Abstract
Prior 13C magnetic resonance spectroscopy (MRS) experiments, which simultaneously measured in vivo rates of total glutamate-glutamine cycling (Vcyc(tot)) and neuronal glucose oxidation (CMRglc(ox), N), revealed a linear relationship between these fluxes above isoelectricity, with a slope of ~1. In vitro glial culture studies examining glutamate uptake indicated that glutamate, which is cotransported with Na+, stimulated glial uptake of glucose and release of lactate. These in vivo and in vitro results were consolidated into a model: recycling of one molecule of neurotransmitter between glia and neurons was associated with oxidation of one glucose molecule in neurons; however, the glucose was taken up only by glia and all the lactate (pyruvate) generated by glial glycolysis was transferred to neurons for oxidation. The model was consistent with the 1:1 relationship between ΔCMRglc(ox), N and ΔVcyc(tot) measured by 13C MRS. However, the model could not specify the energetics of glia and γ-amino butyric acid (GABA) neurons because quantitative values for these pathways were not available. Here, we review recent 13C and 14C tracer studies that enable us to include these fluxes in a more comprehensive model. The revised model shows that glia produce at least 8% of total oxidative ATP and GABAergic neurons generate ~18% of total oxidative ATP in neurons. Neurons produce at least 88% of total oxidative ATP, and take up ~26% of the total glucose oxidized. Glial lactate (pyruvate) still makes the major contribution to neuronal oxidation, but ~30% less than predicted by the prior model. The relationship observed between ΔCMRglc(ox), N and ΔVcyc(tot) is determined by glial glycolytic ATP as before. Quantitative aspects of the model, which can be tested by experimentation, are discussed.
Introduction
Glutamate, the major excitatory neurotransmitter, and y-amino butyric acid (GABA), its conjugate inhibitory neurotransmitter, constitute more than 90% of cortical neurons in the adult mammalian brain (Nicholls, 1989, 1993). Because glutamate is also a precursor of GABA (Roberts and Frankel, 1950), glutamate metabolism plays a central role in glutamatergic and GABAergic function (Nicholls, 1989). Glutamate is a common constituent of neurons and glia but its concentration differs greatly in these cell types; glutamatergic neurons possess ~80% to 88%, whereas GABAergic neurons contain ~2% to 10% and astrocytes ~10% of total tissue glutamate (Storm-Mathisen et al, 1983; Ottersen and Storm-Mathisen, 1984; Korf and Venema, 1983; Van den Berg and Garfinkel, 1971; Patel et al, 1974; Ottersen et al, 1992; Lebon et al, 2002).
14C and 15N isotope labeling studies performed in the 1960s and 1970s (Van den Berg et al, 1969; Van den Berg and Garfinkel, 1971; Van den Berg and Matheson, 1975; Berl et al, 1962) provide an early framework for our current understanding of neuronal-glial trafficking and the glutamate-glutamine cycle (Benjamin and Quastel, 1975). The role of GABA as an inhibitory neurotransmitter in the mammalian brain was firmly established by the mid-1970s (Roberts, 1960; Roberts et al, 1976) with the establishment of glutamate's role as an excitatory neurotransmitter following shortly thereafter (Storm-Mathisen and Iversen, 1979). Results from the modeling of 14C isotope data (Garfinkel, 1966, 1970) were consistent with the existence of two compartments (i.e., ‘large’ and ‘small’ tricarboxylic acid cycles) and the movement of substrates (e.g., glutamate, glutamine, and GABA) between them, although their connection to specific cellular or subcellular locations were unclear. The concept of a glutamate-glutamine neurotransmitter cycle (Benjamin and Quastel, 1975) was solidified by the discovery that key enzymes (e.g., glutamine synthetase (Martinez-Hernandez et al, 1977; Norenberg and Martinez-Hernandez, 1979) and pyruvate carboxylase (Yu et al, 1983; Shank et al, 1985)) were localized in the glia, thus linking the ‘large’ and ‘small’ compartments to neurons and glia, respectively. Little was known, however, about the magnitude of the neurotransmitter cycling flux.
The first quantitative measurements of the glutamate-glutamine cycle in vivo were made during the 1990s using 13C magnetic resonance spectroscopy (MRS). These early 13C MRS experiments simultaneously measured (Sibson et al, 1997) the in vivo rates of total glutamate-glutamine cycling (Vcyc(tot)) and neuronal glucose oxidation (CMRglc(ox), N). Measurements of both rates in the cerebral cortex of anesthetized rats at different levels of electrical activity found a linear relationship above iso-electricity with a slope of ~1 (Sibson et al, 1998). Studies of glial cultures (Pellerin and Magistretti, 1994) showed that glial glutamate uptake (Flott and Seifert, 1991) cotransported with Na+ (Kimelberg et al, 1989) stimulated glial glycolysis and lactate production (Ercinska and Silver, 1987).
Glial metabolism of glucose to lactate produces two ATP equivalents per glucose, in principal sufficient to maintain active glutamate clearance from the extracellular space—one for pumping of Na+/K+ ions and the other for glutamine synthesis. The approximately 1:1 correspondence of ΔCMRglc(ox), N and ΔVcyc(tot) (Figure 1) was proposed (Shulman and Rothman, 1998) to be consistent with a model in which glial glycolytic ATP supports glutamate uptake and subsequent glutamine formation (Magistretti et al, 1994). Considering only these 13C MRS results (Sibson et al, 1998; gray triangles in Figure 1), all the lactate (pyruvate) produced by glial glycolysis was assumed to be transported to the neurons for oxidation. A key factor uniting these components was the energy needed to clear glutamate from the extracellular space (Shulman and Rothman, 1998). In addition to the energy supporting cycling activity, the intercept on the CMRglc(ox), N axis of Figure 1 (i.e., iso-electricity) signified a smaller but necessary oxidative neuronal energy for noncycling activities (Shulman and Rothman, 1998).
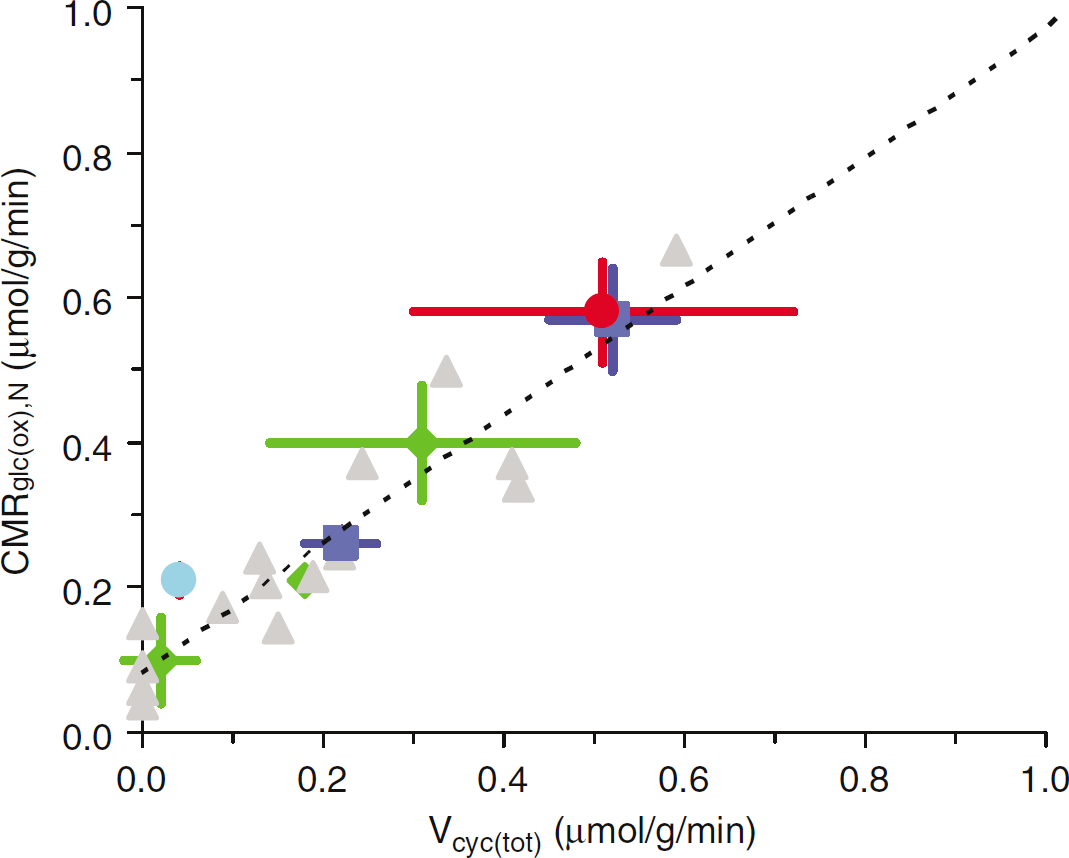
Experimental results of Vcyc(tot) and CMRglc(ox), N. Values of Vcyc(tot) and CMRglc(ox), N for the rat brain reported in studies published between 1998 and 2005. The dark blue squares are from Patel et al (2004a), the red circle is from Oz et al (2004), the light blue circle is from Choi et al (2002), the green diamonds are from de Graaf et al (2004), and the gray triangles are from Sibson et al (1998). See Table 1 for details.
Summary of metabolic fluxes (μmol/g/min) measured using in vivo 13C MRS in rat brain at different activity levels
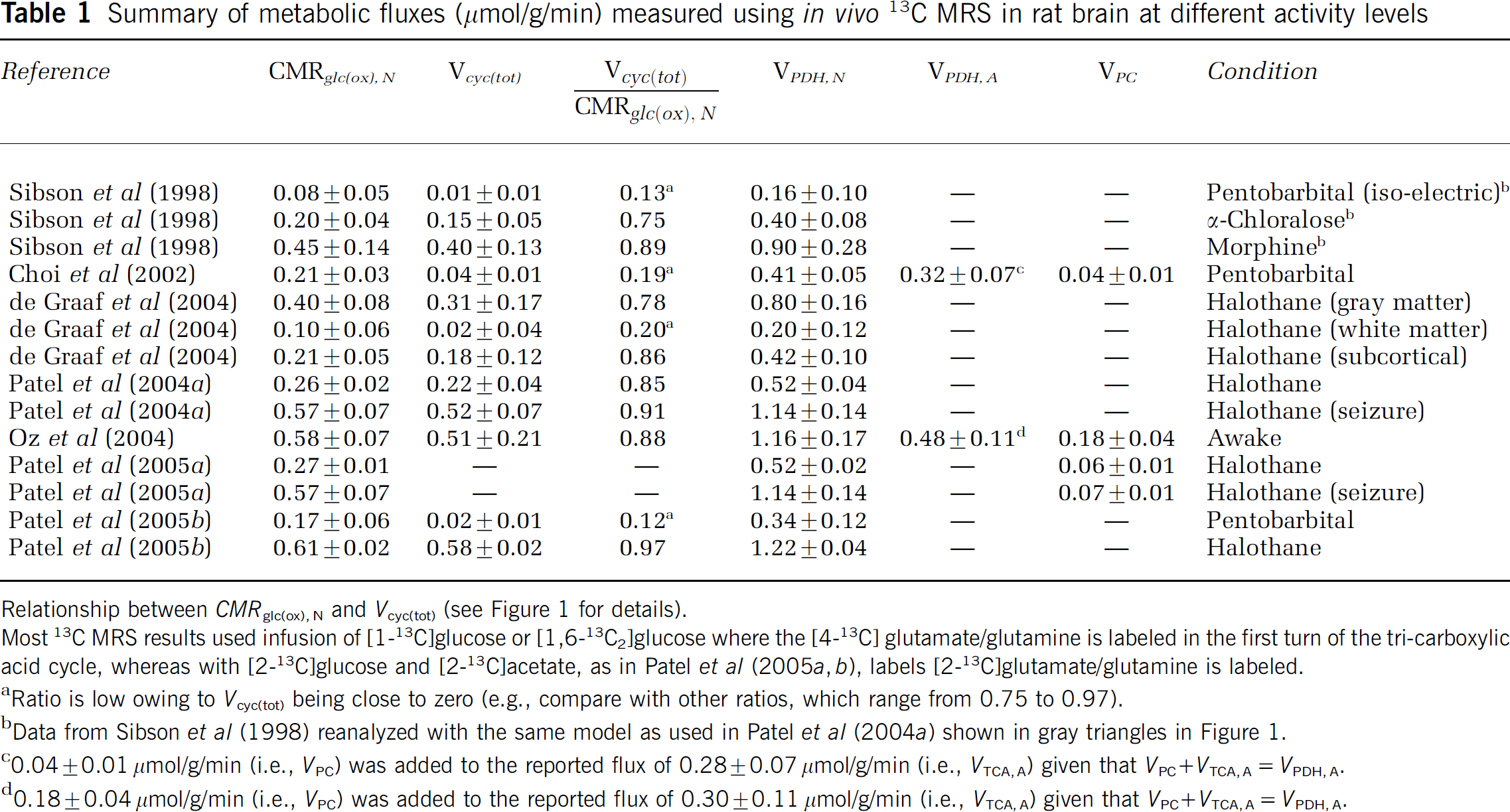
Relationship between CMRglc(ox), N and Vcyc(tot) (see Figure 1 for details).
Most 13C MRS results used infusion of [1-13C]glucose or [1,6-13C2]glucose where the [4-13C] glutamate/glutamine is labeled in the first turn of the tri-carboxylic acid cycle, whereas with [2-13C]glucose and [2-13C]acetate, as in Patel et al (2005a, b), labels [2-13C]glutamate/glutamine is labeled.
Ratio is low owing to Vcyc(tot) being close to zero (e.g., compare with other ratios, which range from 0.75 to 0.97).
Data from Sibson et al (1998) reanalyzed with the same model as used in Patel et al (2004a) shown in gray triangles in Figure 1.
0.04 ± 0.01 μmol/g/min (i.e., VPC) was added to the reported flux of 0.28 ± 0.07 μmol/g/min (i.e., VTCA, A) given that VPC + VTCA, A = VPDH, A.
0.18 ± 0.04 μmol/g/min (i.e., VPC) was added to the reported flux of 0.30 ± 0.11 μmol/g/min (i.e., VTCA, A) given that VPC ² VTCA, A = VPDH, A.
The summary of these results (Magistretti et al, 1999) stimulated much interest (e.g., Coyle and Schwarcz, 2000; Raichle and Gusnard, 2002; Lauritzen and Gold, 2003; Vannucci and Simpson, 2003; Leybaert, 2005) and raised questions about the relationship between brain glucose oxidation and glutamate-glutamine cycling (e.g., Griffin, 1999; Hertz and Robinson, 1999; Chih et al, 2001; Gruetter et al, 2001; Gruetter, 2002; Gjedde et al, 2002; Attwell and Iadecola, 2002; Choi et al, 2002; Chih and Roberts, 2003; Oz et al, 2004). Many were concerned about the significance of energetic pathways that were not measured in the Sibson et al (1998) study, for example, glial glucose oxidation, glucose oxidation in GABAergic neurons, and glial oxidation of neurotransmitter glutamate. There were also concerns about the exclusion of neuronal glucose uptake, and whether the relationship would hold for other conditions.
Here, we review recent results from several laboratories that supplement the data on which the original model of glutamate-glutamine cycling was based (Magistretti et al, 1999). The new data are used to address questions raised about glucose uptake as pictured in the prior model (Magistretti et al, 1999), where all the lactate (pyruvate) that is oxidized (in neurons) to support signaling was produced by glial glycolysis. Not considering glucose oxidation in glia and GABAergic neurons, the prior model (Magistretti et al, 1999) accommodated the 1:1 relationship between ΔCMRglc(ox), N and ΔVcyc(tot) by allowing all the glial lactate (pyruvate) to shuttle to neurons. The revised model takes into account recent measurements of oxidation in glia and GABAergic neurons and retains the essence of the earlier model, for example, lactate (pyruvate) flow from glia to neurons. However, it differs by allowing for oxidation of some of the pyruvate (lactate) in the glia to meet their independent needs for oxidative energy. Consequently, the revised model allows for direct neuronal uptake and phosphorylation of glucose compensating for the reduced glial lactate (pyruvate) shuttled to neurons. In the revised model, the 1:1 stoichiometry between ΔCMRglc(ox), N and ΔVcyc(tot), an experimental measurement, is still consistent with glial glucose uptake. The revised model, as discussed below, makes specific testable predictions of the relative uptake of glucose by neurons and glia, and of lactate (pyruvate) efflux from the brain.
The relationship between ΔCMRglc(ox), N and ΔVcyc(tot) was questioned most often. Was the slope of Figure 1 really unity? This question has been addressed in recent studies (Choi et al, 2002; de Graaf et al, 2004; Patel et al, 2004a; Oz et al, 2004) and is discussed in the next section (Table 1). Another question centered on glial oxidative processes (i.e., pyruvate dehydrogenase and pyruvate carboxylase pathways), for which direct experimental results were lacking earlier. Results from different laboratories have reported on neuronal and glial fluxes (Table 2) which are summarized in the section Glial energetics. Other questions were related to GABA; fluxes associated with GABA were neglected and assumed to be included with the corresponding glutamatergic fluxes because their magnitudes were not known in the 1990s (Rothman et al, 1999). Recent measurements of GABAergic fluxes (Patel et al, 2005b) quantify this pathway in Figure 2 as summarized in the section GABAergic contributions (Table 3). Finally, the issue of glutamate oxidation versus glutamate recycling (Hertz and Robinson, 1999; Lieth et al, 2001) is discussed followed by consolidation of all the data into a revised model. Below are the definitions of the different fluxes:
Summary of metabolic fluxes (μmol/g/min) measured using in vivo 13C MRS in awake human brain
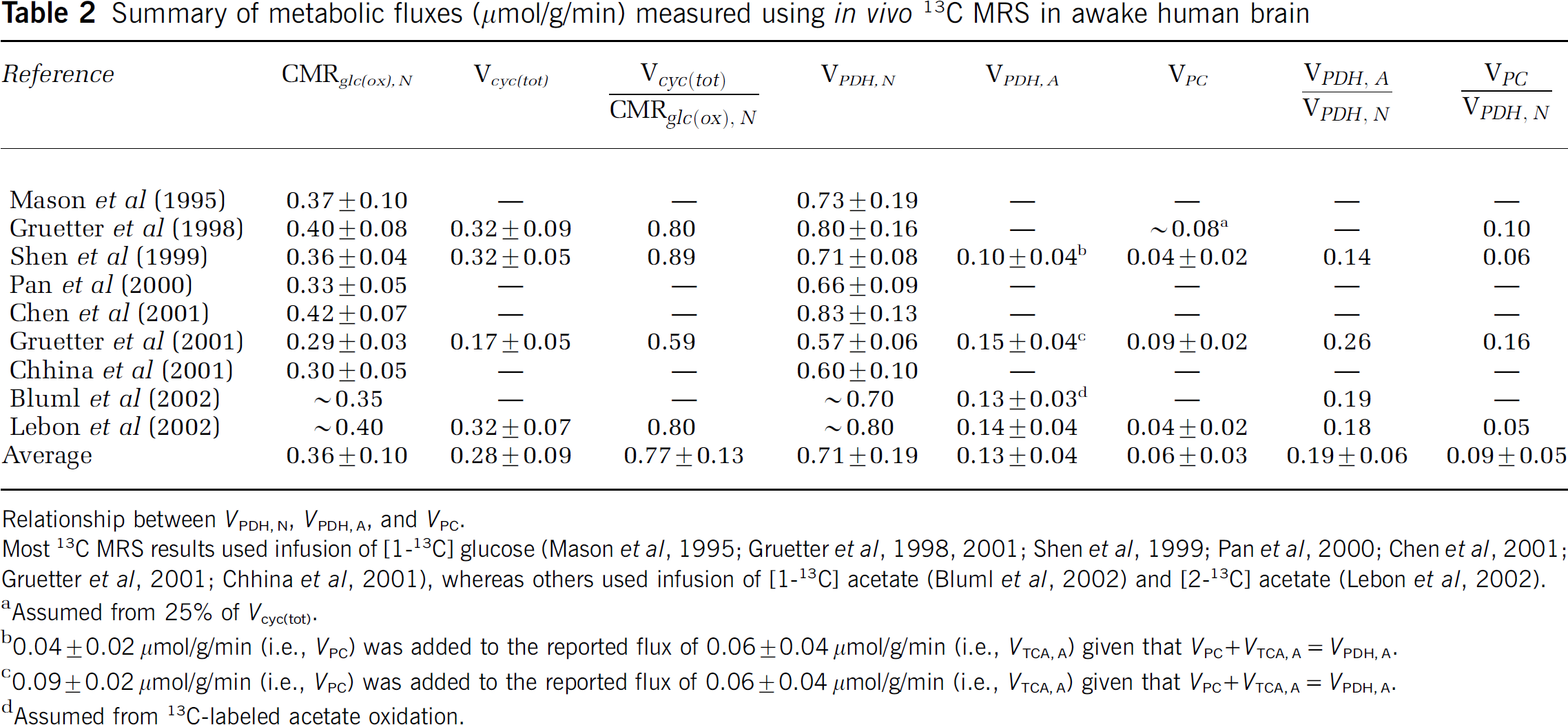
Relationship between VPDH, N, VPDH, A, and VPC.
Most 13C MRS results used infusion of [1-13C] glucose (Mason et al, 1995; Gruetter et al, 1998, 2001; Shen et al, 1999; Pan et al, 2000; Chen et al, 2001; Gruetter et al, 2001; Chhina et al, 2001), whereas others used infusion of [1-13C] acetate (Bluml et al, 2002) and [2-13C] acetate (Lebon et al, 2002).
Assumed from 25% of Vcyc(tot).
0.04 ± 0.02 μmol/g/min (i.e., VPC) was added to the reported flux of 0.06 ± 0.04 μmol/g/min (i.e., VTCA, A) given that VPC ± VTCA, A= VPDH, A.
0.09 ± 0.02 μmol/g/min (i.e., VPC) was added to the reported flux of 0.06 ± 0.04 μmol/g/min (i.e., VTCA, A) given that VPC ² VTCA, A= VPDH, A.
Assumed from 13C-labeled acetate oxidation.
Contributions of GABAergic and glutamatergic fluxes (μmol/g/min) in anesthetized rat cerebral cortex
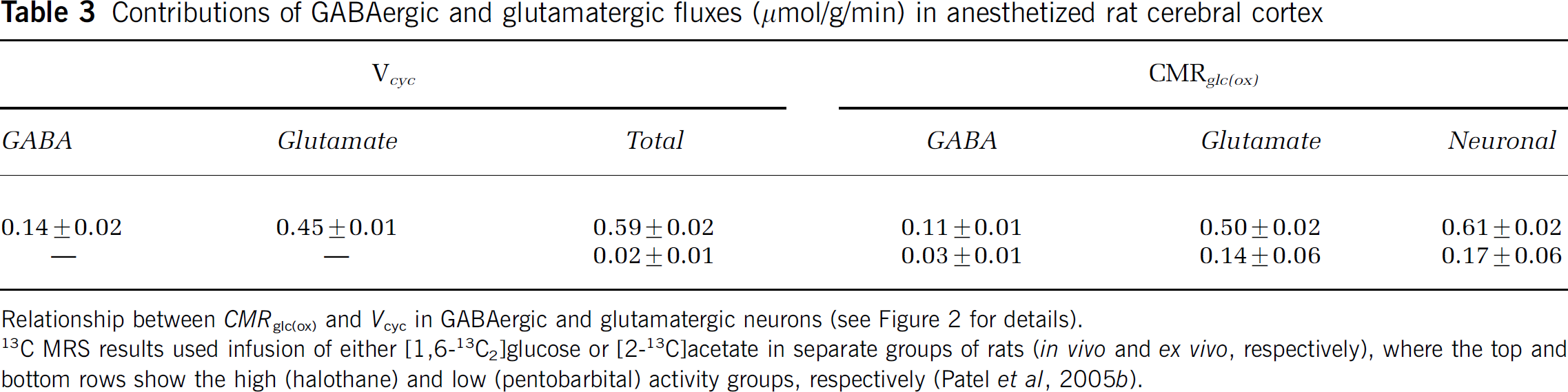
Relationship between CMRglc(ox) and Vcyc in GABAergic and glutamatergic neurons (see Figure 2 for details).
13 C MRS results used infusion of either [1,6-13C2]glucose or [2-13C]acetate in separate groups of rats (in vivo and ex vivo, respectively), where the top and bottom rows show the high (halothane) and low (pentobarbital) activity groups, respectively (Patel et al, 2005b).
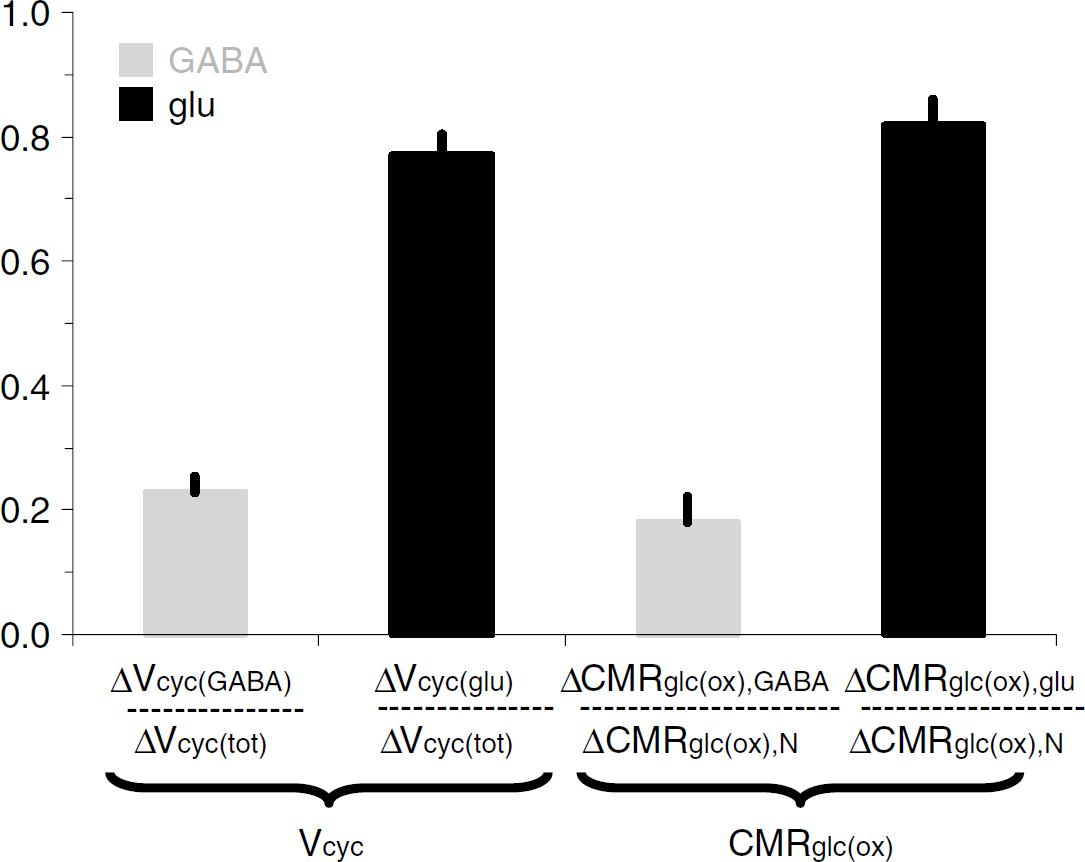
GABAergic versus glutamatergic neurotransmission and neuroenergetics. Summary of results from Patel et al (2005b) where changes in neurotransmission and neuroenergetics are shown in a normalized manner for glutamatergic (dark) and GABAergic (light) neurons.
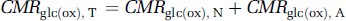
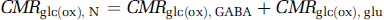
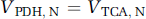
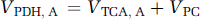
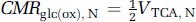
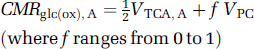
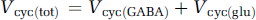
where CMRglc(ox), T is the cerebral metabolic rate of total glucose oxidation, CMRglc(ox), N the cerebral metabolic rate of neuronal glucose oxidation, CMRglc(ox), glu the cerebral metabolic rate of glucose oxidation in glutamatergic neurons, CMRglc(ox), GABA the cerebral metabolic rate of glucose oxidation in GABAergic neurons, CMRglc(ox), A the cerebral metabolic rate of astrocytic glucose oxidation, VPDH, N the neuronal pyruvate dehydrogenase flux, VTCA, N the neuronal tricarboxylic acid cycle flux, VPDH, A the astrocytic pyruvate dehydrogenase flux, VTCA, A the astrocytic tricarboxylic acid cycle flux, VPC the astrocytic pyruvate carboxylase flux, Vcyc(tot) the rate of total neurotransmitter cycling, Vcyc(glu) the rate of neurotransmitter cycling for glutamatergic neurons, and Vcyc(GABA) the rate of neurotransmitter cycling for GABAergic neurons.
Relationship Between CMRglc(ox), N and Vcyc(tot)
Figure 1 and Table 1 list 13C MRS studies of rat brain where values of CMRglc(ox), N and Vcyc(tot) were reported. As shown from the plot of CMRglc(ox), N versus Vcyc(tot), when more recent data are added the slope and intercept of the plot differ insignificantly from the original data. Thus the 1:1 stoichiometry suggested from the increments of CMRglc(ox), N and Vcyc(tot) (Sibson et al, 1998) is supported by subsequent results (Choi et al, 2002; de Graaf et al, 2004; Patel et al, 2004a; Oz et al, 2004).
Glial Energetics
A source of some confusion in comparing rates of glial and neuronal glucose oxidation has been the inclusion of the glial pyruvate carboxylase flux as part of glial glucose oxidation by some authors (e.g., Gruetter et al, 2001; Choi et al, 2002; Oz et al, 2004) and not by others (e.g., Sibson et al, 2001; Lebon et al, 2002; Patel et al, 2005a). The differences in definition affect the calculation of oxidative ATP production in glia from the pyruvate dehydrogenase and pyruvate carboxylase pathways. For the purposes of this paper, the oxidative ATP production pathways in astrocytes—pyruvate dehydrogenase (VPDH, A) and pyruvate carboxylase (VPC)—are treated separately, because these glial fluxes can now be measured independently of neuronal pyruvate dehydrogenase (VPDH, N) using 13C or 14C tracers (Table 2).
The measurement of glial energetics is more difficult than for neurons using 13C or 14C tracers due mainly to the much smaller glutamate pool in glia. Recent 13C studies have measured glial energetics using one of two strategies (Table 2). In the first method, the different pools in the glial tricarboxylic acid cycle are selectively labeled with either [2-13C]glucose or [2-13C]acetate. Acetate is selectively metabolized in glia and thus provides a specific probe of glial metabolism. The second approach has been to measure the time courses of multiple positions of aspartate, glutamate, and glutamine during a [1-13C]glucose infusion and access glial metabolism from differences in the pattern of labeling produced by pyruvate carboxylase. In the awake human brain, both approaches have yielded similar relative values for cortical glial metabolism (Table 2), that is, approximately 20% of neuronal oxidative metabolism (Shen et al, 1999; Gruetter et al, 2001; Lebon et al, 2002; Bluml et al, 2002). Results from different laboratories reported for rodents using both 13C- and 14C-labeled isotopes (Sibson et al, 2001; Oz et al, 2004; Cruz et al, 2005; Patel et al, 2005a) indicate that glial oxidation comprises at least 30% of the total under resting conditions.
GABAergic Contributions
Inhibitory GABAergic neurons consume energy (Garfinkel, 1970; Jueptner and Weiller, 1995; Sokoloff, 1999), just as other neurons do; however, their quantitative contribution to total oxidative energy use has been unclear. Distinctions between glutamatergic and GABAergic fluxes require measurement of 13C turnover from glucose and acetate into glutamate, glutamine, and GABA (Pfeuffer et al, 1999; Choi et al, 2000; Henry et al, 2000; Patel et al, 2005b; Yang et al, 2005). A 13C MRS study combining [1-13C]glucose and [2-13C]acetate infusion with three-compartment metabolic modeling (Patel et al, 2005b) showed that total neurotransmitter cycling (Vcyc(tot)) could be resolved into separate glutamatergic (Vcyc(glu)) and GABAergic (Vcyc(GABA)) components. Similarly, neuronal glucose oxidation (CMRglc(ox), N) could also be separated into glutamatergic (CMRglc(ox), glu) and GABAergic (CMRglc(ox), GABA) components. The study found that both fluxes, cycling and oxidation, increased with activity and that GABAergic neurons account for approximately 20% of the total flux in the mildly anaesthetized rat (Figure 2; Table 3).
Glutamate Oxidation Versus Glutamate Recycling
Glutamate can undergo complete oxidation in the glia. In this pathway, glutamate taken up by the glia is converted, through glutamate dehydrogenase or transamination, to α-ketoglutarate. The glial tricarboxylic acid cycle can further metabolize α-ketoglutarate to malate and then to pyruvate by malic enzyme; complete oxidation of the pyruvate then follows in the usual manner (Hertz and Robinson, 1999; Lieth et al, 2001). Subsequent loss of glutamate to the system by oxidation must be replaced by anaplerosis to maintain homeostasis, starting with the conversion of pyruvate to oxaloacetate by pyruvate carboxylase. Glial α-ketoglutarate (or glutamate) formed by this pathway and further tricarboxylic acid cycle reactions will then be cycled back to the neuron after conversion to glutamine. The theoretical and experimental evidence for this pathway have been reviewed in detail (Hertz and Robinson, 1999; Lebon et al, 2002). For the purpose of this article, we point out that, provided the glutamate carbons are recycled to the neurons as glutamine, the presence of this pathway does not affect the measurements of either glial or neuronal glucose oxidation or glutamate-glutamine cycling.
Standardization of Different Fluxes
CMRglc(ox), N (or VPDH, N) was measured in all recent 13C MRS studies (Tables 1 to 3) so that other fluxes can be expressed as fractions of CMRglc(ox), N. The relative experimental values of total neurotransmitter cycling flux, as well as the GABAergic and glutamatergic cycling fractions, are as follows:
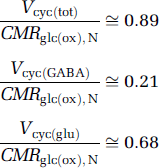
The data considered were measured in anesthetized or awake rats, at moderate to high activity levels (Sibson et al, 1998; de Graaf et al, 2004; Patel et al, 2004a, 2005a, b; Oz et al, 2004) so as to avoid errors arising from the difference of small numbers during extremely low fluxes. These ratios are activity dependent because of the nonzero intercept in Figure 1. The ratios for neuronal glucose oxidation in GABAergic and glutamatergic neurons were similarly derived (Patel et al, 2005b) for moderate activity level (halothane):
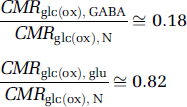
The metabolic pathways in the glia via pyruvate carboxylase and pyruvate dehydrogenase can be similarly calculated from Table 2:
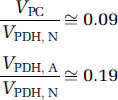
The standardized relationships are shown in Figure 3. In equation (6), the maximum value of f is 1, which occurs when all carbons entering the tricarboxylic acid cycle via pyruvate carboxylase are oxidized in glia. If all pyruvate carboxylase flux, however, is used to synthesize glutamine for export (e.g., ammonia detoxification), then f is ~0.4 because of incomplete tricarboxylic acid cycling (i.e., oxaloacetate → α-ketoglutarate → glutamine).
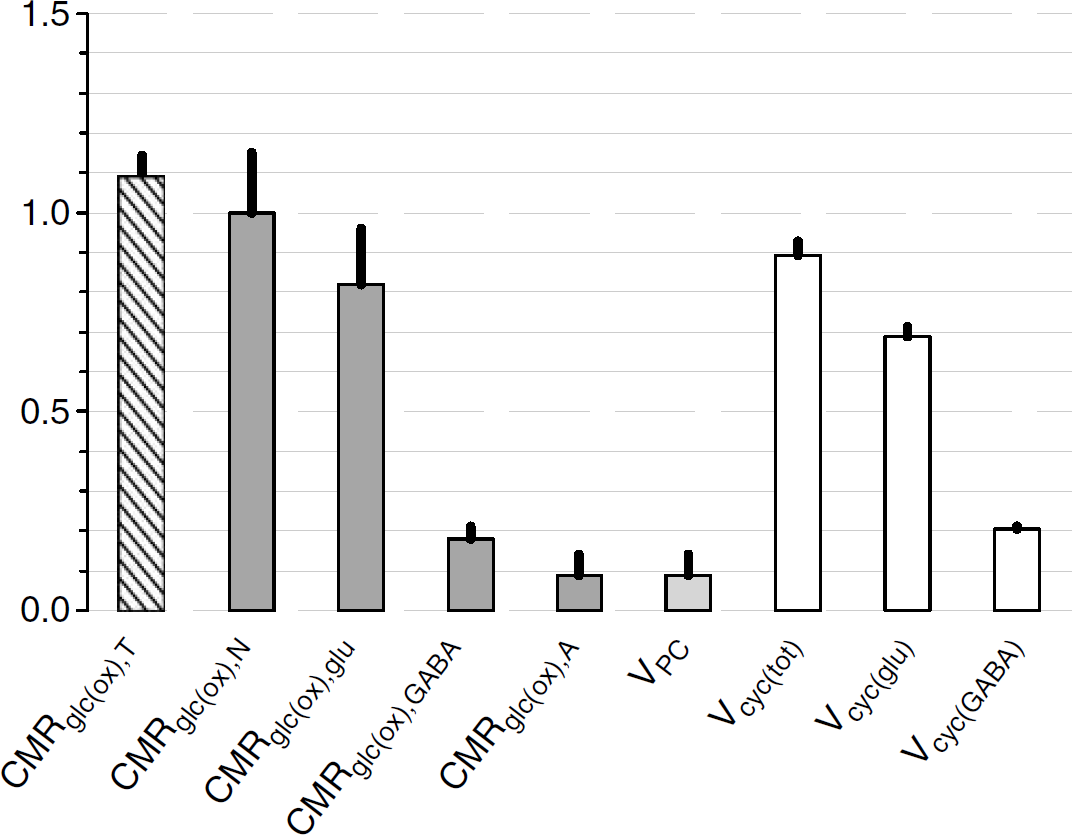
The 13C MRS results (Tables 1 to 3) are plotted in Figure 3 relative to CMRglc(ox), N. In this case, glucose oxidation in glutamatergic and GABAergic neurons is ~82% and ~18%, respectively, of CMRglc(ox), N. In comparison, CMRglc(ox), A is ~9% of CMRglc(ox), N, which means that CMRglc(ox), T is ~109%. Therefore, astrocytes are responsible for ~ 8% of total glucose oxidation (= 0.09/1.09) and neurons consume the rest. These values were calculated with f = 0.4 in equation (6) and with f = 1.0 these values would increase by ~5%. The neurotransmitter fluxes show that Vcyc(tot) is ~89% of CMRglc(ox), N, with contributions for glutamatergic and GABAergic systems of ~68% and ~21%, respectively.
Revised Model of Glucose Uptake/Metabolism and Lactate Exchange/Efflux
In the prior model (Magistretti et al, 1999), all glucose was considered to be metabolized to lactate (pyruvate) in the glia and then exported to and subsequently oxidized in neurons to support neuronal firing. The carbon skeletons all diffused to neurons as lactate (pyruvate). It should be emphasized, however, that glial-selective glucose uptake was primarily based on the anatomic connection between the microvasculature and astrocytes (Tsacopoulos and Magistretti, 1996) and lacked direct experimental evidence (Rothman et al, 2001; Chih et al, 2001; Gjedde et al, 2002). The earlier model neglected the findings that glucose transporters are ubiquitous (Maher et al, 1994; Gerhart et al, 1992; Vannucci et al, 1997) and that both neurons and glia in culture readily metabolize glucose (Schousboe et al, 1993; Hertz and Robinson, 1999; Itoh et al, 2003; Cruz et al, 2005).
The comprehensive 13C MRS results (Figure 3) provide parameter estimates that formed the basis of an amendment to the earlier model. The revised model (Figure 4) presents values of fluxes that represent a control condition (e.g., moderately anesthetized to awake resting), and includes the contributions of GABAergic and glutamatergic neurons (Figure 2). The novel features of the revised model (Figure 4) are derived from the measured values of glial oxidation. The new conclusions are that glucose uptake and oxidation occur in both glia and neurons, and that much but not all of glial lactate (pyruvate) is transported to neurons, with a slight excess being lost to the circulation. The fundamental assumption is that glycolytic ATP produced in glia is needed to support ion pumping and glutamine synthesis is retained in this model. The details of these revisions are discussed below.
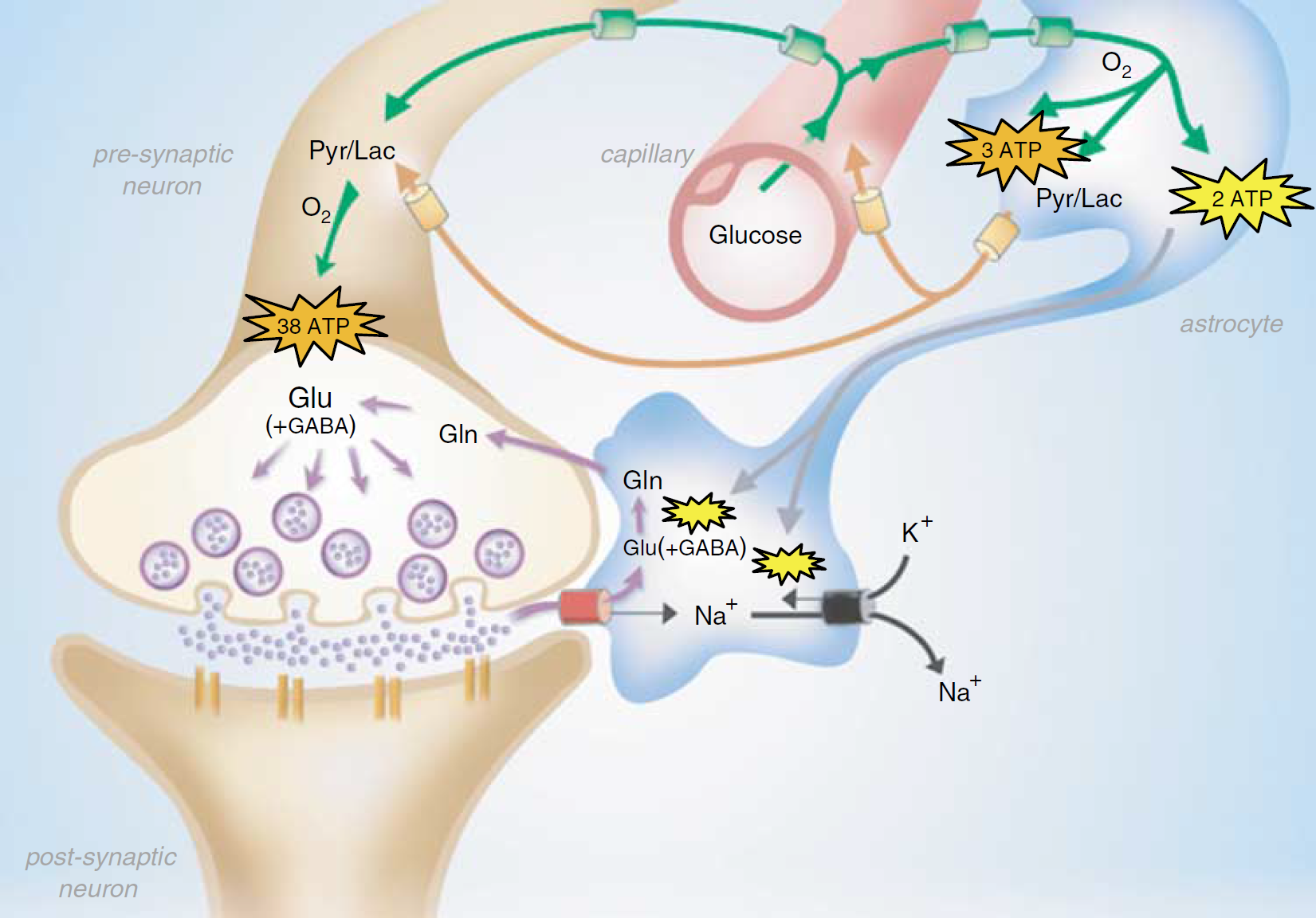
A revised coupling between synaptic activity and glucose metabolism. Glucose uptake (green) is now ubiquitous. Astrocytic lactate flow (orange) to neurons is now less, with a small amount being effluxed to blood. ATP is produced oxidatively (orange) in neurons and astrocytes. ATP produced by glycolysis (yellow) fuels ion pumping and glutamine synthesis in astrocytes. Abbreviations: Glu, glutamate; Gln, glutamine; Pyr, pyruvate; Lac, lactate. See Figure 5 for details.
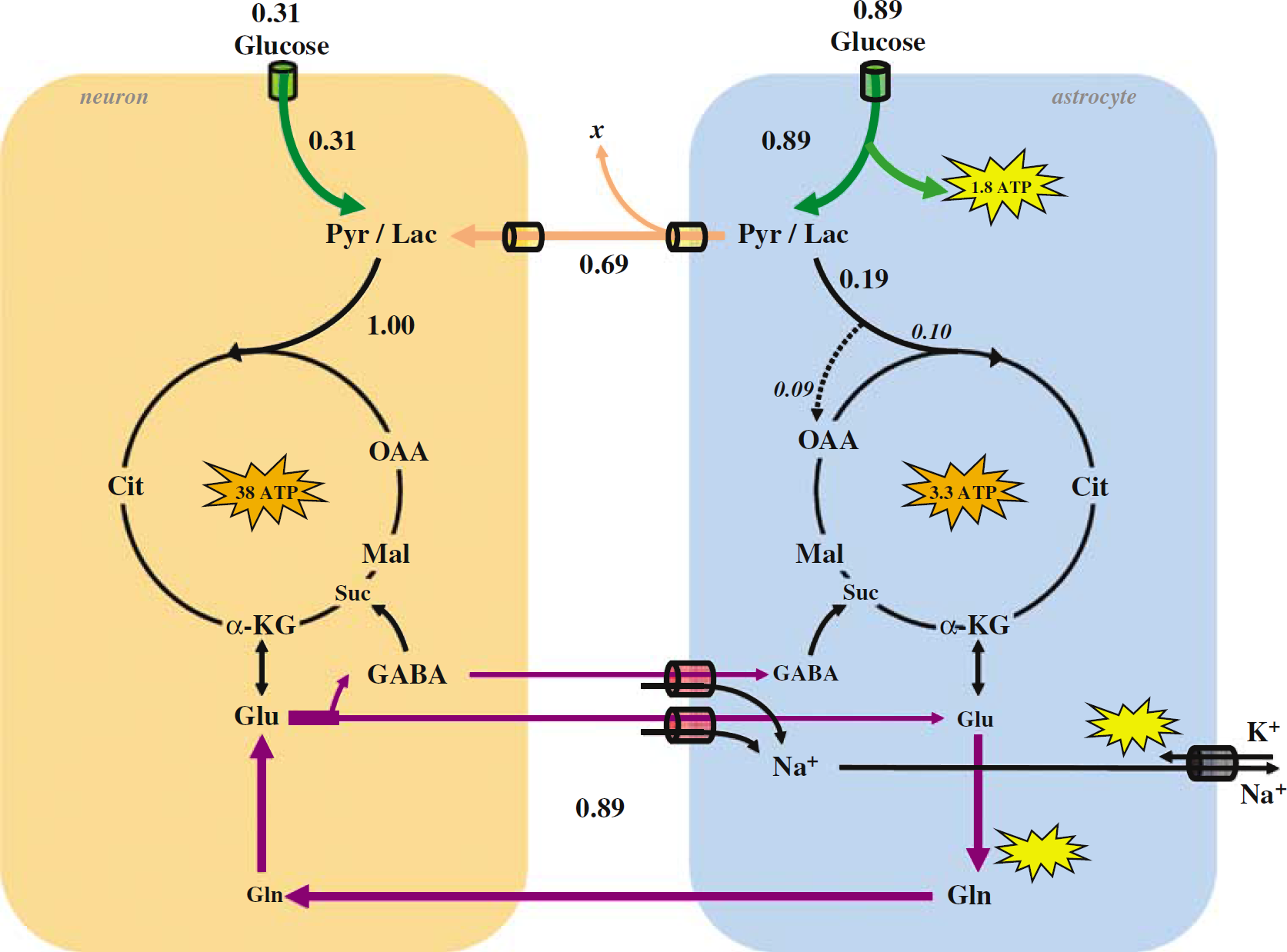
The revised model for coupling of brain glucose metabolism and neuronal activity. The main observables are CMRglc(ox), N and Vcyc(tot). All fluxes are scaled to CMRglc(ox), N of 1.00 μmol/g/min from the fluxes shown in Figure 3 and listed in Tables 1 to 3. Glial uptake of ~0.89 glucose equivalents (green) produces ~1.8 glycolytic ATP (yellow) to fuel ion pumping and glutamine synthesis. The glial tricarboxylic acid cycle consumes ~0.19 glucose equivalents in total, where pyruvate carboxylase flux itself provides ~ 0.09 glucose equivalents, yielding ~3.3 oxidative ATP (orange) in astrocytes. The remaining glial lactate (~ 0.70) is transported to neurons (— 0.69) and blood (x ≈ 0.01). For ~1.00 glucose equivalents to be oxidized in neurons, an additional ~0.31 glucose uptake is needed in neurons, yielding ~38 oxidative ATP (orange) in neurons. This process in neurons and glia results in the cycling of ~0.89 glucose equivalents of neurotransmitter glutamate (~ 82%) and GABA (~ 18%).
To illustrate specific values of the different fluxes, CMRglc(ox), N is set to 1.00 μmol/g/min in Figure 5, thereby generating ~38 ATPs from glucose oxidation in neurons and includes contributions of both glutamatergic (~31 ATPs) and GABAergic (~7 ATPs) neurons. The data in Figure 3 show that a CMRglc(ox), N of 100 μmol/g/min is coupled with Vcyc(tot) of ~ 0.89 μmol/g/min. The other fluxes in Figure 5 are also scaled from the data in Figure 3. Glial uptake of both glutamate (~0.68 μmol/g/min) and GABA (~0.21 μmol/g/min) also transports Na+, but with different stoichiometries.
Glutamate uptake into glia cotransports three Na± ions and one H+ ion, and one K+ ion is moved out of the cell (Zerangue and Kavanaugh, 1996; Levy et al, 1998), whereas two Na+ ions and one Cl−- ion are cotransported with GABA into glia (Kavanaugh et al, 1992). Thus, glial GABA uptake moves less Na+ per transport cycle than does glial glutamate. However, glutamine efflux from glia to the extracellular space is also coupled to Na+ ion transport, with one Na+ ion leaving and one H+ ion entering per molecule of glutamine exiting the cell (Chaudhry et al, 2002). Thus, glial glutamate uptake coupled to glutamine outflow would lead to net entry of two Na+ and two H+ ions, whereas a net entry of one Na+ and one H+ would be expected for glial GABA uptake. If we consider the removal of the H+ ions in exchange for Na+ by the Na+/H+ exchanger (to maintain glial H+ homeostasis), which is very active in glia (Schlue and Deitmer, 1988; Klee et al, 1999), then glial uptake of one molecule of glutamate or GABA coupled to efflux of one molecule of glutamine would lead to net uptake of three Na+ ions or two Na+ ions, respectively. Thus, glial cycling of GABA to glutamine would be 2/3 as active as glial cycling of glutamate with regard to Na+ activation of the plasma membrane Na+/K+ ATPase (Chatton et al, 2003).
For the measured cycling fluxes of glutamate and GABA and the respective stoichiometries given above, the amount of Na+ entering the glia would be ~2.7 μmol/g/min (= 0.68 × 4) and ~0.4 μmol/g/min (= 0.21 × 2) respectively, or ~3.1 μmol/g/min because of their combined cycling. With the 3:1 stoichiometry of Na+ ATP for the ATP-dependent Na+ pump, ~1 μmol/g/min of ATP production (= 3.1/3) would be sufficient to reestablish the Na+ ion gradient. However, an additional ~0.89 μmol/g/min of ATP would still be required for synthesis of glutamine, by recalling that Vcyc(tot) is ~ 0.89 μmol/g/min. Thus, combined cycling of glutamate and GABA would require a total of ~1.89 μmol/g/min of ATP production, which could be supplied by ~0.94 μmol/g/min of glucose undergoing glycolysis in glia. In Figure 5, the glial glycolytic ATP production (~1.78 μmol/g/min) and glucose uptake (~0.89 μmol/g/min) rates, which are derived from 13C MRS measurements, are similar to the theoretical Na+ :ATP stoichiometry for coupling glial glycolysis to neurotransmitter cycling. This more detailed analysis of Na + ion pumping and glutamine synthesis is consistent with fluxes shown in Figure 5.
Two pathways of glucose metabolism in glia have now been included in the revised model (Figure 3). Pyruvate enters the glial tricarboxylic acid cycle via pyruvate dehydrogenase and pyruvate carboxylase. These two glial oxidative pathways, however, have different ATP yields. In total, ~ 0.19 μmol/g/min glucose is oxidized in the glial tricarboxylic acid cycle to produce ~ 3.3 ATP in glia (Figure 5). The remaining fraction (≈ 0.89-0.19) consisting of lactate (pyruvate) is assumed to leave glia and enter neurons (~ 0.61 μmol/g/min) or blood (~ x μmol/g/min), which is consistent with the previous model (Magistretti et al, 1999). The brain lactate level is related to activity (Prichard et al, 1991; Hu and Wilson, 1997; Shulman et al, 2001a, b; Patel et al, 2004a) and the higher intracellular than extracellular concentration drives the passive transport of lactate out of the tissue Quel, 1997; Bonen, 2000; Deitmer, 2002; Brooks, 2002). This lactate efflux has been proposed to be responsible for changes in the oxygen to glucose index (OGI) during activation (Rothman etc al, 2001; Shulman et al, 2001a, b ). An additional ~ 0.39 μmol/g/min of glucose is phosphorylated and metabolized in neurons, thereby reaching the measured CMRglc(ox), N of 1.00 μmol/g/min.
Discussion
A large set of in vivo 13C MRS flux measurements now exists for glucose oxidation in neurons and glia and neurotransmitter cycling (Figures 1 to 3). These results have been consolidated into a revised model (Figure 4), with testable values (Figure 5) and which is based upon the 1:1 relationship measured between ΔCMRglc(ox), N and ΔVcyc(tot).
The separation of the fluxes of cortical glutamatergic and GABAergic neurons in the revised model (Figure 5) is an advance compared with the prior model (Magistretti et al, 1999), because earlier 13C MRS results did not include 13C label flow into GABA (e.g., Rothman et al, 1999, 2001). An important finding indicated from 13C MRS studies for glutamatergic and GABAergic systems (Patel et al, 2004b, 2005b) is that fluxes through both neuron types change commensurately with activity, where the GABAergic system may account for as much as ~ 18% of total neuronal glucose oxidation to support ~ 23% of total neurotransmitter (i.e., glutamate plus GABA) cycling. Because glutamate and GABA together constitute ~ 90% of cortical neurons (Nicholls, 1989, 1993; Peters and Jones, 1984; Beaulieu and Colonnier, 1985), current 13C MRS studies measure the dominant activities and their associated energetic costs. These types of 13C MRS measurements, therefore, have implications for the interpretation of neuroimaging data (Shulman et al, 2002, 2004).
The inclusion of glial oxidative fluxes into the revised model (Figure 5) rectifies another simplification made previously (Magistretti et al, 1999). Quantitative 13C MRS data, with reasonable error limits, are now available for these pathways (Table 2; Figure 3) and studies suggest that these pathways may contribute to functional activity (Oz et al, 2004; Cruz et al, 2005). The carbon source for glial oxidation is assumed to be pyruvate produced by glial glycolysis. In the revised model (Figure 5), as before we assume that glial glycolysis provides the ATP needed for restoration of Na+ gradients due to neurotransmitter cycling and glutamine synthesis.
There are, of course, several assumptions in the revised model, which can be tested. Because of glial oxidation of pyruvate, ~30% less glial lactate (pyruvate) would be transferred to the neurons in this case (Figure 5). Thus, to achieve the full oxidative metabolism in neurons required by the 1:1 stoichiometry, ~31% of pyruvate generation would come from neuronal glycolysis. The partitioned glucose uptake is ~26% by neurons and ~74% by glia.
The experimental relationship between ΔCMRglc(ox), N and ΔVcyc(tot) (Figure 1) requires that any model describing these fluxes must present a mechanism consistent with the 1:1 stoichiometry between neuronal energy consumption and neurotransmitter cycling. Although the answer was implicit in the prior model (Magistretti et al, 1999), in view of the new data summarized above (Figures 1 to 3) and the new proposal with its underlying assumptions (Figures 4 and 5), the solution can now be explored far more explicitly. The basic postulate is that only the glycolytic ATP (in glia) is produced rapidly enough to clear glutamate from the extracellular space in the time needed to prepare the synapse for the next synaptic event. The time needed for synaptic clearance of glutamate was shown by Dzubay and Jahr (1996) to be on the order of a few milliseconds: one rapid glycolytic ATP is required to pump the Na+/K+ ions and one to energize glutamine synthesis. If two glycolytic ATPs (in glia) were not needed to clear and cycle one neurotransmitter molecule, then the glucose uptake by glia would have no fixed relationship to neurotransmission. The assumption that rapid glycolytic ATP in glia is needed for glutamate clearance agrees with the 1:1 stoichiometry between CMRglc(ox), N and V cyc(tot), which is an observable. The ‘fast’ ATP (in glia) could be restored by the buffering action of phosphocreatine, which is known to be very rapid (Chen et al, 1997; Chung et al, 1998). However, phosphocreatine must be resynthesized, which uses ATP, before the synapse is set for the next pulse of neurotransmitter release. A possible role for glial glycogenolysis in the production of rapid glycolytic ATP was discussed earlier (Shulman et al, 2001a).
The hypothesis that reestablishing ion gradients and replenishing neurotransmitter pools must be powered by glycolytically produced ATP in glia finds some support in previous explorations, but remains an assumption to be tested experimentally. Increased lactate concentration under conditions of increased brain activity (Prichard et al, 1991) is generally consistent with this idea. Numerous studies using blood oxygenation-dependent functional imaging have shown that during local activation the tissue actually experiences an increased oxygen tension (Hedera et al, 1995; Ances et al, 2001; Hyder et al, 2002; Trübel et al, 2006). We have explained the lactate increase in the presence of plentiful oxygen by suggesting that the rapid energy requirements for ATP cannot be met by normal oxidative metabolism, which proceeds more slowly (Shulman et al, 2001b). Hence, glial glucose consumption rates are set by the rate of rapid anerobic (glycolytic) energy delivery needed to power neurotransmitter clearance rather than by the energetic need of neuronal firing and neurotransmitter release. In this event, the extra glucose consumed beyond the glial oxidative requirement leaves as lactate. This idea has received considerable support in studies of the brain where the decrease in the OGI during increased activity is consistent with the observed increase in lactate (Siesjö, 1978; Gjedde et al, 2002; Shulman et al, 2002). The higher level of lactate observed during activation would drive efflux of lactate from brain to blood (e.g., Juel, 1997) and this ‘lactate shuttle’ (e.g., Brooks, 2002) shows an apparent, but not actual, support of neuronal firing by anerobic (glycolytic) energy. However, in the revised model (Figure 5), both pyruvate carboxylase flux and lactate efflux decrease the OGI and their contributions can be determined from data.
Because evidence for the need of anerobic (glycolytic) ATP in glia is hypothetical, this suggestion must be judged by its linked predictions, of which several can be found in the proposed model (Figure 5). First, because the relative glucose uptake by neurons and glia depends on this assumption, experiments could test this prediction by localizing glucose uptake for these two cell types (Itoh et al, 2004). Second, because changes in lactate have only been measured in a limited number of cases (Prichard et al, 1991; Sappey-Marinier et al, 1992; Frahm et al, 1996; Patel et al, 2004a), the ability to relate changes in brain lactate concentrations, which is the driving force for the efflux into blood, to the energetic demands on stimulation should be examined more closely (Kuwabara et al, 1995; Mangia et al, 2003; Urrila et al, 2003). Third, because the relation between the rates of total glucose uptake and of the amounts oxidized, as reflected by the OGI, also depends on this assumption, more accurate measurements of total glucose oxidation (Chen et al, 2001; Chhina et al, 2001) and total glucose uptake (Merboldt et al, 1992; Chen et al, 1993) and kinetics (Choi et al, 2001; de Graaf et al, 2001) are needed. Of course, these complex 13C turnover data require careful compartmental modeling (Gruetter et al, 2001; Mason and Rothman, 2004). In addition to these particular tests of the assumptions, other measurements of cellular and subcellular thermodynamics and kinetics are needed to explore the important relations between neuronal/glial energetics and neurotransmission.
Conclusion
The present review provides an argument for the usefulness of models in brain research, an approach that has been questioned recently (Kimelberg, 2004). The present revised version (Figure 5) of the earlier model (Magistretti et al, 1999) takes into account the wealth of data that have been provided by recent research from different laboratories. As such, it retains the experimental results formerly used, all of which have been strengthened by subsequent measurements, and now includes new insights from measurements formerly not available. In its comprehensive form, the model leads readily to certain crucial questions, which can be tested experimentally: for example, the possibility of direct neuronal uptake of glucose and the extent that glial oxidative processes contribute to functional energetics. The relations between energy and work, which played an important role in understanding muscle physiology (Shulman and Rothman, 2001a, b ), are beginning to display a similar centrality in neurophysiology (Shulman et al, 2002, 2004). Brain imaging methods localize and measure the neurophysiologic parameters of glucose and oxygen consumption, as well as the accompanying cerebral blood flow, with increasing sensitivity and spatial resolution. Furthermore, cellular localization of metabolites and enzymes provide the basis for cell-specific fluxes and energetics. Noninvasive measurements of energy consumption, mainly from glucose oxidation, have been related to neurotransmitter fluxes, in both rodents and humans, and thereby link these imaging results to neuronal activity (Hyder et al, 2002). These relations are based on the rigorous laws of thermodynamics and as such provide a sure foundation for further interpretation and experimentation. Experimental results from a variety of laboratories are converging and, as reviewed here, provide the basis for an understanding of how cerebral energy supports the work of neuronal activity.
Footnotes
Acknowledgements
We thank Drs Robin de Graaf, Graeme Mason, Kamil Ugurbil, WeiChen and Golam Chowdhury for helpful discussions and extend appreciation for continued support of scientists and engineers at DBS (bioimaging.yale.edu), MRRC (mrrc.yale.edu), and QNMR (![]() ).
).
