have the greatest impact (Ferro et al., 1999).
Impact of the morphologic lesion
The morphologic lesion significantly affects type and severity of aphasia, which are the major determinants of outcome (Pedersen et al., 1995). Improvement rate is inversely correlated with lesion size (Kertesz et al., 1979; Selnes et al., 1983; Knopman et al., 1983; Naeser et al., 1987; Ferro, 1992), a dependency, which is most significant for recovery of fluency and less marked for comprehension. Infarcts limited to Broca's area generally produce a nonfluent aphasia with good prognosis (Mohr et al., 1978), whereas recovery is rather limited if lesions extend from the Wernicke area to surrounding temporal regions (Selnes et al., 1985; Naeser et al., 1987). All of these morphologic studies applying computed tomography in stable states of poststroke aphasia supported the importance of intact ipsilateral structures surrounding the lesions (Kertesz, 1988; Vallar, 1990; Naeser and Palumbo, 1994), which played a larger role than right hemispheric regions in the recovery of language function (Gainotti, 1993).
Design and rational of functional imaging studies
The role of areas in the left and right hemisphere in aphasic recovery and the reorganization of the intra- and interhemispheric network after ischemic lesion can be studied by functional imaging methods. Several designs can be applied to such studies including the following: measurements at rest, comparing baseline, or follow-up, or both, to deficit and outcome; measurements during activation tasks, comparing changes in activation patterns to functional performance; and measurements at rest and during activation tasks early and late in the course after stroke to demonstrate recruiting and compensatory mechanisms in the functional network responsible for complete or partial recovery of language function. Only a few studies were performed applying this last and most complete design together with neuropsychological testing for the evaluation of the quality of finally achieved language performance.
Single studies of metabolism and flow: correlates to severity and prognosis of disturbed language function
Studies of glucose metabolism after stroke have shown that the left temporo-parietal cortex is crucial for language perception (Metter, 1987; Karbe et al., 1989) and that the metabolic disturbance in these areas is related to outcome (Heiss et al., 1993b). Investigations in the subacute state after stroke showed a highly significant correlation with language performance assessed at follow-up after two years (Karbe et al., 1995). The receptive language disorder correlated with rCMRglc in the left temporal cortex and word fluency correlated with rCMRglc in the left prefrontal cortex. These results indicate that the functional disturbance as measured by rCMRglc in speech-relevant brain regions early after stroke is predictive of the eventual outcome of aphasia. However, not only functional deactivation (diaschisis) (Feeney and Baron, 1986), but neuronal loss, may contribute to metabolic and perfusional changes in the neighborhood of the infarct, and the condition of the surrounding tissue may affect the recovery of individual patients. On this basis it is not surprising that, in patients with a poor outcome of poststroke aphasia, metabolism in the hemisphere outside the infarct was significantly less than in those with good language recovery, indicating significant cell loss caused by the ischemic episode outside the ischemic core (Heiss et al., 1993b). In addition, the functionality of the network was reduced in patients with an eventual poor outcome; during task performance, patients with an eventual good recovery predominantly activated structures in the ipsilateral hemisphere. Activation studies in small groups of patients resulted in controversial results that were related to the time elapsed since the stroke and to the type of aphasia in the selected patients. Weiller et al. (1995) studied six chronic patients with Wernicke's aphasia and observed bilateral activation in left prefrontal regions and right areas homotopic to language areas. This finding was interpreted as an indication of parallel processing within a bilateral network for functional reorganization of language performance. However, the development of reorganized processing with reestablishment of language performance was not demonstrated, because repeat studies in the acute and chronic phase were not performed.
Serial studies in the course of aphasia: flow and metabolic changes related to recovery
Serial 133Xe-CBF studies demonstrated favorable recovery of comprehension associated with diffuse right hemispheric and regional left parietotemporal activation, whereas incomplete recovery was associated with activation in right frontal areas (Knopman et al., 1984). In patients with good recovery, flow activation in the left hemisphere involved wider areas than in those with an unfavorable outcome (Demeurisse and Capon, 1987). In chronic aphasic patients, the right postero-inferior frontal and postero-superior temporal areas were activated during a word repetition task, but nonfluent aphasic patients recruited the undamaged left postero-inferior frontal region for spontaneous speech (Ohyama et al., 1996). Language recovery in the first months was associated with regression of functional deactivation of glucose metabolism in intact anatomic areas in both hemispheres (Cappa et al., 1997), but abnormal activation patterns (for example, reactivated Broca's area and deactivated right Wernicke's counterpart) were not necessarily related to recovery (Belin et al., 1996). Overall, adults seem not to have a great potential for the homotopic interhemispheric reorganization for language described in children (Müller et al., 1998b).
The involvement of various structures of the functional network in recovery from poststroke aphasia can only be demonstrated by repeated activation studies at intervals after stroke. A few recent studies indicate that preserved left hemispheric speech areas are crucial for satisfactory outcome; regional glucose metabolism at rest and during word repetition was studied by PET in 6 aphasic patients at 4 weeks and 12 to 18 months after the ischemic stroke (Heiss et al. 1997). Satisfactory recovery was related to activation of left hemispheric speech areas surrounding the infarct, especially the left superior temporal gyrus. Activation of right hemispheric regions was not efficacious for a considerable recovery from aphasia. Regional cerebral blood flow—PET studies showed left inferior temporal activation, but only a small right hemispheric response on a word retrieval task in six patients who had at least some recovery from aphasia (Warburton et al., 1999).
Based on these preliminary data, language performance was related to 15O-water PET activation patterns in 23 right-handed aphasic patients 2 and 8 weeks after stroke (Heiss et al. 1999a). In patients classified according to the site of lesion (frontal, n = 7; subcortical, n = 9; temporal, n = 7) and in 11 control subjects, flow changes caused by a word repetition task were calculated in 14 regions representing eloquent and contralateral homotopic areas, which were defined on coregistered MRI. As in previous studies (Frackowiak, 1994; Raichle, 1996), normal subjects showed a symmetric activation pattern of the superior temporal regions bilaterally and asymmetric activations in the inferior part of the left precentral gyrus and (with low intensity [4.13% relative difference]) in the left inferior frontal gyrus. In the three aphasic groups, the extent of recovery differed and was reflected in the activation pattern. The patients with frontal and subcortical lesions improved substantially; they showed activation of the right inferior frontal gyrus and the right superior temporal gyrus during the first scan and had regained left superior temporal gyrus activation at follow-up. The temporal group improved only in word comprehension; they showed activation of the left Broca area and supplementary motor areas in the first scan and had added the precentral gyrus bilaterally as well as the right superior temporal gyrus at follow-up, but the left superior temporal gyrus showed no activation. The shift of activation pattern necessary for satisfactory recovery could also be observed when frontal and subcortical groups were combined (because of their similar activation pattern and preservation of temporal structures) and subdivided into two subgroups, one with improvement of more than 50% and the other with improvement of less than 50% in Token test scores. Both subgroups had a significant activation of the right inferior frontal gyrus in the first scan but not in the follow-up (Fig. 3). The group with more than 50% improvement, however, had a significant activation of left temporal regions in the initial measurement, whereas the group with less than 50% improvement failed to activate these areas in either the first or second scans, despite the fact that these areas are morphologically intact. Patients with a destruction of the left temporal language areas achieve the lowest grade of improvement and always incur a permanent deficit. The most striking compensatory mechanism in this group is the activation of the left opercular region and the supplementary motor area for the task of word repetition during which these areas are either uninvolved or only slightly involved in normals. In the course of time, and with some improvement in comprehension, these patients activate right temporal and left frontal areas. In selected cases, these strategies can be partially successful in improving some language function (Weiller et al., 1995), but usually the destruction of the left superior temporal gyrus prevents sufficient recovery of comprehension (Naeser et al., 1990) and is associated with persistent impairment of repetition, naming, and sentence comprehension (Selnes et al. 1985). Similar results were reported using fMRI and picture naming and verb generation tasks in 7 right-handed aphasic patients at least 5 months after onset of left hemispheric stroke (Cao et al., 1999). As in the previous studies, restitution of damaged left hemisphere language networks was associated with a better outcome than compensatory right hemisphere language activity. After focal left hemispheric damage, a bilaterally organized network worked more effectively than a right predominant network. Other follow-up studies with fMRI showed a good correlation between increase in task-related signals in Broca's area and the recovery of language function in one case (Miura et al., 1999) and a shift of activation to homologous areas in the right hemisphere with improvement of aphasia (Thulborn et al., 1999).
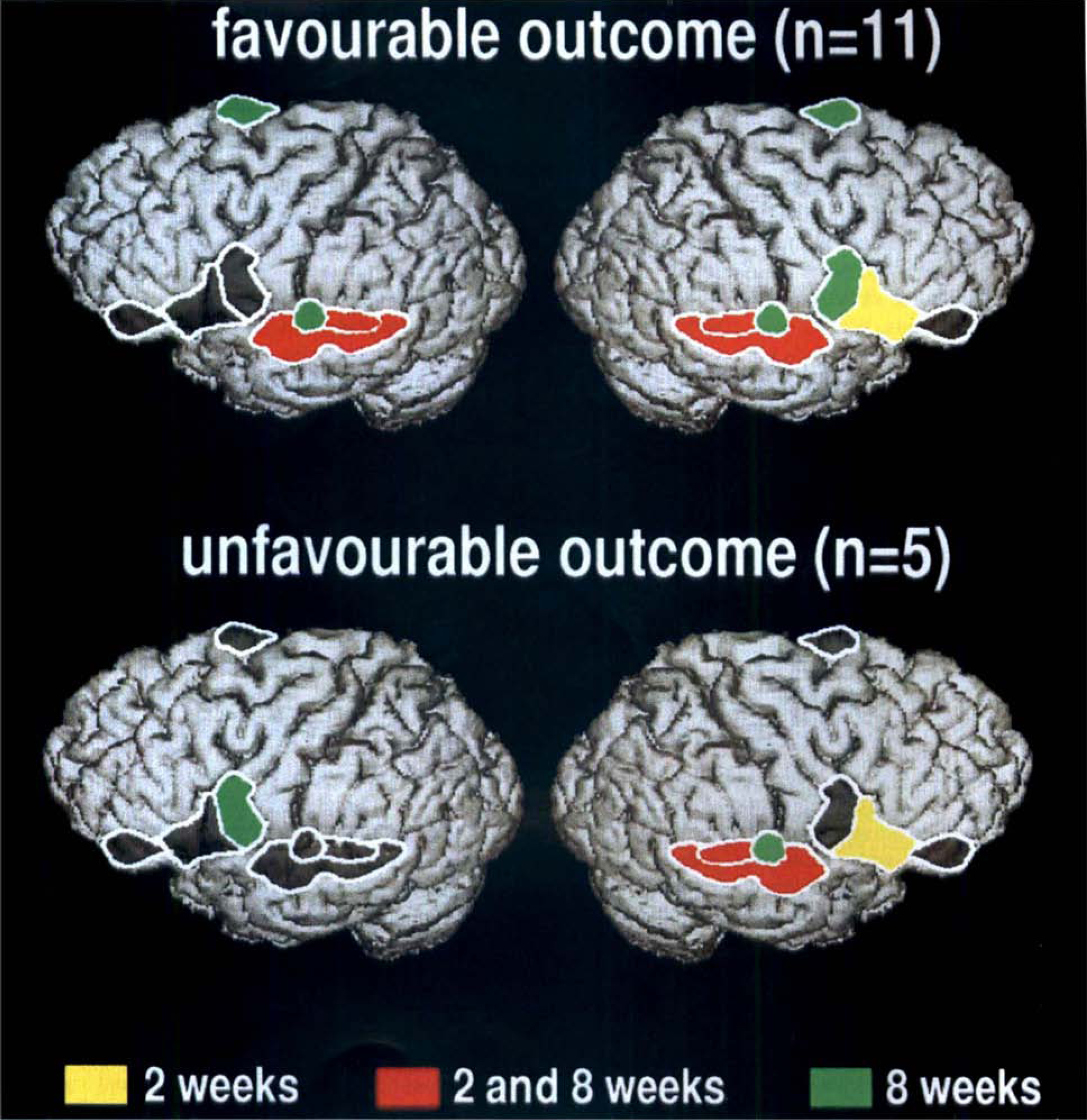
Significantly activated regions during word repetition were identified 2 weeks and 8 weeks after stroke as indicated by colors: yellow regions were activated at 2 weeks only, green at 8 weeks only, red at 2 and 8 weeks. Grey regions did not show significant activation. Patients with favorable outcome (relative improvement in Token test greater than 50%, top row) showed left superior temporal activation that was not present in patients with unfavorable outcome (bottom row). There was transient activation of right Broca's area and persistent activation of right Wernicke's area in both groups.
Importance of left versus right hemispheric areas for restoration of speech
In summary, evidence extracted from those studies suggests a hierarchy within the language-related network regarding effectiveness for improvement of aphasia, a conclusion also reached in other recent reviews (Ferro et al. 1999; Samson et al., 1999; Khatri and Hier, 2000). In the early period, some recovery might reflect the regression of diaschisis in areas that usually participate in language processing. In the long-term, outcome of aphasia is mainly dependent on the size and location of the left hemispheric lesion and the functional reorganization of the left hemisphere. If the more important left hemispheric areas are destroyed, the right hemispheric network can contribute to improvement to some extent (Gainotti, 1993). Satisfactory outcome with restoration of language function, however, correlates with the possibility to activate left temporal areas; but, it can only be achieved within a rather short period if the temporal cortical areas of the dominant hemisphere are morphologically preserved (Kertesz et al., 1993) and can be reintegrated within the functional network. Because repeated activation studies over a longer rehabilitation period are still scarce (Heiss et al., 1997), and up to now did not contain a significant number of patients with different types of poststroke aphasia, the contribution of slow processes involved in the engagement of the nondominant hemisphere for long-term language recovery (Mimura et al., 1998) is still not determined (Selnes, 1999).
Effects of various therapeutic strategies on activation patterns
The evidence that speech therapy is efficacious is rather limited, and the justification for the application of various modalities of speech therapy to different disturbances of language is rather controversial (Ferro et al., 1999). Functional imaging studies, therefore, may be of value for the demonstration of activating effects in defined locations of the network and of related improvements in language performance. In a PET-CBF study of seven nonfluent aphasic patients during hearing and repetition of single words, the language tasks abnormally activated right hemispheric regions, homotopic to those activated in the normal subject, and deactivated the left hemisphere language zone (Belin et al., 1996). In contrast, repeating words with melodic intonation therapy reactivated Broca's area and the left prefrontal cortex, while deactivating the counterpart of Wernicke's area in the right hemisphere. The recovery process induced by melodic intonation therapy in these patients probably coincides with this reactivation of left prefrontal structure, whereas the right hemisphere regions abnormally activated during single language tasks seem to be associated with the initial persistence of the aphasia. However, four patients with chronic Wernicke's aphasia benefited from intense language comprehension training. The training induced improvement in verbal comprehension correlated to changes in activated blood flow in the posterior part of the right temporal gyrus and the left precuneus (Musso et al., 1999).
The efficacy of adjuvant drug therapy in the rehabilitation of aphasic patients is even more controversial, because well-designed, large-scale—controlled clinical trials are lacking. For some drugs, beneficial effects of adjuvant pharmacotherapy were shown (Small, 1994; Wallesch et al., 1997). For example, a significant improvement of aphasia in patients who received piracetam has been repeatedly demonstrated (Huber et al.,1997; Orgogozo, 1998). In a small prospective, double-blind, placebo-controlled trial in 24 patients with poststroke aphasia (Kessler et al., 2000), the piracetam group (12 patients, receiving 2.4 g piracetam twice daily for 6 weeks) improved in 6 language functions of the test battery and showed a significantly higher activation during word repetition in left temporal and inferior frontal areas than the placebo group (12 patients), which improved in 3 subtests and activated the left vocalization area only. This study not only suggested a beneficial adjuvant effect of piracetam, but again stressed the eminent role of undamaged and reactivated left hemispheric regions for relevant restoration of language function after poststroke aphasia.
LIMITATIONS AND CONFOUNDS OF FUNCTIONAL IMAGING STUDIES
The morphologic changes caused by ischemic lesions impose considerable methodologic challenges for the analysis of functional imaging data, because standard regional templates and automated stereotactic normalization routines for statistical parametric mapping are no longer applicable. Individual identification and delineation of anatomic structures is a time consuming process that also requires high quality morphologic images. It is to be expected that further progress in three-dimensional image processing techniques including coregistration of functional and structural images will facilitate these analyses, thus providing tools to study larger samples and to overcome the limitations of current studies that are frequently based on very few subjects. Such small samples often do not permit valid statistical conclusions with regard to the population of interest, but findings must be regarded as significant only in that particular sample that was studied (Friston et al., 1999).
Interpretation of signal changes is not always straight-forward or possible because factors such as practice and changes of mental processes that accompany motor performance (for example, monitoring of movement) may be difficult to control (Poldrack, 2000). Mere imagination of complex movements may activate SMA and premotor cortex (Rao et al., 1993; Roth et al., 1996). The influence of the more easily measurable factors of accuracy of performance, speed (Rao et al., 1996; Jancke et al., 1998) and exerted force (Dettmers et al., 1995), could be controlled for by study design or use of covariates. The latter may also be controlled by pacing with constant frequency. Motor actions are accompanied by proprioceptive sensorimotor perception, which also causes significant activation effects that may be difficult to separate from motor activation proper (Weiller et al., 1996).
The increase of CBF because of functional activation occurs on top of resting CBF, which is often impaired in extensive brain areas after stroke (Pawlik et al., 1985; Weiller et al., 1992). An interaction between resting CBF and activation effects (stronger CBF activation in areas with low resting CBF) has been shown (Weder and Seitz, 1994), but was neglected in many other studies. Because enough details about the mechanism of coupling between CBF changes and changes of neuronal activity (Kuschinsky, 1991) are not known, the physiologic interpretation of altered activation intensity in areas with reduced baseline CBF is unclear. In addition, it is not possible to distinguish increased activation from less deactivation without absolute quantitation (Raichle et al., 1994).
In summary, several functional imaging studies, in particular those that used high resolution techniques capable to look closely at individual anatomy, demonstrated that the best recovery from paresis and aphasia is achieved if the primary areas that are normally responsible for these functions regain functional activity. This finding underlines the importance of immediate treatment in acute stroke to rescue as much of that tissue as possible.
Many studies also indicate that neuronal networks are adjusting to damage of the primary areas. These adjustments often involve increased activation of functionally related areas, often in frontal cortex, and of corresponding areas in the nonaffected hemisphere. However, it is not clear how and whether these adjustments contribute to clinical recovery, or whether they are simply the consequence of the functional disabilities caused by the damage to the primary areas or fiber tracts. The use of the term “neuronal plasticity” currently does not have a clear definition in this clinical context and further research is needed to clarify these issues.
