Abstract
The aim of this study was to investigate whether postoperative hyperperfusion is associated with preoperative cerebral hemodynamic impairment due to chronic ischemia and with acute cerebral ischemia during clamping of the internal carotid artery (ICA) during carotid endarterectomy (CEA). Transcranial cerebral oxygen saturation (SO2) was monitored intraoperatively using near-infrared spectroscopy in 89 patients undergoing CEA for ipsilateral ICA stenosis (> 70%). Cerebral blood flow (CBF) and cerebrovascular reactivity (CVR) to acetazolamide were also measured using single photon emission computed tomography (SPECT) before CEA. In addition, CBF was measured immediately after CEA and on the third postoperative day. Hyperperfusion (CBF increase > 100% compared with preoperative values) was observed immediately after CEA in 10 of 18 patients (56%) with reduced preoperative CVR. Also, post-CEA hyperperfusion was observed in nine of 16 patients (56%) whose SO2 during clamping of the ICA decreased to less than 90% of the preclamping value. Logistic regression analysis showed that reduced preoperative CVR and reduced SO2 during ICA clamping were significant independent predictors of the development of hyperperfusion immediately after CEA. In fact, all patients with reduced preoperative CVR and reduced SO2 during ICA clamping developed post-CEA hyperperfusion, and two of these patients developed cerebral hyperperfusion syndrome. These data suggest that development of cerebral hyperperfusion after CEA is associated with preoperative hemodynamic impairment and intraoperative cerebral ischemia.
Introduction
Cerebral hyperperfusion after carotid endarterectomy (CEA) is defined as a major increase in ipsilateral cerebral blood flow (CBF) following surgical repair of carotid stenosis that is well above the metabolic demands of the brain tissue (Sundt et al, 1981; Piepgras et al, 1988). Cerebral hyperperfusion syndrome following CEA is a complication of cerebral hyperperfusion, which is characterized by unilateral headache, face and eye pain, seizure, and focal symptoms that occur secondary to cerebral edema or intracerebral hemorrhage (Solomon et al, 1986; Sundt et al, 1981; Bernstein et al, 1984; Piepgras et al, 1988). Although the incidence of intracerebral hemorrhage is relatively low (0.4% to 1.8%), the prognosis for patients with this condition is poor (Piepgras et al, 1988; Pomposelli et al, 1988; Jansen et al, 1994; Riles et al, 1994; Dalman et al, 1999; Ouriel et al, 1999).
Risk factors for this syndrome include longstanding hypertension, high-grade stenosis, poor collateral blood flow and contralateral carotid occlusion, which often impair cerebral hemodynamic reserve (Reigel et al, 1987). A rapid restoration of normal perfusion pressure following CEA may result in hyperperfusion in regions of the brain that have impaired autoregulatory capacity secondary to chronic ischemia. This hypothesis is similar to the ‘normal perfusion pressure breakthrough' theory described by Spetzler et al (1978). In fact, several investigators have showed that decreased cerebrovascular reactivity (CVR) to acetazolamide is a significant predictor of post-CEA hyperperfusion (Yoshimoto et al, 1997; Hosoda et al, 2001; Ogasawara et al, 2003b). However, while post-CEA hyperperfusion is not observed in patients with normal preoperative CVR, patients with impaired cerebral hemodynamic reserve do not always develop post-CEA hyperperfusion (Yoshimoto et al, 1997; Hosoda et al, 2001; Ogasawara et al, 2003b). Thus, additional factors may be associated with the development of post-CEA hyperperfusion.
Clamping of the internal carotid artery (ICA) during CEA results in a transient decrease in CBF in the ipsilateral cerebral hemisphere in many patients (Sundt et al, 1981; Halsey, 1992). If the decrease in the hemispheric CBF is significant enough to impair autoregulation, then ipsilateral cerebral hyperperfusion can occur after ICA declamping. Thus, the purpose of this study was to investigate whether acute cerebral ischemia during clamping of the ICA is associated with development of post-CEA hyperperfusion. Preoperative and postoperative CBF and intraoperative cerebral hemodynamics were measured using single photon emission computed tomography (SPECT) and transcranial cerebral oxygen saturation (SO2) monitoring with near-infrared spectroscopy, respectively.
Materials and methods
Patient
In total, 89 patients with ipsilateral ICA stenosis (> 70%) and useful residual function (modified Rankin disability scale 0, 1 or 2), who underwent CEA were enrolled in the present study. Of the 89 patients, 80 were men and nine were women. The mean age of the patient population was 68.1 ± 6.8 years (mean ± s.d.), ranging from 47 to 78 years. Concomitant disease states and symptoms were recorded, including 68 patients with hypertension and 26 patients with diabetes mellitus. A total of 54 patients evinced ipsilateral carotid territory symptoms, with 16 patients displaying transient ischemic attacks, 10 patients with transient ischemic attacks and subsequent stroke, and 28 patients with stroke alone. In total, 35 patients exhibited asymptomatic ICA stenosis.
Preoperative magnetic resonance (MR) imaging showed infarction in the hemisphere ipsilateral to the ICA stenosis in 46 patients and no infarction in 43 patients. In total, 44 patients had symptomatic infarction, with transient ischemic attack and/or stroke, while the remaining two patients displayed asymptomatic infarction.
All patients underwent preoperative angiography with arterial catheterization. The average degree of ICA stenosis was 83% ± 9%, with a range of 70% to 95%, according to the method of the North American Symptomatic Carotid Endarterectomy Trial (1991). The contralateral ICA was occluded in six patients, and 20 additional patients had 60% to 95% stenosis.
This study was reviewed and approved by the institutional ethics committee. Informed consent was obtained from all patients or their next of kin.
Single photon emission computed tomography
Cerebral blood flow was assessed using N-isopropyl-p-[123I]iodoamphetamine (IMP) and SPECT before and immediately after CEA. Cerebrovascular reactivity to acetazolamide was also measured for evaluation of cerebral hemodynamic reserve before CEA. In addition, patients with post-CEA hyperperfusion underwent a third CBF measurement in the same manner, 3 days after CEA. Preoperative SPECT study was performed more than 1 month after the last ischemic event and 7 to 10 days before CEA.
Single photon emission computed tomography studies were performed using a ring-type SPECT scanner (Headtome-SET080, Shimadzu Corp., Kyoto, Japan), which provided 31 tomographic images simultaneously. The spatial resolution of the scanner with a low-energy, all-purpose collimator was 13 mm FWHM at the center of the field of view, and the slice thickness was 25 mm FWHM at the field of view center. Image slices were taken at 5 mm center-to-center spacing, parallel to the orbitomeatal line. The images were reconstructed using the weighted-filtered backprojection technique, in which the attenuation correction was made by detecting the edge of the object. An attenuation coefficient of 0.065 cm−-1, a Butter-worth filter (cutoff = 0.45 cycle/cm; order = 3) and a ramp filter were used for image reconstruction.
The IMP SPECT study with acetazolamide challenge was performed as described previously (Ogasawara et al, 2003a), and the CBF images were calculated according to the IMP-autoradiography method (Iida et al, 1994a, b; Ogasawara et al, 2003a). In each image, slice obtained immediately after CEA, a large irregular region of interest (ROI) of 16 cm2 or more was manually and bilaterally drawn in the entire cerebral cortex. These ROIs were placed in regions where infarction was not present, as confirmed by MR imaging. After the CBF was determined in each ROI, the ratio of ipsilateral regional CBF (I) to contralateral regional CBF (C) (I/C ratio) was calculated in each image slice. The tomographic plane with the highest I/C ratio was selected and analyzed for each patient. Next, the tomographic plane that was determined in image slices obtained immediately after CEA was manually selected in image slices obtained preoperatively and 3 days after CEA, and a ROI was set in the entire cerebral cortex in the same manner. Computed tomography (CT) or MRI-SPECT imaging coregistration was not used. Preoperative CVR to acetazolamide was calculated as follows (Ogasawara et al, 2003a): CVR (%) = [(acetazolamide challenge CBF-resting CBF)/resting CBF] × 100. Using the IMP-autoradiography method, 10 normal subjects (eight men and two women) aged 35 to 65 years (mean, 52.3 years) were studied to obtain control values (Ogasawara et al, 2003a). The control values of CBF at resting state and CVR were 35.9 ± 4.4 ml/100 g/min and 36.8% ± 9.2%, respectively. When the values of CVR were less than the mean-2s.d., (i.e., 18.4%), they were rated as reduced CVR. Post-CEA hyperperfusion was defined as CBF increase of > 100% (i.e., a doubling) compared with preoperative values, according to Piepgras et al (1988).
Transcranial Cerebral Oxygen Saturation
A near-infrared spectroscope, TOS 96 (Tostec, Tokyo, Japan), with a dual-channel system was used to measure SO2 throughout CEA (Litscher et al, 1998; Nara et al, 1998). After the patient's skin was thoroughly cleaned, the sensors were bilaterally and symmetrically placed over the forehead, according to the manufacturer's instructions. After a stable SO2 reading was achieved, the margins of the sensors were secured with opaque tape. Transcranial cerebral oxygen saturation readings were recorded at 30-secs intervals and stored for later analysis. The following data points regarding cerebral oximetry for the side ipsilateral to CEA were gathered and recorded: the mean value in the last 5 mins before ICA clamping (SO2−0) and the lowest value during clamping of the ICA (SO2−1). The SO2 data for each patient were normalized such that the SO2−0 was assigned as a value of 1.0 and the SO2−1 was calculated as a ratio to the SO2−0 (SO2 ratio).
Intraoperative and Postoperative Management
All patients underwent surgery under general anesthesia more than 1 month after the last ischemic event. Patients were premedicated with midazolam (7.5 mg orally). Anesthesia was induced with fentanyl (2 to 3 μg/kg intravenously), propofol (1.5 to 3 mg/kg intravenously), vecuronium (0.1 mg/kg intravenously) and was maintained by repeated boluses of fentanyl (1 to 2 μg/kg intravenously), vecuronium, and 0.4% to 1.0% inspired isoflurane. All patients were artificially ventilated with an air-oxygen mixture (inspired fraction of oxygen ~0.30). Analysis of intermittent drawn arterial blood gas samples ensured normoventilation (4.7 to 5.2 kPa). Routine monitoring during anesthesia included standard electrocardiography, placement of an intraarterial catheter for direct arterial blood pressure measurement, pulse oximetry, and capnography. Blood pressure was kept stable in a range ± 20% of the preoperative level throughout the procedure by adjusting the depth of anesthesia or, if needed, by intravenous administration of a vasodilator (nitroglycerin) or a vasoconstrictor (theoadrenalin).
An intraluminal shunt was not used in these procedures. The mean duration of ICA clamping was 33 mins, ranging from 17 to 47 mins. A bolus of heparin (5000 U) was administered prior to ICA clamping, and protamine was administered at the conclusion of CEA.
All patients underwent CT imaging on the first postoperative day and T1- and T2-weighted MR imaging on the third postoperative day to confirm the presence or absence of additional ischemic lesions.
In all patients with post-CEA hyperperfusion, intensive control of arterial blood pressure between 100 and 140 mm Hg was instituted using intravenous administration of antihypertensive drugs immediately after SPECT. When CBF decreased and hyperperfusion resolved on the third postoperative day, pharmacologic control of blood pressure was discontinued. However, when hyperperfusion persisted, systolic arterial blood pressure was maintained below 140 mm Hg. When hyperperfusion syndrome developed, the patient was placed in propofol coma. A diagnosis of hyperperfusion syndrome required: (1) seizure, deterioration of consciousness level and/or development of focal neurological signs such as motor weakness, and (2) hyperperfusion on the SPECT performed after CEA without findings of any additional ischemic lesion on postoperative CT scan or MR imaging.
Data Analysis and Statistics
Data are expressed as means ± s.d. Logistic regression analysis was used to determine the joint effect of multiple variables on hyperperfusion immediately after CEA. Covariates included age, gender, complications (hypertension and diabetes mellitus), symptomatic lesion, infarction on MR imaging, bilateral lesions (contralateral ICA stenosis or occlusion), degree of ICA stenosis, duration of ICA clamping, SO2 ratio during ICA clamping, and preoperative CVR. Differences were deemed statistically significance if P < 0.05.
Results
Two patients experienced new postoperative neurological deficit (slight hemiparesis) on the side contralateral to the CEA. However, this deficit resolved completely within 12 h in both patients. Postoperative CT scan or MR imaging did not detect additional ischemic lesions in any of the patients.
Of the 89 patients, 10 patients (11%) met the CBF criteria for post-CEA hyperperfusion on SPECT imaging performed immediately after CEA. In these 10 patients, hyperperfusion was observed in the entire ipsilateral hemisphere on the SPECT images (Figure 1A).
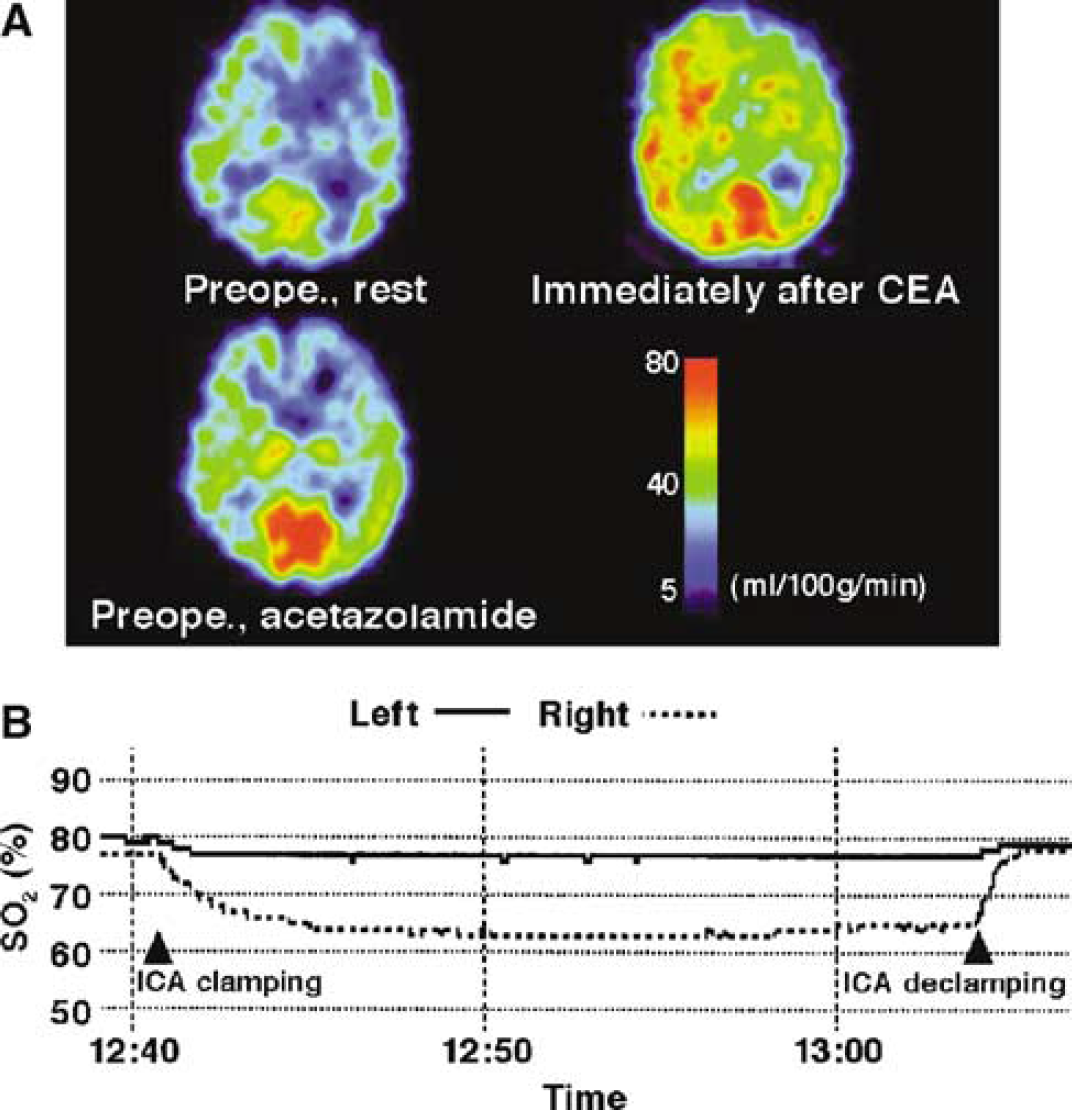
A 72-year-old man with symptomatic right internal carotid artery stenosis (90%) and asymptomatic left internal carotid artery stenosis (95%) exhibiting hyperperfusion syndrome after carotid endarterectomy. (
Post-CEA hyperperfusion was observed in 10 (56%) of 18 patients with reduced preoperative CVR. Further, none of patients with normal preoperative CVR (n = 71) developed post-CEA hyperperfusion.
While seven patients exhibited no change in SO2 before and during clamping of the ICA, 82 patients experienced a decrease in SO2 during clamping of the ICA. In these 82 patients, mean arterial blood pressure fluctuated ± 14% to −5% between the time points bounded by SO2−0 and SO2−1, and the time between clamping of the ICA and measurement of SO2−1 ranged from 0.5 to 19.5 mins (3.9 ± 3.9 mins). Further, the SO2 ratio in 66 of the 82 patients was between 0.9 and 1.0, while the remaining 16 patients exhibited a SO2 ratio less than 0.9. Post-CEA hyperperfusion was observed in 9 (56%) of these 16 patients. Both patients that experienced postoperative transient neurological deficits exhibited a SO2 ratio less than 0.9, and one of these patients experienced post-CEA hyperperfusion.
Logistic regression analysis showed that reduced preoperative CVR and reduced SO2 ratio during ICA clamping were significant independent predictors of the development of hyperperfusion immediately after CEA (Table 1). Other variables showed no significant association with post-CEA hyperperfusion. All nine patients with reduced preoperative CVR and SO2 ratio less than 0.9 developed post-CEA hyperperfusion (Figure 2). Only one of the nine patients with a combination of reduced preoperative CVR and a SO2 ratio greater than 0.9 developed post-CEA hyperperfusion. No patients with a combination of normal preoperative CVR and SO2 ratio less than 0.9, or with normal CVR and SO2 ratio more than 0.9 developed post-CEA hyperperfusion. Typical SPECT imaging and the time course of SO2 in a patient with post-CEA hyperperfusion is illustrated in Figure 1.
Risk factors for cerebral hyperperfusion immediately after carotid endarterectomy
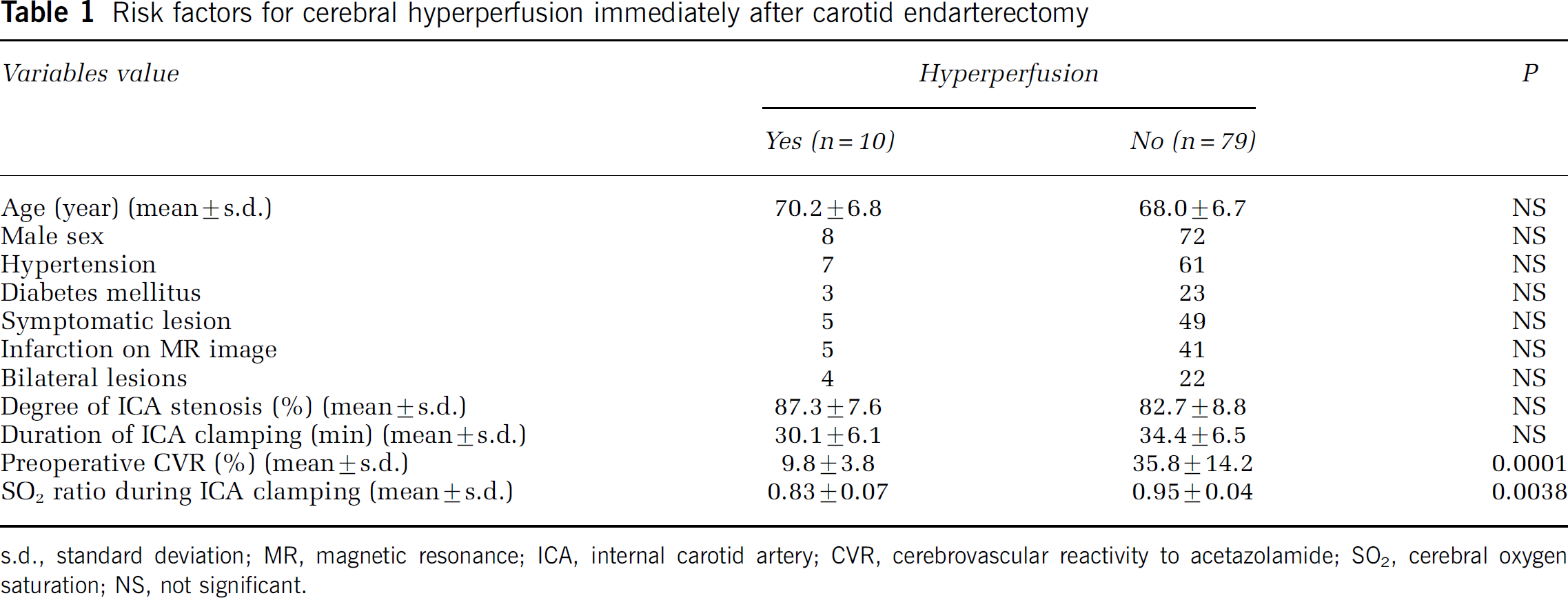
s.d., standard deviation; MR, magnetic resonance; ICA, internal carotid artery; CVR, cerebrovascular reactivity to acetazolamide; SO2, cerebral oxygen saturation; NS, not significant.
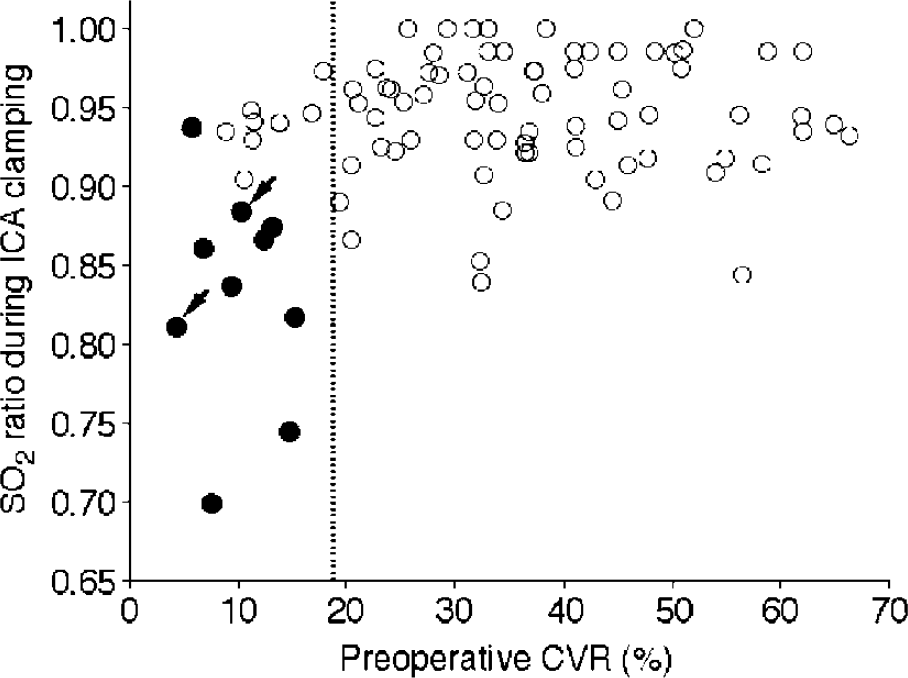
Preoperative cerebrovascular reactivity (CVR) and cerebral oxygen saturation (SO2) ratio during internal carotid artery clamping. Nine of 10 patients with postoperative cerebral hyperperfusion are located in the quadrant with preoperative CVR less than the mean-2s.d. of the normal control value and SO2 ratio less than 0.9. Closed and open circles indicate patients with and without cerebral hyperperfusion immediately after surgery, respectively. Arrows indicate patients with cerebral hyperperfusion syndrome. The dashed vertical line indicates the mean-2s.d. of the normal control value.
In eight of 10 patients with post-CEA hyperperfusion, hyperperfusion resolved in the SPECT performed on the third postoperative day, and pharmacologic control of blood pressure was discontinued. However, in the remaining two patients, hyperperfusion persisted and hyperperfusion syndrome developed (Figure 2). One of these two patients experienced a focal seizure as evidenced by motor disturbances of the left upper extremity 6 days after surgery. The other patient experienced confusion and left motor weakness on the sixth postoperative day. Propofol coma was induced in both patients. Eventually, both patients experienced full recovery following termination of propofol coma. Patients without hyperperfusion immediately after CEA (n = 79) did not develop hyperperfusion syndrome for the remainder of the study.
Discussion
This study showed that development of cerebral hyperperfusion after CEA is associated with preoperative hemodynamic impairment and intraoperative cerebral ischemia.
Previous studies have reported that patients with reduced preoperative CVR to acetazolamide have a high risk of developing post-CEA hyperperfusion (Yoshimoto et al, 1997; Hosoda et al, 2001; Ogasawara et al, 2003b), which is consistent with results from the present study. This supports the theory that hyperperfusion results from loss of normal vasoconstriction secondary to chronic cerebral ischemia and maladaptive autoregulatory mechanisms (Bernstein et al, 1984). However, patients with reduced preoperative CVR do not always develop post-CEA hyperperfusion (Yoshimoto et al, 1997; Hosoda et al, 2001; Ogasawara et al, 2003b). In fact, the incidence of post-CEA hyperperfusion in patients with reduced preoperative CVR ranges from 20% to 70% (Yoshimoto et al, 1997; Hosoda et al, 2001; Ogasawara et al, 2003b).
The present study showed that a reduction in the SO2 ratio during ICA clamping is a significant independent predictor of post-CEA hyperperfusion. Indeed, all patients with reduced preoperative CVR and a SO2 ratio less than 0.9 developed post-CEA hyperperfusion. These findings suggest that in addition to impairment of cerebrovascular autoregulation due to chronic ischemia, acute ischemia during clamping of the ICA contributes to the pathogenesis of post-CEA hyperperfusion.
Several investigators have suggested that acute ischemia and reperfusion by clamping and declamping of the ICA may produce oxygen-derived free radicals (Soong et al, 1996; Weigand et al, 1999; Holm et al, 2001). The free radicals may impair cerebrovascular autoregulation, resulting in postischemic hyperperfusion or brain edema (Patt et al, 1988; Phillis and Sen, 1993; Karibe et al, 1994). In addition, pretreatment with a novel free radical scavenger, edaravone, which is widely used in Japan to improve functional outcomes in patients suffering from acute ischemic stroke (The Edaravone Acute Brain Infarction Study Group, 2003), can prevent development of cerebral hyperperfusion after CEA (Ogasawara et al, 2004). Thus, free radicals produced by clamping and declamping of the ICA may adversely affect cerebrovascular autoregulation that is already impaired by preoperative chronic ischemia. This process likely culminates in post-CEA hyperperfusion.
In the present study, no patients with a combination of normal preoperative CVR and a SO2 ratio less than 0.9 developed post-CEA hyperperfusion. Further, all of these patients recovered from surgery without new major neurological deficits, and postoperative CT scan or MR imaging did not detect additional ischemic lesions. Thus, acute cerebral ischemia alone during ICA clamping may not be sufficient to provoke post-CEA hyperperfusion if the cerebral ischemia is not so severe.
In the present study, two of 10 patients with hyperperfusion immediately after CEA developed hyperperfusion syndrome. In the remaining eight of 10 patients with post-CEA hyperperfusion, CBF decreased, and hyperperfusion resolved by the third postoperative day. Previous studies with postoperative SPECT or intraoperative transcranial Doppler monitoring showed that only 11% to 25% of patients with post-CEA hyperperfusion went on to develop hyperperfusion syndrome and that patients who did not experience hyperperfusion immediately after CEA did not develop hyperperfusion syndrome (Dalman et al, 1999; Ogasawara et al, 2003b), which is consistent with findings from the present study.
The present study possesses several limitations that require discussion. First, the cerebral oximetry sensor used in this study was situated over the forehead and, thus, detected frontal lobe SO2. The frontal lobe is mainly supplied by the anterior cerebral artery, but some portions of the region exist in the watershed area between the anterior and middle cerebral arteries (van der Zwan et al, 1992). Thus, SO2 may not always detect hypoperfusion in the regions outside anterior cerebral artery territory. The SO2 is also influenced by extracranial blood flow. Despite improvements in sensor technology, the compensation modeling used by the apparatus to eliminate the effect of extracerebral tissue is still inadequate (Germon et al, 1999). Thus, the issue of extracranial contamination in SO2 monitoring remains a topic of considerable debate, particularly when SO2 monitoring is used for recognition of cerebral ischemia during ICA and external carotid artery clamping (Duncan et al, 1995; Kirkpatrick et al, 1995; Beese et al, 1998; Samra et al, 2000; Takeda et al, 2000). Most investigators have also observed that the absolute changes of SO2 after ICA clamping were highly variable from patient to patient (Beese et al, 1998; Cho et al, 1998; Grubhofer et al, 2000; Samra et al, 2000) and that the specificity of these values are not sufficient for satisfactory identification of patients suffering from severe cerebral ischemia during ICA clamping (Beese et al, 1998; Grubhofer et al, 2000). However, other investigators suggest that the intersubject variability in the absolute changes of SO2 could be addressed by normalizing the data and recording the decrease in SO2 after ICA clamping as a percentage of preclamp value (Cho et al, 1998; Samra et al, 2000). Such normalization would account for intersubject variability as well as allow comparisons between different investigations. In fact, using this strategy, these investigators concluded that a SO2 ratio less than 0.9 was indicative of cerebral ischemia (Cho et al, 1998; Samra et al, 2000).
Second, all our patients underwent CEA without use of an intraluminal shunt and suffered cerebral ischemia for a relatively long time, which may increase the risk of post-CEA hyperperfusion. However, the duration of ICA clamp was not a significant independent predictor of post-CEA hyperperfusion, while the severity of cerebral ischemia, which peaked at an average of 3.9 mins after ICA clamping, was a significant independent predictor of post-CEA hyperperfusion. Weigand et al (1999) reported that even reperfusion following short-term cerebral ischemia (ICA clamping for 6 mins) significantly enhanced cerebral lipid peroxidation. In fact, the incidence of hyperperfusion in patients undergoing CEA with the use of an intraluminal shunt is 8% to 11% (Piepgras et al, 1988; Dalman et al, 1999; Hosoda et al, 2001), which is consistent with data from the present study.
In conclusion, development of cerebral hyperperfusion after CEA is associated with preoperative hemodynamic impairment and with intraoperative cerebral ischemia. Further study to investigate the relationship between production of free radicals during CEA and development of post-CEA hyperperfusion would be of benefit.
