Abstract
[11C]FLB 457 is a very high-affinity radiotracer that allows the measurement of dopamine D2/3 receptor availability in regions of the brain where densities are very low, such as the cerebral cortex. It is not known if [11C]FLB 457 binding is sensitive to the concentration of endogenous dopamine in humans in a manner analogous to [11C]raclopride and [123I]IBZM in the striatum. To test this possibility, extrastriatal [11C]FLB 457 binding was measured at baseline and after the oral administration of 40 to 60 mg of the psychostimulant methylphenidate (MP) in 12 healthy volunteers using positron emission tomography (PET) in a balanced-order, double-blind design. The dynamic PET data were quantified using a two-tissue compartment model with a metabolite-corrected arterial plasma input function. Two volunteers were excluded because of excessive head movement. In the remainder, MP caused significant reductions in the volume of distribution (VD) in temporal and frontal cortical regions and thalamus, suggesting that [11C]FLB 457 binding is sensitive to endogenous dopamine concentration. Moreover, the change in [11C]FLB 457 binding after MP correlated with the dose of MP (in mg/kg body weight) in all regions assessed. We conclude that MP in doses within the therapeutic range for the treatment of attention deficit hyperactivity disorder causes increases in dopamine concentrations in extrastriatal regions and that [11C]FLB 457 PET may be a useful tool for the assessment of change in dopamine concentration in these areas in humans.
Introduction
The observation that the binding of some radiotracers is inhibited by increased concentration of endogenous ligand (Dewey et al, 1993) has allowed novel measurements to be made in the living human brain (Laruelle, 2000). In particular, the dopamine D2/3 receptor radiotracers [11C]raclopride and [123]I-IBZM have been utilised to index perturbation of dopamine concentrations in the striatum of the normal (Koepp et al, 1998) and abnormal brain (Breier et al, 1997; Laruelle et al, 1996). However, both [11C]raclopride and [123I]-IBZM are relatively low-affinity radiotracers with nanomolar affinity for the D2/3 receptor, thereby limiting the detection of dopamine changes to regions with high concentrations of D2/3 receptors.
Very high-affinity radiotracers, such as the substituted benzamide [11C]FLB 457, which has picomolar affinity for the D2/3 receptor (Halldin et al, 1995; Suhara et al, 1999), bind with sufficient signal-to-noise ratio to extrastriatal D2/3 receptors to allow quantitative measurements of receptor binding in low-density regions such as the temporal and frontal cortices (Olsson et al, 1999). However, it is not known if the binding of [11C]FLB 457 is sensitive to perturbations of endogenous concentrations of dopamine in humans. Two studies performed in ketamine anesthetised primates examining the effect of dopamine concentrations on [11C]FLB 457 binding reached different conclusions. In the first study, three cynomolgus monkeys were examined before and after treatment with 2 mg/kg amphetamine i.v. (Chou et al, 2000). The authors reported reductions in [11C]FLB 457 binding in the thalamus (TH) and cortical regions of between −1.2% and −16.3% using a reference tissue approach. In contrast, in rhesus monkeys administered 1 mg/kg methamphetamine i.v., there was no significant reduction in binding in cortical regions (Okauchi et al, 2001). Two studies in humans have reported changes of [11C]FLB 457 binding in cortical regions after alfentanil (an opiod μ-receptor agonist) (Hagelberg et al, 2004), and a working memory task (Aalto et al, 2005). Although these studies used a variety of different models to analyze their data, they all employed a cerebellar reference region. Using the iodinated analogue of FLB 457 (the single photon emission tomography (SPET) ligand [123I]epidepride) one study in rhesus monkeys found a reduction in striatal binding after 1 to 2 mg/kg amphetamine (Kessler et al, 1993), but another study in baboons reported no striatal change (al-Tikriti et al, 1994) with 1 mg/kg d-amphetamine. One pilot study using [123I]epidepride in humans showed no statistically significant change in striatal binding after low oral dose (20 mg p.o.) d-amphetamine (Tibbo et al, 1997). However, none of these SPET studies examined extrastriatal regions. The alternative approach of reducing dopamine concentrations in the brain with α-methyl-para-tyrosine increased [123I]epidepride binding by 13% in the temporal cortex (TC) of healthy controls suggesting that the tracer is sensitive to extracellular dopamine concentrations (Fujita et al, 2000). All of these SPET studies utilised a reference region approach.
In this study changes in [11C]FLB 457 binding induced by oral administration of 40 or 60 mg of methylphenidate (MP) were measured in healthy volunteers. MP is a dopamine transporter (DAT) blocker which has been shown to increase extracellular concentrations of dopamine in striatal regions in humans (Volkow et al, 1994) and in extrastriatal brain regions in rodents (Wayment et al, 2001). To avoid the confounding influence of potential changes in [11C]FLB 457-specific binding in the cerebellum (CB) (Asselin et al, 2003, 2005) a compartmental model with an arterial plasma input function was used to derive the volume of distribution (VD) in cortical and subcortical (TH) brain regions.
Materials and Methods
Volunteers
Volunteers were recruited by newspaper advertisement and were screened by a trained psychiatrist (AJM). Exclusion criteria were presence of current or previous psychiatric or neurologic disorder, any previous exposure to stimulant drugs, any history of cardiovascular disease or abnormality on ECG. The study was approved by the Hammersmith, Queen Charlotte's & Chelsea and Acton Hospitals Research Ethics Committee. All subjects provided written informed consent before inclusion in the study.
Radiotracer Synthesis
[11C]FLB 457 was prepared as previously described by methylation of the desmethyl precursor using [11C]methyl triflate (Lundkvist et al, 1998) at a specific activity ranging over 25 to 120 MBq/nmol (see Table 1 for details) and a radiochemical purity higher than 95%.
Demographic and radiopharmaceutical data for group of n = 10 and smaller subgroups defined by difference in stable FLB 457 coinjected (see Figure)
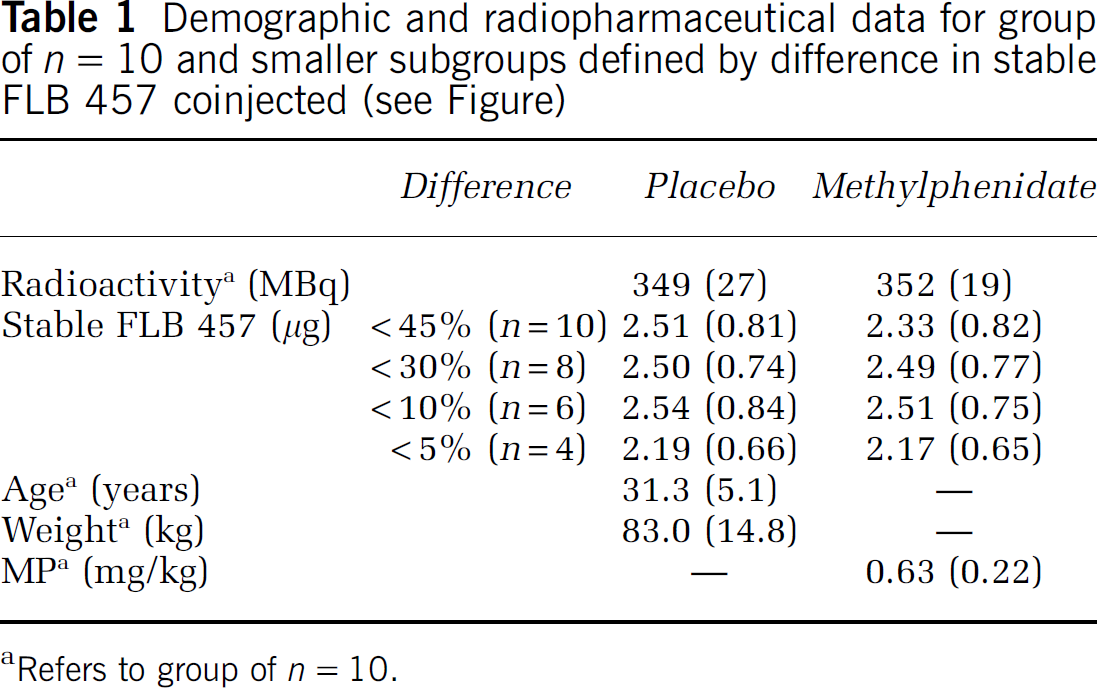
Refers to group of n = 10.
Positron Emission Tomography Scanning Protocol
Twelve male volunteers were scanned on two occasions 21 (mean, range 4 to 102) days apart. MP was administered orally in a balanced order, placebo-controlled, doubleblind manner. Volunteers received either MP 40 mg (n = 6) or 60 mg (n = 6) 60 mins before radiotracer injection. Arterial and venous cannulation were performed 1 h before scanning. Half-an-hour before injection, volunteers were positioned in the CTI/Siemens ECAT EXACT3D tomograph (Spinks et al, 2000). Head movement was minimised using a padded head support and a strap over the forehead. After a 5 mins transmission scan using a 137Cs rotating point source, 280 to 380 MBq of [11C]FLB 457 was manually administered as a bolus over 45 secs. Emission data were acquired over 90 mins in list mode and were subsequently rebinned into the following sequence of 26 frames: variable background (30 to 45 secs), 1 × 30, 1 × 15, 1 × 5, 1 × 10, 1 × 30, 4 × 60, and 17 × 300 secs. The images were corrected for attenuation (Xu et al, 1994) and scatter (Watson et al, 1996) and reconstructed using a reprojection algorithm (Kinahan and Rogers, 1989) with the ramp and Colsher filters set to Nyquist frequency. The transaxial and axial spatial resolutions of the reconstructed images were 5.1 mm and 5.9 mm FWHM, respectively (Spinks et al, 2000). Emission data were corrected after reconstruction for head movement by realigning wavelet denoised images (Battle, 1987) to a 5 mins frame acquired 35 mins after the radiotracer injection using an automated mutual information method (Studholme et al, 1997). For each subject a 3D T1-weighted magnetic resolution imaging (MRI) (TR=21 ms, TE = 6 ms, 35° flip angle, 256 × 256 image matrix, 25 cm FOV, 114 sagittal slices of 1. 6 mm thickness) was obtained using a 1.0T Picker HPQ system.
Subjective Measures of Affect
Five items (‘tenseness‘, ‘alertness', ‘mind-racing’, ‘happiness,’ and ‘buzz’) were self-rated on a scale of 1 to 10 (1 not at all, 10 most ever) before, 70 and 120 mins after MP/placebo administration, the latter two corresponding to 10 and 60 mins after [11C]FLB 457 injection, respectively.
Blood Analyses
After i.v. injection of [11C]FLB 457 the radioactivity concentration in blood was measured continuously using a bismuth germanium oxide (BGO) radiation detection system (Ranicar et al, 1991). In addition, serial discrete blood samples were taken at increasing time intervals throughout the study for the measurement of the radioactivity in blood and plasma in a NaI well counter cross-calibrated to the tomograph. The concentration of [11C]FLB 457 and its radioactive metabolites in plasma were measured using a semiautomated system with on-line sample enrichment, reverse phase high-pressure liquid chromatography, with radioactivity and absorbance detection (Luthra et al, 1993). To generate the input function the plasma over whole blood ratio was fitted to a monoexponential increase to a constant and the parent fraction to a sigmoid decrease to a straight line using nonlinear least squares. The plasma input function was obtained by multiplying these two curves with the blood time–activity curve (TAC).
Plasma protein binding was determined by an ultrafiltration method using centricon units with 30 kD cut-off filter (Amicon). Plasma sample (1 ml) was mixed with [11C]FLB 457and incubated at 37°C for 10 mins. An aliquot (20 to 50 ml) was taken for the measurement of radioactivity in the sample using an automatic gamma counter. The remainder of the mixture was loaded into the centricon unit and centrifuged at 1,000g for 15 mins. An aliquot (20 to 50 ml) was taken from the ultra-filtrate and the radioactivity measured as described above. The amount of nonspecific binding of [11C]FLB 457 to the centricon filter was also determined by addition of [11C]FLB 457 to saline and following the same procedure described above for plasma sample. The free unbound fraction in plasma corrected for nonspecific binding to the filter was calculated by using the following formula:
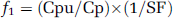
SF is the stick factor (Csu/Cs) that is radioactivity in a known volume of saline that was ultra-filtered divided by radioactivity in the same volume of saline loaded into the unit.
Kinetic Analyses
Regions of interest (ROI) were derived from a probabilistic brain atlas, described in Hammers et al (2003). Using SPM99 (Wellcome Department of Cognitive Neurology, University College, London, UK (Ashburner and Friston, 1999)), the atlas was transformed to the individual's MR space by applying parameters derived from the transformation (linear and nonlinear) of the Montreal Neurological Institute (MNI) single subject brain to the individual's MRI. The individual's MRI was then segmented into grey and white matter and cerebrospinal fluid (CSF) components using SPM99 with the grey matter probability map thresholded at 30%. Using this segmentation the atlas was then divided into grey and white matter and CSF components. Finally, the segmented atlas was resliced into the individuals PET image using parameters derived from the coregistration of the individual's MRI to the summated PET image (frames 1 to 26) with a mutual information method (Studholme et al, 1997). The ROI selected from the atlas were amygdalla/hippocampal formation (AH), frontal cortex (FC), TC, anterior cingulate gyrus (AC), TH, and CB. Regions of interest from the right and left hemispheres were combined. Tissue TAC's were generated by overlaying these ROI on the dynamic PET images using the image analysis software Analyze AVW 3.0 (Biomedical Imaging Resource, Mayo Foundation). Time-activity curves were fitted to a two-tissue compartment, four-rate constant model (Asselin et al, 2005; Olsson et al, 1999) with fixed delay and variable blood volume using a metabolite-corrected plasma input function. The rate constants were used to estimate the total VD = K1/k2(k3/k4 + 1), where K1 and k2 describe the forward and reverse transport of the radioligand between plasma and the combined free and nonspecific binding pool, and k3 and k4 are the rate constants to and from the specific binding pool.
For comparisons with previous primate studies (Chou et al, 2000; Okauchi et al, 2001), data were also analysed with the simplified reference tissue model (SRTM) (Lammertsma and Hume, 1996) using the cerebellar TAC as an indirect input function.
Statistical Analyses
All values are expressed as mean (s.d.). Statistical analyses were performed using the Statistical Package for Social Sciences 12.0 (SPSS Inc., Chicago, IL, USA). Differences across conditions were compared using repeated measures analysis of variance (ANOVA). Post hoc t-tests were two tailed. Analysis of covariance (ANCOVA) was used to assess the relationship between covariates and dependent variables. Significance was set at the P < 0.05 level.
Results
Volunteers
Of the 12 subjects scanned, two were excluded because of excessive head movements during scanning which was not adequately compensated for by the frame-by-frame realignment. Five of the remaining 10 subjects received 40 mg methylphenidate and five 60 mg. Six volunteers had placebo followed by MP, whereas the order was reversed in the other four volunteers. Unless otherwise stated the following results refer to the group of 10 volunteers.
Radiochemistry
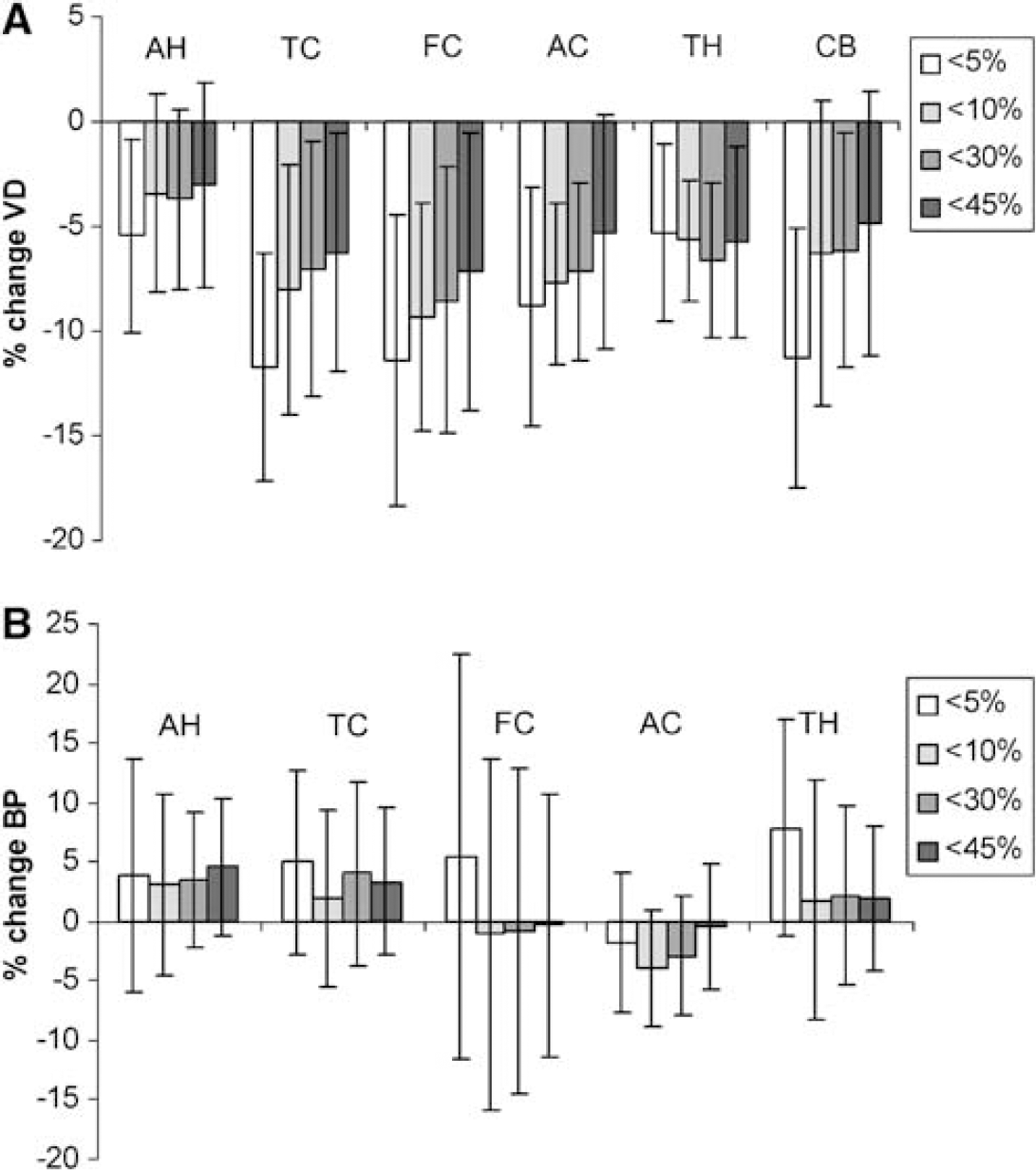
Data are presented along with demographic data in Table 1. There were no significant differences in either radioactivity or total mass injected of ligand between the two conditions. Percentage difference in stable FLB 457 for individual subjects is shown in Figure 2, and used for subject grouping.
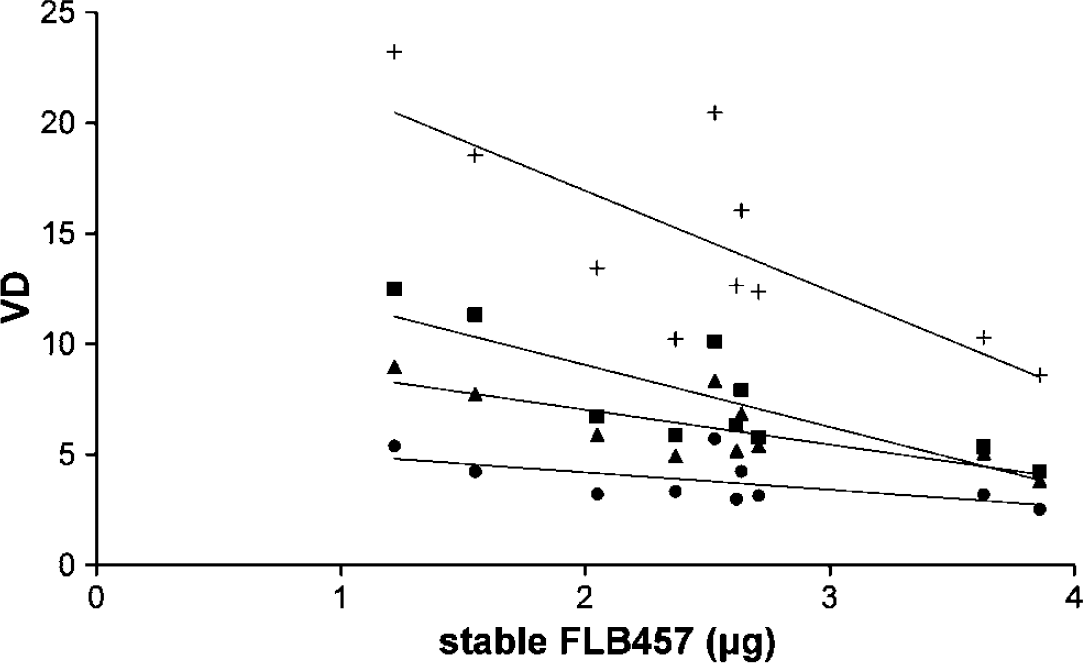
Decrease in regional VD with increasing mass of coinjected stable FLB 457 (TC: temporal cortex (■); FC: frontal cortex (▲); TH: thalamus (+); CB: cerebellum (●)).
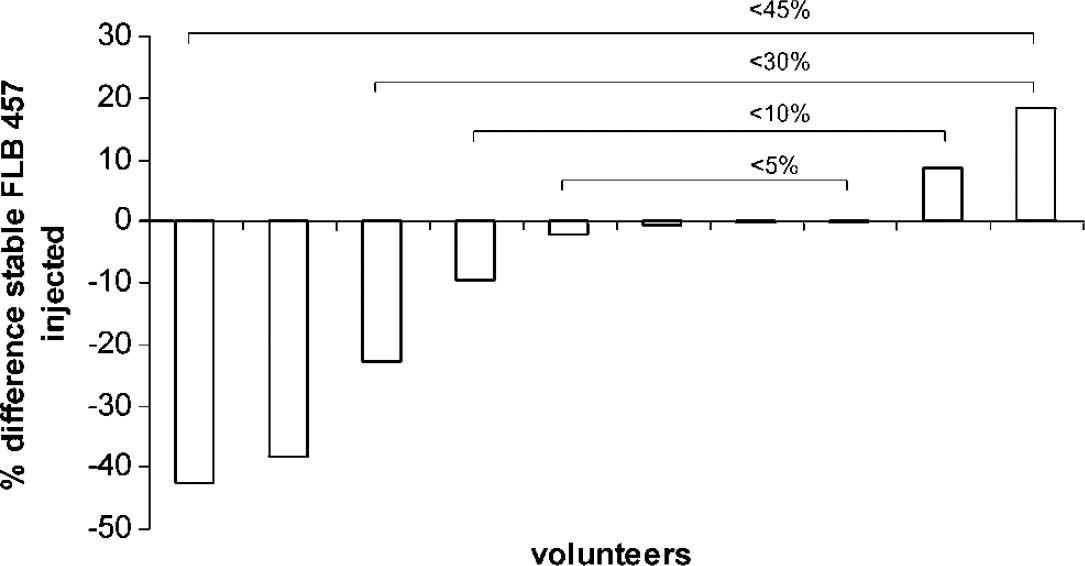
Percentage difference in stable FLB 457 between the placebo and methylphenidate (MP) conditions (= 100 x [{stable MP—stable placebo}/{(stable MP +stable placebo)/2}]). Four different subject groups (indicated by brackets) comprising subjects with different degrees of match in stable ligand were compared (results shown in Figure 3). Negative values indicate less stable FLB457 injected in the MP scan compared with the placebo scan.
Blood Data
The concentration of unchanged [11C]FLB 457 in plasma decreased with time and represented 94 (2)% and 20 (4)% of the total radioactivity in plasma at 5 and 60 mins post-injection, respectively. The radioactive metabolites were more polar than [11C]FLB 457. There was no significant difference in the time course of [11C]FLB 457 in plasma between placebo and MP conditions (rmANOVA: df=1,9, F = 2.88, P =0.12). There was no significant difference in the free fraction of [11C]FLB 457 in plasma (f1) between placebo (8.0 (2.4)%) and MP (8.6 (1.0)%) conditions (t = −0.28, df=5, P = 0.79) (complete data available only for six subjects for technical reasons).
Tissue Data
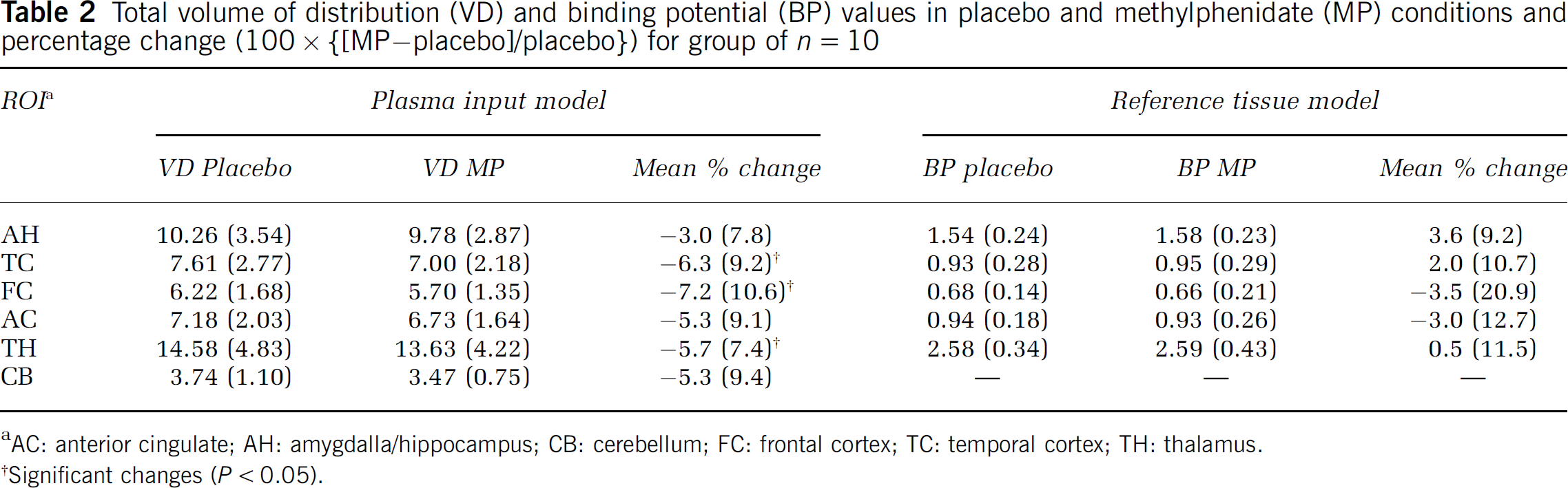
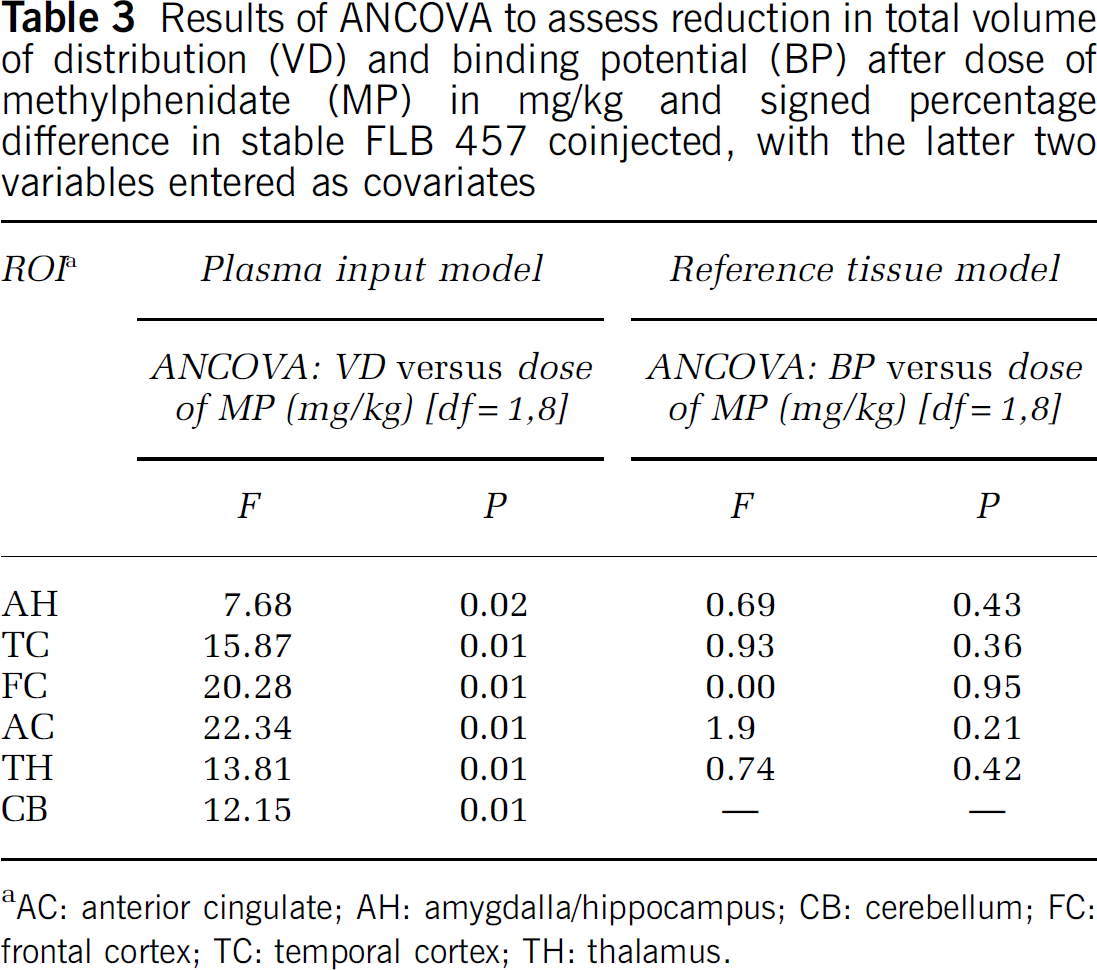
Plasma input model: A significant negative correlation was observed between VD and the mass of coinjected stable FLB 457 in all regions (AH r= −0.83, P < 0.01; TC r =−0.84, P < 0.01; FC r = −0.78, P = < 0.01; AC r = −0.74, P < 0.01; TH r = −0.78, P < 0.01; CB r = −0.63, P =0.05) (Figure 1). To control for potentially confounding effects of differences in stable FLB 457 between conditions a series of analyses were performed on different groups of subjects with variable degrees of matching between conditions (Figure 2). The effects of methylphenidate on [11C]FLB 457 binding are shown in Figure 3A in these groups. With < 5% difference in coinjected stable FLB 457 significant reductions in VD in all ROI were found. However, as the sample size increased with greater differences in stable FLB 457 significance was lost in the smaller AH and AC ROI. Volumes of distribution for the group of 10 are shown in Table 2. There was a significant effect of condition (placebo/methylphenidate) on VD (rmANOVA: F = 5.96 df=1,9 P =0.04) with a significant interaction between condition and ROI (rmANOVA: F = 3.18 df = 5,45 P =0.01). ANCOVA was used to assess reduction in VD after dose of MP in mg/kg and signed percentage difference in stable FLB 457 coinjected, with the latter two variables entered as covariates. Relative reductions in VD were significantly correlated with increasing dose of MP in the amygdala/hippocampus, FC, anterior cingulate, TC, TH, and cerebellar ROI (Table 3).
Change in (
Total volume of distribution (VD) and binding potential (BP) values in placebo and methylphenidate (MP) conditions and percentage change (100 x {[MP–placebo]/placebo}) for group of n =10
AC: anterior cingulate; AH: amygdalla/hippocampus; CB: cerebellum; FC: frontal cortex; TC: temporal cortex; TH: thalamus.
Significant changes (P < 0.05).
Results of ANCOVA to assess reduction in total volume of distribution (VD) and binding potential (BP) after dose of methylphenidate (MP) in mg/kg and signed percentage difference in stable FLB 457 coinjected, with the latter two variables entered as covariates
AC: anterior cingulate; AH: amygdalla/hippocampus; CB: cerebellum; FC: frontal cortex; TC: temporal cortex; TH: thalamus.
Subjective measures of affect: Within the group of 12 subjects there was a trend effect of drug condition on change in rating scales between baseline (predosing) and 10 and 60 mins after injection (rmANOVA: df = 1,11, F = 3.94, P = 0.07). In the subgroup of n = 10, the effect of drug no longer reached trend level (rmANOVA: df = 1,9, F = 2.75, P = 0.13). Exploratory analyses with ANCOVA were performed to examine the relationships between changes in VD and changes in rating scales after MP within the subgroup of 10 (change in VD and difference in stable FLB 457 coinjected as covariates). There was a significant relationship between increased ‘mind racing’ between the baseline measurement and 10 mins after [11C]FLB 457 injection, and reduction of VD after MP in the AH (F = 6.93, P =0.03), TC (F = 16.29, P =0.01), AC (F = 21.17, P =0.01), FC (F = 21.11, P =0.01), TH (F = 16.51, P =0.01), and CB (F = 5.94, P= 0.04) ROI. These relationships were not present when comparing baseline to the rating scales 60 mins after the start of scan. There were no significant correlations between dose of MP and change in rating scales.
Reference tissue model: Binding potential (BP) also negatively correlated with the mass of coinjected stable FLB 457, but only in the amygdala/hippocampus (r = −0.83, P < 0.01) and TC (r = −0.70, P = 0.02) ROIs. There was no main effect of treatment with MP on BP (rmANOVA: df = 1,9, F = 0.026, P= 0.88) (Figure 3B, Table 2), nor were there significant correlations between change in BP and dose of MP on ANCOVA (Table 3).
Discussion
We have shown that MP, in doses within the normal therapeutic range for the treatment of attention deficit hyperactivity disorder (40 to 60 mg) (Volkow et al, 2001), reduces the binding of [11C]FLB 457 in cortical and subcortical regions of healthy controls, presumably because of the increase in extracellular dopamine concentration associated with MP. Moreover, there is a correlation between the dose of MP and the change in radiotracer binding between placebo and MP. The result is important for two reasons: First it supports preclinical evidence of an effect of DAT inhibition on dopamine concentrations in cortical regions (Marsteller et al, 2002; Wayment et al, 2001). Second, it demonstrates the potential to measure changes in dopamine concentration in these regions. Neuropsychiatric disorders such as schizophrenia have been hypothesised to involve dysregulation of dopamine activity, particularly in the FC (Weinberger et al, 1988). Positron emission tomography examinations with [11C]FLB 457 may allow such hypotheses to be tested.
In the group of 10 subjects mean decreases in [11C]FLB 457 binding after MP administration (mean dose 0.63 mg/kg) were around 5% to 7%. Larger cortical changes were seen in the subgroup which had the most closely matched concentrations of stable FLB 457 (e.g. 12% in frontal and temporal cortices) (see Figure 3A). This compares with values of 11.1 ± 7.0% in a recent paper measuring the effect of 0.3 mg/kg amphetamine in the human striatum (Martinez et al, 2003) using the PET tracer [11C]raclopride (values ranging from 6.0 ± 6.9% to 16.0 ± 11.6% were found in subdivisions of the striatum). In all the subgroups the amount of coinjected stable FLB 457 was greater in the placebo than in the treatment conditions, possibly diminishing the expected MP effect. Methylphenidate increases extracellular dopamine concentrations by DAT inhibition alone rather than also increasing dopamine release as amphetamine does by facilitated diffusion (Kuczenski and Segal, 1997) suggesting that larger changes may be seen with an amphetamine challenge. The observed changes in [11C]FLB 457 binding after oral MP supports preclinical evidence that MP increases extracellular dopamine concentrations in cortical regions, despite the fact that DAT are found in much lower concentrations in cortical than striatal regions (Hitri et al, 1995; Sesack et al, 1998).
In this study, oral methylphenidate was used as the dopaminergic challenge because of its favourable safety profile and ready availability in the UK. The two primate studies conducted examining the effect of increasing extracellular dopamine concentration on binding of [11C]FLB 457 (Chou et al, 2000; Okauchi et al, 2001) both used amphetamines administered intravenously at relatively large doses (2 mg/kg amphetamine and 1 mg/kg methamphetamine, respectively). Doses of this magnitude significantly increase dopamine in microdialysates from the FC in rodents (Moghaddam et al, 1990) and in the striatum in primates (Breier et al, 1997), suggesting that the negative finding of Okauchi et al was not because of an inadequate challenge. Alternatively, the use of a reference tissue approach may be responsible for the result as discussed in detail below.
There is an imprecise relationship between dose of MP and occupancy of DAT, with no correlation reported between striatal DAT occupancy and striatal dopamine release or change in visual analogue scores (VAS) scores (Volkow et al, 2002). We found a relationship between increase in ‘mindracing’ and change in VD (when controlling for difference in FLB 457 injected). However, no such correlation was found with related measures such as ‘alert’ or ‘buzz‘. In contrast to previous studies (for example (Volkow et al, 2001)) we found minimal changes in other psychological measures.
Confounding Factors
The amount of coinjected stable FLB 457 presents a potential confound because of its significant occupancy effects at the specific radioactivities used in this study (Figure 1). To avoid occupancy of the receptors by the ligand itself resulting in significant changes in the measured VD it is necessary to produce the radioligand to a very high specific radioactivity thus allowing relatively small doses (< 0.5 μg) of stable FLB 457 to be coinjected (Olsson et al, 2004). Such high specific radioactivity is a challenge for radiochemistry and has not been required for previously developed low affinity radiotracers such as [11C]raclopride. Based on their simulated data, Olsson et al (2004) estimated that 1 to 3 μg of FLB 457 occupies 5% to 20% of D2/3 receptors. This occupancy is high enough to cause a reduction in VD as the mass of coinjected stable FLB 457 increases, but is not high enough to allow the in vivo KD to be estimated. In cases when very high specific radioactivity cannot be assured it is necessary to adopt alternative strategies to compensate for any occupancy effect.
In this study, we have reported a variety of different analyses comparing VD between groups of subjects with varying differences in the amount of radioligand injected in each scan (Figures 2, 3A and 3B). With n = 4 the scans were highly matched for stable FLB 457 (< 5% difference) and the changes in VD attributed to MP were relatively large. As the number of subjects increased to 6 and 8 (< 10% and 30% difference, respectively), the mean changes seen were smaller because of the inclusion of subjects in whom the effect of coinjected stable FLB 457 opposed the MP-induced changes. In these groups the amount of stable FLB 457 was closely matched at the group level, although in some subjects there were differences (Table 1 and Figure 2). When the analysis was expanded to include all 10 subjects the changes lost significance in some regions because the mass of coinjected stable FLB 457 was greater in the control than in the treatment conditions, opposing the changes in VD produced by the MP. Correlational analyses revealed a significant dose–response relationship between MP and change in VD in several regions, when difference in stable FLB 457 was covaried (Table 3). Significant correlations between dose of MP and change in VD in the anterior cingulate, FC and CB were revealed by limiting the difference in coinjected stable FLB 457 (and hence its effect on VD) to < 10% (data not shown).
In humans MP (0.5 mg/kg) has been shown to decrease cerebral blood flow (CBF) globally by 20% to 25% compared with baseline after i.v. administration of the drug (Wang et al, 1994). The parameters K1 and k2 have the same flow dependence such that their ratio, as it appears in VD, is flow independent. In the present study, the value of K1 decreased by up to 25% after the highest dose (1 mg/kg) of MP (data not shown). Changes in CBF may affect the estimation of VD if CBF varies markedly during the scan (Logan et al, 1994). Brain (Volkow et al, 1998) and blood (Wargin et al, 1983) concentrations of MP have been measured to be relatively stable after oral administration during our scanning period (between 60 and 150 mins after drug administration) suggesting that changes of CBF because of MP are unlikely to have occurred during the scans. Furthermore, after i.v. methylphenidate administration (which will have induced more rapid changes in blood and brain concentrations of MP than in our oral study) there was no significant difference in the reduction in CBF between 5 to 10 and 30 mins in five healthy human volunteers (Wang et al, 1994) supporting the view that significant changes in CBF are unlikely to have occurred during our scans.
Choice of Model
Analysis of this data set only revealed effects of MP on [11C]FLB 457 binding when a compartmental model with an arterial plasma input function was used. In contrast, the SRTM did not reveal the treatment effect. This is probably because of some of the assumptions of the SRTM are violated with [11C]FLB 457, namely, that the target and reference regions are appropriately fitted by a one-tissue compartment model, and particularly that there is no specific binding in the reference region (Delforge et al, 1999). D2/3 receptors are present in the CB in non-negligible concentrations in comparison to cortical levels. For example, using the D2/3 antagonist [3H]spiroperidol and dopaminergic agonist [3H]CV 205 to 502 and autoradiography, D2/3 concentrations within the CB were between 10 and 20% of those found in the FC (Camps et al, 1989). With the very high-affinity tracer [125I]epidepride concentrations of D2/3 receptors in the CB were 75% of frontal cortical concentrations (Hall et al, 1996). Moreover, D3 receptors to which [11C]FLB 457 binds are found in significant concentrations in regions of the CB (Stanwood et al, 2000). The functional significance of these receptors is unknown but there is evidence of dopaminergic innervation of the cerebellum (Panagopoulos et al, 1991). In our study, the VD of [11C]FLB 457 in the CB after MP administration was reduced by −5.3(9.4)% which obscured changes in binding in target regions when a reference tissue method was applied. This would suggest caution is necessary in the interpretation of PET studies which have used the SRTM with FLB 457.
The VD was chosen over the k3/k4 ratio even though, in theory, the latter is a more specific binding parameter than the former which includes nonspecific binding. However, it is difficult to estimate the k3/k4 ratio reliably. The individual rate constants tend to be correlated and for this reason, VD is more robust to noise. Volume of distribution is linearly dependent on k3/k4, and can be used as an index of specific binding as long as nonspecific binding (K1/k2) and the free fraction in plasma (f1 are not altered between conditions. Because of the lack of a suitable brain reference tissue, it was not possible to assess changes in non-specific binding after MP. However, the potential contribution of such putative change is likely to be small since studies in rats demonstrate that nonspecific binding makes a relatively small contribution to total binding, even in regions of low specific binding (Asselin et al, 2005). Furthermore, MP did not change the plasma-free fraction compared with placebo.
[18F]Fallypride is another very high-affinity D2/3 PET radioligand which has been investigated as a possible tool for the assessment of extrastriatal dopamine release. An early study in rhesus monkeys suggested that striatal [18F]fallypride binding may be sensitive to amphetamine challenge (Mukherjee et al, 1997). More recently, it has been demonstrated that [18F]fallypride binding in baboons is reduced after amphetamine, in a dose-dependent fashion, in striatal and several extrastriatal regions (Slifstein et al, 2004a, b ). No significant change in binding was found in the cingulate cortex, which was the only frontal cortical region examined. Binding potential decreased with stable fallypride and increased at higher specific activity (Slifstein et al, 2004a), suggesting that stable fallypride may have a significant effect on binding in a manner similar to FLB 457. A recent study in rhesus monkeys also reported reduced binding in extra-striatal regions after amphetamine (0.6 to 1.1 mg/kg), but could not assess changes in the FC (Mukherjee et al, 2005). Compared to [18F]fallypride the advantages of [11C]FLB 457 include lower dosimetry, sensitivity to binding changes in the FC and relatively short scan duration. [18F]Fallypride allows striatal and extrastriatal measures to be obtained in one scan, and if a bolus followed by infusion method is adopted, two measurements under different conditions may be made during one single scan (Slifstein et al, 2004b). However, the scanning time of 480 mins required for this purpose limits the use of [18F]fallypride for applied clinical studies. In addition, [18F]fallypride will also require a plasma input function if the CB is shown to have significant specific binding, which is likely for this ligand having an affinity for D2/3 receptors similar to that of [11C]FLB 457.
In conclusion, we have demonstrated that [11C]FLB 457 binding in extrastriatal regions, including FC, is sensitive to the influence of local changes in dopamine concentration. Moreover, the change in [11C]FLB 457 binding was proportional to the administered dose of MP. Larger changes in VD may be seen with an amphetamine challenge or with higher specific activity of the radioligand. [11C]FLB 457 PET studies may prove to be a valuable tool for the assessment of extrastriatal dopamine function in neuropsychiatric disorders.
Footnotes
Acknowledgements
We thank all volunteers for their time and gratefully acknowledge the technical support of the staff of Hammersmith Imanet. We also thank Drs Sue Hume and Vin Cunningham for invaluable discussions.
