Abstract
Experimental stroke models exhibit robust protection after prior preconditioning (PC) insults. This study comprehensively examined cerebral blood flow (CBF) responses to permanent middle cerebral artery (MCA) occlusion in spontaneously hypertensive rats preconditioned by noninjurious transient focal ischemia, using [14C]iodoantipyrine autoradiography at varied occlusion intervals. Preconditioning was produced by 10-min occlusion of the MCA and ipsilateral common carotid artery under halothane anesthesia. These vessels were permanently coagulated 24 h later in naïve, PC, and sham-operated rats. Infarct volumes were determined from hematoxylin-eosin-stained frozen sections after 1 or 3 days. Edema-corrected infarct volume was reduced from 127±21 in naïve rats to 101 ±31 and 52±28 mm3 in sham and PC groups, respectively, at 1 day, with similar results at 3 days. All animals exhibited a consistent CBF threshold for infarction (approximately 30mL/100g/ min). Tissue volumes below this threshold were identical in naïve and PC groups after 15-min occlusion. However, by 3 h the volume of ischemic cortex decreased in the PC group but remained unchanged in naïve rats, predicting final infarct volumes. Cerebral blood flow recovery was confirmed in brains of individual rats evaluated by repeated laser Doppler perfusion imaging during the same 3-h interval. Modest sham protection correlated with better-maintained global perfusion, detectable also in the contralateral cortex, apparently reflecting the PC effects of prior anesthesia. These results establish that timely reperfusion of penumbra, achieved by synergistic mechanisms, is a primary determinant of PC-induced protection in experimental stroke.
Introduction
Preconditioning (PC) effects of prior insults have been described in many tissues, including brain (Dirnagl et al, 2003; Kirino, 2002). In the context of focal ischemia, infarct volumes are reduced by prior global (Simon et al, 1993) or focal ischemia (Barone et al, 1998; Chen et al, 1996; Matsushima and Hakim, 1995), by inflammatory signaling molecules such as lipopolysaccharide (LPS) (Tasaki et al, 1997), by spreading depression (Matsushima et al, 1996; Otori et al, 2003), or even by remote cortical injury that does not induce spreading depression (Muramatsu et al, 2004). A role of perfusion changes in PC has been largely discounted, based on numerous studies that failed to identify cerebral blood flow (CBF) differences between naïve and preconditioned animals during acute intervals after subsequent occlusion, using autoradiographic (Alkayed et al, 2002; Chen et al, 1996; Dawson et al, 1999), hydrogen clearance (Matsushima and Hakim, 1995; Matsushima et al, 1996) or laser Doppler measurements (Barone et al, 1998). Recent results indicate that basal CBF might even be reduced in a preconditioned hemisphere, perhaps secondary to decreases in metabolic rate after treatments that produce cortical injury (Muramatsu et al, 2004; Otori et al, 2003). However, no previous studies combined quantitative CBF measurements with volumetric assessments of the territory below ischemic flow thresholds over an extended time course.
The observation that prior ischemic PC reduced infarct volume even after permanent occlusion in such a robust model as the spontaneously hypertensive rat (SHR) (Barone et al, 1998), and the finding of progressive recovery of microvessel filling during ischemia in LPS-preconditioned rats (Dawson et al, 1999), strongly implicated a perfusion component in the protection mechanism. The present re-examination of CBF in this model establishes that, although the initial distribution of perfusion deficits is unchanged, preconditioned animals show a significant reduction in the volume of severely ischemic tissue by 3 h, which reliably predicts the final infarct volume.
Materials and methods
Experimental Design
These studies compared histopathology and CBF changes after permanent focal ischemia in naïve SHRs, in preconditioned (PC) animals that experienced brief focal ischemia on the previous day, and in a corresponding sham-operated group. The PC insult consisted of 10 mins transient focal ischemia, previously shown to reduce the infarct volume after subsequent permanent occlusions in a comparable model (Barone et al, 1998). Cortical infarct volumes were measured 24 h after permanent test occlusions in each group (naïve, n = 17; sham n = 19; PC, n = 22), and additional animals were evaluated at 3 days survival (naïve, n = 13; sham n = 5; PC, n = 14). Regional CBF was evaluated by quantitative autoradiography in a total of 87 animals in the following experimental groups: normal controls (n = 3); sham alone (n = 5) and PC alone (n = 5) at 27 h after surgery; animals subjected to permanent occlusions and evaluated after 15 mins (naïve, n = 6; PC, n = 6), 90 mins (naïve, n = 3; PC, n = 2), 3 h (naïve, n = 6; sham, n = 9; PC, n = 9), 6 h (naïve, n = 3; sham, n = 3; PC, n = 4), and 24 h (naïve, n = 6; sham, n = 8; PC, n = 9). Those animals in which CBF was evaluated at 24 h were also used to assess infarct volume and contributed to the group totals indicated above. In addition, 12 rats were used for acute laser Doppler perfusion imaging after permanent focal ischemia (naïve, n = 7; PC, n = 5). A total of eight animals (five sham, three PC) exhibited detectable cortical injury or surface hemorrhage due to the initial surgery and were excluded from the above analyses.
Ischemia Model
Male SHR (250 to 350 g, Charles River Laboratories, Wilmington, MA, USA) were allowed free access to food and water before and after all procedures. Focal brain ischemia was produced by tandem occlusion of the right middle cerebral artery (MCA) and ipsilateral common carotid artery (CCA) essentially as described previously (Brint et al, 1988; Kaplan et al, 1991), according to a protocol approved by the Animal Care and Use Committee, University of Tennessee. In brief, animals were anesthetized and ventilated with 0.5% to 1% halothane in 70% N2, 30% O2, and fitted with a tail artery cannula (PE-50) to monitor the blood pressure, and for periodic sampling to measure blood gases, pH, and glucose. Body temperature was monitored and maintained at 37°C using a rectal probe with a feed-back-controlled heating pad and infrared lamp throughout the surgical procedure until complete recovery from anesthesia. The right CCA was exposed and cauterized for permanent ischemia, or a Teflon/silastic occluding device was used to produce temporary flow disruptions. The right temporalis and masseter muscles were dissected and a burr hole was drilled through the temporal-squamous bone to expose the MCA at the level of the rhinal fissure. The temperature of the brain surface was maintained at 37°C during drilling and all subsequent steps with a thermostated saline drip (TC-324B, Warner Instruments Inc., Hamden, CT, USA). The MCA was snared with a micromanipulator-controlled wire hook and raised approximately 1 mm. The vessel was cauterized to produce permanent ischemia or kept elevated for the 10-min duration of PC occlusions, in which case recirculation was achieved by sequentially releasing the MCA and CCA occlusions. Wounds were closed with surgical suture and swabbed with povidone/iodine and lidocaine ointment. Anesthesia was discontinued, animals were weaned from the respirator and allowed to recover consciousness and thermal equilibrium. Sham surgery involved exposure and manipulation of both arteries without occlusion.
Histologic Analysis
Animals were decapitated 1 or 3 days after permanent occlusion and brains were rapidly dissected, frozen in hexane at −40°C, and stored at −70°C. Coronal sections (20 mm) were cut in a cryostat at −20°C and collected at 1-mm intervals throughout the extent of the MCA territory. Slides were briefly fixed in 95% ethanol and stained with hematoxylin and eosin, after which calibrated images were collected (NIH Image). The pale infarct area of each section was summed across all sections to determine the lesion volume in mm3. Edema volume was calculated by the difference in cortical volume between ischemic and nonischemic hemispheres, and subtracted from the total lesion volume to yield the corrected infarct volume (Jacewicz et al, 1990; Swanson et al, 1990).
Autoradiographic Cerebral Blood Flow Measurement
Cerebral blood flow was measured by an indicatorfractionation technique (Brint et al, 1988; Van Uitert et al, 1981) using 4-iodo-[N-methyl-14C]antipyrine (56.2 mCi/mmol; Perkin-Elmer Life Sciences, Inc., Boston, MA, USA) as the diffusible tracer. Aliquots of the label (15 μCi), obtained in ethanol solution, were evaporated to dryness in a stream of air and dissolved in 0.4 mL of physiologic saline. Animals were anesthetized as described above for placement of femoral artery and vein cannulae. For measurements at early time points, this was performed during the course of surgery to produce occlusion. Blood flow measurements were performed in awake animals under brief restraint. The tracer was injected as a bolus into the femoral vein, while femoral artery blood was withdrawn into a syringe containing heparinized saline at a constant rate of 1 mL/min using a programmable pump (SP230iw, World Precision Instruments, Inc., Sarasota, FL, USA). The animal was decapitated approximately 6 secs after injection and the pump was simultaneously stopped. The brain was rapidly removed, frozen in hexane at —40°C, and stored at —70°C. Sampled blood was transferred with saline rinses to a preweighed tube and reweighed to determine the total volume, assuming a density of 1.05 g/mL. Aliquots (20 mL) of the withdrawn blood were decolorized with 0.2 mL H2O2 and counted in 7mL of scintillation fluid (Ultima Gold, Packard Instrument Company, Meriden, CT, USA). Coronal brain sections (20 μm) were cut in a cryostat at —20°C and collected at 0.5-mm intervals throughout the extent of the MCA territory. These were thaw mounted on slides and exposed to a Kodak Biomax MR film for 7 days, together with 14C standards (Amersham Biosciences, Piscataway, NJ, USA), after which calibrated digital images were captured and saved using NIH Image.
The CBF threshold for infarction was determined by aligning histologic images with autoradiographic blood flow images of the same sections, and averaging the CBF values obtained on each side of the infarct border at five depths in cortex. Volumetric assessment of ischemic territory was made by integrating across the collected images from each brain the area of cortex in each section in which the majority of pixels fell below the mean CBF threshold for infarction (30 mL/100 g/min).
Perfusion Imaging
Rats were anesthetized, ventilated, and prepared for occlusion as described above. In addition, the dorsal surface of the skull was exposed and thinned until surface vasculature was uniformly visible. A stream of warmed saline was delivered to maintain a surface temperature of 37°C. Baseline images were collected using the LDI-HR laser Doppler perfusion imaging system (Moor Inc., Wilmington, DE, USA) over a field of 125 × 125 pixels at a sampling rate of 4 ms/pixel and a nominal resolution of 100 mm/pixel. Arteries were then occluded and the animal repositioned for further imaging at 2-min intervals for several hours, with periodic sampling to measure blood gases. Data were successfully collected from seven naïve and five PC rats in which stable contralateral perfusion values were maintained throughout the recording period. Regions of interest (1.2 × 1.4 mm, 168 pixels) were chosen at a consistent anatomical location at the margin of the MCA territory (2 mm lateral, 4 to 5 mm caudal to bregma as required to avoid major blood vessels in individual animals). Mean pixel distributions were determined for 5 consecutive images captured during 10 to 20 and 175 to 185 mins (15 mins and 3-h time points), and differences in these histograms between the two time points were calculated and plotted for each animal.
Temperature Telemetry
Several 3-day survival animals (4 rats each from naïve and PC groups) were previously fitted with intraabdominal transmitters (PDT-4000 E-Mitter, Mini Mitter Co., Inc., Sunriver, OR, USA) under brief halothane anesthesia approximately 1 week before occlusion surgery. Thereafter, cages were placed on ER-4000 receivers, and temperature was continuously monitored using VitalView data acquisition software. Data were collected at 2-min intervals, and values obtained during 30 mins were averaged for presentation.
Statistical Analysis
Group values for all parameters are stated as mean 7 standard deviation (s.d.). Statistical comparisons of infarct and ischemic territory volumes and physiologic parameters among naïve, sham, and PC groups were made using analysis of variance followed by Scheffe' F-test. Comparisons of changes in pixel distributions between naïve and PC animals in the perfusion imaging studies used an unpaired t-test. Both analyses were implemented in StatView 5.0 (SAS Institute, Inc., Cary, NC, USA), with P < 0.05 considered statistically significant.
Results
Preconditioning Efficacy
Striking reductions in infarct volume were observed in preconditioned animals after subsequent permanent MCA/CCA occlusion, with comparable effects noted at 1- and 3-day survival (Figure 1A). Sham surgery resulted in modest protection that nevertheless reached statistical significance for the larger 1-day group. Comparable sham effects were also seen in several animals subjected to exposure and manipulation of either CCA or MCA alone, as well as to procedures involving only halothane anesthesia and tail vessel cannulation (not shown). Telemetry showed no influence of prior PC on core temperature, which remained within the range of diurnal variation in both naïve and PC animals (Figure 1B). Other physiologic parameters were identical across groups during PC and test insults, with the exception of slightly higher pH during and after the test surgery in sham and PC groups, which also exhibited slightly lower blood pressures than naïve animals after recovery from anesthesia (Table 1).
Physiological variables during preconditioning and test insults
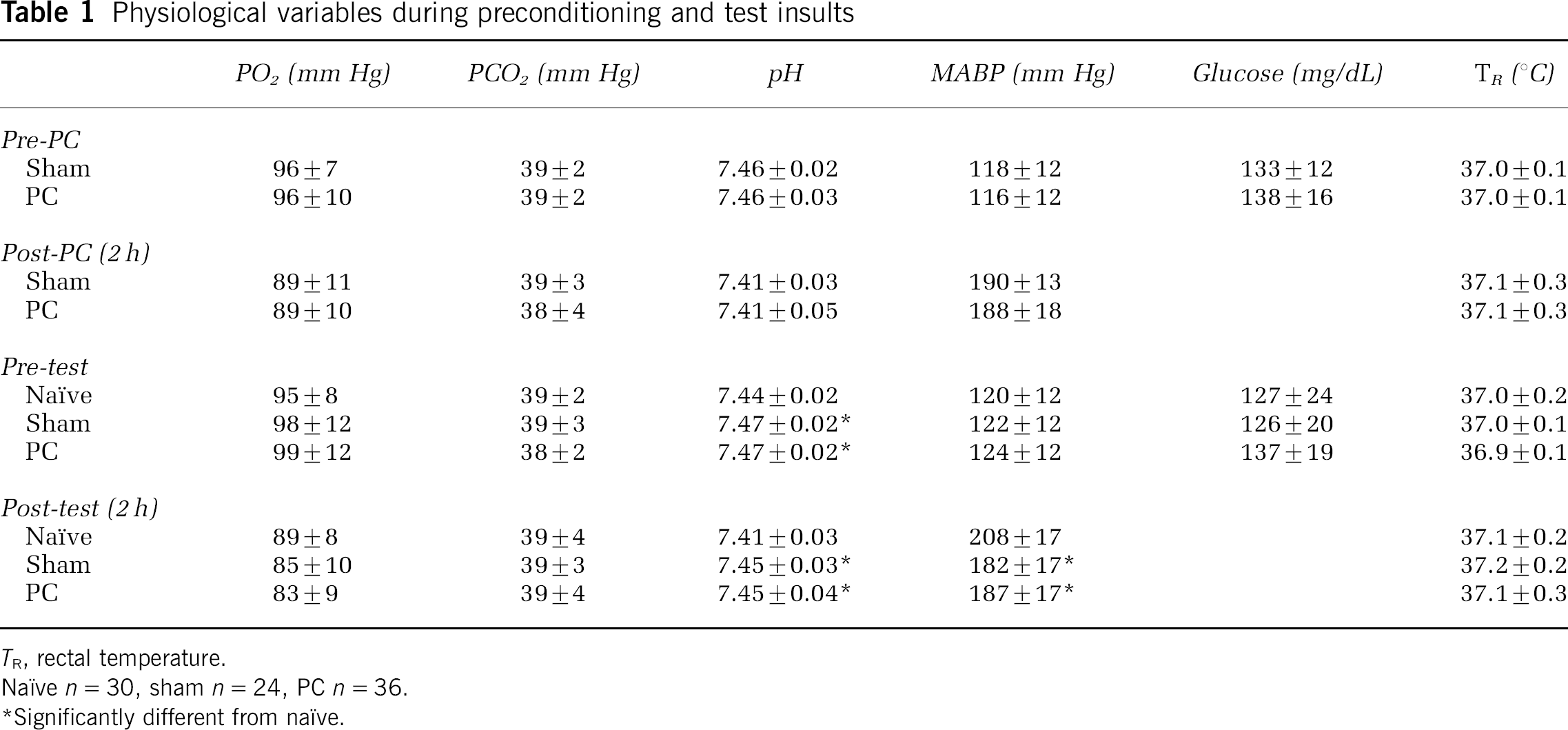
TR rectal temperature.
Naïve n = 30, sham n = 24, PC n = 36.
Significantly different from naïve.
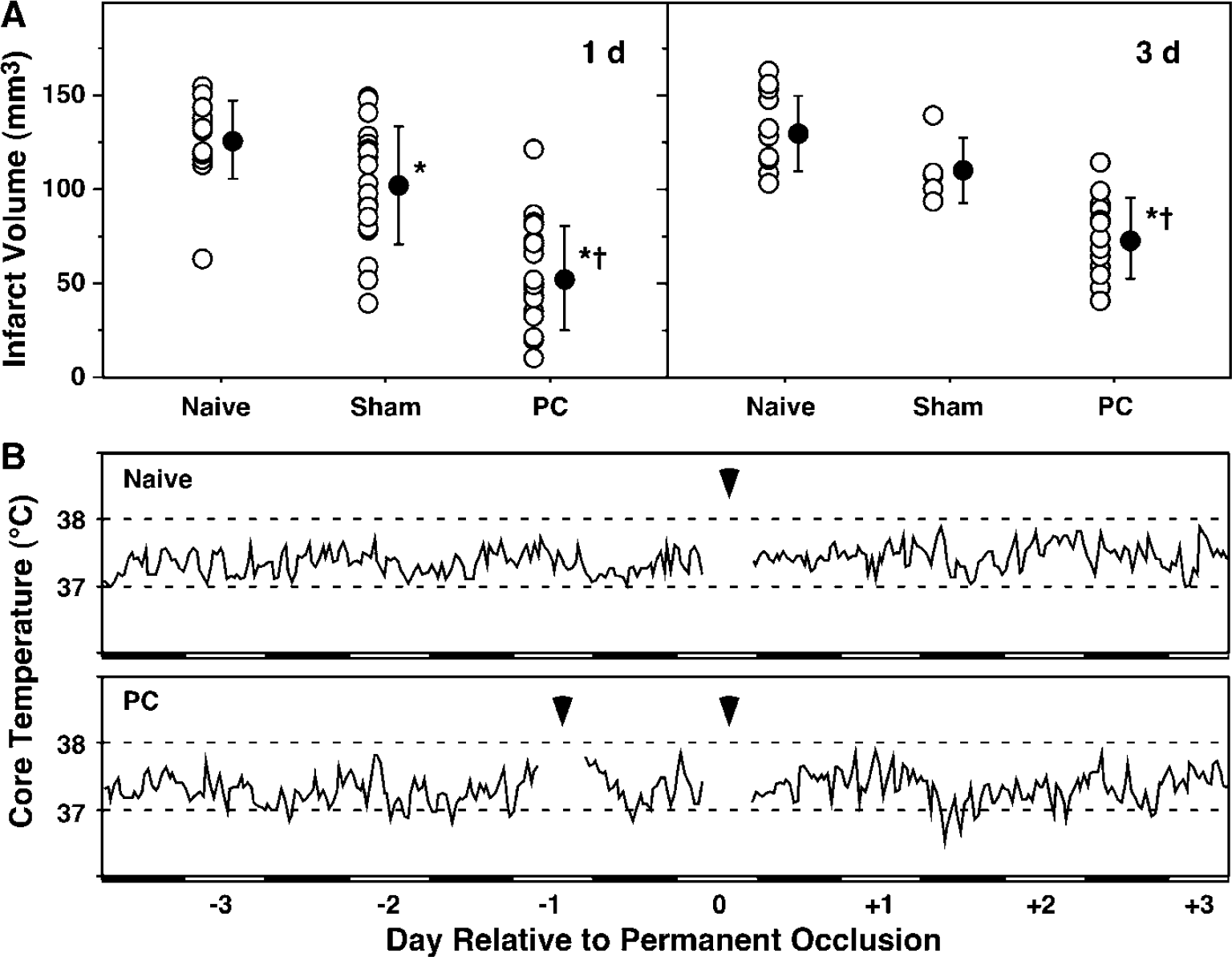
Efficacy of ischemic PC and absence of a temperature effect.
Perfusion and Preconditioning
Cerebral blood flow autoradiograms at the same 24-h time point showed a striking difference in the distribution of severe perfusion deficits in naïve and preconditioned animals (Figure 2A). Quantitative measurements established a consistent CBF threshold for infarction of 30 mL/100 g/min, and the volume of cortex falling below this flow threshold was well-correlated with lesion volume for individual animals of all experimental groups (Figure 2B). An analysis of tissue volumes falling below this threshold as a function of time after MCA occlusion is shown in Figure 2C. The ischemic territory was identical in naïve and preconditioned rats evaluated 15 or 90 mins after occlusion, and this volume remained constant through 24 h in the naïve group (with correction for edema at 3, 6, and 24 h). In contrast, PC led to a marked decrease in the perfusion deficit at 3 h that persisted at 6 and 24 h. Sham groups presented intermediate flow deficits at the 3-, 6-, and 24-h time points at which they were examined (not shown). Physiologic measurements in the CBF study again showed trends toward higher pH and lower blood pressure in awake sham and PC animals during the initial hours after occlusion, only reaching statistical significance for pH at the 3h time point (Table 2).
Physiological variables in quantitative CBF study
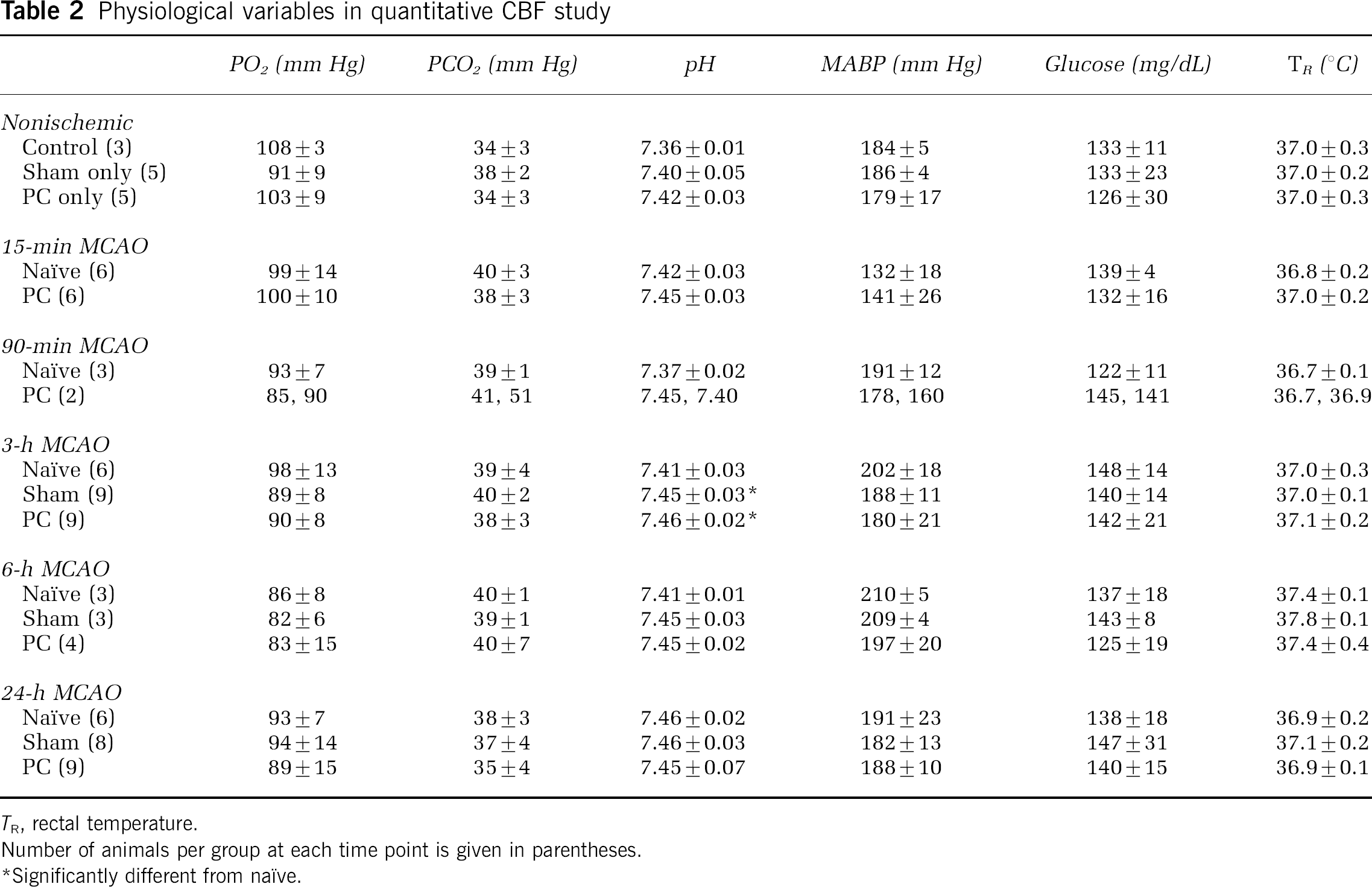
TR rectal temperature:
Number of animals per group at each time point is given in parentheses.
Significantly different from naïve.
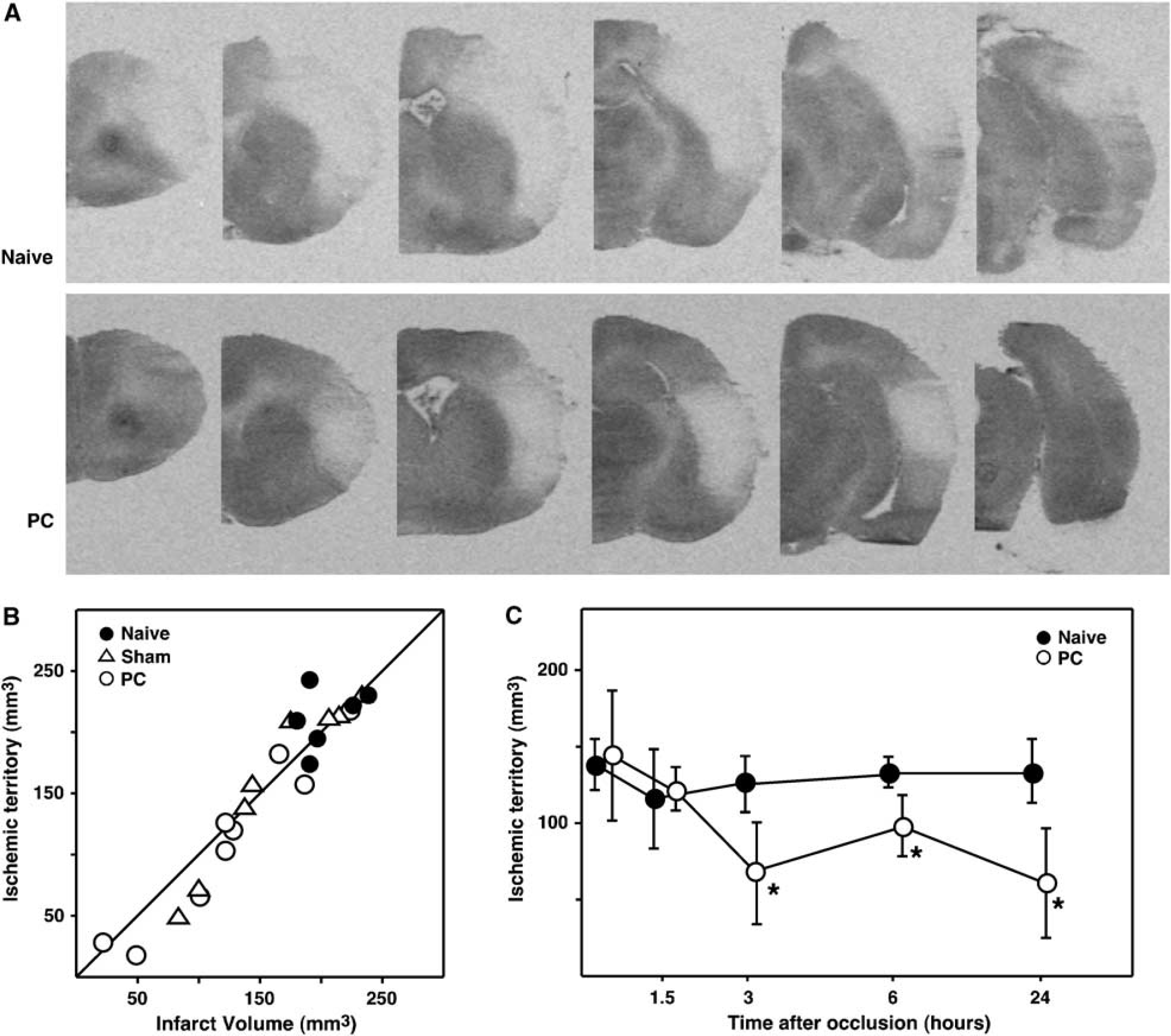
Cerebral blood flow changes associated with PC.
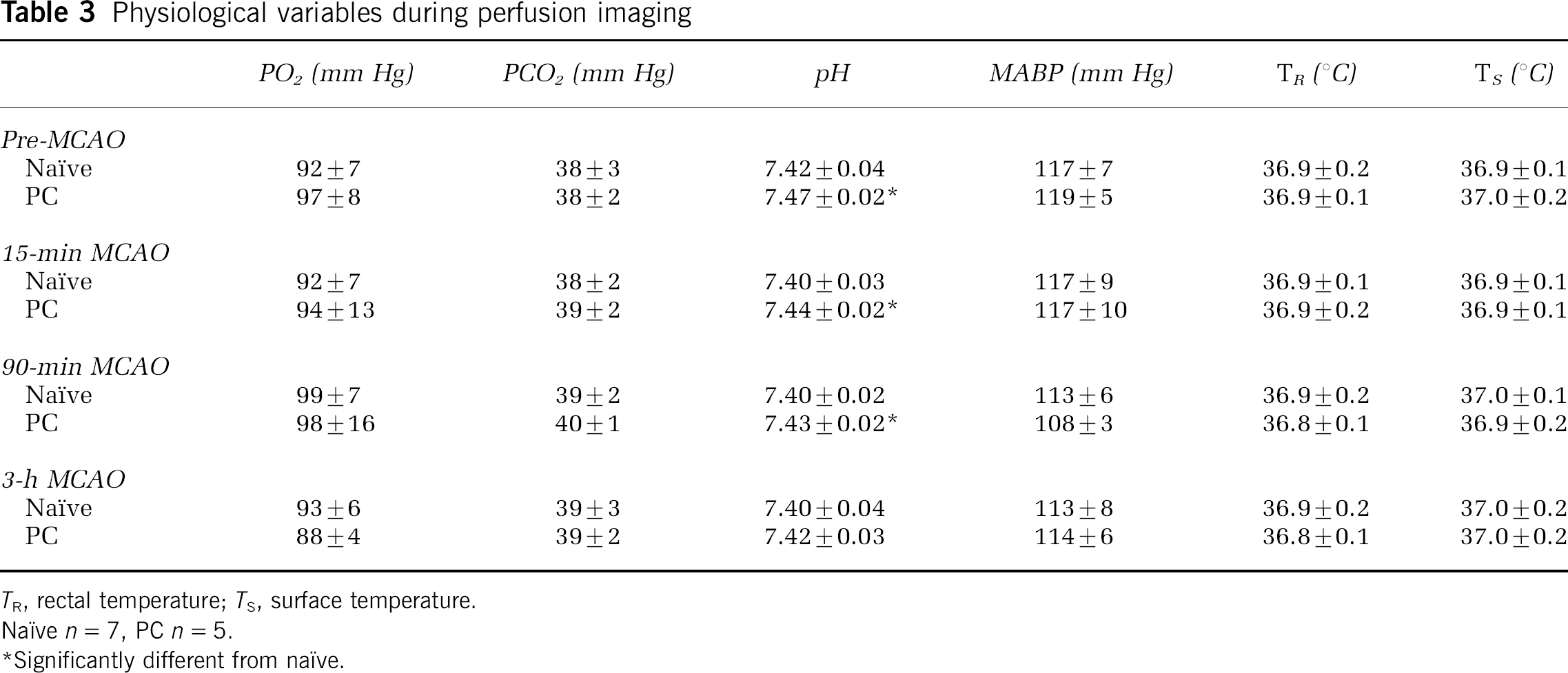
Perfusion Imaging
Laser Doppler perfusion imaging documented dynamic CBF changes during the initial hours after occlusion in individual animals. In contrast to naïve cortex, preconditioned brain showed progressive reperfusion in the margin of the MCA territory, illustrated for representative animals in Figure 3A. Difference plots of the change in mean pixel distribution for the indicated region of interest in Naïve and preconditioned groups are shown in Figure 3B. In naïve animals the perfusion distribution shifted to lower flow values between 15 mins and 3 h, but the preconditioned group showed a consistent shift toward higher perfusion during the same interval, with a decrease in the number of pixels at low flows. Contralateral perfusion remained constant during this interval in both groups. The change in pixel number at flow values in the ischemic range (i.e., below the lower limit of the contralateral flow distribution) differed significantly between naïve and PC animals (P = 0.0013, unpaired t-test). Physiologic parameters did not differ, with the exception of the slightly but consistently higher pH in the PC group (Table 3).
Physiological variables during perfusion imaging
TR, rectal temperature, Ts n = 5
Significantly different from naïve.
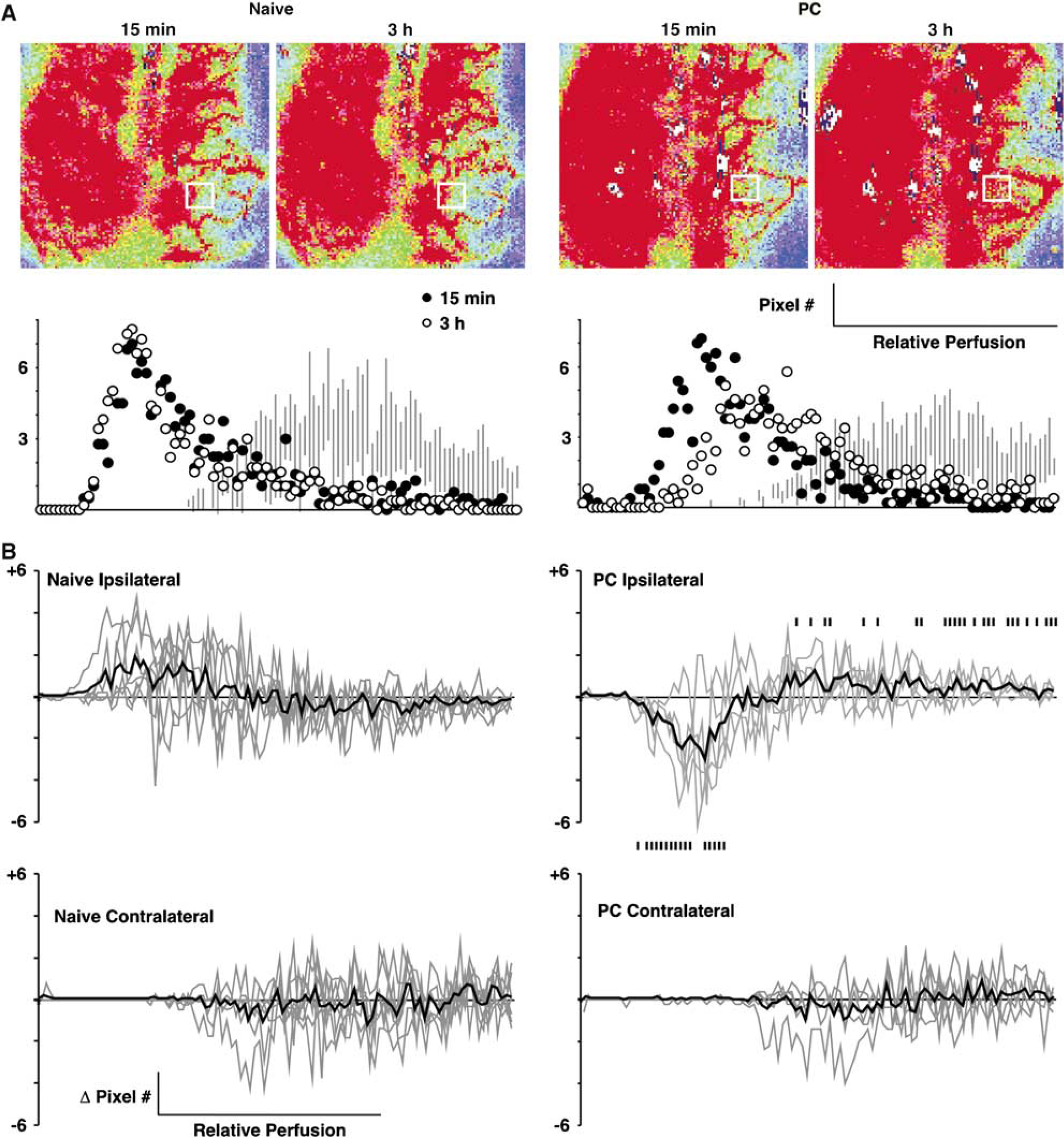
Perfusion imaging of CBF recovery in preconditioned brain.
Differentiating Sham and Preconditioning Effects
The quantitative CBF data yielded several additional observations. Prior sham or PC treatments alone tended to reduce cortical CBF on the subsequent day, more prominently ipsilateral to the manipulation. Mean flow values (mL/100g/min) for control, sham, and PC animals were 160 ± 10, 116 ± 26, and 138 ± 11 in the contralateral (left) hemisphere, and 161 ± 12, 92 ± 40, and 93 ± 35 in the ipsilateral (right) hemisphere, all measured 3 h after brief anesthesia for femoral cannulae placement. Systematic differences in global CBF were noted in contralateral cortex at various intervals after occlusion surgery. In both naïve and PC animals CBF was low at 15 mins, presumably reflecting metabolic suppression by the recent anesthesia, but by 90 mins both had recovered to flow values comparable to those obtained in awake control animals (Figure 4A). Thereafter, CBF was generally well maintained in the PC and sham groups, but tended to be lower in naïve animals, reaching statistical significance at the 3-h time point. These differences in overall perfusion clearly contributed to group differences in the volume of severely ischemic cortex. As shown in Figure 4B, the volume of tissue falling below the infarct threshold was inversely correlated with global CBF in naïve animals, and most of the sham animals also fell along the regression line calculated for the naïve group, but at higher flow values. This is reflected in the distribution of ischemic volumes for shampretreated animals at the low end of the distribution observed for naïve animals (Figure 4C). In contrast, approximately half of the PC group and 2 of 12 sham animals exhibited ischemic territories distributed at much smaller volumes (Figures 4B and 4C), indicating protection independent of global perfusion differences. The distribution of ischemic territories obtained at early postocclusion intervals after the onset of flow differences (3 and 6 h) reliably predicted the distribution of edema-corrected infarct volumes in pooled 1- and 3-day survival groups (Figure 4C). Infarct volumes were systematically lower than the ischemic volumes determined at early intervals after occlusion, reflecting the slight overcorrection for edema that arises from the assumption that swelling is restricted to the lesion territory.
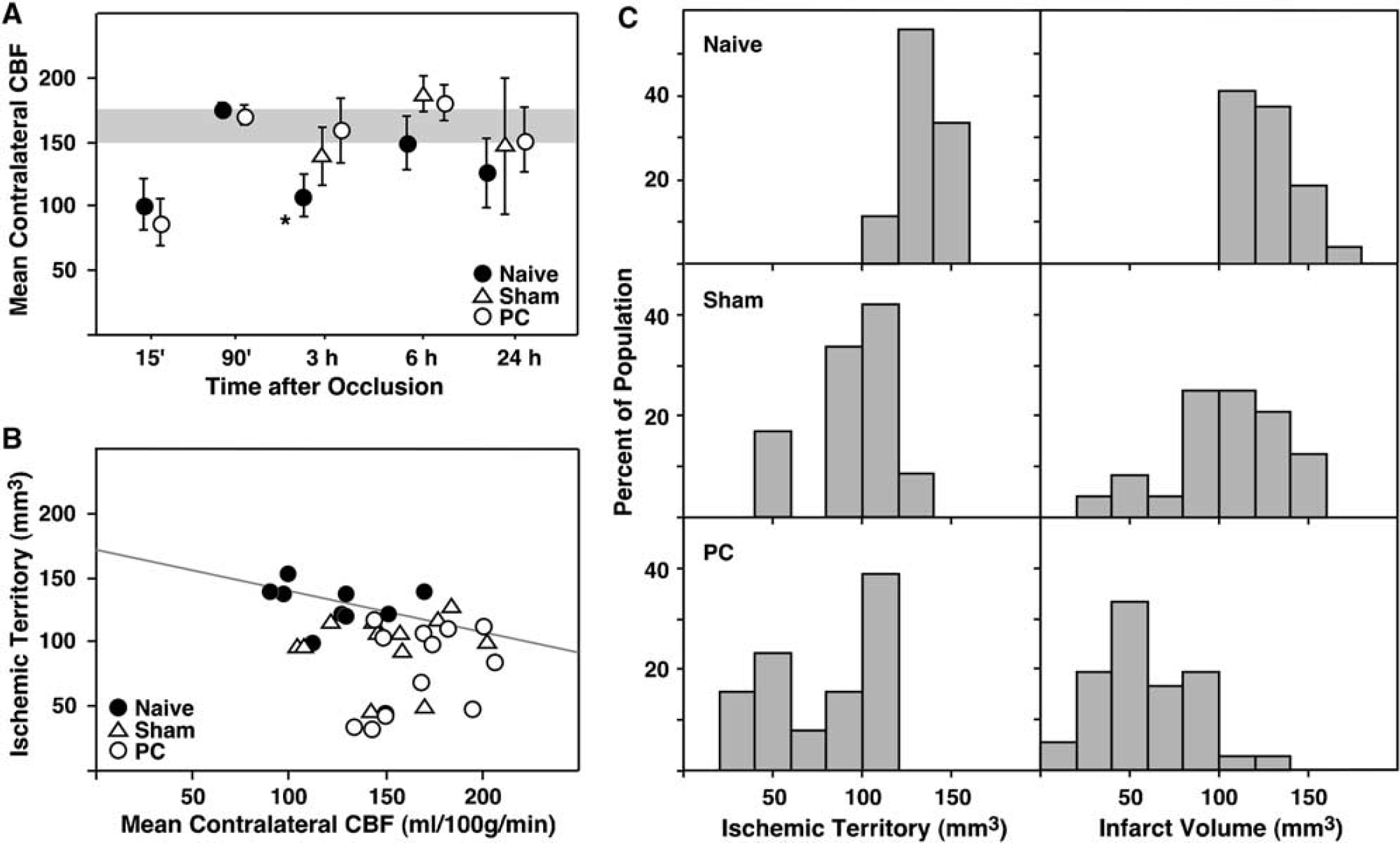
Global CBF changes during focal ischemia and the impact of sham and PC treatments.
Discussion
These results confirm previous observations that PC insults can be protective in models of permanent focal ischemia in the SHR (Barone et al, 1998; Dawson et al, 1999; Tasaki et al, 1997). In contrast to earlier blood flow studies that examined only acute occlusion intervals (Alkayed et al, 2002; Barone et al, 1998; Chen et al, 1996; Dawson et al, 1999; Matsushima and Hakim, 1995; Matsushima et al, 1996), the present work identifies a delayed increase in CBF at the margin of the ischemic territory in PC animals. This is consistent with improvement in microvascular filling noted in LPS-preconditioned rats (Dawson et al, 1999), and with recent laser Doppler measurements in brains preconditioned by cortical lesions (Muramatsu et al, 2004). There appear to be two distinct mechanisms underlying the perfusion effects relevant to PC. Better maintained CBF after recovery from occlusion surgery, detectable contralaterally, is observed in PC animals as well as those previously subjected to sham procedures, and appears to be largely a consequence of prior exposure to halothane anesthesia. Superimposed on this is a progressive recovery of CBF in penumbra, decreasing the volume of the ischemic territory independent of generalized changes in CBF, which occurs selectively in the PC group.
Sham Effects and the Contribution of Anesthetic Preconditioning
The impact of sham surgery on subsequent ischemic injury contributed modestly to overall protection in the model (Figures 1 and 4C). This appears consistent with previously described PC by halogenated anesthetics (Kapinya et al, 2002; Xiong et al, 2003). The effect seen here was much less robust, perhaps reflecting shorter exposure times and potential strain differences in sensitivity. A role for inducible nitric oxide synthase (iNOS) in anesthetic PC has been suggested based on its increased expression after such exposure, as well as elimination of PC effects by the iNOS inhibitor, aminoguanidine, given before initial anesthesia (Kapinya et al, 2002). Parallel mechanisms might be relevant to ischemic PC models (see below), and the potential for synergistic effects must therefore be considered.
Both sham and PC groups avoided the delayed decline in global CBF seen in the contralateral cortex of naïve animals after occlusion surgery (Figure 4A), and this difference was sufficient to account for the small reductions in infarct volume seen in the sham group (Figures 4B and 4C).
Decreases in contralateral perfusion have been described in previous CBF evaluations during and after focal ischemia in naïve animals (Tamura et al, 1981; Zhao et al, 1997), and were also evident in both sham and PC rats in a previous PC study (Chen et al, 1996). The absolute contralateral blood flow measured in the naïve group at 3 h is identical to that obtained at 2.5 h in a separate study from this laboratory (Kamiya et al, 2005). The inverse relationship between contralateral CBF and ischemic volume is simply correlative, and many factors may differentially impact perfusion proximal to the occluded MCA territory. However, CBF in ipsilateral frontal cortex remained well correlated with mean contralateral CBF but averaging approximately 50% lower (not shown), perhaps reflecting responses to cortical injury as well as the effect of persistent CCA occlusion to limit flow. We cautiously interpret contralateral CBF as a general indicator of cerebral vascular tone on which additional local mechanisms would be superimposed.
The importance of physiologic control in focal ischemia models is well recognized (Zausinger et al, 2002), and the consistent blood gas values throughout these studies indicate both adequate ventilation and good recovery of spontaneous respiration. However, both sham and PC groups showed slight increases in systemic pH and decreases in blood pressure relative to the naïve group at early postocclusion intervals (Tables 1 to 3). Blood pressure and pH were identical among the groups of awake animals evaluated at 24 h after occlusion (Table 2), and were comparable to the values obtained at earlier intervals in the sham and PC groups. Therefore, it seems appropriate to interpret the sham effect as an avoidance of mild systemic acidosis, together with a blunted hypertensive response to subsequent MCA occlusion. It is unlikely that such slight physiologic variations would be directly relevant to either perfusion differences or ischemic injury, but they may serve as indicators of mechanistically relevant underlying processes.
The increase in blood pressure during occlusion in the naïve group must reflect sustained vascular constriction, and attenuation of this response could be a correlate to the effect of sham treatment to better maintain global perfusion. There is a considerable literature documenting the acute vasomotor effects of halothane and other anesthetics in specific vascular beds, but interactions between focal ischemia and anesthesia withdrawal have not been extensively examined, much less in the context of repeated anesthesia. Halothane and related anesthetics increase CBF (Farber et al, 1997; Hansen et al, 1988; Jensen et al, 1992; Koenig et al, 1993; McDowall, 1967). Spontaneously hypertensive rats and Wistar-Kyoto strains exhibit particularly prominent perfusion increases during halothane exposure with metabolic suppression by pentobarbital (Takahashi et al, 1996). Sustained halothane administration increased blood flow and reduced infarct volume relative to cats switched to α-chloralose anesthesia after MCA occlusion (Saito et al, 1997). Halothane withdrawal may therefore amplify the underlying vasoconstrictive responses to occlusion. Blood flow progressively decreases during prolonged halothane anesthesia (Warner et al, 1985), but vascular responses during delayed reexposure has not been examined. The effect observed here could be viewed as a blunting of postischemic vasoconstrictive responses to halothane withdrawal.
Subtle differences in blood pH between naïve and sham groups emerge during preparative surgery (Tables 1 and 3) and are therefore independent of occlusion per se Halothane is recognized to induce an extreme hypermetabolic state in susceptible humans and animal strains that results in malignant hyperthermia, arising through the effects on sarcoplasmic reticulum calcium efflux, most often via activation of mutant ryanodine receptors (Litman and Rosenberg, 2005). The subtle pH differences observed here could conceivably reflect weak effects on wild-type receptor, the halothane sensitivity of which can be unmasked at low Mg2+ concentration (Duke et al, 2003, 2004). However, it is increasingly appreciated that such anesthetics impact a wide range of channel activities in brain and other tissues (Franks and Honore, 2004). Whatever the mechanism, the differential effect in naïve versus sham/PC groups again suggests that it exhibits desensitization after prior anesthesia exposure.
Perfusion Changes and Ischemic Preconditioning
Dramatic reductions in infarct volume were observed in the PC group relative to both naïve and sham-operated animals (Figure 1A), consistent with previous studies using diverse PC methods in permanent focal occlusion models (Barone et al, 1998; Otori et al, 2003; Tasaki et al, 1997). This occurred in the absence of detectable differences in physiologic parameters relative to the sham group (Tables 1 to 3), and diurnal temperature rhythms were likewise unchanged by the PC treatment (Figure 1B). However, the PC group exhibited robust CBF recovery in the margin of the ischemic MCA territory, clearly evident within 3 h after occlusion (Figures 2 and 3), independent of global perfusion change (Figures 3 and 4). It remains possible that CBF increases occurred secondary to enhanced functional recovery of preconditioned tissue, and the present studies did not formally distinguish the cause/effect relationship between perfusion and protection. Nevertheless, the increase in blood flow occurred within a temporal window during which reperfusion can be of benefit in this model (Jacewicz et al, 1992; Kaplan et al, 1991; Ren et al, 2004). Together with the demonstration that the ischemic territory defined at 3 to 6 h predicts subsequent infarct volume (Figure 4C), this strongly implicates timely reperfusion as a primary mechanism underlying protection by ischemic PC.
This result is consistent with the similarly delayed recovery of vascular filling in LPS-preconditioned rats (Dawson et al, 1999), which also employed an SHR permanent occlusion model. Previous quantitative perfusion studies had either examined acute occlusion intervals during which no effect is evident (Chen et al, 1996; Dawson et al, 1999), or had involved hydrogen clearance measurements at one point within the MCA territory (Matsushima and Hakim, 1995; Matsushima et al, 1996). A laser Doppler evaluation in the SHR had been interpreted to exclude perfusion effects (Barone et al, 1998), although a trend toward increased relative flow could be detected during the 2-h interval evaluated. More recently, significant flow recovery has been noted in the Sprague-Dawley strain after PC by cortical injury (Muramatsu et al, 2004). The volumetric analysis applied in the present study avoids the limitations of monitoring at specific anatomical loci, and shows a consistent flow threshold for infarction in naïve, sham, and preconditioned brain (Figure 2). A similar approach did not identify a significant difference in perfusion between naïve and preconditioned Wistar rats subjected to intraluminal filament occlusions (Al-kayed et al, 2002). However, there was a trend toward larger tissue volume at higher blood flows in the PC group in that study, and the 2-h occlusion interval examined corresponds to a point at which CBF differences would have just begun to emerge in the present experiments. The absolute CBF threshold for infarction identified here is lower than the 40 to 50 mL/100g/min reported in earlier studies in this model (Jacewicz et al, 1992), but somewhat above thresholds for overt energy failure (Hossmann, 1994; Kamiya et al, 2005), falling within a ‘low penumbral’ range (Ginsberg, 2003; Zhao et al, 1997). The measured flow values and infarct thresholds varied with cortical depth in this study (not shown), and the averaging method applied here gave equal weight to deeper layers with lower flows. The perfusion gradient in this model is sufficiently steep that variations in the precise threshold applied would not greatly impact the calculated lesion volumes.
A reduction in baseline CBF was reported in hemispheres preconditioned by spreading depression in which relative perfusion was found to be increased (Otori et al, 2003), leading to the suggestion that absolute intraischemic flow values could actually be similar in sham and preconditioned brain. In the present study, we also found evidence of blood flow reductions 24 h after surgical manipulations, but this effect was comparable in sham and PC groups and was highly variable, and somewhat less evident flow reductions were also noted in the contralateral hemisphere. As noted above, there is strong evidence that contralateral CBF after permanent MCA occlusion was in fact better maintained in the sham and PC groups (Figure 4A). Nevertheless, it is possible that small cortical lesions could have been produced occasionally by either sham or PC surgery, and by analogy with the PC effects of intentional cortical lesioning (Muramatsu et al, 2004) these might account for the rare small infarcts seen in the sham group (Figure 4C). As noted in the previous study, decreased baseline CBF in such hemispheres would be expected to be associated with reduced metabolic rate in response to the prior injury (Pappius, 1981; Yoshino et al, 1991), which would also contribute to overall protection.
The potential contribution of perfusion effects to protection against transient ischemic insults remains uncertain. Reperfusion would accomplish the primary event identified in the present study, presumably unmasking other potential protection mechanisms, as well as introducing the possibility for additional pathologies (Aronowski et al, 1997; Ren et al, 2004). Preconditioning effects are frequently studied with transient occlusions as test insults (Chen et al, 1996; Matsushima et al, 1996), particularly in mice (Rosenzweig et al, 2004). Preconditioning in such models can involve a range of heterogeneous phenomena. As an extreme example, some studies have identified only very delayed effects emerging weeks after PC (Yanamoto et al, 1998, 2004). In the context of reperfusion, other identified PC effects such as increased mitochondrial DNA repair activity (Chen et al, 2003) or attenuated lipid peroxidation (Chimon and Wong, 1998) might be expected to predominate. However, postischemic hypoperfusion is attenuated in preconditioned retina (Lin and Roth, 1999), and improved CBF recovery could also be a factor in brain PC against transient focal ischemia.
The mechanisms underlying dynamic perfusion changes in preconditioned brain remain to be defined. A number of observations implicate nitric oxide (NO) in PC (Huang, 2004). Vascular nitric oxide synthase (NOS) expression is increased, reported for the endothelial isoform (eNOS) after LPS PC in rats (Puisieux et al, 2000), and for the inducible isoform (inducible nitric oxide synthase (iNOS)) after ischemic PC in mice (Cho et al, 2005). A temporally associated increase in vasodilatory tone, showed in rat aorta after LPS treatment (Pu et al, 1999), might be a functional correlate of increased NOS expression. Available pharmacologic evidence is even more indicative of NO involvement in the initial mechanisms that trigger PC, with model-specific effects of various inhibitors to eliminate PC when given during the priming insult (Cho et al, 2005; Gidday et al, 1999; Puisieux et al, 2000). Failure to observe acute PC in both neuronal NOS and eNOS knockout mice (Atochin et al, 2003) or delayed protection in iNOS knockout mice (Cho et al, 2005) could reflect effects on both the development and the expression of PC. Nitric oxide is also implicated in cellular PC mechanisms (Nandagopal et al, 2001), independent of vascular effects. Conversely, induction of the arachidonic acid epoxygenase, P454 2C11, would be expected to increase levels of vasodilatory epoxyeicosatrienoic acids in preconditioned brain (Alkayed et al, 2002), providing a further mechanism that could contribute to perfusion effects of PC.
Summary
These results document striking recovery of CBF during MCA occlusion in preconditioned SHR, presaging reductions in infarct volume. This arises via two apparently independent mechanisms. First, PC rats better maintain global CBF during recovery from the test insult, detectable in contralateral cortex. This is observed in both PC and sham animals and appears to account entirely for the protection seen in the latter group. In addition, there is progressive reperfusion of penumbra in the ipsilateral cortex of the PC group, observable also by perfusion imaging of individual brains, independent of global perfusion changes. The underlying mechanisms and the relative contributions of perfusion effects to PC in other focal ischemia models remain to be delineated.
