Abstract
During the past decade, studies on the manipulation of various inhaled inert gases during ischemia and/or reperfusion have led to the conclusion that inert gases may be promising agents for treating acute ischemic stroke and perinatal hypoxia-ischemia insults. Although there is a general consensus that among these gases xenon is a golden standard, the possible widespread clinical use of xenon experiences major obstacles, namely its availability and cost of production. Interestingly, recent findings have shown that helium, which is a cost-efficient inert gas with no anesthetic properties, can provide neuroprotection against acute ischemic stroke in vivo when administered during ischemia and early reperfusion. We have investigated whether helium provides neuroprotection in rats subjected to middle cerebral artery occlusion (MCAO) when administered after reperfusion, a condition prerequisite for the therapeutic viability and possible clinical use of helium. In this study, we show that helium at 75 vol% produces neuroprotection and improvement of neurologic outcome in rats subjected to transient MCAO by producing hypothermia on account of its high specific heat as compared with air.
Introduction
Acute ischemic stroke is one of the most common causes of death and long-term neurologic morbidity in the adult population. The primary cause of acute ischemic stroke is a massive disruption of cerebral blood flow through thromboembolism that leads to an oxygen and glucose deprivation and to a subsequent overstimulation of the excitatory N-methyl-d-aspartate glutamatergic receptor (Endres and Dirnagl, 2002; Parsons et al, 1998), whose postsynaptic activation is known as a critical event in neuronal death and brain damage induced by acute ischemic stroke (Parsons et al, 1998). Although limitation of the vascular insult by early reperfusion through thrombolysis has benefited ischemic stroke patients if administered within 3h of symptoms onset, strategies of neuroprotection by the use of glutamatergic receptor antagonists have not yet been proven efficient in humans because these compounds possess an intrinsic neurotoxicity (Olney et al, 1989, 1991; Davis et al, 2000) and fail to reach the site of brain injury (Arrowsmith et al, 1998). During the past decade, studies on the manipulation of various inhaled inert gases during ischemia and/or reperfusion have led to the conclusion that the use of such agents may be a promising therapy for treating acute ischemic stroke. Among these gases, the remarkably safe anesthetic gas, xenon, which possesses pharmacological properties of potentially therapeutic interest (Franks et al, 1998; Yamakura and Harris, 2000; Gruss et al, 2004), has been studied extensively in various models of brain injury, such as acute ischemic stroke, perinatal hypoxia-ischemia, and cardiopulmonary bypass-induced neurologic and neurocognitive dysfunctions (David et al, 2003, 2008; Homi et al, 2003; Ma et al, 2003; Abraini et al, 2005a; Ma et al, 2005, 2006; Dingley et al, 2006, 2008; Martin et al, 2007; Hobbs et al, 2008). Contrasting with the controversial results on neuroprotection by nitrous oxide (David et al, 2003; Homi et al, 2003; Yokoo et al, 2004; Abraini et al, 2005a, b , Haelewyn et al, 2008; Taninishi et al, 2008; McGregor et al, 2008), which shares many pharmacological properties with xenon (Franks et al, 1998; Yamakura and Harris, 2000; Gruss et al, 2004; Jevtovic-Todorovic et al, 1998; Colloc'h et al, 2007), studies on xenon have led to the general consensus that this gas has unique neuroprotective properties with no adverse effects when used at nonanesthetic concentrations. However, although closed xenon delivery systems for humans are now being developed to resolve some of the major obstacles to the widespread clinical use of xenon, such as its scarceness and excessive cost of production, it remains true that the availability and volume of production of xenon are limited.
Alternatively and interestingly, recent findings have shown that helium, which is a cost-efficient inert gas with no anesthetic properties, may provide neuroprotection against traumatic injury in vitro and acute ischemic stroke in vivo when administered during or early after a brain insult, such as ischemia (Pan et al, 2007; Coburn et al, 2008). In this study, because helium on account of its high specific heat as compared with air could produce hypothermia (Tapper et al 1974)—a condition known to be neuroprotective by itself (Kollmar et al, 2007)—we investigated whether helium can produce neuroprotection in rats subjected to middle cerebral artery occlusion (MCAO) when administered after reperfusion, a condition prerequisite for the therapeutic viability and possible clinical use of helium.
Materials and methods
Animals
All animal-use procedures were approved by the local ethics committee in accordance with the framework of the French Legislation and The European Communities Council Directive of 24 November 1986 for the use of laboratory animals in biomedical experimentation. Adult male Sprague-Dawley rats (Janvier, Le Genest Saint-Isle, France) weighing 250 to 280 g were used. Before being used, rats were housed in groups of six at 21°C ± 0.5°C in Perspex home cages with free access to food and water on a light-dark reverse cycle with lights on from 2000 to 0800 hours.
Effects of Helium on the Body Temperature of Rats
We investigated the effects of helium at 75 vol% at various gas temperatures (aimed temperature: 32°C, 28°C, and 21°C; n = 4 per group) on the body temperature of the rats. On the day of testing, the body temperature of the rats was measured immediately before and after a 3-h exposure to helium at 75 vol%. For more details, see the section ‘Gas Pharmacology and Treatment’.
Transient Cerebral Ischemia Induced by Middle Cerebral Artery Occlusion in Rats
Rats were subjected to transient cerebral ischemia for 60 mins by the intraluminal MCAO method. They were anesthetized with 5% isoflurane in oxygen alone for induction, and then maintained anesthetized with 1.5% isoflurane in oxygen alone, while breathing spontaneously throughout the surgical intervention. A midline neck incision was made, and the right common carotid artery was exposed. After coagulation of the branches of the external carotid artery, a nylon thread (0.18 mm in diameter), including a distal cylinder (3 mm long and 0.38-mm diameter) was inserted into the lumen of the external carotid artery, directed into the internal carotid artery up to the origin of the middle cerebral artery, and secured to the external carotid artery. During surgery, body temperature was kept normothermic at 37.5°C ± 0.5°C using a feedback-controlled thermostatic heating pad. After surgery, rats were allowed to move freely in their home cages with free access to food and water. The total duration of the surgery was no more than 25 mins, thereby allowing rapid isoflurane desaturation from the rat's body and the presence of little residual isoflurane during the MCAO period (White et al, 1974). One hour after MCAO, the nylon thread was removed under short isoflurane anesthesia of no more than a 10-min duration to restore blood flow, thereby allowing rapid isoflurane desaturation from the rat's body and the virtual absence of residual isoflurane during the period of treatment with helium (White et al, 1974). Thereafter, the rats were returned to their home cages before being treated 2 h after MCAO induction, i.e., 1 h after reperfusion, with medical air (controls, n = 18) or helium at 75 vol% at a gas temperature of 25°C (n = 17) or 33°C (n = 6) for 3 h, according to a blinded procedure. This consisted in giving each group of animals a ‘secret’ code unknown to the experimenters in charge of assessing the neurologic and histologic outcome of the rat until the end of the study. Sham rats (n = 8) were subjected to the same surgical protocol, except that the nylon thread was introduced into the external carotid artery and then directed up to the origin of the internal carotid artery. Sham rats were exposed to air for 3 h. In addition, arterial blood samples were taken before and after the treatment to assess the possible effects of helium on a series of physiologic parameters, including arterial pH, paCO2, paO2, and SaO2. All values fell within normal physiologic ranges (data not shown).
Assessment of Motor Functions and Neurologic Outcome
Motor functions and neurologic outcome of the rats were investigated as described earlier (David et al, 2008; Haelewyn et al, 2008) by recording the their rearing activity and motor coordination according to the blinded procedure described above. The rearing activity of the rats was quantified using a bank of eight individual activity cages of 30 × 20 × 20 cm, equipped with infrared beams; the motor coordination of the rats was quantified using a bank of eight individual nonmotorized activity wheels of 34 cm in diameter, equipped with 1/8 rotation sensors (Imetronic, Pessac, France). Beam interruptions and rotation signals were detected through an electrical interface and recorded over 5-min intervals on a personal computer. On day 3 before MCAO, the rats were placed in the activity cages and the activity wheels for 15mins to familiarize them with the experimental procedures and environments. On day 2 (48 h) before MCAO, and on day 1 (24 h) and day 2 (48 h) after MCAO, the animals were placed again in the activity cages and the activity wheels, and recorded for 15 mins in each type of environment. Post-MCAO scores of rearing activity and motor coordination recorded on day 1 and day 2 after MCAO were expressed as a percentage change of the pre-MCAO scores recorded on day 2 before MCAO, using each animal as its own control. Thereafter, a global score of the neurologic outcome on rats was calculated by averaging post-MCAO scores of rearing activity and motor coordination, and expressed as percentage of the global scores of the sham rats on day 1 and day 2.
Gas Pharmacology and Treatment
Oxygen, nitrogen, and helium of medical grade were used. Gas mixtures containing medical air composed of 25 vol% oxygen and 75 vol% nitrogen, or helium at 75 vol% with the remainder being oxygen were obtained using calibrated flowmeters and an oxygen analyzer. During the treatment period, the rats were allowed to move freely in a closed chamber (10 L volume) fitted with viewing windows to allow observation, and treated for a 3-h period with either medical air (controls) or helium at 75 vol% at a flow rate of 6 L/min. This allows maintaining carbon dioxide at <0.03 vol% and humidity at ∼60% to 70%. The gas temperature was controlled by placing the last part (1.5-m length) of the gas admission pipe in a heating/cooling bath (see Figure 1A).
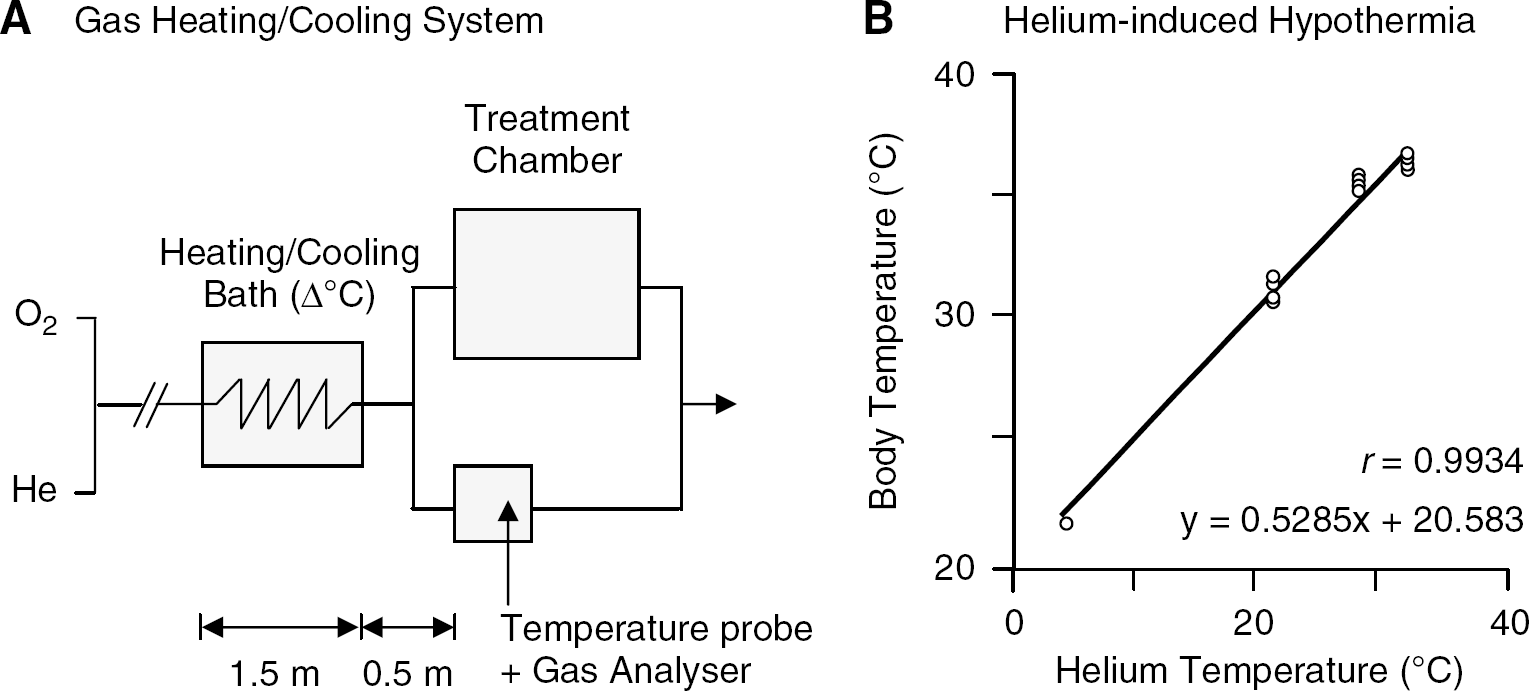
Studying the effect of helium on body temperature: device and data. (
Histologic Analysis
Fifty hours after the induction of MCAO, the rats were killed by decapitation under isoflurane narcosis. The brain was rapidly removed, frozen in isopentane, and placed at −80°C. Coronal brain sections (20 μm) were cryostat-cut, mounted on slides, stained with thionin, dehydrated with serial alcohol and cleared with xylene, and coverslipped with Eukitt mounting. Brain sections colored with thionin were digitized on a personal computer and analyzed using an image analyzer (ImageJ Software, Scion Corp., MD, USA) by one blinded observer, according to the blinded procedure described above. The lesioned areas were delineated by the pallor of staining in the necrotic tissue as compared with the surrounding healthy tissue; MCAO-induced brain damages were calculated by the integration of the infarcted surfaces over the whole brain, corrected for brain tissue edema, and expressed in mm3 of infarction volume.
Statistical Analysis
Data are given as mean ± s.e.m. Data were analyzed according to size of the samples. Changes in body temperature (n < 6) were analyzed using the Kruskall-Wallis nonparametric ANOVA (analysis of variance) and the post hoc Mann-Whitney nonparametric unpaired U-test. Middle cerebral artery occlusion-induced brain infarction and behavioral alterations (n ≥ 6) were analyzed by parametric ANOVA; following a significant F-value, post hoc analysis was performed using the Fisher test for unpaired comparisons.
Results
Helium Produces Hypothermia When Administered at Certain Gas Temperatures
In excellent agreement with earlier data in rat pups (Tapper et al, 1974; see Figure 1B), we found that exposure to 75 vol% helium at 32°C, 28°C, and 21°C produced hypothermia (P<0.02 for each group). The amplitude of the reduction of the body temperature of the rat depended highly on the gas temperature at which helium was administered, as indicated by the high correlation coefficient between gas and body temperature (n = 13, r = 0.9934, P< 0.0001; Figure 1B).
Helium at 25°C Reduces Middle Cerebral Artery Occlusion-Induced Brain Infarction and Behavioral Motor Deficits
Sham rats had no brain damage (data not shown). Middle cerebral artery occlusion control rats treated with air at 25°C had normal body temperature (38.1°C ± 0.05°C) and brain damage at the cortical level (P < 0.0001; Figure 2A and 2B) in frontal, parietal, and temporal areas (from A = + 4.20 mm to A = −5.80 mm from the bregma) and at the subcortical level (P< 0.0001; Figure 2A and 2B). Compared with sham rats, control rats subjected to MCAO further showed behavioral deficits in motor coordination and rearing activity, as well as lower scores of global neurologic outcome on day 1 and day 2 after MCAO (0.0001 ≤ P ≥ 0.005; Figure 2C).
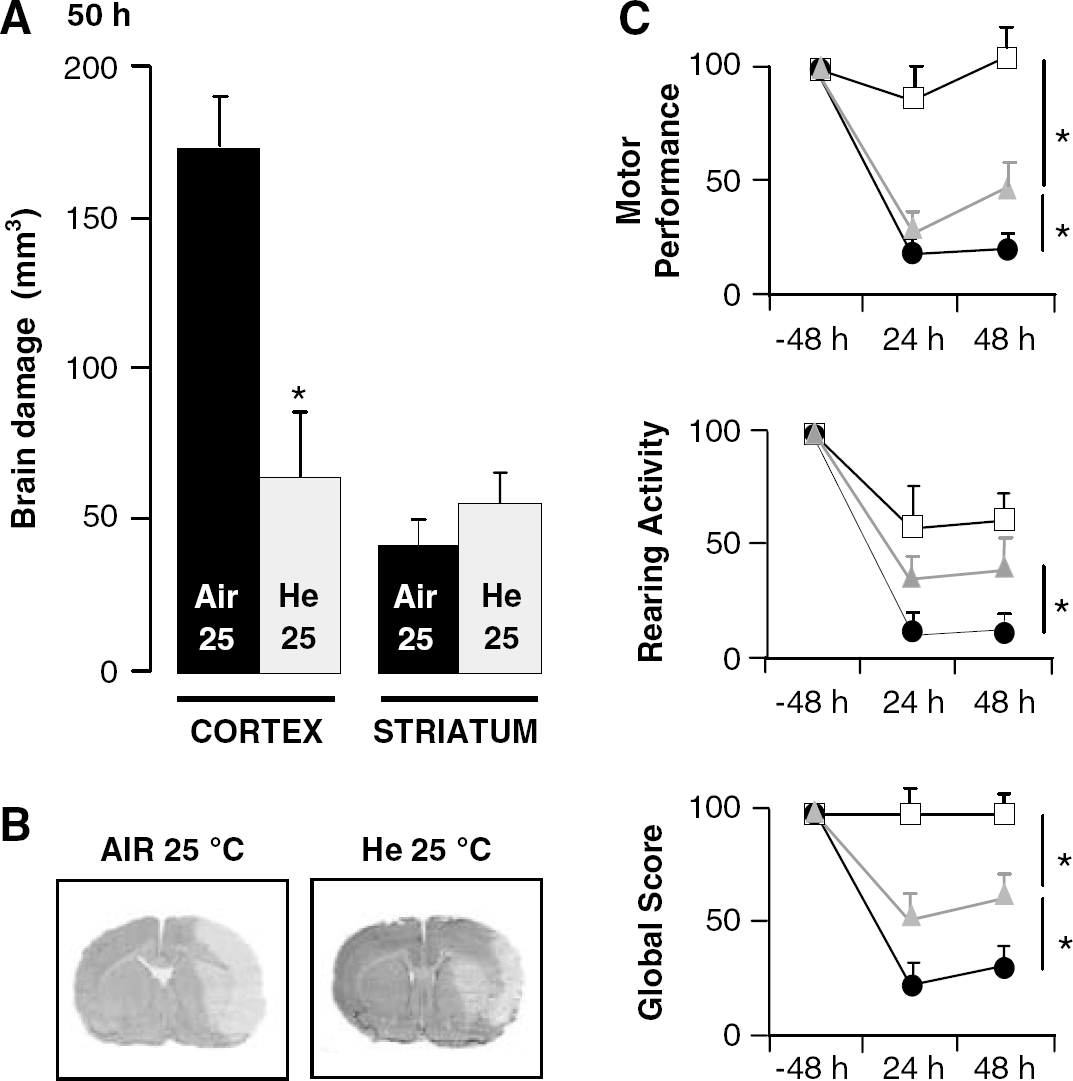
Tissue neuroprotection and improvement of neurologic outcome provided by a gas temperature of 25°C of helium at 75 vol% in rats subjected to transient MCAO. (
Compared with MCAO control rats treated with air at 25°C, those treated with helium at 25°C showed both reduced body temperature (34.3°C ± 0.08°C versus 38.1°C ± 0.05°C; P < 0.0001) and reduced cortical (P < 0.0001), but not subcortical, infarction volumes (Figure 2C). Helium-treated rats further showed higher scores of rearing activity and global neurologic outcome on day 1 (P < 0.02), as well as higher scores of motor coordination, rearing activity, and global neurologic outcome on day 2 after MCAO (0.002 ≤ P ≥ 0.02; Figure 2C). However, as compared with sham rats, helium-treated rats still showed lower scores of motor coordination and global neurologic outcome on day 1 (0.0001 < P < 0.02) and day 2 (0.002 < P < 0.02) after MCAO, indicating a partial but not a full functional recovery.
Helium at 33°C Fails to Reduce Middle Cerebral Artery Occlusion-Induced Brain Infarction
Compared with rats treated with helium at 25°C, those treated with helium at 33°C had normal body temperature (38.2°C ± 0.27°C versus 34.3°C ± 0.08°C; P < 0.0001) and greater cortical (P < 0.0001), but not subcortical, brain damage. Ischemic lesions in these animals were similar to those observed in MCAO control rats treated with medical air at 25°C (Figure 3A and 3B).
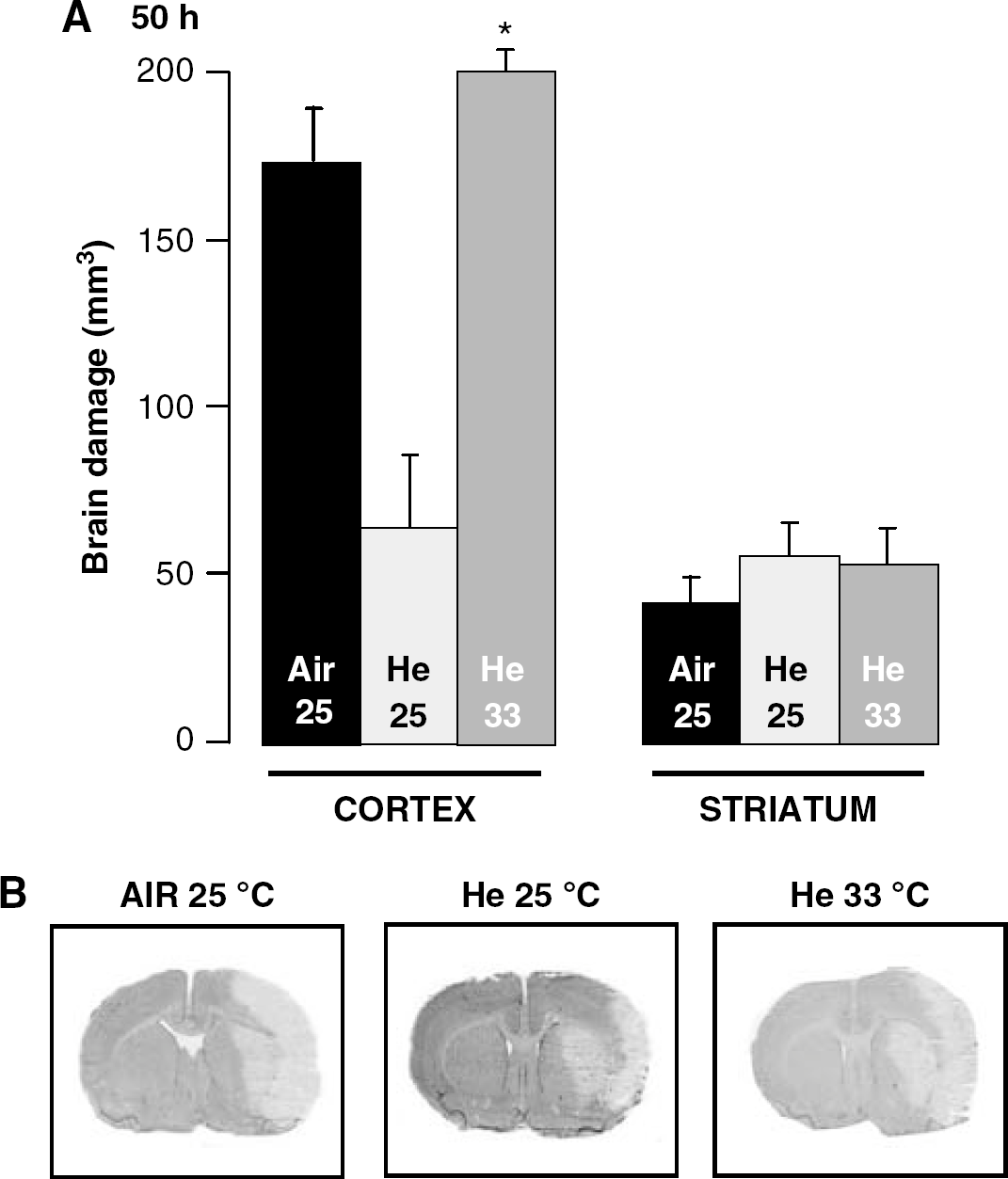
Lack of tissue neuroprotection provided by a gas temperature of 33°C of helium at 75 vol% in rats subjected to transient MCAO. (
Discussion
In this study, we provide experimental evidence, for the first time to the best of our knowledge, that helium under certain conditions of gas temperature can provide neuroprotection and improvement of the neurologic outcome in rats subjected to MCAO-induced brain ischemia by producing hypothermia, a condition known to be neuroprotective by itself (Konstas et al, 2006; Kollmar et al, 2007).
We showed that helium at a gas temperature of 25°C allows reduction of body temperature by > 3°C from 38.1°C to 34.3°C. Interestingly, it should be emphasized that the amplitude of neuroprotection that produced such conditions of helium-induced hypothermia is in good agreement with earlier findings on hypothermia-induced tissue neuroprotection and improvement of the neurologic outcome in rats subjected to MCAO (Kollmar et al, 2007). Contrasting with the beneficial effect of helium at 25°C, we found that helium at a gas temperature of 33°C that allows maintaining normothermia in rats failed to produce tissue neuroprotection in rats subjected to MCAO-induced brain ischemia. Taken together, these data suggest that hypothermia may have mainly contributed to the neuroprotective action of helium in addition to other possible theoretical effects on prosurvival signaling kinases and mitochondrial permeability (Pagel et al, 2007; Heinen et al, 2008).
Our findings of postischemic helium-induced neuroprotection extend earlier data that have first shown that intraischemic helium has neuroprotective properties in rats subjected to MCAO (Pan et al, 2007). In line with these data, we found that helium has neuroprotective action at the cortical, but not at the subcortical, level. The lack of effect of helium for providing subcortical neuroprotection seems consistent with the fact that the striatum, which constitutes the main part of the subcortical areas subjected to ischemia, is well known to be difficult in protecting against MCAO-induced stroke on account of its lack of collateral vasculature that results in a complete blood flow cessation as compared with the cortex (Tyson et al, 1984). Despite this, we found that helium did improve the neurologic outcome as evaluated using a quantitative assessment of behavioral motor function. As helium provides cortical, but not subcortical, neuroprotection, it is likely that the improvement of the neurologic outcome produced by helium can be attributed to its ability in reducing cortical brain damage. This finding may be related to the fact that most of silent brain infarctions, which are ischemic episodes free of neurologic deficit, are well known to occur in deep brain structures, including the basal ganglia, pons, and subcortical white matter (Fisher, 1998).
On the basis of these findings, we suggest that combining helium with xenon may help in reducing the excessive cost of treatment with xenon while providing neuroprotection. Supporting this are results obtained from rat pups subjected to perinatal hypoxia-ischemia that have shown that combining xenon at 20 vol% with a mild hypothermia of 35°C is sufficient enough for providing effective neuroprotection (Ma et al, 2005; Martin et al, 2007). However, although helium like xenon has an excellent safety profile in humans and in animals when administered alone or in combination with other inert gases, even at high hyperbaric concentrations (Rostain and Naquet, 1974; Abraini and Rostain, 1991; Abraini et al, 1994; Harris and Barnes, 2008; Ward and Dushay, 2008), further experiments are needed to determine the respective concentration of helium and xenon to be used to provide neuroprotection with helium-xenon gas mixtures, and to define the optimal indications, duration, and time points for treatment, as well as the actual concentration of helium and xenon to be used in humans.
Footnotes
Acknowledgements
HND performed the behavioral studies; BH and LC performed the MCAO experiments; BH and ML performed histologic analysis of the rat brains; MD and JHA performed the calibration experiments on helium-induced hypothermia; JJR provided medical gases through the DGA programs on medical gases and neuroprotection. JHA designed the experiments, analyzed the data, and wrote this paper.
