Abstract
Estrogens are believed to provide females with endogenous protection against cerebrovascular events although clinical trials studying long-term hormone replacement have yielded disappointing results. In contrast, estrogens might be neuroprotective after experimental ischemia. We performed a systematic review of controlled experimental studies that administered estrogens before, or after, cerebral ischemia and measured lesion volume. Relevant studies were found from searching PubMed, Embase and Web of Science. From 161 identified publications, 27 studies using 1304 experimental subjects were analyzed using the Cochrane Review Manager software. Estrogens reduced lesion volume in a dose-dependent manner, after either transient (P < 0.001) or permanent (P < 0.001) ischemia and whether administered before or up to 4 h after ischemia onset; no studies assessed efficacy for later time periods. The effect size for estrogens decreased with increasing quality scores for studies of transient ischemia. Estrogens reduced lesion volume when administered to ovariectomized females and young adult males, but had no effect in intact females. Limited data were present for aged animals and the full dose-response relationship was not available in all experimental groups. On the basis of these data, estrogens are a candidate treatment for ischemic stroke, although further preclinical studies are also warranted.
Introduction
Before the menopause, women have a lower risk of stroke relative to men of the same age (Kannel and Thom, 1994; Sacco et al, 1997). After the menopause, the incidence of stroke in women rapidly increases (Wenger et al, 1993), coincident with diminished circulating levels of estrogens and progesterone. Thus, steroid hormones appear to provide females with a certain degree of endogenous protection against stroke occurrence and stroke damage.
Experimental studies also support the concept that steroid hormones provide females with endogenous protection against stroke. These animal studies show that females suffer less ischemic damage than males, the protection in females is absent after ovariectomy, and exogenously applied estrogens reduce ischemic damage in males and females (for reviews, see Murphy et al, 2004; Simpkins et al, 2005). Such observations have been replicated across a variety of species, strains, and research laboratories, and the outcomes from experimental animal studies have been fundamental in supporting the need for clinical trials of estrogens.
Hormone replacement therapy (HRT) has been widely tested in clinical trials aimed at reducing the occurrence of vascular events. While some studies reported a significant reduction in stroke occurrence after HRT (Finucane et al, 1993; Henderson et al, 1991), others found no significant benefit (Bushnell et al, 2001; Pedersen et al, 1997; Petitti et al, 1998; Viscoli et al, 2001). More recently, the Women's Health Initiative (WHI) trial, assessing the effect of HRT on incidence and outcome after stroke and other events, was halted prematurely because of increased hazard (Wassertheil-Smoller et al, 2003). A systematic review of completed clinical trials found that HRT was associated with an elevated risk of stroke, which was ischemic in type and of increased severity (Bath and Gray, 2005).
The discrepancy in findings between long-term clinical use (HRT increases stroke) and short-term use in preclinical models of ischemia (estrogens might be neuroprotective) remains unexplained. Key factors include the relationship between the design of clinical and experimental studies and interpretation of the latter in terms of dose, timing of administration, sex, and age.
To evaluate the neuroprotective potential of estrogens, we have performed a systematic review to investigate the neuroprotective properties of exogenously applied estrogens on lesion volume after experimental stroke, including with regards to timing of treatment, therapeutic dose, and effectiveness according to sex and age.
Materials and methods
Study Identification
Experimental controlled studies of the effects of exogenously applied estrogens on infarct size in animal models of stroke were identified from Pubmed, Embase, and Web of Science by searching for all articles published from 1980 to the end of 2004. The earliest study included for analysis was from 1997 (Simpkins et al, 1997). Additional publications were identified from reference lists of all identified publications and nonsystematic review articles (Garcia-Segura et al, 2001; Green and Simpkins, 2000; Hurn and Brass, 2003; Hurn and Macrae, 2000; McCullough and Hurn, 2003; Wise et al, 2001; Wise, 2002; Yang et al, 2001). The search strategy employed the following keywords: estrogens or estradiol, ischemia, and cerebral. However, studies were only included if they met all of the following criteria: ischemia induced, estrogens administered, and infarct volume measured (Figure 1).
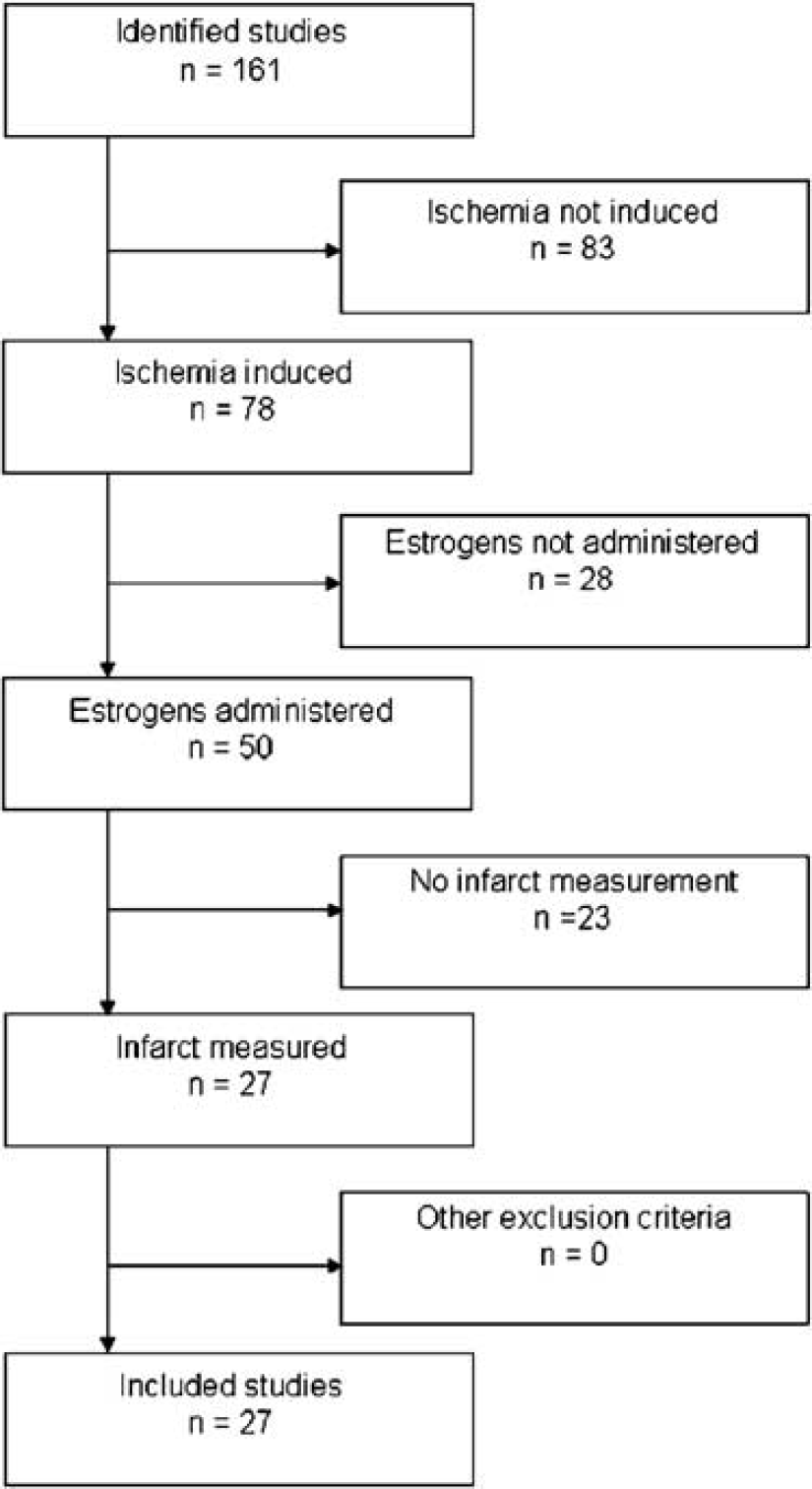
Search process showing reasons for exclusions of studies. A total of 27 studies were included. Studies were excluded if they did not report the following: induction of ischemia, administration of estrogens, or measurement of infarct volume. N, number of studies.
Data Extraction
Two authors (CG, LG) independently extracted data from relevant publications on animal species, number, gender and estrogen status, model of ischemia (permanent, transient), intervention (dose of estrogens, timing relevant to induction of ischemia), and infarct volume (mm3,% of normal brain, mean, standard deviation). A comparison (C) was defined as the assessment of outcome (i.e. lesion volume) in treatment and control groups after treatment with an administered dose of estrogens starting at a stated time before or after the onset of cerebral ischemia. For each comparison, the data for mean outcome, standard deviation, and number of animals per group were extracted. Where the number of animals per group was reported as a range, the lowest numerical value given was used. If published studies (S) used multiple groups, for example, to assess dose-response relationships, then the data from each group were individually extracted for analysis. Infarct volumes were classified as total, and, if available, subcortical and cortical. Occasionally, numerical data were not reported in text and these were extracted from enlarged, photocopied figures.
The methodological quality of each study was assessed using an 8-point Stroke Therapy Academic Industry Roundtable (STAIR) (1999) rating, as described previously (Horn et al, 2001; Macleod et al, 2005; Willmot et al, 2005a, b). One point was given for written evidence of each of the following criteria: presence of randomization; monitoring of physiological parameters; assessment of dose-response relationship; assessment of optimal time window; masked outcome measurement; assessment of outcome at days 1 to 3; assessment of outcome at days 7–30; combined measurement of lesion volume and functional outcome.
Data Analysis
The data were analyzed using Cochrane Review Manager (version 4.2) and Stata (version 7). The effect of estrogens compared with control on total, cortical, and subcortical lesion volume was assessed using the standardized mean difference (SMD); here the difference in effect between the estrogens and control group is divided by the total standard deviation. This allows comparisons to be made across different methods of measurement. These estimates were pooled using the DerSimonian and Laird (1986) random effects model, which is a more conservative method than using a fixed-effects model and takes into account the statistically significant heterogeneity found between studies.
To examine the effects of study characteristics and potential sources of heterogeneity on outcome, stratified metaanalyses were performed with experiments grouped according to: (i) study quality score; (ii) population grouping—all animals, adult males, ovariectomized females, intact females, aged females, aged males; (iii) lesion location; (iv) dose of estrogens; and (v) timing of administration in relation to onset of ischemia.
Publication bias was assessed by visually examining a funnel plot (Figure 2) of precision (reciprocal of standard error) against the SMD, with asymmetry being formally assessed using the Egger test (STATA function ‘metabias’; Egger et al, 1997). Significance was set at P < 0.05 and 95% confidence intervals (CI) are quoted throughout.
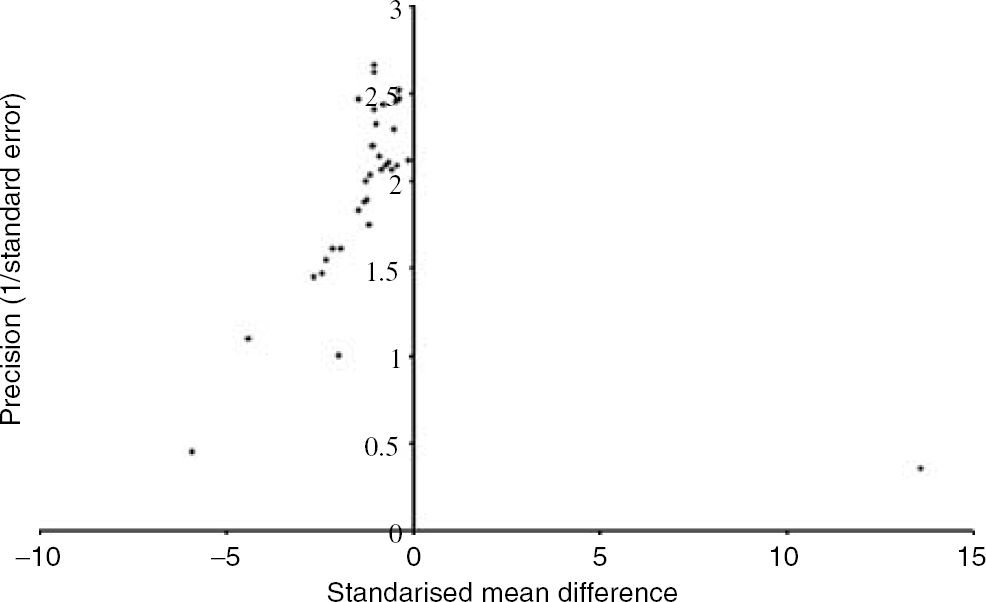
Funnel plot showing the existence of publication bias. Publication bias was assessed by visually examining a funnel plot of precision (reciprocal of standard error) against the SMD and asymmetry was formally assessed using the Egger test. In the above funnel plot publication bias was present for studies reporting the effect of 17/β-estradiol administration on lesion volume in permanent ischemia (Egger's test statistic = −1.65, P = 0.047).
Results
Methodological Design
The literature search identified 161 potential articles, although a large number of these were excluded for the reasons given in Figure 1. The characteristics of the remaining 27 studies are reported in Table 1. All of the included studies reported the effect of exogenously applied estrogens on infarct volume after cerebral ischemia. All of the studies except one (McCullough et al, 2001) administered estradiol, which is the most potent naturally occurring member of the estrogens (Table 1). All of the studies that administered estradiol did so in the form of 17 β-estradiol, apart from one study (Fukuda et al, 2000). In addition, all studies using the model of permanent ischemia applied 17 β-estradiol, whereas transient ischemia studies applied either estradiol or estrone. All included studies compared the effect of administering estrogens versus no estrogens (or vehicle) on infarct volume. The 27 studies represented the outcome from 9 independent research groups. Six of the studies came from 6 different research groups, while the remaining 21 studies represent work from 3 research groups. Within the 27 studies, data from a total of 1304 experimental subjects were included for analysis.
Characteristics of included studies
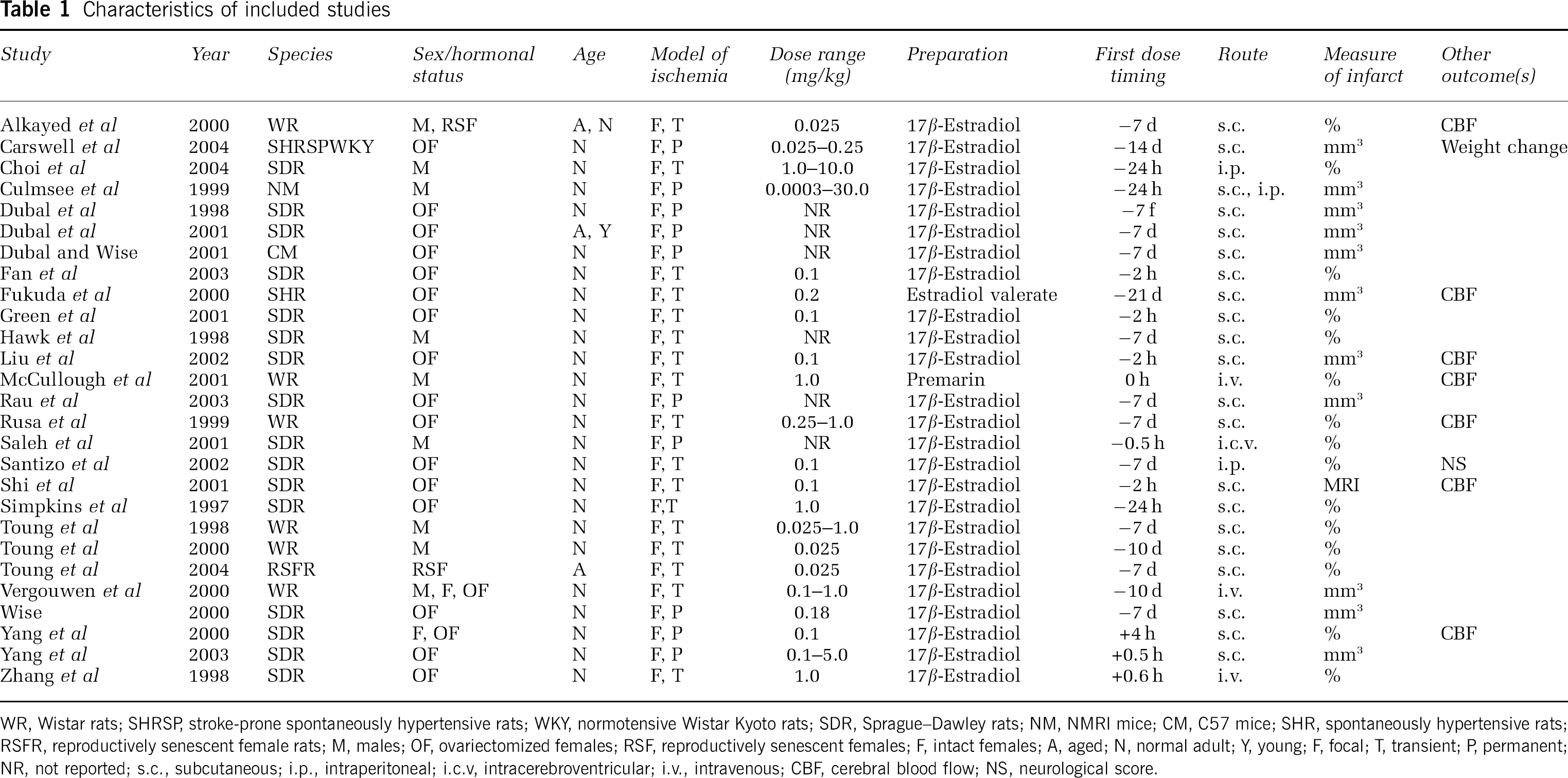
WR, Wistar rats; SHRSP, stroke-prone spontaneously hypertensive rats; WKY, normotensive Wistar Kyoto rats; SDR, Sprageu–Dawley rats; NM, NMRI mice; CM, C57 mice; SHR, spontaneously hypertensive rats; RSFR, reproductively senescent female rats; M, males; OF, ovariectomized females; RSF reproductively senescent females; F, intact females; A, aged; N, normal adult, Y, young F, focal; T, transient, P, permanent; NR, not reported; s.c., subcutaneous; i.p., intraperitioneal; i.c.v. intracerebroventricular; i.v., intravenous; CBF, cerebral blood flow, NS, neurological score.
The majority of studies employed a model of transient focal ischemia (17 studies), with 10 studies reporting the effect of 17 β-estradiol after permanent focal ischemia. A study of estrogens after global ischemia (Horsburgh et al, 2002) was excluded, since data on lesion volume were not reported.
Various rat strains (Wistar, Sprague–Dawley, spontaneously hypertensive, and reproductively senescent) were used in 25 out of the 27 included studies; 2 studies used mice. Methodological design was variable as far as drug administration was concerned. Several routes of administration (subcutaneous, intraperitoneal, intravenous, intracerebroventricular) were used with first dose timings in relation to onset of ischemia varying from 14 days before, to 4 h after.
In terms of outcome measure, lesion volume was assessed by histological staining and reported as follows: lesion volume (mm3), % of total crosssectional area, or % of ipsilateral nonischemic total/region. An exception to this was the study by Shi et al (2001), which used structural MRI to determine lesion size.
Publication bias was present for studies reporting the effect of administering estrogens on lesion volume in permanent (Egger's test statistic = −1.65, P = 0.047, Figure 2) and transient (Egger's test statistic= – 2.30, P = 0.011) ischemia models.
Reported Study Quality
The median STAIR rating for included articles was 3 (range 1–4 out of 8). Animals allocated treatment by randomization were reported in only two studies (Toung et al, 1998, 2004). All but two studies (Wise, 2000; Yang et al, 2003) reported the monitoring of physiological parameters, with most only monitoring body temperature. Nine studies assessed dose-response relationships (Carswell et al, 2004; Choi et al, 2004; Culmsee et al, 1999; Dubal et al, 1998, 2001; Rusa et al, 1999; Toung et al, 1998; Vergouwen et al, 2000; Yang et al, 2000) and eight studies investigated the optimal time window of administering estrogens (Dubal et al, 1998; Fukuda et al, 2000; McCullough et al, 2001; Rau et al, 2003; Saleh et al, 2001; Simpkins et al, 1997; Vergouwen et al, 2000; Yang et al, 2000). All but two studies (Saleh et al, 2001; Shi et al, 2001) assessed the outcome at days 0 to 3; one study (Simpkins et al, 1997) assessed the outcome at days 7 to 30, and another (Santizo et al, 2002) reported the combined measurement of lesion volume and functional outcome.
The latter was the only study to report outcome measures being masked to treatment.
After permanent ischemia, a beneficial effect of 17 β-estradiol treatment was observed regardless of reported quality score (P < 0.001, Figure 3A). The majority of studies utilized the model of transient focal ischemia and the beneficial effect of administering estrogens decreased with increasing reported quality score (Figure 3B). In fact, there was no beneficial effect of administering estrogens in studies that obtained a quality score of 4, which was the highest score awarded (P = 0.39).
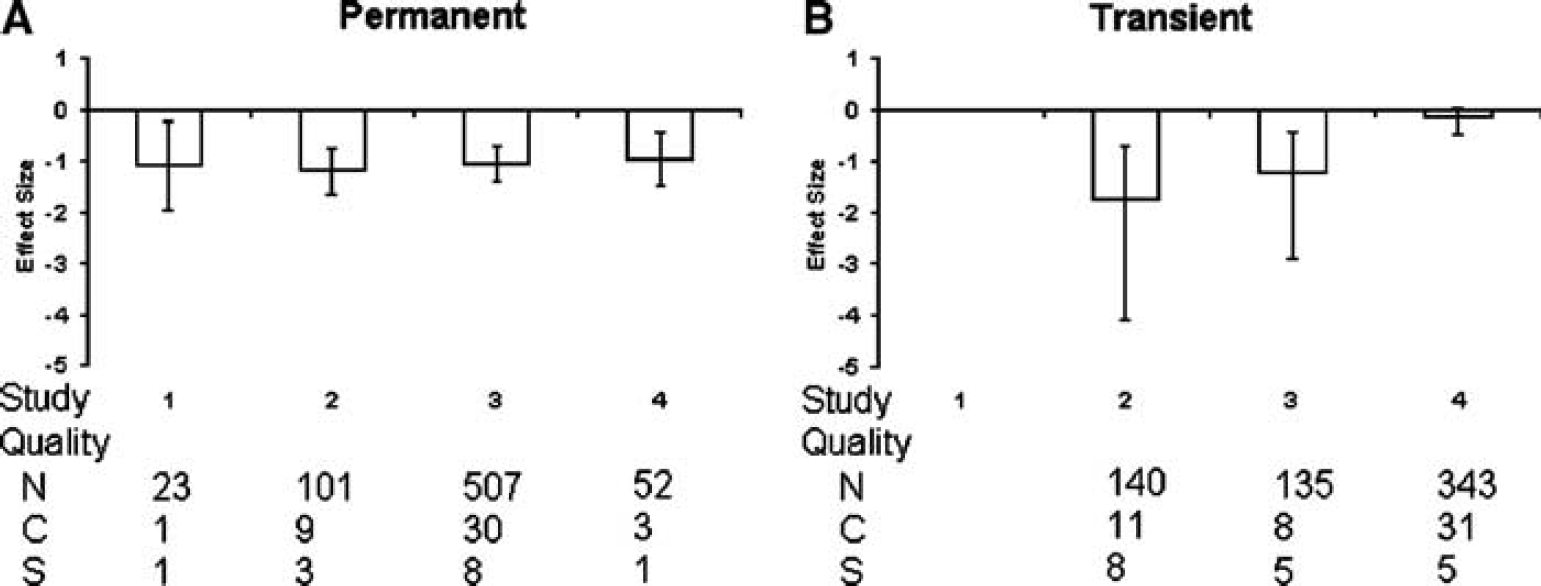
Standardized mean difference and 95% CI by reported STAIR score after permanent
Infarct Volume According to Hormonal Status/Age
The effects of estrogens on total, cortical and subcortical lesion volume were analyzed (Figure 4) according to population groupings based on hormonal status/age, that is, males, ovariectomized females, intact females, aged females, and aged males.
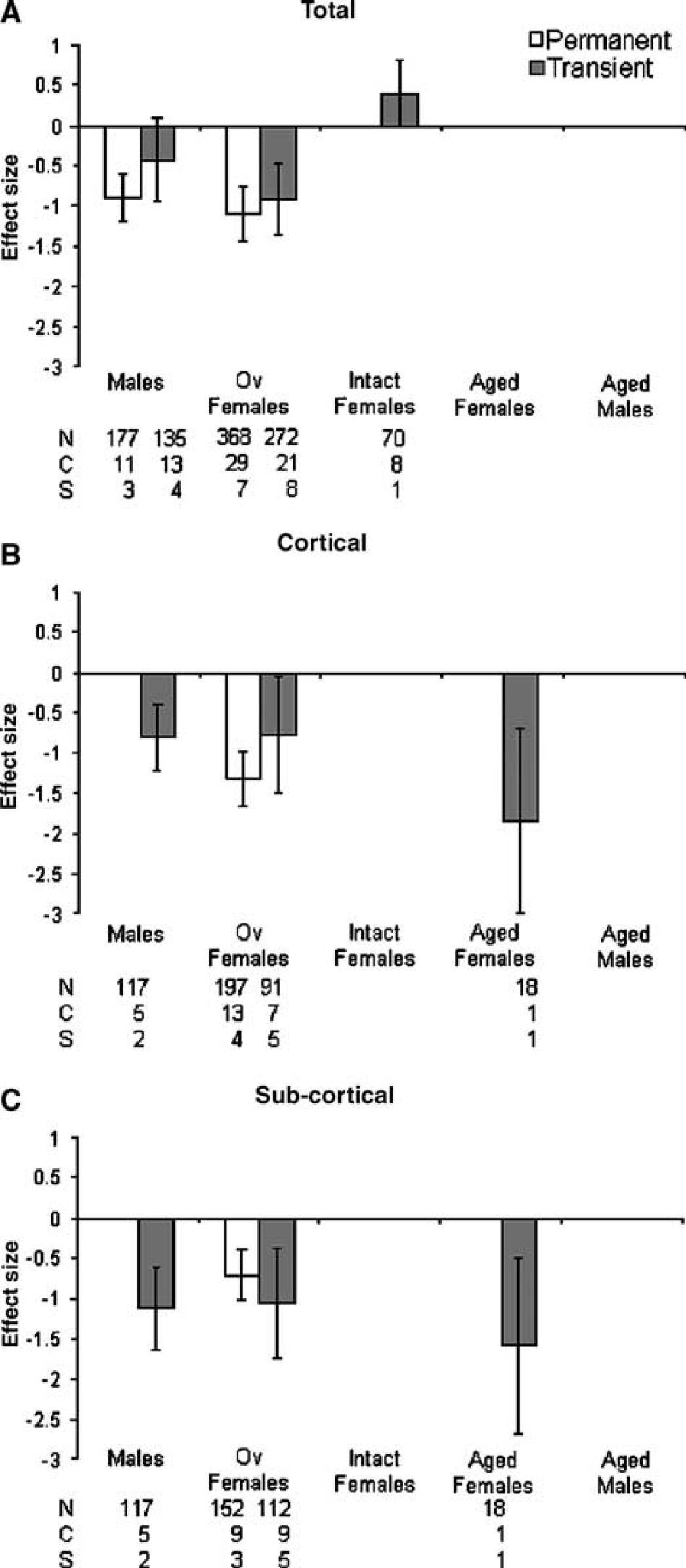
Standardized mean difference and 95% CI for total
Administration of estrogens significantly reduced the total lesion volume after permanent (effect size = −1.05, 95% CI = −1.31 to −0.78, P < 0.001) and transient ischemia (−0.48, −0.78 to −0.17, P = 0.002, Figure 4A). After permanent ischemia, 17 %T-estradiol had a slightly greater beneficial effect in ovariectomized females compared with males. After transient ischemia, the largest effect of estrogens on total lesion volume was also seen in ovariectomized females. However, estrogens appeared to have a detrimental effect when administered to intact females (Figure 4A), although this was not significant (P = 0.06). Significantly different estimates were found within hormonal status groups (males, ovariectomized females, intact females) for total lesion volume after permanent (χ2 = 99.49, df=39, P ≤ 0.001) and transient ischemia (χ2 = 109.19, df=41, P < 0.00001).
Exogenously applied estrogens significantly reduced cortical lesion volume when measured after permanent (−1.32, −1.66 to −0.98, P < 0.001) and transient ischemia (−0.86, −1.24 to −0.48, P < 0.001, Figure 4B). The effects of 17 β-estradiol on cortical lesion volume after permanent ischemia have only been reported in ovariectomized females, and between-group heterogeneity analysis was not possible. After transient ischemia, administration of estrogens was most effective at reducing cortical lesion volume in aged females, although this was based on data extracted from one published study (Figure 4B). Significantly different estimates were found within hormonal status groups for cortical lesion volume (χ2 = 21.9, df = 11, P = 0.02).
In addition, estrogen treatment significantly reduced subcortical lesion volume when measured after permanent (−0.72, −1.03 to −0.40, P < 0.001) and transient ischemia (−1.10, −1.49 to −0.72, P < 0.001, Figure 4C). Again, the effects of 17 β-estradiol on subcortical lesion volume after permanent ischemia were only examined in ovariectomized females. After transient ischemia (one study), estrogen treatment was most effective at reducing cortical lesion volume in aged females (Figure 4C). Significantly different estimates were found within hormonal status groups for subcortical lesion volume (χ2 = 23.6, df = 13, P = 0.04).
Dose of Estrogens
Taking all animals into account, that is, regardless of age/hormonal status, estrogens significantly reduced lesion volume after permanent (−1.05, −1.31 to −0.78, P < 0.001) and transient ischemia (−0.45, −0.75 to −0.14, P <0.004, Figure 5A). The effect of estrogens on reducing lesion volume appeared to increase with increasing dose. Significantly different estimates were found within estrogen dose groups for permanent (χ2 = 99.35, df = 38, P ≤ 0.001) and transient ischemia (χ2 = 112.44, df = 41, P < 0.001).
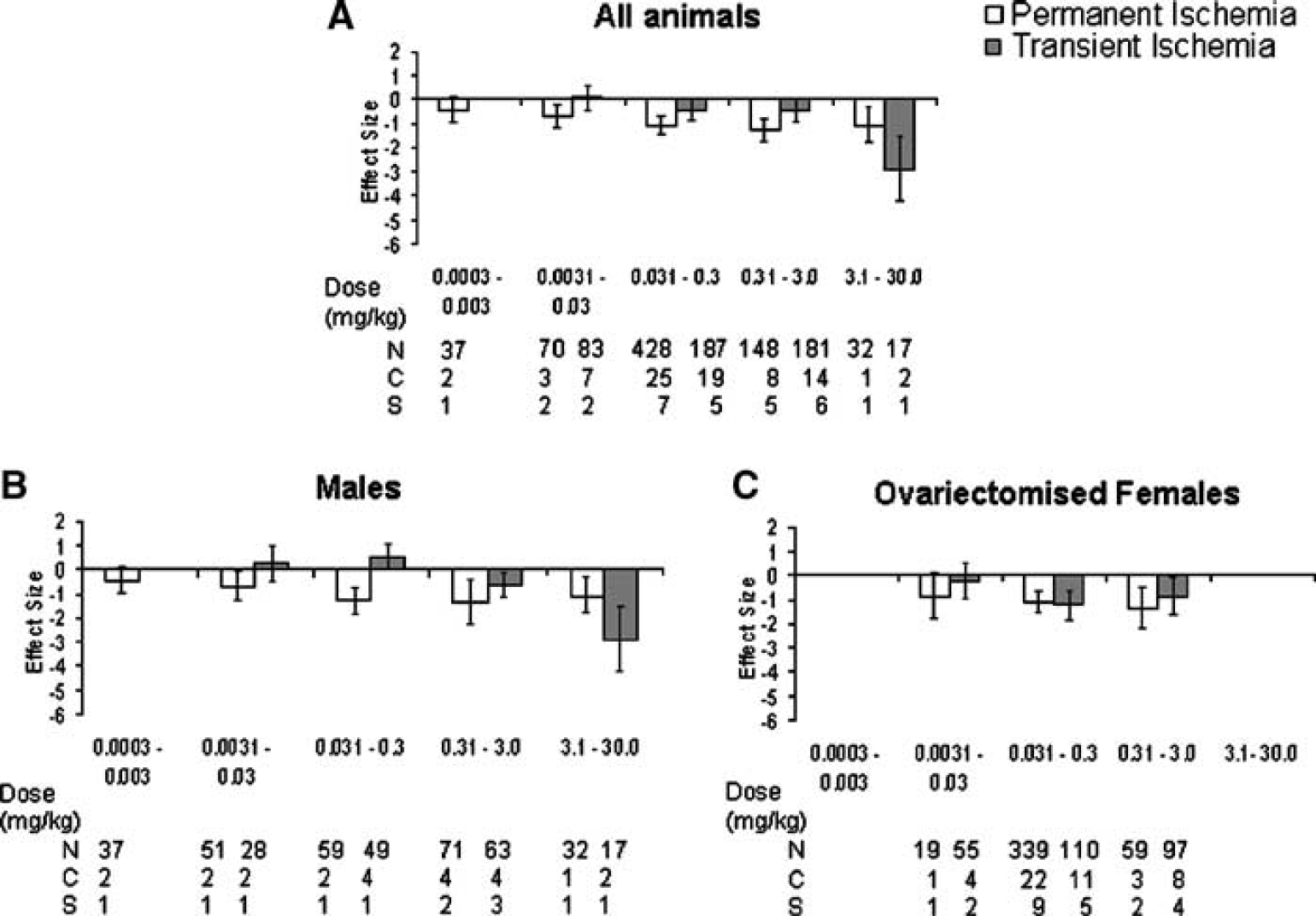
Standardized mean difference and 95% CI for total lesion volume after permanent and transient ischemia in all animals
In an attempt to identify any gender differences in response to different doses of estrogens, the data were analyzed for males (Figure 5B) and ovariectomized females (Figure 5C). In males, the effect of estrogens on reducing lesion volume appeared to increase with increasing dose (Figure 5B); however, these comparisons were made with data from only a few available studies. However, in ovariectomized females no studies have reported the effect of administering estrogens within the dose range (3.1–30.0 mg/kg) that gave the best result in males.
Timing of Estrogen Administration in Relation to Onset of Ischemia
Both pre- and postischemic administration of estrogens were effective at reducing lesion volume in all animals after permanent (−1.05, −1.31 to −0.78, P < 0.001) and transient ischemia (−0.46, −0.78 to −0.14, P = 0.004, Figure 6A). However, no studies examined the effects of administering estrogens later than 6 h after onset of ischemia. Significantly different estimates were found within the timing of estrogen groups for permanent (χ2 = 99.35, df = 38, P ≤ 0.001) and transient ischemia (χ2 = 112.2, df = 40, P ≤ 0.001).
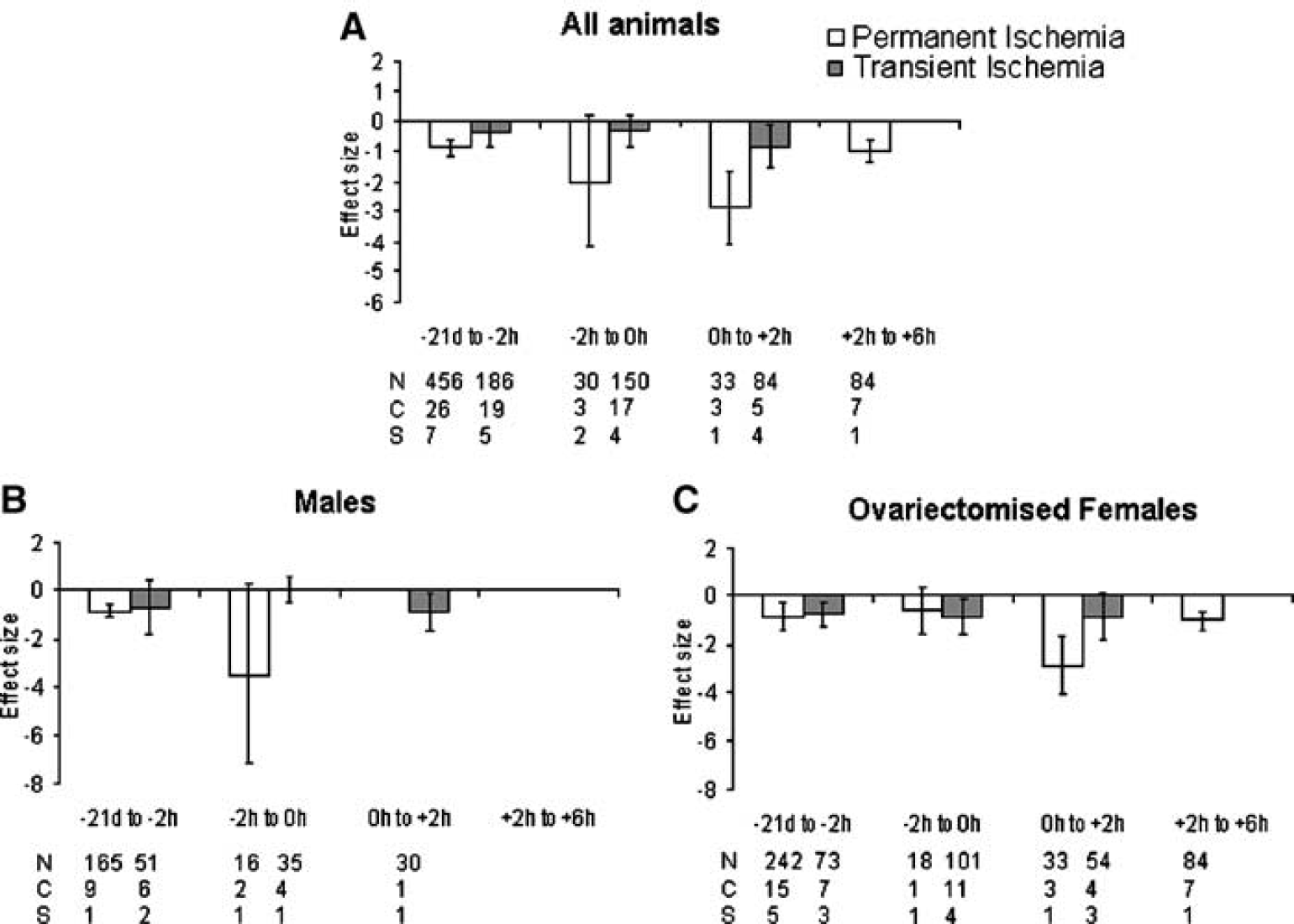
Standardized mean difference and 95% CI for total lesion volume after permanent and transient ischemia Data are grouped according to time of first administration of estrogens. Both pre- and postischemic administration of estrogens were effective at reducing lesion volume although no studies report administering estrogens later than 6 h after onset of ischemia. N, number of animals; C, number of comparisons; S, number of published studies.
For permanent ischemia, 17 β-estradiol only significantly reduced lesion volume in males when administered -21 days to -2 h before surgery (Figure 6B). Due to few studies investigating the neuroprotective effects of estrogens in males after transient ischemia, it is not possible to identify an ideal time of administration.
In ovariectomized females, estrogens had the greatest effect when administered immediately after (i.e. 0 h to +2 h) permanent ischemia (P < 0.001) or immediately before (i.e. −2 h to 0 h) transient ischemia (P = 0.03, Figure 6C). However, the majority of studies administered estrogens between 21 days and 2 h before ischemia, where it was effective at reducing lesion volume after permanent and transient ischemia.
Discussion
This systematic review has found that overall administration of estrogens reduces the lesion volume present after either permanent or transient ischemia. The term ‘estrogens’ encompasses three naturally occurring estrogens that is, estradiol, estriol, and estrone. Only one of the studies administered estrone, whereas all of the others administered estradiol. When studies were grouped according to reported quality score, the effectiveness of estrogen treatment after transient ischemia decreased with increased quality score. To identify any limitations of the effectiveness of estrogens, data were grouped according to hormonal status and age, dose and timing of estrogen administration in relation to onset of ischemia. When studies were grouped according to hormonal status and age, treatment with estrogens was only effective in ovariectomized females and young adult males.
Importantly, studies of estrogens in aged animals and administration later than 4 h after ischemia were lacking. Additionally, adequate dose-response relationships of estrogens have not been fully investigated in all experimental groups.
Systematic review and metaanalyses are fundamental tools in the interpretation of the effectiveness of a particular treatment across a large number of studies. However, they are used extensively for reviewing clinical trials and the application of systematic reviews to data obtained from animal experiments is rare (Sandercock and Roberts, 2002). Systematic reviews allow for a more objective appraisal of the research evidence than the traditional narrative reviews that are commonly associated with animal research. It would appear, from the papers analyzed in this review, that the methodological quality of a large number of animal studies is poor (in comparison to clinical trials), in particular with respect to randomization and blinding. Systematic reviews allow an insight into the cause of any bias present in animal experiments. The long-term aim of animal studies examining candidate neuroprotective factors is to inform clinical trials. Some would argue the need for a much more systematic review of animal data before proceeding to clinical trials (Pound et al, 2004) to reveal not only that a drug can have neuroprotective activity under certain conditions but also to give an insight into the limits of the efficacy of the drug (e.g. time to treatment), which are likely to effect its clinical usefulness.
However, systematic reviews do have various limitations. Firstly, analyses can only include available data, usually only available in published studies. Negative or neutral studies are less likely to be published; so the results meta-analysis may overstate effect size. In fact, Egger's asymmetry test did identify that publication bias was present, which resulted from the lack of reporting negative studies. Consequently, the benefits of estrogens on infarct volume might have been overestimated. Additionally, nonpublication will limit available information on the effect of treatment within certain protocol aspects such as dose or time of administration. The technique of extracting multiple pieces of information from single publications has the potential to introduce bias into the review because the results have been generated from the same laboratories and investigators.
This review focuses only on the effect of estrogens on lesion volume after stroke, due to insufficient data regarding other outcomes such as behavior; only one study examined the functional benefits of 17 β-estradiol administration in both males and females up to 14 days after experimental ischemia (Li et al, 2004). However, lesion volume is of limited value when interpreting whether a treatment is beneficial. Functional outcome, in combination with histopathological outcome, is as important in terms of assessing benefit (STAIR, 1999). Infarct size may (Rogers et al, 1997) or may not (Hattori et al, 2000; Reglodi et al, 2003; Wahl et al, 1992) correlate with neurological impairment. The STAIR criteria (STAIR, 1999) emphasize the need to determine the functional effects of interventions.
The purpose of this review is to show whether estrogens are effective at reducing lesion volume after stroke. It also aims to give an insight into the limitations of the effectiveness of estrogens. Overall, it was possible to conclude that estrogens were effective at reducing lesion volume after permanent or transient ischemia. However, the effect of estrogens after transient ischemia decreased with increasing quality score, which we assessed after previously published protocols (Horn et al, 2001; Macleod et al, 2005; Willmot et al, 2005a, b). However, in respect of study quality, we could only judge the studies as reported; a low-quality score could reflect either that the authors did not undertake that procedure, for example, randomize animals to treatment, or that they did not report it. Authors, journal reviewers and editors need to be more stringent in reporting key methodological details. A particular area of concern is that the majority of studies did not report randomization and/or masked assessment of outcome. Lack of randomisation and masked assessment of outcome are key sources of bias and will overemphasize treatment efficacy. Additionally, while most studies reported physiological parameters, the majority only assessed body temperature. Although body temperature is a useful indicator, additional physiological parameters also give invaluable information about the physiological effects of a particular treatment, which could be crucial when considering the design of clinical trials.
To assess the limitations of the effectiveness of estrogens we evaluated the effects of hormonal status and age. While estrogens appeared to be effective in young adult males and ovariectomized females, they might have been hazardous in intact females. However, very few studies have been performed using young adult males with the majority being conducted using ovariectomized females. However, assessments in ovariectomized females are not an ideal model of postmenopausal women. In experimental studies young female animals are subjected to a sudden surgical removal of estrogen supply, whereas humans who enter the menopause, at a mean age of 51 years in the USA (Creasman et al, 2003), experience a slow decline of circulating estrogen levels over months or even years. Importantly, there is a substantial lack of data in the literature reporting the effects of administering estrogens in aged animals; yet, the incidence of stroke in humans is strongly age-dependent (Wolf et al, 1992). Although studies on aged animals are not usually undertaken because of expense, these represent but a fraction of the costs of failed clinical trials.
The neuroprotective effects of estrogens are dose-dependent, with studies administering estrogens at doses considered to be either physiological (Dubal et al, 1998) or pharmacological (Simpkins et al, 1997). In males and ovariectomized females, the effect of estrogens on reducing lesion volume increased with increasing dose. However, the higher doses administered in males have not been applied to ovariectomized females. Thus, there is a need to further explore the dose–response relationship of estrogens and neuroprotection. Additionally, the long-term benefits of administering estrogens are not fully understood. In fact, where histological outcome was assessed at 7 days after ischemia, physiological levels of 17 β-estradiol had a detrimental effect on cell death in the vulnerable CA1 region of the hippocampus (Harukuni et al, 2001). Importantly, no studies reported administering estrogens at later than 4 h after ischemia. As the majority of those at the greatest risk of suffering a stroke are not receiving HRT (i.e. men and postmenopausal women), such a time window of application, that is, later than 4 h after ischemia, would be clinically relevant. Thus, this review emphasizes the need for bidirectional translational research between experimental and clinical studies.
Although it is well recognized that premenopausal women have an endogenous protection against vascular events, the importance of other steroid hormones must also be considered. For example, progesterone has been shown to reduce lesion volume after both permanent (Gibson et al, 2005) and transient experimental ischemia (Gibson and Murphy, 2004; Murphy et al, 2002), and also to improve functional outcome (Gibson and Murphy, 2004). Although experimental studies have shown that progesterone is neuroprotective when administered before or after cerebral ischemia in males and ovariectomized females, no clinical studies to date have investigated the effects of progesterone on outcome after stroke.
There is a need to determine precisely the cellular targets and molecular events triggered by the administration of estrogens. Animal studies can be fundamental in determining the optimal estrogens treatment in terms of timing and dosing among different populations. However, this review has highlighted fundamental areas where experimental evidence showing a protective effect of estrogens is lacking. It is these areas that should focus research to determine the most relevant design of further clinical studies.
Footnotes
Acknowledgements
CG is funded by a Wellcome Trust Value in People Award. Support from the NIH (NS 29226) to SM is gratefully acknowledged. PMWB is Stroke Association Professor of Stroke Medicine.
