Abstract
The aim of our study was to visualize developing vessel occlusion in focal cerebral ischemia
Introduction
Cerebral infarction is caused by temporary or permanent occlusion of brain vessels, which leads to reduced intracerebral blood flow and decreased oxygen supply. Within minutes, irreversible tissue damage develops in brain areas which hemodynamically depend on the occluded vessels, and further extends into the periphery (‘penumbra‘) with time (Dirnagl et al, 1999). This secondary and delayed infarct growth has been attributed to increased excitotoxicity, inflammatory tissue damage and neuronal apoptosis (Dirnagl et al, 1999). Salvation of the penumbra is of particular therapeutic interest in stroke research. Diffusion-weighted magnetic-resonance imaging (DW-MRI) provides valuable information regarding the extent of ischemic tissue damage, but cannot depict the underlying vessel occlusion (Warach et al, 1995). Perfusion-weighted MRI (PW-MRI) in conjunction with DW-MRI gives an estimate of a potential mismatch between the infarct core and the surrounding tissue at risk (Schlaug et al, 1999; Neumann-Haefelin et al, 2000). Development of MR techniques allowing direct
In our present study, we report on ‘early’ iron enhanced MRI as
Materials and methods
Animal Experiments
All animal experiments were performed in accordance with institutional guidelines and were approved by Bavarian state authorities. Focal cerebral ischemia was induced in 43 adult male Wistar rats (200 to 220 g) by PT of cortical microvessels under inhalation anesthesia with enflurane in a 2:1 nitrogen/oxygen atmosphere, as described previously (Watson et al, 1985; Jander et al, 1995). A fibre-optic bundle of a cold light source was centred stereotactically 4 mm posterior and 4 mm lateral from Bregma on the intact skull. After that, 0.4 mL of a sterilefiltered Rose Bengal solution (10 g/L; Sigma, Deisenhofen, Germany) was administered via a femoral vein catheter, and the brain was illuminated for 20 mins. This procedure resulted in cone-shaped cortical infarctions without clinically visible symptoms. At the end of the operation, subcutaneous fat and skin were sutured in anatomical layers and animals were placed in separate cages for recovery. During the operation procedure, rectal temperature was kept between 36.5°C and 37.5°C by a servocontrolled heating blanket. In a subgroup of animals (
Magnetic Resonance Imaging
All measurements were performed on a clinical 1.5 T MRT unit (Magnetom Siemens Vison®, Erlangen, Germany). Rats were anesthetized with intraperitoneal injections of 100 mg/kg body weight ketamin (Ketanest®, Pfizer Inc., New York, NY, USA) and 10 mg/kg body weight xylazin (Rompun®, Bayer, Leverkusen, Germany). Superparamagnetic iron oxide particles (Resovist®, Schering, Berlin, Germany) were applied via the femoral vein at a dosage of 0.8 mmol Fe/kg body weight at the beginning of the illumination (
For all MRI scans, animals were lying in supine position with their heads fixed in a custom-made dual channel surface coil designed for investigations of the rat brain (A063HACG, Rapid Biomedical, Würzburg, Germany). The MR protocol included a scout sequence in three planes and a coronal T2-w sequence (TR 2.500 ms, TE 92 ms) with a slice thickness of 2 mm and a coronal CISS sequence (TR: 16.4 ms, TE: 8.2 ms) with a slice thickness of 1 mm. For data postprocessing, MR images were transferred to an external workstation (Leonardo®, Siemens, Erlangen, Germany). In seven animals that had received SPIO particles at the beginning of the illumination, the proportional volumes of the hypointense infarct core and the hyperintense rim surrounding the core were calculated over time by manually delineating the respective areas in subsequent slices as described previously (Kleinschnitz et al, 2003). Likewise, infarct volumes after 4 and 24 h were calculated in six rats without SPIO application. Infarct volumes were normalized to the total volume of the ipsilateral hemisphere.
Histology
After MRI, rats were killed by C02 narcosis. Brains were rapidly removed and immersion-fixed overnight in 4% paraformaldehyde, washed in phosphate-buffered saline (PBS), and embedded in paraffin. Subsequently, 5-μm-thick coronal paraffin sections were cut at multiple levels through the infarcts, deparaffinized with xylene, rehydrated, and washed in deionized water and PBS. Iron detection was performed as described previously (Kleinschnitz et al, 2003). Briefly, tissue sections were immersed in Perl's solution containing 2% potassium ferrocyanide and 2% HCl at a 1:1 concentration for 30 mins, washed and coverslipped. For evaluation of ischemic tissue and cell damage, hematoxylin-eosin (HE) staining was used. To further analyze the iron distribution in different brain compartments after PT, 400 to 600 serial coronal sections (5 μm) covering the whole photothrombotic lesion (mean diameter 2 to 3 mm) from the frontal to the occipital pole were cut from the brains of the animals that had received SPIO particles at the beginning of the illumination. Every 10th section was stained with Perl's solution (40 to 60 slices per animal) and analyzed for intravascular, interstitial and cell-bound iron deposits. Sections were examined with a Zeiss-Axiophot microscope.
Statistical Analysis
For statistical analysis the GraphPad Prism software package, version 3.0. (GraphPad Prism, USA) was used. Data are expressed as mean ± s.d. To analyze the hypo- and hyperintense lesion volumes after simultaneous injection of SPIO particles over time, nonparametric Kruskal—Wallis test followed by Dunn's post test for multiple comparisons was applied. Infarct volumes between 4 and 24 h were compared using nonparametric Mann—Whitney test.
Results
In accordance with previous studies (Schroeter et al, 2001; Kleinschnitz et al, 2003), control animals without SPIO particle injection exhibited hyperintense cortical lesions on T2-w MRI sequences after PT (Figures 1A and 1B). These lesions significantly increased in size between 4 and 24 h, indicating secondary photothrombotic infarct growth after cessation of illumination (Figures 1A—C). In animals receiving SPIO particles at the beginning of illumination, a markedly hypointense lesion was seen on T2-w images when MRI was performed immediately after PT (Figure 2A). After 3 h, a hyperintense rim appeared around the hypointense core that gradually increased until 48 h after PT (white arrows in Figures 2B and 2C). Both the early hypointense core and the hyperintense peripheral rim persisted until day 14 in animals that were scanned serially by MRI, but gradually declined in size, indicating shrinkage of the ischemic lesion (Figure 2D). Quantification of our data revealed that the relative volume of the hypointense infarct core remained stable between days 0 and 5 and slightly decreased afterwards (Figure 3). In contrast, the volume of the hyperintense peripheral rim changed with maximum levels at day 1 (6.4% of the ipsilateral hemisphere) and a subsequent decrease until day 14 (2.3% of the ipsilateral hemisphere;
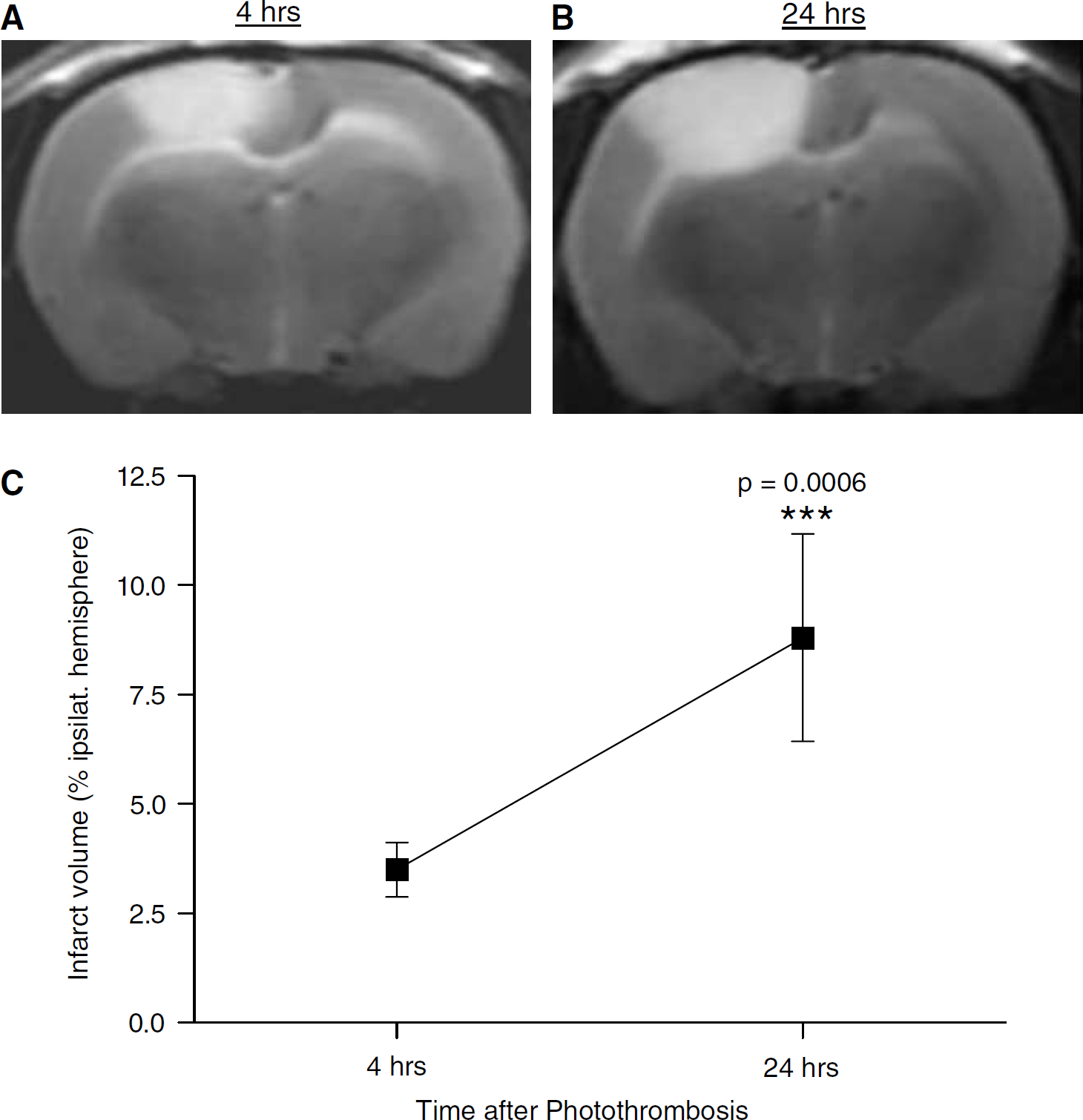
Sequential coronal T2-w MRI in a control animal without SPIO application and quantification of infarct volumes. A hyperintense cortical lesion is present 4 h after PT (
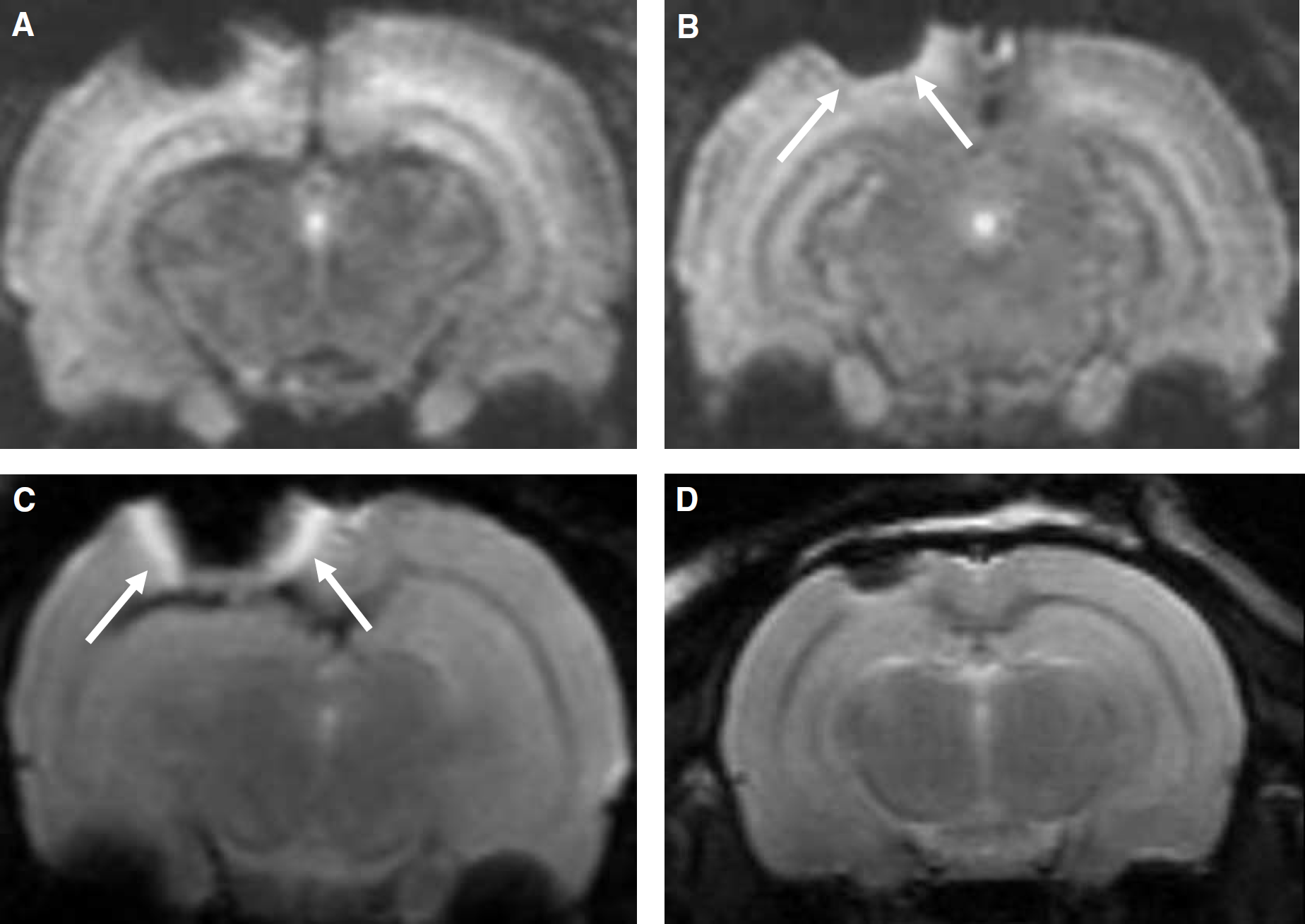
Sequential T2-w MRI in an individual animal that had received SPIO particles at the beginning of the illumination. Immediately after PT, the ischemic infarct is depicted as a markedly hypointense cortical lesion on MRI (
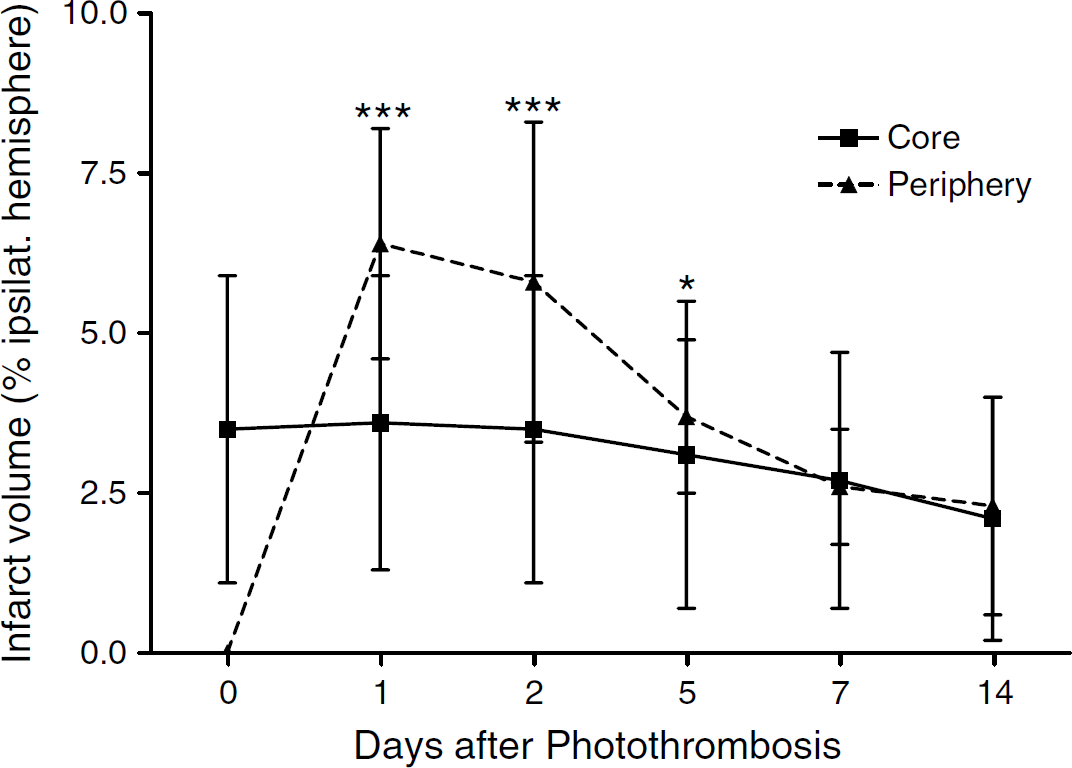
Quantitative analysis of the hypo- and hyperintense lesion portions after simultaneous injection of SPIO particles to PT over time. The relative volume (normalized to the ipsilateral hemisphere) of the hypointense part of the lesion (—) virtually remains stable over time, while the hyperintense rim in the periphery (—) displays a maximum at day 1 (6.4% ± 1.8%) and subsequently decreases until day 14 (2.3% ± 1.7%). Mean ± s.d. (
When SPIO particles were injected by a 2-h delay after completion of PT, the cortical lesions appeared mainly hyperintense on T2-w images, but displayed a small hypointense rim at their outer margin, which was indicative of ongoing vessel thrombosis (white arrows in Figure 4A). In contrast, more delayed injection of SPIO particles 24 h after PT showed merely hyperintense lesions on T2-w MRI without signal loss, which indicated cessation of SPIO accumulation at this advanced stage of infarct development (Figure 4B).
On corresponding histological sections stained by Perl's solution, cortical areas with a hypointense MRI signal representing the animals that had received SPIO at the beginning of the illumination showed a massive and homogenous intravascular accumulation of iron particles which were trapped within the vessel thrombi (Figure 5A). Most notably, multiple serial sections covering the whole photothrombotic lesion revealed an exclusively intravascular staining pattern, while diffuse, interstitial iron deposits, for example, due to the disrupted blood—brain barrier, were not present. When SPIO particles were injected with a delay of 2 h after PT, SPIO particles were localized exclusively in vessels at the outer margins of the infarcts (white arrows in Figure 5B), while vessel thrombi in the center of the lesions were devoid of iron particles (black arrows in Figure 5B). These data indicate ongoing vessel occlusion exclusively in the penumbra of photothrombotic lesions. When SPIO particles were applied 24 h after PT, the hyperintense cortical lesions on MRI consisted of necrotic tissue on HE-stained sections, and no SPIO particles could be detected by Perl's stain (Figure 5C). This finding indicates termination of thrombus formation already before SPIO application.
To clarify what happens with the SPIO deposits within thrombi in the long term, animals that had received SPIO particles during induction of PT were allowed to survive for prolonged periods. In these animals, in addition to intravascular SPIO accumulation, iron particles were found in increasing numbers of phagocytic cells from day 3 onward within the infarcted areas (Figures 5D and 5E). These phagocytes most likely cleared debris from destroyed and occluded vessels, and thereby accumulated iron particles intracellularly.
Discussion
As a principal finding, we provide a novel technique that allows detection of developing vessel occlusion
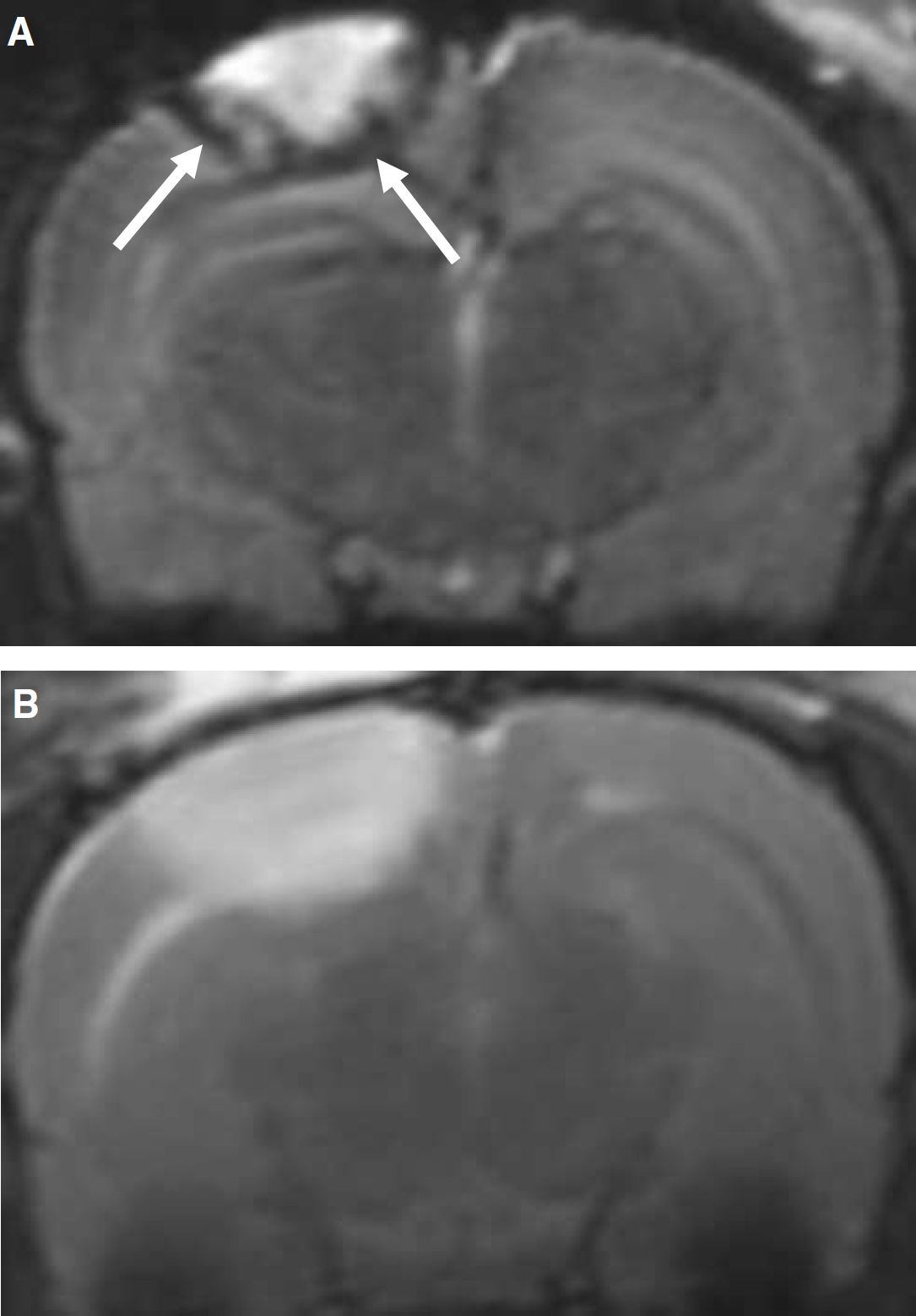
T2-weighted MRI after delayed SPIO particle application. When SPIO are injected 2 h after PT (
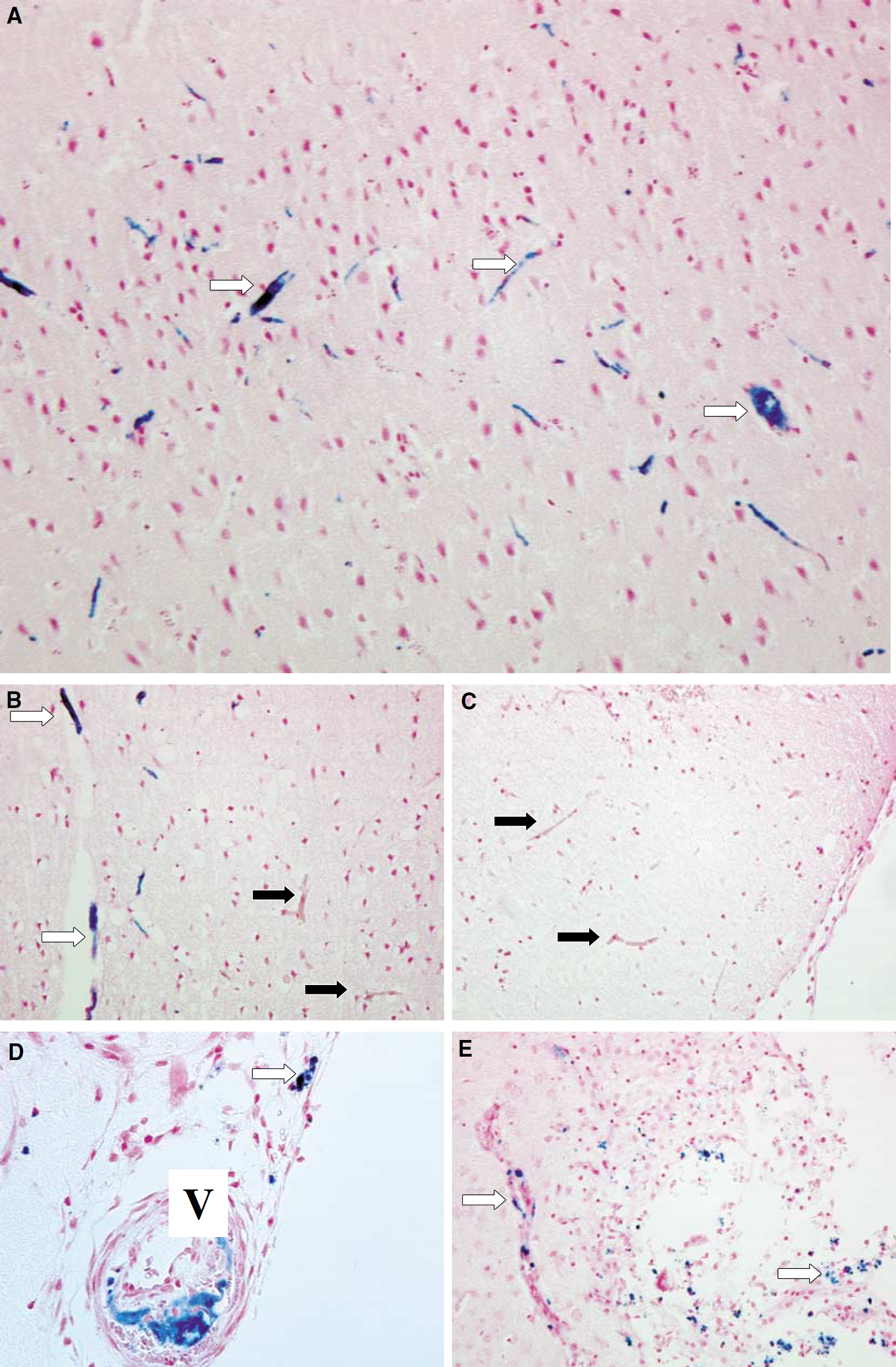
Histological analysis of the photothrombotic infarcts after SPIO particle application. Localization of SPIO particles by Perl's stain in focal cerebral infarcts injected during illumination (
By comparing infarct areas showing hyperintensities (structural damage) and hypointensities (iron accumulation) on T2-w images, we could further show that in PT vessel occlusion is still ongoing despite cessation of illumination, and spreads from the core of the lesion to the ‘penumbra’. Thus, ongoing thrombosis as shown here provides a partial explanation and possible mechanism for secondary infarct growth in the PT model documented by several groups (Grome et al, 1988; Verlooy et al, 1993; Lee et al, 1996; this study). The concept of an early thrombotic lesion core surrounded by a zone of ongoing vessel thrombosis in the periphery as revealed by iron-enhanced MRI in the present study further substantiates previous findings on the temporal profile of hemodynamic alterations after PT. Dietrich et al (1986) described a severely hypoperfused area 30 mins after cortical irradiation, which further enlarged after 4 h, indicating dynamic secondary infarct growth via expanding cerebral hypoperfusion.
Paramagnetic iron oxide particles have recently been introduced as a new class of cell-specific MRI contrast agent. They are taken up by circulating macrophages when applied directly to the circulation (Kleinschnitz et al, 2003). Moreover, T cells (Arbab et al, 2003) and stem cells (Hoehn et al, 2002) can be loaded
Ischemic stroke is caused by the cessation of cerebral blood flow. Brain regions which hemodynamically depend on the occluded vessels are irreversibly damaged within a few minutes. The surrounding peripheral tissue, where blood flow is reduced but reaches a certain threshold to maintain structural integrity (‘penumbra’), is at high risk to be lost by secondary infarct growth (Dirnagl et al, 1999; Garcia et al, 1993; Furlan et al, 1996; Marchal et al, 1996). Today, DW-MRI is the gold standard for the diagnosis of acute brain ischemia (Warach et al, 1995). Within minutes after the onset of experimental ischemia restricted diffusion can be shown (Moseley et al, 1990). Diffusion-weighted magnetic-resonance imaging only provides information regarding cytotoxic brain edema, but not regarding the extent of the underlying intravascular thrombus formation (Warach et al, 1995). Recently, a fibrinbinding gadolinium-labeled peptide was used as MR-contrast agent to visualize intravascular thrombus formation
In the PT model, vessel occlusion and ischemic brain injury occur acutely in a highly synchronized and reproducible fashion. Further studies are required to clarify whether our novel technique can also be applied to other stroke models with different lesion dynamics, such as mechanical middle cerebral occlusion or the complex pathophysiology of thrombosis or fibrinolysis in humans.
As another important issue, cerebral ischemia might be complicated by intracerebral hemorrhage, resulting in a similar signal hypointensity on MRI as local trapping of iron particles. This was no confounding factor in our present study, since we found no bleedings on our thorough histological examination. Moreover, at this early stage hemorrhage does not appear hypointense on all MR sequences (Gustafsson et al, 1999) and thus cannot account for the observed signal changes in our study. In future studies employing our technique, hemorrhage probably can also be distinguished from iron-particle-induced signal loss by combined analysis of different MR sequences.
Photothrombotic brain injury is accompanied by strong parenchymal edema (Dietrich et al, 1987a). Therefore, intravascular accumulation of iron particles may not exclusively be caused by thrombosis but vascular compression as well. Although vasogenic edema develops within minutes after PT, maximum water content is reached with a delay of 24 h (Dietrich et al, 1987a; Pratt et al, 1998). At this time, however, intravascular iron trapping was absent in our study. This argues against a substantial contribution of transmural vascular compression.
Taken together, we have shown that SPIO-enhanced MRI is a valid
Footnotes
Acknowledgements
The authors thank Gabi Köllner for expert technical assistance. MB holds an endowed professorship donated by the Schering Deutschland GmbH, Berlin, to the University of Würzburg, but has no financial interests in this study.
