Abstract
We measured the long-term test–retest reliability of [11C]raclopride binding in striatal subregions, the thalamus and the cortex using the bolus-plus-infusion method and a high-resolution positron emission scanner. Seven healthy male volunteers underwent two positron emission tomography (PET) [11C]raclopride assessments, with a 5-week retest interval. D2/3 receptor availability was quantified as binding potential using the simplified reference tissue model. Absolute variability (VAR) and intraclass correlation coefficient (ICC) values indicated very good reproducibility for the striatum and were 4.5%/0.82, 3.9%/0.83, and 3.9%/0.82, for the caudate nucleus, putamen, and ventral striatum, respectively. Thalamic reliability was also very good, with VAR of 3.7% and ICC of 0.92. Test-retest data for cortical areas showed good to moderate reproducibility (6.1% to 13.1%). Our results are in line with previous test–retest studies of [11C]raclopride binding in the striatum. A novel finding is the relatively low variability of [11C]raclopride binding, providing suggestive evidence that extrastriatal D2/3 binding can be studied
INTRODUCTION
[
11
C]Raclopride is a commonly-used dopamine (DA) D2/3 receptor antagonist in positron emission tomography (PET) studies assessing D2/3 receptor availability. It dissociates rapidly from receptors allowing binding equilibrium to be established
Even though the expression of D2/3 receptors is highest in the striatum, dopaminergic projections show widespread efferents terminating in limbic, thalamic, and cortical regions. 7 In extrastriatal areas, [ 11 C]raclopride has been considered to be less adequate for measuring DA activity because [ 11 C]raclopride binding is far lower in extrastriatal than in striatal areas because of the lower density of D2/3 receptors resulting in low signal-to-noise ratios that hamper the reliability of the measurement. 8 Despite these concerns, there is evidence that decreases in [ 11 C]raclopride binding potential (BPND) can also be observed in extrastriatal regions after drug or behavioral challenges.9–11
Changes in extrastriatal [ 11 C]raclopride binding have also been associated with brain pathologies such as Parkinson's, Huntington's, and Alzheimer's diseases.12–14 The loci of the extrastriatal reductions of [ 11 C]raclopride binding in the abovementioned studies are also consistent with the distribution of extrastriatal D2/3 receptors reported in postmortem studies, where the thalamus has the highest density of D2/3 receptors followed by the lateral temporal cortex, the anterior cingulate, and the frontal cortex. 15
The region of interest (ROI) measurement of striatal D2/3 receptor binding using single-bolus [
11
C]raclopride injection is highly reliable.16–18 In most test–retest studies, scans have been performed on the same day17–19 and only one study has documented long-term reproducibility.
16
Long-term stability is essential for follow-up studies using pharmacological or behavioral challenges, where the interval between measurements can be relatively long. The reproducibility of [
11
C]raclopride binding in cortical areas has not yet been fully assessed. The ability to reliably measure DA release
MATERIALS AND METHODS
The study protocol was approved by the Ethics Committee of the Hospital District of Southwestern Finland. Subjects were given written information about all relevant issues involved in the study. Written consent was obtained from each subject. This study was performed according to the ethical guidelines given by the Declaration of Helsinki.
SUBJECTS
Seven healthy, right-handed, nonsmoking, and nonmedicated Finnish male university students took part in an ongoing study on the effects of working-memory training on striatal DA release. These subjects belonged to the control group who received no training, and they were chosen for the present test–retest analyses based on optimal motion data. Each subject underwent two [ 11 C]raclopride PET scans, 5 weeks apart. Each scan started with 55 minutes of baseline measurement during which subjects performed a very simple computerized letter-recall task, followed by 25 minutes working memory task. Only the 55 minutes of baseline data was analyzed in this study. During the baseline measurement the subjects were shown 7 to 15 letter sequences, where all the letters were identical (e.g., A – A – A – ...). When the sequence ended, the subjects were to report the last four letters by pressing buttons corresponding to A, B, C, and D. To exclude any structural brain abnormalities and to obtain anatomic references, all subjects underwent 3-T magnetic resonance imaging. Age, height, and weight of the subjects were 24±2 years, 176±1 cm, and 69±3 kg, respectively (mean ± s.d.).
Preparation of [ 11 C]Raclopride [ 11 C]Raclopride was prepared as described in our previous papers.19,20 Positron Emission Tomography Imaging
Positron emission tomography experiments were performed using a brain-dedicated high-resolution PET scanner, the ECAT high-resolution positron emission scanner (HRRT; Siemens Medical Solutions, Knoxville, TN, USA). The HRRT is a dual-layer, crystal-detector scanner allowing for depth-of-interaction measurement for coincident photons. It yields an isotropic 2.5-mm intrinsic spatial resolution. Spatial resolution in reconstructed images varies in radial and tangential directions from ~2.5 to 3 mm and in axial directions from 2.5 to 3.5 mm in the 10-cm field of view covering most of the brain.
21
The left antecubital vein was cannulated and [
11
C]raclopride bolus injections were administered intravenously, followed immediately by continuous infusion of [
11
C]raclopride for 80 minutes (data from the first 55 minutes of acquisition was used for test–retest analyses). A bolus-to-infusion rate ratio (Kbol) of 105 minutes and a bolus component of 50% (of the total volume of the tracer) were chosen according to an optimization study by Watabe
Image Processing
List-mode format PET measurements were histogrammed into series of three-dimensional sinograms using span 9 and a maximum ring difference of 67, resulting in 2,209 sinograms in 16 segments. Image reconstructions were made using an ordinary Poisson ordered subset expectation maximization algorithm with 16 subsets and 8 iterations and a voxel size of 1.22 mm × 1.22 mm × 1.22 mm. The time-frame sequence was 8 × 2, 4 × 3, 2 × 4, 1 × 5, 1 × 6, and 1 × 8 minutes. Dynamic image data were first corrected for frame-by-frame motion and between-scan misalignment using the realign function in SPM8 (Wellcome Trust Centre for Neuroimaging, London, UK). Second, anatomic T1-MR images were coregistered to a PET sum image, which was followed by MR tissue segmentation and spatial normalization using the unified segmentation algorithm in SPM8 (Wellcome Trust Centre for Neuroimaging). 24 Unified segmentation yielded tissue probability maps for gray and white matter and deformation fields for mapping individual images into Montreal Neurological Institute space and, conversely, for mapping images in Montreal Neurological Institute space into individual space.
Motion Data
An individually fitted thermoplastic mask was used for each subject to minimize head movements. To monitor head movements, an external position detector (Polaris Vicra, Northern Digital, Waterloo, Ontario, Canada) was used. The Polaris Vicra system (Northern Digital) tracks the position of passive infrared reflectors placed on top of the mask. The position of the reflectors was averaged over the transmission scans, which were used as reference positions for later tracking. External-motion estimates were obtained by calculating the position difference between the reference and a given time point. Points tracked by the Polaris system (Northern Digital) were located at the forehead close to the central line. Frame-to-frame motion estimates were obtained using the realign function of Statistical Parametric Mapping, version 8 (SPM8, revision 4290, Wellcome Trust Centre for Neuroimaging), which applies a mutual information maximization algorithm for rigid image registration. The frame at 4 to 6 minutes was used as a reference to obtain low within-frame motion and high-count statistics. Each frame was registered to the reference frame and a transformation matrix was used to generate motion-corrected image sequences, and to calculate displacement of a reference point at the forehead close to the central line. Frame-to-frame displacement of the reference point within SPM8 (Wellcome Trust Centre for Neuroimaging) was compared with average displacement of the Polaris (Northern Digital) reference point with very good agreement. Thus, frame-to-frame motion estimates from SPM8 (Wellcome Trust Centre for Neuroimaging) were used to exclude suspect data from final analysis. We considered displacements above 2.0 mm from the reference position to be critical, and if this threshold was exceeded in more than 10% of all frames in either of the scans, the subject was excluded from test–retest analysis.
Regions of Interest
Subcortical ROIs and reference region were drawn using in-house software (Carimas 2.8, Turku PET Centre, Turku, Finland) on coregistered magnetic resonance imagings. Thus, an identical set of ROIs was applied for both [
11
C]raclopride scans of the same subject. Regions of interest were defined in coronal plane for the caudate nucleus, putamen, and ventral striatum (VST). The caudate nucleus and putamen were divided anatomically into precommissural and postcommissural sections. The VST was defined according to Mawlawi
Quantification of [ 11 C]Raclopride Binding
Specific [ 11 C]raclopride binding to D2/3 receptors was estimated using the simplified reference tissue compartmental modeling (SRTM) 27 at both ROI and voxel levels. As reference tissue, we selected the cerebellum that is devoid of D2/3 receptors. 2 Simplified reference tissue compartmental modeling was used to obtain BPND values to estimate specific binding. 28 For ROI-level model fitting, we used nonlinear, weighted fitting based on actual count statistics implemented in an in-house software (http://www.turkupetcentre.net/software/doc/fit_srtm_3_1_0.xml). For voxel-level model fitting, we used a linearized model using a basis-function approach 29 implemented also in in-house software (http://www.turkupetcentre.net/programs/doc/imgbfbp.html). As a comparison, we also used ratios of area under the curve (from this point onward denoted as RM = ratio method) from 36 to 55 minutes to calculate BPND values representing specific binding of [ 11 C]raclopride during equilibrium condition. In this method, radioactivity concentration ratios of the target region and cerebellar reference region are calculated using the formula BPND = target–reference/reference. 30
Statistical Analyses
Test–retest variability (VAR%) was calculated as follows:
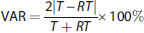
where T is the first (test) estimate and RT is the second (retest) estimate. Test–retest consistency was measured with an intraclass correlation coefficient (ICC). The ICC can have a value between −1 and 1. Values closer to 1 indicate that most of the variance is because of between-subject rather than within-subject variation, which indicates good reliability. Values below 0 indicate greater within-subject than between-subject variation indicating poor reliability. Paired two-tailed
RESULTS
Regional [
11
C]raclopride BPND values and their test–retest characteristics are presented in Table 1. BPND values are expressed as the mean ± s.d. In the striatum, [
11
C]raclopride BPND values were high in all subregions, ranging from 3.48±0.27 (VST) to 4.55±0.37 (posterior putamen). In the thalamus, the mean BPND was 0.70±0.06 and in the cortical areas BPND values ranged from 0.30±0.06 (superior frontal gyrus) to 0.38±0.07 (dorsolateral prefrontal cortex). There were no reliable differences in [
11
C]raclopride binding between scans in any of the regions measured (
SRTM-based regional [11C]raclopride BPND values and their test–retest characteristics (
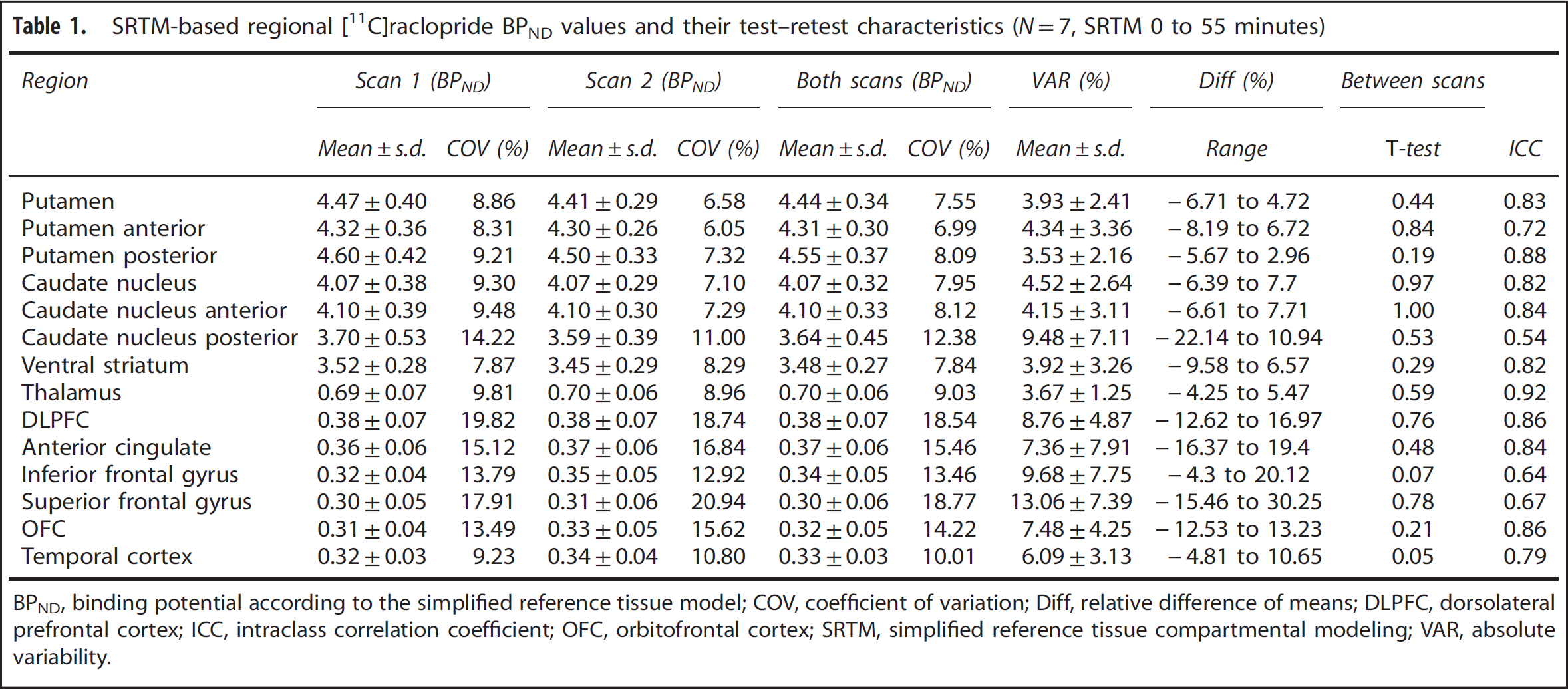
BPND, binding potential according to the simplified reference tissue model; COV, coefficient of variation; Diff, relative difference of means; DLPFC, dorsolateral prefrontal cortex; ICC, intraclass correlation coefficient; OFC, orbitofrontal cortex; SRTM, simplified reference tissue compartmental modeling; VAR, absolute variability.
Maps of voxel-level VAR and ICC are shown in Figures 1A and 1B, respectively. Table 2 shows regional [
11
C]raclopride BPND values and their test–retest characteristics using RM-based modeling. There were no statistically significant (
RM-based regional [11C]raclopride BPND values and their test–retest characteristics (
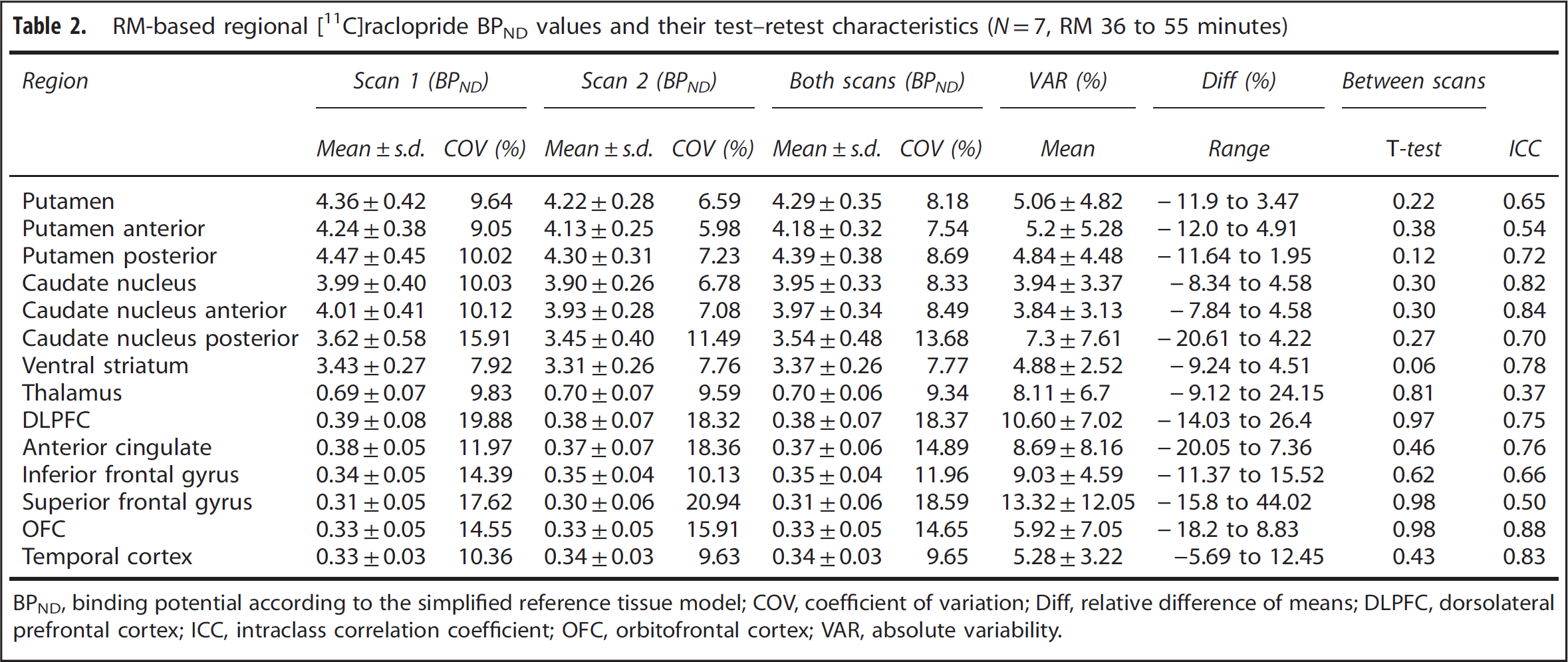
BPND, binding potential according to the simplified reference tissue model; COV, coefficient of variation; Diff, relative difference of means; DLPFC, dorsolateral prefrontal cortex; ICC, intraclass correlation coefficient; OFC, orbitofrontal cortex; VAR, absolute variability.
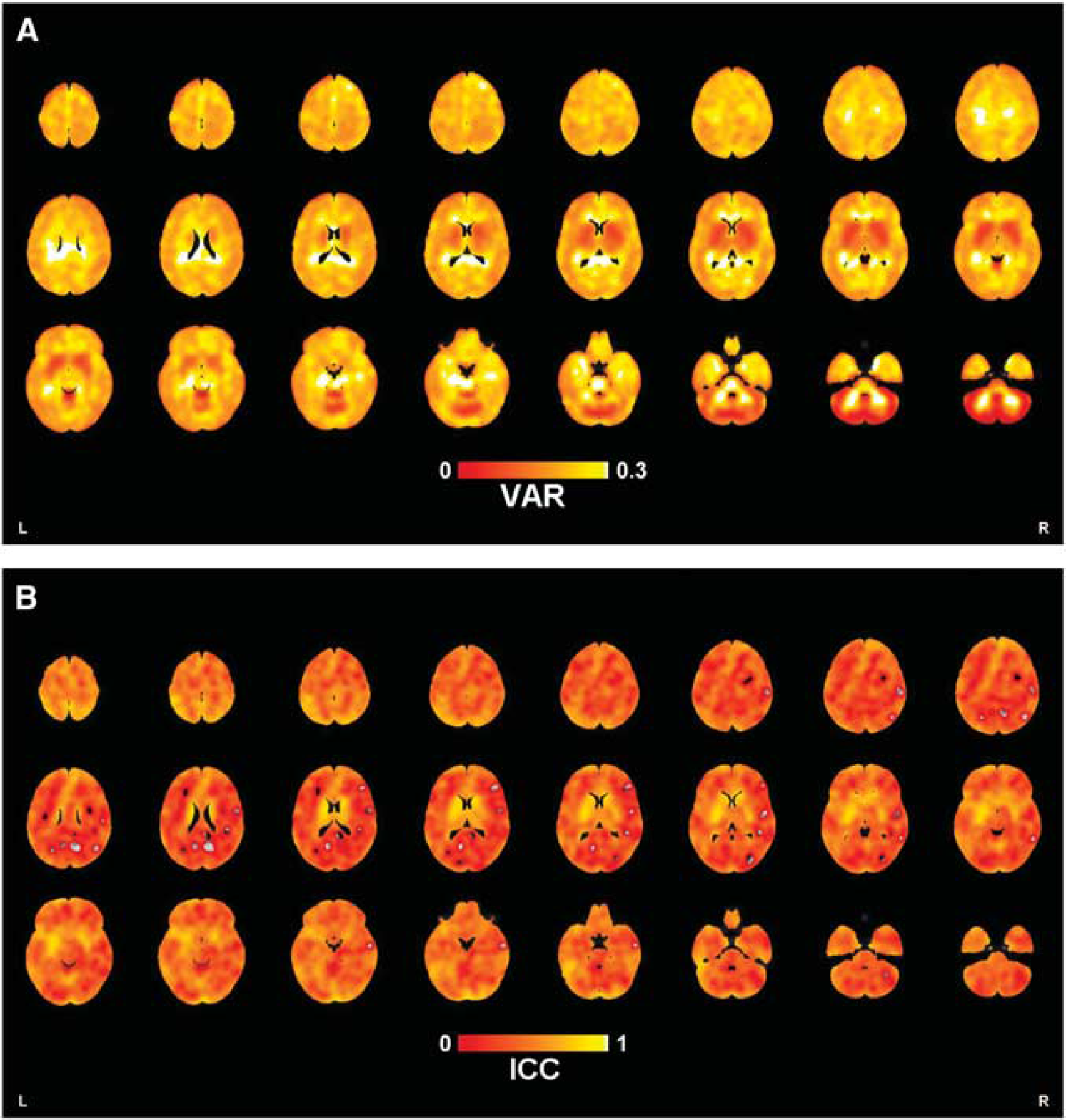
(
DISCUSSION
We explored the long-term test–retest reliability of striatal, thalamic, and neocortical DA D2/3 receptor binding using the B/I method with [ 11 C]raclopride and the HRRT scanner. A cohort of seven male subjects took part in an ongoing study on the effects of working-memory training on striatal DA release and they were scanned twice with a 5-week interval while they performed same, very simple computerized letter-recall task during both PET measurements. They were selected for the present test–retest analyses based on optimal motion data. The analyses were performed using SRTM modeling and by calculating ratios of areas under the curve (RM; see Materials and Methods section). In a previous test–retest study during rest, we showed that the use of the HRRT scanner increased the signal from the striatum and thalamus, resulting in higher BPND values compared with previous studies using scanners with lower resolution. 18 In the current study, we found that also in the regions characterized with low densities of D2/3 receptors such as the neocortex, the measurement of [ 11 C]raclopride binding turned out to be fairly robust. Protocol optimization by homogenizing the scanning conditions (simple cognitive task) and by eliminating the effect of movement may have contributed to this positive outcome. The rank order of observed BPND values was in line with the known density of both striatal and extrastriatal D2/3 receptors. 15 Moreover, the rostrocaudal trend of striatal [ 11 C]raclopride binding detected in our previous study was replicated, as the highest BPND values were observed in the posterior putamen and in the anterior caudate nucleus. 20
The reproducibility of a single bolus of [
11
C]raclopride binding in human striatum has been evaluated in various studies using different time intervals.16–18 In the earlier studies, the striatum was evaluated as a whole structure
16
and later subdivided into the putamen and caudate nucleus.
19
Mawlawi
The density of D2/3 receptors in extrastriatal regions is only 2% to 8% of that in the striatum.
31
Because [
11
C]raclopride has moderate
Although [
11
C]raclopride is suboptimal for measuring extrastriatal DA transmission, there is evidence that decreases in [
11
C]raclopride BPND can be observed in extrastriatal regions after drug or behavioral challenges.9–11 Changes in extrastriatal [
11
C]raclopride binding have also been associated with brain pathologic assesment, such as Parkinson's, Huntington's, and Alzheimer's diseases.12–14 Moreover, a recent study using diffusion-weighted magnetic resonance imaging and PET found that connectivity-based subdivision of human striatum improved the evaluation of regional differences in DA transmission, supporting an association between striatal DA release and cortical connectivity.
35
Thus, as major neurodegenerative disorders have been associated with alterations in corticostriatal circuits, imaging DA transmission simultaneously in striatal and extrastriatal regions is a major advantage. Stokes
In addition to SRTM
27
estimation of the BPND values, we also calculated ratios (=RM, as described earlier in the Materials and Methods section) of area under the curve from 36 to 55 minutes. RM is considered to be a gold standard in activation-type studies when using a B/I protocol.
30
The validity of both SRTM and RM modeling using B/I of [
11
C]raclopride has previously been evaluated by Mawlawi
The PET method is very sensitive to motion during image acquisition and greatly biases BP estimates. 38 Thus, the optimization of a study starts with careful planning of the setup, including optimal head support during data acquisition, which is especially critical when studying small structures or areas with a low signal-to-noise ratio. At the stage of analysis, if data are found to be affected by movement, all necessary steps to correct the bias should be taken. There is no unambiguous threshold for the amount of motion that could be tolerable during a PET scan. However, it is conceivable that an amount of motion that is less than the scanner resolution may be regarded as tolerable. The high-resolution scanner used in the present study is characterized by an isotropic 2.5-mm intrinsic spatial resolution. Thus, we considered displacements of >2.0 mm from the reference position to be critical, and if this threshold was exceeded in more than 10% of all frames in either scan, the subject was excluded from the test–retest analysis. By using this rather rigorous criterion for exclusion, we sought to ensure that head movements were not hampering the analysis of cortical D2/3 receptor binding, which is known to be challenging using [ 11 C]raclopride. In light of our results, this optimization appears to have been beneficial.
Subjects who participated in this study were initially recruited with the goal of studying the impact of working-memory training on DA transmission. The seven male subjects who were chosen for test–retest analyses were from the control group, so during the 5-week retest interval they did not practice any cognitive task. Thus, the scan pairs were identical as the same extremely simple letter-recall task was administered, and the same scan protocol was used during both measurements. We considered this to be a better alternative than rest, because functional magnetic resonance imaging studies have showed that the rest condition presumably might give subjects an opportunity for day dreaming, self-reflection, problem solving, and other uncontrolled mental activities that may affect the measurement.
39
Conceivably, the fact that our subjects focused their attention on the same simple cognitive task served to homogenize their DA transmission, increasing the detectability of radioligand binding. It may also have helped us to achieve good reproducibility of inherently weaker cortical [
11
C]raclopride D2/3 receptor binding. In our previous test–retest study, we showed that the use of the HRRT scanner increased the signal from the striatum and thalamus, resulting in higher BPND values compared with previous studies using scanners with lower resolution.
19
Stokes
Some potential limitations of the study should be noted. First, the sample size was small (
CONCLUSIONS
We examined long-term reliability of [ 11 C]raclopride as a DA D2/3 receptor marker in the human striatum, thalamus, and neocortex. Our results are in good agreement with past research and show the feasibility of the B/I method and good long-term stability of [ 11 C]raclopride BPND estimates. To our knowledge, this is the first test–retest study on cortical D2/3 receptor binding using [ 11 C]raclopride and suggests that extrastriatal D2/3 binding might be possible to study with this ligand although further validation is first needed. Careful planning of the study protocol and well-optimized conditions during scanning, e.g., reducing subject motion, could improve the possibilities to evaluate interactions between striatal and extrastriatal DA transmission using [ 11 C]raclopride.
AUTHOR CONTRIBUTIONS
KA contributed to study design, acquisition of data, interpretation of data, and drafting the article. JJJ was responsible for study design, analyzing the data, interpretation of data, and drafting the article. JJ was involved in study design, data analyses, and drafting the article. ML, LB, LN, and JOR have all contributed to study design, interpretation of data, and in revising the article critically. All authors have given their final approval of the version to be published.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGMENTS
The authors thank the personnel of Turku PET Centre for their skillful assistance during scanning.
