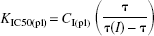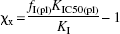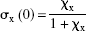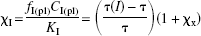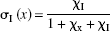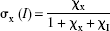Abstract
The half-inhibition concentration (IC50) of a drug indicates its ability to inhibit the binding of other ligands of a receptor. The authors used positron emission tomography to test the hypothesis that haloperidol's IC50 toward the binding of tracer N-[11C]methylspiperone ([11C]NMSP) in brain must be increased in patients in whom more dopamine is bound to receptors than in healthy volunteers. The IC50 of haloperidol was significantly elevated from 1.5 nmol/L in healthy volunteers and patients with bipolar disease without psychosis to 4.5 nmol/L in patients with schizophrenia or bipolar disease with psychosis. The higher IC50 values in psychosis are consistent with an 8-fold increased binding of dopamine and a 16-fold elevated concentration of synaptic dopamine in psychosis. At the 80% haloperidol blockade of the receptors, the calculated amount of neurotransmitter bound in the patients with psychosis declined to twice the value estimated in the nonpsychotic subjects, that is, 5 pmol cm−3.
Previous studies (Wong et al., 1986c, Dewey et al., 1991, 1993; Laruelle et al., 1996) have claimed an effect of endogenous dopamine on the binding of neuroleptics and analogous radioligands to D2 -like receptors in primate (human and monkey). Recently, Abi-Dargham et al. (2000) reported findings suggestive of increased dopamine concentration in brain of patients with schizophrenia, compared with healthy volunteers. When increased baseline dopamine concentration is associated with increased dopamine binding to receptors, it must raise the half-inhibition concentration of exogenous antagonists of this binding. The authors tested this prediction by comparing measures of the half-inhibition concentration of haloperidol in healthy volunteers and in volunteers with psychosis.
To test the prediction, the authors derived a theoretical basis for the in vivo determination of the half-inhibition constant of an exogenously supplied inhibitor. To specifically test the hypothesis that the apparent affinity to haloperidol must decline in states of psychosis in which the competition from dopamine is increased, the authors determined the apparent affinity for this neuroleptic in previously examined healthy volunteers, volunteers with bipolar disease with or without psychosis, and volunteers with schizophrenia (Wong et al., 1997b).
The results are consistent with an elevation of the concentration of dopamine in patients with psychosis, whether from schizophrenia or bipolar illness, but decline of plasma protein binding or intrinsic receptor affinity for haloperidol also explain this finding.
MATERIALS AND METHODS
Operational equation
In the Appendix, the operational equation is derived, which was used to determine the half-inhibition plasma concentration of an unlabeled neuroreceptor antagonist by means of a tracer radioligand. The in vivo estimation of the half-inhibition concentration (IC50) of an inhibitor of specific neuroreceptors requires (at least) two measurements of the rate or degree of binding of the radioligand, for example, in the presence and absence of the inhibitor (Gjedde et al., 1986; Gjedde and Wong, 1990a, b ). The authors used the radioligand 3-N-[11C] methylspiperone ([11C]NMSP) to determine tho degree of dopamine D2 -like receptor occupancy of the neuroleptic haloperidol in caudate nucleus, the dopamine D2 -like receptors being the only significant neuroreceptors shared by [11C]NMSP and haloperidol (Wong et al., 1986b). The following six equations were derived in the Appendix and used to interpret the experimental results. They are listed here to show how estimates of the quantities of endogenous ligand and competitor can be made from the binding rate constant (k3) or time constant (τ = 1/ k3).
The assumptions underlying the equations include the claims that haloperidol and [11C]NMSP bind to the same binding site with single affinities, no change of the maximum binding capacity Bmax occurs between the two administrations of the tracer, no average differences of haloperidol free fractions in plasma and brain tissue exist between the groups of subjects, the true aqueous receptor affinity for haloperidol in vivo (KI) can be known (although it may not be known correctly) and is the same for all groups and both injections, haloperidol is in steady state, the plasma aqueous-free haloperidol concentrations equal the synaptic aqueous-free haloperidol concentrations, no differences of free fractions exist between the groups, and the binding of [11C]NMSP is unidirectional (that is, the dissociation constant koff is effectively nil) in all groups and at both injections. The effect(s) of departures from these assumptions are examined in the Appendix.
The operational equation relates the plasma IC50 of haloperidol to the binding time constant of the tracer,
where KIC50(pl) symbolizes the plasma IC50, CI(pl) symbolizes the steady-state plasma concentration of haloperidol, and τ symbolizes the time constant of unidirectional binding of the tracer [11C]NMSP, equal to the reciprocal of the binding rate constant (1/ k3) in the absence (τ) and presence (τ(I)) of the exogenous inhibitor. The symbol in parentheses, I, refers to the presence of the exogenous inhibitor haloperidol, that is, at the second tomography session, 4 hours after the oral administration of haloperidol. To calculate KIC50(pl), the authors measured the plasma concentration of the inhibitor and the binding time constants of the tracer. With this value, the authors estimated the concentrations and occupancies of hypothetical endogenous competitors.
Equations of inference
To evaluate the influence of changes of the half-inhibition concentration of the inhibitor, five additional equations were derived, which allowed the authors to make inferences about several factors that affect the interpretation of the results of the study. These equations include:
The equation relating the concentration of one or more postulated endogenous competitors to the binding of haloperidol, the concentration expressed relative to the competitors' unknown Michaelis half-inhibition concentrations,
where χx is the sum of the putative endogenous competitors, relative to their half-saturation constants, ΣC x/K x, and where f I(pl) is the plasma “free” fraction of haloperidol at steady state. K I is the aqueous half-inhibition concentration of haloperidol in the absence of other inhibitors of the binding, that is, in the absence of the putative endogenous inhibitors. Calculation of χx requires that K IC50(pl) is known from Eq. 1 and that the free fraction of the inhibitor and the aqueous half-inhibition concentration of the inhibitor is known from the literature or measured separately in plasma.
The equation for the occupancy achieved by the putative endogenous competitor(s) in the absence of the exogenous antagonist,
where σ x (0) is the occupancy at the receptors achieved by the putative endogenous competitor(s) in the absence of exogenous inhibition. The calculation of σ x (0) required that χ x is known from Eq. 2.
The equation relating the concentration of the exogenous inhibitor relative to its inhibitory constant to the binding time constants and the sum of the relative concentrations of other competitors,
where χ I is the concentration of the exogenous inhibitor relative to its inhibitory constant. The calculation of χ I requires that χ x is known from Eq. 2.
The equation for the occupancy achieved by the exogenous antagonist in the presence of the endogenous competitor(s),
where σ I (x)is the occupancy achieved by the exogenous inhibitor in the presence of the endogenous competitor(s). The calculation of σ I (x) requires that χ x is known from Eq. 2 and that χ I is known from Eq. 4.
The equation for the occupancy achieved by the putative endogenous competitor(s) in the presence of the exogenous antagonist,
where σ x (I)is the occupancy achieved by the putative endogenous competitor(s) after the blockade of the receptors by the exogenous inhibitor. The calculation of σ x (I) requires that χ x is known from Eq. 2 and that χ I is known from Eq. 4.
These equations were derived in the Appendix for a radioligand that binds unidirectionally to the neuroreceptors during the time available for positron emission tomography. In addition, in the Appendix, the criteria are established that must be fulfilled for the authors to claim that binding of the radioligand was in fact unidirectional for the duration of the positron emission tomography.
Volunteers
All healthy subjects and patients volunteered and gave their informed consent to the study, in compliance with the Johns Hopkins Committee on Clinical Investigation. The details of the volunteers participating in this study were presented previously (Wong et al., 1986a, c , 1997b). The distribution of the subjects between the years 1986 and 1997 is listed in Table 1.
Number of subjects in 1986 and 1997 cohorts

Briefly, 14 patients with bipolar affective disorder (7 men and 7 women, mean age = 43 years), 22 drug-naive patients with schizophrenia (17 men and 5 women, mean age = 36 years), 5 drug-free but previously medicated patients with schizophrenia (mean age = 27 years), and 22 healthy volunteers (17 men and 5 women, mean age = 33 years) were recruited. Of the patients with bipolar disease, 11 were drug-naive and 3 drug-free for at least 2 months. Of the patients with bipolar disease, 3 were depressed and 11 were manic at the time of study.
On the basis of DSM-III, -IIIR, and -IV criteria as described in Wong et al. (1986a, c , 1997b), the patients with bipolar disease were divided in two groups—patients without episodes of psychosis and patients with episodes of psychosis during the tomography. Subjects were divided categorically into those who were psychotic by definition at all times for the drug naive schizophrenic patients or specifically at the time of scanning for the psychotic bipolar patients, in contrast with the nonpsychotic bipolar patients. In patients with bipolar disease, the episodic nature necessary required the identification of active hallucinations and delusions during the positron emission tomography scan time. Patients with bipolar illness who exhibited active hallucinations and delusions were considered psychotic, whereas those that did not present with these symptoms were considered nonpsychotic. Patients and healthy volunteers did not differ significantly with respect to blood pressure, pulse, weight, and nutritional status at the time of study. Further clinical details were given by Tune et al. (1993) and Pearlson et al. (1993, 1995).
Tomography
The tomography is described in details in previous articles by Wong et al. (1986c, 1997a). Briefly, in each patient, the binding time constant of N-[11C]methylspiperone, labeled according to Burns et al. (1984) to the high specific activity of 2 mCi pmol −1, was measured twice by positron emission tomography after 10- to 20-second injection of 20 mCi into an antecubital vein. The injected mass averaged 30 pmol (kg body weight) −1. The specific activities did not differ between the injections.
Each tomography session lasted 90 minutes. Emitted radioactivity was recorded with the NeuroECAT tomograph (CTI; Knoxville) in its highest resolution mode. Radioactivity was simultaneously detected in caudate nucleus and cerebellum in 5 frames of 2 minutes, 5 frames of 5 minutes, and single frames of 15 and 30 minutes each. Between the 2 tomography sessions, 4 hours before the second session, all subjects ingested 300 to 400 nmol kg −1 haloperidol resulting in steady-state plasma concentrations that averaged less than 10 nmol/L in subjects without psychotic episodes and more than 10 nmol/L in subjects with such episodes.
Blood samples for radioactivity and haloperidol concentration measurements were obtained from the dorsal vein of the hand contralateral to the injection hand, after “arterialization” by heating to 44°C, and, in some cases, from the radial artery. The sampling schedule consisted of 4 to 6 samples the first minute after the tracer injection, 3 to 6 samples the second minute, 2 samples the third and every subsequent minute until the tenth minute, and samples at 25, 30, 45, 60, 75, and 90 minutes. Blood samples were centrifuged and samples of the plasma were counted in a gamma-scintillation spectrometer. In previous comparisons, results obtained with arterialized venous sampling did not differ significantly from results obtained with arterial sampling (Wong et al., 1997a).
Quantitative analysis
Quantitative analysis of the studies was performed by an investigator blinded to the clinical diagnosis, as described by Wong et al. (1986a, 1986b, 1986c). Calculations of IC50 and χx were made from values of the time constants of unidirectional binding of tracer [11C]NMSP in the caudate nucleus in the absence and presence of haloperidol in all subject groups, according to Eqs. 1 and 2. Analysis of variance detected the significant differences among the patient groups.
Time constants (τ = 1/ k3) of unidirectional binding of tracer [11C]NMSP were directly estimated from the time-radioactivity curves in brain tissue, as described by Gjedde et al. (1986). Plasma haloperidol concentrations were determined as described by Wong et al. (1997b) and the grand average of the measurements was used as listed in Table 2. The average free fractions [fI(pl), mean ± SD (n)] for [18F]haloperidol of the individual groups were 0.035 ± 0.002 (10) for schizophrenia, 0.041 ± 0.008 (5) for bipolar illness, and 0.033 ± 0.005 (7) for controls.
Constants used in analysis of NMSP and haloperidol binding
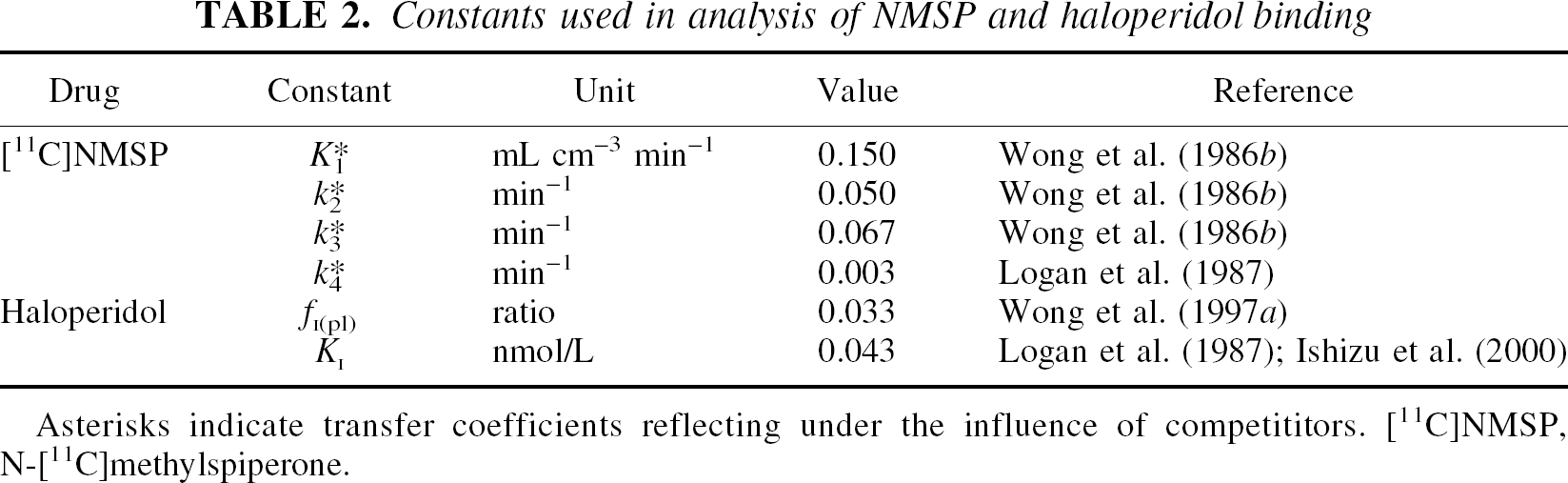
Asterisks indicate transfer coefficients reflecting under the influence of competititors. [11C]NMSP, N-[11C]methylspiperone
Putative endogenous competitor concentration ratios were computed from Eq. 2, using the value of the haloperidol “free” plasma fraction (fI(pl)) of 0.033 reported by Wong et al. (1997a), and an aqueous haloperidol half-inhibition concentration of 0.043 nmol/L in the absence of endogenous or other exogenous inhibitors, as reported by Ishizu et al. (2000) for the pig and listed in Table 2.
Equations 3 to 6 were used to predict the degrees of receptor occupancy established by the putative endogenous competition and exogenous inhibition and the actual neurotransmitter binding to the receptors in the groups of volunteers.
RESULTS
The authors confirmed the unidirectionality of [11C]NMSP uptake into the caudate nucleus by modeling the uptake with and without dissociation from the receptors at the rate reported by Logan et al. (1987) and listed in Table 2. The plasma free fraction of haloperidol listed in Table 2 was measured in the second group of subjects added to the Wong et al. (1986c) group by Wong et al. (1997a). The KI of haloperidol in aqueous solution (listed in Table 2) was calculated from studies of people by Logan et al. (1987) and pigs by Ishizu et al. (2000).
The [11C]NMSP dissociation rate constant k4 of 0.003 min−1(0.18 h−1) was not sufficient to render the binding less than approximately unidirectional during the 90-minute tomography, assuming unlimited specific activity of the tracer. In the 90 minutes, the accumulation was 95% of the accumulation expected with complete unidirectionality. In terms of the total amounts of radioligand, the authors estimated that the s/σ ratio (Appendix, Eqs. 12 and 14) never exceeded 0.05 during the 90 minutes and that the transfer therefore must be effectively unidirectional, even if the inclusion of dissociation would yield different estimates of the magnitude of τ. Therefore, the authors limited the regression for τ to three parameters.
In agreement with the previous analysis (Wong et al., 1997b), the average estimates of the magnitude of τ did not differ among the groups, except for the nonpsychotic patients with bipolar disease. Plasma haloperidol concentrations did not vary significantly among the subject groups, although the concentration averages were twice as high in the psychotic patients, as shown in Table 3. This variation is well known from the literature (Midha et al., 1989; Goff et al., 1991) but remains unexplained. When the measurements were lumped according to the patients' condition at the positron emission tomography measurement (nonpsychotic or psychotic), the plasma haloperidol concentrations were significantly elevated in the psychotic subjects. The average neuroleptic blockade corresponded to group mean haloperidol occupancies of 70% to 87%.
Average IC50 of haloperidol in psychosis
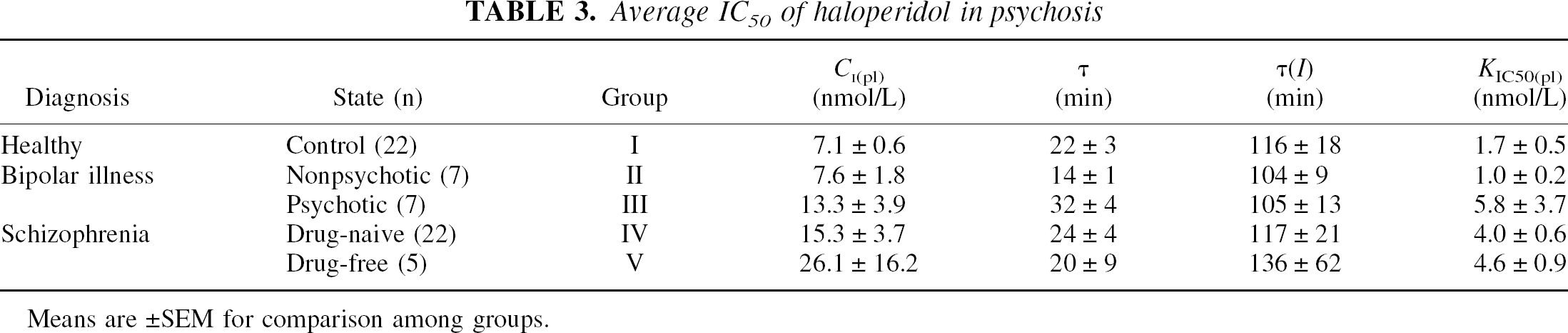
Means are ± SEM for comparison among groups.
The values of the haloperidol IC50 (KIC50(pl)) were calculated from Eq. 1 as normally distributed, because denominator values generally were large rather than small. Averages differed significantly among the five groups (F = 2.81, P = 0.05). The apparent affinity was significantly decreased in the drug-naive patients with schizophrenia, compared with healthy volunteers (t = 2.84, P = 0.008). Multiple regression against age showed a significant relation between the IC50 and age in the patients with bipolar disease who had had psychotic episodes (K IC50(pl) = 1.5 nmol/L + [0.1 nmol/L year−1] [age in years], P = 0.01). No other group showed a significant change of IC50 with age, the values deviating insignificantly from the 1.5 nmol/L average for healthy volunteers and patients with bipolar disease without episodes of psychosis, and 4.5 nmol/L for patients with schizophrenia or psychosis of other origin.
DISCUSSION
In 1986, the authors reported that therapeutic doses of haloperidol block 70% to 90% of the dopamine receptors imaged by [11C]NMSP (Wong et al.,1986b), but they also found that the IC50 of haloperidol was higher in patients suffering from psychosis than in subjects not so afflicted (Wong et al.,1986c). Hence, the results of these experiments do not reject the hypothesis that patients with psychosis have a higher endogenous neurotransmitter binding to dopamine receptors than subjects without psychosis (Abi-Dargham et al., 2000). Nonetheless, numerous assumptions underlie the equations used, and multiple factors influence the results. Increased endogenous neurotransmitter level is only one among a multitude of possible interpretations of the data. A theoretical description of some of the many possible interpretations is given in the Appendix.
Increased intrasynaptic dopamine has been hypothesized since the earliest formulation of the dopamine theory of schizophrenia. Early evidence originated from an in vivo study of the binding of [11C]NMSP in ventral striatum and nucleus accumbens in schizophrenia (Wong et al., 1986c). Using the procedure described here, the authors noted an apparent increase of the haloperidol affinity constant in the patients, which was interpreted as a possible increase of endogenous dopamine (footnote 16, Wong et al., 1986c). In the current study, the authors extend the calculation to include bipolar illness and provide a theoretical basis for the interpretation of the results in terms of dopamine binding, as shown in Table 4.
Predicted neurotransmitter binding in psychosis

CI(pl) is the concentration of the exogenous inhibitor in arterial plasma. KIC50(pl) is the apparent half-inhibition concentration of the inhibitor in arterial plasma. χx is the sum of putative endogenous competitors, relative to their half-saturation concentrations. Bmax(40) is the maximum binding capacity in ventral caudate nucleus at age 40 reported by Wong et al. (1997b). σx(0) is the occupancy of the endogenous competitor(s) in the absence of exogenous inhibition. Bx(40) is the predicted binding of neurotransmitter x at age 40. χI is the concentration of exogenous inhibitor, relative to its inhibitory constant. σI(x) is the occupancy of the exogenous inhibitor in the presence of the endogenous competitor(s). σI(x) is the occupancy of the putative endogenous competitor(s) in the presence of the exogenous inhibitor. Bx(1)(40) is the predicted binding of neurotransmitter x after neuroleptic blockade at age 40. Remaining symbols are defined in relation to the equations indicated in the table.
For the purpose of inference about dopamine binding, the group averages were lumped into the three classes listed in Table 4. The class averages formed the basis of speculations on the quantities of free and bound neurotransmitter and antagonist, incorporating previously published estimates of the maximum binding capacities (Bmax) of the three subject classes (Wong et al., 1997 b). Using the same value of the plasma “free” fraction fI(pl) for all groups, and the aqueous half-inhibition concentration KI of 0.043 nmol/L for haloperidol listed in Table 2, the total relative concentration of putative endogenous competitors averaged 0.15 in the nonpsychotic or healthy subjects, corresponding to an average receptor occupancy of 13%. The quantity of bound endogenous competitors was estimated to be 2.5 pmol cm−3 at a total [11C]NMSP receptor density of 19 pmol cm−3 at age 40.
In the psychotic patients with bipolar disease or drug-naive schizophrenic patients, the total relative concentration of putative endogenous competitors averaged 2.45, a 16-fold increase, corresponding to an 8-fold increase of receptor occupancy (71%, or 21 pmol cm−3 of a total [11C]NMSP receptor density of 29 pmol cm−3 at age 40). After haloperidol blockade, the putative endogenous competitor occupancy declined to 2.2% in the combined groups I and II and to 16% in the combined groups III and IV (0.4 pmol cm−3 and 4.7 pmol cm−3, respectively), as shown in Table 3. Thus, even at the higher plasma haloperidol concentration, patients in groups III and IV had a lower haloperidol occupancy and a 12-fold higher putative endogenous neurotransmitter binding than the subjects in groups I and II.
Notably, the predicted residual neurotransmitter binding in the haloperidol-blocked patients in groups III and IV was only twice as high as the predicted neurotransmitter binding in the volunteers in groups I and II (those without psychosis). These results show that the haloperidol blockade tended to normalize the neurotransmitter binding in the patients with psychosis.
The analysis predicted that patients with schizophrenia who had received neuroleptics previously did not have a higher neurotransmitter binding than did the patients who never received medication (4 vs. 4.7 pmol cm−3). Thus, the plasma haloperidol concentration was not elevated by greatly increased agonist competition at the receptor sites, as the bound endogenous competitors differed little between the two groups (χx 2.53 vs. 2.45).
The theoretical treatment (Appendix) revealed that estimates of the apparent affinity of an administered receptor antagonist can be used to infer changes of neurotransmitter concentration in brain only on certain conditions. This means that the significant elevation of the haloperidol plasma IC50 in patients with psychotic potential has other possible explanations. In the Appendix, an expression for KIC50(pl) (Eq. 47) is derived that identifies several factors that could have contributed to the measured increase,
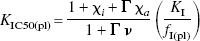
including the free plasma fraction of the inhibitor (fI(pl)), the intrinsic affinity constant of the inhibitor itself (KI), the possible endogenous nonagonist competitors (χi), the endogenous agonists (χa), the G-protein-induced affinity change of the agonists (Γ), and the inhibitor-induced relative increase of the endogenous agonists (ν).
The claim that the level of endogenous competition of the haloperidol binding (exerted by χi, or χa, or both) is elevated in the patients assumes that neither the inhibitor's plasma “free” fraction nor the intrinsic affinity of its receptors has changed. The current calculation used the same plasma “free” fraction of haloperidol in all the subject groups. As previously discussed by Wong et al. (1997 b), it is unlikely that the change of the IC50 arose because of different degrees of protein binding.
The calculation assumes that haloperidol binds competitively to the receptors, which also bind NMSP, and that the inhibition in theory can reach 100%. Although the group mean occupancies averaged 70% to 87%, the calculated occupancy reached 100% in some subjects. Lyon et al. (1986) and Frost et al. (1987) recorded near-100% occupancies of NMSP receptors in rodents, and Ishizu et al. (2000) showed that haloperidol bound competitively over a range of multiple concentrations, although no concentration reached as high as recorded in some patients in this study.
The calculation also assumes a single value for the intrinsic haloperidol half-inhibition concentration (KI), but there is no direct evidence that the affinities of the relevant neuroreceptors for haloperidol in fact remain unchanged. For this reason, it can not be ruled out that a fundamental change of the receptors occurred in patients with the psychotic potential that caused the inherent affinity toward haloperidol to decline. In the final analysis, however, it is difficult to distinguish between an intrinsic decline of affinity and a decline imposed by the elevation of a competitor, because it is not known how the affinity of dopamine receptors is regulated.
The interpretation that the level of endogenous competitors of the haloperidol binding is elevated in the patients is consistent with previous claims that extracellular dopamine levels may be high in patients with active psychosis, or that the administration of a neuroleptic leads to greater extracellular release of dopamine in patients with psychotic tendencies than in patients without these tendencies.
Rather than making distinctions among the different roles of nonagonists, agonists, and G-proteins, the current analysis yielded a single estimate of the “lumped” baseline endogenous competitor concentration relative to its affinity constant (χx). In Eq. 7, the apparent concentration of this “lumped” competitor, relative to a weighted affinity, is
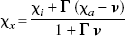
according to which the calculated increase is an estimate of an extracellular dopamine elevation only when the level of other competitors and G-protein action remain unchanged and when the inhibitor itself induces no change of dopamine or G-protein action. Thus, it was not possible to give a direct estimate of the absolute agonist concentration because the apparent affinities of partial and complete agonists probably do change as functions of the agonist concentration itself.
Independently of changes of membrane potential, dopamine action at the D2, D3, and D4 receptors in the striatum is mediated primarily by G o proteins that form ternary complexes with the agonist-activated receptor (Wang et al., 1995; Beindl et al., 1996; Jiang et al., 2001). The striatal D2 receptor is predominantly of the D2L “long” isoform in which the long third cytoplasmic loop mediates coupling to the G-proteins (Guivarc'h et al., 1995). As derived in the Appendix, this coupling leads to an increase of the apparent affinity of the receptor to the agonist, which contributes to an increase of KIC50(pl).
Laruelle et al. (1996, 1997) reported a decline of the binding potential of a labeled benzamide tracer induced by administration of amphetamine to healthy volunteers and patients with schizophrenia. The decline of the binding potential was inferred from calculations of the ratio of specific-to-nonspecific binding of the tracer that would not reveal the baseline concentration of the endogenous neurotransmitter, as argued above, but would reveal the transient change induced by the administered amphetamine. In schizophrenic patients, the decline was significantly more pronounced than in the healthy control subjects. On the basis of this finding, Laruelle et al. (1997) claimed that the release of an endogenous competitor of the tracer binding was more pronounced in schizophrenia. Equation 7 shows that not only the release of the agonist but also a G-protein-induced increase of the agonist's affinity could explain the finding.
Amphetamine is not the only agent capable of releasing dopamine to the extracellular space; both haloperidol and raclopride are neuroleptics known to have this effect (Westerink and de Vries, 1989). Equation 7 shows that it is possible to miss increases of receptor density and half-inhibition concentration if pathologically elevated but transient increases of the endogenous competitor occur as a consequence of the administration of the exogenous inhibitor, as has been shown for schizophrenia. This is also possible when the exogenous inhibitor is itself a tracer administered at low specific activity, as is the case when raclopride is used as a tracer of dopamine D2 -like receptor density (Farde et al., 1990, 1995). In the current experimental design, the authors were not in a position to ascertain whether any such “hidden” agonist increase persisted long enough after the administration of the exogenous antagonist to affect the estimates reported. However, if the estimates were affected, it would lower rather than further increase the estimates of the Michaelis constants. Thus, a “hidden” agonist surge would render the current estimates of the IC50 of haloperidol too low.
Recently, Abi-Dargham et al. (2000) attempted to measure the baseline endogenous competitor level by increasing the binding of an exogenous competitor with drugs that deplete the dopamine (for example, AMPT). The outcome of this study was interpreted as showing a greater baseline dopamine level in patients with schizophrenia than in healthy volunteers. As such, that study agrees with the conclusions drawn from the current study, although the change calculated by Abi-Dargham et al. (2000) is much smaller than determined in the current study.
On the basis of the observations reported here, the authors speculate that the propensity for psychosis is associated with increased endogenous neurotransmitter binding to dopamine D2 -like receptors in the most ventral parts of the caudate nucleus. Variations of the baseline level of endogenous competition may invalidate measures of binding rate or potential as index of receptor density (Wolf et al., 1996), and transiently increased endogenous competition, as a result of exogenous inhibitor administration, may mask changes of receptor density, or Michaelis half-saturation concentration, or both (Farde et al., 1990, 1995).
Dopamine-induced inhibition of haloperidol binding speaks against the ‘denervation hypersensitivity’ theory of low extracellular dopamine as the explanation of episodes of psychosis in these disorders, unless it is also argued that the extracellular dopamine is lowered outside the synapses and elevated in the synaptic cleft. Such a differential change would represent a shift of dopamine from a more static pool outside vesicles and synapses to the more dynamic pool in the vesicles and the synapses.
The apparent (‘measurable’) affinity of the D2 -like receptors to dopamine in the high-affinity state is 10-fold greater than the affinity of the D1-like receptors, approximately 1 nmol/L versus 10 nmol/L (Seeman and Van Tol, 1993, 1994). The intrinsic affinity is unknown. Speculating on the basis of the G-protein effect discussed above that the intrinsic affinity is no more than 1% of the apparent agonist-induced affinity (that is, that Ka is no less than 100 nmol/L), the intrasynaptic dopamine concentration is ≥15 nmol/L in the healthy volunteers studied here, and 10-fold greater in the individuals with psychosis. This speculation about the concentration of dopamine on the basis of occupancy figures must take into account agonist-induced affinity changes.
Footnotes
Acknowledgments:
The authors thank Godfrey Pearlson and Larry Tune for patient recruitment and support, and Bob Dannals, Alan Wilson, and Hayden Ravert for radiochemistry assistance.
1
This formalism ignores the role of G-proteins, the effect of which will be discussed briefly below.
2
But only when no steady-state changes of the binding capacity or endogenous competitors occur, and persist, after the administration of the exogenous inhibitor. The effect of such a change is discussed below.
3
The maximum quantity of the ternary G-protein complexes and the apparent half-maximum agonist concentration can be predicted from this equation. The maximum quantity of the ternary complexes is
and the apparent half-saturation concentration is