Abstract
Oligodendrocytes are sensitive to ischemic damage. The Sonic hedgehog (Shh) pathway is critical in oligodendrogenesis; Gli1 is the principal effector of Shh signaling. We investigated oligodendrogenesis and Shh/Gli1 pathway activation after bone marrow stromal cell (BMSC) treatment of stroke in rats. Rats were subjected to the middle cerebral artery occlusion (MCAo). BMSCs have been shown to promote functional recovery post stroke. A therapeutic dose of BMSC (3times106 cells) treatment was initiated 1 day after MCAo. Immunohistochemistry was carried out to measure the oligodendrocyte progenitor cells, oligodendrocytes, myelin, and expressions of Shh and Gli1 at 14 days after MCAo. Gene expression of Shh and Gli1 was tested at 2 days after MCAo. An in vitro study was used to investigate the effects of BMSC on a premature oligodendrocyte cell line (N20.1 cells). BMSC treatment significantly increased 04+ oligodendrocytes, MBP+ area, and bromodeoxyuridine (BrdU)+, NG2+, BrdU+-NG2+ cells, and mRNA and protein expressions of Shh and Gli1 in the ipsilateral brain of the MCAo rats than that in phosphate buffered saline (PBS)-treated rats. BMSCs promoted N20.1 cell proliferation and Gli1 mRNA expression, and these effects were abolished by the Shh pathway inhibitor cyclopamine. These data indicate that the BMSC treatment stimulates oligodendrogenesis by activation of the Shh/Gli1 pathway post stroke.
Introduction
Oligodendrocytes generate the myelin sheaths that enwrap axons, thereby facilitating saltatory conduction (McTigue and Tripathi, 2008); they are vulnerable to damage in a variety of neurologic diseases, such as ischemic cerebrovascular diseases, and oligodendrocyte damage leads to demyelination, which contributes to neurologic functional deficits (Dewar et al, 2003; McTigue and Tripathi, 2008; Micu, et al 2006). Recruited oligodendrocytes can remyelinate axons, raising the possibility of therapeutic intervention (Chang et al, 2000). Mature oligodendrocytes in the adult mammalian central nervous system (CNS) were considered to be postmitotic and are unable to proliferate in response to injury (McTigue and Tripathi, 2008). However, abundant oligodendrocyte progenitor cells exist in the white and gray matter of normal CNS, and are present in the lesions (Komitova et al, 2006; McTigue and Tripathi, 2008; Tanaka et al, 2003), and proliferating oligodendrocyte progenitor cells contribute to remyelination (Carroll and Jennings, 1994; Gensert and Goldman, 1997; Jiang et al, 2008; Keirstead et al, 1998; Prineas et al, 1989).
Our previous in vivo studies found that bone marrow stromal cells (BMSCs) stimulate the production of restorative factors by parenchymal cells and evoke white matter remodeling in the injured brain (Chopp and Li, 2002; Chopp et al, 2009), which lead to improvement in neurologic function post stroke (Chen et al, 2000; Chen et al, 2001a; Shen et al, 2007; Zhang et al, 2004), and our in vitro results suggest that BMSCs protect oligodendrocytes subjected to oxygen-glucose deprivation (Zhang et al, 2008). These studies provide insight into white matter damage and the therapeutic benefits of BMSC cell based-remyelinating therapy after stroke. In this study, we investigated the effects of BMSC treatment of stroke on oligodendrogenesis.
To elucidate the underlying mechanisms of oligodendrocyte progenitor cell proliferation after treatment, we used N20.1 cells, which are premature oligodendrocytes (Paez et al, 2004). These cells are widely used to study the cellular and molecular mechanisms involved in the development, maturation, and formation of myelin by oligodendrocytes in the CNS (Garcia et al, 2007; Paez et al, 2004; Zhang et al, 2008).
A signaling pathway by which BMSCs induce oligodendrogenesis was investigated. Sonic hedgehog (Shh) is a member of the family of the hedgehog proteins; it plays a critical role in the induction, survival, proliferation, and migration of oligodendrocytes (Marti and Bovolenta, 2002; Seifert et al, 2005; Sussman et al, 2002). The Shh signaling pathway is well conserved (Marti and Bovolenta, 2002), Shh binds to the transmembrane receptor protein, patched, to activate another transmembrane receptor, smoothened (Ingham and McMahon, 2001), and induces intracellular reactions that target the Gli family of transcription factors (Ruiz i Altaba et al, 2002). Gli1 is the principal effector of Shh signaling in neural progenitor cells (Ahn and Joyner, 2005; Wang et al, 2007). In this article, we show that BMSC treatment, as an effective restorative treatment for stroke, enhances white matter remodeling and stimulates the Shh pathway.
Materials and methods
All experimental procedures have been approved by the Institutional Animal Care and Use Committee of Henry Ford Health System.
Bone Marrow Stromal Cell Preparation
Rat (r) and mouse (m) BMSCs were generously provided by Cognate Therapeutics Inc. (Baltimore, MD, USA). BMSCs were isolated, grown, and tested, as described previously (Zhang et al, 2004). Briefly, the bone marrow was obtained from adult donors and placed into media with Dulbecco's modified Eagle's medium-low glucose and 10% selected fetal bovine serum (FBS). The adherent cells were fed and generally lasted for about 2 weeks. When dense colonies of spindle-shaped cells covered greater than 80% of the dish, the cells were passaged into the secondary culture. When dose and purity levels were achieved, cells were harvested and cryopreserved in appropriate dose-related aliquots in Plasma-Lyte (Baxter Healthcare Corporation, Deerfield, IL, USA) containing serum albumin and dimethyl sulfoxide.
Animal Model
Adult male Wistar rats (270 to 300 g, n = 27; Charles River Laboratories, Wilmington, MA, USA) were used in our experiments. Middle cerebral artery occlusion (MCAo) was induced by a method of intraluminal vascular occlusion modified in our laboratory (Zhang et al, 2004). Briefly, a length of 4–0 monofilament nylon suture (18.5 to 19.5 mm) was advanced from the external carotid artery into the lumen of the internal carotid artery until it blocked the origin of the MCA. Rats were randomly divided into the following:
Normal control group: This control group consisted of naive rats (n = 9) without surgery.
MCAo control group: Rats (n = 9) were subjected to permanent MCAo and 1 mL phosphate buffered saline (PBS) was injected into a tail vein at 1 day after MCAo.
BMSC treatment group: After washing with PBS and subsequent centrifugation, rBMSCs (rat BMSC) (3 × 106) in 1 mL of PBS were injected into a tail vein at 1 day after MCAo (n = 9). This dose of cells was shown earlier to be highly effective in improving functional recovery after stroke (Chen et al, 2001a, 2003; Zhang et al, 2004). Bromodeoxyuridine (BrdU, Sigma Chemical) was used for mitotic labeling. It (100 mg/kg) was intraperitoneally injected twice a day for 7 consecutive days into normal or ischemic rats starting 1 day after MCAo.
Tissue Preparation
To measure the expressions of Shh and Gli1 mRNA, normal rats (n = 3) and ischemic rats with BMSC or PBS treatment were killed at 2 days after MCAo (n = 3 per treatment group). The ischemic boundary zone (IBZ) was extracted from the brains of the rats subjected to stroke (Zhang et al, 2004). For immunohistochemical evaluation, normal rats (n = 6) were killed at 7 days after BrdU injection. Rats treated with BMSCs or PBS were killed at 14 days (n = 6 per treatment group) after MCAo. Brains were fixed in 4% of paraformaldehyde and embedded in paraffin. A coronal slide (6 μm thick) from the center of the ischemic core (bregma 0.2 mm) (Zhang et al, 2004) was used for immunohistochemical staining.
Real-Time Reverse Transcriptase-PCR Analysis
Quantitative PCR was carried out using the SYBR Green real-time PCR method. Total RNA was isolated from the IBZ using the RNeasy Lipid Tissue Mini Kit (Qiagen, Valencia, CA, USA). Quantitative reverse transcriptase-PCR (RT-PCR) was carried out on an ABI 7000 PCR instrument (Applied Biosystems, Foster City, CA, USA) using three-stage program parameters provided by the manufacturer as follows: 2mins at 50°C, 10 mins at 95°C, and then 40 cycles of 15 secs at 95°C and 1 min at 60°C. Specificity of the produced amplification product was confirmed by the examination of dissociation reaction plots. A distinct single peak indicated that a single DNA sequence was amplified during PCR. Polymerase chain reaction products were run on 2% agarose gels to confirm that correct molecular sizes were present. Each sample was tested in triplicate, and samples obtained from three independent experiments were used for analysis of relative gene expression using the 2−ΔΔCT method (Livak and Schmittgen, 2001).
Primer sets used to detect Shh and Gli1 mRNAs were listed as follows: Shh (Fwd, ATAACC TTGCCTGCTGTTGC; Rev, GAGACCCAACTCCGA TGTGT), Gli1 (Fwd, CAGCTCAAAGCTCAGCTCCT; Rev, CTTGGGGCTCTGATATGGAA), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Fwd, AGAACATCATCCCTGCATCC; Rev, CACATTGGG GGTAGGAACAC). One-way ANOVA (analysis of variance) followed by the Student-Newman-Keuls test was carried out. The data are presented as means ± s.d. A value of P<0.05 is considered significant.
Immunohistochemistry and Quantification
Oligodendrocytes were identified by antibodies 04 (1:100, Chemicon, Billerica, MA, USA) or myelin basic protein (MBP) (1:50, Abcam, Cambridge, MA, USA). The NG2 antibody (1:100, Chemicon) was used to mark oligodendrocyte progenitor cells. To test the protein expressions of Shh and Gli1 in the MCAo brains with or without BMSC treatment, immunohistochemistry stainings were carried out using a goat polyclonal antibody (pAb) against Shh (1:50, Santa Cruz Biotechnology) and a rabbit pAb against Gli1 (1:200, Santa Cruz Biotechnology).
Double immunostaining for NG2 with BrdU (a mouse monoclonal antibody, 1:100, Dako, Carpinteria, CA, USA) was used to identify oligodendrocyte progenitor cell proliferation. To clarify the relationship of the expressions of Shh and Gli1 in neural cells, double immunostaining for Shh with NeuN (marker of neurons, Chemicon), glial fibrillary acidic protein (GFAP, marker for astrocytes, Dako), and 04, as well as Gli1 with NG2 were used.
Immunostaining was carried out following standard protocols. Slides were treated first with the primary antibody and then with the antibody conjugated to fluorescein isothiocyanate (FITC, Jackson Immuno-Research, West Grove, PA, USA). These slides were then treated with a second primary antibody and then incubated with an antibody conjugated to Cy3 (Jackson ImmunoResearch). Negative control slides for each animal received identical preparations for immunostaining, except that primary antibodies were omitted.
To measure immunoreactive cells, numbers of 04+, Shh+, Gli1+, NG2+, BrdU+, and NG2+-BrdU+ cells were counted in eight fields of view from the IBZ of a standard section from the center of the ischemia core (bregma 0.2 mm) and in all the fields from the ipsilateral subventricular zone (SVZ) in the same slide. The fields were digitized under the light microscope (Olympus BX40, Tokyo, Japan) using a 3-CCD color video camera (Sony DXC-970 MD, Tokyo, Japan) interfaced with Micro Computer Imaging Device (MCID) analysis system (Imaging Research, Saint Catharines, ON, Canada). The immunopositive cells were calculated and divided by the measured areas, and presented as numbers per mm2. The density of the MBP+ area was measured in eight fields of view in the IBZ and presented as a proportional area by dividing the measured MBP+ area by the total scanned area. Double immunostaining images were taken using Zeiss (Oberkochen, Germany) laser-scanning confocal microscopy (LSM 510 META). 4′,6-diamidino-2-phenylindole (DAPI)-stained nuclei were visualized by two-photon microscopy using a Chameleon pulsed laser (Coherent Inc., Santa Clara, CA, USA) set at 720 nm excitation. These regions were scanned in 512 × 512 pixel (279 × 279 μm2) format in the x−y direction using a × 5 frame-scan average, and five optical sections along the z-axis with a l- μm step size were obtained under a × 63 objective.
Data are presented as mean ± s.d. Significance between the two groups was examined using ANOVA analysis. A value of P < 0.05 was considered significant.
In Vitro Oligodendrocyte Proliferation
We used an immortalized mouse premature oligodendrocyte cell line (N20.1, generously provided by Dr Anthony Campagnoni, University of California at Los Angeles) to measure oligodendrocyte proliferation and differentiation. N20.1 cells were obtained from mouse primary cultures of oligodendrocytes conditionally immortalized by transformation with a temperature-sensitive large T-antigen (Paez et al, 2004). N20.1 cells were cultured in Dulbecco's modified Eagle's medium/F12 (Invitrogen, Carlsbad, CA, USA) with 3.6 g/L of dextrose anhydrous, 3.38 g/L of HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid), 2.16 g/L of sodium bicarbonate, 90 mg/L of gentamicin, 1% of FBS, and G418 (100 μg/mL) at 39°C (Paez et al, 2004).
N20.1 cells were incubated in the following groups (n = 3 per group, three wells were used in each group): (1) regular culture medium for control; (2) mouse BMSCs (mBMSCs) cocultured with normal N20.1 cells (oligodendrocytes: mBMSCs = 10:1), an insert (0.4 μm, BD Biosciences, Franklin Lakes, NJ, USA) was used to contain mBMSCs. Oligodendrocytes were plated on the base of the culture wells and the upper transwell compartments were seeded with mBMSCs; (3) mBMSCs cocultured with 5 μmol/L cyclopamine (Calbiochem, Gibbstown, NJ, USA)-treated N20.1 cells. Cyclopamine is a specific inhibitor of smoothened (Wang et al, 2007).
For investigation of oligodendrocyte proliferation, N20.1 cells were treated for 12 h and 50 μg/mL BrdU (Sigma) was added to the cell cultures for 1 h. BrdU immunostaining of the N20.1 cells was measured. Numbers of BrdU+ cells were calculated by counting 10 random fields in each well with six wells per group. The results are presented as a percentage (positive cells divided by total cells).
In Vitro Activation of the Shh/Gli Pathway in Oligodendrocytes
An additional set of in vitro experimental groups was used for mRNA analyses (n = 3 per group). Primer sets used to detect mouse NG2 and Gli1 mRNAs were listed as follows: Gli1 (Fwd, TGTGTGAGCAAG AAGGTTGC; Rev, TTGCACACGTATGGCTTCTC), NG2 (Fwd, CGGCCAACAGTGGTTTCAAGT; Rev, CTTCTGTGAAGGCTGTCGATG), GAPDH (Fwd, AG AACATCATCCCTGCATCC; Rev, CACATTGGGGGT AGGAACAC). One-way ANOVA followed by the Student–Newman–Keuls test was carried out. The data are presented as means ± s.d. A value of P < 0.05 is considered significant.
Statistical Analyses
Data were evaluated by an investigator blinded to the treatment status of each animal. Data were evaluated for normality, and data transformation or nonparametric analysis was considered if data were not normal. Repeated analysis of covariance was used to test the BMSC effects on measurements. The analysis began testing for BMSC by time interaction, followed by testing the main effect of BMSCs if no interaction was observed at the 0.05 level.
Results
BMSC Treatment Increases the Area of Myelin in the Brain of Rats Subjected to MCAo
Given the significant improvement in neurologic outcome when rodents subjected to MCAo were treated with BMSCs (Chen et al, 2001a; Chopp et al, 2009; Shen et al, 2007; Zhang et al, 2004), we sought to address whether BMSC treatment affects myelin. As indicated by MBP immunostaining, myelin damage was obvious in the IBZ of MCAo rats at 14 days (Figure 1A). After BMSC treatment, the proportional area of myelin (MBP+) density was significantly increased in the IBZ of BMSC-treated rats than that of PBS-treated rats (73.7% ± 4.8% versus 64.9% ± 2.1%, P<0.05, Figures 1B and 1C). These data indicate that BMSC treatment protects myelin and/or induces oligodendrogenesis and the remyelination in the white matter of MCAo rats.
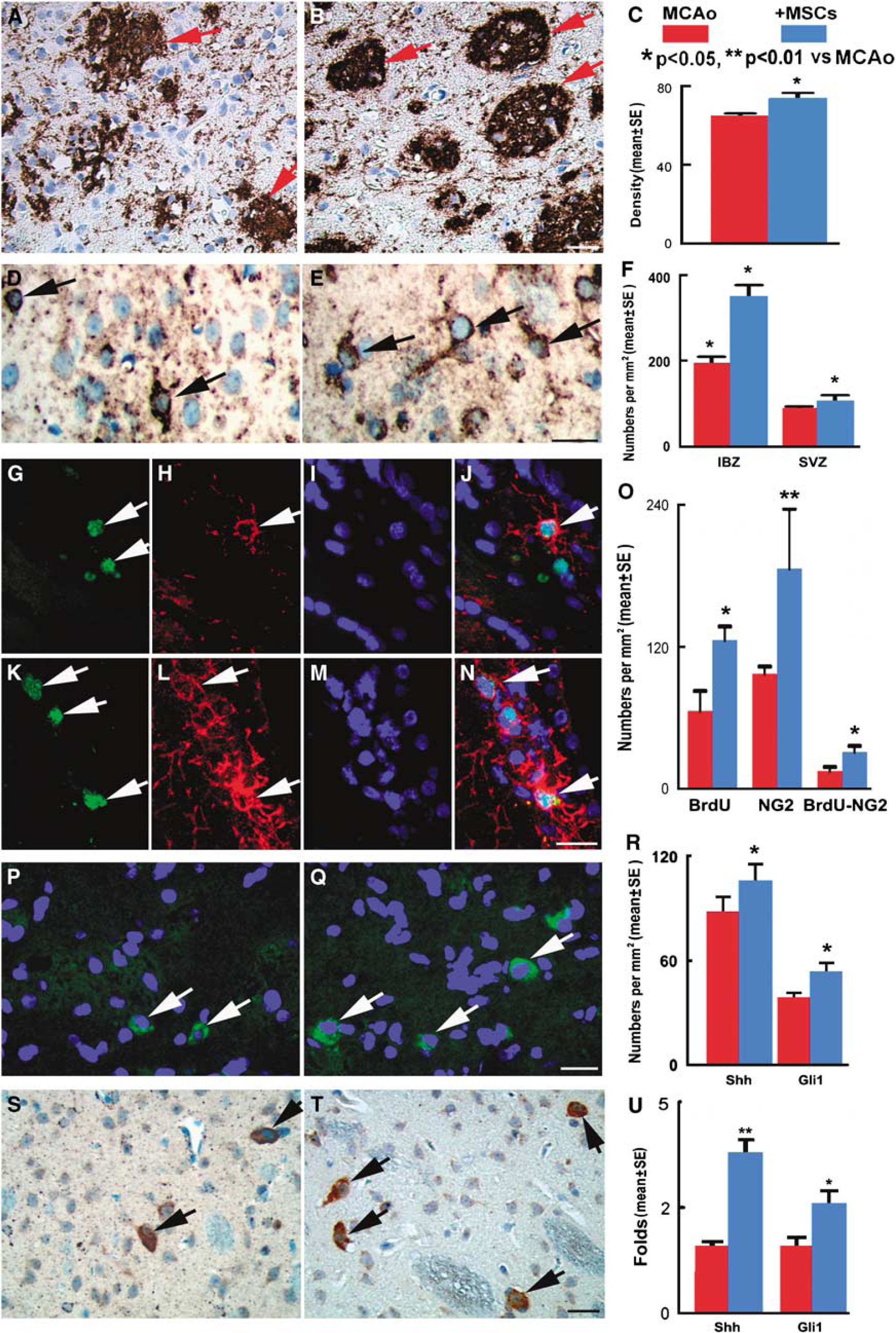
(
BMSC Treatment Increases Oligodendrogenesis in the Brain of MCAo Rats
Given the significant increase of the myelin area, we sought to address whether BMSC treatment affects oligodendrocyte proliferation and remyelination. After ischemic injury, 04+ oligodendrocytes were significantly increased in the IBZ and the SVZ of BMSC-treated rats than those of the PBS treatment group (350.7 ± 44.9 per mm2 versus 195 ± 25 per mm2 in IBZ; 107 ± 23.1 per mm2 versus 90 ± 7.5 per mm2 in SVZ, P<0.05, Figures 1D to 1F).
Using double BrdU and NG2 immunostaining, we found that BrdU+ proliferating cells, NG2+ oligodendrocyte progenitor cells, and BrdU+-NG2+ proliferating oligodendrocyte progenitor cells were significantly increased in the ipsilateral brain of BMSC-treated MCAo rats than that of PBS-treated rats (125.9 ± 11.5 per mm2 versus 65.8 ± 17 per mm2; 186.1 ± 20.7 per mm2 versus 97.2 ± 6.3 per mm2; 31.3 ± 5.1 per mm2 versus 15.2 ± 3.3 per mm2, respectively, P<0.05, Figures 1G to 1O). These data indicate that BMSC treatment stimulates oligodendrogenesis and evokes white matter remyelination in the injured brain.
BMSC Treatment Activates the Shh/Gli1 Signaling Pathway
The Shh/Gli1 signaling pathway regulates oligodendrogenesis in the adult rodent CNS (Wang et al, 2007). To determine whether BMSCs induce Shh/Gli1 signaling activation, we examined the expression of Shh and its effector Gli1. Immunostaining showed that Shh+ and Gli1+ cells are present in the ipsilateral brain at 14 days after MCAo, and significantly increased after the BMSC treatment compared with the PBS treatment (Figures 1P to 1T). Real-time RT-PCR analysis showed that Shh and Gli1 mRNA expressions are present in the IBZ at 2 days after MCAo. After BMSC treatment, gene expressions of Shh and Gli1 were significantly increased in the IBZ of BMSC-treated rats than that of the PBS-treated controls (Figure 1U).
Using double immunostaining of Shh with NeuN, GFAP, and 04, respectively, we found that Shh+ cells colocalized with NeuN+ neurons (Figures 2A to 2D), GFAP+ astrocytes (Figures 2E to 2H), and 04+ oligodendrocytes (Figures 2I to 2L). Thus, Shh protein may be released from neurons, astrocytes, and oligodendrocytes (Jiao and Chen, 2008; Paintlia et al, 2005; Seifert et al, 2005; Wallace, 1999; Wallace and Raff, 1999; Wechsler-Reya and Scott, 1999). Double immunostaining data showed that Gli1+ cells colocalized with NG2+ oligodendrocyte progenitor cells (Figures 2M and 2N), implying that Gli1 activity was involved in oligodendrogenesis after BMSC treatment. These data suggest that BMSCs stimulate the production of Shh by parenchymal cells, evoke oligodendrogenesis, and lead to white matter remodeling in the injured brain.
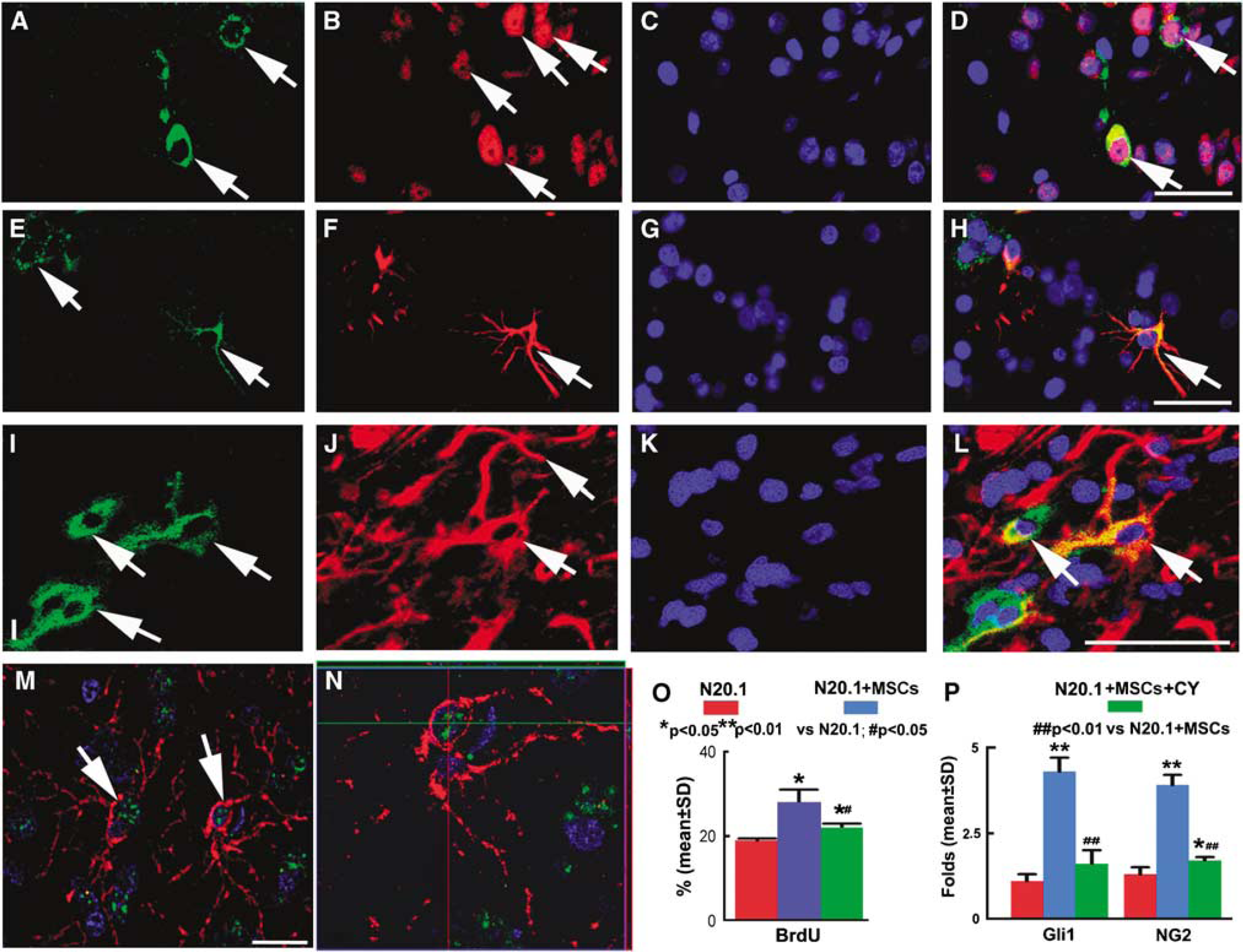
Double immunofluorescence staining (nuclei, DAPI, blue) showed that Shh+ cells (FITC, green) were reactive to NeuN+ (CY3, red) (
BMSC Treatment Promotes Oligodendrocyte Proliferation in Cell Culture
As in vivo data suggest that activation of the Shh/Gli1 pathway induces oligodendrogenesis, and BMSC treatment after MCAo promotes Shh/Gli1 gene and protein expression leading to remyelination, we carried out an in vitro study to clarify the relationships among proliferation of N20.1 cells, the Shh/Gli1 pathway, and BMSC treatment. N20.1 cells are premature oligodendrocytes (Paez et al, 2004). Under 1% FBS and 39°C conditions, most of N20.1 cells exhibited a significantly decreased growth rate. BMSC treatment significantly increased the N20.1 cell proliferation, as indicated by BrdU immunostaining, compared with the normal medium group (P<0.01, Figure 20). BMSC-induced N20.1 cell proliferation was significantly reduced by cyclopamine, a pharmacological inhibitor of the Shh signaling pathway (P<0.05). These data indicate that BMSCs induce N20.1 cell proliferation through the Shh pathway.
BMSC Treatment Activates the Shh/Gli1 Signaling Pathway in Oligodendrocyte Cell Culture
To test the hypothesis that the effect of BMSCs on oligodendrogenesis is mediated through the Shh/Gli pathways, we used RT-PCR to measure NG2 and Gli1 gene expressions. RT-PCR data showed that the NG2 mRNA level was increased significantly after 12 h of BMSC coculture, implying an increase in the proliferation of N20.1 cell. Gli1 mRNA expression was increased significantly in N20.1 cells after coculture with BMSCs. All these BMSC effects were blocked by the Shh/Gli1 pathway inhibitor, cyclopamine (Figure 2P). These data suggest that activation of the Shh/Gli1 pathway is involved in oligodendrogenesis, and BMSC treatment promotes this activation.
Discussion
BMSCs, including multipotential mesenchymal stem and precursor cells, have broad therapeutic applications to the treatment of neurologic diseases (Chopp and Li, 2002; Chopp et al, 2009). Functional recovery is evident after BMSC treatment of rodents with stroke from 14 days (Chen et al, 2001a, 2003; Shen et al, 2007; Zhang et al, 2004) and up to 1 year (Shen et al, 2007) after ischemic injury. BMSCs stimulate the production of restorative factors by parenchymal cells, evoke white matter remodeling in the injured brain, and lead to improvement in neurologic function post stroke (Chopp and Li, 2002; Chopp et al, 2009). Similar restorative benefits were also obtained using other cell sources for transplantation in stroke (Borlongan, 2009; Chen et al, 2001b). In this study, we show that BMSC treatment of rats subjected to MCAo induces oligodendrogenesis and activates the Shh/Gli1 pathway, which may contribute to functional recovery. We note, however, that our data do not show a causal relationship between oligodendrocyte proliferation and functional benefit. There are likely many factors that contribute to the BMSC-mediated functional improvement post stroke (Chen et al, 2002; Chopp and Li, 2002; Chopp et al, 2009; Qu et al, 2007), among which are angiogenesis, neurogenesis, and synaptogenesis (Chopp and Li, 2002; Chopp et al, 2009). As axonal integrity and myelination are necessary for brain function, and stroke often disrupts and damages white matter, it is reasonable to consider the BMSC-mediated induction of oligodendrogenesis as a potential contributing factor in promoting recovery post stroke. To our knowledge, this is the first study to show that a cell-based therapy stimulates oligodendrogenesis and that the Shh pathway may mediate this restorative process.
Oligodendrocytes are the only myelin-producing cells in the CNS that enable rapid electrical conduction of impulses, and they are vulnerable to the ischemic injury (Dewar et al, 2003; Domercq and Matute, 2004). The lack of oxygen and glucose, as well as free radicals, contributes to oligodendrocyte damage after stroke (Shibata et al, 2000). The death of oligodendrocytes after MCAo leads to demyelination, and axonal conduction will be subsequently impaired or lost (Dewar et al, 2003; McTigue and Tripathi, 2008; Micu et al, 2006).
In this study, we focus on the effects of BMSCs on oligodendrogenesis and myelin remyelination. Abundant oligodendrocyte progenitor cells (NG2+) are present in the adult CNS (McTigue and Tripathi, 2008). BrdU and NG2 double immunostaining showed oligodendrocyte progenitor cell proliferation in the CNS after MCAo, and BMSC treatment significantly enhanced this proliferation. This proliferation may contribute to the remyelination, which is suggested by the significantly increased 04+ cells and MBP+ area. These data suggests that BMSC treatment promotes oligodendrocyte progenitor cell proliferation and differentiation into myelinated oligodendrocytes. Remyelination is likely a requisite for functional recovery, and recruited oligodendrocytes can replace the damaged oligodendrocytes, remyelinate axons, and reduce axonal loss.
Shh plays a critical regulatory role in oligodendrogenesis and facilitates remyelination (Marti and Bovolenta, 2002; Murray et al, 2002; Nery et al, 2001; Orentas et al, 1999; Seifert et al, 2005; Sussman et al, 2002), and Gli1 is the principal effector of Shh (Ahn and Joyner, 2005; Wang et al, 2007). Our data show Shh and Gli1 mRNAs are present in the IBZ at 2 days, and protein expressions of Shh and Gli1 were present in the ipsilateral brain at 14 days after MCAo, and BMSC treatment significantly enhanced these expressions. Double immunostaining data suggest that the Shh protein expressed in neurons, astrocytes, and oligodendrocytes and in the Gli1 activity are involved in oligodendrogenesis. These data suggest that BMSC treatment-mediated enhancement of oligodendrogenesis may occur by promoting the production of Shh by parenchymal cells and activation of the Shh/Gli1 pathway in the ischemic brain. Also, our in vitro study shows that BMSCs stimulate N20.1 cell proliferation and Gli1 mRNA expression. Blockage of the Shh pathway with the Shh antagonist, cyclopamine, abolished the BMSC-induced proliferation of N20.1 cells, and also decreased Gli1 mRNA expression in N20.1 cells, implying that BMSCs promote oligodendrocyte proliferation by activation of the Shh/Gli1 pathway. These data suggest that activation of the Shh/Gli1 pathway underlies BMSC-induced oligodendrogenesis.
In summary, our data indicate that BMSC treatment promotes oligodendrogenesis and remyelination. Activation of the Shh/Gli1 pathway may underlie the restorative effects of BMSCs after MCAo in rats.
Footnotes
Acknowledgements
The authors thank Mr. Siamak Pourabdollah, Dr Lei Wang, Cynthia Roberts, Supata Santra and Qinge Lu for their technical assistance.
The authors declare no conflict of interest.
