Abstract
Hypoperfusion after traumatic brain injury may exacerbate damage. Adenosine, a vasodilator, regulates cerebral blood flow (CBF). Treatment with adenosine receptor agonists has shown benefit in experimental CNS trauma; however, their effects on CBF after injury remain undefined. We used magnetic resonance imaging to assess CBF in uninjured rats both early and at 24 h after intrahippocampal administration of either the nonselective adenosine receptor agonist 2-chloroadenosine (2-CA, 12 nmol) or the A2A-receptor agonist 2-p-(2-carboxyethyl)-phenethylamino-5'-N-ethylcarbox-amidoadenosine (CGS 21680, 6 nmol). We also assessed the effects of these agents on cerebral metabolic rate for glucose (CMRglu). We then assessed the effect of 2-CA on CBF at 3.5 to 5 h after controlled cortical impact (CCI). Injection of 2-CA into uninjured rat brain produced marked increases in CBF in ipsilateral hippocampus and cortex versus vehicle (P < 0.05); CBF increases persisted even at 24 h. Measurement of hippocampal levels of 2-CA showed persistent increases to 24 h. CGS 21680 produced even more marked global increases in CBF than seen with 2-CA (2–6-fold versus vehicle, P < 0.05 in 10/12 regions of interest (ROIs)). Neither agonist altered CMRglu versus vehicle. After CCI, 2-CA increased CBF in ipsilateral hippocampal and hemispheric ROIs (P < 0.05 versus vehicle), but the response was attenuated at severe injury levels. We report marked increases in CBF after injection of adenosine receptor agonists into uninjured rat brain despite unaltered CMRglu. 2-Chloroadenosine produced enduring increases in CBF in uninjured brain and attenuated posttraumatic hypoperfusion. Future studies of adenosine-related therapies in CNS injury should address the role of CBF.
Introduction
Traumatic brain injury (TBI) acutely reduces cerebral blood flow (CBF) in experimental models and humans (Yamakami and McIntosh, 1989; Bouma et al, 1992). This may mediate secondary damage, since posttraumatic metabolic demands are increased (Hovda et al, 1995). Few experimental studies have targeted posttraumatic hypoperfusion. These studies have assessed effects of vasodilators (ethanol,
Adenosine receptor agonists increase CBF in normal brain. This occurs across species with intracarotid (Joshi et al, 2002), intravenous (Soricelli et al, 1995), topical (Ibayashi et al, 1991), or parenchymal (Van Wylen et al, 1989) administration. However, some investigators have reported differing results depending on the dose administered (Sollevi et al, 1987; Stange et al, 1997). Vasodilation by adenosine occurs through activation of adenosine-2a (A2A) receptors with adenyl cyclase activation in vascular smooth muscle (Kalaria and Harik, 1988). A2B receptor activation linked to nitric oxide synthase activity may also participate (Ngai et al, 2001; Shin et al, 2000).
Beneficial effects of the nonselective agonist 2-chloroadenosine (2-CA) on cerebral energy charge and functional outcome were shown after intraventricular or intraparenchymal administration in experimental TBI (Headrick et al, 1994; Varma et al, 2002). Benefit was also observed in experimental stroke (Evans et al, 1987) and epilepsy (Dunwiddie and Worth, 1982). Cerebral blood flow was not assessed in these studies. The A2A receptor agonist ATL-146e reduced damage in experimental spinal cord injury (Cassada et al, 2002a, b ), but CBF was not reported. Although the benefits of CBF promotion are anticipated, A2A receptor activation might be either beneficial or detrimental in CNS injury—including stroke (Simpson et al, 1992; Higashi et al, 2002; Chen et al, 1999; Aden et al, 2003), spinal cord injury (Cassada et al, 2002a), Parkinson's (Chen et al, 2001), and Huntington's disease (Popoli et al, 2002). Putative detrimental consequences of A2A receptor activation include augmented presynaptic glutamate release (Popoli et al, 2002), A1 receptor desensitization (Dixon et al, 1997) and leukocyte mediated effects (Yu et al, 2004). Since adenosine levels are high early after TBI—with activation of lower-affinity A2A receptors (Pedata et al, 2001; Bell et al, 1998), effects on CBF deserve investigation. To our knowledge, the effects of adenosine receptor agonists on CBF have not been reported in experimental TBI.
We previously used magnetic resonance imaging (MRI)—with continuous arterial spin labeling—to serially map CBF in traumatically injured rat brain (Forbes et al, 1997; Hendrich et al, 1999) and show that cortical injection of 2-CA acutely increased CBF in normal rat brain (Kochanek et al, 2001).
We currently report MRI measurements of CBF in uninjured sham rats early and at 24 h after intrahippocampal administration of either 2-CA or the selective A2A receptor agonist 2-p-(2-carboxyethyl)-phenethylamino-5'-N-ethylcarbox-amidoadenosine (CGS 21680). We also studied the effects of these agents on cerebral glucose utilization in uninjured shams using autoradiography. Finally, we studied the effect of 2-CA on CBF in the controlled cortical impact (CCI) model of TBI in rats. We hypothesized that adenosine receptor agonists would increase CBF in uninjured and injured rat brain, and that A2A receptor activation played an important role.
Materials and methods
This study protocol was approved by the University of Pittsburgh Animal Care and Use Committee. A total of 61 adult, male Sprague–Dawley rats (353 ± 24 g, Harlan, Indianapolis, IN, USA) were studied.
Specific Treatment Protocols
Uninjured rats: Four separate protocols were performed in uninjured rats subjected to craniotomy and intrahippocampal injection of adenosine receptor agonist or vehicle (Table 1). These included assessment of CBF at either ~3.5 and ~5 h or ~24 h after injection by MR or CMRglu at 3 h after injection by autoradiography.
Treatment protocols for all studies
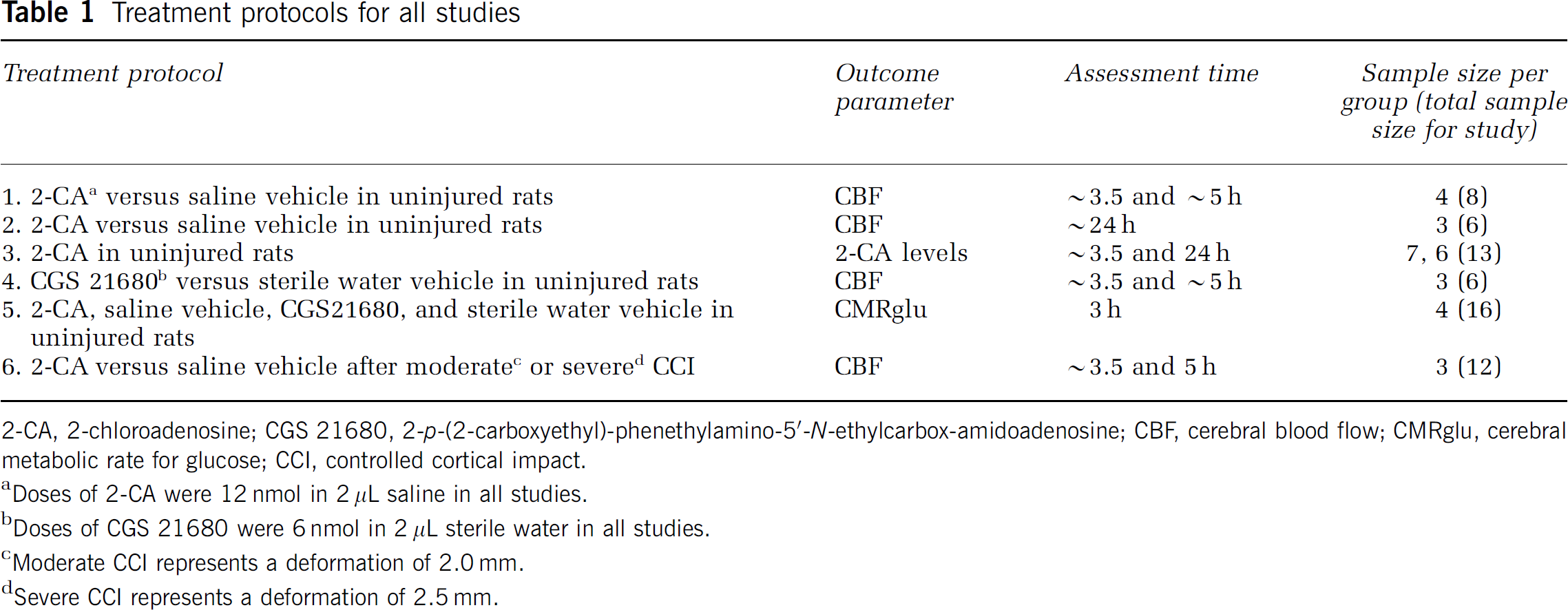
2-CA, 2-chloroadenosine; CGS 21680, 2-p-(2-carboxyethyl)-phenethylamino-5'-N-ethylcarbox-amidoadenosine; CBF, cerebral blood flow; CMRglu, cerebral metabolic rate for glucose; CCI, controlled cortical impact.
Doses of 2-CA were 12nmol in 2 µL saline in all studies.
Doses of CGS 21680 were 6 nmol in 2 µL sterile water in all studies.
Moderate CCI represents a deformation of 2.0 mm.
Severe CCI represents a deformation of 2.5 mm.
Controlled cortical impact model: A protocol comprised of four groups (Table 1) was used to study rats treated with either 2-CA or saline vehicle after CCI. Each treatment was assessed in groups subjected to either 2.0 mm deformation (moderate) or 2.5 mm deformation (severe) injury levels, with CBF assessed at ~3.5 and ~5 h after injury. An investigator (KH) masked to treatment performed MR data acquisition and analysis.
Surgical Preparation
For all protocols, male Sprague–Dawley rats had free access to food and water until the time of study. Anesthesia was induced in a plastic jar with 4% isoflurane (Anaquest, Memphis, TN, USA) in oxygen. The trachea was intubated with a 14-gauge angiocath and mechanically ventilated with 2% isoflurane in N2O:O2 (2:1). Sterile prep and drape were used for all surgical procedures. Femoral arterial and venous catheters (PE-50) were surgically inserted for continuous monitoring of blood pressure, arterial blood sampling (CCI protocol only), and administration of pancuronium bromide (0.1 mg/kg, Elkins-Sinn, Cherry Hill, NJ, USA). A rectal probe was inserted for continuous monitoring and maintenance of temperature at 37.0°C ± 0.5°C. Bicillin (100,000 U, Upjohn, Kalamazoo, MI, USA) and gentamicin (10 mg/kg, Elkins-Sinn, Cherry Hill, NJ, USA) were administered intramuscularly.
Rats in all protocol groups were randomized to treatment with an adenosine receptor agonist or appropriate vehicle, which was stereotactically injected (immediately after impact for the CCI group), through the craniotomy into the brain to a depth of 3.5 mm (−4.3 mm AP, −2.0 ML, relative to bregma) at a rate of 1 µL/min using a 30-gauge, 10 µL syringe (Hamilton, Reno, NV, USA). All injection volumes were 2 µL. Based on pilot studies, using Evans blue dye, this yielded drug or vehicle delivery to the center of the dorsal hippocampus. After injection, the needle was slowly withdrawn. The craniotomy was closed by replacing the bone flap and sealing the site with dental cement (Koldmount, Vernon Benshoff, Albany, NY, USA). The scalp incision was then closed. After surgery, anesthesia was discontinued, the temperature probe was removed, and the rats were weaned from mechanical ventilation and extubated within 1 h. In acute studies, catheters were fastened externally to the rat using a specially designed thoracic harness and thus maintained for re-use at the NMR center. 2-Chloroadenosine and CGS 21680 were purchased from RBI (Natic, MA, USA) and Sigma (St Louis, MO, USA), respectively. Drug doses are provided in Table 1.
The head was fixed in a stereotactic device (David Kopf, Tujunga, CA, USA). After retraction of the scalp, a craniotomy was made over the left parietal cortex with a dental drill, using the coronal and interparietal sutures as margins. For rats in the CCI model protocol groups, the bone flap was removed to zero the piston: it was then replaced, and the rats were allowed to equilibrate on isoflurane (1.1%) and N2O:O2 (2:1) for 30 mins. At 15 mins before CCI, an arterial blood sample was obtained to verify whether arterial blood gas tensions were within physiological limits (PaCO2 = 30 to 45 mm Hg; PaO2 > 90 mm Hg). Controlled cortical impact was performed as previously described (Dixon et al, 1999). Briefly, rats were subjected to injury to left parietal cortex (5-mm impactor tip, velocity of 4 m/secs, deformation depth of either 2.0 or 2.5 mm, impact duration of ~50 secs).
MR Studies of Cerebral Blood Flow
At the predefined time after either craniotomy or CCI, rats were transferred to the NMR center. Anesthesia was again induced using 4% isoflurane in O2. Rats were re-intubated with a 14-gauge angiocath and mechanically ventilated on 1% isoflurane in N2O:O2 (1:1), and pancuronium bromide (0.1 mg/kg h) was administered intravenously for immobilization. If rats were in the 24 h postinjection group (Protocol 2, Table 1), femoral arterial and venous catheters were surgically re-inserted at the NMR center. For rats studied in the acute postinjection or postinjury phase, vascular catheters were removed from the harness at the NMR center and reconnected to appropriate infusion or monitoring devices. Rats were placed in a cradle in the prone position. Motion was prevented by gently securing their heads in a custom-built holder with round-tipped ear bars and an adjustable bite bar. After placement in the magnet, the isoflurane and N2O were discontinued and a gas mixture of O2 in N2 (1:1) was initiated. An infusion of pentobarbital (37.5 mg/kg h, Abbot Laboratories, Chicago, IL, USA) was begun and rats were equilibrated on this anesthetic for a minimum of 30 mins before CBF acquisitions were initiated. Rectal temperature was controlled at 37°C ± 0.5°C and target PaCO2 was between 30 and 45 mm Hg (manipulated by adjusting ventilator rate and tidal volume). Arterial pO2, pH, and MABP were also measured immediately before and after each perfusion assessment.
Imaging was performed at 4.7 T on a 40-cm horizontal-bore Bruker AVANCE-DBX system, equipped with a 15-cm diameter shielded gradient insert and a 74-mm diameter RF coil. Localized shimming optimized homogeneity within a coronal slice ~3 mm posterior to bregma (field of view = 4 cm, slice thickness = 2 mm). Consistent positioning was confirmed on T2-weighted images and all subsequent data were acquired from this same plane.
Images were perfusion encoded by arterial spin labeling with continuous flow-induced adiabatic inversion before each phase-encoding step (Detre et al, 1992; Williams et al, 1992). A constant amplitude RF pulse was applied for 2 secs in the presence of an axial B0 field gradient (1 G/cm). Perfusion data were acquired in duplicate (spin-echo, TR = 2,000 ms, summation of TE = 10, 20, and 30 ms echoes, 2 averages, 128 × 70 zero-filled to 128 × 128). The inversion plane was positioned ± 2 cm from the detection plane for control and labeled images by offsetting the frequency of the labeling pulse ± 8,500 Hz. Spin-labeling efficiency (α) was measured in each study (Zhang et al, 1993). The spatial dependence of the spin-lattice relaxation time of tissue water in the presence of perfusion (T1 obs) was measured within the same slice from a series of spin-echo images acquired with variable TR (range = 100 to 8,000 ms, TE = 8.1 ms, 2 averages). T1 obs maps were generated for each study (Hendrich et al, 1999). At the completion of imaging, and while anesthesia was continued, rats were killed with an intravenous injection of a saturated solution of KCl.
Images were analyzed with the software package ‘Stimulate’ (Strupp, 1996) kindly provided by John Strupp (Center for MR Research, University of Minnesota Medical School, Minneapolis, MN, USA). Anatomical regions of interest (ROIs) within the slice were defined on the control perfusion image. An additional region was also identified that corresponds to the contused area in CCI studies; this ROI encompasses the portion of cortex extending 1 mm from midline to approximately halfway down the rhinal fissure, and is referred to as the medial cortical segment. Pixel-by-pixel maps of (MC—ML)/MC were generated, where MC is the magnetization intensity from each control image of the perfusion study and ML is the magnetization intensity from each labeled image. Negative pixel values within the brain were retained for quantification. To visually confirm that CBF calculations were not artifactually affected by potential changes in 1H density after injection, a map of (MC—ML)/MC was also generated using the first labeled image and the second control image—effectively reversing the acquisition order. The duplicate (MC—ML)/MC maps were then averaged and mean regional values were computed for each ROI within both the (MC—ML)/MC maps and the T1 obs maps. Cerebral blood flow was then calculated for each ROI from (Zhang et al, 1995):
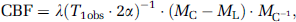
where λ is the brain–blood partition coefficient of water, with a spatially constant value of 0.9 mL/g assumed. A value of α = 0.7 was used for CBF quantification (mean and standard deviation of α in all MR studies was 0.68 ± 0.03). Pixels outside the brain were assigned to zero intensity for figure presentation.
Assessment of Brain Tissue Levels of 2-Chloroadenosine
Brain tissue levels of 2-CA were assessed in rats decapitated at either 3.5 (n = 7) or 24 h (n = 6) after intrahippocampal injection of 2-CA (12 nmol) using the protocol for injection exactly as described above. In each rat, the brain was rapidly removed and tissue was harvested from the left and right hippocampi and cortices mirroring the ROIs selected for imaging. The tissue was rapidly frozen in liquid nitrogen. Wet brain tissue was weighed (~50 mg), 100 µL of methanol and 10 µL of 200 pg/µL of phenacetin (internal standard) added, and the tissue homogenized. The homogenate was centrifuged and the supernatant collected. An equal volume of water was added to the supernatant, and 50 µL was assayed. A standard curve was performed in brain tissue from rats that had no treatment.
The assay was performed using a Thermofinnigan high-pressure liquid chromatograph system coupled to a Thermofinnigan LCQ Duo ion trap mass spectrometer equipped with an electrospray ionization (ESI) source. The analytical column was a C-18 Varian Microsorb. The mobile phase flow rate was 0.5 mL/min. The mobile phase changed from 100% water to 50% water/50% methanol over 5 mins with a linear gradient. This composition was kept constant for 2 mins, and then changed to 100% methanol over 1 min, and kept at this composition for 2 mins. The mobile phase was acidified with 0.1% formic acid. The mass spectrometer was operated in the ESI-positive ion mode. Nitrogen was used both as sheath and auxiliary gas at a pressure optimized automatically for each of the analytes during tuning of each analyte. The spray voltage and the heated capillary temperature were set at 5 kV and 270°C, respectively. Analysis was performed with two stages of ionization (MSMS), monitoring m/z of 170 for 2-CA (molecular ion m/z of 302 at 26% collision energy), and m/z of 138 for phenacetin (molecular ion m/z of 180 at 34% collision energy).
Deoxyglucose Autoradiography of Cerebral Glucose Utilization
Using anesthesia, surgery, and monitoring methods nearly identical to those in the experiments studying CBF by MR, separate groups of uninjured rats (craniotomy only) were injected with adenosine receptor agonists or respective vehicle and CMRglu was assessed by deoxyglucose autoradiography by the method of Sokoloff et al (1977), as modified by Kelly et al (2000), with minor additional modifications. At 3 h after injection of either 2-CA, CGS 21680, or their respective vehicles, 50 µCi of 14C-2-deoxy-
Statistical Analysis
Data are presented as mean ± s.d. or median (range), where appropriate. Physiological data were compared between groups using either a two-way (treatment and time) analysis of variance (ANOVA) for repeated measures or Kruskal–Wallis test. In each of the protocols examining uninjured rats with serial assessment of CBF, data were analyzed with a two-way ANOVA for repeated measures. In protocols with analysis of CBF or CMRglu at a single time point, a Kruskal–Wallis test was used. Because the effect of treatment, injury severity, and time after injury were simultaneously assessed in the CCI protocol, CBF data from that study were assessed using a multivariate analysis. Two multivariate models were created, one for absolute CBF values from each group and the second for left (ipsilateral) minus right (contralateral) CBF values. A P-value < 0.05 was considered significant.
Results
Uninjured Rats
Protocol 1. Effect of 2-chloroadenosine on cerebral blood flow in uninjured rats early after injection: Injection of 2-CA produced a marked, localized, and consistent 2–3-fold increase in CBF- versus saline vehicle-injected rats (Table 2, Figure 1). Significant increases in CBF for treated versus vehicle were seen ipsilateral to injection in the hippocampal, cortical, medial cortical segment, and hemispheric ROIs (two-way ANOVA for repeated measures). There was no significant effect of time or time–treatment interaction in any ROI in the injected hemisphere. Controlled and observed physiological parameters did not differ between 2-CA- and saline-injected groups, with two exceptions (Table 2). MABP was lower at both ~3.5 and 5 h in rats injected with 2-CA versus saline (P < 0.05), possibly a result of central A1 receptor activation (Tseng et al, 1988). However, this reduction was not below a MABP of 75 mm Hg in any rat. In addition, a statistically significant, albeit small (< 0.5°C), difference in rectal temperature was seen between groups. There was also no effect of time or treatment–time interaction for either MABP or temperature.
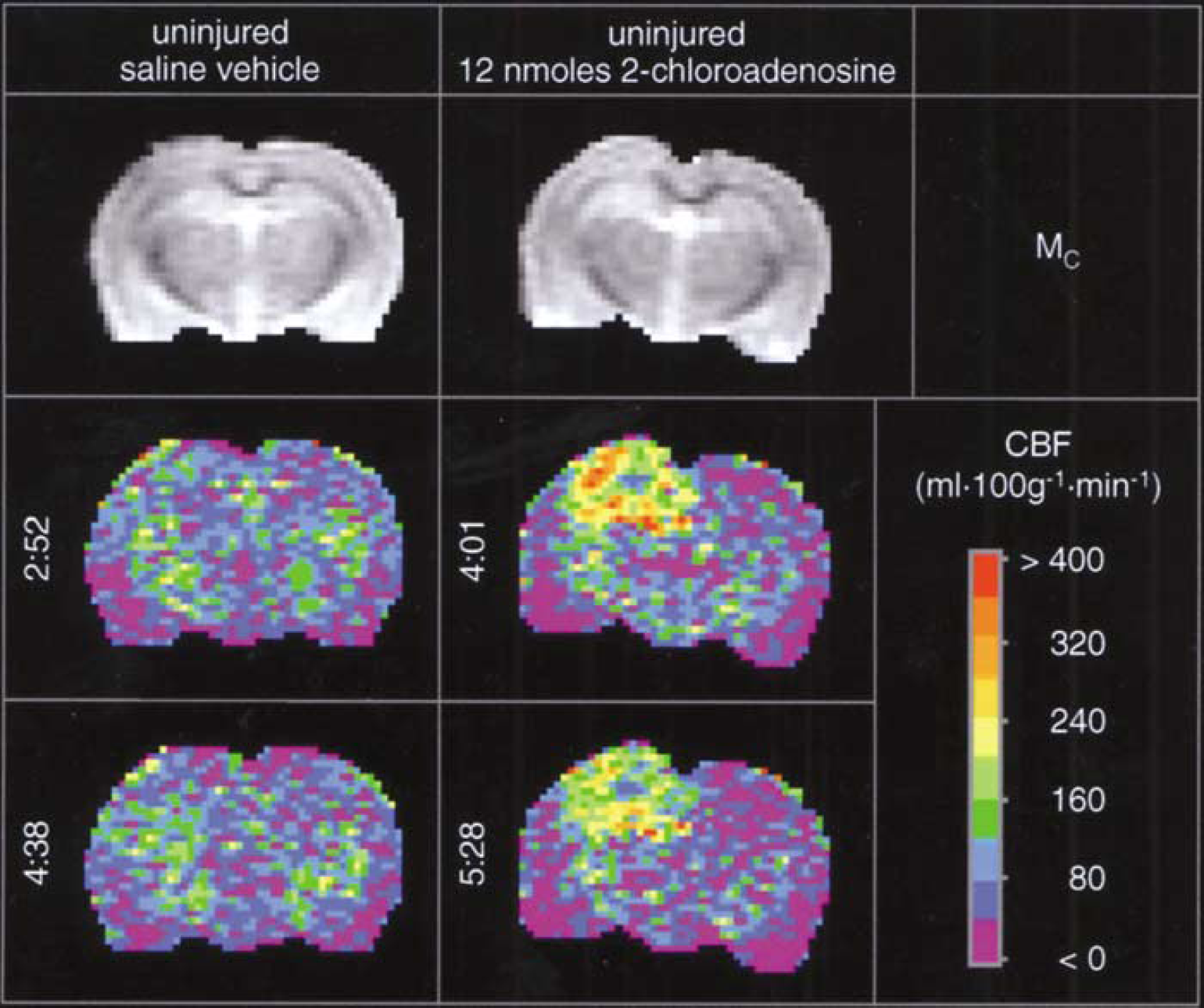
Acute effects of injection of either saline vehicle (left three vertical panels) or the nonselective adenosine receptor agonist 2-CA(12 nmol, right three vertical panels) into the left dorsal hippocampus of uninjured rats on CBF(mL 100 g−1 min−1) assessed by the continuous arterial spin-labeled MRI. The left hemisphere appears as the left side of image. The top horizontal panels show unlabeled control (Mc) images reflecting anatomy. Pseudocolor images are CBF maps. Numbers to the left of the images are times after injection. After injection of saline, CBF was not affected at either 2 h 52 mins or 4 h 38 mins, but at both 4 h 1 min and 5 h 28 mins after 2-CA administration CBF was increased in the ipsilateral hippocampus and overlying cortex in these representative examples.
Regional CBF (mL/100 g min) a and physiological parameters early after injection of either 2-CA or vehicle in uninjured rats
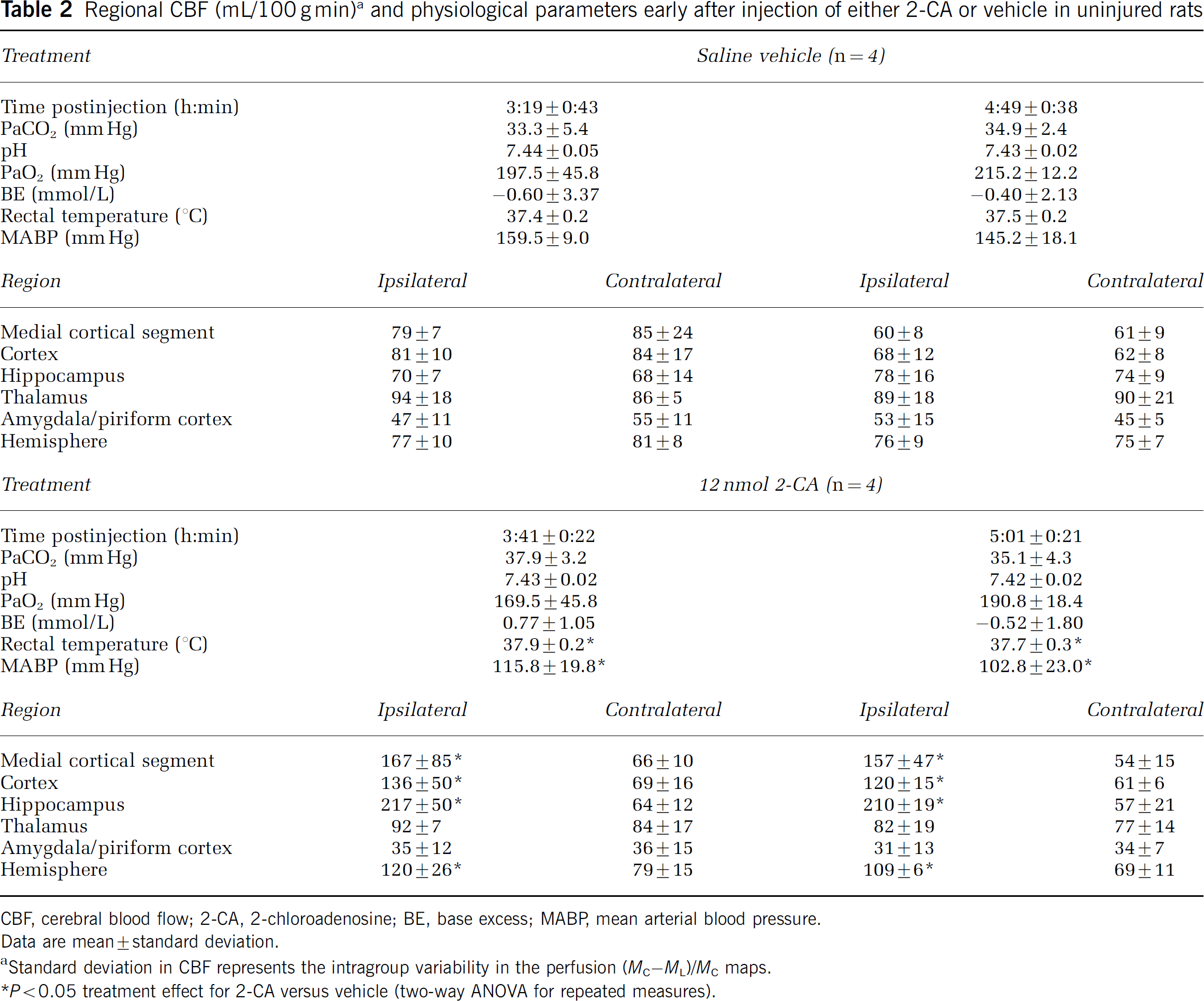
CBF, cerebral blood flow; 2-CA, 2-chloroadenosine; BE, base excess; MABP, mean arterial blood pressure.
Data are mean ± standard deviation.
Standard deviation in CBF represents the intragroup variability in the perfusion (MC–ML)/MC maps.
P < 0.05 treatment effect for 2-CA versus vehicle (two-way ANOVA for repeated measures).
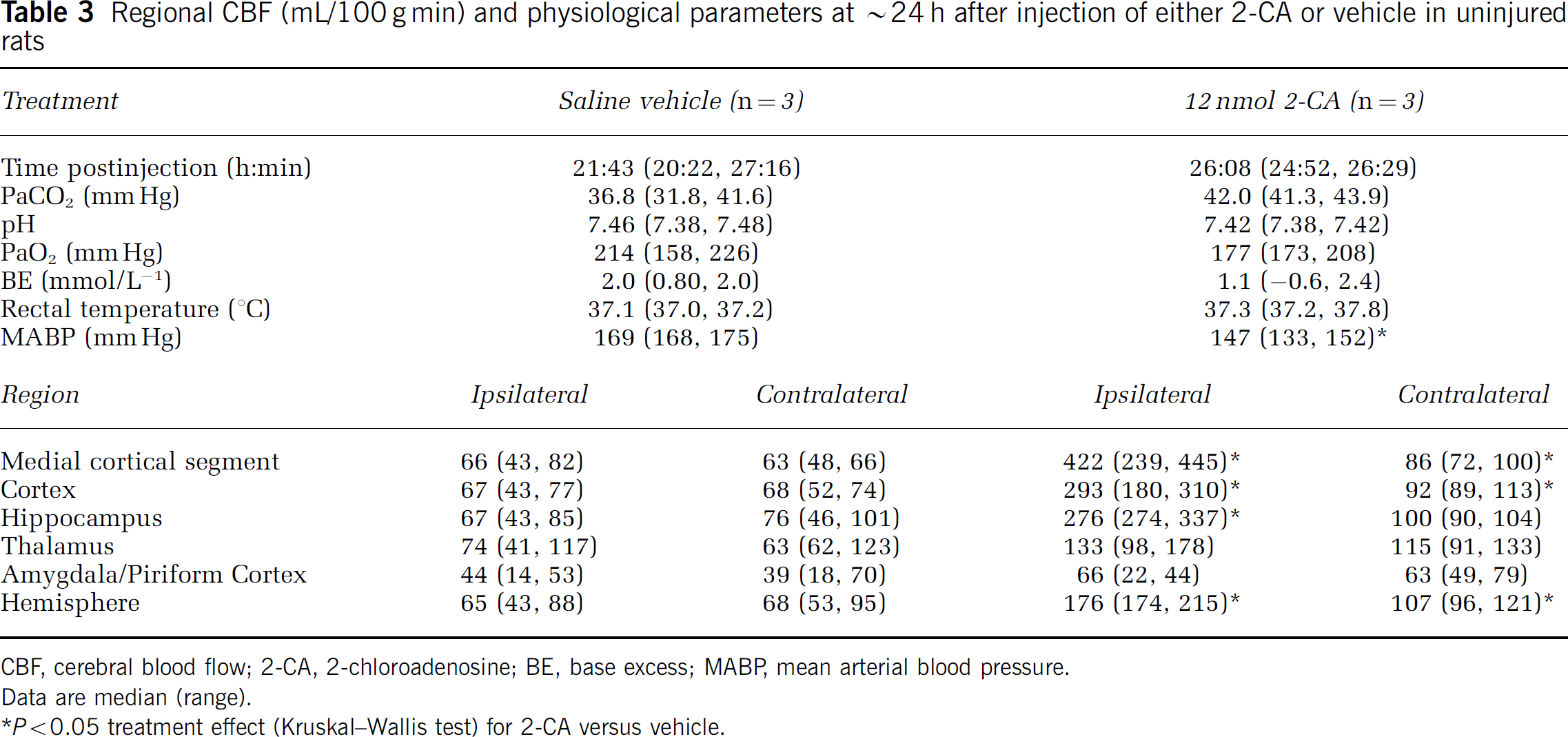
Protocol 2. Effect of 2-chloroadenosine on cerebral blood flow in uninjured rats at ~24 h after injection: Injection of 2-CA produced a remarkably sustained increase in CBF- versus saline vehicle-injected rats (Table 3, Figure 2). As in the studies assessing CBF early after injection, a consistent ~4-fold increase in CBF was seen in the hippocampal ROI ipsilateral to the injection site at ~24 h. Significant increases in CBF were also seen in cortical, medial cortical segment, and hemispheric ROIs ipsilateral to injection (Table 3, Kruskal–Wallis test). At 24 h after injection, significant albeit modest increases in CBF were also seen in three ROIs contralateral to injection (cortical, medial cortical segment, and hemispheric). Controlled and observed physiological parameters did not differ between 2-CA- and saline-injected groups, except again for an ~15% lower MABP at the time of CBF acquisition in 2-CA-versus vehicle-injected rats.
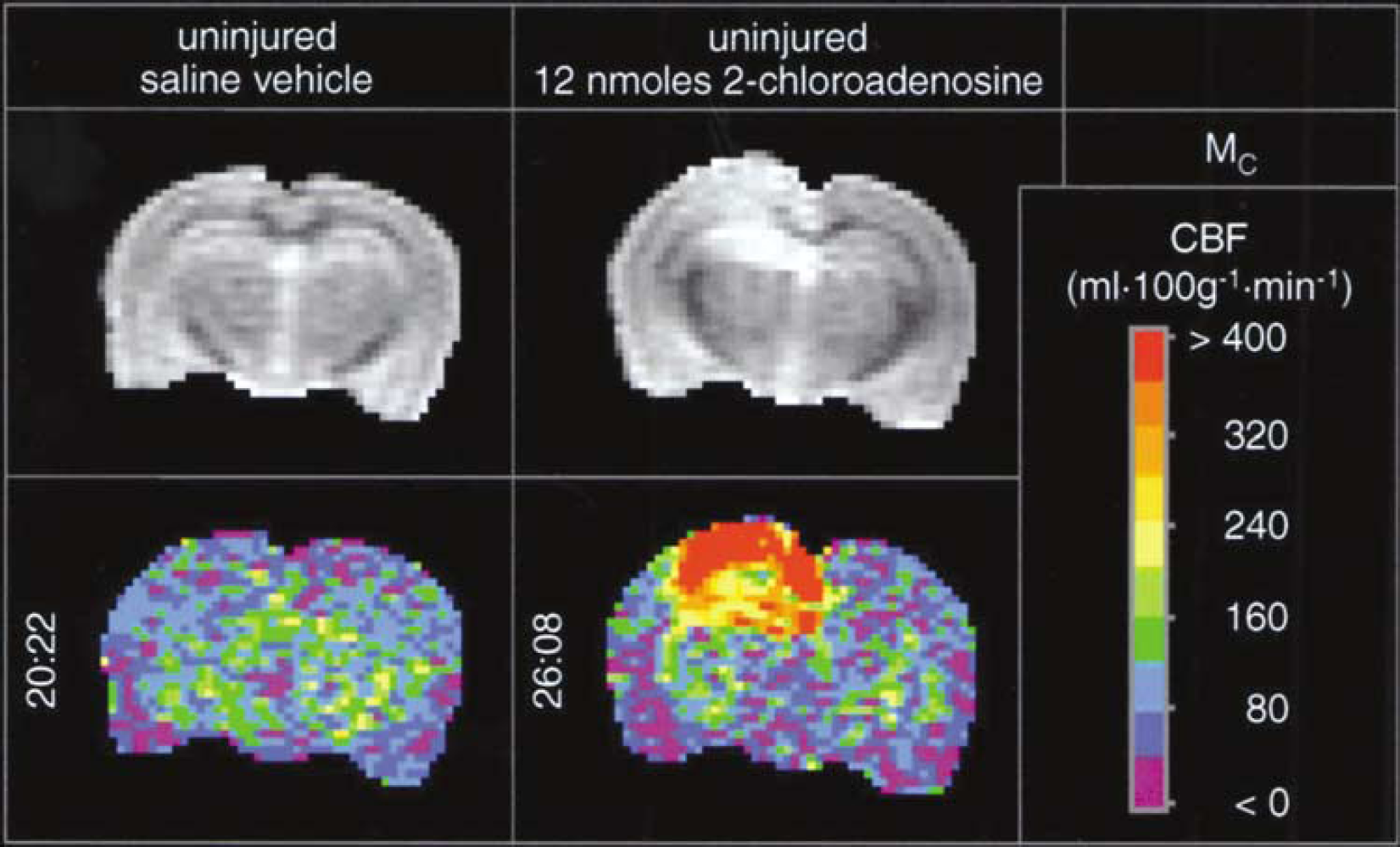
Delayed effects of injection of either saline vehicle (left two vertical panels) or the nonselective adenosine receptor agonist 2-CA(12 nmol, right two vertical panels) into the left dorsal hippocampus of uninjured rats on CBF (mL 100g−1 min−1), assessed by the continuous arterial spin-labeled MRI method. The left hemisphere appears as the left side of image. The top horizontal panels show unlabeled control (Mc) images reflecting anatomy. Pseudocolor images are CBF maps. Numbers to the left of the images are times after injection. After injection of saline, CBF was not affected at 20 h 22 mins, but at 26 h 8 mins after 2-CA administration CBF was increased in both the ipsilateral hippocampus and overlying cortex in these representative examples. This enduring effect on CBF was not anticipated, but is consistent with the sustained increases in brain tissue levels of 2-CA even at 24 h after injection.
Regional CBF (mL/100 g min) and physiological parameters at ~24 h after injection of either 2-CA or vehicle in uninjured rats
CBF, cerebral blood flow; 2-CA, 2-chloroadenosine; BE, base excess; MABP, mean arterial blood pressure.
Data are median (range).
P < 0.05 treatment effect (Kruskal–Wallis test) for 2-CA versus vehicle.
Protocol 3. Assessment of 2-chloroadenosine levels in brain tissue at either 3.5 h or 24 h after injection in uninjured rats: Brain tissue levels of 2-CA were 12,567 ± 6,702, 1,381 ± 1,231, 1,604 ± 3,511, and 274 ± 215 pg/mg wet brain tissue in the left hippocampus, right hippocampus, left cortex, and right cortex at 3.5 h after left hippocampal injection, P < 0.05 for left hippocampus versus all other regions. Similarly, brain tissue levels of 2-CA were 2,095 ± 951, 210 ± 148, 262 ± 144, and 44 ± 57 pg/mg wet brain tissue in the left hippocampus, right hippocampus, left cortex, and right cortex at 24 h after left hippocampal injection, P < 0.05 for left hippocampus versus all other regions.
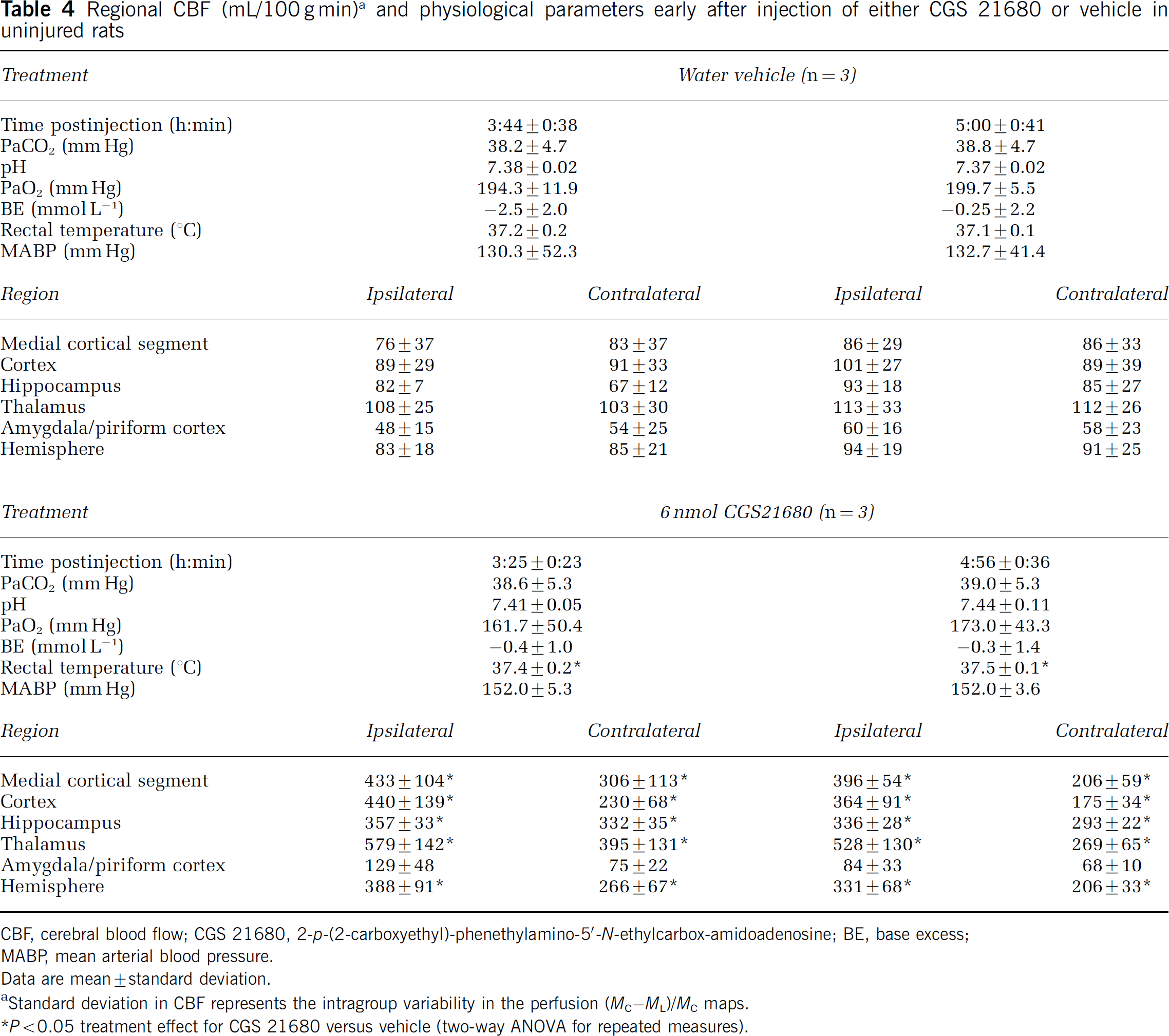
Protocol 4. Effect of CGS 21680 on cerebral blood flow in uninjured rats early after injection: intrahippocampal injection of the selective A2A receptor agonist CGS 21680 produced even more marked increases in CBF than seen after 2-CA injection. The dramatic increases in CBF in treated versus vehicle-injected rats were nearly global in the slice-reaching significance in 10 of the 12 ROIs that were assessed—with the exception of the ipsilateral and contralateral amygdala (Table 4, Figure 3, two-way ANOVA for repeated measures). For example, four-and five-fold increases in CBF for treatment versus vehicle were observed in ispilateral hippocampal and ipsilateral hemispheric ROIs, respectively. A 2–3-fold increase in CBF for treatment versus vehicle was seen in the hemispheric ROI contralateral to injection. Similarly, a significant effect of time was seen in the ipsilateral hippocampus, thalamus, amygdala, and hemisphere, and contralateral thalamus. A significant treatment–time interaction was also seen in 9 of the 12 ROIs, with higher CBF at ~3.5 versus ~5 h after CGS 21680 injection in these ROIs. Controlled and observed physiological parameters did not differ between CGS 21680- and vehicle-injected groups, except for a statistically significant, albeit small (< 0.5°C), difference in rectal temperature between groups at ~5 h after injection. There were no time effects or treatment–time interactions on physiological parameters.
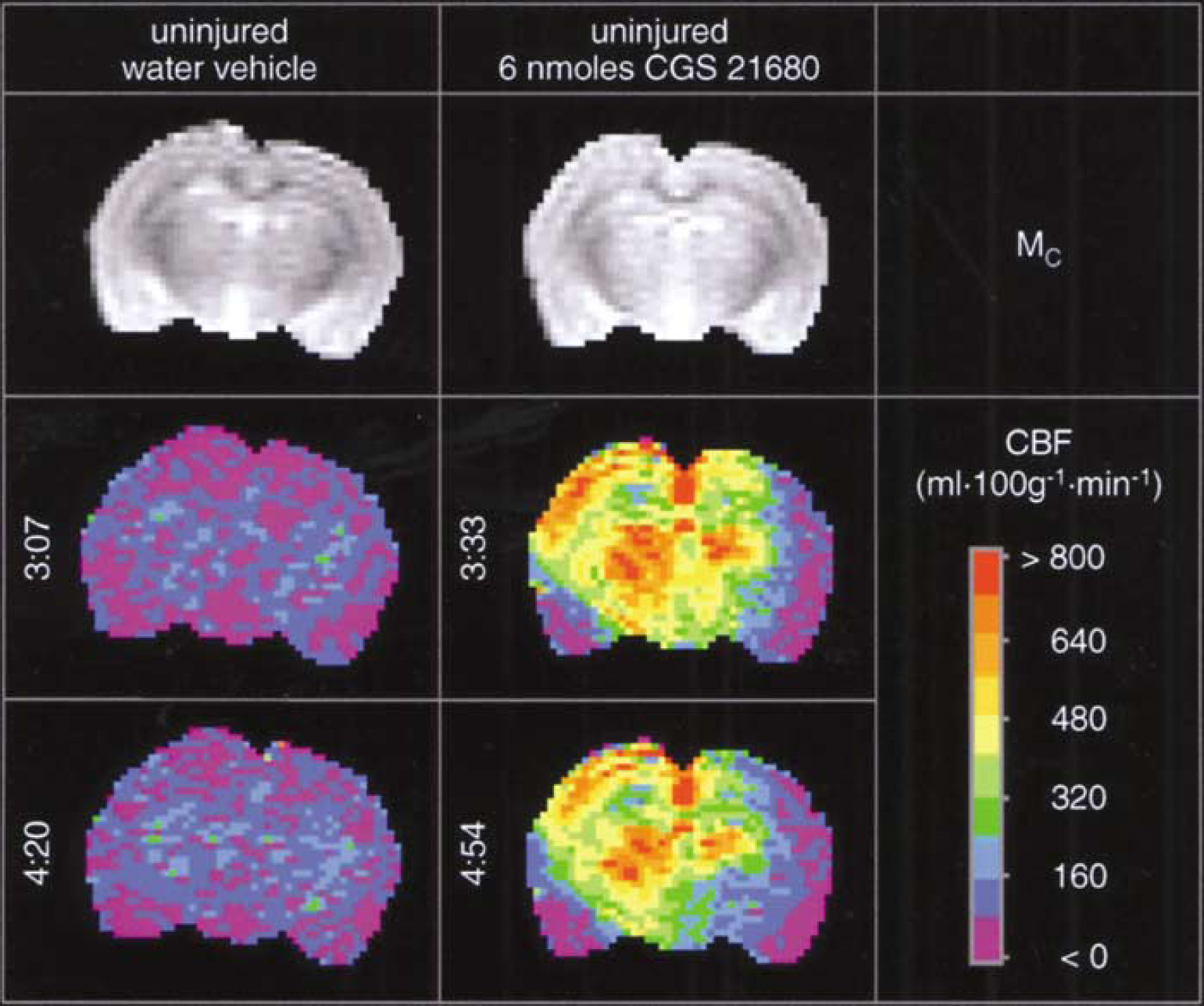
Acute effects of injection of either sterile water vehicle (left three vertical panels) or the selective adenosine2A receptor agonist, CGS 21680 (6 nmol, right three vertical panels) into the left dorsal hippocampus of uninjured rats on CBF (mL 100 g−1 min−1) assessed by the continuous arterial spin-labeled MRI method. The left hemisphere appears as the left side of image. The top horizontal panels show unlabeled control (Mc) images reflecting anatomy. Pseudocolor images are CBF maps. Numbers to the left of the images are times after injection. Cerebral blood flow was not affected at either 3 h 7 mins or 4 h 20 mins after injection of vehicle, but at both 3 h 33 mins and 4 h 54 mins after CGS 21680 administration a marked and nearly global increase in CBF is seen in most ROIs within the imaging slice in these representative examples.
Regional CBF (mL/100 g min) a and physiological parameters early after injection of either CGS 21680 or vehicle in uninjured rats
CBF, cerebral blood flow; CGS 21680, 2-p-(2-carboxyethyl)-phenethylamino-5‘-N-ethylcarbox-amidoadenosine; BE, base excess;
MABP, mean arterial blood pressure.
Data are mean ± standard deviation.
Standard deviation in CBF represents the intragroup variability in the perfusion (MC–ML)/MC maps.
P < 0.05 treatment effect for CGS 21680 versus vehicle (two-way ANOVA for repeated measures).
Protocol 5. Effect of 2-chloroadenosine or CGS 21680 on CMRglu in uninjured rats early after injection: Despite marked increases in CBF at ~3 h after injection of either 2-CA or CGS 21680 versus the respective vehicle, neither treatment significantly altered CMRglu in any ROI in pentobarbital anesthetized rats—including ipsilateral hippocampus, cortex, and thalamus (Table 5, Kruskal–Wallis test). Controlled and observed physiological parameters did not differ between groups.
Regional CMRglu (µmol/100 g min) and physiological parameters at 3 h after injection of either adenosine agonists or vehicle in uninjured rats
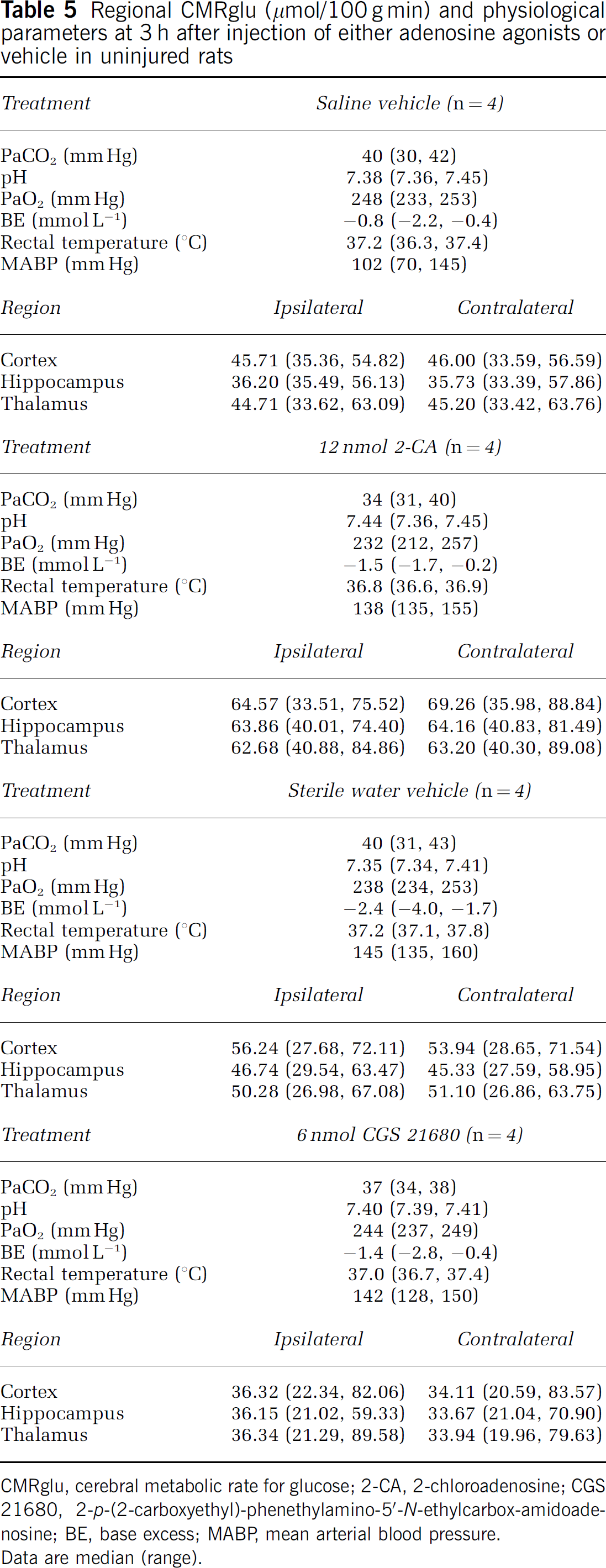
CMRglu, cerebral metabolic rate for glucose; 2-CA, 2-chloroadenosine; CGS 21680, 2-p-(2-carboxyethyl)-phenethylamino-5‘-N-ethylcarbox-amidoade-nosine; BE, base excess; MABP, mean arterial blood pressure.
Data are median (range).
Controlled Cortical Impact Model
Protocol 6. Effect of 2-chloroadenosine on cerebral blood flow in rats after CCI: effect of injury severity and time: Cerebral blood flow in individual ROIs is shown for studies in vehicle- and 2-CA-treated rats after moderate (Table 6) and severe (Table 7) CCI. Representative examples show that the focal increases in CBF produced by 2-CA after CCI (Figures 4 and 5) were more heterogeneous than those previously shown in uninjured rats. Multivariate analysis (Table 8) revealed that posttraumatic increases in CBF mediated by 2-CA were local, reaching significance in the ipsilateral hippocampal and hemispheric ROIs (both P < 0.005 for 2-CA versus vehicle). Multivariate analyses also confirmed that CBF recovery in ipsilateral medial cortical segment, cortex, and hemispheric ROIs after CCI was dependent on the severity of the injury (all P < 0.005 for moderate versus severe injury), with lower flows being observed at the more severe injury level. Similarly, CBF recovery in the ipsilateral hippocampus, medial cortical segment, cortex, and hemispheric ROIs after CCI was also dependent on time after injury (all P < 0.005 for first (~3.5 h) versus second (~5 h) CBF assessment) when assessing this parameter using injured minus noninjured values in respective ROIs. Physiologic parameters were not different between treatment groups, except for a statistically significant, albeit modest difference in temperature between treatment and control groups at the time of CBF acquisition (two-way ANOVA for repeated measures). Mean rectal temperature was never more than 0.7°C different between groups (Tables 6 and 7).
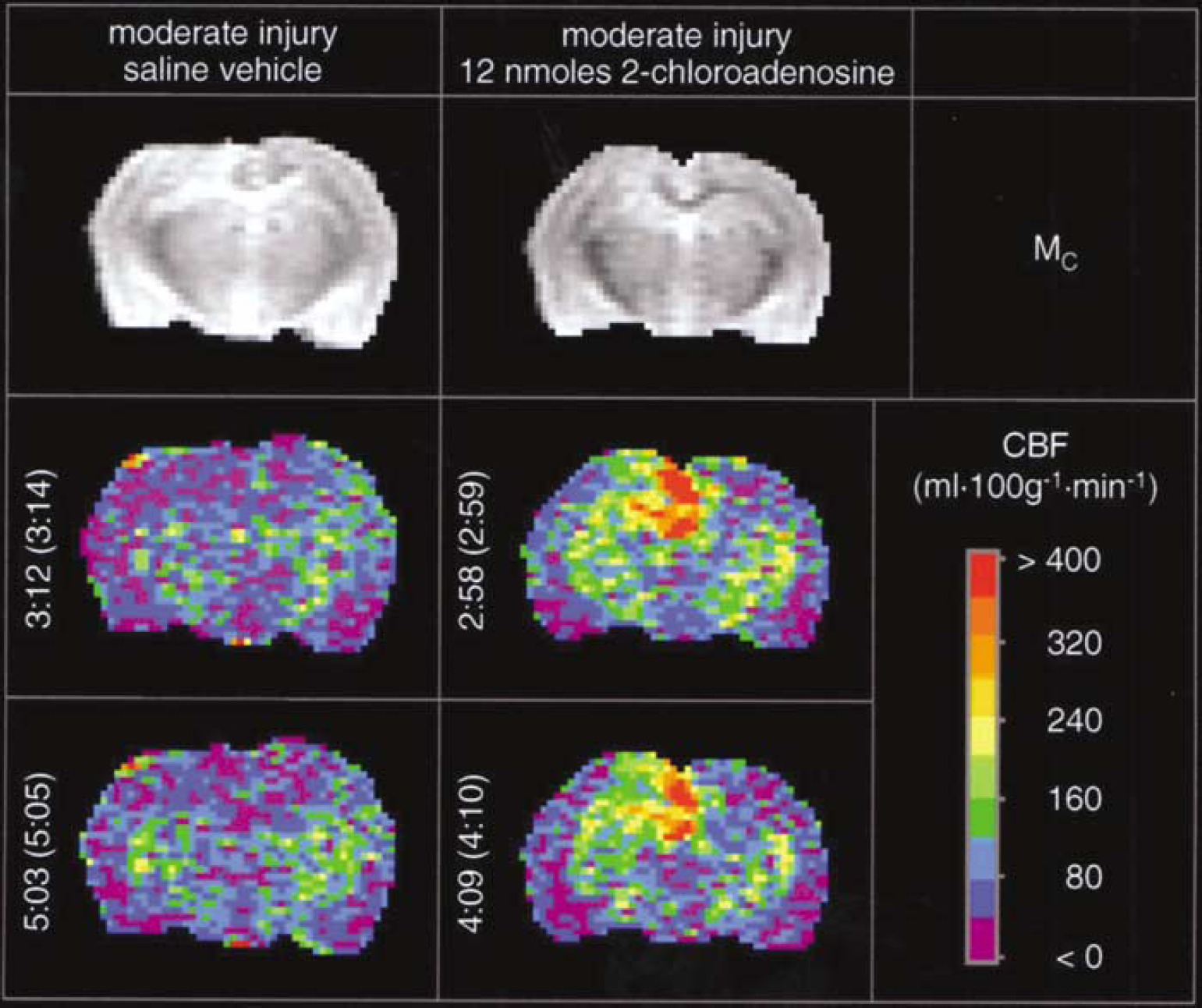
Acute effects of injection of either saline vehicle (left three vertical panels) or the nonselective adenosine receptor agonist 2-CA (12 nmol, right three vertical panels) into the left dorsal hippocampus of rats on CBF (mL 100 g−1 min−1, assessed by the continuous arterial spin-labeled MRI) after CCI at a moderate (2.0 mm deformation) injury level. The left hemisphere appears as the left side of image. The top horizontal panels show unlabeled control (Mc) images reflecting anatomy. Pseudocolor images are CBF maps. Numbers to the left of the images are times after injection, with time after CCI indicated in parentheses. Some local swelling in the Mc images at the injury site is seen after moderate injury. Cerebral blood flow was not affected at either 3 h 12 mins or 5 h 3 mins after injection of saline, but at both 2 h 58 mins and 4 h 9 mins after 2-CA administration CBF was increased in the ipsilateral hippocampus, overlying cortex, and brain regions in proximity to the contusion in these representative examples at the moderate injury level.
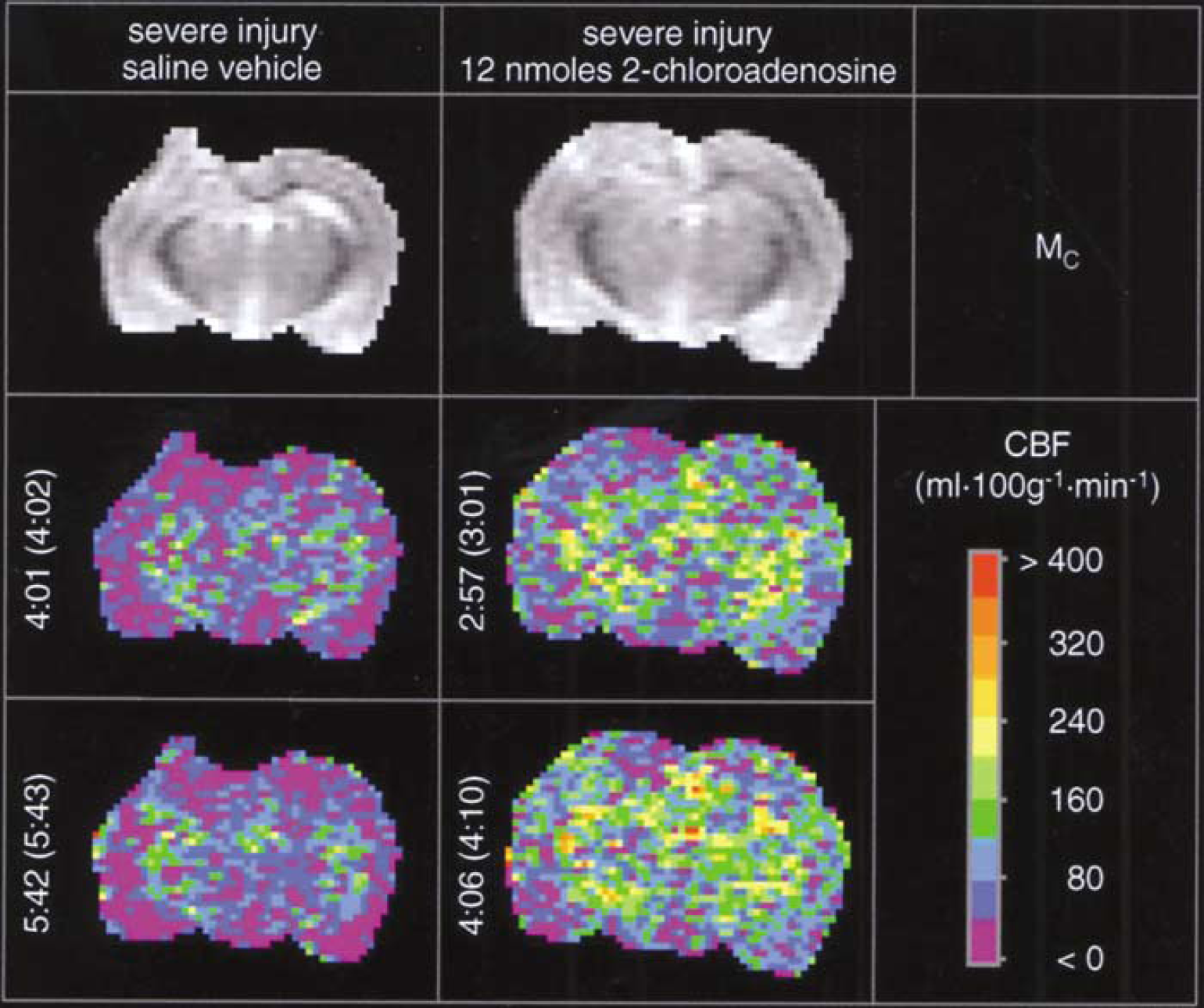
Acute effects of injection of either saline vehicle (left three vertical panels) or the nonselective adenosine receptor agonist 2-CA (12 nmol, right three vertical panels) into the left dorsal hippocampus of rats on CBF (assessed by the continuous arterial spin-labeled MRI) after CCI at a severe (2.5 mm deformation) injury level. The left hemisphere appears as the left side of image. The top horizontal panels show unlabeled control (Mc) images reflecting anatomy. Pseudocolor images are CBF maps. Numbers to the left of the images are time after injection, with time after CCI indicated in parentheses. Marked local swelling in the Mc images at the injury site is readily seen after severe injury. Cerebral blood flow was not affected at either 4 h 1 min or 5 h 42 mins after injection of saline, and hypoperfusion is seen at the contusion site. At both 2 h 57 mins and 4 h 6 mins after 2-CA administration, less posttraumatic hypoperfusion is evident in brain regions in proximity to the contusion in these representative examples at the severe injury level.
Regional CBF (mL/100 gmin) a and physiological parameters after CCI (deformation depth = 2.0 mm) of moderate injury level: studies of 2-CA versus vehicle
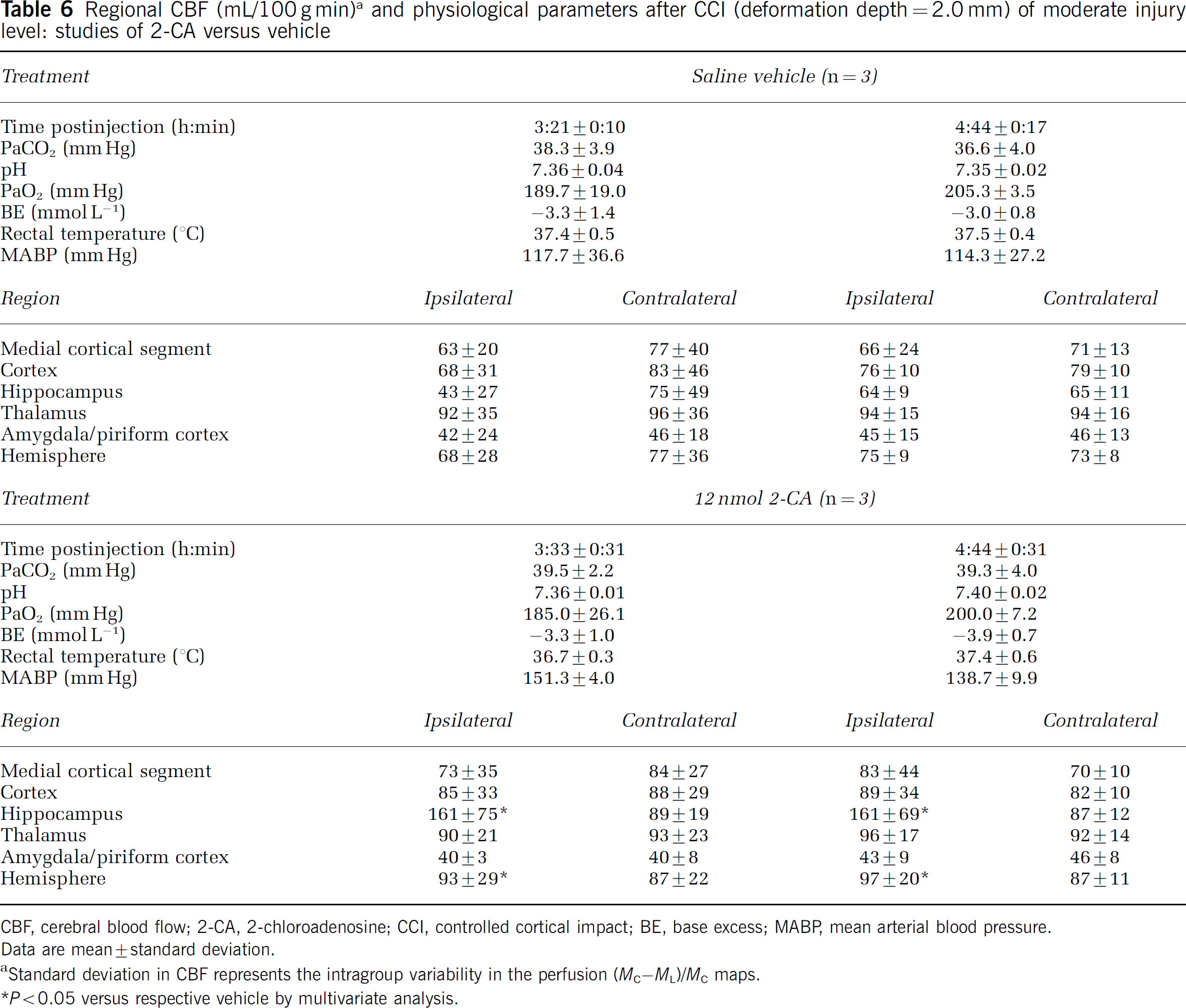
CBF, cerebral blood flow; 2-CA, 2-chloroadenosine; CCI, controlled cortical impact; BE, base excess; MABP, mean arterial blood pressure.
Data are mean ± standard deviation.
Standard deviation in CBF represents the intragroup variability in the perfusion (MC–ML)/MC maps.
P < 0.05 versus respective vehicle by multivariate analysis.
Regional CBF (mL/100 g min) a and physiological parameters after CCI (deformation depth = 2.5 mm) of severe injury level: studies of 2-CA versus vehicle
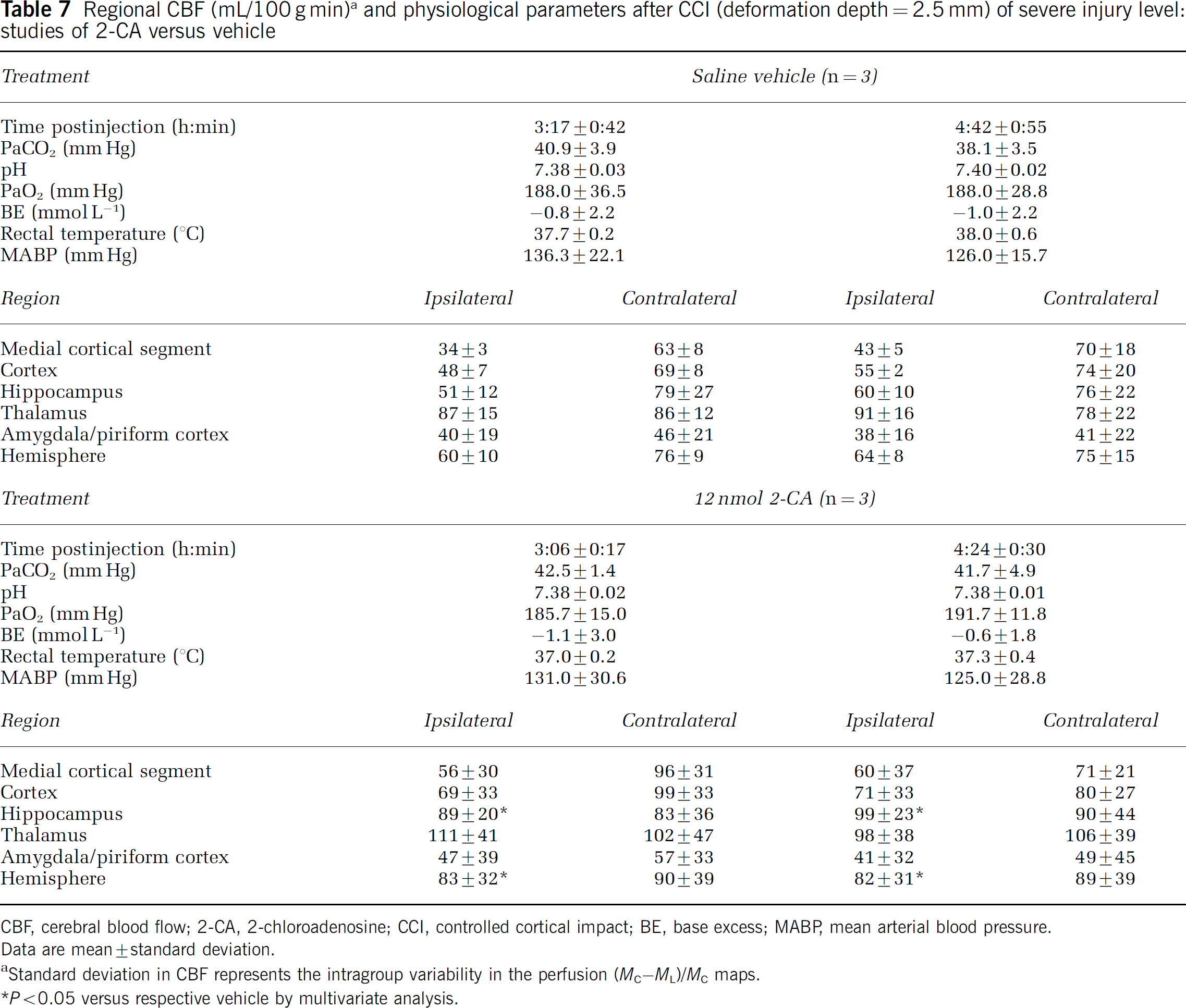
CBF, cerebral blood flow; 2-CA, 2-chloroadenosine; CCI, controlled cortical impact; BE, base excess; MABP, mean arterial blood pressure.
Data are mean ± standard deviation.
Standard deviation in CBF represents the intragroup variability in the perfusion (MC–ML)/MC maps.
P < 0.05 versus respective vehicle by multivariate analysis.
Discussion
Effect of 2-Chloroadenosine on Cerebral Blood Flow in Uninjured Rat Brain
The marked increase in regional CBF produced by intrahippocampal injection of 2-CA in uninjured rat brain expands the findings of our prior study using cortical injection (Kochanek et al, 2001). 2-Chloroadenosine is a nonselective adenosine receptor agonist with receptor affinities similar to those of adenosine. Despite the use of identical doses in this and our prior report (Kochanek et al, 2001), intrahippocampal administration of 2-CA produced a more localized distribution of increase in CBF than cortical administration. Early increases in CBF were restricted to the injected hemisphere. This increase in CBF in our study is consistent with the ~2.9-fold increase produced by infusion of a 10− 4 mol/L solution of 2-CA into the caudate nucleus in rats (Van Wylen et al, 1989). Our 24 h studies revealed that the increase in CBF is remarkably enduring. Cerebral blood flow was at least as high in the ipsilateral hippocampus and cortex at 24 h after injection as seen early after injection. Prolonged effects of 2-CA on CBF have not received prior study. However, 2-CA is a relatively nonmetabolizable analog and parenchymal injection into caudate nucleus produces anticonvulsant effects that last up to three days (Abdul-Ghani et al, 1997). Consistent with those studies, 2-CA levels were still 2,095 ± 951 pg/mg wet brain tissue in hippocampus ipsilateral to injection at 24 h. This is equal to 6.94 pmol/mg tissue—which is ~1 µL of tissue volume. This represents a 2-CA concentration of 6.94 µmol/L. The equilibrium dissociation constant of 2-CA for A2A receptors is 0.46 µmol/L. Therefore, the concentration of 2-CA in hippocampus at 24 h after injection is 15-fold greater than the equilibrium dissociation constant and would provide near-saturation levels at A2A receptors. Even cortical levels were in a range that would activate A2A receptors at 3.5 and 24 h after injection. The low lipid solubility of 2-CA may also delay parenchymal clearance (De Sarro et al, 1991). Our data, thus, implicate sustained A2A receptor activation in mediating the sustained increase in CBF. MABP was lower in rats after intrahippocampal injection of 2-CA versus saline vehicle. This finding was surprising since our prior report did not reveal an effect of intracortical injection of 2-CA on MABP (Kochanek et al, 2001). Few prior experiments studied the 12 nmol dose. This effect likely results from central A1-receptor-mediated effects on sympathetic tone (Tseng et al, 1988). This modest MABP reduction would not be expected to increase CBF in uninjured rat brain. The focal nature of the increases in CBF supports local effects of 2-CA on CBF. Marked increases in CBF, without effects on MABP in the studies of CGS 21680, support potent local effects of adenosine agonists on CBF.
Effect of the A2A Receptor Agonist CGS 21680 on Cerebral Blood Flow in Uninjured Rat Brain
Cerebrovasodilatory effects of adenosine are mediated by actions at A2A and A2B receptors located on vascular smooth muscle and endothelium (Ngai et al, 2001; Shin et al, 2000). Before studying 2-CA in experimental TBI, we thought it important to show that local administration of a selective A2A receptor agonist increases CBF in our paradigm. CGS 21680 exhibits ~140-fold selectivity for the A2A versus A1 receptor (Hutchison et al, 1989). Applied to the cortical surface of the rat brain, CGS 21680 increased CBF (Coney and Marshall, 1988). The CGS 21680 dose of 6 nmol was based on pilot studies. Parenchymal injection produced marked increases in CBF in nearly every ROI—in both hemispheres—suggesting rapid diffusion throughout the brain and the dramatic effects of A2A receptor activation. In isolated rat cerebral arterioles, the A2A receptor antagonist ZM-241385 only partially reversed vasodilation by adenosine, suggesting a possible contribution of A2B receptor activation (Ngai et al, 2001). We did not study the effect of A2B receptor activation because of the lack of availability of a selective agonist. We also did not carry out studies combining A2A agonists and antagonists in our model. Such studies could contribute additional mechanistic insight. CGS 21680 does not cross the blood–brain barrier (Jacobson and Van Rhee, 1997) and no effect on MABP was seen.
Effect of Adenosine Receptor Agonists on CMRglu in Uninjured Rat Brain
Neither 2-CA nor CGS 21680 affected CMRglu in any ROI in pentobarbital-anesthetized rats. This suggests that the vasodilatory effects of intrahippocampal injection of either 2-CA or CGS 21680 do not result from an increase in CMR. Our CMRglu values in vehicle-treated rats are similar to those reported by Warner et al (1996). Metabolic suppression would be expected after administration of the nonselective agonist 2-CA, based on studies showing potent anticonvulsant actions of 2-CA (Pourgholami et al, 1997; De Sarro et al, 1991). Although studies of the effects of adenosine receptor agonists on CMRglu using a similar paradigm (i.e., intraparenchymal administration, pentobarbital anesthesia) have not, to our knowledge, been reported, McBean et al (1989) reported that a 15-min intracarotid infusion of 2-CA (10−9 mol/min) had no effect on CMRglu in halothane-anesthetized rats. An increase in CMRglu might be expected after parenchymal injection of CGS 21680, based on its ability to facilitate synaptic transmission by ~14% in rat hippocampal slices (Cunha and Ribeiro, 2000). However, intravenous administration of CGS 21680 reduced CMRglu in unanesthetized rats (Nehlig et al, 1994). We saw no effect of intrahippocampal administration of this agent on CMRglu—despite a marked global increase in CBF.
Effect of 2-Chloroadenosine on Cerebral Blood Flow after Traumatic Brain Injury
We examined 2-CA rather than the selective A2A receptor agonist CGS 21680 after TBI because 2-CA has been reported to reduce functional deficits in both the fluid percussion and CCI models (Headrick et al, 1994; Varma et al, 2002). We observed that CBF promotion by 2-CA after TBI depends on injury severity. After moderate CCI, 2-CA produced supranormal CBF in the ipsilateral hippocampal ROI—however, this response is less pronounced than that seen after injection of 2-CA in uninjured hippocampus. After severe CCI, mean CBF levels in hippocampus ipsilateral to 2-CA injection were increased versus vehicle-injected groups. However, CBF was similar to uninjured values in our model. This suggests that 2-CA was able to restore CBF only to the normal range early after severe injury. Early posttraumatic hypoperfusion is common in experimental and clinical TBI, and may mediate secondary damage (Yamakami and McIntosh, 1989; Bryan et al, 1995; Bouma et al, 1992); however, the optimal level of CBF early after injury is not known, and changes in metabolic demands are temporally complex. Kelly et al (2000), using a moderate CCI injury level, reported a marked initial increase in CMRglu, followed by metabolic depression by 3 h. Headrick et al (1994) reported that ICV administration of 5 nmol of 2-CA in rats attenuated the loss in phosphorylation potential at 2 to 4 h after fluid-percussion injury. Taken with our study and the work of Varma et al (2002), after experimental TBI, beneficial effects of local administration of 2-CA are seen on CBF, cerebral metabolism, and functional outcome. Multivariate analysis also revealed an effect of time after injury on CBF. Most investigators have reported an initial reduction in CBF, followed by progressive recovery after experimental TBI (Yamakami and McIntosh, 1989; Kochanek et al, 1995). In saline-treated rats in this study, after severe CCI, CBF recovered by ~5% to 50% between ~3.5 and ~5 h in ROIs beneath the impact site. Finally, the spatial pattern of the effect of 2-CA on CBF was less consistent after CCI versus the uninjured condition. This could result from variable vascular responsivity, vascular disruption, or alterations in drug distribution after injury. Brain interstitial levels of adenosine are increased 61-fold after severe CCI in our model in the initial 30 mins, however; they nearly normalize by 40 mins (Bell et al, 1998). The increase in CBF seen after 2-CA administration in our paradigm suggests that, by 3.5 h after injury, A2A receptors are unlikely to be saturated by endogenous adenosine. However, the time course of changes in the distribution and response of adenosine receptors in rat brain after TBI remain undefined.
Limitations
First, we administered the adenosine receptor agonists by intrahippocampal injection to minimize systemic effects. This may limit the clinical relevance of these findings. Although we are not suggesting clinical testing of this approach based on these initial studies, ventriculostomy drainage is routinely used in clinical TBI. Intraventricular administration of adenosine agonists would be feasible. Second, we did not assess ICP. Some studies of CBF after experimental TBI in rats have assessed ICP (Kroppenstedt et al, 2002; Cherian et al, 1999); most have not (Yamakami and McIntosh, 1989; Bryan et al, 1995; Kelly et al, 2000; Thomale et al, 2002). The use of MRI placed additional limits on monitoring capabilities. We cannot prove that the local increase in CBF by 2-CA after CCI was not limited by intracranial hypertension. Third, CGS 21680 is the most selective commercially available A2A agonist. However, it also binds to a second high-affinity site in the hippocampus and cortex, distinct from the A2A receptor (Cunha et al, 1996). Nevertheless, it is used in most studies of the role of this receptor (Ngai et al, 2001; Shin et al, 2000). Fourth, we did not study CBF immediately after injury. Future studies should pursue earlier CBF effects. Fifth, as discussed above, the increase in CBF after administration of 2-CA was attenuated at the more severe injury level. This could importantly limit its efficacy in severely contused brain regions, where the vasculature might be unresponsive. The responsivity of isolated cerebral vessels in rat is directly related to injury severity (Golding et al, 2002). Fluid percussion injury has been shown to blunt both G-protein-mediated vasodilatation and agonist-mediated elevations in cyclic nucleotides (Armstead, 1998, 2003), both of which could be relevant to the attenuation of 2-CA-mediated effects on CBF at the higher injury level. Sixth, we chose the dose of 2-CA based on our prior studies in rats assessing effects on CBF (Kochanek et al, 2001). However, the optimal dose of any adenosine receptor agonist after CCI in our model remains to be determined in studies assessing outcome. Finally, the suggestion to augment CBF after TBI through via A2A receptor activation must be reconciled with findings that in some studies A2A receptor antagonists are neuroprotective in models of cerebral ischemia and Parkinson's and Huntington's disease (O'Regan et al, 1992; Higashi et al, 2002; Popoli et al, 2002).
Multivariate analyses of a CBF after CCI; effects of treatment, time after injury, and severity
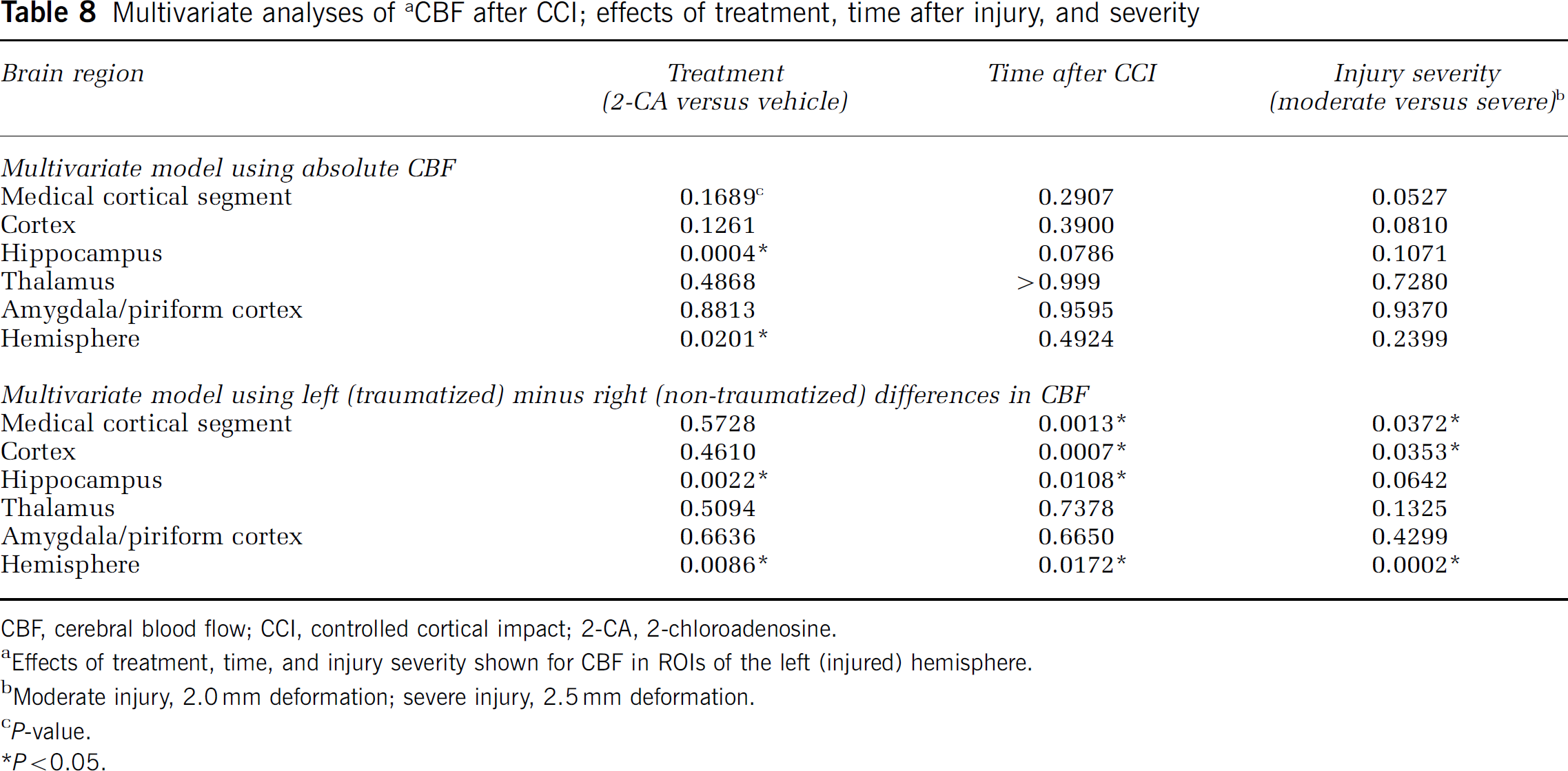
CBF, cerebral blood flow; CCI, controlled cortical impact; 2-CA, 2-chloroadenosine.
Effects of treatment, time, and injury severity shown for CBF in ROIs of the left (injured) hemisphere.
Moderate injury, 2.0 mm deformation; severe injury, 2.5 mm deformation.
P-value.
P < 0.05.
Conclusions
Using MRI, we report marked, regional, and enduring increases in CBF after intrahippocampal injection of the nonselective adenosine receptor agonist 2-CA into uninjured rat brain. Marked global increases in CBF are seen after administration of the selective A2A receptor agonist CGS 21680. Neither agonist affected CMRglu. 2-Chloroadenosine attenuated posttraumatic hypoperfusion after experimental TBI. Future studies assessing the effect of adenosine-related therapies in CNS injury should address the potential contribution of important effects on CBF.
Footnotes
Acknowledgements
The authors thank Dr Larry Jenkins for helpful review and Marci Provins for assistance with preparation of the manuscript.
