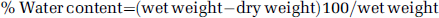Abstract
Experimental evidence indicates that long-term exposure to moderately high ambient temperature (heat acclimation, HA) mediates cross-tolerance to various types of subsequently applied stress. The transcriptional activator hypoxia-inducible factor 1 (HIF-1) has been implicated in playing a critical role in HA. It also regulates the expression of Erythropoietin (Epo), whose neuroprotective effects have been shown in a variety of brain injuries. The aim of the present study was to examine whether HA exerts a beneficial effect on the outcome of closed head injury (CHI) in mice and to explore the possible involvement of HIF-1 and Epo in this process. Heat acclimated mice and matched normothermic controls were subjected to CHI or sham surgery. Postinjury motor and cognitive parameters of acclimated mice were compared with those of controls. Mice were killed at various time points after injury or sham surgery and brain levels of HIF-1α, the inducible subunit of HIF-1, Epo, and the specific erythropoietin receptor (EpoR) were analyzed by Western immunoblotting. Motor and cognitive functions of acclimated mice were significantly better than those of controls. Heat acclimation was found to induce a significant increase in expression of nuclear HIF-1α and EpoR. The EpoR/Epo ratio was also significantly higher in acclimated mice as compared with controls. Nuclear HIF-1α and EpoR were higher in the acclimated group at 4 h after injury as well. The improved outcome of acclimated mice taken together with the basal and postinjury upregulation of the examined proteins suggests the involvement of this pathway in HA-induced neuroprotection.
Keywords
Introduction
Traumatic brain injury is one of the leading causes of mortality and morbidity in Western countries (Waxweiler et al, 1995), inducing tissue edema formation, cell death and loss of function. Many of these long-term effects result from the complex cascade of deleterious events initiated at the moment of injury. Using an experimental model of closed head injury (CHI) developed in our laboratory (Shapira et al, 1988; Chen et al, 1996), we have shown that in rats previously exposed to prolonged moderate ambient heat (heat acclimation, HA), secondary damage was reduced and functional recovery was faster (Shohami et al, 1994). Heat acclimation has also been shown to induce tolerance on ischemia—reperfusion insult to the heart (Levi et al, 1993) and on hyperoxia in the brain (Arieli et al, 2003), suggesting that HA produces a wide scale protective effect (Horowitz, 2002), yet the mechanisms underlying the neuroprotective effects remained poorly defined. Heat acclimation leads to the development of a new physiologic homeostasis, characterized by a reduced basal metabolic rate and improved heat tolerance (Horowitz, 2001). The process also leads to an increase in basal levels of cytoprotective proteins such as heat shock proteins (HSPs) (Horowitz, 2002). Establishment of this new physiologic state involves both molecular and integrative mechanisms including changes in gene expression (Horowitz et al, 2004).
Hypoxia-inducible factor 1 (HIF-1), a transcriptional activator which mediates homeostatic responses to reduced oxygen availability, has been shown to be essential for the development of HA (Treinin et al, 2003) and is also required for hypoxia induced cardio-protection against ischemia—reperfusion injury in mice (Cai et al, 2003). Hypoxia-inducible factor 1 regulates the expression of over 70 known target genes, leading to an overall increase in tissue oxygen delivery and ATP production (Semenza, 2004). This adaptive mechanism is preserved among eukaryotic species, indicating its importance in enabling adjustment to changes in environmental pO2 conditions. Hypoxia-inducible factor 1 is a heterodimer consisting of an inducible α subunit and a constitutively expressed β subunit. Only a dimer containing both subunits can undergo translocation to the cell nucleus. One of the genes regulated by HIF-1 is the erythropoietin (Epo) gene. Erythropoietin was first recognized as a hematopoietic cytokine. It enhances erythrocyte production by inhibiting apoptosis of erythroid progenitor cells and promoting their survival and proliferation. Owing to this physiologic function, it was thought that both the cytokine and its specific receptor, the erythropoietin receptor (EpoR), are exclusively expressed in the hematopoietic system and in the fetal liver and adult kidney where Epo is produced. However, several lines of evidence now show that both Epo and EpoR are expressed by other tissues, including the nervous system (Buemi et al, 2002). Erythropoietin receptor is expressed by neuronal and glial cells in the brain as well as by brain capillary endothelial cells, and its expression is upregulated by various types of stress including ischemia and metabolic stress (Bernaudin et al, 1999; Bernaudin et al, 2000). In addition, neuroprotective effects of Epo have been shown in several in vitro and in vivo brain injury models: it increases cell viability of differentiated PC12 cells in the absence of serum and neuronal growth factor (Koshimura et al, 1999) and protects neuronal primary cultures against glutamate neurotoxicity (Morishita et al, 1997). Erythropoietin is also protective in experimental in vivo models of brain injury including transient and permanent ischemia (Bernaudin et al, 1999; Catania et al, 2002), subarachnoid hemorrhage (Grasso et al, 2002), and experimental autoimmune encephalomyelitis (EAE) (Agnello et al, 2002). Other studies indicate that Epo also mediates neuroprotection in retinal ischemia (Junk et al, 2002) as well as in spinal cord injury and peripheral nerve injury models (Gorio et al, 2002; Iwasaki et al, 2002). A clinical trial by Ehrenreich et al (2002) indicated that rhEpo is also beneficial in stroke patients. We have previously established the neuroprotective effect of Epo in our model of CHI. Our results show a beneficial effect of treatment with rhEpo on functional recovery after injury (Yatsiv et al, 2002).
The neuroprotective effect of Epo, a HIF-1 target gene, taken together with the critical role of HIF-1 in HA, led us to further investigate the involvement of this pathway in the induced neuroprotective effect in HA mice. Our CHI model, which was initially developed and characterized using rats, has been modified to enable the use of mice, which resemble rats in their ability to heat acclimate (Bromberg and Horowitz, 2004). Thus, the purpose of the current study was two-fold: (1) to establish whether HA is neuroprotective in mice and (2) to examine the effect of HA and CHI on the expression of HIF-1α, Epo and EpoR.
Materials and methods
Animals and Maintenance
The study was approved by the Institutional Animal Care Committee of the Hebrew University. Male Sabra mice weighing 35 to 50 g were used in this study. Animals were kept under controlled light conditions with a 12 h/12 h light/dark cycle. Food and water were provided ad libitum. The animals were divided into two groups: control (CON), maintained at an ambient temperature of 24°C±1°C and acclimated (HA), held in a climatic chamber at 34°C±1°C for 30 days, ensuring the achievement of a stable acclimated state.
Trauma Model
Experimental CHI was induced under ether anesthesia using a modified weight drop device developed in our laboratory (Chen et al, 1996). Briefly, after anesthesia, a midline longitudinal incision was performed and the skull exposed. A Teflon tipped cone (2 mm diameter) was placed 1 to 2 mm lateral to the midline in the mid-coronal plane. The head was manually held in place and a 75 g weight was dropped on the cone from a height of 18 cm, resulting in a focal injury to the left hemisphere. After recovery from anesthesia, the mice were returned to their home cages with postoperative care and free access to food and water. Sham controls received anesthesia and skin incision only.
Neurobehavioral Evaluation
The functional status of the mice was evaluated according to the Neurological Severity Score (NSS), by an observer unaware of the given treatments. This score is a 10-point scale that assesses the functional neurologic status based on the presence of some reflexes and the ability to perform motor and behavioral tasks such as beam walking, beam balance and spontaneous locomotion (Beni-Adani et al, 2001). Animals are awarded one point for failure to perform a task, such that scores range from zero to ten, increasing with the severity of dysfunction. The NSS obtained 1 h after CHI reflects the initial severity of injury. Therefore, the extent of recovery (ΔNSS) can be calculated as the difference between the NSS at 1 h and at any subsequent time point. A positive ΔNSS indicates recovery, zero reflects no change and a negative ΔNSS indicates neurologic deterioration. NSS values were measured at 1 h after injury and in 24 h intervals during the first 3 days thereafter. Additional measurements were performed at 7 and 14 days after injury. ΔNSS was calculated for each time point.
Evaluation of Edema Formation
Frontal segments weighing ~20 mg were taken from both hemispheres of mice killed 24 h after injury and their wet weight was determined. Segments were then dried in an oven for 24 h at 95°C and dry weight was measured. The percent water content was calculated according to the formula:
Evaluation of Cognitive Performance in the Object Recognition Test
The object recognition test (ORT) was performed on groups of CON and HA mice 2 and 3 days after CHI as previously described (Ennaceur and Delacour, 1988), and shown in our model (Biegon et al, 2004). In the first part of the test (2 days after injury), mice (n = 6/group) were placed in the testing cage (a glass aquarium-like transparent box of 60 × 60 cm) for 1 h habituation. On the following day, they were put back into the same cage with two identical objects. The cumulative time spent by the mouse at each of the objects was recorded manually during a 5-min interval by an observer masked to the treatment received. After 4 h, the mice were reintroduced into the cage, where one of the two objects was replaced by a new one. The time (of 5 mins total) spent at each of the objects was recorded. The basic measure is the percent of the total time spent by mice in exploring an object during the testing period, whereby normal healthy rodents will spend relatively more time exploring a new object than a familiar, that is, ‘memorized’ object.
Western Immunoblotting
Immediately after the 30-day acclimation or normothermic period, either CHI or sham surgery were performed on mice from both CON and HA groups. Mice from both groups were killed at either 4 or 24 h after CHI or sham surgery. After decapitation, brains were quickly removed and frontal segments from both hemispheres were separated and frozen at −70°C until analysis. Sample preparation was performed as previously described (Bernaudin et al, 2002) with minor modifications. Both nuclear and cytosolic extracts were prepared from each sample. Tissue segments were incubated in a buffer containing 10 mmol/L HEPES (pH 7.9), 1.5 mmol/L MgCl2, 1 mmol/L EDTA, 10% glycerol, 1 mmol/L dithiothreitol (DTT), 1 mmol/L phenylmethylsulfonyl fluoride (PMSF), 1 mmol/L Na3VO4, and 10% complete EDTA-free protease inhibitor (Roche Diagnostics, Mannheim, Germany) for 30 mins at 4°C and then homogenized. Homogenates were centrifuged at 1,000g for 15 mins and supernatants (cytosolic extracts) were kept at −70°C whereas pellets were further processed to prepare the nuclear extract fraction. Pellets were resuspended in the homogenization buffer and suspensions were centrifuged at 12,000g for 10 mins. These pellets were resuspended and incubated for 30 mins at 4°C in a buffer containing 20 mmol/L HEPES (pH 7.9), 25% glycerol, 0.42 M NaCl, 0.2 EDTA, 1 mmol/L DTT, 1 mmol/L PMSF, 1 mmol/L Na3VO4, and 10% complete EDTA-free protease inhibitor for nuclear protein extraction. Protein concentrations of both cytosolic and nuclear extracts were determined using the Bradford method (BioRad Laboratories, Munich, Germany). Equal protein samples (50 μg) were separated on 7.5% SDS-polyacrylamide gels with 4.5% SDS stacking gels and electrotransferred onto nitrocellulose membranes (0.2 μm, Schleicher and Schuell, Dassel, Germany). Nuclear extract blots were probed with a rabbit polyclonal anti-HIF-1α antibody (1:200; Santa Cruz Biotechnology, Santa Cruz, CA, USA) and cytosolic blots were probed with rabbit polyclonal antibodies for either Epo (1:200) or EpoR (1:200; both from Santa Cruz Biotechnology, Santa Cruz, CA, USA). Appropriate peroxidase-coupled immunoglobulin G (1:5,000; Jackson Immunoresearch, Soham, Cambridgeshire, UK) was used in secondary incubations. Reactive bands were visualized using the enhanced chemiluminescence system (Biological Industries, Beit Haemek, Israel). All membranes were also incubated with a rabbit polyclonal antibody for β-actin (1:200; Santa Cruz Biotechnology, Santa Cruz, CA, USA). Subsequent washes and visualization procedures were as described for HIF-1α, Epo, and EpoR antibodies. Blots were quantified using the Tina software (Raytest, Straubenhardt, Germany). Protein level was expressed as relative optical density, representing the optical density of HIF-1α, Epo, or EpoR divided by the optical density of β-actin within the same lane.
Statistical Analysis
Data are expressed as mean ± s.e.m. A commercial statistics package (Graph Pad Prism version 3.03) was used for determining statistical significance. Significance was determined using the Mann—Whitney test for ΔNSS values and one-way analysis of variance (ANOVA) followed by Student's t-tests for water content data, optical density values, and ORT results. P-values <0.05 were considered significant for all comparisons.
Results
Functional Recovery Edema Formation and Memory
To examine the effect of HA on functional recovery and edema formation after CHI, the parameters of injured HA mice were compared with those of injured CON ones. The initial severity of injury (NSS at 1 h) was similar in both groups (data not shown) and the results of the functional recovery are shown in Figure 1A. Markedly greater recovery was observed in the HA group 24 h after injury as compared with CON (0.92 ± 0.14 versus 0.44 ± 0.16, P<0.05). ΔNSS values increased with time in both HA and CON mice as a result of spontaneous recovery occurring in both groups. ΔNSS values continued to tend to be higher in HA mice at all subsequent time points as well, yet the statistical significance of the effect of HA observed 24 h after injury diminishes with time.
Beneficial effect of heat acclimation (HA) on neurobehavioral recovery tissue, edema formation and cognitive function after closed head injury (CHI).
As shown in Figure 1B, a pronounced increase in tissue water content was observed in the left (ipsilateral) hemisphere of all injured mice (from 78.42% ± 0.24% and 77.78% ± 0.24% in noninjured mice to 81.81% ± 0.45% and 83.09% ± 0.37% in HA and CON mice, respectively, P<0.001). In both HA and CON injured mice, a moderate (nonsignificant) increase occurred in the right (contralateral) hemisphere as well. Although the effect of injury on ipsilateral hemisphere is clearly showed in both groups, water accumulation was significantly smaller in HA mice (83.09% ± 0.37 in CON versus 81.81% ± 0.45 in HA, P<0.05) indicating that HA markedly reduces edema formation.
Cognitive function was assessed by ORT and the results are depicted in Figure 1C. When tested 3 days after injury, both groups spent equal time at the two objects (~50%) at the baseline measurements. However, at the test, 4 h later, HA mice spent most of their exploration time at the new object (73.26% ± 3.37% P<0.05 versus baseline measurement) while the CON ones failed to prefer the ‘novel’ object over the familiar one (exploration time of the new object 58.33%±10.66%, not different from baseline).
Effect of HA on HIF-1α, Epo, and EpoR Expression
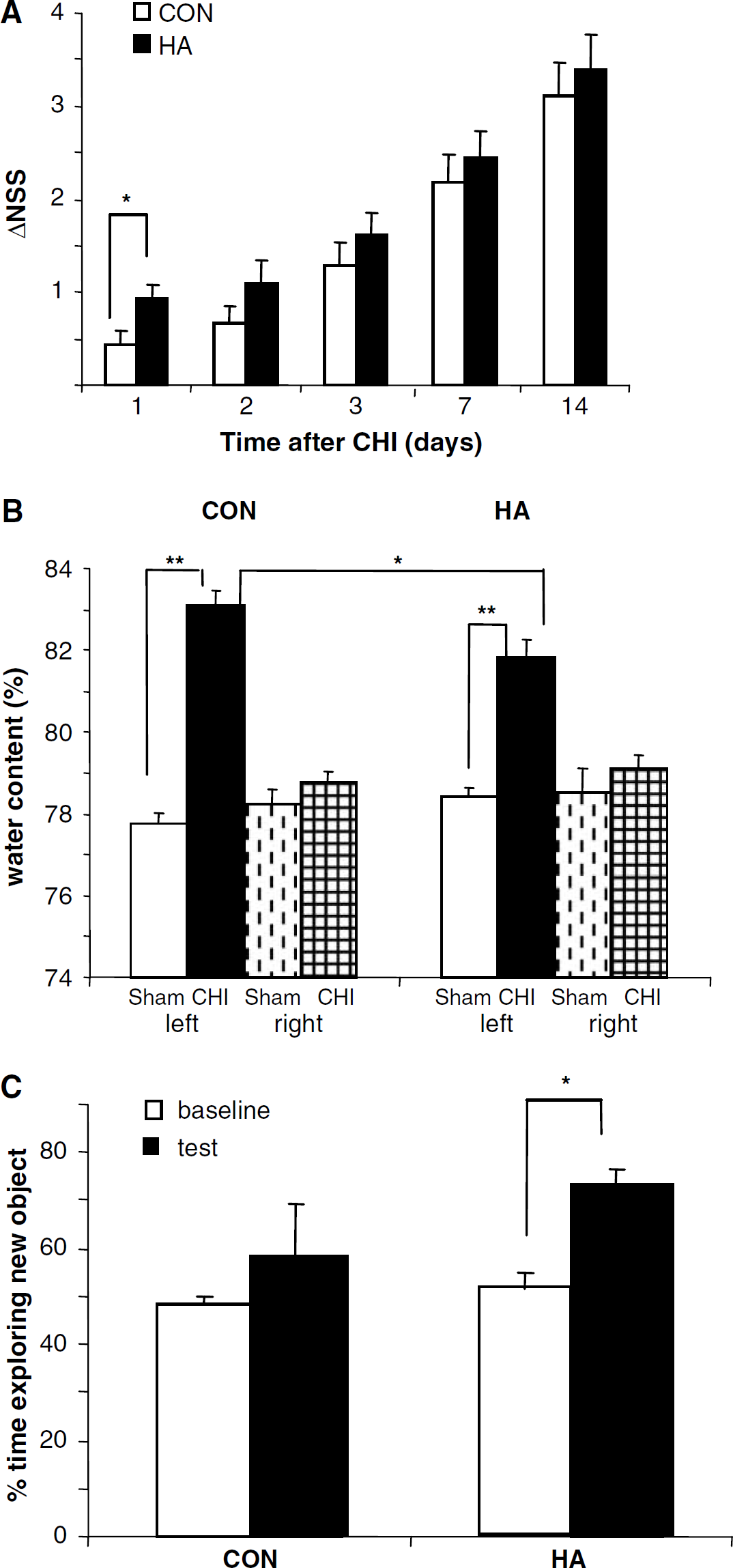
Our initial analysis of nuclear and cytosolic levels of HIF-1α and nuclear/cytosolic HIF-1α ratios in CON and HA sham mice indicated that cytosolic HIF-1α levels were similar in both groups (Figure 2, inset), while differences were clearly observed in the nuclear fraction (Figure 2) and between the nuclear/cytosolic ratios (data not shown). These results taken together with the large body of evidence indicating that the effect of HIF-1 on its target genes is mediated through its binding to hypoxia responsive elements on their DNA, a mechanism which requires the presence of this transcription factor within the cell nucleus, led us to proceed using nuclear extracts for the analysis of HIF-1α levels during the rest of the study. Therefore, throughout the study, the analysis of HIF-1α levels was performed only in the nuclear extracts. To establish the effect of HA on protein expression, protein levels of HA and CON noninjured sham mice were compared (Figure 2). Heat acclimated mice displayed a three-fold increase in HIF-1α within the cell nucleus as compared with CON (85 ± 18.53 and 27 ± 3.61, respectively, P < 0.05). Erythropoietin receptor was also markedly upregulated in these mice (24.96 ± 2.52 versus 12.67 ± 1.72, respectively, P 0.01). Erythropoietin expression was similar in both groups, yet because the neuroprotection afforded by Epo, depending on the activity of the Epo pathway, is reflected by the EpoR/Epo ratio, we calculated this ratio and found a significant increase because of HA (93.91 ± 3.94 and 46.03 ± 11, respectively, P < 0.01).
Heat acclimation (HA) increases the expression of nuclear HIF-1α, EPOR, and EpoR/Epo ratio. Nuclear or cytosolic extracts from tissue segments taken from frontal cortex of sham mice were separated on SDS-PAGE gels. Nuclear blots were probed with antibodies against HIF-1α and β-actin. Cytosolic blots were probed with antibodies against Epo, EpoR, and β-actin. Relative optical density was defined as the calculated optical density of HIF-1α, Epo, or EpoR divided by the calculated optical density of β-actin within the same lane. Heat acclimation led to an upregulation of HIF-1α within the cell nucleus and of EpoR leading to increased EpoR/Epo ratio. *P < 0.05, **P < 0.01, as determined by one-way ANOVA followed by Student's t-tests. n = 3 to 4 per group. Inset: cytosolic HIF-1α levels are not affected by HA or closed head injury (CHI). 1—CON sham, 2—CON 4 h after CHI, 3—HA sham, 4—HA 4 h after CHI. Cytosolic extracts from tissue segments taken from the frontal cortex of sham mice or from the site of injury were separated on SDS-PAGE gels. Blots were then probed with antibodies against HIF-1α and β-actin. Relative optical density was defined as the calculated optical density of HIF-1α divided by the calculated optical density of β-actin within the same lane. No difference was observed in the cytosolic content of HIF-1α after HA or CHI. n = 4 per group.
HIF-1α, Epo, and EpoR Expression After CHI
Next, the effect of CHI on nuclear HIF-1α, Epo, and EpoR expression was examined by comparing post-CHI protein levels in both HA and CON mice. Because there were marked differences in HIF-1α, EpoR, and EpoR/Epo ratio between HA and CON sham mice, it was necessary to normalize the optical density values of postinjury samples relative to sham-operated mice of the same group (HA to HA sham and CON to CON sham) at each time point to distinguish the effect of CHI from that of HA. Data are therefore represented as values respective to sham control.
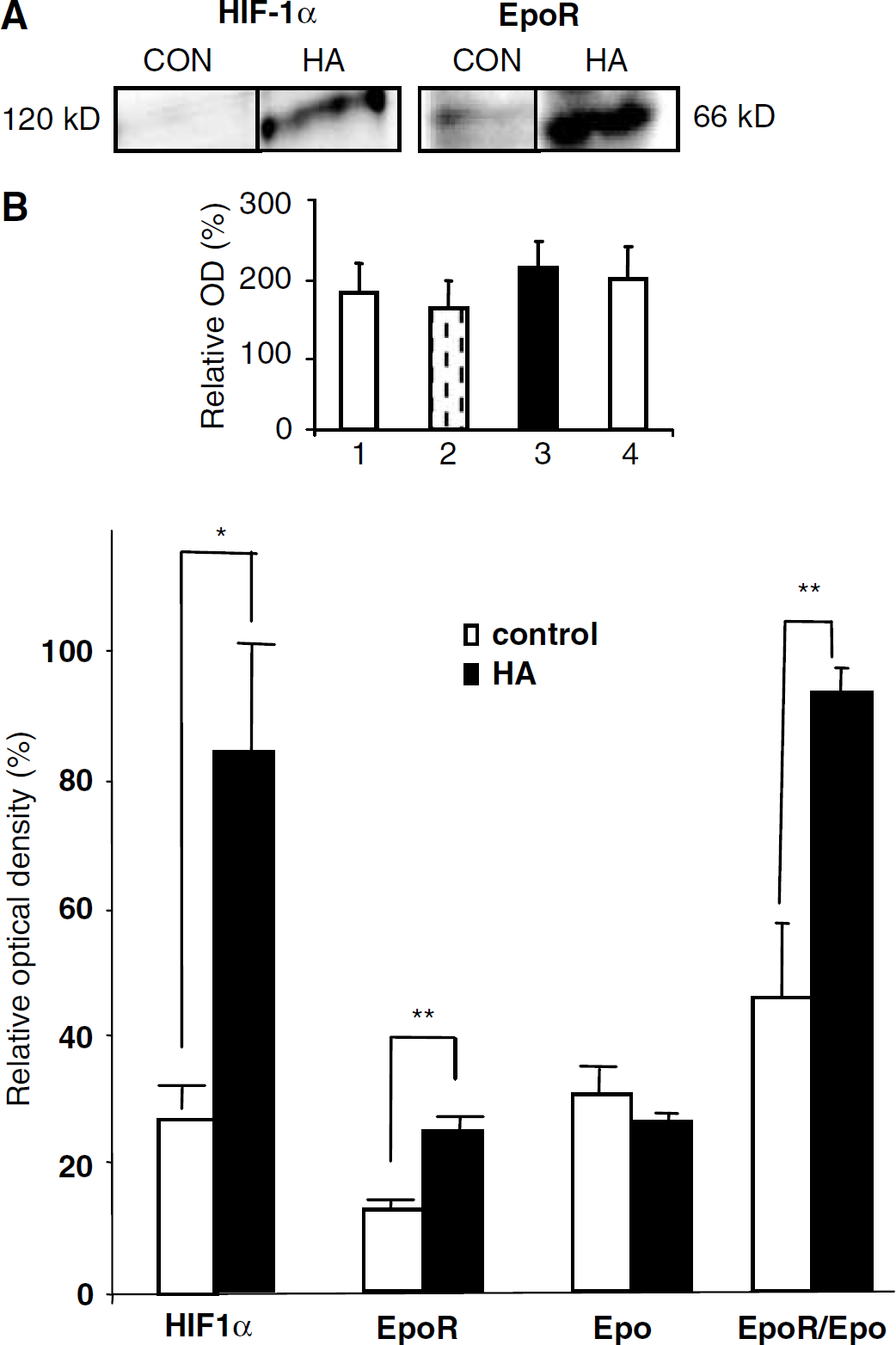
As shown in Figure 3, 1 h after injury, HIF-1α expression within the cell nucleus is slightly decreased in both CON and HA groups as compared with sham controls (0.89 ± 0.11 and 0.69 ± 0.10 for CON and HA, respectively). While this level continues to decline in CON mice, a marked two-fold increase in protein expression (versus 1 h) occurs in the HA group (0.69 ± 0.10 and 1.37 ± 0.21, P < 0.05), resulting in a significantly higher level of nuclear HIF-1α in HA mice 4 h after injury as compared with CON (1.37 ± 0.21 and 0.69 ± 0.06, respectively, P < 0.05). Cytosolic levels of HIF-1α remain unchanged after CHI (Figure 2, inset).
HIF-1α expression is increased in heat acclimated (HA) mice 4 h after injury but not in CON. Nuclear extracts prepared from tissue segments taken from the site of injury were separated on SDS-PAGE gels. Blots were incubated with anti-HIF-1α and anti-β-actin antibodies. Relative optical density was calculated and values were corrected relative to sham-operated mice at the same time point. At 1 h after injury, a slight decrease in expression was observed in both groups as compared with sham. Heat acclimation levels then increased while CON expression continued to decline. *P < 0.05 as determined by one-way ANOVA followed by Student's t-tests. n = 4 to 7 per group.
The levels of Epo and EpoR after injury are depicted in Figure 4. At 4 h after injury, EpoR levels are higher in HA mice as compared with CON (1.89 ± 0.18 and 1.15 ± 0.09, respectively, P < 0.05). Although Epo levels remain similar between the two groups with a slight decrease observed as compared with sham-operated mice of the same group (0.89 ± 0.13 and 0.79 ± 0.13 at 4 and 24 h after injury in CON versus 0.67 ± 0.24 and 0.79 ± 0.13 at the same time points in HA), the EpoR/Epo ratio 4 h after injury was much higher in HA mice than in CON ones (3.59 ± 0.08 versus 1.33 ± 0.07, P < 0.05). The increase in EpoR and therefore of the EpoR/Epo ratio occurring in the HA group is transient, such that at 24 h after CHI they reach levels similar to those of nonacclimated mice (1.14 ± 0.15 and 1.18 ± 0.25, 1.54 ± 0.32 and 1.45 ± 0.17 for EpoR and EpoR/Epo ratio in CON and HA, respectively). As opposed to the robust transient increase seen in HA mice, EpoR is only slightly increased after injury in CON as compared with sham mice and remains relatively unchanged at both 4 and 24 h after injury (1.15 ± 0.09 and 1.14 ± 0.15, respectively).
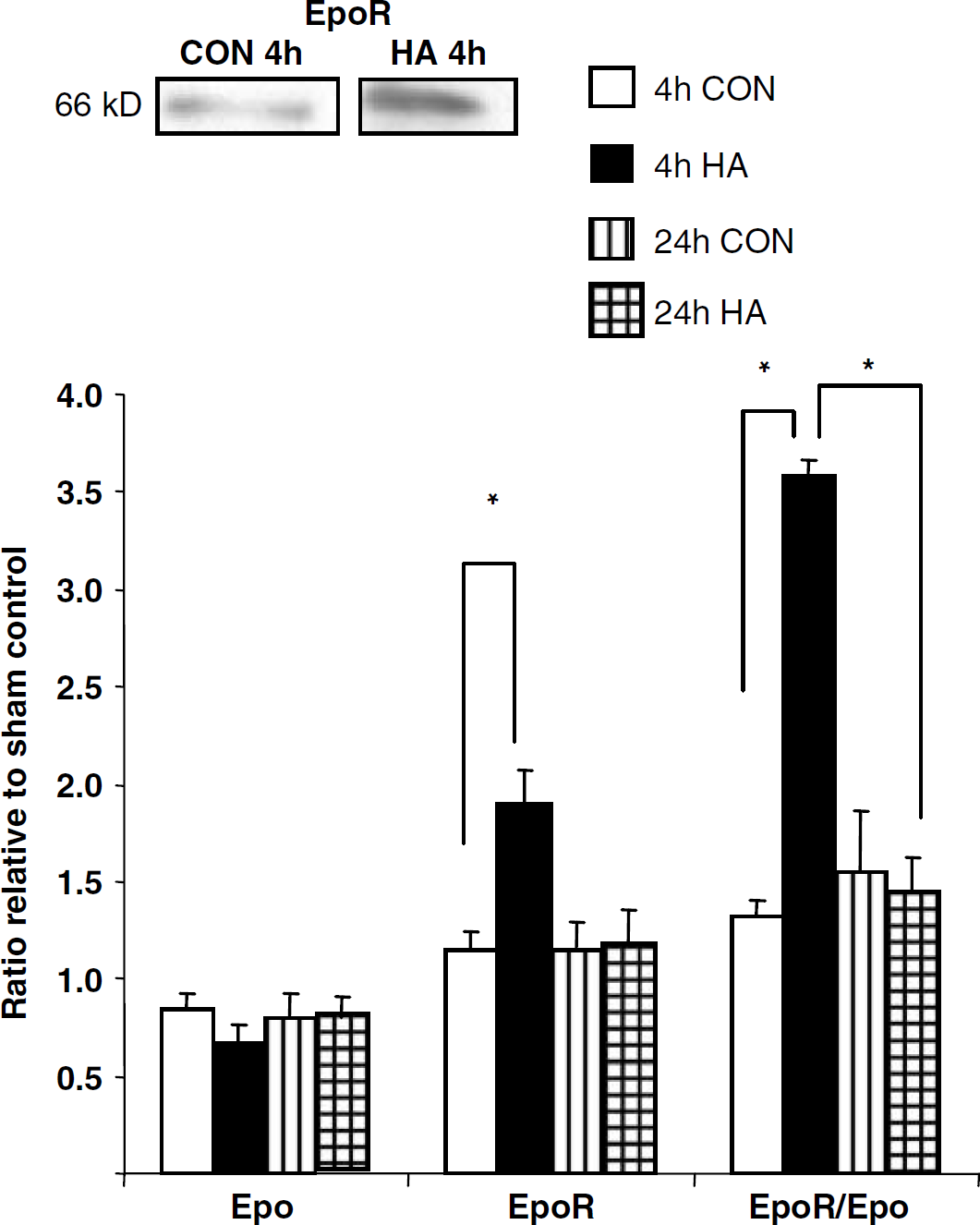
EpoR and EpoR/Epo ratio are increased in heat acclimated (HA) mice after closed head injury (CHI). Cytosolic extracts from ipsilateral tissue segments taken from the site of injury were separated on SDS-PAGE gels. Blots were incubated with antibodies against Epo, EpoR and β-actin. Optical density values were corrected relative to the sham-operated mice at each time point and represented as values relative to control. A marked transient increase was observed 4 h after CHI in HA mice but not in CON. *P < 0.05 as determined by one-way ANOVA followed by Student's t-tests. n = 3 to 4 per group.
Discussion
In the first part of this study, we confirmed that HA improves functional recovery after CHI in mice, extending our earlier findings in the rat. We also showed that HA induces increased expression of nuclear HIF-1α and EpoR. The documented evidence on Epo as a neuroprotective agent and the causal evidence between better clinical recovery and the upregulation of these factors in HA mice suggest their possible involvement in neuroprotection.
The weight drop device model of CHI was originally developed for utilization on rats and was subsequently modified for induction of injury in mice. Here we used the modified model to induce injury on HA and CON mice and compare their postinjury recovery, edema formation, and protein levels. We established the neuroprotective effect of HA against CHI in mice by showing that recovery of motor ability in HA mice is improved as compared with that of CON 24 h after injury. In addition, we have also showed that their cognitive function is better preserved, as showed by their performance in the ORT and that ipsilateral edema formation is decreased in HA mice. The ΔNSS values, which reflect the extent of recovery, of HA mice show a tendency to be higher than those of CON at all subsequent time points, although the significant difference seen clearly 24 h after injury is lost with time because of spontaneous recovery occurring in both groups. These findings are in agreement with results from previous experiments performed using the rat model, which also showed a beneficial effect of HA on the outcome of injury (Shohami et al, 1994). We have also previously showed the existence of augmented concentrations of low molecular weight antioxidants in the brain of HA rats after CHI (Beit-Yannai et al, 1997), a finding which suggests that better ability of dealing with postinjury oxidative stress may contribute to the improved recovery observed in HA animals. The similarity between the results obtained in mice and rats indicates that the effects of HA on CHI outcome are conserved regardless of the species used and that the modification of the injury model did not affect the interaction between the two processes. However, differences in the duration of HA-induced neuroprotection do exist between the two rodent species, probably because rats are larger than mice and therefore have a lower metabolic rate per gram of body mass (Schmidt-Nielsen, 1964). The fact that HA affects clinical outcome after CHI indicates that at least part of the molecular mechanisms involved in the development of the integrative adaptive response of HA are interactive with the pathophysiologic cascade of CHI. Characterization of these protective pathways will possibly enable their use in the development of exogenic neuroprotective agents. This is particularly important in light of the lack of a specific therapy for the treatment of CHI patients in the clinical setting (Narayan et al, 2002). Recent studies from our laboratory show that treatment of mice after CHI with rhEpo is neuroprotective, and, among other effects, improves motor and cognitive functions (Yatsiv et al., 2005, submitted for publication). Preliminary experiments that we have conducted using the ORT, indicate that the extent of protection against CHI, which is induced by HA, is similar to that offered by treatment with rhEPO. Interestingly, treatment of HA mice with this agent did not lead to an additive effect (data not shown).
It has been previously suggested that exposure to sublethal stress can induce protection against subsequent more severe exposure to a second stressor of a different kind. This phenomenon is known as cross-tolerance and has been shown in a variety of experimental models (see Dirnagl et al (2003) for a variety of stressors, Glantz et al (2005) for cerebral ischemia, Horowitz et al (2004) for heat stress and cardiac ischemia, and Arieli et al (2003) for hyperoxia). The development of cross-tolerance indicates the involvement of common signaling pathways in the physiologic reaction, which is alerted by different stressors. cross-tolerance toward CHI resulting from HA is similar to other observations indicating that this physiologic adaptation conveys protection against subsequently applied stress of various types in addition to improving heat tolerance (Levi et al, 1993; Horowitz et al, 2004).
The central role of HIF-1 in HA has been previously established. It has been shown that HA could not be induced in Hif-1 knockout Caenorhabditis elegans nematodes (Treinin et al, 2003). Other studies have shown that a marked increase in HIF-1α expression occurs in the heart after HA and have raised the question of the involvement of this transcription factor in HA-induced ischemic tolerance (Maloyan et al, 2001). Because the protective effect observed in our study is induced by HA, it is conceivable that HIF-1 plays a role in this case as well. This notion is further strengthened by the fact that the expression of Epo, known to mediate neuroprotection in numerous models of brain injury including our own model (Yatsiv et al, 2002), is regulated by HIF-1. Our findings show that brain HIF-1α levels present in the cell nucleus are markedly increased by HA, similar to the increase previously reported in the heart. It is also noteworthy that in the heart, greater transcriptional activation of HIF target genes such as vascular endothelial growth factor (VEGF) and heme oxygenase (HO) was observed (Horowitz et al, 2005). We have also shown that after injury, the dynamic changes in nuclear HIF-1α expression differ in HA and CON mice. While the subunit levels steadily decrease in CON, the initial decrease in expression seen in the HA is followed by an increase, resulting in much higher levels of HIF-1α in HA 4 h after injury. This indicates that nuclear HIF-1α expression is enhanced by both HA and CHI. The increase in expression, which occurs because of HA, is sustained after injury as well. Pre- and post-injury upregulation of HIF-1α implies that it could be involved in the observed protective effect. Furthermore, the fact that the changes are pronounced in the nuclear content of the transcription factor, but do not occur in the cytosolic fraction, is consistent with the hypothesis that HIF-1 mediates protection via transcriptional activation of its target genes, because this mechanism would require its presence in the cell nucleus. Our focus in the present study was on the potential role of Epo-mediated neuroprotection in the beneficial effect of HA in CHI. However, it is noteworthy that HIF-1 also regulates the expression of several glycolytic enzymes, the GLUT1 glucose transporter and Transferrin, all of which can potentially promote cell survival (Eynan et al, 2002). Other factors such as VEGF and HO-2, which are also the products of HIF-1-regulated target genes, may also function as downstream mediators of protection involving HIF-1 (Storkebaum et al, 2004; Chang et al 2003). Surprisingly, the increase in HIF-1α after HA and CHI did not lead to a subsequent increase in Epo expression. This is unexpected because many studies have described the connection between these two factors during hypoxic stimuli. However, despite the substantial evidence indicating that increased expression HIF-1 is followed by enhanced Epo expression, other studies show that this is not necessarily always the case. It has been previously shown that the behavior of HIF-1 target genes varies among different genes and is also greatly dependent on the experimental model used. Jones and Bergeron (2001) have described the behavior of several HIF-1 target genes including Epo after the activation of HIF-1 by hypoxia or exposure to CoCl2. Similar to our results, in both models used in their study, the activation of HIF-1 was not followed by the expression of Epo. This was the case in both protein expression and mRNA level. They clearly show that differences between the different targets of HIF-1 exist in what is considered to be the classical setting of its induction. Furthermore, their work indicates that such differences occur even in two very similar models: hypoxia and a hypoxia mimetic agent. Therefore, it is conceivable that the lack of change in Epo expression is characteristic of this specific HIF-1 target gene in our model of CHI.
EpoR may also be upregulated by ischemia or hypoxia (Genc et al, 2004a,b; Siren et al, 2001). The involvement of ischemia in the pathophysiology of CHI could thus explain the increase in EpoR expression after injury. Because HA has been shown to mediate both heat tolerance and cross-tolerance via enhancement of the responsiveness of various genes to the applied stress (Horowitz et al, 2004), it is conceivable that the marked increase in EpoR in acclimated mice after CHI is because of an additive effect of both injury-induced ischemia and of increased responsiveness resulting from HA.
The precise mechanism by which Epo fosters neuroprotection is not entirely clear. However, it has been suggested to promote an antiapoptotic effect by triggering crosstalk between the Janus-tyrosine kinase (JAK-2) and nuclear factor κB (NFκB) signaling cascades, both of which are activated after Epo binding to EpoR (Digicaylioglu et al, 2001). Ligand binding to the receptor induces the phosphorylation of JAK-2, subsequently leading to dimerization of signal transducer and activator of transcription 5 (STAT5) and to the activation of both the phosphatidylinositide-3-kinase (PI3K) and the mitogen-activated protein kinase (MAPK) pathways. The NFκB pathway is activated via JAK-2-dependent phosphorylation of the inhibitory factor κB (IκB) (Chong et al, 2002). Another important consequence of increased EpoR expression is that enhanced activation of this receptor, as in the case of other growth factor receptors, has been shown to induce the upregulation of HIF-1α (Chun et al, 2002). Our results show that EpoR is upregulated after HA. A slight increase in receptor levels was observed in both CON and HA mice after injury, yet this increase was far less pronounced as compared with the robust upregulation induced by HA (two-fold increase). Erythropoietin receptor levels continue to be higher in HA than in CON up to 24 h after injury, indicating that the preinjury difference is sustained after injury as well. Although Epo levels of HA and CON were similar both before and after injury, overall pathway signaling, dependent on both receptor and ligand expression, was increased both by HA itself and by injury. Similar to the changes observed in HIF-1 and EpoR expression, EpoR/Epo ratio is also initially enhanced in HA animals and continues to be higher in this group after injury as well. The marked changes we have showed in both HIF-1 and EpoR expression imply the possible involvement of this pathway in neuroprotection occurring as a result of HA.
Although both acclimation and injury affect protein expression, the changes observed after acclimation were far more pronounced than those induced by injury. Acclimated animals are physiologically different than nonacclimated ones regardless of the subsequently induced insult (Horowitz, 2002). These basal differences in cytoprotective proteins are preserved after injury as well and may therefore explain cross-tolerance occurring after HA. This behavior is similar to the dynamics described for HSPs after HA (Maloyan et al, 1999; Horowitz et al, 2004). We therefore suggest that the improved outcome of CHI in HA animals is largely because of changes in protein levels occurring during HA, more so than those induced by injury itself.
Footnotes
Acknowledgements
ES is affiliated with the David R Bloom Center for Pharmacy of the Hebrew University School of Pharmacy, Jerusalem.