Abstract
Isoflurane preconditioning (IsoPC) neuroprotection in experimental stroke is male-specific. We determined whether estradiol alters ischemic outcomes in IsoPC brain and examined the role of estrogen receptors (ERs). Seven to 10 days before preconditioning, ovariectomized (OVX) mice were implanted with estradiol, vehicle, or ER subtype agonists. OVX ± estradiol, OVX ± vehicle, OVX ± ER agonists, and ER subtype wild-type (WT) and knockout (KO) mice were preconditioned for 4 h with sham anesthetic preconditioning (sham PC) or 1% IsoPC and recovered for 24 h. Mice then underwent 2 h of middle cerebral artery occlusion followed by 22 h of reperfusion. Infarct volumes were determined by 2,3,5-triphenyltetrazolium chloride staining, with comparisons between IsoPC and corresponding sham PC for each treatment group. Decreased infarct injury was seen in IsoPC OVX ± vehicle, whereas estradiol in IsoPC OVX mice enhanced ischemic damage. In ER studies, increased infarct volumes were seen in IsoPC ERWT mice regardless of ER subtype. IsoPC in ERαKO mice had no effect on infarction volume but reduced only cortical ischemic damage in ERβKO mice. In OVX + ERβ agonist, IsoPC had no effect on infarction volume. In OVX + ER/α agonist, IsoPC increased cortical infarct volume. Estradiol depresses the brain's protective response to IsoPC and may exacerbate cortical ischemic injury mainly through an ERβ-dependent mechanism.
Introduction
Perioperative stroke is a potential complication for cardiovascular surgical procedures such as carotid endarterectomy (Kitano et al, 2007a; Wang et al, 2008). However, women may have a greater perioperative stroke risk than men for these procedures (Kitano et al, 2007a; Wang et al, 2008). Recent clinical studies have raised the issue that there are unanticipated and paradoxical effects of estrogen and hormone replacement therapy relative to stroke risk as it is currently administered to women (Grodstein et al, 2000; Macrae and Carswell, 2006; Rossouw et al, 2002; Viscoli et al, 2003; The Women's Health Initiative Steering Committee, 2004). Whether hormone replacement therapy alters perioperative stroke risk is currently unknown. Only one study has suggested that women on hormone replacement therapy have a higher risk of perioperative stroke when undergoing carotid endarterectomy than women who are not on hormone replacement therapy (Lane et al, 2003).
Anesthetic selection may be an important consideration for procedures at risk for perioperative stroke as agents such as isoflurane are known to affect experimental ischemic outcomes (Kitano et al, 2007a; Wang et al, 2008). Anesthetic exposure or preconditioning may consequently prevent or delay neurologic complications from perioperative stroke. Experimental studies examining anesthetic preconditioning and its neuroprotective mechanisms in ischemic brain have used predominantly male animals, leaving open the question of the effect of gender and of sex steroids such as estrogen. We have previously shown that isoflurane preconditioning (IsoPC) is neuroprotective only in male ischemic mouse brain and can exacerbate ischemic injury in young female ischemic mouse brain (Kitano et al, 2007b).
Estrogen is a pleiotropic hormone that exhibits acute and chronic effects and acts by genomic and nongenomic mechanisms. Classical estrogen signaling occurs through binding of estrogen receptors (ERs), followed by transcriptional regulation of target genes. Two receptor subtypes, ER-alpha (ERα) and ER-beta (ERβ), have been identified. Although estrogen has largely reduced ischemic brain injury in rodents (Macrae and Carswell, 2006; Murphy et al, 2004), several clinical and experimental studies would suggest that estrogen may not be universally neuroprotective in experimental cerebral ischemia (Macrae and Carswell, 2006) and may not be beneficial for ischemic preconditioning in experimental myocardial ischemia models (Song et al, 2003; Wang et al, 2006). Little work has been done evaluating the effects of estrogen and the role of ERs in anesthetic preconditioning in the ischemic brain.
Using a mouse IsoPC model, we determined whether the lack of IsoPC neuroprotection in ischemic female brain is because of estradiol, the primary estrogen observed in rodents and in women. We also used ER subtype-specific knockout mice and selective ER subtype agonists to determine whether estradiol acts by an ER-dependent mechan-ism in the IsoPC ischemic female brain.
Materials and methods
Animal Model
The Oregon Health and Science University Animal Care and Use Committee approved all experiments. Young (9 to 20 weeks of age, 20 to 25 g) female (n = 79) C57BL/6 mice (Charles River Laboratories, Wilmington, MA, USA) were used for infarct volume determinations. Infarct volumes were also determined in ERα wild-type (ERαWT; n = 19), ERα knockout (ERαKO; n = 21), ERβ wild-type (ERβWT; n = 18), and ERβ knockout (ERβKO; n = 23) female mice. Female C57BL/6 (n = 16), ERβWT (n = 8), and ERβKO (n = 8) mice were used for physiologic studies evaluating mean arterial blood pressure, blood gases (pH, PaO2, and PaCO2), and blood glucose values during ischemia and reperfusion.
Hormone and Drug Treatments
Estradiol implants were administered subcutaneously under isoflurane anesthesia (induction 4%; maintenance 1.5%) to female mice (n = 13) concurrently undergoing ovariectomy (OVX) 1 week before preconditioning and transient focal cerebral ischemia (Figure 1). Estradiol was administered via a subcutaneous silastic implant containing 35 μL of 180 μg/mL estradiol in sesame oil (6.3 μg total dose). This estradiol dose has been used previously by our group and yields physiologic levels of plasma estradiol comparable with cycling female mice (Ardelt et al, 2005).
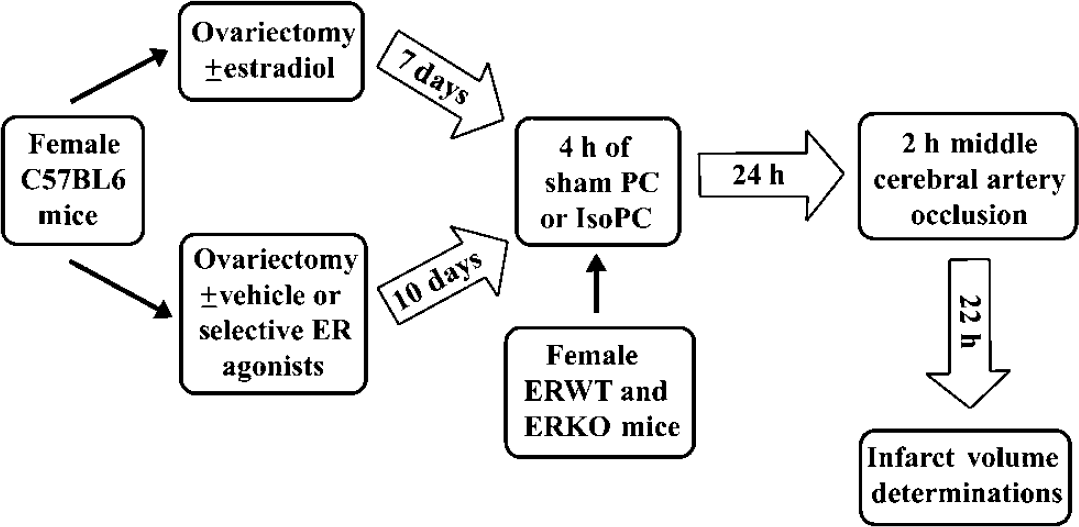
Schematic overview of the time line and experimental design for ovariectomy, hormone or drug administrations, preconditioning, and ischemia. ER, estrogen receptor; ERKO, estrogen receptor knockout; ERWT, estrogen receptor wild-type; IsoPC, isoflurane preconditioning; Sham PC, sham preconditioning.
Propyl pyrazole triol (PPT; 2 mg/kg/day), diarylpropionitrile (DPN; 8 mg/kg/day), and vehicle (50% DMSO (dimethyl sulfoxide) in 0.9% saline) were dispensed via osmotic pumps (Model 1002, Alzet Osmotic Pumps®; Durect Corporation, Cupertino, CA, USA), which were implanted in female mice (n = 69) concurrently undergoing OVX 10 days before preconditioning and transient focal cerebral ischemia (Figure 1). Propyl pyrazole triol is an ERα agonist with an approximately 400-fold binding selectivity over ERβ and can cause ER-mediated transcription (Meyers et al, 2001; Stauffer et al, 2000). Systemically administered PPT has also been shown to cross the blood—brain barrier and exert central effects (Harris et al, 2002). Diarylpropionitrile is an ERβ agonist with 70-fold higher relative binding affinity over ERα and 170-fold higher relative estrogen potency in transcription assays with ERβ than with ERα (Meyers et al, 2001). Diarylpropionitrile administered systemically was also reported to have effects centrally (Le Saux and Di Paolo, 2005). The PPT and DPN doses selected for the proposed studies are based on effective doses used in ischemic rodent brain studies (Carswell et al, 2004; Farr et al, 2007) as well as in other rodent studies (Fasor et al, 2003; Harris et al, 2002).
Isoflurane Preconditioning Model
For preconditioning experiments, mice were placed for 4 h in an air-tight, temperature-controlled chamber flushed with 1.0% isoflurane (IsoPC) or no isoflurane (sham preconditioning, sham PC) in oxygen-enriched air as previously described (Kitano et al, 2007b). We have previously shown that physiologic systemic variables (glucose, pH, and PaCO2) evaluated after 1 and 4 h of IsoPC showed no major perturbations imposed by IsoPC other than mild hypotension and that PaO2 values were consistent with animals exposed to oxygen-enriched air (Kitano et al, 2007b). Twenty-four hours after preconditioning, mice (n = 192) subsequently underwent transient focal cerebral ischemia (Figure 1).
Transient Focal Cerebral Ischemia
Preconditioned (sham PC and IsoPC) female mice were subjected to 2 h of reversible middle cerebral artery occlusion by the intraluminal filament technique as previously described (Ardelt et al, 2005). Mice were anesthetized with isoflurane (induction 4%; maintenance 1.5%) during surgery until unilateral middle cerebral artery occlusion was achieved and then allowed to awaken during ischemia. Toward the end of ischemia, mice were briefly reanesthetized with isoflurane and reperfusion was initiated by intraluminal filament with-drawal. Rectal temperatures were monitored throughout middle cerebral artery occlusion surgery and during manipulations to induce reperfusion. Anesthetized mice were warmed as needed using a heating lamp. Cortical blood flow was monitored throughout middle cerebral artery occlusion surgery, initial induction of ischemia, end of ischemia, and the initiation of reperfusion by laser Doppler flowmetry (Moor Instruments Ltd, model MBF3D, Oxford, UK) to confirm occlusion and reperfusion. Neurologic deficit scores were determined during ischemia and at 22 h of reperfusion to confirm ischemia and to assess equivalence of ischemic injury, respectively, rather than to determine specific functional outcomes according to the following graded scoring system: 0, no deficit; 1, forelimb weakness and torso turning to the ipsilateral side when held by tail; 2, circling to the affected side; 3, unable to bear weight on the affected side; and 4, no spontaneous locomotor activity or barrel rolling. Any animal without a deficit was excluded.
After 22 h of reperfusion, animals were euthanized, brains removed for infarct volume determinations, and blood obtained for hormone analysis (Figure 1). Tissue infarction was identified by the lack of 2,3,5-triphenyl-tetrazolium chloride (Sigma, St Louis, MO, USA) staining in 2-mm-thick coronal sections (total five slices) (Bederson et al, 1986). Both sides of each stained coronal slice were photographed by a digital camera and then evaluated by digital image analysis (SigmaScan Pro, Jandel, San Rafael, CA, USA). Infarcted area was integrated across sections, and volume expressed as percentage of contralateral structure (cortex and caudate—putamen). Plasma estradiol levels were measured in duplicate by radioimmunoassay (Diagnostic Products Corp., Los Angeles, CA, USA) in all treatment groups. The lower limit of detection for the estradiol radioimmunoassay was 10 pg/mL. Samples with estradiol levels < 10 pg/mL were not used in calculating mean estradiol levels per treatment group.
Femoral arterial catheters were placed in separate groups of sham PC and IsoPC ERβWT, ERβKO, vehicle (50% DMSO in 0.9% saline), and DPN mice (n = 4 per group) to determine mean arterial blood pressure, blood gases (pH, PaO2, and PaCO2), and blood glucose values at 1 h of ischemia and at 10 mins of reperfusion. Laser Doppler flowmetry and rectal temperature were also monitored. Animals were anesthetized with isoflurane throughout ischemia and reperfusion. Body temperature was controlled as needed using a heating lamp. Mice were euthanized after the final blood sample was taken 10 mins into the reperfusion period.
Estrogen Receptor α- and Estrogen Receptor β-Deficient Mice
Generation of ERα- and ERβ-targeted mice is as previously described (Krege et al, 1998; Lubahn et al, 1993). In these animals, the ERα or ERβ gene has been disrupted and is physiologically nonresponsive to estrogens by several classical bioassays of estrogenic activity. Both strains originated on a B6;129 background and have been back-crossed to C57BL/6 for at least 10 generations. ERα+/− and ERβ+/− (heterozygote) breeder pairs were obtained from Dr Kenneth S Korach (Laboratory of Reproductive and Developmental Toxicology, National Institute of Environmental Health Sciences, Research Triangle Park, NC, USA). Breeding colonies were maintained using the following breeding harems (two females and one male): ERα+/− harems, ERβ+/+ (WT) harems, and harems with ERβ+/− females and ERβ−/− (KO) males.
Estrogen receptor α mice were genotyped by PCR using the following primers in a single reaction: Neo F (5ʹ-GCT GACCGCTTCCTCGTGCTTTAC-3ʹ), ER 2382 (5ʹ-CGGTCTA CGGCCAGTCGGGCACC-3ʹ) and mER intron 2 Rev (5ʹ-CA GGCCTTACACAGCGGCCACCC-3ʹ) (Krege et al, 1998; Lubahn et al, 1993). The expected sizes of the PCR products are 281 bp (base pairs) for ERα+/+ (WT), 760 bp for ERα−/− (KO), and the presence of both PCR products for ERα+/−. Estrogen receptor β animals were genotyped by PCR using the following primers: the intron 2 primer (5ʹ-GTGATGAGCTGAGGTGGTGCTT-3ʹ), the Neo primer (5ʹ-GCAGCCTCTGTTCCACATACAC-3ʹ), and a third primer from exon 3 (5ʹ-CATCCTTCACAGGACCAGACAC-3ʹ) (Krege et al, 1998). A 1,435-bp band (intron 2 and exon 3 primers) is amplified for homozygous WT (ERβ+/+) mice, a 1,479-bp band (intron 2 and Neo primers) for homozygous mutant (ERβ−/−) mice, and both bands for heterozygous (ERβ+/-) mice (Krege et al, 1998). Every set of PCR reactions contained a negative control (no DNA) and a positive control (heterozygous DNA). Female ERα+/+, ERα−/−, ERβ+/+, and ERβ−/− mice underwent the same preconditioning (sham PC and IsoPC) and ischemia protocols as described above for young female C57BL/6 mice (Figure 1).
Statistics
Values were expressed as mean ± s.e.m. Differences in infarction volumes and plasma hormone levels in each IsoPC group as compared with its corresponding sham PC group were determined with Student's t–test. Differences in hormone levels between OVX and OVX + estradiol; between female ER subtype-specific WT and KO mice; and between OVX treated with vehicle, PPT, or DPN regardless of preconditioning status were determined by one-way analysis of variance with the post hoc Newman—Keuls test. Laser Doppler flowmetry (% baseline), neurologic deficit scores, rectal temperatures, mean arterial blood pressure, blood gases (pH, PaO2, and PaCO2), and blood glucose were subjected to two-way analysis of variance with the post hoc Newman—Keuls test. Statistical significance was P 0.05. All statistical analyses were performed using SigmaStat Statistical Software (version 2.0, SPSS Inc., Chicago, IL, USA).
Results
Estradiol Alters Infarct Volumes in Isoflurane Preconditioned Brain
Laser Doppler flowmetry (percentage of preischemic baseline) at the induction of ischemia was comparable between estradiol-treated and untreated OVX regardless of the preconditioning group (data not shown). At the initiation of reperfusion, all experimental groups reperfused to > 55% of end-ischemic baseline laser Doppler flowmetry measurements (data not shown). Neurologic deficit scores were determined to confirm ischemia and to assess equivalence of injury rather than specific functional outcomes. Neurologic deficit scores (data not shown) and rectal temperatures were comparable between estradiol-treated and untreated OVX preconditioning groups at all time points evaluated. However, there were some differences in rectal temperature within each experimental group (Table 1). No mortality occurred in the estradiol-treated and untreated OVX groups, and no animals were excluded from these groups because of failure to reperfuse or lack of neurologic deficits.
Rectal temperatures during experimental focal cerebral ischemia and plasma estradiol levels in preconditioned OVX C57BL/6 mice treated with or without estradiol (6.3 μg total dose) for 7 days
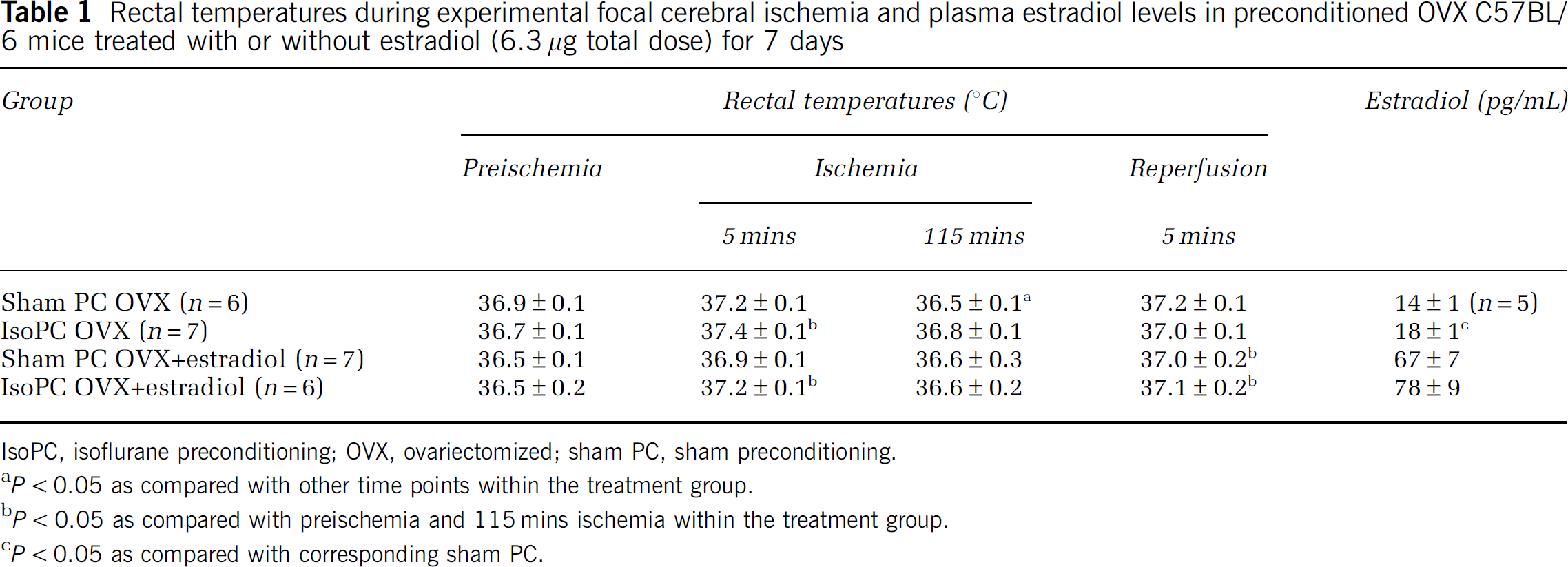
IsoPC, isoflurane preconditioning; OVX, ovariectomized; sham PC, sham preconditioning.
P < 0.05 as compared with other time points within the treatment group.
P < 0.05 as compared with preischemia and 115 mins ischemia within the treatment group.
P < 0.05 as compared with corresponding sham PC.
As expected, plasma estradiol levels were higher in estradiol-treated animals (OVX + estradiol, 72 ± 6 pg/mL, n = 13) compared with OVX (17 ± 1 pg/mL, n = 12) mice regardless of the preconditioning group. There were no differences in estradiol levels between IsoPC and the appropriate corresponding sham PC for OVX + estradiol (Table 1). Isoflurane preconditioning OVX had higher estradiol levels than sham PC OVX (Table 1). There was one sample from sham PC OVX compared with no samples from other treatment groups that was below < 10 pg/mL.
Significant decreases in infarct injury were seen in IsoPC OVX mice (cortex, 23.2% ± 4.6%; caudate—putamen, 73.1% ± 3.4%) as compared with sham PC OVX mice (cortex, 46.5% ± 3.2%; caudate—putamen, 101.9% ± 4.2%) (Figure 2). Estradiol treatment in IsoPC OVX mice led to enhanced cortical and caudate—putamen ischemic damage (cortex, 55.1% ± 5.4%; caudate—putamen, 99.3% ± 3.5%) as compared with sham PC OVX ± E2 mice (cortex, 19.3% ± 2.7%; caudate—putamen, 71.6% ± 2.6%) (Figure 2).
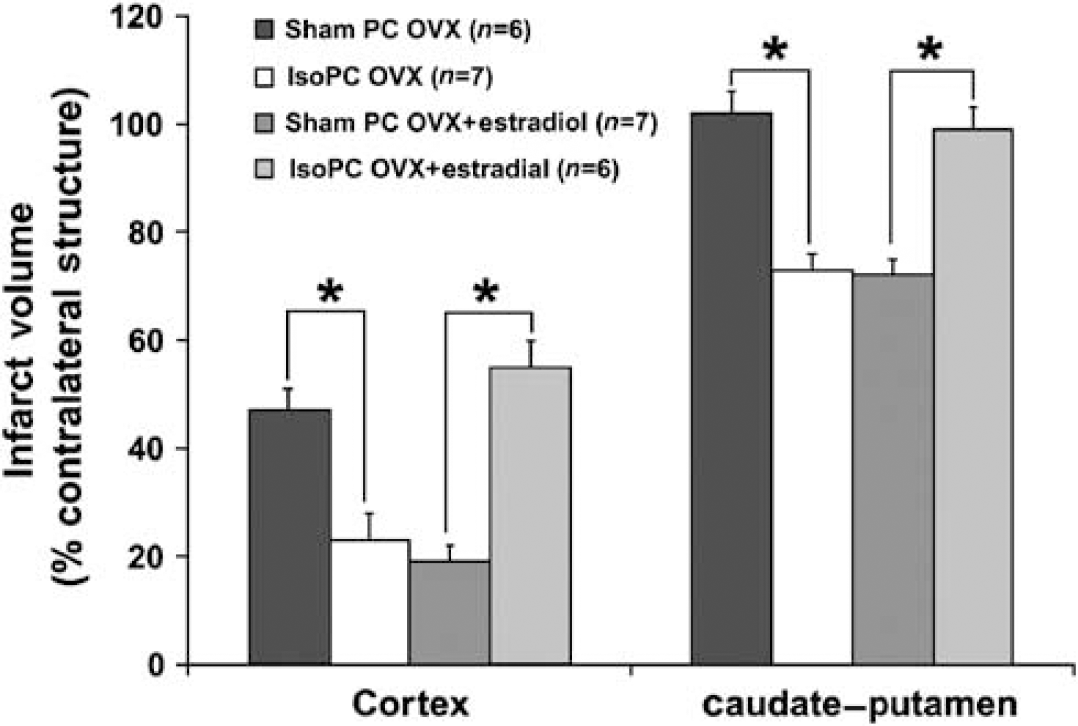
Cortical and caudate—putamen infarction volumes (percentage of contralateral structure) determined by 2,3,5-triphenyltetrazolium chloride staining in C57BL/6 mice preconditioned for 4 h with 0% (sham PC) or 1% isoflurane (IsoPC). Preconditioning occurred 24 h before 2 h of MCAO followed by 22 h of reperfusion. Subcutaneous estradiol silastic implants (6.3 μg total dose) were placed in selected groups of female mice concurrently undergoing ovariectomy (OVX) 1 week before preconditioning. Effects of OVX ± estradiol in females on the response to IsoPC in the ischemic mouse brain are shown. Values are mean ± s.e.m. *P < 0.05.
Estradiol's Effects in Isoflurane Preconditioning Ischemic Brain is Estrogen Receptor-Dependent
Laser Doppler flowmetry (percentage of preischemic baseline) at the induction of ischemia was comparable between ERα- and ERβWT and -deficient mice regardless of the preconditioning group (data not shown). At the initiation of reperfusion, all experimental groups reperfused to > 55% of end-ischemic baseline laser Doppler flowmetry measurements (data not shown). Neurologic deficit scores were determined to confirm ischemia and to assess equivalence of injury rather than specific functional outcomes. Neurologic deficit scores during and after ischemia were equivalent between ERα- and ERβWT and -deficient mice preconditioning groups (data not shown). Rectal temperatures recorded at 5 mins of ischemia and at 5 mins of reperfusion were higher in ERαWT and ERαKO groups as compared with ERβWT and ERβKO mice (Table 2). There were differences in rectal temperature within ERαWT and ERαKO groups as well as within the IsoPC ERβWT (Table 1) group. No mortality occurred in any of the treatment groups, and no animals were excluded from these groups because of failure to reperfuse or lack of neurologic deficits.
Rectal temperatures during experimental focal cerebral ischemia and plasma estradiol levels in ERαWT and ERαKO mice and ERβWT and ERβKO mice
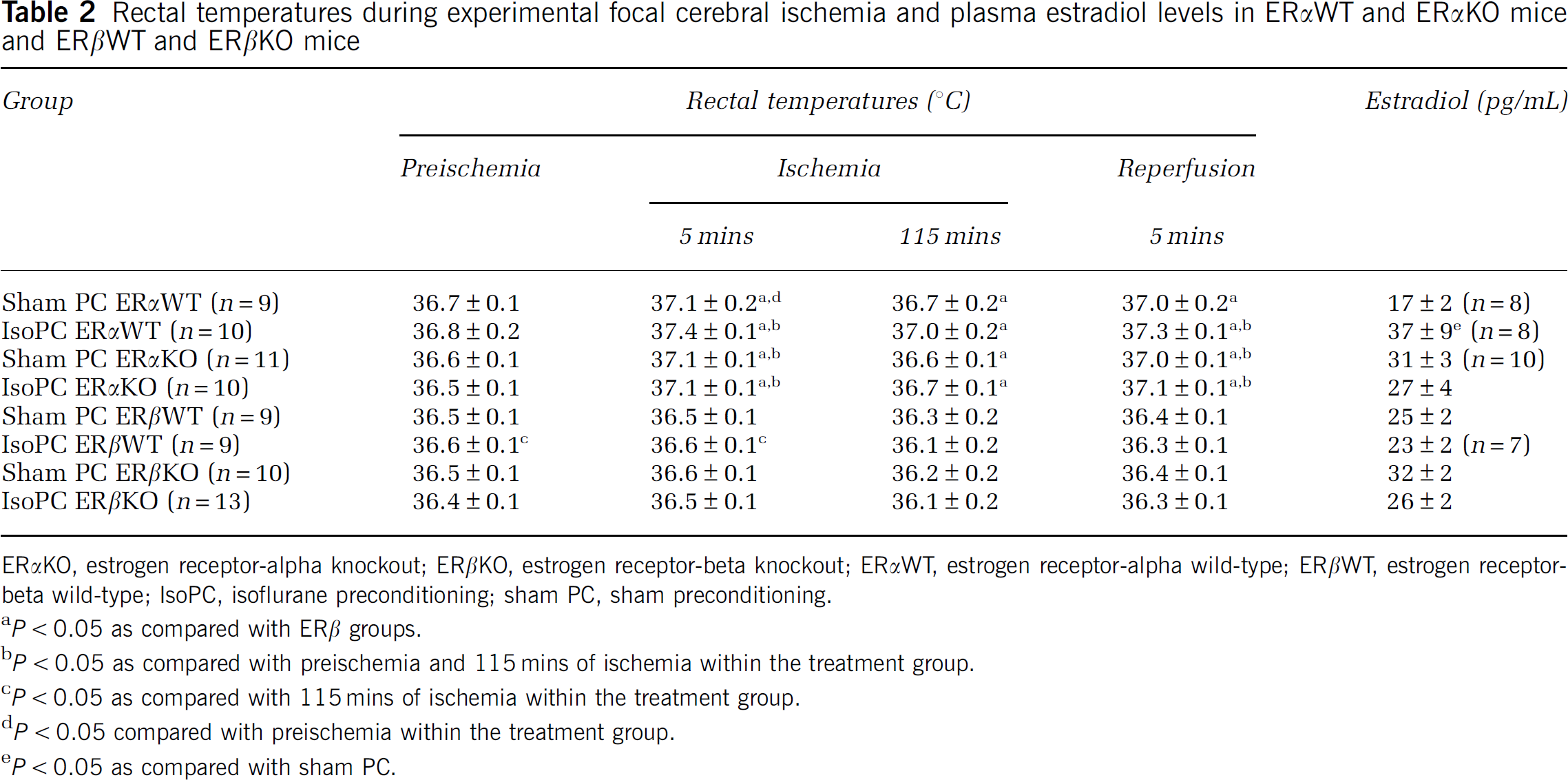
ERαKO, estrogen receptor-alpha knockout; ERβKO, estrogen receptor-beta knockout; ERαWT, estrogen receptor-alpha wild-type; ERβWT, estrogen receptor-beta wild-type; IsoPC, isoflurane preconditioning; sham PC, sham preconditioning.
P < 0.05 as compared with ERβ groups.
P < 0.05 as compared with preischemia and 115 mins of ischemia within the treatment group.
P < 0.05 as compared with 115 mins of ischemia within the treatment group.
P < 0.05 compared with preischemia within the treatment group.
P < 0.05 as compared with sham PC.
There were no differences in estradiol levels between IsoPC and the appropriate corresponding sham PC for female ERα-deficient mice and female ERβ-WT and -deficient mice (Table 2). There was a difference in estradiol levels between sham PC and IsoPC ERαWT mice (Table 2). However, regardless of the preconditioning group, there were no differences in estradiol levels between female ERWT (ERα, 27 ± 5, n = 16; ERβ, 24 ± 1, n = 16) and ER-deficient (ERα,29 ± 2, n = 20; ERβ, 30 ± 2, n = 23) mice. There were two samples from female IsoPC ERαWT mice compared with one sample from female sham PC ERαKO mice that were below < 10 pg/mL.
In ERWT mice, increases in infarct volume were seen in IsoPC ERαWT (cortex, 52.6% ± 5.4%; caudate—putamen, 93.8% ± 5.7%) and ERβWT (cortex, 49.8% ± 2.8%; caudate—putamen, 99.1% ± 2.5%) mice as compared with sham PC ERαWT (cortex, 20.9% ± 5.1%; caudate—putamen, 73.6% ± 3.1%) and ERβWT (cortex, 36.7% ± 5.2%; caudate—putamen, 85.3% ± 4.1%) mice (Figure 3). Isoflurane preconditioning in ERαKO had no effect on infarction volume (cortex, 35.8% ± 3.3%; caudate—putamen, 91.8% ± 2.1%) as compared with sham PC ERαKO mice (cortex, 39.7% ± 4.2%; caudate—putamen, 83.0% ± 4.7%) (Figure 3A). However, in ERβKO mice, IsoPC reduced cortical (41.4% ± 3.7%) infarct volume (Figure 3B) but had no effect on caudate—putamen (90.2% ± 4.0%) infarct volume (Figure 3B) as compared with sham PC mice (cortex, 51.0% ± 1.8%; caudate—putamen, 100.0% ± 2.1%).
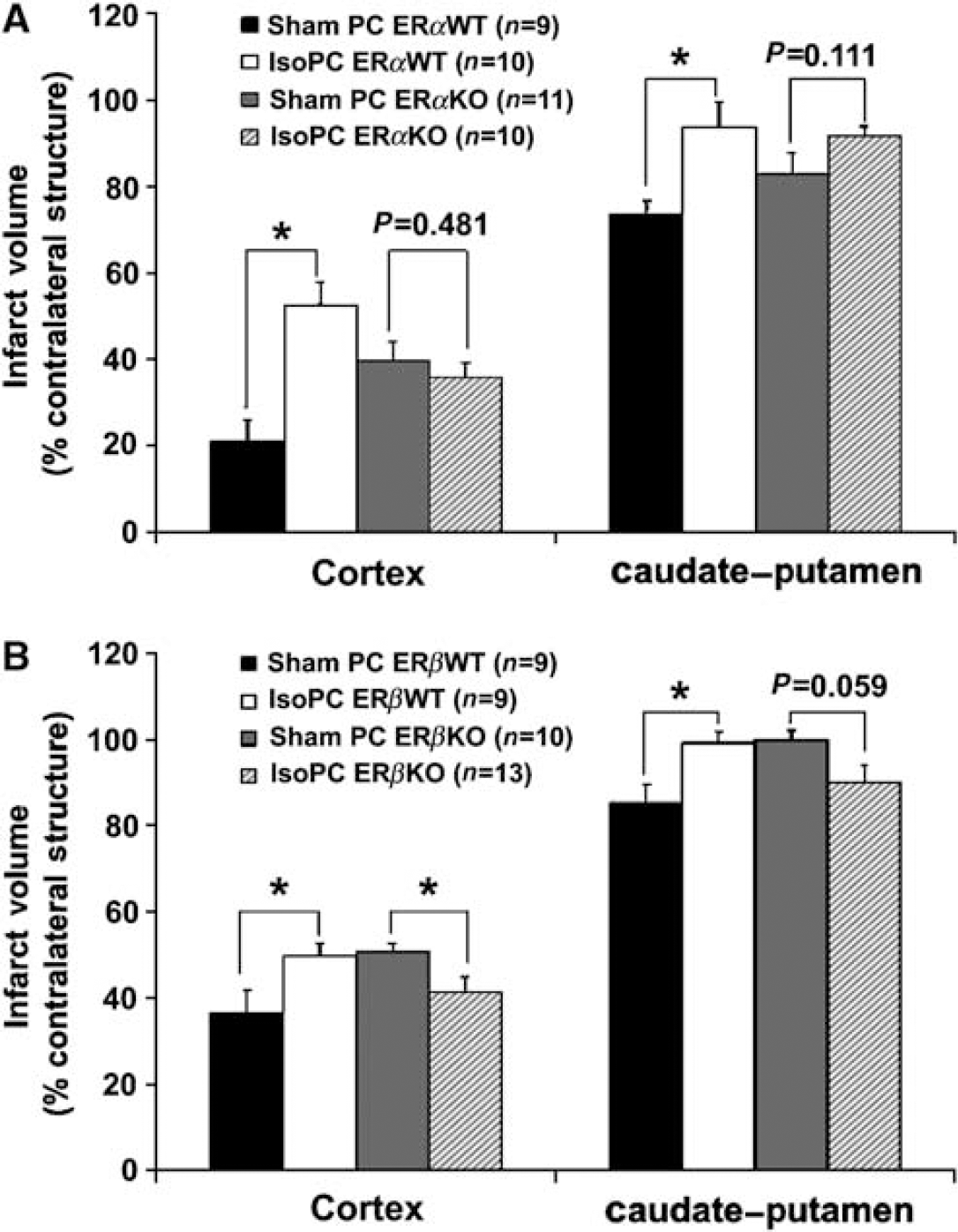
cortical and caudate—putamen infarction volumes (percentage of contralateral structure) determined by 2,3,5-triphenyltetrazolium chloride staining in estrogen receptor subtype alpha (ERα) and beta (ERβ), knockout (KO), and wild-type (WT) female mice preconditioned for 4 h with 0% (sham PC) or 1% isoflurane (IsoPC). Preconditioning occurred 24 h before 2 h of middle cerebral artery occlusion followed by 22 h of reperfusion. Effects of the presence or absence of ERα (
Laser Doppler flowmetry (percentage of preischemic baseline) at the induction of ischemia was comparable between vehicle and ER agonist-treated OVX mice regardless of the preconditioning group (data not shown). At the initiation of reperfusion, all experimental groups reperfused to > 55% of end-ischemic baseline laser Doppler flowmetry measurements (data not shown). Neurologic deficit scores were determined to confirm ischemia and to assess equivalence of injury rather than specific functional outcomes. Neurologic deficit scores (data not shown) and rectal temperatures during and after ischemia were equivalent between vehicle and ER agonist-treated OVX preconditioning groups. How-ever, rectal temperature at 115 mins ischemia was different between sham PC vehicle and sham PC PPT mice (Table 3). There were some differences in rectal temperature within sham PC vehicle and DPN groups (Table 3). No mortality occurred in any of the treatment groups, and no animals were excluded from these groups because of failure to reperfuse or lack of neurologic deficits.
Rectal temperatures during experimental focal cerebral ischemia and plasma estradiol levels in OVX mice treated with vehicle (50% DMSO in 0.9% saline), PPT (estrogen receptor α agonist, 2 mg/kg/day), or DPN (estrogen receptorβ agonist, 8 mg/kg/day) for 10 days
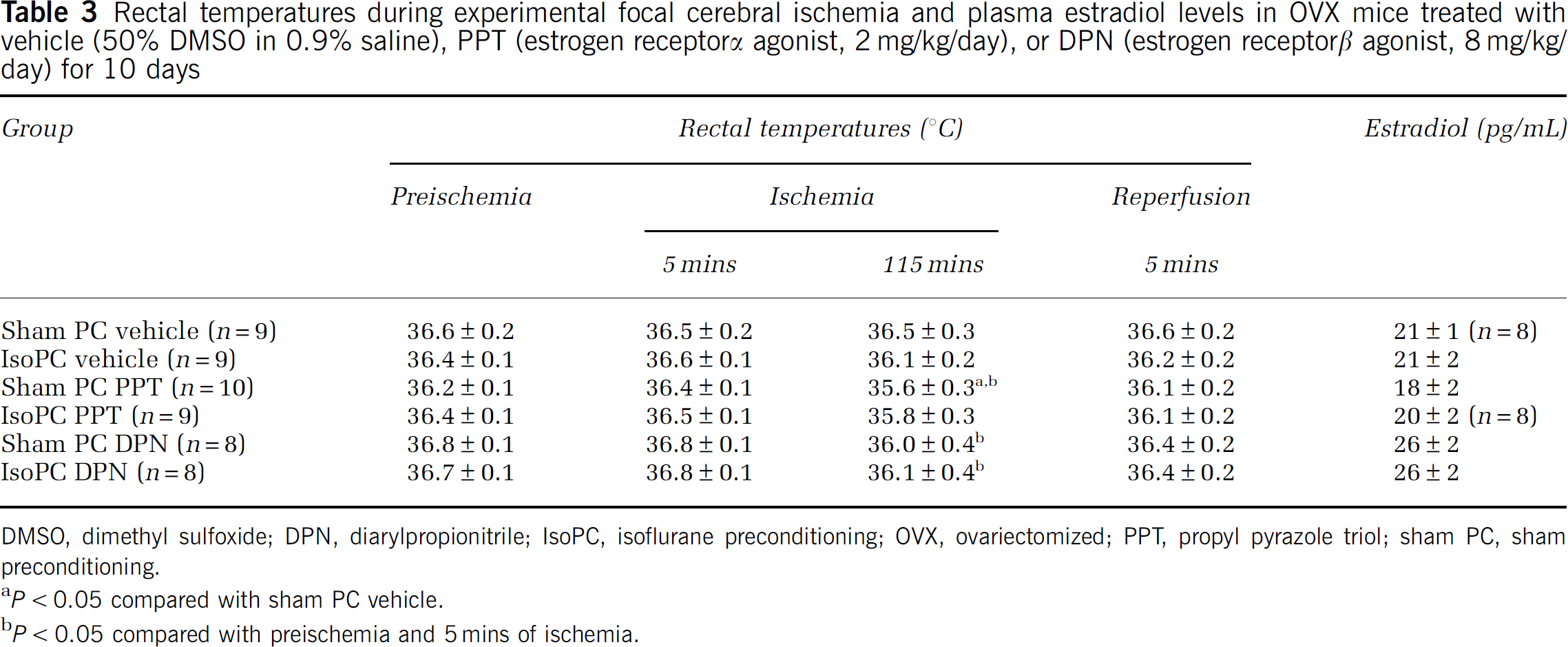
DMSO, dimethyl sulfoxide; DPN, diarylpropionitrile; IsoPC, isoflurane preconditioning; OVX, ovariectomized; PPT, propyl pyrazole triol; sham PC, sham preconditioning.
P < 0.05 compared with sham PC vehicle.
P < 0.05 compared with preischemia and 5 mins of ischemia.
There were no differences in estradiol levels between IsoPC and the appropriate corresponding sham PC for vehicle and ER agonist-treated OVX mice (Table 3). Regardless of the preconditioning group, there were also no differences in estradiol levels among vehicle (21 ± 1 pg/mL, n = 17), PPT (19 ± 1 pg/mL, n = 18), and DPN (23 ± 2 pg/mL, n = 16) mice. There were no samples below < 10 pg/mL for any of these treatment groups.
In studies evaluating the effects of ER agonists on the response to IsoPC in the ischemic brain, decreases in infarct volume were observed in IsoPC vehicle (cortex, 40.4% ± 4.8%; caudate—putamen, 93.3% ± 3.7%) as compared with sham PC vehicle (cortex, 56.3% ± 2.8%; caudate—putamen, 107.5% ± 2.7%) mice (Figure 4). In OVX mice treated with the ERα-agonist PPT, IsoPC had no effect on infarction volume (cortex, 45.9% ± 6.7%; caudate—putamen, 90.3% ± 2.9%) as compared with sham PC mice (cortex, 54.2% ± 6.1%; caudate—putamen, 95.6% ± 3.7%) (Figure 4). In OVX mice treated with the ERβ agonist DPN, IsoPC increased cortical (56.4% ± 3.0%) infarct volume (Figure 4) but had no effect on caudate—putamen (97.1% ± 4.6%) infarct volume (Figure 4) as compared with sham PC mice (cortex, 40.0% ± 7.1%; caudate—putamen, 97.2% ± 2.2%).
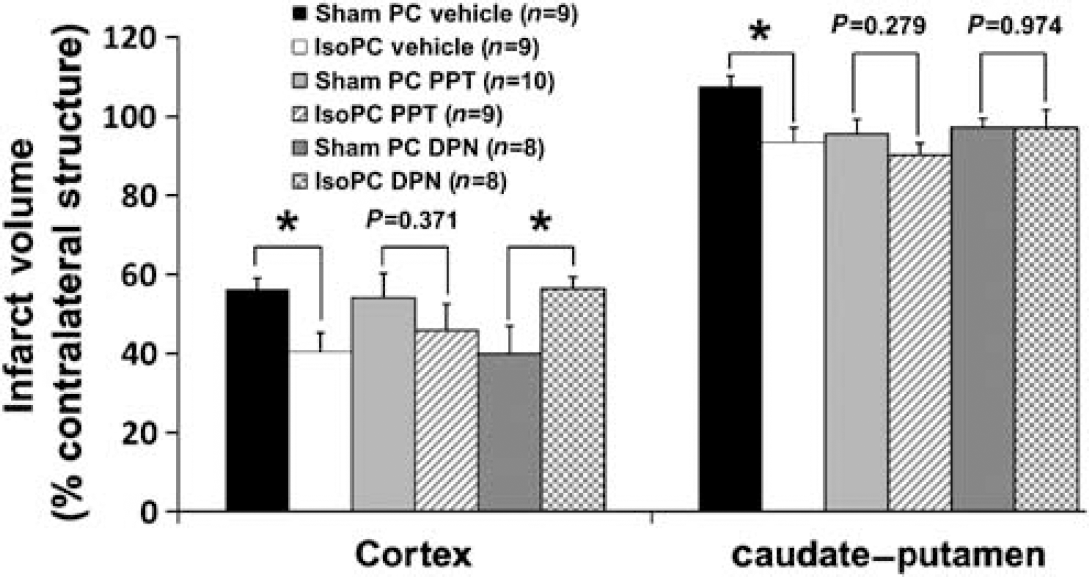
Cortical and caudate—putamen infarction volumes (percentage of contralateral structure) determined by 2,3,5-triphenyltetrazolium chloride staining in ovariectomized female C57BL/6 mice preconditioned for 4 h with 0% (sham PC) or 1% isoflurane (IsoPC). Preconditioning occurred 24 h before 2 h of middle cerebral artery occlusion followed by 22 h of reperfusion. Subcutaneous osmotic implants containing the estrogen receptor subtype-α agonist propyl pyrazole triol (PPT, 2 mg/kg/day), the estrogen receptor subtype-β agonist diarylpropionitrile (DPN, 8 mg/kg/day), or vehicle (50% DMSO in 0.9% saline) were placed in selected groups of female mice concurrently undergoing ovariectomy 10 days before preconditioning. Effects of PPT, DPN, and vehicle in the cortex and caudate—putamen on the response to IsoPC in the ischemic mouse brain are shown. Values are mean ± s.e.m. *P < 0.05.
Mean arterial blood pressure, blood gases (pH, PaO2, and PaCO2), and blood glucose were comparable among ERβWT, ERβKO, vehicle, and DPN preconditioning groups at each time point evaluated (Table 4). There were differences between ischemic and reperfusion pH values within ERβKO preconditioning groups, sham PC vehicle, and IsoPC DPN; between ischemic and reperfusion PaCO2 values within IsoPC ERβKO and sham PC vehicle; and between ischemic and reperfusion glucose values within sham PC DPN (Table 4).
Physiological parameters in Sham PC and 1% IsoPC ERβWT and ERβKO mice as well as OVX mice treated with either vehicle (50% DMSO in 0.9% saline) or DPN (ERβ agonist, 8 mg/kg/day) for 10 days at 1 h of ischemia and 10 mins of reperfusion (n = 4 per group)
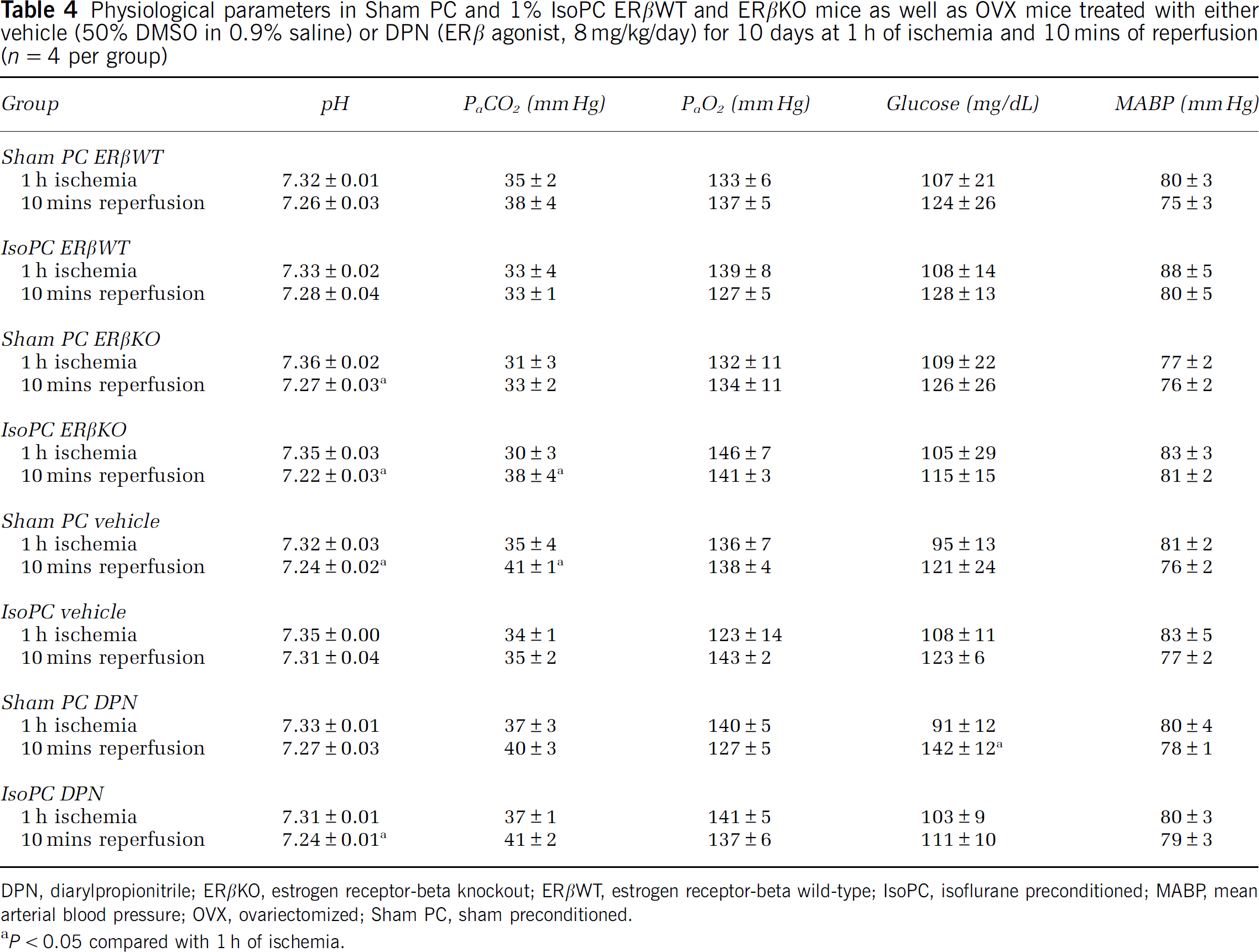
DPN, diarylpropionitrile; ERβKO, estrogen receptor-beta knockout; ERβWT, estrogen receptor-beta wild-type; IsoPC, isoflurane preconditioned; MABP, mean arterial blood pressure; OVX, ovariectomized; Sham PC, sham preconditioned.
P < 0.05 compared with 1 h of ischemia.
Discussion
This study shows two important findings. First, estradiol attenuates the neuroprotective response to IsoPC and increases cortical and caudate—putamen injury in the IsoPC ischemic brain. Second, estradiol's effects on the response to IsoPC in the ischemic cortex are mediated primarily through ERβ. These findings suggest that estradiol depresses the brain's protective response to IsoPC and exacerbates cortical ischemic injury mainly through an ERβ-dependent mechanism.
Sex differences in the pharmacokinetics and pharmacodynamics of anesthetics are well recognized (Pleym et al, 2003). However, sex-based differences in anesthetic requirements and responses could be because of a number of sex-dependent and sex-specific effects. Sex-dependent effects include body weight, height, basal metabolic rate, body fat, and muscle mass, whereas sex-specific effects are focused on receptor responses, cyclical variation, neurotransmitter differences, cytochrome enzyme changes, and sex hormone-induced events (Ciccone and Holdcroft, 1999). In this study, we focused on sex-specific outcomes involving estradiol's effects on the IsoPC ischemic female brain and whether these effects were mediated through ERs.
We have previously reported a gender-specific response to IsoPC in experimental focal cerebral ischemia, with IsoPC being beneficial in ischemic male brain and aggravating ischemic injury in the young female brain (Kitano et al, 2007b). We performed studies examining the role of estradiol in the female-specific response to IsoPC. Our results suggest that estradiol has two potential effects on the response to IsoPC in the ischemic female brain: the loss of IsoPC neuroprotection and the exacerbation of ischemic injury in the cortex and caudate—putamen. We observed that ovariectomizing females, and thus reducing endogenous estradiol levels, led to decreased injury in IsoPC OVX mice as compared with sham PC OVX mice, a response to IsoPC similar to that previously seen in male rodents (Kitano et al, 2007a, 2007b). We then found that estradiol administration in OVX mice enhanced cortical and caudate—putamen ischemic damage in IsoPC mice as compared with sham PC mice. This pattern of response to IsoPC is similar to what we had previously observed in intact females (Kitano et al, 2007b). Therefore, our results show that estradiol can alter the neuroprotective response to IsoPC in females and enhance infarction volume.
Estradiol has for the most part reduced rodent brain injury after an experimental ischemic insult (Macrae and Carswell, 2006; Murphy et al, 2004). The overall neuroprotective results of these animal studies on stroke outcomes have been reflected in a few of the earlier clinical trials that focused on stroke outcomes (Langer, 2002). However, the results of more recent clinical studies, which have focused largely on stroke risk rather than outcomes, have raised the issue that there are unanticipated and paradoxical effects of estrogen as it is currently administered to women (Rossouw et al, 2002; The Women's Health Initiative Steering Committee, 2004). In this study, we have shown that estradiol treatment not only attenuates IsoPC neuroprotection but can also enhance infarction volumes in the IsoPC brain. Several recent experimental studies support our observation that estradiol may not be beneficial in ischemic brain injury outcomes (Bingham et al, 2005; Carswell et al, 2004a; Gordon et al, 2005; Harukuni et al, 2001; Santizo et al, 2002; Xu et al, 2004) and for ischemic preconditioning in experimental myocardial ischemia models (Song et al, 2003; Wang et al, 2006).
We used ER-deficient mice and selective ER agonists to determine whether estradiol acts by an ERα- or ERβ-dependent mechanism to attenuate IsoPC neuroprotection and augment cortical and caudate—putamen infarction volume in the IsoPC ischemic female brain. Our results with ER subtype-deficient mice would suggest that although both ER subtypes may be involved, estradiol's effects on the response to IsoPC in the cortex are largely mediated through ERβ, as the absence of ERβ resulted in decreased cortical infarction volumes in the IsoPC brain. The involvement of chiefly ERβ in mediating estradiol's effects in the IsoPC ischemic cortex is further supported by our experiments with ER subtype agonists, in that no neuroprotection was observed and increased cortical injury was seen only with the ERβ agonist DPN. Because brain ER expression can be affected weeks to months after estrogen deprivation by OVX (Iivonen et al, 2006; Jesmin et al, 2003; Jin et al, 2005; Mohamed and Abdel-Rahman, 2000; Rose'Meyer et al, 2003; Zhou et al, 2002), it is possible that ER expression could have been altered 7 to 10 days after OVX in our studies. Such changes might explain the partial corroboration we observed between the studies using ER subtype-specific knockout mice and selective ER subtype agonists. Because ERβ was implicated by our results in mediating estradiol's effects in the IsoPC ischemic cortex, we evaluated physiologic parameters during ischemia and early reperfusion in preconditioned ERβ-WT and -deficient mice as well as in vehicle and ERβ agonist-treated mice. Our findings relative to infarct volume are not explained by differences in physiologic parameters among treatment groups involving ERβ manipulations.
There is currently controversy as to which ER subtype mediates ischemic outcomes in response to estradiol, with some studies suggesting one or the other receptor subtype, both receptor subtypes, or neither receptor subtype playing a critical role in mediating estradiol neuroprotection (Dai et al, 2007; Farr et al, 2007; Morissette et al, 2008; Singh et al, 2006). Our study did not address this controversy but used fundamental paradigms to test estradiol signaling and IsoPC mechanisms, as little work has been done evaluating the role of ERs in anesthetic or other forms of preconditioning in the ischemic female brain. For example, in a model of hypoxic tolerance using mice preconditioned with 3-nitro-propionate, Von Arnim et al (2002) have observed that ERα mRNA levels were found to be similar in males and females and were unaltered by 3-nitropropionate preconditioning. Interestingly, increased protein levels of ERα but not ERβ in the cortex were observed 24 h after hypoxic preconditioning in neonatal rats (Cimarosti et al, 2005). Our results suggest that in the cortex, ERβ may be predominantly involved in response to anesthetic preconditioning in contrast to other forms of brain preconditioning.
Our results imply that ERβ may have direct effects in the cortex because both ER subtypes are present in the cortex but are scarce in the caudate—putamen (Shughrue et al, 1997). Estrogen receptor β may also mediate vascular outcomes in the cortex and caudate—putamen because both ER subtypes have been observed in the cerebral vessels (Duckles and Krause, 2007; Gonzales et al, 2007). Although we did not observe any significant changes in parietal cortical laser Doppler flowmetry in ERWT, ER-deficient, vehicle, and ER agonist-treated OVX mice, laser Doppler flowmetry measures only relative changes in cortical reperfusion rather than absolute cerebral blood flow in the cortex and other brain regions and may not be sensitive enough to determine whether ERs were involved with peri-ischemic preservation of regional cerebral blood flow in the IsoPC ischemic brain. Future studies using more quantitative measures of regional cerebral blood flow are necessary to determine whether ER-mediated vascular effects are involved with estradiol's actions in the IsoPC ischemic female brain.
This study has some limitations with regard to temperature. There was no attempt to measure or to monitor brain temperature as mice were briefly anesthetized before and the initiation of middle cerebral artery occlusion and upon the induction of reperfusion. Mice were awake throughout most of the ischemic period. Rectal temperature was monitored only while animals were anesthetized, with relatively modest variations in rectal temperature observed. However, rectal temperature does not always correlate with brain temperature. For example, some studies have reported brain temperature to be either approximately 1°C higher or lower than rectal temperature (for review, see van der Worp et al, 2007). Therefore it is possible that brain temperatures may have been transiently hypothermic in our experimental groups. However, rectal temperatures were comparable between sham PC and IsoPC groups for each experimental manipulation (OVX, hormone or drug administration, and ER subtype status). Consequently, any effect of transient rectal temperature changes during the initiation of ischemia and reperfusion would be similar and present in each IsoPC group and its corresponding sham PC group, still allowing us to examine the effect of the presence or absence of the 4 h IsoPC stimulus 24 h before cerebral ischemia under different experimental conditions.
Our previous study suggests gender differences in the response to the IsoPC in ischemic cortex may be mediated through the activation of Akt, a neuronal survival signaling pathway, and the basal expression of neuronal inducible cell-death putative kinase (NIPK), a negative modulator of Akt activation (Kitano et al, 2007b). Recently, NIPK was identified as an estrogen-responsive gene by microarray analysis (Ise et al, 2005; Terasaka et al, 2004), suggesting that the presence or absence of estradiol could alter NIPK levels, thus providing a novel transcriptional mechanism through which estradiol could alter cortical Akt activation in the IsoPC brain. Future studies from our laboratories are necessary to determine whether estradiol acts by an ERβ-dependent or ERβ-independent mechanism to attenuate cortical Akt activation and increase NIPK gene expression in the IsoPC mouse brain, thus leading to increased ischemic damage.
Our observations challenge the assumption that the general response to preconditioning is similar in male and female brains and raise concerns about the effect of sex steroids on preconditioning in the ischemic brain. More studies are needed to further delineate the role of estradiol and ERs in the brain's response to anesthetic preconditioning as well as to determine which pathways are involved down-stream of ERs or independently of ERs. Clinically, our findings suggest that isoflurane anesthesia during “at-risk” cardiovascular surgical procedures may alter perioperative stroke risk and outcomes in women, particularly women on hormone replacement therapy, relative to men.
Footnotes
Acknowledgements
We thank Dr Kenneth S Korach (Laboratory of Reproductive and Developmental Toxicology, National Institute of Environmental Health Sciences, Research Triangle Park, NC, USA) for providing ERα+/− and ERβ+/− (heterozygous) breeder pairs and also acknowledge the excellent service and care provided by the Department of Anesthesiology and Peri-Operative Medicine Mouse Colony Core, which oversaw management of the ERα- and ERβ-deficient mouse breeding colonies. Lastly, we recognize the Endocrine Service Laboratory at the Oregon National Primate Research Center (Beaverton, OR) that performed all plasma hormone measurements.
