Abstract
The endothelial nitric oxide synthase (eNOS) gene harbors three well-characterized genetic variants, which have been reported to be associated with various vascular diseases. Recently, conflicting results have been published relating to the role of these genetic variants in the pathogenesis of intracranial aneurysms (IA). Therefore, we analyzed these variants in a large European population of IA patients and controls. In all, 142 patients with IA and 190 controls were enrolled in our study. The −786T > C and 894G > T single-nucleotide polymorphisms (SNPs) were analyzed by direct sequencing of the corresponding sections in the genomic DNA. A variable number tandem repeat (VNTR) located in intron 4 of the gene and consisting of either four or five 27-base pair (bp) repeats was analyzed by polymerase chain reaction amplification and electrophoresis using ALF sequencer™ equipment. Genotype and allele frequencies were determined, and the frequencies in cases and controls were compared. In addition, haplotypes were constructed. There were no deviations from Hardy–Weinberg equilibrium. Genotype and allele frequencies did not differ significantly between cases and controls in any sample group or after stratification for multiple IA or aneurysm size. No single haplotype was significantly associated with the phenotype of an IA. The −786T > C, 894G > T, and 27-bp VNTR genetic variants of the eNOS gene are not associated with IA in the European population.
Introduction
The prevalence of intracranial aneurysm (IA) (MIM105800) is about 2% to 6% in Western Europe and the United States (Rinkel et al, 1998). Rupture of an IA occurs with an overall incidence of about 10/100,000/year in these countries (as against up to 25/100,000/year in Finland and Japan) and causes subarachnoidal hemorrhage (SAH), a disease with high morbidity and mortality constituting a major public health problem (Longstreth Jr et al, 1993; Inagawa et al, 2000; Inagawa, 2001). The treatment of, unruptured, incidental IA is associated with a much better prognosis (Inoue, 2002; Vindlacheruvu et al, 2005), but with current radiologic methods screening for IA in the general population is not justified, because of risks associated with an invasive technique like the digital subtraction angiography (DSA), or the costs in the case of magnetic resonance imaging-based screening. Identification of those at risk by biochemical means requires an understanding of the molecular pathogenesis of IA and the identification of molecular markers.
There are several lines of evidence to show that endothelial nitric oxide synthase (eNOS) has a major role in the early development of aneurysms (Faraci and Brian Jr, 1994). Endothelial nitric oxide synthase is one of three isoforms that are the source of NO production (Moncada et al, 1991), which in turn has a central role within the extracellular matrix, being a potent vasodilator and a powerful inhibitor of inflammation, platelet aggregation, and smooth muscle cell proliferation (Garg and Hassid, 1989; Gurjar et al, 1999; Chiang et al, 2001). Several studies have shown that the NO pathway is impaired, and NO levels in cerebrospinal fluid are elevated in experimentally induced SAH in animals (Weir and MacDonald, 1993; Pluta, 2005). Most strikingly, NO knockout mice are prone to various vascular diseases, and particularly to the development of aortic aneurysms (Kuhlencordt et al, 2001). Although the molecular pathogenesis of aortic and IA seems to be different, these data are indicative for the role of eNOS in extracellular matrix remodeling.
The eNOS gene is located on chromosome 7q35–36 (OMIM163729) and harbors three well-defined polymorphisms. One, −786T > C, located in the promoter region of the gene, was previously found to be associated with the occurrence of coronary vasospasm (Nakayama et al, 1999). Another, 894G > T, is associated with coronary artery disease (CAD), acute myocardial infarction (AMI), and carotid atherosclerosis (Lembo et al, 2001; Hingorani, 2003), and the third genetic variant, a variable number tandem repeat (VNTR) located in intron 4 of the gene, consists of either four or five 27-base pair (bp) repeats and is associated with CAD and AMI (Wang et al, 1996) and with the occurrence of aortic aneurysms (Kotani et al, 2000). Whether these polymorphisms have a functional impact on the gene product is a matter of an ongoing debate (for a review see Wang and Wang, 2000).
Recently, Khurana et al (2004) analyzed these three genetic variants in 51 SAH patients admitted to the Mayo Clinic and in 90 controls. In their study, the four-repeat allele of the VNTR was found to be associated with increased susceptibility to aneur-ysm rupture and the C-allele of the −786T > C single-nucleotide polymorphism (SNP), was found to be significantly associated with the occurrence of post-SAH vasospasm. In a previous study by the same group, an association was found between the −786T > C SNP and IA size (Khurana et al, 2003), suggesting that the heterozygous state is significantly more frequent in patients with aneurysms measuring > 10mm. However, a large cooperative Japanese and Korean study has not confirmed these results (Akagawa et al, 2005).
Therefore, we conducted the present case–control study to investigate whether these three eNOS genetic variants were associated with the phenotype for single or multiple IA in a large European population and whether we could confirm an association between genotype and IA size.
Materials and methods
Study Population
The study group consisted of 142 unrelated, consecutively recruited patients, each with at least one IA that had been confirmed by cerebral angiography. All patients were resident in the Dresden urban area and were operated on or the IA was coiled by an endovascular approach in the Department of Neurosurgery or Neuroradiology, respectively, of the University of Technology in Dresden. The clinical characteristics of the patient group are listed in Table 1.
Patients' characteristics
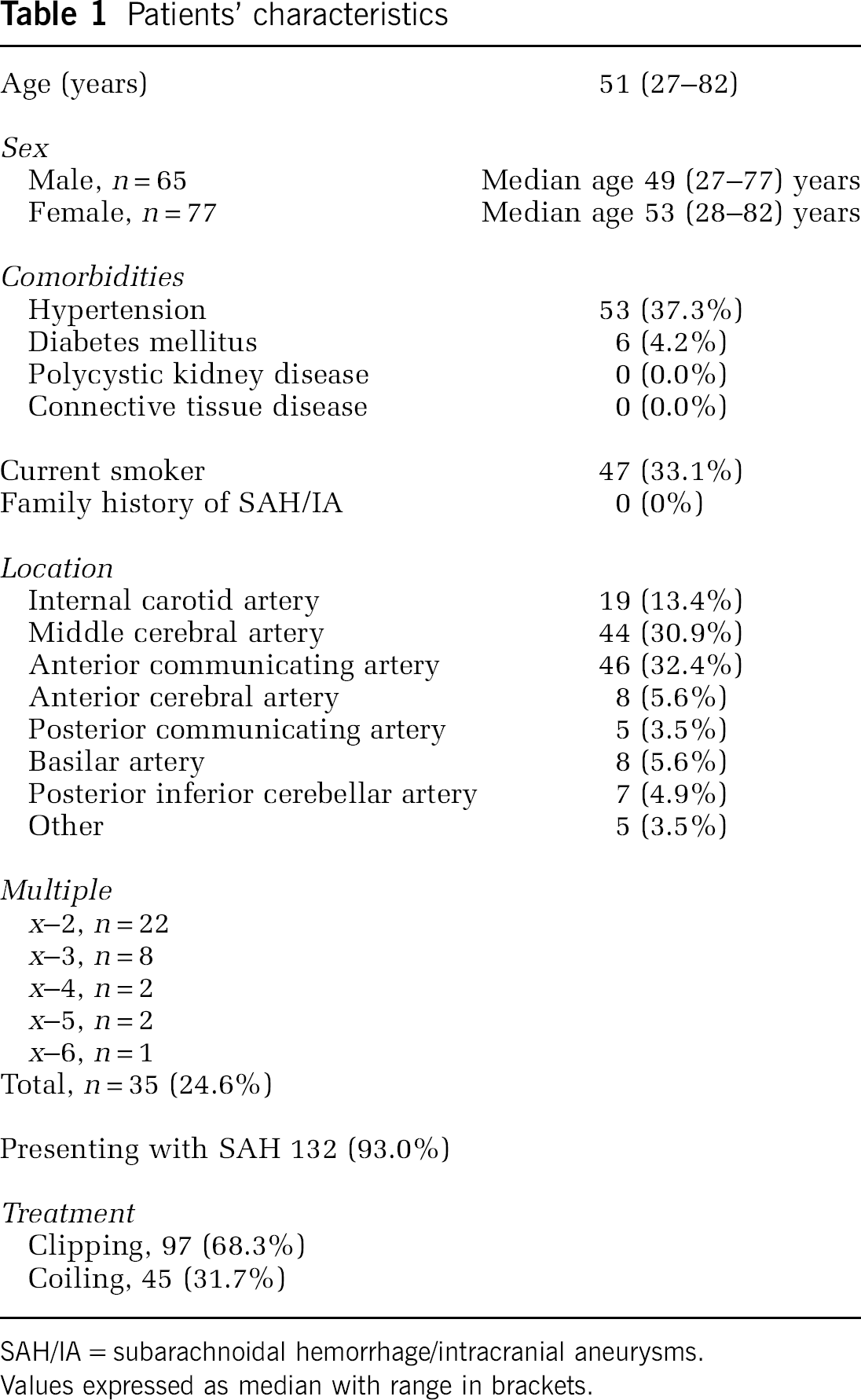
SAH/IA = subarachnoidal hemorrhage/intracranial aneurysms.
Values expressed as median with range in brackets.
The control group consisted of 190 anonymous, healthy blood donors. It should be emphasized that the control subjects were enrolled from the same geographical area as the patients. Because of its historical background, the city of Dresden is populated by a homogenous European population. In general, blood is donated by people of all social classes in Germany. All potential blood donors are routinely screened for disease by means of a questionnaire, red blood cell and white blood cell counts, and liver function tests (details are available from the authors on request). This study was approved by the local ethics committee. Informed written consent to genetic analysis was obtained from all nonanonymous individuals.
Sequence Analysis
Genomic DNA was extracted from peripheral blood lymphocytes using either the QIAmp® DNA Blood Minikit (Qiagen, Hilden, Germany) or the MagNa Pure LC nucleic acid isolation kit® (Roche Applied Science, Mannheim, Germany).
For amplification of genomic DNA, primer pairs were used as published previously (Table 2).
Primer sequences
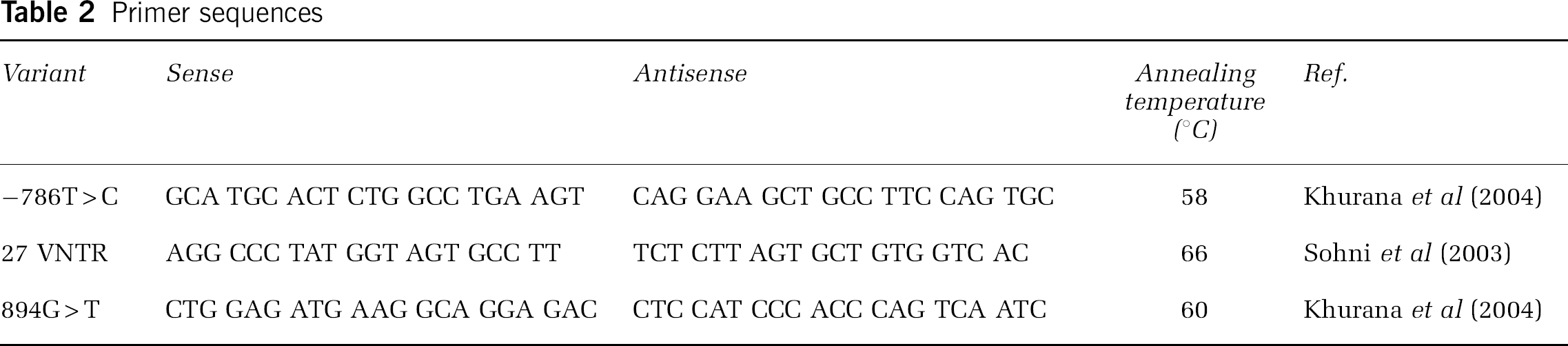
Polymerase chain reaction (PCR) was performed using a thermal cycler (Perkin-Elmer, Applied Biosystems GmbH, Weiterstadt, Germany): 1 μmol/L of each primer was mixed with 100 ng genomic DNA, 200 μmol/L deoxynucleotide triphosphates (Promega, Madison, WI, USA), 2.0 mmol/L MgCl2, 10 × PCR buffer (50 mmol/L KCl, 500 mmol/L Tris buffer, 160 mmol/L (NH4)2SO4, pH 8.8, and 0.1% Tween 20), and 0.5 U Taq polymerase (all from InViTek GmbH, Berlin, Germany) to give a final volume of 25 μL. For a negative control, water was used instead of genomic DNA in the PCR. Amplifying conditions were: initial denaturation at 94°C for 5 mins, followed by 35 cycles of denaturation at 94°C for 30 secs, annealing at various temperatures (see Table 2) for 30 secs, and extension at 72°C for 90 secs, with a final extension step at 72°C for 7 mins.
Polymerase chain reaction products were electrophoresed on a 0.8% agarose gel, and bands were cut out and purified on Sephadex G-50 Fine columns (Amersham Pharmacia Biotech AB, Uppsala, Sweden). Direct sequencing was performed using an Applied Biosystems 3777 automated DNA sequencer according to the manufacturer's instructions.
For detection of the VNTR in intron 4, a cyanine5 endlabeled sense primer was used for PCR. Products were run on an ALF Express® sequencer using denaturing 6.5% Long Ranger™ gels, which were run at 40 W (1000 V, 38 mA) for 4 h. Runs were analyzed using ALF Evaluation™ software. Random samples were selected for sequence analysis (see above).
Data Analysis
All variants investigated were evaluated for deviations from Hardy–Weinberg equilibrium by comparing observed and expected genotype frequencies by means of an exact goodness-of-fit test, which was applied separately for cases and controls. Fisher's exact test was used in both patients and controls to check for an association of any eNOS genetic variant genotype with IA.
Haplotype frequencies were estimated with the expectation–maximization algorithm and the frequencies in cases and in controls were compared, calculating the χ2 statistic with simulated P-values.
Nomenclature
Polymorphisms are referred to as in the system of nomenclature recommended by den Dunnen and Antonarakis (2001) (http://www.hgvs.org/mutnomen). The alleles harboring four and five repeats of the VNTR are termed 4a and 4b, respectively (Wang et al, 1996).
Results
All genotype frequencies detected, whether in the patients, in the controls, or in any subgroup, were in keeping with those predicted by Hardy-Weinberg equilibrium.
Genotype and allele frequencies did not differ significantly between cases and controls for any of the three genetic variants analyzed: −786T > C, 27-bp VNTR, and 894G > T (Table 3).
Comparison of genotype and allele frequencies in cases and in controls
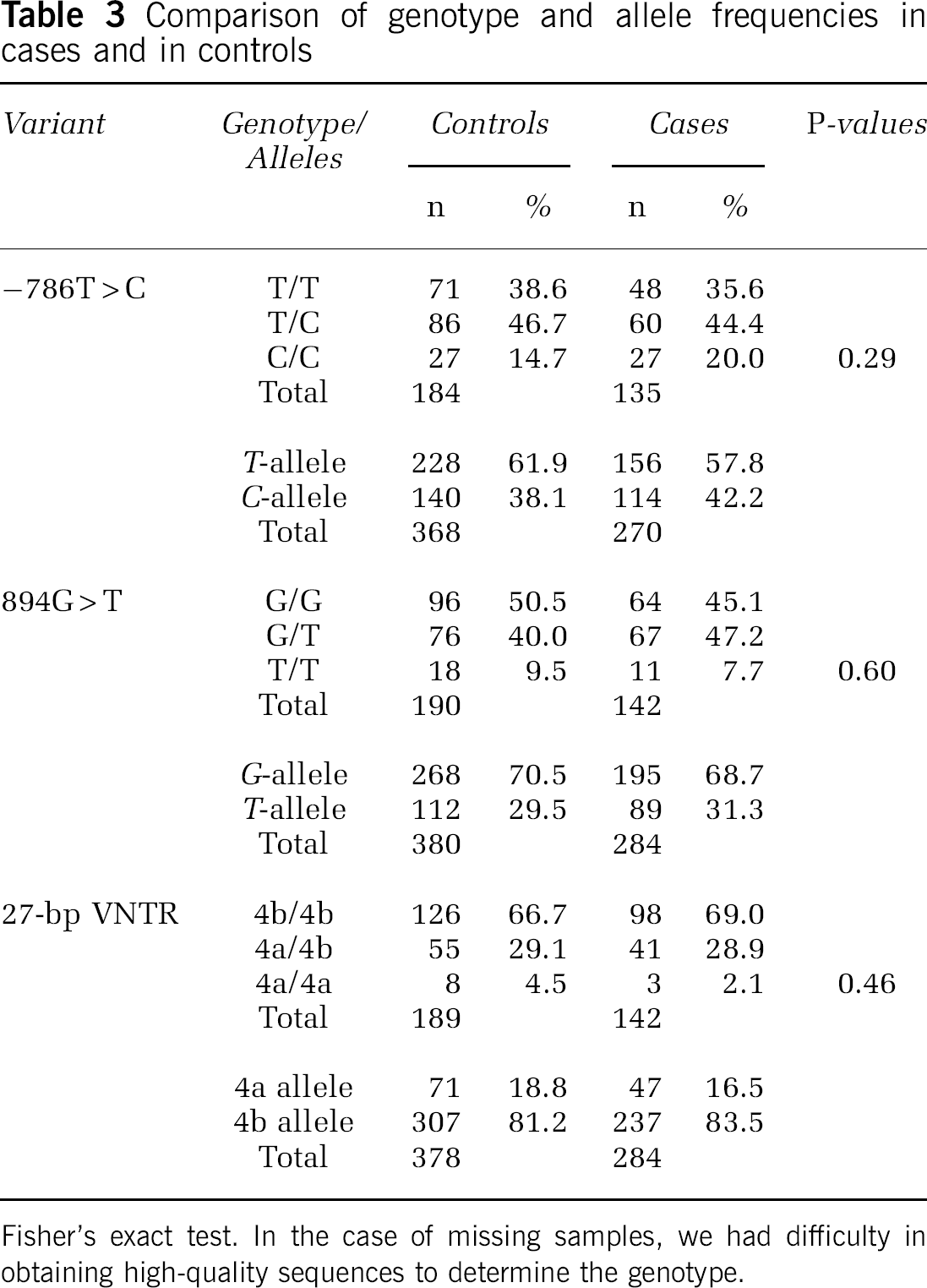
Fisher's exact test. In the case of missing samples, we had difficulty in obtaining high-quality sequences to determine the genotype.
As a multiple IA phenotype suggests that genetic variants might be more pronounced in the particular group with this phenotype, we analyzed all 35 patients who had had more than one IA again separately. However, there was also no difference in any of the three polymorphisms between cases and controls in this subgroup (Table 4).
Comparison of genotype and allele frequencies in patients with multiple IA and in controls
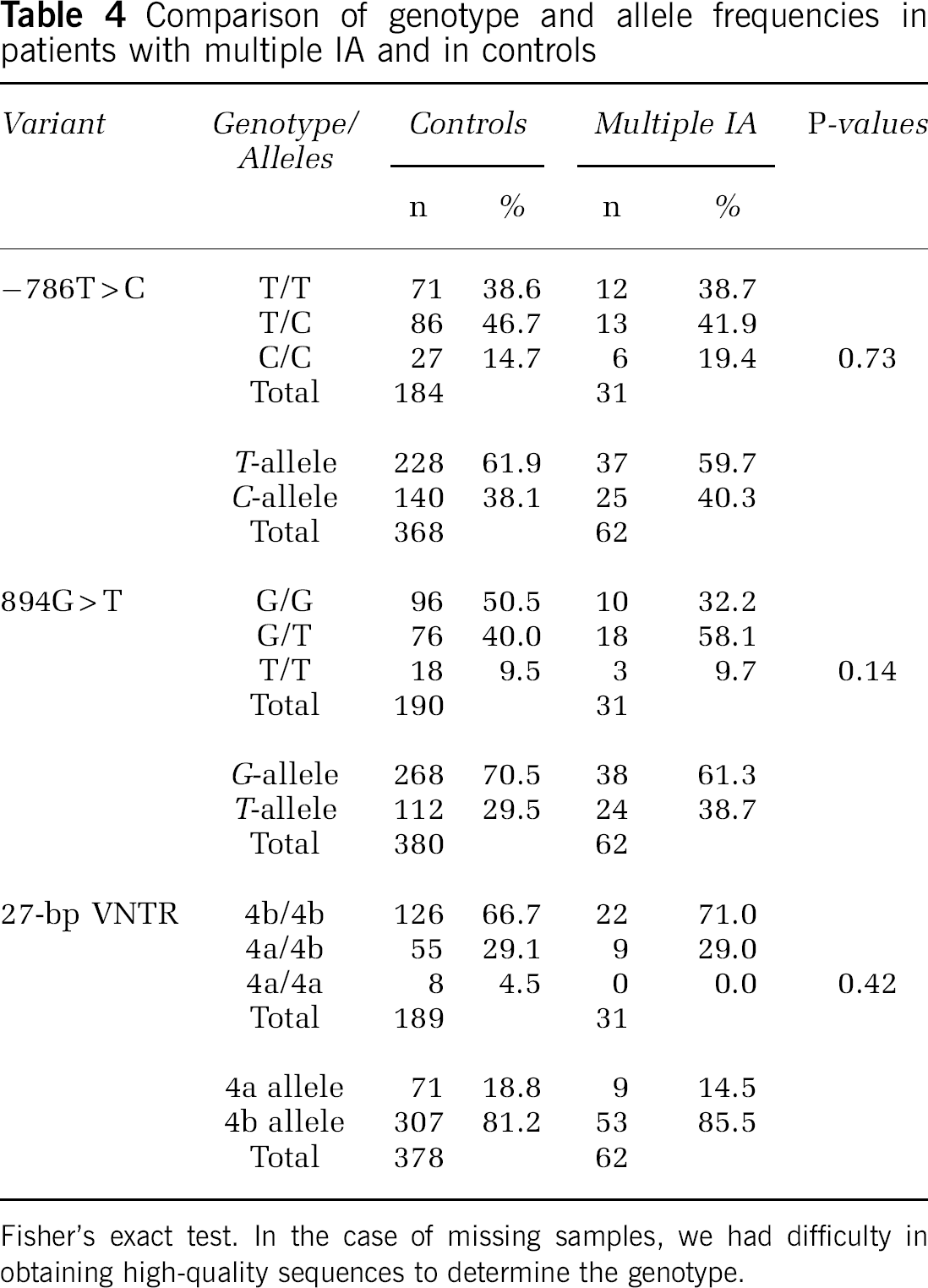
Fisher's exact test. In the case of missing samples, we had difficulty in obtaining high-quality sequences to determine the genotype.
The −786 T > C SNP has been associated with the size of IA. Therefore, the largest diameter of the IA that had caused an SAH was determined in 95 patients. Genotypes and allele frequencies of the −786T > C SNP in patients with IA diameters of 0 to 5, 6 to 9, and > 10 mm are presented separately in Table 5. Again, genotype frequencies did not differ in any of the three groups (0 to 5, 6 to 9, and > 10 mm). However, the T-allele, which is the more common allele of this SNP, trends to be over-represented in the group with IA 0 to 5 mm in diameter, but this difference did not achieve statistical significance (P = 0.27).
Genotype and allele frequencies of −786T > C SNP in IA patients by IA diameter
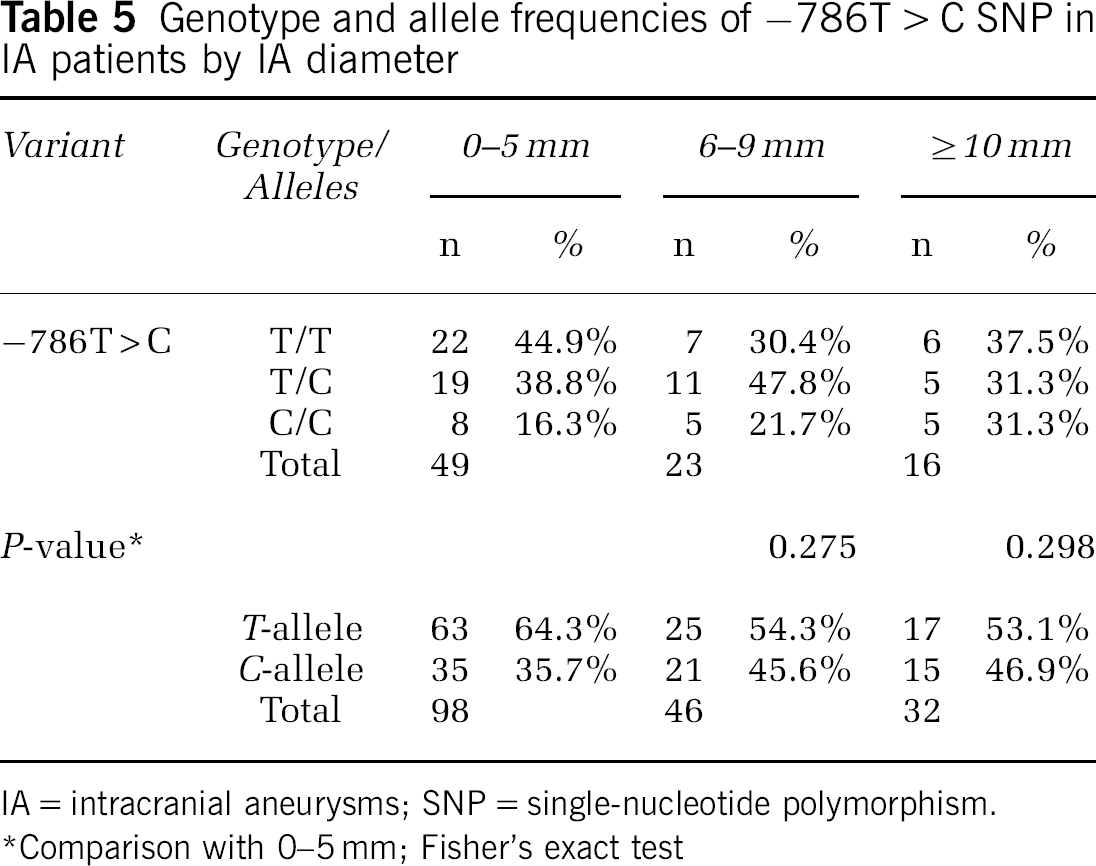
IA = intracranial aneurysms; SNP = single-nucleotide polymorphism. *Comparison with 0-5 mm; Fisher's exact test
Haplotypes were constructed, and the T-4b-G haplotype (i.e., that with variants located in promoter–intron 4–exon 7) was identified as the most common. Haplotype C-4a-G was over-represented, whereas haplotype C-4b-G was found less frequently than expected, but there was no association with phenotype (simulated P = 0.090).
Discussion
In our study, we have analyzed the −786T > C SNP, located in the promoter region, the 894G > T SNP located in exon 7, and a 27-bp VNTR located in intron 4 of the eNOS gene, where conflicting results have been published with regards to the role of these variants in the pathogenesis of IA.
The minor allele (4a) containing four repeats of the 27-bp VNTR was found to be over-represented in patients with ruptured IA (P = 0.002) in a North American study, suggesting that this genetic variant might be a risk factor for aneurysmal SAH (Khurana et al, 2004). In our study, genotype and allele frequencies in cases and in controls were almost identical, indicating that this particular variant might not be involved in the pathogenesis of IA in the European population. Our study population was large enough to detect the differences indicated in the North American study with a power of more than 99% on the 0.05 significance level. All detected genotype frequencies are in agreement with the Hardy–Weinberg equilibrium. Although we did not use a population-based control group or specifically selected controls as described by Morton and Collins (1998), our control population was well suitable. Because of the low population prevalence of IA, which is about 2% in Western Europe (excluding Finland), it is not necessary to use a sex- and age-matched control population proven to be free of IA as discussed by Owen et al (1997).
There is no difference in genotype frequencies between our controls and the North American control population, underlining the comparability of both samples.
In both studies, patients and controls were of Caucasian origin. Therefore, a different ethnic background, which is a well-known source of varying genotype frequencies, can largely be excluded. Genotype distribution might also be affected by environmental factors, particularly smoking, which, in turn, might be an explanation for the different results found in the North American and European populations, respectively.
In another of their papers, the same authors suggest that the −786T > C SNP of the eNOS gene is associated with larger aneurysm size (Khurana et al, 2003). In this study, all 13 patients with aneurysms ≥ 10 mm in diameter were heterozygous for that SNP, whereas only 13 (59%) of 22 patients with IA 0 to 5 mm in diameter and 9 (53%) of 17 patients with IA 6 to 9 mm in diameter were found to be heterozygous for the same SNP. These results could not be confirmed by Akagawa et al (2005) in their study investigating the −786T > C SNP in 336 Japanese and 191 Korean patients, and 214 Japanese and 191 Korean controls. In their study, genotype frequencies did not differ significantly between subgroups with aneurysms of different sizes in either the Japanese or the Korean study populations. There is only an insignificant tendency for the C-allele to be more frequent in the presence of IA measuring ≥ 10 mm across than in smaller ones, and for the T-allele to be more frequent in IA 0 to 5 mm in diameter. These data are in agreement with our findings. We did not find a difference between cases and controls in genotype frequencies for the −786T > C SNP after stratification for size, whereas there was a tendency for the T-allele to be more frequent in smaller (0 to 5 mm) IA.
Interestingly, while there is no difference in genotype frequency for the −786T > C SNP between our European population and the North American population of Caucasian origin, there is a tremendous difference in genotype frequency between our European population and the Asian control populations (P < 0.001; Table 6). This is indicative of the well-known influence of different ethnicity on genotype expression, and, this genotype distribution—a difference between two ethically different populations, but no difference between cases and controls—supports the hypothesis that there is no association between −786T > C genotype frequencies and an IA phenotype.
−786T > C SNP in various studies differing in ethnicity of study population

Difference between the two Caucasian populations combined and the Japanese and Korean populations combined. There are no differences between the individual Caucasian populations or between the Japanese and the Korean populations.
In addition, it had been observed that the −786T > C SNP is also associated with post-SAH vasospasm (Khurana et al, 2004). We cannot confirm this result, as in our database the occurrence of vasospasm diagnosed by clinical presentation, elevated flow velocities in transcranial Doppler ultrasound, and/or DSA is not uniformly recorded. The evaluation of these data would have led to a major bias. Therefore, we decided not to stratify for that factor; however, our ongoing data sampling will enable us to answer this question in the near future.
The 894G > T SNP leading to a conservative substitution from glutamate to aspartate has been shown to be functional (Tesauro et al, 2000). Aspartate, but not a glutamate, at amino-acid position 298 is subject to cleavage in normal tissue and in cells overexpressing eNOS. Although case–control studies have shown an association with CAD, AMI, and carotid atherosclerosis (Lembo et al, 2001; Hingorani, 2003), no association has been found between that SNP and ischemic stroke in two studies (Markus et al, 1998; MacLeod et al, 1999). Like in the above-mentioned North American studies, we too could not substantiate an association between the 894G > T SNP and an IA phenotype in the European population.
In our study, we concentrated on three well-characterized genetic variants of the eNOS gene, which were not found to be associated with IA in a European population. Our approach was not suitable for analysis of the entire coding sequence or regulative elements of the gene. Therefore, we cannot exclude the possibility that other genetic variants in those regions might be associated with the development of IA in the European population.
Footnotes
Acknowledgements
We would like to thank Janet Dodsworth and Kartik Krishnan for their assistance in preparing the manuscript, and Ina Esswein for her help in collecting clinical data. We thank all members of the Department of Radiology and Neuroradiology for their collaboration.
There is no conflict of interest. All authors disclose any involvement, financial, or otherwise.
