Abstract
Acute ischemic stroke is a leading cause of mortality and disability in the elderly. Age is the most important nonmodifiable risk factor for stroke, yet many preclinical models continue to examine only young male animals. It remains unclear how experimental stroke outcomes change with aging and with biologic sex. If sex differences are present, it is not known whether these reflect an intrinsic differing sensitivity to stroke or are secondary to the loss of estrogen with aging. We subjected both young and aging mice of both sexes to middle cerebral artery occlusion (MCAO). Young female mice had smaller strokes compared with age-matched males, an effect that was reversed by ovariectomy. Stroke damage increased with aging in female mice, whereas male mice had decreased damage after MCAO. Blood–brain barrier (BBB) permeability changes are correlated with infarct size. However, aging mice had significantly less edema formation, an effect that was independent of sex and histologic damage. Differences in the cellular response to stroke occur across the life span in both male and female mice. These differences need to be considered when developing relevant therapies for stroke patients, the majority of whom are elderly.
Introduction
Acute ischemic stroke is a disease that primarily affects the elderly, and the strongest independent risk factor for stroke is age (Rojas et al, 2007). With increasing life expectancy, the world is facing a rapid expansion in its elderly population, and old subjects will soon constitute the majority of stroke victims (Rojas et al, 2007). The prevalence of ischemic stroke increases from 11% to 43% as patients' age increases from 55 to 85 years (Howard et al, 1998). Owing to its high mortality and morbidity in elderly patients, stroke is a major health-care problem worldwide, exerting an enormous financial toll and using vast medical resources. Despite clear evidence that stroke is a disease of the elderly, the majority of preclinical studies continue to use young male animals. As the most promising neuroprotective agents have failed when moved to clinical trial, stroke researchers need to address possible flaws in our experimental models if we hope to develop efficacious therapies. Clearly, age is an important factor in determining how the brain responds to stroke. For example, edema is more robust in the young brain, suggesting that the cellular response to ischemia may differ across the life span (Desbordes and Cohadon, 1987; Wagner and Lutsep, 2005).
Several earlier studies have shown that histologic stroke damage may be surprisingly less in aging male rodents, despite higher mortality and poorer functional outcomes compared with that in young males (Shapira et al, 2002; Wang et al, 2003). Although the majority of strokes now occurs in elderly women (Rosamond et al, 2008), there is a paucity of information on the response to stroke in aging female rodents, and no earlier study has examined stroke outcomes in aging female mice.
It is becoming increasingly recognized that stroke is a sexually dimorphic disease, both experimentally and clinically, although these sex differences are still poorly understood. Most international databases consistently show that women enjoy a lower incidence of stroke relative to men until advanced age (Broderick et al, 1998), yet functional outcomes are worse in women than in men after stroke (Glader et al, 2003), with higher associated disability and mortality presumably because of, in part, the advanced age at which strokes occur in women. Sex differences in stroke epidemiology have been attributed to the vascular and neuroprotective effects of premenopausal estrogen (17β-estradiol or E2) exposure. However, recent clinical trials examining the effect of hormone replacement have shown a surprising increase in vascular risk in hormone-treated women (Prentice et al, 2006). A major criticism of these trials was that E2 treatment was begun years after menopause. Recent animal studies have shown that a sustained period of hypoestrogenicity before the initiation of hormone replacement ameliorates the protective effects of E2 in female mice; however, only young animals subjected to surgical menopause (through ovariectomy) were examined (5 to 6 months) (Suzuki et al, 2007). Whether E2 supplementation can reduce infarction in aging female mice is not known.
The overall goal of this study was to investigate how age and sex affect the ischemic stroke outcome in male and female mice. As clinical data suggest that older women have worse outcomes after stroke and that the loss of E2 may contribute to these differences, we hypothesized that aging female mice would have larger strokes than younger female mice and that this increase in injury could still be ameliorated with peri-menopausal E2 replacement. Effects on histologic and functional outcomes, blood–brain barrier (BBB) permeability, and edema formation were assessed in mice of both sexes.
Materials and methods
Animals
C57BL/6 mice were purchased from Charles River Laboratories (Willimantic, CT, USA). All experiments were performed according to the National Institutes of Health guidelines for the care and use of animals in research, and under protocols approved by the University of Connecticut Animal Care and Use Committee. Both young mice (9 to 12 weeks; 21 to 25 g) and aging mice (16 months, males: 35 to 50 g, females: 26 to 38 g) of both sexes were used.
Ischemic Model
Focal transient cerebral ischemia was induced by middle cerebral artery occlusion (MCAO) (0.21mm silicone-coated suture) for 90 mins followed by reperfusion as described earlier (McCullough et al, 2005). In aging mice, a larger 0.23-mm silicone-coated suture was used to achieve occlusion. In sham animals, the suture was not advanced into the MCA. Cerebral blood flow (CBF) was measured by laser Doppler flowmetry (LDF, Moor Instruments Ltd, Wilmington, DE, USA) during the surgery as described earlier (McCullough et al, 2005). Only the mice in which CBF in the MCA area showed a sharp decrease of over 85% of control immediately after MCA occlusion were included.
Neurologic deficit was confirmed, and scored as follows: (0) no deficit; (1) forelimb weakness and torso turning to the ipsilateral side when held by tail; (2) circling to the affected side; (3) unable to bear weight on the affected side; and (4) no spontaneous locomotor activity or barrel rolling. Monitoring of physiologic variables was performed in companion cohorts for all groups before MCAO and 60 mins after reperfusion as described earlier (McCullough et al, 2005).
Terminal Histopathology
At 24h after stroke, the mice were killed and the brains were removed and cut into 52-mm slices. The slices were stained with 1.5%2,3,5-triphenyltetrazolium chloride (TTC) at 37°C for 10mins. The stained slices were fixed with 4% formalin, images were digitalized, and the infarct volumes (percentage of (%) contralateral hemispheric structure (to correct the different brain sizes in male and female mice and to correct edema)) were analyzed using a computer software (Sigmascan Pro5) as described earlier (McCullough et al, 2005). All infarct analysis was performed by an investigator blinded to the treatment group.
Evans Blue Extravasation
The integrity of the BBB was investigated using Evans blue (EB) extravasation, according to Bahcekapili et al (2007). One hour before the mice were killed, 4 mL/kg of 2% EB (Sigma) in normal saline was injected intravenously. For quantitative measurements, brain hemispheres were homogenized in 1mL of 50% trichloroacetic acid (Sigma), and centrifuged. Evans blue extravasation was quantified in the supernatants by spectrophotometry.
Measurement of Edema
At 24 h after stroke, brain edema was measured by comparing the wet to dry tissue weight ratios as described earlier (Liu et al, 2008). Briefly, the brain was quickly removed after the animal was killed. Then the brain was blotted to remove residual absorbent moisture, and dissected through the interhemispheric fissure into right and left hemispheres. Wet weight was determined with a resolution of 0.1 mg. Dry weight of whole ipsilateral and contralateral hemispheres was determined after heating the tissue for 3 days at 100°C in a drying oven. The tissue water content was then calculated as % H2O=(1–dry wt/wet wt) × 100%.
Western Blotting
At 24h after stroke, mouse brain samples were obtained by rapid removal of the brain from the skull, resection of the cerebellum, followed by immediate dissection into right (R; ischemic) and left (L; nonischemic) hemispheres. The brain samples were further dissected into core and penumbral regions as follows: The hemisphere was cut 1mm from the midline and 2mm from either olfactory/frontal or cerebellum/occipital pole, then a wedge of tissue was removed from a coronal section as infarct core along the boundary between infarct and noninfarcted tissue, and the remaining section was assessed as the penumbra (Figure 4A). The samples were flash-frozen and stored in −80°C until use. The samples were homogenized in 750µL of ice-cold RIPA buffer containing a protease inhibitor tablet (Roche Diagnostics, Indianapolis, IN, USA) and 1mmol/L of phenylmethylsulfonyl fluoride using a dounce homogenizer on ice, and briefly sonicated on ice. The extracts were immediately centrifuged at 14,000 rpm, for 20 mins at 4°C. The resulting supernatant was removed and protein concentrations were determined using a BCA kit (Bio-Rad, Hercules, CA, USA). The homogenates were mixed with equal volumes of 2 × sample buffer and boiled for 5mins before being loaded onto a 4% to 15% gradient SDS-PAGE gel. Equal amounts of protein (40 µg) were loaded onto each lane and then transferred to polyvinylidene difluoride membranes (Bio-Rad). The membranes were blocked and then incubated with Aquaporins (AQP1 and AQP4; 1:1,000, Chemicon, Billerica, MA, USA), Claudin-5 (1:1,000, Chemicon), Connexin 43 (Cx43; 1:1,000, Cell Signaling) or β-actin (1:5,000, Sigma). Actin was used as a loading control. All blots were incubated overnight in a primary antibody at 4°C in TBS buffer containing 5% bovine serum albumin and 0.1% Tween 20. The secondary antibody (1:2,000) was either goat anti-rabbit or goat anti-mouse depending on the primary antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA). The signal was visualized with SuperSignalWest Pico Chemiluminescent Substrate (ThermoScientific, Waltham, MA, USA). The densitometry of western blotting images was performed with computer software (Scion Image).
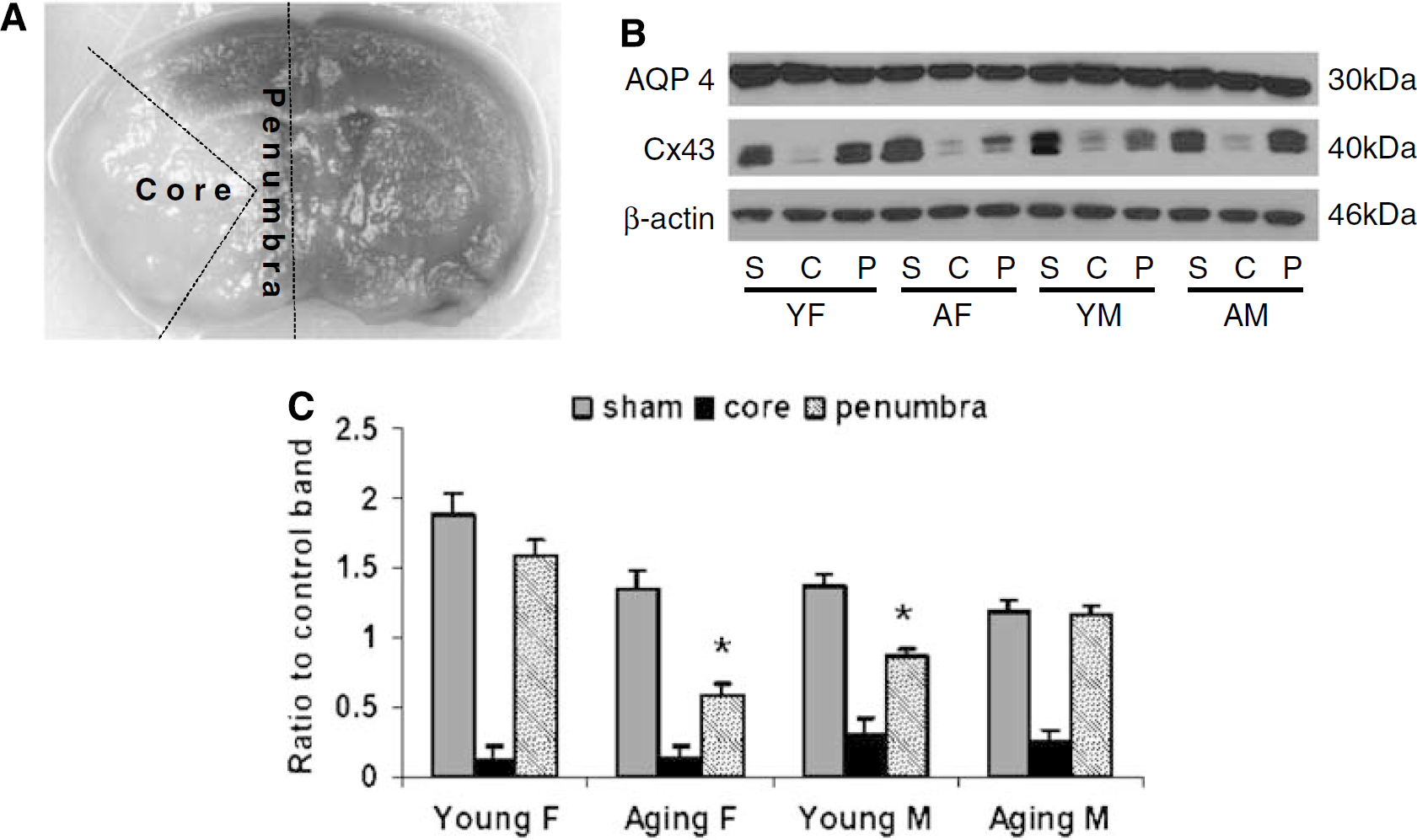
Cx43 expression in infarct core (C), penumbra (P), and sham (S). (
Statistics
All values are expressed as mean±s.e.m. and analyzed with a
Results
Physiologic Parameters are Normal and Equivalent in All Study Groups
To assess whether young and aging mice have equivalent physiologic parameters, mean arterial pressure, pH, pO2, pCO2, and blood glucose were measured. There was no difference in preischemic or intraischemic arterial blood pressure or blood gas measurements between young and aging mice (Table 1). The intraischemic LDF signal showed that CBF in the area blood-supplied by MCA was reduced equivalently in all groups (Figure 1A). Three aging mice and two young mice were excluded from the experiment because their CBF did not decrease below 85% of the baseline after MCAO.
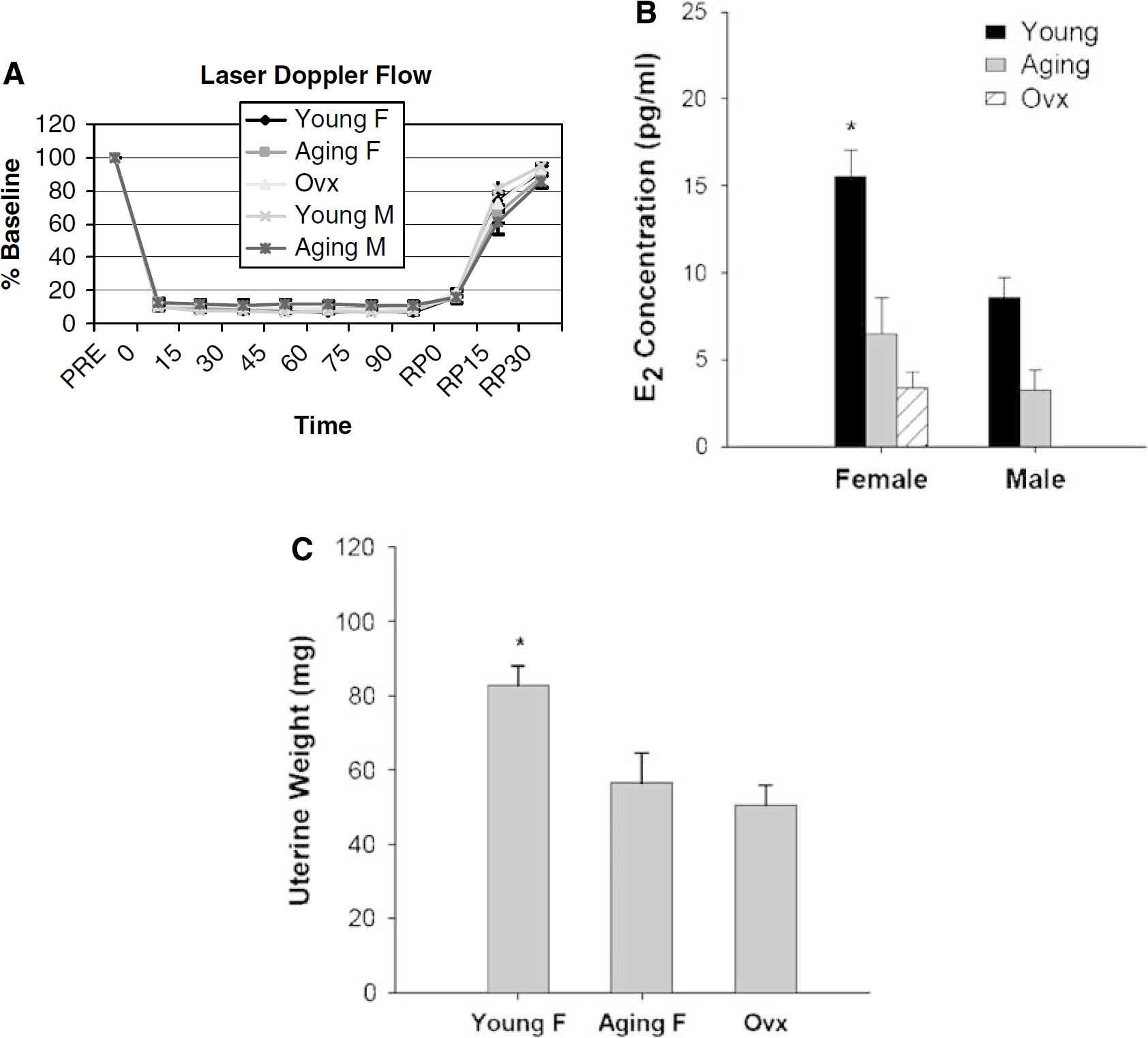
Cerebral blood flow, E2 concentration, and uterine weights measurement. (
Physiologic measurements in young and aging mice
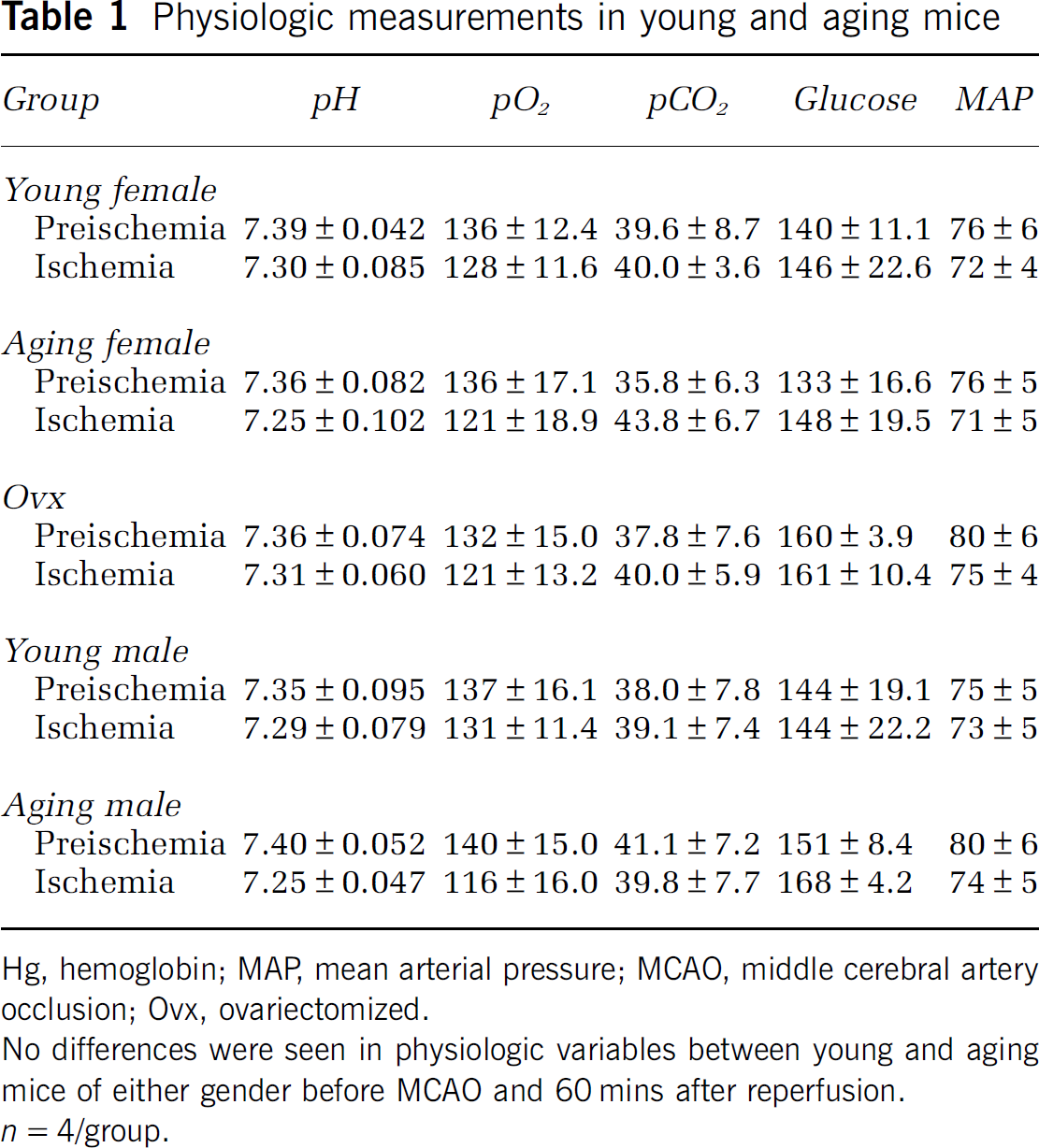
Hg, hemoglobin; MAP, mean arterial pressure; MCAO, middle cerebral artery occlusion; Ovx, ovariectomized.
No differences were seen in physiologic variables between young and aging mice of either gender before MCAO and 60mins after reperfusion.
E2 Concentration and Uterine Weights were Higher in Intact Young Female Mice
To evaluate the contribution of estrogen to stroke outcome, we measured the levels of E2 and uterine weights in both young and aging mice. Young intact female mice had significantly higher levels of E2 (15.6±3.7 pg/mL,
Age and Sex Differences were Found in Infarct Volumes Induced by Middle Cerebral Artery Occlusion
To examine histologic outcomes after stroke, we used standard staining with 1.5% TTC (Figure 2A). A quantitative analysis (Figure 2B) showed that aging female mice had significantly larger infarct volumes than young females (total infarct: aging versus young: 36.86%±5.49% versus 20.49%±2.20%;
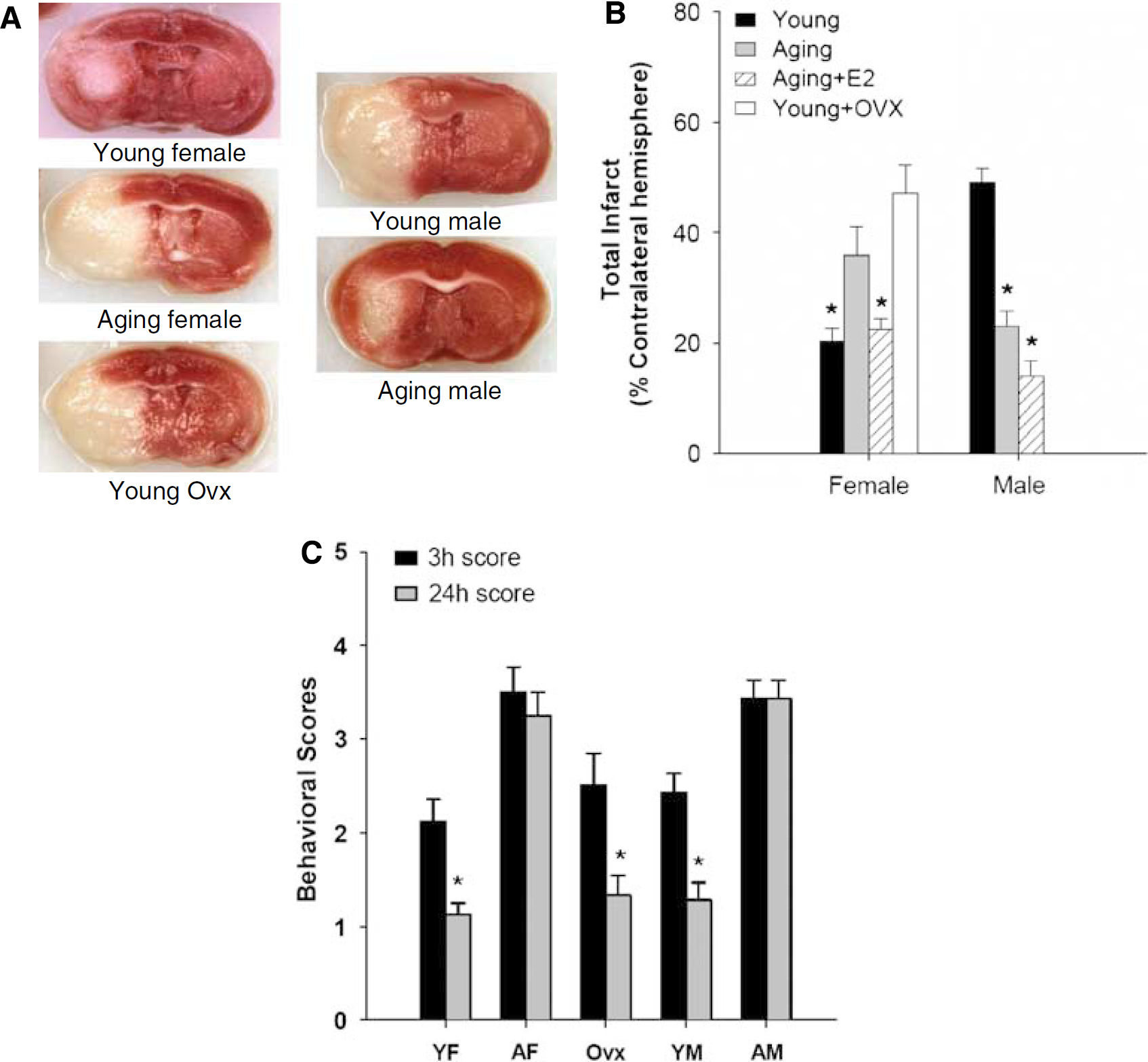
Stroke outcomes in young and aging mice after 24 h of MCAO. (
Estrogen was Neuroprotective in Aging Animals of Both Sexes
To determine whether the increase in infarct damage in aging females was because of the loss of estrogen, a separate cohort of young females was ovariectomized before MCAO (surgical menopause). As expected, young Ovx females had significantly larger strokes (total: 47.11%±5.24%,
Neurologic Deficits are Severe in Aging Mice
Behavioral deficits were scored at either 3 or 24 h after stroke. Both young and aging mice showed clear neurologic deficits at 3 h of stroke; however, young mice had a significant improvement in their deficits as rapidly as 24 h after stroke (female—3h: 2.1±0.2 versus 24 h: 1.1±0.1,
Evans Blue Extravasation and Expression of Cx43 Levels Reflected the Degree of Infarction
As it has been shown that reproductive hormones regulate the permeability of BBB (Wilson et al, 2008), we hypothesized that the loss of estrogen with aging would lead to increased permeability of BBB after stroke. The EB concentration in both the ipsilateral and contralateral hemispheres of mice after MCAO was measured to determine an EB index (EBI: ipsilateral/contralateral hemisphere EB concentrations). As expected, the EBI reflected the degree of ischemic damage, with large infarcts leading to a higher amount of dye extravasation. The EBI in both aging and Ovx female mice was significantly higher than that in young intact female mice (
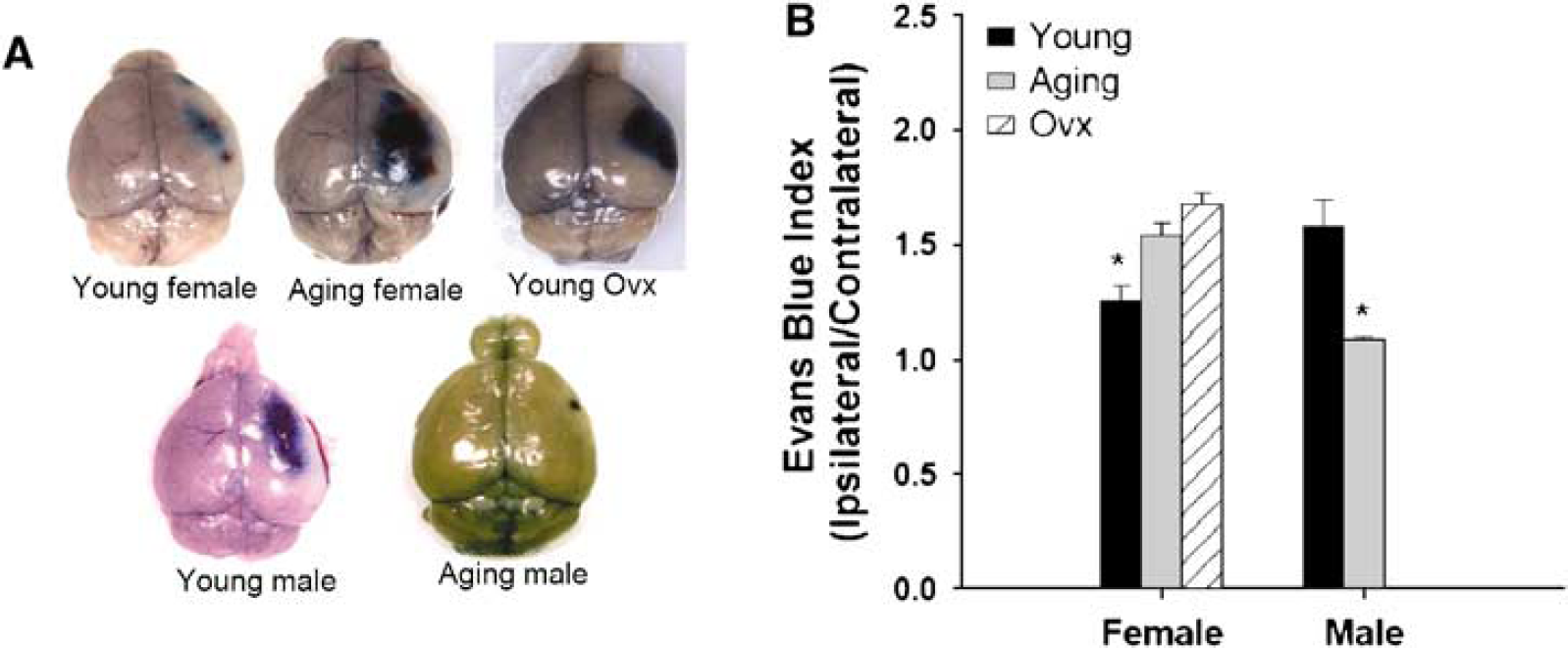
Evans Blue (EB) extravasation in brains after 24 h of stroke. (
To further assess the integrity of BBB after stroke, we also examined the expression of Cx43, a major gap junction protein that is hormonally regulated (Wilson et al, 2008), involved in the response to stroke. The Cx43 expression was very low in the core of the infarct in all groups of mice suggesting a complete loss of BBB integrity. However, an analysis of the penumbral tissue showed striking differences between groups. Aging female mice had a significantly decreased expression of Cx43 in the penumbra compared with that of sham, as did young male mice, whereas the Cx43 levels were relatively preserved in both young females and aging males. This was consistent with both infarction volumes and EB leakage (Figures 4B and 4C). As tight junction proteins may also play a role in the response to stroke, we also examined Claudin-5 levels and found no differences with aging (data not shown).
Aging Mice had Less Edema Formation than Young Mice
Edematous reactions surrounding brain lesions are less extensive in elderly patients (Desbordes and Cohadon, 1987). Young patients who experience extensive middle cerebral artery strokes are more likely to develop fatal brain edema than older patients (Wagner and Lutsep, 2005). To determine whether there were differences in edema formation induced by MCAO, we measured the water content in a separate cohort of animals after stroke. Young animals had dramatically more edema even upon gross visual inspection of the brain compared with sex-matched aging mice. Water content in both the ipsilateral and contralateral hemispheres was measured and an edema index (EI) was calculated (EI = water content in the ipsilateral/the contralateral hemisphere). Edema indices were significantly higher in young mice of both sexes compared with those in aging mice (
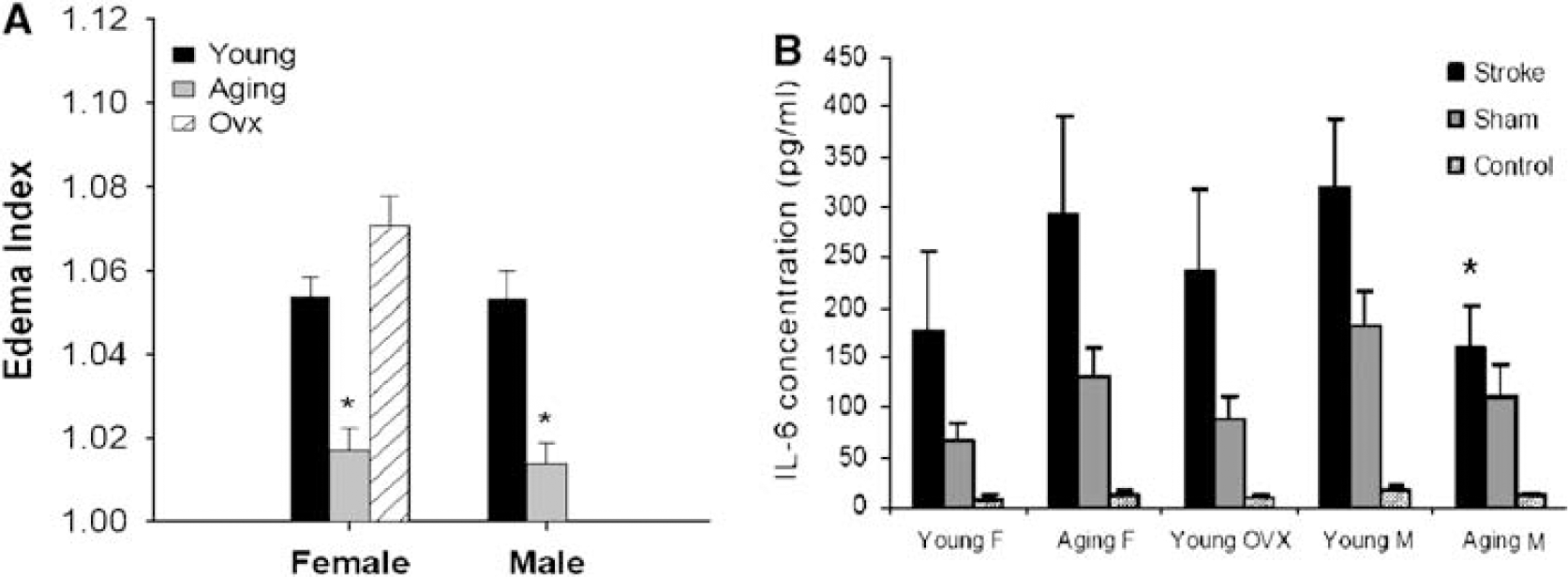
Edema in brains and serum IL-6 levels in young and aging mice after stroke. (
Serum and Brain Levels of the Proinflammatory Cytokine, Interleukin-6, were Lower in Aging Males
Inflammation plays an important role in the response to ischemic stroke (Huang et al, 2006). To assess the inflammatory response induced by stroke, and to determine whether levels were related to the striking reduction in edema volume in aging mice, we measured the serum levels of IL-6, a well-known proinflammatory cytokine, in mice of all groups 24 h after stroke. As expected, the serum concentrations of IL-6 increased in all stroke groups compared with those in sham (Figure 5B). Aging males had the smallest increase in IL-6. Although IL-6 levels tended to be lower in young females compared with aging females, this difference was not significant. However, IL-6 levels in aging females were higher than those of aging males (aging female versus aging male: 292.6±97.6 versus 159.9±41.2 pg/mL,
Discussion
This study represents the initial steps in the investigation of sex and age differences in response to focal stroke in mice, and shows several important findings. First, age-related differences exist in behavioral and histologic outcomes after MCAO in mice. Aging female mice had significant exacerbation in ischemic damage compared with young female mice. This appears to be more related to estrogen loss rather than to aging
Estrogen has numerous effects on neurons, astrocytes, endothelial cells, and microglia, and has potent anti-inflammatory and antiapoptotic actions in the injured brain (Brann et al, 2007). Stroke outcomes in female mice show a clear estrogen-dependent response in our studies, that is both aging and Ovx females had increased infarct volumes compared with intact females. Plasma estrogen levels in both aging and Ovx females were significantly lower and the uteruses were lighter than those of intact females. Despite the robust preclinical evidence for E2, recent clinical trials have questioned the ability to translate this effect into human populations. The Woman's Health Initiative (WHI), the largest clinical trial of E2 replacement for stroke prevention, showed a surprising increase in stroke incidence in E2-treated women (Wassertheil-Smoller et al, 2003). The explanation for these findings has been debated extensively in the literature, but the timing of replacement may be a major factor. For example, women involved in the WHI trial were considerably older (average age of 63 years) and well past menopause (which occurs at an average age of 51 years) before randomization and initiation of therapy.
A recent animal study (Suzuki et al, 2007), designed in an attempt to explain these results, found that E2 exerted profound neuroprotective effects when administered immediately on ovariectomy, but not when administered after 10 weeks of hypoestrogenicity. Robust stroke-induced inflammatory changes were only seen in mice that had been without E2 for months before E2 replacement. A second recent study found that ‘mature’ (7 to 8 months) females with normal but lengthened estrous cycles had a neuroprotective response to E2 after ovariectomy and E2 replacement, whereas slightly older rats that were acyclic and ‘reproductively senescent’ (9 to 11 months) had an exacerbation of injury after ovariectomy and E2 replacement (Selvamani and Sohrabji, 2008). These studies support the hypothesis that the neuroprotective efficacy of E2 depends on the timing of initiation of therapy and that E2 replacement can be deleterious in the aging brain.
However, our studies suggest that E2 plays an important protective role in aging mice, even in aging males, and replicate earlier study that showed E2 retained its neuroprotective efficacy even in aging animals (Alkayed et al, 2000; Alonso de Lecinana and Egido, 2006; Toung et al, 2004). There are several explanations for these findings. In the negative studies discussed above, much younger animals were used (i.e., 5 to 11 months) and all were subjected to surgical ovariectomy before injury. We examined older mice (15 months) that had progressed through ‘natural menopause,’ as confirmed by low serum E2 levels and uterine atrophy. It is important to note that these animals are reproductively senescent and aging, but not ‘elderly.’ Rodents undergo a transition in midlife (at 12 to 14 months of age) referred to as the ‘estropause,’ which is characterized by irregular, usually prolonged, estrous cycles and eventual acyclicity (Chakraborty and Gore, 2004). Therefore, our studies reflect the effect of relatively early E2 replacement initiated just beyond middle age. This design more accurately reflects the ongoing clinical trial, the Kronos Early Estrogen Prevention Study (KEEPS; available at: http://www.keepsstudy.org/), in which younger women are given estrogen replacement therapy within months after acyclicity, the results of which will be available in 2012.
The difference in infarct volumes between aging and young male animals is in agreement with earlier study by others that showed increased histologic damage in young (3 months), compared with old (24 to 26 months) male rats (Shapira et al, 2002). However, a controversy exists in the literature regarding this issue, with some studies showing larger strokes in aging animals (Sutherland et al, 1996) and many others showing no difference between aging and young ones (Wang et al, 2003). However, the majority of these earlier studies did not assess CBF, even with LDF—a method widely used to secure a successful occlusion of MCA. We used LDF as well as behavioral testing in every mouse peristroke to ensure that CBF was decreased equivalently (below 85% of baseline). Furthermore, most investigators utilized rats in their studies, and the results in female mice were unknown before this study. The sex of the animal evaluated is also a major issue in interpretation. For example, a recent study that showed an exacerbation of infarct damage with aging only examined females (DiNapoli et al, 2008). The larger infarct may represent the effect of hormonal loss rather than an effect of aging
Despite the smaller infarcts, our aging mice had significantly higher mortality (29%) and more severe behavioral deficits than young mice, again consistent with several other studies (Badan et al, 2003; Popa-Wagner et al, 2007), which showed that aging is associated with rapid infarct development and a delayed, suboptimal functional recovery in the poststroke period. However, although a selective acceleration of infarct development in the aging brain could account for the larger strokes seen in our aging female mice, aging male mice had smaller infarcts than young males. This makes acceleration of infarct an unlikely explanation for the changes in infarct size that occur with aging. The mechanism of the decreased histologic damage in aging male mice is unclear, but may be related to a decline in NMDA receptor binding or a decrease in NMDA receptor-mediated responses in aged, as compared with young male rats (Wenk et al, 1991; Gonzales et al, 1991).
It is now widely accepted that inflammation plays a critical role in stroke, and postischemic inflammatory responses strongly contribute to the extent of ischemic brain injury (Huang et al, 2006). In the elderly, immunodeficiency has been suggested because of low detected quantities of T cells that play critical roles in the development of ischemic injury by producing proinflammatory cytokines, such as IL-6, IL-1β, and TNF-α (Cakman et al, 1996). However, there is considerable controversy in the literature regarding the inflammatory responses in aging, possibly because many studies were performed only in male animals or sex-mixed subjects (Cakman et al, 1996; Gabriel et al, 2002). Young female animals are known to have a decreased inflammatory response to injury compared with males and a considerable body of evidence strongly implicates E2 as a major regulator of inflammatory pathways (Maggi et al, 2004). Both serum and brain levels of the proinflammatory marker, IL-6, were significantly lower in our aging males compared with those in aging females. Obviously, many cell adhesion molecules, cytokines, and chemokines contribute to the inflammatory response to stroke. Interleukin-6 was initially evaluated, as data from human studies show higher CSF and serum levels of IL-6 are correlated with larger infarcts after stroke (Smith et al, 2004; Tarkowski et al, 1995). Studies are being conducted even to date to examine the effect of aging on the inflammatory response to stroke in both sexes.
Ischemic stroke induces impairment of the BBB, and increased BBB permeability has been shown to play an important role in the pathophysiology of postischemic reperfusion injury (Belayev et al, 1996). Our EB results suggest that BBB disruption may be more reflective of the degree of injury or hormone loss rather than an enhanced intrinsic BBB dysfunction with aging. Several other studies have shown that E2 loss enhanced brain injury and BBB permeability after ischemic insults (DiNapoli et al, 2008; Wilson et al, 2008). Surprisingly, there is a paucity of data in the literature examining BBB disruption in aging animals after focal stroke. The most recent and complete study to date showed an increase in BBB breakdown, stroke damage, and enhanced behavioral deficits after MCAO but used only female rats (DiNapoli et al, 2008). No mention was made of hormonal status, and the enhanced damage is likely a reflection of the loss of E2, consistent with our findings.
The integrity of BBB is maintained by both tight junction proteins, such as zona occludens 1 (ZO-1 and ZO-2), occludins and claudin-5, and gap junction proteins, such as Cx43, Cx45, Cx32 and Cx26 between endothelial cells (Nagasawa et al, 2006). We examined Cx43 as it is a major astrocytic gap junction protein that is upregulated after injury (Daleau et al, 2001), and is hormonally responsive (Wilson et al, 2008). Earlier study by others has shown that the Cx43 protein is decreased in the postischemic cerebral cortex, suggesting an impairment of gap junction intercellular communication (Haupt et al, 2007). In this study, the Cx43 protein expression in the penumbra was consistent with both our EB and infarction data. Aging female mice had the lowest expression of penumbral Cx43, suggesting the greatest disruption of the BBB after MCAO. Interestingly, we did not find any difference in the Claudin-5 expression between young and aging mice of either sex, suggesting that the effects on Cx43 are specifically affected by sex and age.
Extravasation of EB is related to leakage of proteins after acute destruction of the endothelium. In contrast, edema formation is more complex, and differentiation of cytotoxic versus vasogenic brain edema can be difficult (Xiao, 2002). To our surprise, there was no sex difference in edema formation in our study despite the striking sex differences in infarction and BBB disruption, as measured by EB and Cx43. We saw no reduction in the AQP4 (or AQP1, data not shown) expression in the aging brain, despite the significant reduction in edema in both male and female aging mice (Figure 5A). This is consistent with several other studies showing a lack of sex differences in cerebral edema formation and in the AQP4 expression after brain injury (Carswell et al, 2000; Liu et al, 2008). Interestingly, our data show that EI was not correlated with the degree of injury, as aging females had much larger strokes than young females, yet younger mice continued to have significantly more cerebral edema. This suggests that BBB breakdown and infarct are dissociated from the mechanism that leads to cerebral edema, and that the most important factor for edema development is age. Among patients with massive cerebral infarcts, older patients (> 70 years) had a surprisingly lower in-hospital mortality (Chen et al, 2007) and died after discharge, often of systemic complications. In contrast, patients under 70 tended to die within the first 2 weeks, coinciding with the peak of cerebral edema. This concept of more robust edema formation in the young brain has been confirmed in postmortem studies (Jaramillo et al, 2006), and is part of the rationale for proposing an upper age limit of 60 years for hemicraniectomy.
This study has several limitations. We only examined the outcome at 24 h after stroke. Former studies (Dereski et al, 1993; Lu and Sun, 2003) have shown that the infarct is complete by 24 h of transient MCAO in young animals, which we have confirmed in our laboratory. However, the effect of E2 or aging on the evolution of infarct needs to be further investigated. It can be noted that we have found similar histologic changes in the brains of aging males and females at 48 h (Liu et al, 2009). In this study, we measured absolute water content in the brain as an indicator of edema formation. This cannot reliably differentiate cytotoxic versus vasogenic edema, and MRI measurements may be superior in this regard. Finally, we compared E2-implanted aging mice with intact aging mice rather than comparing them with an ovx-vehicle-implanted aging cohort in an attempt to limit animal usage and model clinical menopause (where ovarian function gradually declines with age). No study has reported that sesame oil has an effect on stroke, and E2 levels are equivalent in aging males and aging females (Liu et al, 2009); ovariectomizing females before implantation of both oil and E2 would have been a more appropriate control. On account of the limited availability and high cost of aging animals, as well as their relative fragility, we chose to forego ovariectomy in the aging females.
In conclusion, dramatic sex- and age-related differences occur after experimental stroke, which are reflected in the outcomes of injury, BBB disruption, and edema formation. Female mice go through a stroke-resistant, followed by a stroke-vulnerable, pattern of response to MCAO across their life span, and this pattern appears to be estrogen-dependent. Surprisingly, males have the opposite pattern, which may be related to a reduction in the inflammatory response to stroke with aging. E2 retains its neuroprotective effect in aging animals of both sexes. Evans blue extravasation and Cx43 expression in penumbra are consistent with the degree of ischemic damage in both sexes, whereas edema formation shows age-related differences. These results strongly suggest that therapeutic interventions for stroke need to be tailored to the sex and age of the patient.
